Abstract
Data previously reported on the whole-body retention of aluminium-26 (26Al) in a male volunteer are extended to 8 years after intravenous administration as citrate, when only ~2% of the injected tracer remained. The extended data, combined with a report elsewhere of the late urinary and faecal excretion of 26Al by this subject, reinforce indications that transdermal losses contribute to the clearance of systemic aluminium and mitigate its long-term accumulation from chronic exposure.
Background
Previously, AEA Technology conducted a study of the metabolism of the radioactive tracer aluminium-26 (26Al; half-life 716,000 years) injected as citrate into a volunteer. 1 Data were presented up to 2.5 years on the whole-body retention of the tracer and on its concentration in blood, with tabulations of urinary and faecal excretion up to 13 days. Subsequently, the retention study continued up to 8 years and an equation mistakenly representing these unpublished results was adopted in a review by Priest, 2 attracting criticism 3 that details of the extended investigation were unavailable for scrutiny. In this account, we respond to the criticism and report that the long-term urinary and faecal excretion rates reported by Priest 2 fall well short of the contemporary rates of loss from the whole body.
Methods
A detailed account of the administration procedures was given previously. 1 In summary, 0.7 μg of 26Al, carrier free as citrate, was injected into an antecubital vein. The volunteer was a healthy Caucasian male, of height 1.83 m, weight 77 kg and age 41 years, who had given informed assent following approval of the study by AEA Technology’s independently constituted ethics committee. The anticipated radiation dose, assessed by a Dosimetry Service certificated by the UK Health and Safety Executive, was 65 μSv, similar to that incurred from enhanced cosmic-ray exposure during a return transatlantic flight. 4
The previous account 1 detailed the methods adopted in studying the biokinetic behaviour of the injected tracer. We shall, however, reiterate and enlarge upon the analytical procedures employed in assessing the whole-body retention; we do this because, in the later stages of the study, the reliable quantification of the residual deposit became increasingly complicated as it approached the limit of detection.
The equipment 5 comprised an array of six scintillation detectors, each of 152 mm diameter × 89 mm thickness; four of the detectors were mounted above and two below the supine subject, inside a room shielded on all sides by 100 mm lead. We did not use the 54-detector geometry mentioned in the review. 2 Assessment of the retained 26Al was based on the spectral response recorded in 30 min from its 1.81 MeV γ-radiation, after subtraction of contributions from the environmental background radiation. The latter were obtained on each occasion from the spectra recorded, shortly before and/or after the subject’s spectrum, in the presence of an inactive ‘phantom’; this consisted of a series of water-filled plastic cylinders of circular or elliptical cross section, which together simulated the human form. 6 The retention was given by the net response attributable to 26Al, expressed as a fraction of that recorded shortly after the injection, before any voiding of urine or faeces.
Results
Estimates of whole-body retention are shown in Figure 1, together with the statistical uncertainties (1σ) arising from the random nature of radioactive decay. With the declining levels of 26Al retained, these uncertainties increased during the study, resulting in a widening scatter of neighbouring points, but positive values were nevertheless obtained on every occasion.
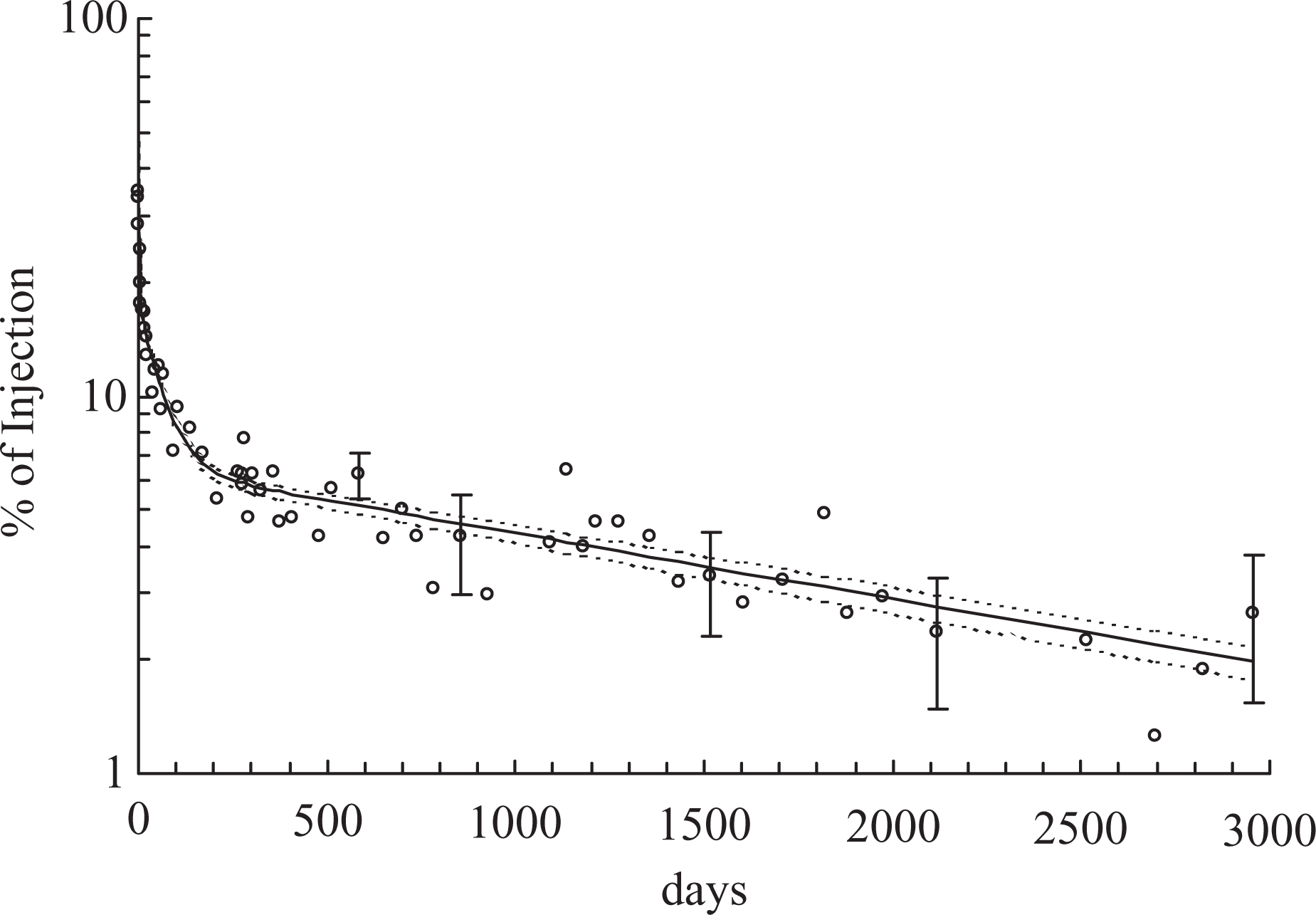
Whole-body retention of injected 26Al. Error bars show uncertainties (1σ) from counting statistics. The solid curve is the three-component exponential function (equation (1)). Broken curves show similar functions fitted to the data with differing assumptions about background effects from cosmic-ray-induced neutrons.
In addition to these random errors, the possibility of bias had to be considered. This was potentially important in the later stages, when the 26Al signal provided only a marginal increment to the background. In the relevant 1.81 MeV region, there would be no detectable interference from other γ-ray emitters in the body: the natural radionuclides of the radium and thorium series do contribute in this region but are ordinarily present at such low levels 7 as to be irrelevant in this context.
However, there was an additional source of potential bias. One component of the background response arises from evaporation neutrons generated by cosmic ray interactions in the lead shielding; these neutrons in turn interact with hydrogen nuclei in the subject (or phantom) leading to γ-radiation that interferes in the 1.81-MeV region. 8 In principle, if the phantom and subject are similar in physique and in hydrogen content, there should be little or no effect on the response after background subtraction, but confirmation was important. This was achieved in two ways. First, records were examined of 15 spectra obtained from the subject in other studies, 9 with their associated background measurements, during the 2 years before he received the 26Al. The mean response after background subtraction, expressed as the equivalent percentage of the subsequent injection, was 0.17 ± 0.23 (SEM); the SD (0.88) accorded satisfactorily with the uncertainty (±1.1) in individual measurements from counting statistics. Second, spectra from 17 other men, who had no exposure to 26Al and who closely matched the subject in weight and height, were treated similarly. These showed an average response equivalent to −0.25 ± 0.25 (SEM)% of the injection, with again close agreement between counting statistics and observed scatter about this mean.
Neither mean result was large in relation to the observed late percentage retention of 26Al (mean of 5 results, days 2115–2954 = 2.25 ± 0.24) but an assumed ‘bias’ of 0.17%, derived from the pre-exposure spectra from the subject himself, was nevertheless subtracted before the data of Figure 1 were plotted.
Discussion
Analysis of retention data
The values in Figure 1 are represented adequately by a three-component exponential function of time, shown as the solid curve in Figure 1
The final term in equation (1) represents an apparent component with a clearance half-life approaching 5 years but cannot reliably be attributed to clearance from a single biokinetic compartment. Skeletal deposits are reported to account for more than half of the total body aluminium in Reference Man. 10 The long-term loss of the tracer will be influenced by its removal from bone, with a range of rate constants reflecting inter alia the broad spectrum of resorption rates 11 within an individual skeleton. Consequently, the curves in Figure 1 might be expected to flatten over the subsequent decade, as is found 9 with other bone seekers, such as, barium-133. Nevertheless, a short extrapolation of equation (1), beyond the 8-year period of observation, is unlikely to be seriously in error.
Comparison with reported excretion
The daily reduction in the retained tracer, again expressed as a percentage of the injected quantity, is given by the first derivative of equation (1)
Implied transdermal losses?
This disparity points to some additional, dominant mechanism in the clearance of systemic aluminium at late times after deposition. Some potential routes are readily dismissed, if the information given for Reference Man 10 is credible. Losses through hair growth are viewed as negligible, and removal through the shedding of dead skin appears an unlikely candidate. The desquamating epidermis of Reference Man 10 is estimated to lose about 200 g of tissue annually, or about 8% of his total mass of skin, which is assumed to contain 5.2 mg aluminium. We found no data for the relative concentrations of endogenous aluminium in dermis and epidermis; if these were assumed equal, the daily loss would amount to ~1 µg, or roughly what is implied 10 would typically be present in 10–20 mL of urine.
Transdermal uptake of topically applied aluminium is known to occur,12,13 and losses via sweat offer a more credible explanation. The estimated balance for Reference Man 10 shows 10 times greater daily perspired aluminium (1 mg) than is found in urine (0.1 mg), although this estimate of sweat loss is extrapolated from observation under unusual environmental conditions. 14 Recently, Genuis et al. 15 have reported median concentrations of aluminium, for 20 subjects, of 4.6 mg L−1 (induced sweat) and 0.9 mg L−1 (urine), implying much greater daily excretion by both routes than is suggested for Reference Man 10 ; these results would also imply that transdermal losses dominate. By contrast, Omokhodion and Howard 16 found mean levels of aluminium in sweat of only 15 µg L−1 in 15 subjects exercising at room temperature. These various, apparently conflicting data may reflect differences in methodology, particularly relating to precautions against contamination with adventitious aluminium. We note, in the study of Genuis et al., 15 a much reduced intersubject variability in the data for blood, compared with that found for their samples of urine and sweat, which their subjects collected unsupervised.
Reliable and consistent evidence may at some stage emerge from analyses of the stable element in body fluids, but could have only indicative bearing on the 17-fold mismatch between our subject’s measured long-term excretion 2 of the tracer and the result from equation (2). The systemic speciation of stable aluminium acquired from chronic exposure cannot be expected to resemble that of the tracer 8–9 years after administration. Indeed, there was no evidence, during the early stages of this study, 1 of any large-scale inconsistency between measured excretion and whole-body retention—a strong indication that the supposed losses via sweat became dominant only after a changing speciation of the tracer had impaired its renal clearance. The beginnings of this impairment can be seen in the early stages of this study 1 and also in a similar study 17 of reduced duration.
Implications for long-term accumulation of systemic aluminium
The inferred dominance of transdermal losses in the long-term elimination of systemic aluminium is based on observations in one subject only, but the magnitude of the excess suggests that it is unlikely to be wholly absent in others. Curiously, the review 2 entertains the possibility of transdermal losses when discussing the behaviour of ingested aluminium (Priest’s Figure 21), but they do not feature in Middlesex University’s biokinetic model 2 for systemic aluminium (his Figure 19) based on the discrepant data for this one subject. This model was used 2 to predict that chronic exposure to aluminium would lead after 50 years to an accumulated deposit of 417 times the daily systemic intake. Whether or not we choose to relate the 17-fold disparity in these data to losses in sweat, this estimate is patently unreliable, potentially exaggerating the long-term accumulation.
Footnotes
Acknowledgements
Dr ND Priest instituted the initial study 1 during his previous affiliation to AEA Technology, with funding from the Aluminum Association, Washington, DC, and from the International Primary Aluminium Institute, London. The extended study reported here received no formal external funding, but employed data secured incidentally by AEA Technology during an unrelated project supported by the UK Department of Health. The authors acknowledge the sustained cooperation of the volunteer and benefited from stimulating discussions with Dr Chris Exley of Keele University and with Prof. David Taylor of Cardiff University.
Authors’ Note
Current address of RJ Talbot is Nuvia Ltd, 351.28 Harwell, Didcot, Oxon, OX11 0QJ, UK.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.


