Abstract
Objective:
An RU486-inducible recombinant adenovirus-Nrf2 construct (Ad-RUNrf2) was constructed and expressed in H460 cells to determine whether Nrf2 gene expression can be regulated and to observe the effect of the adenovirus Ad-RUNrf2 on inflammatory cytokines, oxidative stress and apoptotic factors that mediate paraquat (PQ)-induced A549 cell injury.
Methods:
The Nrf2 gene within the RU486 (mifepristone)-inducible system was introduced into an adenovirus vector. A549 cells were transfected with Ad-RUNrf2, and Nrf2 expression was detected using Western blotting and real time-polymerase chain reaction (RT-PCR). RT-PCR, Western blots and enzyme-linked immunosorbent assay were used for observing the effect of RU486-induced Nrf2 expression on the inflammatory cytokines (interleukin-6 (IL-6), IL-10 and tumor necrosis factor-α (TNF-α)), oxidative stress factors (catalase (CAT) and malondialdehyde (MDA)) and apoptosis factors (caspase-3, caspase-9 and cytochrome C) that mediated PQ-induced A549 cell injury.
Results:
After infection of H460 cells by Ad-RUNrf2, RT-PCR and Western blot analyses showed that Nrf2 expression increased with additional RU486 doses. IL-6 and TNF-α protein and gene expression levels were significantly reduced, and IL-10 protein levels were significantly increased. Although IL-10 expression increased, it remained significantly lower than that of noninduced adenovirus infection and the simple virus exposure group. RU486 induced a significant reduction in MDA expression and increased CAT protein levels. Caspase-9 and caspase-3 protein and gene expression levels decreased in the RU486 induction group (p < 0.05). Cytochrome C protein levels were not significantly reduced, but its gene expression was significantly decreased (p < 0.05).
Conclusion:
Ad-RUNrf2 adenovirus was successfully constructed and can be stably expressed and regulated in cells. Ad-RUNrf2 can reduce PQ-induced inflammation, oxidative stress and apoptosis in A549 cells.
Introduction
Paraquat (PQ) is a quaternary ammonium bipyridyl compound (1,1′-dimethyl-4, 4′-bipyridylium dichloride), which is a fastacting, nonselective compound that destroys tissues of green plants on contact and by translocation within the plant. The mechanism of PQ poisoning remains unclear but is often associated with increased oxidative stress and activation of inflammatory and immune responses. Our previous research has found that inflammation and oxidation play important roles in PQ-induced lung injury. Although studies found that antioxidants can reduce the toxic effects of PQ, 1 their efficacy is not ideal. Nrf2 can bind with the antioxidant response element (ARE) in oxidative stress and plays an important role in the regulation of liver phase I and phase II detoxification enzymes. 2,3 We speculate that if intracellular Nrf2 gene expression can be increased and maintained at a certain level, the generation of endogenous antioxidants, such as CAT and superoxide dismutase (SOD), may be improved to enhance cellular antioxidant capacity, thereby reducing or treating lung injury induced by PQ. 4 –6
We inserted the Nrf2 gene into an adenovirus vector and regulated its expression with an RU486 (mifepristone)-inducible system in order to prevent potential adverse effects due to continued activation and overexpression of the gene. 7,8 The RU486-inducible adenovirus-Nrf2 construct (Ad-RUNrf2) was used to regulate and study Nrf2 expression in H460 cells. The effects of adenovirus Ad-RUNrf2 on inflammatory cytokines, oxidative stress and apoptosis factors that mediate PQ-induced A549 cell injury were studied.
Materials and methods
Materials
Cell lines and bacterial strains
The pCMV-XL5-Nrf2 plasmid was purchased from Origene. Plasmids pRS17-RUNEP, pKC-EF1α, pRS17-RURED and pRS17-RULUC, the adenovirus vector pDC312 and the adenovirus backbone plasmid pPE3 were constructed by the Viruses and Gene Therapy Laboratory of Shanghai Eastern Hepatobiliary Surgery Hospital (Shanghai, China). Escherichia coli strain DH5α, 293 cells, H460 cells, A549 cells were obtained from the Viruses and Gene Therapy Laboratory.
Reagents and instruments
Taq polymerase, deoxyribonucleotide triphosphate (dNTP) mix, T4 DNA ligase and DNA molecular weight markers were purchased from TaKaRa (Japan). Restriction enzymes were purchased from Neb (USA). Fetal calf serum and F12-Dulbecco's Modification of Eagle's Medium (DMEM) and 1640 media were purchased from Gibco (Britain). The viral DNA extraction kit, plasmid DNA preparation kit, polymerase chain reaction (PCR) product purification kit and Lipofectamine 2000 liposomes were purchased from Qiagen (Germany). The Dual-Luciferase® Reporter Assay kit was purchased from Promega (USA). The Nrf2 antibody was purchased from Origene (USA). The caspase-3, caspase-9 and cytochrome C antibodies were purchased from Abcam (Britain). RU486 was purchased from Sigma Corporation (USA). Tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and IL-10 detection kits were purchased from R&D (USA).
Primers
Primers were designed by Primer Premier 5.0 software (Table 1).
Primers

Vector construction
Construction of the adenovirus shuttle vector pDC312-RUNrf2
The IHSNEP gene (2367 bp) fragment from pRS17-RUNEP was digested with NheI and KpnI and ligated into the vector pKC-EFα1 (5786 bp) to construct pKC-IHSNEP. The plasmid pCMV-XL5-Nrf2 was used to amplify Nrf2 gene fragments for NheI + SalI double digestion and ligation with the vector pKCIHS (7676 bp). The product was named pKC-IHSNrf2. The IHSNrf2 fragment (3720 bp) in pKC-IHSNrf2 underwent NheI + KpnI double digestion and ligation into the pRS17-RUNEP vector, resulting in plasmid pRS17-RUNrf2. The RUNrf2 gene (7653 bp) from PRS17-RUNrf2 was digested with NotI and inserted into the pDC312-RUNEP vector, yielding plasmid pDC312-RUNrf2 (Figure 1(a)).

The flow charts of the vector construction: (a) pDC312-RUNrf2 construction; (b) pDC312-RULUC construction; (c) pDC312-RUDsred construction.
Construction of the pDC312-RULUC vector plasmid
The RULUC fragment (7821 bp) of pRS17-RULUC was digested with NotI and cloned into the PDC312 vector. The product was named pDC312-RULUC (Figure 1(b)).
Construction of pDC312-RUDSred vector plasmid
The RURed fragment (6526 bp) of pRS17-RUDSred was digested with NotI and cloned into the pDC312 vector, resulting in pDC312-RUDsred (Figure 1(c)).
Adenovirus Ad-RUNrf2 reconstruction and identification
The pDC312-RUNrf2 plasmid and the adenovirus backbone plasmid pPE3 were cotransfected into 293 cells using Lipofectamine 2000. The recombinant virus was obtained by homologous recombination. Virus plaques were observed 12 days after the cotransfection. The isolated plaques were picked and used to infect 293 cell cultures that were then overlaid with 5% agarose. After 10 days, well-defined plaques can be observed under the microscope and the isolated plaques were picked again. New round plaque purification was carried out on the picked plaques again.
After three rounds of plaque purification on 293 cells, the viral DNA was extracted using a Qiagen DNA Blood Mini Kit. Identification of the recombinant adenovirus was performed using PCR. Nrf2 primers were used for the detection of Nrf2 gene. Wild-type virus primers were used for the identification of wild-type virus.
Red fluorescent qualitative detection and LUC quantitative detection of the RU486 regulation system
pDC312-RUDsred was transfected into 293 cells in six-well plates using the Lipofectamine 2000 liposome transfection method. After 24 h of transfection, cells were treated with different doses of RU486 to induce Dsred expression. The concentrations of RU486 used were 0, 10−6, 10−7, 10−8, 10−9 and 10−10 mol/L. After 48 h of incubation, the pDC312-RUDsred plates were observed under a fluorescence microscope.
pDC312-RULUC was transfected into 293 cells in six-well plates using the Lipofectamine 2000 liposome transfection method. After 24 h of transfection, different doses of RU486 were given to induce luciferase (LUC) expression. The concentrations used were 0, 10−6, 10−7, 10−8, 10−9 and 10−10 mol/L. After 48 h of incubation, luciferase activity was detected using a Dual-Luciferase reporter gene assay kit.
Detection of Nrf2 mRNA and protein expression levels in Ad-RUNrf2-infected H460 cells
H460 cells were grown in 24-well plates for 24 h, and the Ad-RUNrf2 virus was added at an multiplicity of infection (MOI) = 5. The cells were further incubated with 5% CO2 at 37°C for 24 h and divided into six treatment groups. A final concentration of 0, 10−6, 10−7, 10−8, 10−9 and 10−10 mol/L of RU486 was used for the induction of four-wells each. In three wells, the supernatant was removed, cells were lysed and proteins were collected after treatment with RU486 for 24, 48 or 96 h. In the final well, the cell supernatant was removed 48 h after RU486 administration and was replaced with culture medium without RU486. After an additional 48-h incubation, the supernatant was removed, cells were lysed and proteins were collected. Nrf2 protein levels were evaluated by Western blotting. All the cells were collected for RNA extraction, and real time-polymerase chain reaction (RT-PCR) was used for the detection of Nrf2 messenger RNA (mRNA) expression.
Nrf2 gene and protein expression levels in Ad-RUNrf2-infected A549 cells
A549 cells were seeded into six-well plates at a concentration of 5 × 106 cells/well. After culturing for 24 h, cells were infected with Ad-RUNrf2 at an MOI = 50 during the exponential growth phase. After a 6-h incubation with 5% CO2 at 37°C, a final concentration of 0, 10−9, 10−8 or 10−7 mol/L of RU486 was given to induce Nrf2 expression. After 48 h, A549 cells were collected, and cellular proteins were extracted. Western blotting was used to detect Nrf2 protein levels.
The effect of Nrf2 expression on PQ-induced A549 cell injury
A549 cells were seeded into six-well plates at a concentration of 5 × 106 cells/well. After culturing for 24 h in the exponential growth phase, A549 cells were infected with Ad-RUNrf2 at an MOI = 50. After a 6-h incubation with 5% CO2 at 37°C, the cells were treated with 10−7 mol/L RU486 (AdRUNrf2 + RU486 group) or saline (Ad-RUNrf2 + NS group). Six hours after RU486 was added to the cells, a final concentration of 10−3 mol/L of PQ was added, and a blank control group was established. Cells were further incubated for 48 h, and the cells and supernatant were collected to evaluate the indicators of cell injury.
Detection of inflammatory cytokines by ELISA
The levels of TNF-α, IL-6 and IL-10 in the supernatant were detected by enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions.
Chemical colorimetric assay to measure levels of oxidative stress factors
Catalase (CAT), malondialdehyde (MDA) and superoxide dismutase (SOD) protein levels in cell supernatants were detected by chemical assay according to the kit’s instructions.
Western blot analysis to measure expression of apoptotic factor proteins
Proteins were extracted after the cells were collected. Sodium dodecyl sulfate-polyacrylamide gel electrophoresis was performed after protein denaturation, and proteins were transferred to nitrocellulose membrane. Blots were blocked with 5% skim milk and then incubated with rabbit anti-human caspase-9, caspase-3 (1:500) or cytochrome C antibodies (1:250) for 2 h. After washing, horseradish peroxidase-conjugated anti-rabbit immunoglobulin G (1:4000) was added to the blots for 2 h, and a chemiluminescence reagent was used for protein detection. Quantitative analysis was performed using an ImageJ analysis system.
RT-PCR detection of Nrf2, inflammatory cytokines and apoptotic factors
Total cellular RNA was extracted, and mRNA levels of Nrf2, TNF-α, IL-6, IL-10, caspase-3, caspase-9 and cytochrome C were detected using RT-PCR with glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as an internal control.
Statistical analysis
SPSS16.0 statistical software was used for statistical analysis. The data are presented as the mean ± SD (mean ± SE). Comparison between the groups was completed using analysis of variance, and a p < 0.05 was considered as a significant difference.
Results
Red fluorescence and LUC expression induced by RU486
When no RU486 was present, only a small amount of red fluorescence could be seen under a 40 × field. As the concentration of RU486 increased, red fluorescence also gradually increased (Figure 2(a)). Luciferase activity rose with increasing concentrations of RU486 (Figure 2(b)). LUC expression induced by RU486 was 2–5 times higher than LUC expression without RU486 induction (p < 0.001), but there were no significant differences in LUC expression among the different concentrations of RU486 used.

Qualitative and quantitative evaluation of the efficiency of the RU468 control system. (a) Qualitative detection of red fluorescence as an indication of RU486 control system efficiency, magnification × 40. As the concentration of RU486 increased, red fluorescence also gradually increased. (b) LUC quantitative evaluation of RU486 control system efficiency. Luciferase activity was low in the absence of RU486, but rose with increasing concentrations of RU486. Note: These data were subjected to a SPSS16.0 statistical analysis. a: p < 0.05 when compared with the NS group.
Nrf2 expression in Ad-RUNrf2-infected H640 cells
Twenty-four hours after H640 cells were infected with Ad-RUNrf2 at an MOI = 5, different concentrations of RU486 were added to the cells to induce Nrf2 expression. Western blots were used to examine the relative amount of Nrf2 protein expression (ratio of Nrf2 and β-actin) at each time point and concentration of RU486 (Figure 3(a)). Induction of Nrf2 protein expression increased with the RU486 concentration. The expression level increased 10–50 times when compared with Nrf2 levels in the absence of RU486 (p < 0.05). With the same concentration of RU486, Nrf2 protein expression gradually decreased over time (p < 0.05). After 48 h of RU486 induction, the supernatant was removed, and the culture medium without RU486 was added for an additional incubation of 48 h. The results indicated that Nrf2 protein expression was maintained and that the 96-h RU486 induction group had the lowest level of expression. Consistent with the LUC gene expression data, Western blots showed that the adjacent concentrations of RU486 did not induce statistically significant differences in Nrf2 protein expression.

Nrf2 protein expression in H460 cells transfected with Ad-RUNrf2 induced with different concentrations of RU486. (a) Western blot showing Nrf2 protein expression at different time points in H460 cells transfected with Ad-RUNrf2 under different concentrations of RU486. Induction of Nrf2 increased with the RU486 concentration, and the expression level increased 10–50 times when compared with Nrf2 levels in the absence of RU486 (p < 0.05). (b) Real time-polymerase chain reaction detection of Nrf2 protein expression at different time points in H460 cells transfected with Ad-RUNrf2 under different concentrations of RU486. Nrf2 mRNA expression was lowest in the absence of RU486, with an expression level only one-eighth that of cells induced with a high concentration of RU486. Under each RU486 concentration, the relative value of mRNA expression decreased gradually over time (p < 0.05). All data were analyzed by SPSS16.0. a: p < 0.05 when compared with the NS group at the same point.
RT-PCR showed that Nrf2 mRNA transcription increased in Ad-RUNrf2-infected H460 cells. The Nrf2 mRNA relative values (ratios of Nrf2 and GAPDH) increased with RU486 concentration at the same time point (p < 0.05; Figure 3(b)). Nrf2 mRNA expression was lowest in the absence of RU486, with an expression level of only 1/8th that of cells induced with a high concentration of RU486. Under each RU486 concentration, the relative value of mRNA expression decreased gradually over time (p < 0.05). When the supernatant was removed after 48 h of RU486 induction and culture medium without RU486 was used for an additional 48-h incubation, Nrf2 protein expression was sustained, and the RU486-induced 96 h group had lowest level of expression. RU486-induced Nrf2 protein expression changed as the concentration of RU486 was changed. The mRNA level peaked at 24 h and gradually decreased with time, but the expression levels at 48 and 96 h were not significant different (p > 0.05). When the supernatant was removed after 48 h of RU486 induction and replaced with culture medium without RU486 for an additional 48 h incubation, Nrf2 mRNA expression was sustained but slightly reduced when compared with cultures with sustained RU486.
Nrf2 expression in Ad-RUNrf2-infected A549 cells
Nrf2 protein expression in A549 cells was similar to that in H460 cells. As shown in Figure 4, Nrf2 protein expression increased with RU486 concentration and peaked at 10−7 mol/L with an expression level of three times that of untreated cells. Nrf2 protein expression in cells with RU486 induction was significantly increased (p < 0.01) over that of noninduced cells. Nrf2 protein induction was significantly increased when compared with control cells without RU486 (p < 0.05).

Western blot showing Nrf2 protein expression in A549 cells transfected with Ad-RUNrf2 under different concentrations of RU486. 1. 0 mol/L of RU486; 2. 10−9 mol/L of RU486; 3. 10−8 mol/L of RU486; 4. 10−7 mol/L of RU486; 5. Negative control group (Ad-RUNrf2 and RU486 were replaced by the same amount of saline). Nrf2 expression in cells with RU486 induction was significantly increased (p < 0.01) over that of noninduced cells. Nrf2 protein induction was significantly increased when compared with control cells without RU486 (p < 0.05).
RU486-induced Nrf2 expression reduced PQ-induced inflammatory cytokine expression in A549 cells
The group A549 cells infected with Ad-RUNrf2, induced with RU486, then treated with PQ (Ad + RU486 + PQ), IL6 and TNFα protein expression was significantly lower than in the Ad + NS + PQ and NS + PQ groups, with the expression levels amounting to one third to one half of the latter two groups (p < 0.05)(Figure 5(a)). IL6 and TNFα gene and protein expressions were consistent. IL-10 protein levels in the Ad + RU486 + PQ group increased three-fold with RU486 induction, but even with this increase, IL-10 levels were significantly lower than the noninduced adenovirus infection group and simple exposure group (p < 0.05; Figure 5(a) and (b)). We found that the Ad + RU486 + PQ group levels of inflammatory cytokines closer to that of the NS group, while the nonadenovirus infection group and simple exposure group had a more significant expression of inflammatory cytokines.

Chemical colorimetric assay for measuring the expression of oxidative stress factors in A549 cells. All data were analyzed using SPSS16.0. a: p < 0.05 when compared with the NS group. b: p < 0.05 when compared with the PQ + NS group. IL6 and TNFα protein expression was significantly lower than in the Ad + NS + PQ and NS + PQ groups (p < 0.05). IL-10 protein levels in the Ad + RU486 + PQ group increased three-fold with RU486 induction, IL-10 levels were significantly lower than the noninduced adenovirus infection group and simple exposure group (p < 0.05).
RU486-induced Nrf2 expression reduced PQ-induced oxidative stress level in A549 cells
MDA expression in the Ad + RU486 + PQ group was significantly lower than in the Ad + NS + PQ and NS + PQ groups (p < 0.05; Figure 6). The CAT protein level in the Ad + RU486 + PQ group was 1.5–4 times higher than in the other groups. When compared with the control group, CAT expression in the Ad + NS + PQ and NS + PQ groups was significantly reduced.

RU486-induced expression of inflammatory cytokines in A549 cells. (a) Enzyme-linked immunosorbent assay for RU486-induced expression of inflammatory cytokines; (b) real time-polymerase chain reaction detection of RU486-induced expression of inflammatory cytokines. All data were analyzed using SPSS16.0. a: p < 0.05 when compared with the NS group. b: p < 0.05 when compared with the PQ + NS group. MDA expression in the Ad + RU486 + PQ group was significantly lower than in the Ad + NS + PQ and NS + PQ groups (p < 0.05). The CAT protein level in the Ad + RU486 + PQ group was 1.5–4 times higher than in the other groups. When compared with the control group, CAT expression in the Ad + NS + PQ and NS + PQ groups was significantly reduced.
RU486-induced Nrf2 expression reduces PQ-induced apoptosis in A549 cells
As shown in Figure 7, caspase-9 and caspase-3 levels in the Ad + RU486 + PQ group decreased significantly (p < 0.05) and were close to that of the control group (p > 0.05). The gene and protein expression levels were both elevated to approximately two-times the expression levels of the control group (p < 0.05). Although Western blot results showed that cytochrome C expression in the Ad + NS + PQ and NS + PQ groups was only slightly higher than that of the Ad + RU486 + PQ group (p < 0.05), RT-PCR results showed that the expression level of cytochrome C in the Ad + RU486 + PQ group was significantly lower than in Ad + NS + PQ and NS + PQ group (p < 0.05) with no significant difference when compared with the control group (p > 0.05).
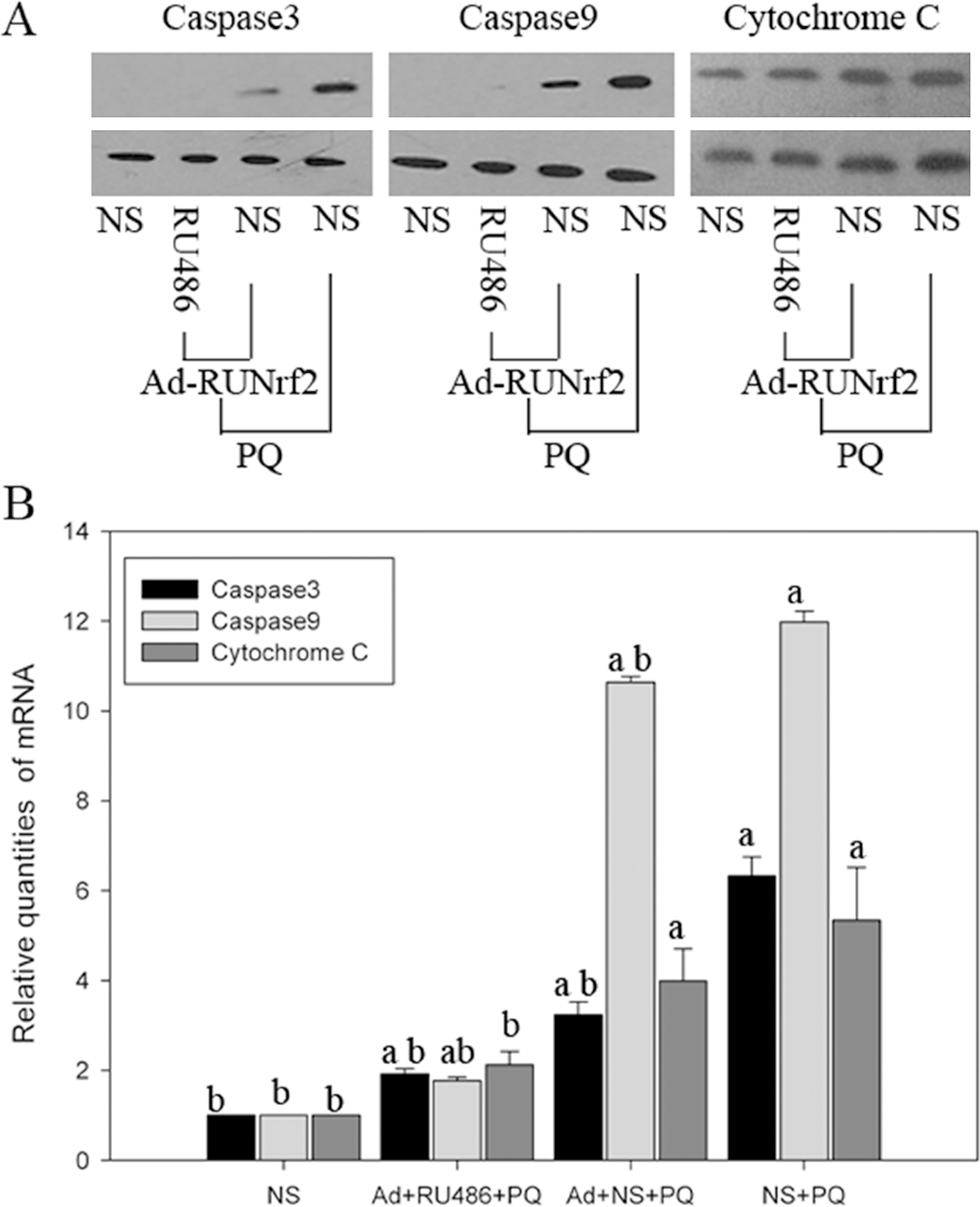
RU486 induced apoptosis factor expression in A549 cells. (a) Western blot of RU486-induced expression of apoptosis factors; (b) Real time-polymerase chain reaction detection of RU486-induced expression of apoptosis factors. All data were analyzed using SPSS16.0. a: p < 0.05 when compared with the NS group. b: p < 0.05 when compared with the PQ + NS group. Caspase-9 and caspase-3 levels in the Ad + RU486 + PQ group decreased significantly (p < 0.05) and were close to that of the control group (p > 0.05). The gene and protein expression levels were both elevated to approximately two-times the expression levels of the control group (p < 0.05).
Discussion
Nrf2 is key factor in regulating the expression of antioxidants. Nrf2 separates from Keap1 under oxidative stress and translocates to the nucleus. Once in the nucleus, Nrf2 forms heterodimers with a small Maf protein, and these heterodimers bind to AREs, thus activating a variety of antioxidant and detoxification enzymes. 9,10 In addition, Nrf2 plays an antiapoptotic role by inhibiting the Fas pathway. 9,11,12 We speculated that cells infected with an adenovirus carrying an inducible Nrf2 gene would have an increase in expression of antioxidant and detoxification enzymes, thus enhancing resistance to PQ-induced oxidative stress. To enhance Nrf2 activity in cells but avoid excessive activation of Nrf2, the Nrf2 gene under the control of an RU486 (mifepristone)-inducible system was introduced in an adenovirus vector to achieve the regulation of Nrf2 gene expression and protein activity.
The RU486 system is an inducible expression system used in mammalian cells. 13 –15 Its activation and expression levels depend on the concentration of RU486 used for induction. Within a certain range, target gene expression is proportional to the RU486 dose. The security of the system is due to the absence of the GAL4 in mammalian cells, so RU486 cannot activate any endogenous genes. Additionally, the amount of RU486 required to activate the system is very low, approximately 1000 times lower than the dose used for abortion (10 mg/kg).
In this experiment, we successfully constructed an RU486-inducible Nrf2 gene expression system using an adenovirus vector. LUC and Dsred were used for the verification of the RU486 regulation system. The results showed that the Renilla luciferase (RLUC) red firefly luciferase enzyme and red fluorescent expression was low in the absence of RU486. As the RU486 dose gradually increased, either firefly luciferase enzyme or red fluorescent increased, suggesting that the RU486 regulation system was stable and that the expression was dependent on the concentration of RU486. In addition, RT-PCR and Western blots showed that the expression of Nrf2 peaked after 24 h of RU486 induction. The expression value of Nrf2 decreased over time and depended on the amount of RU486 used for induction, which is consistent with previous reports. 15 Therefore, Nrf2 gene and protein concentration were RU486 dose- and time-dependent within the RU486 regulation system.
PQ can produce a large number of free radicals and upsets the intracellular oxidative/antioxidant balance, injuring cells and tissues directly 16 and induces inflammatory and immune responses, eventually leading to pulmonary fibrosis. 17 PQ poisoning caused oxidative stress increases and activation of inflammatory and immune responses. 18 Matters and Scandalios found PQ treatments resulting in a 40% increase in superoxide dismutase activity and catalase activity. Then Komada et al found a protective effect of transfection with SOD genes against extracellular superoxide anion-induced cytotoxicity. 19 In addition, Some research found that PQ can increase the incidences of Parkinson's disease. The Nrf2/Trx axis as a new target to block these pathways and protect from oxidant exposure such as that found in Parkinson. 20 A literature review revealed that A549 cells are similar to alveolar type II cells in terms of cell specificity. 21 Alveolar type II epithelial cells can uptake PQ actively through amine channels. 22 Alveolar type II epithelial cells are one of the main targets of PQ poisoning, and their severe injury is a main cause of alveolar hemorrhage, edema and hypoxemia. Therefore, we studied A549 cells and observed cellular injury induced by PQ after Ad-RUNrf2 adenovirus infection and Nrf2 induction. Examination of inflammatory cytokine protein and gene expression levels found that IL-6 and TNF-α gene and protein expression is the lowest during RU486 induction, while anti-inflammatory cytokine IL-10 showed the opposite trend and increased after RU486 induction of Nrf2. Therefore, Nrf2 induction balanced the expression of anti-inflammatory and proinflammatory cytokines. Thus, we concluded that A549 cells with Ad-RUNrf2-mediated Nrf2 gene expression were resistant to the PQ-induced inflammatory response, which is consistent with the view that Nrf2 and ARE play an important role in immune and inflammatory pathways. 5,23 Although SOD protein concentrations were not detected, the significant increase in CAT expression and decrease in MDA expression suggest that Ad-RUNrf2 can reduce the oxidative stress induced by PQ. A549 apoptotic factors (caspase-3, caspase-9 and cytochrome C) were reduced when Nrf2 expression was induced, suggesting that Nrf2 expression plays a significant role in reducing A549 apoptosis. These results provide technical support and a theoretical basis for further animal experiments.
Footnotes
Acknowledgments
The authors thank Professor Qijun Qian, the director of viral gene therapy laboratory, and staff of Shanghai hepatobiliary surgery hospital.
Funding
This work is supported by Natural Science Foundation of Zhejiang Province (grant No. Y2080977) and medical key construction project of Zhejiang Province (07-F04).
