Abstract
High blood pressure, obesity, abnormal lipid profile, which often coexist with diabetes, tend to be associated with preclinical cardiovascular abnormalities and may contribute to the association of diabetes with cardiovascular events. Many studies have proved that streptozotocin (STZ) is responsible for type-2-diabetes-induced cardiovascular complications. Long-term perindopril therapy in patients with hypertension and diabetes has been observed to correct carotid remodeling by reducing hypertrophy. We studied the effect of perindopril (1 mg/kg/d orally [po]) on cardiovascular complications in neonatal model of rats, which was induced by administering STZ (90 mg/kg, intraperitoneally [ip]), in 5-d-old wistar rats and cardiac hypertrophy induced by isoprenaline (ISO; 5 mg/kg, ip) for 10 d. Various biochemical, cardiac, and hemodynamic parameters were measured at the end of 8 weeks of treatment in diabetes model and 10 d in hypertrophy model. STZ produced hyperglycemia, hyperinsulinemia, dyslipidemia, hypertension, bradycardia, increased creatinine kinase (CK-MB), lactate dehydrogenase enzymes (LDH) and C-reactive protein (CRP) levels, cardiac hypertrophy, and oxidative stress. Chronic treatment with perindopril significantly prevented STZ-induced hyperglycemia and hyperinsulinemia and controlled dyslipdemia in diabetic rats. Further, perindopril produced a significant reduction in elevated levels of CRP, LDH, and CK. STZ-induced hypertension and bradycardia were also prevented by perindopril treatment. Perindopril also produced beneficial effect by preventing cardiac hypertrophy as evident from cardiac hypertrophy index and left ventricular hypertrophic index. Perindopril also prevented STZ-induced oxidative stress. Similar results were obtained in ISO-induced cardiac hypertrophic model, which confirms the beneficial role of perindopril in cardiac hypertrophy. In conclusion, our data from both studies suggest that perindopril produced beneficial effect on cardiac complications.
Introduction
Hypertension is very frequently associated with diabetic subjects, that is about 50% of diabetes patients, irrespective of whether they are type-1 or type-2, are hypertensive, and they contribute appreciably to cardiovascular morbidity and mortality by the acceleration of diabetic microvascular and macrovascular complications. 1 High blood pressure (BP), obesity, and abnormal lipid profile, which often coexist with diabetes, tend to be associated with preclinical cardiovascular abnormalities and may contribute to the association of diabetes with cardiovascular events. 2 Among patients with type-2 diabetes, coronary heart disease, myocardial infarction (MI), stroke and peripheral vascular disease are few important causes of mortality and morbidity. 3 Individuals with hypertension and diabetes have accelerated progression of these complications, and management of hypertension in these patients reduces the risks and progression of diabetes-associated complications. 4
Many studies have proved that streptozotocin (STZ) is responsible for type-2-diabetes-induced cardiovascular complications. STZ enters the β cell via a glucose transporter (GLUT2) and causes alkylation of DNA. DNA damage induces activation of poly–adenosine diphosphate (ADP) ribosylation, a process that is more important for the diabetogenicity of STZ than DNA damage itself. Poly-ADP ribosylation leads to depletion of cellular nicotinamide adenine dinucleotide (NAD+) and adenosine triphosphate (ATP). Enhanced ATP dephosphorylation after STZ treatment supplies a substrate for xanthine oxidase, resulting in the formation of superoxide radicals. Consequently, hydrogen peroxide and hydroxyl radicals are also generated. Furthermore, STZ liberates toxic amounts of nitric oxide that inhibits aconitase activity and participates in DNA damage. As a result of the STZ action, β cells undergo the destruction by necrosis. 5
In hypertensive patients, increased angiotensin II (Ang II) is known to impair insulin intracellular signaling 6 and pancreatic islet structure. 7 It has been suggested that Ang II may have a deleterious effect on glucose metabolism by increasing reactive oxygen species (ROS) and inducing inflammation, decreasing blood flow in many tissue beds, and stimulating the sympathetic nervous system. 8 Hypertension and impaired glucose tolerance leads to decrease in arterial compliance and structural and functional cardiac changes such as left ventricular hypertrophy (LVH) and left ventricular diastolic dysfunction.9,10
The antihypertensive efficacy of perindopril, a potent angiotensin-converting enzyme inhibitor (ACE) inhibitor, is well documented. Approximately 65% of patients attained a diastolic BP of <90 mmHg after 4 or 6 weeks of treatment; after 6 months, this had increased to more than 94% of patients in two of the studies.11,12 Long-term perindopril therapy in patients with hypertension and diabetes has been observed to correct carotid remodeling by reducing carotid artery wall hypertrophy and increasing internal diameter. 13 During the Diabetes Artery Perindopril Hypertension Normalization Excess Stiffness (DAPHNET) study, in which 57 patients with hypertension and type-2 diabetes were randomized to perindopril 4 mg/d or 8 mg/d, a dose-dependent reduction in carotid artery stiffness was observed that was independent of BP reduction. 14 In a double-blind, 1-year, parallel trial comparing perindopril to atenolol in untreated hypertensive subjects, perindopril increased small artery diameter and reduced the ratio of media thickness to lumen diameter, whereas atenolol had no effect. 15 In experimental animal studies, perindopril demonstrated protective effects against aortic endothelial cells 16 and endothelium-dependent relaxation. 17 Perindopril also enhanced myocardial angiogenesis in a model of metabolic syndrome in rats 18 and demonstrated an antitrophic effect in mesenteric arteries in diabetic rats. 19 In rats with STZ-induced diabetes, perindopril attenuated mesenteric vascular hypertrophy and it was suggested that this antitrophic effect resulted from the inhibition of transforming growth factor (TGF) expression. 20 In SHR diabetic rats, perindopril produced an improvement in insulin sensitivity, prevented dyslipidemia and cardiac dysfunction associated with STZ-induced diabetes in Wistar and SHR rats. 21 Despite these, direct study of perindopril in type-2 diabetic complications is not reported. Hence, in the present investigation, we have evaluated the effect of perindopril on cardiovascular complications associated with type-2 diabetes. Simultaneously we have analyzed the direct effect of perindopril on cardiac hypertrophy.
Materials and methods
The protocol of the experiment was approved by institutional animal ethics committee as per the guidance of committee for the purpose of control and supervision of experiments on animals (CPCSEA), Ministry of social justice and Empowerment, Government of India (Protocol No: IPS/PCOL/MPH10/003, Dated January 23, 2010). Animals used in the experiment were obtained from Zydus-Cadila Pharmaceuticals Ltd. All biochemical diagnostic kits were procured from Labcare Diagnostics Pvt. Ltd., India.
Isoprenaline-induced cardiac hypertrophy
Cardiac hypertrophy was induced by intraperitonial injection of isoprenaline (ISO; 5mg/kg/d) once daily for 10 d. Sprague Dawley rats of either sex were randomly divided into 4 groups: control (CON), control treated with perindopril (COP), disease control (DIC) and disease treated with perindopril (DIP). Perindopril was dissolved in distilled water and administered orally at a dose of 1 mg/kg/d with food and water ad libitum.
Neonatal type-2 diabetes
Five-day-old Wistar neonates of either sex were injected with 90 mg/kg intraperitoneal (ip) STZ (Sigma Ltd., USA) in 0.9% sodium chloride solution. CON neonates received equivalent amount of isotonic saline. The neonates were left with their own mothers and weaned at 4 weeks of age. Twelve weeks after the injection of STZ, the animals were checked for fasting glucose levels with the help of available diagnostic kit (Labcare Diagnostics Pvt. Ltd., India). The animals showing fasting glucose levels >170 mg/dl were considered as diabetic and divided into 4 groups: CON, COP, DIC and DIP. Perindopril was dissolved in distilled water and administered orally at a dose of 1 mg/kg/d with food and water ad libitum for 8 weeks.
Blood sample collection and serum analysis
At the end of 10th day and 8 weeks of treatment, blood was collected from the retro orbital plexuses under light ether anesthesia. Serum samples were analyzed for glucose (GLUCOSE-SLR enzymatic colorimetric test GOD-POD), cholesterol (CHOLESTEROL-SLR enzymatic colorimetric test), high-density lipoprotein–cholesterol (HDL-C; HDL CHOLESTEROL direct enzymatic colorimetric, Liquid), triglycerides (TRIGLYCERIDES enzymatic colorimetric test—GPO PAP), C-reactive protein (CRP; CRP-SRL enzymatic colorimetric test), lactate dehydrogenase (LDH; LDH-SLR kinetic test) and creatinine kinase (CK-MB; CK-MB-SLR measurement of CK-MB activity serum and plasma) spectrophotometrically (Shimadzu UV-1601, Japan). Serum insulin was estimated for diabetic model by radioimmunoassay technique using diagnostic kits obtained from Board of Radiation and Isotope Technologay, Mumbai, India, using gamma counter (Packard, USA).
Hemodynamic and cardiac parameters
Hemodynamic parameter viz. BP, heart rate, rate of pressure development and decay were recorded by invasive method of carotid artery cannulation (Iworx 118 System Inc., USA). After withdrawal of blood samples from retro-orbital plexus and recording hemodynamic parameters, animals were sacrificed, heart and femur were excised, and extraneous tissues were separated. Wet weight of the entire heart, left ventricle and length of femur was recorded to calculate the index of cardiac hypertrophy as wet heart weight to femur length ratio and the LVH index as left ventricular weight (LVW) to heart weight ratio. As described earlier, quantification of LV glutathione (GSH) 22 and malondialdehyde (MDA) levels 23 was carried out.
Reduced GSH measurement
LV tissue was homogenized in 50 mmol/L Tris-HCI buffer (pH 7.4). The homogenate was centrifuged sequentially at 10,000g for 20 min, then at 100,000g for 60 min. To 0.5 ml of tissue supernatant, 1.5 ml of 0.2 mol/L Tris-HCI buffer (20 mmol/L EDTA, pH 8.2), 0.1 ml of 0.01 mol/L of 5,5’-dithiobis-(2-nitrobenzoic acid) and 7.9 ml of methanol were added. The mixture was incubated at 37°C with occasional shaking for 30 min. The mixture was then centrifuged at 3,000g for 15 min and the absorbance of the supernatant was determined at 412 nm. 22
MDA measurement
The supernatant, 0.5 mL, of homogenized LV tissue and 2.5 ml of 10% trichloracetic acid were added into the tubes and mixed. After incubating for 15 min at 90°C and cooling with cold water the mixture was centrifuged at 3000 rpm for 10 min. Two milliliters of supernatant were taken and 1 ml of 0.675% TBA was added. The tubes were sealed and incubated at 90°C for 15 min and then cooled to room temperature. The optical density was measured at 532 nm by a spectrophotometer. 23
Statistical analysis
Results are expressed as mean ± SEM. Statistical differences between the means of the various groups were evaluated using Student’s t test by SPSS 16 software. Data were considered statistically significant at p < 0.05.
Results
General features
Diabetic rats showed significant decrease in body weight at the end of eighth week as compared to the CON group. Chronic treatment with perindopril did not alter the body weight of CON rats. Diabetic rats also showed significant increase in food and water intake at the end of eighth week as compared to the CON group. Chronic treatment with perindopril produced a significant decrease in food and water intake (Table 1).
Effects of perindopril on glucose, insulin and cardiac parametersa

Each group contains 6 numbers of animals.
a Values are expressed as Mean ± SEM.
b Significantly different from control (P < 0.05).
c Significantly different from diabetic control group (P < 0.05).
CON, control; COP, control treated with perindopril; DIC, diseased control; DIP, diseased treated with perindopril; CRP, C-reactive protein; LDH, lactate dehydrogenase; CK-MB, creatinine kinase.
Biochemical parameters
STZ-induced diabetic rats were found to exhibit significant hyperglycemia and hyperinsulinamia as compared to the CON rats. Chronic treatment with perindopril produced significant decrease in elevated serum glucose and insulin levels.
There was a significant increase in total cholesterol, very-low-density lipoprotein (VLDL), atherogenic index and triglycerides levels, and significant decrease in HDL-cholesterol levels in STZ diabetic rats as well as hypertrophic rats compared to the CON rats. Chronic treatment with perindopril showed a significant reduction in the levels of total cholesterol, VLDL, atherogenic index and TG, with a significant increase in HDL-C levels in both the models (Table 2).
Effect of perindopril on lipid profilea

Each group contains 6 numbers of animals.
a Values are expressed as Mean ± SEM.
b Significantly different from control (p < 0.05).
c Significantly different from diabetic control group (p < 0.05).
CON, control; COP, control treated with perindopril; DIC, diseased control; DIP, diseased treated with perindopril; HDL, high-density lipoprotein; VLDL, very-low-density lipoprotein.
Cardiovascular parameters
Serum cardiac biomarkers
STZ-induced diabetes produced a significant increase in the levels of serum LDH, CK and CRP as compared to CON rats. Chronic treatment with perindopril significantly reduced the elevated levels of serum LDH, CK and CRP in diabetic rats (Table 1).
Cardiac parameters
The cardiac hypertrophy and left ventricular weight (LVW) to heart weight ratio which is a measure of LVH index was significantly high in hypertrophic and diabetic animals as compared to DIC animals (Figure 1A and B). Chronic treatment with perindopril significantly reduced cardiac hypertrophy index and LVH index of diabetic and hypertrophic rats. LVW to total body weight (LVW/TBW) and LVW to right ventricle weight (LWV/RVW) ratios were also found to be significantly higher in both hypertrophic and diabetic rats than that of the the CON rats. Chronic treatment with perindopril significantly reduced the LVW/TBW and LWV/RVW ratios of diabetic and hypertrophic rats (Table 3).

A, Effect of perindopril on the change in cardiac hypertrophy index. Each bar represents mean ± SEM of six experiments. *Significantly different from control. #Significantly different from DIC. B, Effect of perindopril on the change in left ventricular hypertrophy index. Each bar represents mean ± SEM of six experiments. *Significantly different from control. #Significantly different from DIC. CON, control animals; COP, control animals treated with perindopril (1 mg/kg/d); DIB, diseased animals; DIP, diseased animals treated with perindopril (1 mg/kg/d).
Effect of perindopril on hemodynamic and cardiac parametersa

Each group contains 6 numbers of animals.
a Values are expressed as mean ± SEM.
b Significantly different from control (p < 0.05).
c Significantly different from diabetic control group (p < 0.05).
CON, control; COP, control treated with perindopril; DIC, diseased control; DIP, diseased treated with perindopril; LVW, left ventricular weight; RVW, right ventricular weight; BW, body weight.
Oxidative stress
STZ-induced diabetes and ISO produced a significant increase in serum MDA levels as compared to CON rats (Figure 2A). Chronic treatment with perindopril significantly reduced the elevated serum MDA in diseased rats. STZ-induced diabetes and ISO also produced a significant decrease in serum GSH levels as compared to CON rats. Chronic treatment with perindopril significantly increased the reduced serum GSH in diseased rats (Figure 2B).
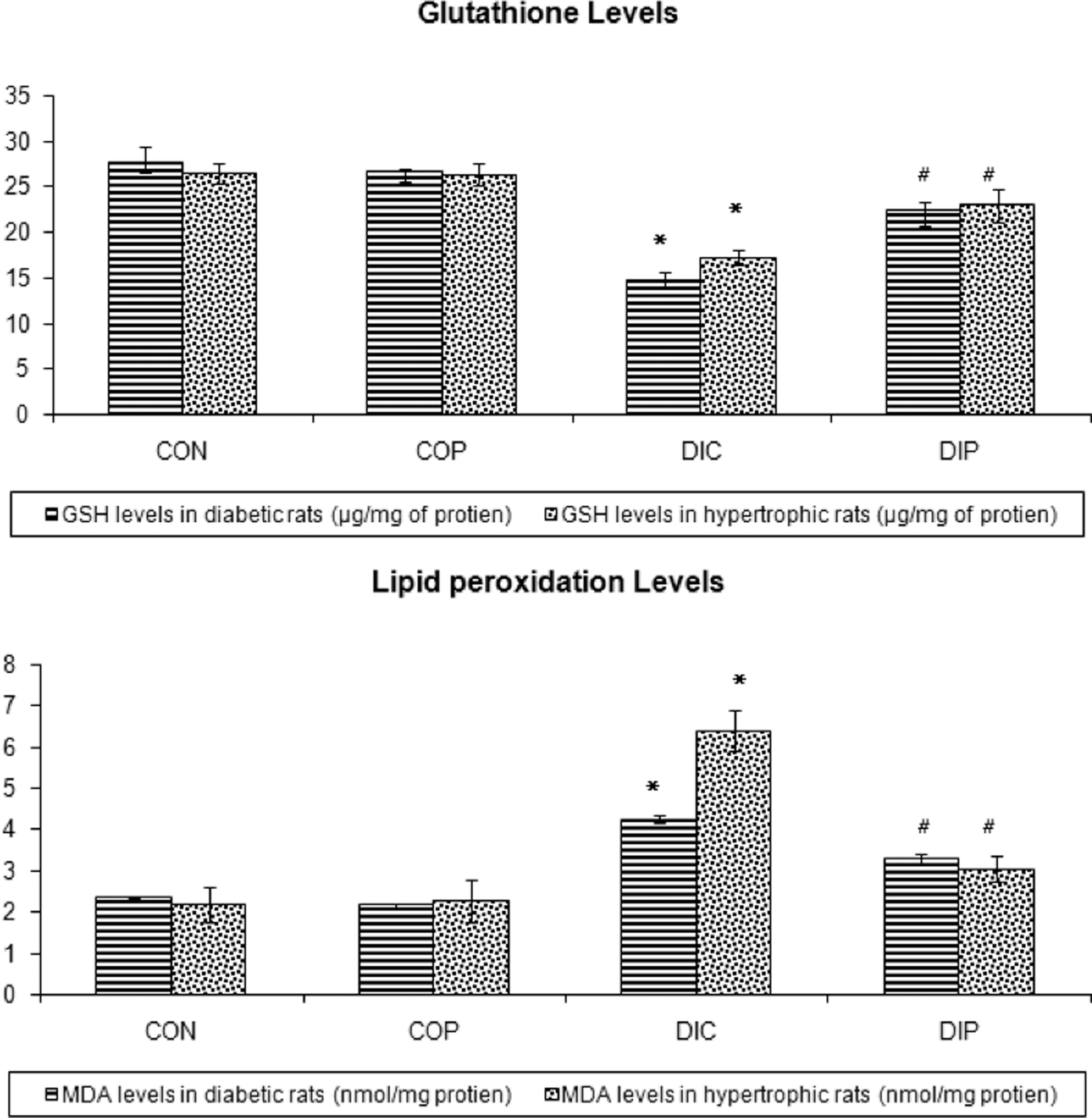
A, Effect of perindopril on serum glutathione levels. Each bar represents mean ± SEM of six experiments. *Significantly different from control. #Significantly different from DIC. Figure .2. B, Effect of perindopril on lipid peroxidation. Each bar represents mean ± SEM of six experiments. *Significantly different from Control. #Significantly different from DIC. CON, control animals; COP, control animals treated with perindopril (1 mg/kg/d); DIB, diseased animals; DIP, diseased animals treated with perindopril (1 mg/kg/d).
Hemodynamic parameters
STZ-induced diabetic rats exhibited significantly increase in BP and significantly decrease in heart rate. Chronic treatment with perindopril in diabetic rats exhibited significant decrease in BP and significant increase in heart rate (Table 3). Rate of pressure development and decay was significantly decreased in diabetic control rats. Treatment with perindopril significantly increased the rate of pressure development and decay (Figure 3). Similar results were also found in ISO-induced cardiac hypertrophy model.

Effect of perindopril on the rate of pressure development and decay. Each bar represents mean ± SEM of six experiments. *Significantly different from Control. #Significantly different from disease control. CON, control animals; COP, control animals treated with Perindopril (1 mg/kg/d); DIB, diseased animals; DIP, diseased animals treated with perindopril (1 mg/kg/d).
Discussion
Hypertensive diabetics have been reported to have more cardiovascular disease with 6–7 time higher mortality rate compared to normotensive diabetics 24–26 In the present investigation, we found that STZ produced cardinal signs and characteristics of diabetes, viz. loss of body weight, polyphagia, polyuria and polydipsia. Chronic treatment with perindopril significantly decreased polydipsia and polyphagia.
Insulin resistance and hyperinsulinemia have been postulated to play a significant role in the etiology of hypertension. 27 Insulin resistance and hyperinsulinemia appear to increase the risks of diabetes, dyslipidemia and hypertension, 28 and are implicated in atherogenesis, either via the increase in proinflammatory cytokines implicated in insulin resistance or via the direct proliferative effects of insulin itself on arterial smooth muscle cells. 29 In the present investigation, STZ-induced diabetic rats exhibited significantly increased glucose and insulin levels, and chronic treatment with perindopril in diabetic rats significantly decreased the insulin and glucose levels. Ang II is responsible for blocking the differentiation of adipocytes, leading to accumulation of lipids in muscle, liver and pancreas and, in turn, reducing insulin sensitivity. 30 Thus, perindopril, being an ACE inhibitor, might prevent ANG-II-induced phosphorylation of insulin, thereby controlling glucose and insulin levels. In a genetically obese animal model of type-2 diabetes, blockade of the renin angiotensin aldosterone system (RAAS) with perindopril attenuated changes in islet cell structure and insulin secretion. 31 Fogari et al. 32 found that perindopril, but not the angiotensin receptor blocker losartan, improved insulin sensitivity and reduced fibrinogen levels in overweight patients with hypertension. Thus, our reports are in consistence with several clinical reports suggesting beneficial role of perindopril in glycemic control.
Hypertriglyceridemia and low HDL-cholesterol levels are common in patients with type-2 diabetes or insulin resistance. 33 In insulin-resistant state, endothelial cell lipoprotein lipase activity is decreased, as is the conversion of cholesterol ester–enriched VLDL to LDL, which is injurious to endothelial cells after receptor-mediated uptake 34 and contribute to endothelial dysfunction. 35 In the present investigation, we have also found that serum cholesterol and triglyceride levels were significantly increased in hypertrophic and STZ-induced diabetic rats and treatment with perindopril significantly reduced the elevated cholesterol and triglyceride levels. Various studies suggest that renin–angiotensin system is responsible for hyperlipidemia and atherosclerotic disease.36,37 Endothelial dysfunction occurs when there is an upset in the balance of Ang II and growth factors. Ang II facilitates oxidation of LDL and its uptake by scavenger receptor on monocytes/macrophages and upregulates lipooxygenase as a basis of enhanced endothelial uptake of oxidized LDL. 38 Thus, perindropril, being an ACE inhibitor, might inhibit the Ang II-mediated oxidation of LDL. Fennessy et al. 39 have reported that administration of perindopril during the 6 weeks of cholesterol feeding reduced lipid accumulation in developing atheromatous plaques of the carotid artery and in fatty streaks of the aorta in rabbit model of cholesterol-induced atherosclerosis. Thus, our reports are in consistence with the other studies that perindopril prevents dyslipidemia, which in turn suggests its beneficial role in endothelial dysfunction and atherosclerotic diseases.
CRP, an acute phase reactant, produced mainly by liver and selectively binds to LDL, is found within atheromatous plaques and enhances complement activation. 40 CRP levels are elevated in type-2 diabetes. 41 In a similar study, Goyal and coworkers found that STZ-administered group possessed high level of CRP, a important cardiac biomarker. 42 In present study, we found a significant rise in CRP level in STZ-induced diabetic rats as compared to normal rats. Treatment with perindopril significantly decreased CRP levels in diabetic rats. Ang II has significant proinflammatory actions in the vascular wall, inducing the production of ROS, inflammatory cytokines, and adhesion molecules, elevating the CRP levels. Enhanced production of angiotensinogen, in turn, supplies more substrate to the activated vascular ras angiotensin system (RAS), wherein the locally produced Ang II synergizes with oxidized lipid to perpetuate atherosclerotic vascular inflammation and increases CRP levels. 43 Perindopril, being an ACE-inhibitor, inhibits Ang II-induced proinflammation and thereby decreases the CRP levels. This suggests that perindopril may serve as an important inhibitor of inflammatory cytokines in the myocardium.
The CK-MB is part of total CK and more specific for cardiac muscle than other striated muscle. CK-MB and LDH levels are reported to be increased in animal model of diabetes induced by STZ. 44 In our study, we found significant increase in LDH and CK levels in STZ-induced diabetic rats as compared to normal rats. Treatment with perindopril significantly reduced LDH and CK levels and further substantiates its beneficial effect by reducing the cardiovascular risk in diabetes mellitus. The activities of LDH and CK as well as lactate concentrations in the coronary venous effluent were reduced by 39.3%, 50%, and 60.6%, respectively, after chronic treatment with perindopril in stroke-prone SHR hypertensive rats, indicating the beneficial effect in cardiac damage. 45
Diabetes is associated with the activation of the RAAS and systemic nervous system (SNS), leading to compensatory changes in the size and shape of the cardiac chambers through cellular hypertrophy or remodeling. It has been reported that STZ induced cardiac hypertrophy, aggravating the cardiovascular complications. 46 A total of 72% of the diabetic patients are found to have LVH, whereas only 32% of the nondiabetic patients have LVH. LVH does not usually occur in diabetes unless hypertension coexists. Consequently, left ventricular mass index was greater in patients with hypertension and diabetes mellitus than in those without diabetes mellitus. 47 Also the ISO injection causes hypertrophic effect on heart via TGF-β 48 and stimulation of protein synthesis. 49 In the present study, cardiac hypertrophy index was found to be increased in both hypertrophic and STZ-induced diabetic hearts. Heart weight to body weight ratio and LVW/RVW ratio were also found to be higher in diseased hearts than control hearts in both models. Treatment with perindopril significantly decreased cardiac hypertrophy index, LVH index, heart weight to body weight ratio and LVW/RVW ratio in diseased rats. Ang II is reported to cause hypertrophy of cardiac myocytes and hyperplasia of cardiac nonmyocytes. 50 In STZ-induced diabetic rats with type-1 diabetes, perindopril attenuated mesenteric vascular hypertrophy, and it was suggested that this antitrophic effect resulted from inhibition of TGF-β expression. 51 Thus, perindopril might inhibit the expression of hypertrophic genes and thus provide beneficial role in cardiac dysfunction.
STZ causes increased production of ROS due to the autooxidation of monosaccharides, leading to the production of superoxide and hydroxyl radicals. 52 Reduced antioxidant capacity has been reported in cardiac tissue from diabetic animals. 53 In present study, we found a significant increase in pro-oxidant (MDA) levels and decrease in antioxidant (GSH) levels in STZ-induced diabetic and hypertrophic rats. Chronic treatment with perindopril showed a significant increase in antioxidant (GSH) and significant decrease in MDA levels in STZ-induced diabetic rats. Both experimental and clinical studies have provided functional evidence for stimulation of the renin–angiotensin system and simultaneously for activation of an NAD(P)H oxidase in the vascular wall. Activation of NAD(P)H oxidase and increased superoxide within the vascular wall have important implications for both the progression of atherosclerosis and the development of clinical event.47,54 The ACE inhibitors reduce Ang II levels and increase bradykinin levels, suggesting that perindopril have important implications for vascular oxidative stress.48,55 It has been established that perindopril reduced oxidative stress and increases the plasma antioxidant capacity in a prospective, randomized, parallel group study assessed in hypertensive patients. 55 Thus, perindopril may be beneficial in oxidative stress and lipid peroxidation during MI.
Increase in BP after treatment with STZ and ISO has been reported by several studies. 56 Hypertension is very frequently associated with diabetic subjects, 2 that is about 50% of patients with diabetes, irrespective of whether they are type-1 or type-2, are hypertensive. Several reasons are postulated for the development of hypertension in diabetes. One of the reasons for hypertension is loss of the autoregulatory (RAAS and SNS) response, which can amplify the damaging effects of systemic BP on small blood vessels. 57 Further, potentiation of bradykinin, a powerful vasodilator that enhances insulin signaling 58 and translocation of the glucose transporter in skeletal muscle, may be responsible for some of the hemodynamic and nonhemodynamic effects of ACE inhibition.59,60 Of the ACE inhibitors, perindopril has the capacity to increase the ratio of bradykinin and Ang II. 61 In the present study of chronic treatment with ACE inhibitors, perindopril significantly decreased the BP in treated rats compared to diseased rats in both models. Oral administration of perindopril has been demonstrated to be an effective BP-lowering agent in adult patients with mild-to-moderate hypertension in numerous randomized clinical trials. 62 Bradycardia has been frequently observed in STZ diabetic rats 63 as well as in ISO-induced rats. 64 The development of STZ-induced bradycardia has been attributed to a downregulation of myocardial beta adrenoceptors, hypothyroidism, 65 depression of myocardial calcium metabolism, 66 reduced uptake of calcium by the sarcoplasmic reticulum and concomitantly depression of SR-calcium ATPase activity. 67 It is well established that Ang II exerts an inhibitory influence on baroreceptor reflex control of heart rate after either intravenous administration in several species. 68 Perindopril is an ACE inhibitor and it is possible that it may attenuate the Ang II-mediated inhibition of baroreceptor reflex. This may be the possible mechanism involved in the increased heart rate by perindopril treatment in hypertrophic rats in both the models.
In the present investigation, STZ and ISO injection decreased the rate of pressure development and decay, which is the direct indicator of myocardial contractility dysfunctioning and cardiac fibrosis. In present study, chronic treatment with perindopril significantly increased the rate of pressure development and decay. The data presented in a very interesting study clearly demonstrate that overexpression of the fast-twitch skeletal muscle SR Ca-ATPase and increase in the rate of pressure development and decay (+dp/dt and −dp/dt) in the mammalian heart results in significantly elevated Ca transport function and cardiac contractile performance. This study suggests that the SERCA pump level is a critical determinant of myocardial contractility. 69 STZ-induced diabetes in animal models causes changes in myocardial cellular calcium transport and contractile proteins, which result in subclinical myocardial contractility dysfunction.70,71 Thus, perindopril appears to be beneficial in cardiac dysfunctioning.
From the above investigation we can concluded that perindopril prevents not only the STZ-induced metabolic abnormalities but also cardiovascular complications. Perindopril is also found to be beneficial in hypertrophy, which can make it an optimistic treatment in type-2 diabetes as well as in cardiac hypertrophy-induced cardiovascular complications.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
