Abstract
Organophosphate-induced delayed neuropathy (OPIDN) is a progressive neuropathic disorder that manifests in days to weeks following exposure to an acute dose of organophosphates. The precise mechanism involved in the development of OPIDN is not clear as it develops after many days of the cessation of cholinergic crisis. The present study has been designed to understand the role of oxidative stress in the development of OPIDN, wherein neuropathy was developed by the administration of acute dose of monocrotophos (MCP) or dichlorvos (2,2-dichlorovinyl dimethyl phosphate (DDVP)) to rats. Significant motor deficits in terms of reduced spontaneous locomotor activity and performance on narrow beam test were observed after 14 days of exposure to MCP or DDVP, which persisted even on day 28, suggesting the development of OPIDN. Rats with OPIDN also exhibited an increase in malondialdehyde levels along with a decrease in thiol content in cerebral cortex, cerebellum and brain stem. Concomitantly, the activities of antioxidant enzymes, superoxide dismutase, catalase, glutathione peroxidase and glutathione reductase were reduced in the three brain regions. The biochemical and functional changes were associated with histological alterations in the brain regions studied. The results clearly indicate that the development of OPIDN is mediated in part through an increased oxidative stress and suggest that the strategies aimed at restoration of antioxidant capacity may be beneficial for the individuals with OPIDN-like symptoms.
Introduction
Organophosphate pesticides (OPs) are of public health concern due to their worldwide use and documented human exposures. 1 ,2 OPs contribute to a substantial burden of mortality and morbidity particularly in developing countries including India. Although no exact estimates are available from India, studies suggest that it is one of the most common poisoning in occupational and nonoccupational cases. 3 One of the well-recognized complication of acute organophosphate poisoning has been organophosphate-induced delayed neuropathy (OPIDN).4 –6 It manifests after weeks of OP exposure and leads to motor paralysis affecting the distal muscles of limbs, minimal sensory involvement and calf pain. 7 The disorder is also characterized by distal degeneration of some long and large diameter axons in peripheral nerves and spinal cord. 8 Axonal swellings containing aggregation of neurofilaments (NFs), microtubules, proliferation of endoplasmic reticulum and multivesicular vesicles have been shown after the appearance of clinical signs of OPIDN; followed by the disappearance of NFs from swollen axons. 9 Exposure to an acute dose of OPs induces early necrotic injury followed by apoptotic neuronal degeneration. 7 Staining with Fluoro-Jade, a dye that detects apoptotic neuronal damage, revealed a time-dependent degeneration of nerve fibers and terminals and cell bodies following development of OPIDN. 10
Inhibition of neuropathy target esterase (NTE) has been well documented in OPIDN and is suggested to be a biomarker of OPIDN development. 8 It has been observed that NTE inhibition/aging occurs within hours after OP exposure. 11 However, NTE activity returns to normal well before the onset of clinical and morphological signs, 11 suggesting that the precise biochemical mechanism involved in the development of OPIDN is not well understood. Besides, studies done with experimental model of OPIDN have shown cytoskeleton abnormalities, 12 alterations in dopaminergic system,13,14 altered membrane function, 15 aberrant activation of cyclin-dependent kinase 5 (CDK5), 16 activation of mitogen-activated protein (MAP) kinase 17 and cAMP-response element binding (CREB), 18 induction of c-jun, 19 decreased ATP production, impaired mitochondrial functions 20 and mitochondrial integrity. 21
Oxidative stress has long been linked to the neuronal cell death that is associated with many neurodegenerative conditions. 22 It is still unclear whether oxidative stress is the primary initiating event or a secondary process associated with neurodegeneration. It is intimately linked with an integrated series of cellular phenomena which all seem to contribute to neuronal demise. Although the involvement of oxidative stress has been studied in OP exposure, 23 little is known about the involvement of oxidative stress in the development of OPIDN. Pazdernik et al. 24 suggested delayed increase in oxidative stress in the development of OPIDN. Recently, Slotkin et al. 25 compared transcriptional response following OP exposure and found that oxidative stress is the major target of OP toxicity. It has been reported that OPs induce oxidative stress by enhancing generation of reactive oxygen species (ROS) and/or by alterations in antioxidant defense mechanisms. 26 Zhang et al, 27 found time-dependent and tissue-specific changes in lipid peroxidation and antioxidant status in cerebrum, spinal cord and sciatic nerve, suggesting that oxidative stress may play an important role in the development of tri-ortho-cresyl phosphate (TOCP)-induced OPIDN in hens.
Monocrotophos (MCP; dimethyl (E) 1-methyl-2-(methylcarbomyl) vinyl phosphate) is an OP that is highly toxic to humans and animals. It is the single largest selling agrochemical in India and its consumption in 1997–1998 was 7500 metric tons, which increased to 42,000 tons during 1999–2004. 28 Dichlorvos (DDVP; 2, 2-dichlorovinyl dimethyl phosphate) is another organophosphate insecticide that has been in widespread use for over 40 years and has also been shown to cause delayed neuropathy in rats. 29 Acute exposure to both MCP and DDVP has been reported to cause delayed neuropathy in humans.30 –32
The present study has thus been designed to study the involvement of oxidative stress in the development of OPIDN following acute MCP or DDVP exposure by examining the levels of lipid peroxidation along with enzymatic as well as nonenzymatic antioxidants and to correlate them with the behavioral and histological changes.
Materials and methods
Chemicals
DDVP (technical grade >95%) was a kind gift from International Testing Center, Panchkula, India. MCP (technical grade > 74%) was a gift from Rallis India Ltd (Mumbai, India). Atropine sulfate (>95%) was obtained from Fluka (Germany), and 2-pralidoxime (2-PAM; >99%) was obtained from Sigma-Aldrich Co. (St.Louis, MO, USA). All other chemicals used were of analytical grade and were procured from reputed Indian manufacturers.
Animals and their treatment
Male Wistar albino rats (200–250 g) were obtained from the central animal house of Panjab University, Chandigarh. They were fed rat pellet diet containing 24% protein, 5% fat, 4% fiber, 55% carbohydrate, 0.6% calcium, 0.3% phosphorous, 10% moisture and 9% ash w/w (Ashirwad Industries, India) and water ad libitum. The protocols used in the study were strictly according to the guidelines on the use of experimental animals and were approved by the Institutional Ethics Committee. Animals were divided into four groups with six animals in each group. Atropine and 2-PAM was administered to the animals prior to administration of OPs to prevent the cholinergic crisis. Control (sham): animals were given pellet diet. Control (antidote): rats received atropine (20 mg/kg body weight i.p.) and 2-PAM (100 mg/kg body weight i.p.) MCP group: rats received MCP (20 mg/kg body weight, orally) after 15–20 min of atropine and 2-PAM. DDVP group: rats received DDVP (200 mg/kg body weight, subcutaneously) after 15–20 min of atropine and 2-PAM.
Body weights of the animals were recorded daily, whereas the plasma acetylcholinesterase activity and neurobehavioral changes were monitored on a weekly basis. After developing OPIDN, animals were euthanatized (day 28 postdosing) by cervical dislocation. Brains were quickly removed, rinsed in ice-cold isotonic saline and dissected into cerebral cortex, cerebellum and brain stem. The tissues were stored in −80°C for further analysis.
Neurobehavioral studies
Locomotor activity
Spontaneous locomotor activity was monitored using actophotometer (IMCORP, India) and the animals were individually placed in a chamber measuring 42 × 42 × 30 cm3 for 5 min for habituation and subsequently the total activity count was recorded for 5 min. The locomotor activity (rearing and ambulatory) was expressed in terms of total photobeams counts per 5 min per animal. 33
Narrow beam test
The narrow beam test was performed to measure hind limb impairment as described by Allbutt and Henderson 34 with slight modifications. Animals were trained to traverse a 150-cm-long wooden beam, divided into three 50-cm segments, from a platform at one end to the animal’s home cage on the other end, the beam was placed horizontally 80 cm above the floor. The number of foot slips onto an underhanging ledge and the time taken to pass the beam were recorded. The maximum time allowed for the task was 2 min; a fall was also recorded as a maximum time taken. Each rat was tested for three times, a score of 1 was given if the animal fell off one segment of the beam (maximum score was 9) and 0 for a rat that readily traverses the beam.
Biochemical estimations
Preparation of samples
Animals were anesthetized under light ether anesthesia. Blood was collected at weekly intervals from supraorbital sinus in tubes containing EDTA as anticoagulant. The samples were centrifuged at 1000g for 10 min and the plasma was separated. A 10% (w/v) tissue homogenate was prepared in 50 mM phosphate-buffered saline (pH 7.4) using Potter Elvehjem type homogenizer. The homogenate was centrifuged at 1200g for 10 min at 4°C and a portion of supernatant was further centrifuged at 12,000g for 20 min at 4°C to obtain the postmitochondrial fraction (PMS).
Acetylcholinesterase
Acetylcholinesterase (AChE) assay was performed in plasma as well as homogenate according to the method of Ellman et al. 35 Briefly, 14.9 mM acetylthiocholine iodide was added to the reaction mixture containing 0.1 M phosphate buffer, 10 mM DTNB (5, 5'-Dithio-bis(2-nitrobenzoic acid)) and appropriate amount of sample to initiate the reaction. The increase in absorbance was followed at 412 nm for 3 min. AChE activity was calculated using the molar extinction coefficient of 5-mercapto-2-nitrobenzoate (13.6 × 103 /M/cm) and the results were expressed as nanomoles of substrate hydrolyzed per minute per milligram of protein.
Lipid peroxidation
Lipid peroxidation (LPO) was ascertained by measuring malondialdehyde (MDA) formed by the method described by Wills. 36 Briefly, homogenates prepared in 0.1 M Tris-HCl buffer (pH 7.4) were incubated at 37°C for 2 h. The reaction was stopped by the addition of ice-cold trichloroacetic acid 10% (w/v) followed by centrifugation at 1000g for 10 min. Thiobarbituric acid, 0.67% (w/v), was added to the supernatant and incubated in a boiling water bath for 10 min. The absorbance was measured at 532 nm and the results expressed as nanomoles of MDA per milligram of protein using molar extinction coefficient of MDA-thiobarbituric acid chromophore (1.56 × 105 /M/cm).
Total thiols
Total thiols (T-SHs) were measured according to the method of Ellman 37 as modified by Sedlak and Lindsay. 38 A reaction mixture containing 0.2 M Tris-HCl, 0.02 M EDTA, pH 8.2, brain homogenate, 0.01 M DTNB in methanol were kept for 15 min at room temperature and then centrifuged at 1200g for 5 min. The absorbance of the supernatant was read at 412 nm. Results were expressed as nanomoles of T-SH per milligram of protein.
Low-molecular-weight thiols
Low-molecular-weight thiols (LMW-SHs; primarily GSH) were quantified according to the method of Ellman. 37 Briefly, proteins in the homogenate were precipitated by 4% sulfosalicylic acid followed by centrifugation at 1200g for 5 min. To the supernatant 0.1 mM DTNB (in 0.1 M phosphate buffer of pH 8) was added and the absorbance was read at 412 nm after 2 min. Results were expressed as nanomoles of LMW-SH per milligram of protein.
Protein thiols
Levels of protein thiols (P-SHs) were calculated from the difference between the values of T-SHs and LMW-SHs.
Superoxide dismutase
Superoxide dismutase (SOD) activity was measured in PMS by the method described by Kono. 39 Addition of SOD inhibited the hydroxylamine hydrochloride–mediated reduction of nitroblue tetrazolium (NBT). The reaction mixture consisted of 50 mM sodium carbonate, 0.1 mM EDTA (pH 10.8) 96 µM NBT, 0.6% Triton X-100, requisite amount of PMS and 20 mM hydroxylamine hydrochloride. The reference contained all the above except PMS. Absorbance was recorded at 560 nm for 3 min. The extent of inhibition was taken as a measure of the enzyme activity. The activity of enzyme was expressed as units per milligram of protein, where one unit of enzyme is defined as the amount of enzyme inhibiting the rate of reaction by 50%.
Catalase
Catalase (CAT) activity was assayed according to the method described by Luck. 40 Appropriate amount of postnuclear supernatant was added to 12.5 mM hydrogen peroxide (H2O2) in 0.067 M phosphate buffer (pH 7.0). The decrease in absorbance was followed at 240 nm for 3 min. Results were expressed as micromoles of H2O2 decomposed per minute per milligram of protein using molar extinction coefficient of H2O2 (71 /M/cm).
Glutathione peroxidase
Glutathione peroxidase (GSH-Px) activity was assayed according to the method of Paglia and Valentine 41 as modified by Lawrence and Burk. 42 The reaction mixture consisting of 50 mM phosphate, buffer 1.5 mM EDTA (pH 7.0), 30 mM sodium azide, 6 mM nicotinamide adenine dinucleotide phosphate (NADPH), 60 mM GSH, glutathione reductase (GR 1 unit) and PMS was allowed to equilibrate at 20°C. The reaction was initiated by addition of 2.2 mM H2O2 and the decrease in absorbance was followed at 340 nm for 3 min. Results were expressed as nanomoles of NADPH oxidized per minute per milligram of protein, using molar extinction coefficient of NADPH (6.22 × 106 /M/cm).
Glutathione reductase
GR activity was determined in PMS by the method of Horn. 43 The assay mixture consisted of 67 mM phosphate buffer, 15 mM EDTA, 6 mM NADPH (dissolved in 1% NaHCO3), 7.5 mM oxidized glutathione and PMS. Absorbance was followed at 340 nm for 3 min. Results were expressed as nanomoles of NADPH oxidized per minute per milligram of protein using molar extinction coefficient of NADPH (6.22 × 106 /M/cm).
Glutathione-S-transferase
Glutathione-S-transferase (GST) activity was assayed in PMS according to Habig et al. 44 GSH, 30mM, was added to a reaction mixture containing 0.3 M phosphate buffer of pH 8, 30 mM CDNB (1-chloro-2,4-dinitrobenzene ) and PMS. Absorbance was followed at 340 nm for 3 min. Results were expressed as nanomoles of GSH-CDNB conjugate formed per minute per milligram of protein using molar extinction coefficient of conjugate (9.6 × 106 /M/cm).
Estimation of protein
The protein content was estimated according to the method of Lowry et al. 45
Histopathological studies
Histopathological studies were carried out by performing routine hematoxylin and eosin (H&E) staining to evaluate the morphological and structural changes. The brain regions were fixed in Bouin’s fluid, embedded in paraffin, sectioned at 5 µm thickness and stained with H&E.
Statistical analysis
All values were expressed as mean ± SEM of six animals per group. Data were analyzed using one-way analysis of variance followed by Newman–Keuls test for multiple pairwise comparisons between the various groups. Values with p < 0.05 were considered as statistically significant.
Results
Locomotor activity
Locomotor activity was assessed in terms of rearing and total activity (rearing and ambulatory) on days 1–28 following exposure to OPs (Figure 1(a) and (b)). MCP-treated animals showed severe impairment in both rearing and total locomotor activity on day 1. The locomotor activity was observed to be deficit on day 28 post MCP exposure. However, the extent of reduction in locomotor activity was less as compared to the activity on day 1. The animals of DDVP group also exhibited motor deficits starting on day 14, which continued up to day 28 postexposure.

Rearing (a) and total (b) locomotor activity in control sham (x), control antidote (O), MCP- (▴) and DDVP (▪)-treated animals. Results are expressed as mean ± SEM; n = 6. Data were analyzed by one-way analysis of variance followed by Student–Newman–Keuls test. *p < 0.05 was considered significant for control animals. #p < 0.05 was considered significant for DDVP (antidote)-treated animals. MCP: monocrotophos; DDVP: 2,2-dichlorovinyl dimethyl phosphate.
Narrow Beam test
Beam walk test was performed to assess hind limb function on a weekly basis in terms of scores and time taken by each rat to traverse the beam (Figure 2(a) and (b)). Control animals transversed the beam at a constant rate, while keeping both the paws on the entire part of the beam; whereas MCP-treated animals showed severe impairment in narrow beam test on day 1. There was a slight recovery at day 7 which, however, did not last long. The deficits in locomotor activity increased and continued from day 14 to day 28 postexposure. The time taken on the beam and the average scores of DDVP-treated animals also clearly indicated impairment in motor functions.

Beam walk test activity expressed as time taken on the beam (a) and average of beam score (b) in control sham (x), control antidote (O), MCP- (▴) and DDVP (▪)-treated animals. Results are expressed as mean ± SEM; n = 6. Data were analyzed by one-way analysis of variance followed by Student–Newman–Keuls test. *p < 0.05 was considered significant for control animals. #p < 0.05 was considered significant for DDVP (antidote)-treated animals. MCP: monocrotophos; DDVP: 2,2-dichlorovinyl dimethyl phosphate.
Acetylcholinesterase
AChE activity was monitored on weekly intervals in both the plasma and in the three brain regions of rats exposed to MCP or DDVP. A significant decrease in the activity was observed. The activity in plasma was inhibited on the days 1, 7 and 14 in the plasma of MCP-exposed animals, whereas in the DDVP-exposed animals the activity of AChE was inhibited on 14, 21 and 28 days postexposure (Figure 3). The activity of AChE was reduced by 13.21% in cortex after 28 days of MCP exposure, whereas cerebellum and brain stem recorded 5.5% and 20.8% reduction in activity as compared with the control animals. Cortex, cerebellum and brain stem of DDVP-treated animals showed 10.87%, 10.03% and 8.92% reduction in AChE activity (Table 1).

Acetylcholinesterase activity in plasma in control sham (x), control antidote (O), MCP-(▴) and DDVP (▪)-treated animals. Results are expressed as mean ± SEM; n = 6. Data were analyzed by one-way analysis of variance followed by Student–Newman–Keuls test. *p < 0.05 was considered significant for control animals. #p < 0.05 was considered significant for DDVP (antidote)-treated animals. MCP: monocrotophos; DDVP: 2,2-dichlorovinyl dimethyl phosphate.
Acetylcholinesterase activity and lipid peroxide levels in the brain regions of OPIDN ratsa
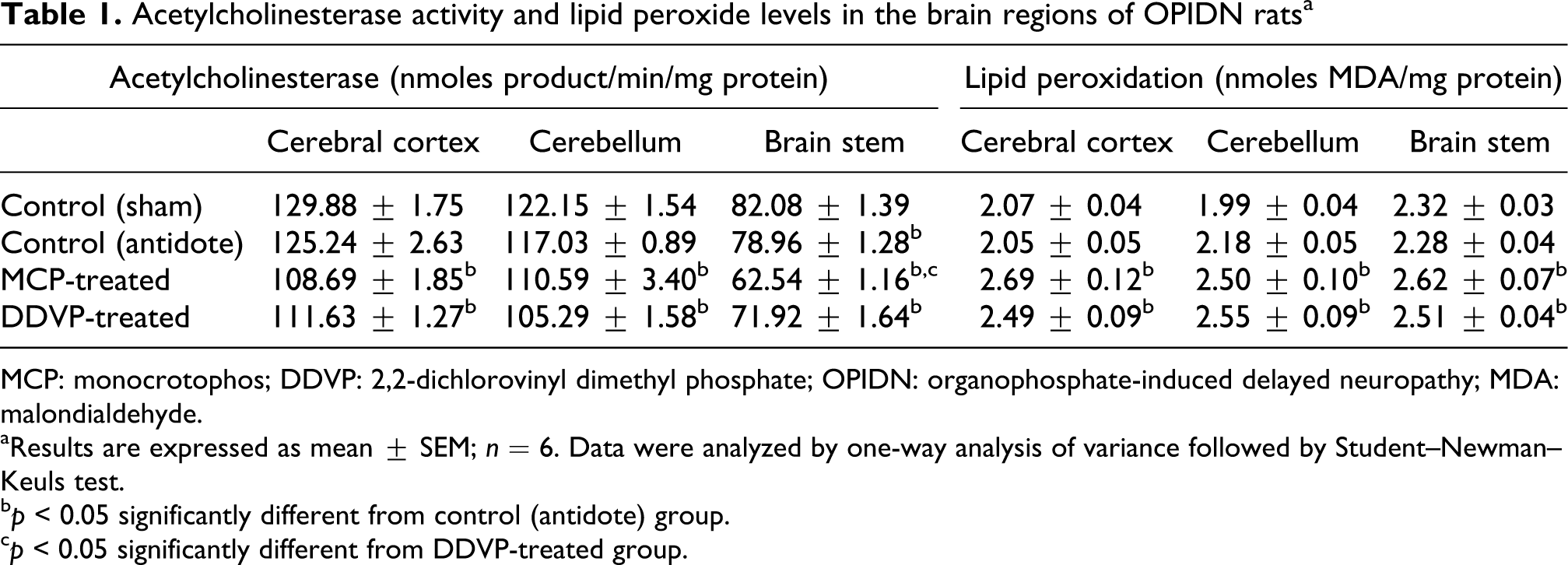
MCP: monocrotophos; DDVP: 2,2-dichlorovinyl dimethyl phosphate; OPIDN: organophosphate-induced delayed neuropathy; MDA: malondialdehyde.
aResults are expressed as mean ± SEM; n = 6. Data were analyzed by one-way analysis of variance followed by Student–Newman–Keuls test.
b p < 0.05 significantly different from control (antidote) group.
c p < 0.05 significantly different from DDVP-treated group.
Lipid peroxidation
A significant increase in the MDA levels were observed in cerebral cortex, cerebellum and brain stem by 31.22%, 14.68% and 14.91%, respectively, in MCP-treated animals as compared to the control group (Table 1). Similarly in DDVP-treated group, a significant increase in the MDA levels were observed in all brain regions (cerebral cortex, cerebellum and brain stem), which corresponded to an increase of 21.46%, 16.97% and 10.09% in control, respectively.
Thiol content
T-SHs were reduced in all the three regions viz. cerebral cortex, cerebellum and brain stem of both DDVP- and MCP-treated animals. MCP-exposed animals had 29.2%, 15.65% and 11.85% reduction in T-SH levels in the brain regions, whereas the DDVP-exposed animals showed 30.29%, 18.32% and 28.91% reduction in thiols (Table 2). LMW-SHs, primarily glutathione, were significantly lowered following OP exposure. In the MCP-exposed brain regions, the LMW-SHs were lowered by 27.84%, 22.47% and 14.88% (Table 2). Similarly, DDVP exposure also caused a significant decrease in LMW-SH content in all the regions of the brain. The decrease was found to be 13.81%, 27.81% and 25.63% in cerebral cortex, cerebellum and brain stem of DDVP-exposed animals, respectively. The levels of P-SH s also showed the similar trend following exposure to MCP or DDVP as observed in the case of T-SH and LMW-SH.
Distribution of total, low-molecular-weight and protein thiols in the brain regions of OPIDN ratsa

MCP: monocrotophos; DDVP: 2,2-dichlorovinyl dimethyl phosphate; OPIDN: organophosphate-induced delayed neuropathy.
aResults are expressed as mean ± SEM; n = 6. Data were analyzed by one-way analysis of variance followed by Student–Newman–Keuls test.
b p < 0.05 was considered significantly different from control (antidote) group.
Antioxidant enzymes
Reduction in SOD activity by 20.19%, 25.47% and 42.46% was seen in cortex, cerebellum and brain stem, respectively, of MCP-treated animals (Table 3), whereas 36.74%, 18.74% and 61.95% decrease in SOD activity was observed in DDVP-treated animals. CAT activity was reduced in all the three regions by 29.2% in cortex, 15.65% in cerebellum and 11.85% in brain stem in MCP-treated animals as compared to the control animals. The enzyme was lowered in the three brain regions of DDVP-treated animals by 30.29%, 18.32% and 28.91%, respectively (Table 3). A significant decrease in the activity of GSH-Px was seen in DDVP-treated animals with 21.38% reduction in cortex, 29.23% in cerebellum and 16.18% in brain stem compared to the control group. The reduction in GSH-Px was 24.87%, 21.79% and 14.23% in the three brain regions, respectively, of MCP-treated animals (Table 4). A decrease in GR activity was seen in the three brain regions of DDVP-treated animals compared to control group, which accounted for 24.1%, 37.14% and 29.17% decrease, respectively. GR was inhibited in brain regions of MCP-exposed animals by 29.88%, 36.8% and 21.07%, respectively (Table 4). The activity of GST was not affected in the MCP- and DDVP-treated animals as compared to the controls (Table 5).
Superoxide dismutase and catalase activities in the brain regions of OPIDN ratsa

MCP: monocrotophos; DDVP: 2,2- dichlorovinyl dimethyl phosphate; OPIDN: organophosphate-induced delayed neuropathy; MDA: malondialdehyde; H2O2: hydrogen peroxide.
aResults are expressed as mean ± SEM; n = 6. Data were analyzed by one-way analysis of variance followed by Student–Newman–Keuls test.
b p < 0.05 significantly different from control animals.
c p < 0.05 significantly different from DDVP (antidote)-treated animals.
Glutathione peroxidase and glutathione reductase activities in the brain regions of OPIDN ratsa

MCP: monocrotophos; DDVP: 2,2-dichlorovinyl dimethyl phosphate; OPIDN: organophosphate-induced delayed neuropathy; NADPH: nicotinamide adenine dinucleotide phosphate.
aResults are expressed as mean ± SEM; n = 6. Data were analyzed by one-way analysis of variance followed by Student–Newman–Keuls test.
b p < 0.05 was considered significantly different from control (antidote) animals.
Glutathione-S-transferase activity in the brain regions of OPIDN ratsa
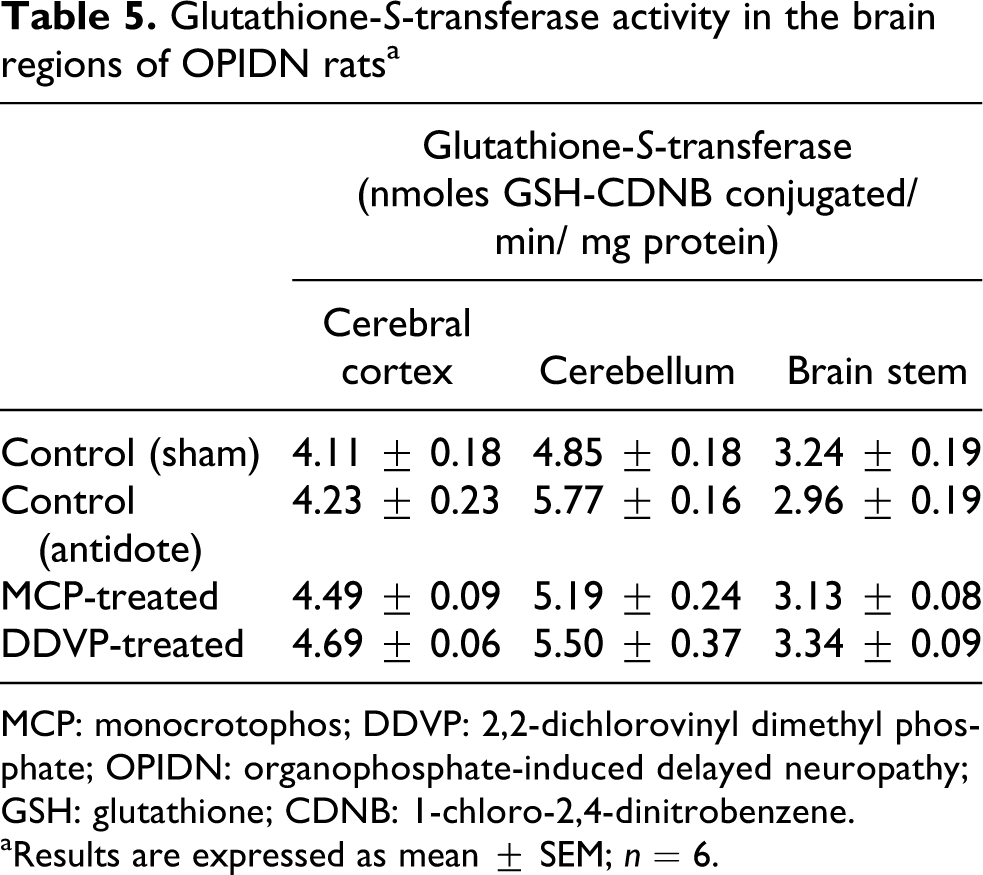
MCP: monocrotophos; DDVP: 2,2-dichlorovinyl dimethyl phosphate; OPIDN: organophosphate-induced delayed neuropathy; GSH: glutathione; CDNB: 1-chloro-2,4-dinitrobenzene.
aResults are expressed as mean ± SEM; n = 6.
Histopathological changes
Histopathological examination of brain regions after staining with H&E showed normal cell morphology in the control (sham) group. Administration of atropine and 2-PAM caused engorging of vessels with rare pyknosis. Cerebral cortex, cerebellum and brain stem from MCP- or DDVP-treated animals showed increased pyknosis, increased number of astrocytes and engorged vessels (Figure 4).

Histopathological changes following MCP or DDVP exposure in (i) cerebral cortex, (ii) cerebellum and (iii) brain stem of control sham (a), control antidote (b), MCP-treated (c) and DDVP-treated (d) (H&E, ×200; inset ×400). P: pyknotic nuclei; EV: engorged vessels; GC: glial cells; MCP: monocrotophos; DDVP: 2,2-dichlorovinyl dimethyl phosphate.
Discussion
Patients hospitalized after accidental or occupational exposures to OPs are treated by administering atropine and 2-PAM to attenuate hypercholinergic crisis. However, some of these patients develop delayed neuropathy a few weeks after recovering from cholinergic syndrome. 46 MCP has been reported to cause delayed neuropathy in humans. 30 In addition, DDVP has also been reported to cause OPIDN in humans30 –32 as well as in experimental animals11,29 along with the inhibition of NTE activity. 13
In the present study, OPIDN was developed in rats by administering atropine and 2-PAM prior to administration of a single acute dose of OP compounds viz. MCP at a dose of 20 mg/kg body weight, orally and DDVP at a dose of 200 mg/kg body weight, subcutaneously. 29 OPIDN is associated with the weakness of the lower limbs; therefore locomotor and narrow beam tests have been performed to confirm the motor deficits. 7 Rats in MCP or DDVP groups in the present study showed a marked impairment of such motor functions. The deficits in motor coordination were observed after 1–2 weeks of exposure of MCP and DDVP and persisted even after 28 days of exposure, indicating the severe development of OPIDN. OPIDN is unique in that it is caused by a single exposure to certain OPs with effects appearing after 14–21 days. 47 This might be because of a high degree of peripheral nerve degeneration, affecting the hind limb function and associated motor deficits. 48 It has been suggested that impaired motor functions in OPIDN rats may involve changes in central nervous system (CNS), which are most likely due to the damage to cerebellum as it controls the balance and fine motor control. 49 Besides, the MCP-treated group showed severe impairment in motor deficits and inhibition in AChE activity at day 1, whereas DDVP exposure had less pronounced effect. These differences might be due to the different routes of exposure of MCP and DDVP. The administration of DDVP subcutaneously would result in a slow release compared to rapid absorption from the gastrointestinal tract in case of MCP. Furthermore, we preferred to administer MCP at an acute dose orally since it is the route by which the population is exposed to OPs. The OPs that cause OPIDN inhibit NTE by more than 70% by phosphorylating the enzyme and leaving the phosphorylated enzyme with a negative charge which is dependent on the alkyl group of OP. 47 The reason that both MCP and DDVP cause OPIDN is that they are structurally similar, both have same alkyl groups attached to the phosphorus atom; however, they have different X groups as shown in Figure 5.

Chemical structure of monocrotophos (a) and dichlorvos (b). The R groups attached to P atom are same in both organophosphates.
MDA, an index of increased oxidative stress, was observed to be increased in the cerebral cortex, cerebellum and brain stem on day 28 following exposure to MCP or DDVP. Many OPs have been reported to accentuate lipid peroxidation in mammalian system after acute and chronic exposure, 50 including MCP 51 and DDVP.52,53 Stotkin et al. 25 also observed that oxidative stress is a major target for OP effects in vivo. Neuronal membranes are characterized by large amounts of polyunsaturated fatty acids (PUFAs) associated with amphipathic lipids and a variety of proteins. The onset of lipid peroxidation within cellular membranes is associated with changes in their physicochemical properties affecting the functions of membrane bound proteins, 54 which might contribute to the development of delayed neuropathy following acute OP exposure. LPO, the end product of ROS, is believed to be largely responsible for the cytotoxic effects observed in various neurodegenerative conditions. 55 The mechanism involved in increased oxidative stress in OPIDN seems to include mitochondrial dysfunctions. 20
The cell detoxifies free radicals via its antioxidant defense system, which includes the antioxidant enzymes: SOD, GSH-Px, GR and CAT and also the nonenzymatic antioxidants like GSH. 56 A significant reduction in the antioxidant status in terms of the decreased thiol levels and activities of CAT, SOD and GR in the brain regions of MCP- and DDVP-treated animals was observed. However, no significant effect in the activity of GST was observed in the three brain regions of MCP- and DDVP-treated groups.
GSH is a water-soluble antioxidant that is central to cellular defense against oxidative stress and potentially toxic chemicals. 57 It directly quenches reactive hydroxyl radicals, other oxygen-centered free radicals and conjugates to the xenobiotic to make them water soluble. 58 We observed that there was a reduction in thiol levels in OPIDN rats. MCP has been reported to lower the GSH levels and other thiols in the hepatic and nonhepatic tissues.59,60 In addition, DDVP has been reported to lower GSH levels in brain and other tissues.61,62 Low levels of GSH could be due to the enhanced generation of ROS which are scavenged by GSH or decreased activity of GR, which converts oxidized glutathione (GSSG) to its reduced form. The reduction in GSH levels would impair thiol-dependent detoxification reactions in brain, whereas reduction in P-SHs may affect conformation/catalysis of –SH-containing enzymes which may be detrimental to the neuronal functions.
Superoxide radical (O2−.) and H2O2 are known to cause membrane lipid peroxidation through Fenton reaction. O2−. is converted into H2O2 by SOD, whereas H2O2 is decomposed to water and oxygen by CAT. SOD showed a reduction in activity in all the regions with a maximum reduction in the brain stem. No report is available on the effect of MCP on SOD activity. Reduction in SOD activity in the brain region has been reported after exposure to DDVP.63,64 However, Julka et al. 61 found induction in both SOD and CAT in brain following chronic DDVP exposure. CAT is an antioxidant enzyme that appears to be less significant due to its relatively low affinity for H2O2, but it becomes an important enzyme when the concentration of H2O2 is raised. 65 The results from the present study showed decreased CAT activity. Decreased CAT activity in brain has been reported by other OPs 66 including DDVP. 63
GR reduces oxidized glutathione GSSG to GSH in a reaction that utilizes NADPH. The inhibition in the activity of GR might involve –SH group that is present at the active site of the enzyme. Alkylation of –SH groups by OPs has been documented. 67 GSH-Px protects the cells from the damaging effect of H2O2 by metabolizing hydroperoxides. 68 We observed a reduction in GSH-Px activity in all the brain regions following the development of OPIDN, suggesting the failure to metabolize toxic hydroperoxides. Inhibition of GSH-Px by OPs has also been reported earlier.69 –71
No effect was observed on the activity of GST in the brain regions of MCP- and DDVP-treated animals. Although induction in GST activity is reported by DDVP in brain, liver, kidney and intestine tissues of rats, 72 the failure to see induction in GST activity can be due to the fact that it might be induced early during the detoxification phase and then gradually returned to baseline by day 28 of exposure as most of the OPs would be detoxified during the early phase.
The decrease in the activities of antioxidants might be attributed to the increase in ROS50,73 or alkylation of –SH groups of proteins. 74 Another reason for increased oxidative stress is downregulation of antioxidant enzymes. Slotkin and Seidler 25 observed that mRNA expression of antioxidant enzymes SOD, GSH-Px and GR was repressed following OP exposure. The inhibition of all the major antioxidant enzymes would lead to increased oxidative stress in nervous system, which is detrimental to cell function/survival. Increased oxidative stress might contribute to a delayed neuronal apoptosis observed in OPIDN. Zhang et al. 27 have reported increased MDA levels and decreased antioxidant defenses (GSH, CAT, SOD, GSH-Px and GR) in cerebrum, spinal cord and sciatic nerve of hens treated with TOCP, a pesticide reported to cause OPIDN. The alterations in antioxidant enzymes were observed to be a time-dependent and tissue-specific effect.
To correlate the biochemical and behavioral alterations with histopathological changes, H&E staining was carried out. Pathological changes were observed in the present study in the brain regions of MCP- or DDVP-treated animals. The vascular endothelium, which regulates the passage of macromolecules and circulating cells from blood to tissue, is a major target of oxidative stress caused by free radicals, playing a critical role in the pathophysiology of the brain. 75 Sarin, a highly toxic ChE inhibitor and OPIDN inducer, has been shown to cause lesions in the mice brain. 76 Spinal cord of mice chronically treated with TOCP showed extensive degeneration of the axon and myelin of the ventral columns in the lumbar cord and of the lateral columns in the cervical cord. Histopathologic lesions in the peripheral nerves of mice chronically treated with TOCP showed axon and myelin degeneration. 77
The results from the study clearly demonstrate that accentuated MDA levels were accompanied by decrease in antioxidant defense (thiols, CAT, SOD, GSH-Px and GR) in the brain regions of OP-treated rats, which contribute in part to the development of OPIDN. The results suggest that antioxidants might be an adjuvant therapy to atropine and pralidoxime for the treatment of acute OP toxicity. Antioxidants like melatonin,78,79 N-acetylcysteine80,81 and vitamin E82,83 have been shown to be effective in attenuating OP-induced toxicity. However, further studies are needed to identify the effective antioxidants that can be beneficial in OPIDN.
Footnotes
Funding
This work was supported by a fellowship provided to Anwar Masoud by the Government of Yemen.
Acknowledgements
The authors thank Prof. S.N. Sanyal, Department of Biophysics, Panjab University, Chandigarh, in the preparation and critical evaluation of manuscript.
Declaration of Conflict of Interest
The authors declared no conflicts of interest
