Abstract
The aim of this study was to investigate the role of hydroxychloroquine (HCQ)-induced oxidative stress on sciatic nerve and muscle tissues of rats. The oxidant/antioxidant parameters in the sciatic nerve and muscle tissues were analyzed, and stereological analysis of the sciatic nerve was performed. Levels of malondialdehyde and nitric oxide in the tissues were significantly higher in the HCQ group than in the control group (p < 0.05). In addition, activities of superoxide dismutase and glutathione peroxidase were found to be significantly higher in the HCQ group than the control group (p < 0.05). There were significant decreases in nerve fiber diameter and myelin sheet thickness in the HCQ group compared with the control group (p < 0.05). These results revealed that HCQ might increase oxidative stress on sciatic nerve and muscle tissues of rats, which may correlate with axonal atrophy in sciatic nerves.
Introduction
Chloroquine (CQ) and hydroxychloroquine (HCQ) are derivatives of 4-aminoquinolone used for antimalarial treatment. In addition, they have been used for the treatment of several autoimmune diseases such as rheumatoid arthritis and systemic lupus erythematosus. 1,2 The molecular structure of HCQ is similar to that of CQ; however, the use of HCQ has increased preferentially because it is more effective and less toxic than CQ. In rare cases, both may cause serious toxicity. 3 The literature reports generally have focused on the well-known complication of retinotoxicity; whereas neuromyotoxicity has received less attention. 4,5 Both CQ and HCQ may cause serious neuromyotoxic side effects involving peripheral nerves and muscles. 5 –7 It has been reported that the long-term use of CQ and HCQ may cause neuromyotoxicity including dysphagia, neuropathy, myopathy, and polyradiculoneuropathy. 5 –9 The rat sciatic nerve is generally used for studying peripheral nerve injuries. 10,11 Drug-induced neuropathy is known to produce reactive oxygen species (ROS) in sciatic nerve and muscle tissues, and increased level of ROS may cause damage to sciatic nerve and muscle tissues. ROS oxidize membrane lipids, proteins, and DNA, thereby, resulting in cellular dysfunction and even in cell death. 12,13
The pathogenesis of HCQ-induced neuromyotoxicity is not clear. Oxidative stress may play an important role in tissue toxicity caused by various drugs such as CQ and methotrexate. 14,15 CQ may mediate oxidative stress and affect antioxidant enzymes in humans. 16 –18 Both HCQ and CQ may increase lysosomal pH leading to lysosomal enzyme inhibition. 4 Although the structures and action mechanisms of HCQ and CQ are similar, the role of HCQ in oxidative stress on the nerve and muscle tissues of rats is unknown. Under normal physiological conditions, tissues produce free oxidative radicals. For example, nitric oxide (NO) is produced by the oxidation of ammonia and malondialdehyde (MDA), which are produced during lipid peroxidation. The tissue injury caused by free radicals is prevented by an antioxidant defense system. The most important antioxidant enzymes are superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px), which react with free oxygen radical molecules to prevent tissue injury. 19,20
To the best of our knowledge, there has been no study on the role of oxidant/antioxidant parameters in the pathogenesis of HCQ-induced toxicity of sciatic nerve and muscle tissues of rats. The aim of this study was to investigate the role of HCQ-induced oxidative stress on sciatic nerve and muscle tissues of rats and to understand the effects of oral administration of HCQ on the sciatic nerve fiber morphology of rats.
Materials and methods
The animals included in the study were maintained and used in accordance with the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals. The Medical Faculty Experimentation Ethics Committee approved the experimental procedures of the study. A total of 14 male rats (Wistar albino type) were used in the experiment. Their age was between 8 and 12 weeks; the average of weight was 230 ± 20 g. The rats were placed in a controlled room, where the temperature was 22 ± 2°C and humidity was 60 ± 5%. Before starting the experiment, 12-h light/12-h dark cycle was maintained for 2 weeks. A commercially balanced diet (Bil Yem, Turkey) and tap water were provided ad libitum. The rats were randomly divided into two experimental groups as follows: Group I, control group (n = 7) and Group II, HCQ group (n = 7). In the control group, 1 ml distilled water was given by intragastric gavage for 12 weeks. Similarly, in the HCQ group, HCQ (160 mg/kg/day) was given by intragastric gavage for 12 weeks. 21 At the end of the study, all rats were anesthetized with an intramuscular injection of 50 mg/kg ketamine hydrochloride (Ketalar, Eczacibasi, Istanbul, Turkey) and killed followed by obtaining 5–10 ml blood. Bilateral 3 cm incisions in gluteal muscles were performed with dissection. Bilateral sciatic nerve and muscle tissues were explored and quickly removed without damaging the tissues. The muscle and sciatic nerve tissues taken from the right side were stored at −20°C until the analysis of MDA, NO levels and the measurements of SOD, paraoxanase (PON1), CAT and GSH-Px activities were performed.
Stereological analyses
The distal blocks of the left sciatic nerves were removed. The nerves were stretched to in situ length by pinning onto a card and then fixed with 2.5% glutaraldehyde in 0.1 M phosphate buffer (pH 7.4) for 4–6 h at 4°C. Once they were fixed, they were rinsed in a phosphate buffer (pH 7.4). After these steps, specimens were postfixed in 1% osmium tetroxide for 2 h, dehydrated in an ascending alcohol series, and put into propylene oxide for 16 min. After these procedures, the tissues were embedded in Epon Embedding Kit (Fluka Chemie Gmbt, Swithzerland). Semithin and ultrathin sections (1 µm and 90 nm thickness, respectively) were cut by an ultra microtome (Super Nova Reichert-Jung, Austria) and with 1% toluidine blue (semithin sections) and uranyl acetate–lead citrate (for ultrathin sections). Ultrathin sections were analyzed using a JEM-1010 transmission electron microscope (JEOL, Tokyo, Japan) equipped with a Mega-View III digital camera and Soft-Imaging System (SIS, Munster, Germany). Sections were stained with 1% toluidine blue for stereological analysis.
Stereological analyses of the sciatic nerves were done according to the principles described previously. 10,11 A manual stereological workstation composed of a digital camera (Nikon COOLPIX5400, Tokyo, Japan), a manual dial indicator-controlled specimen stage, and a light microscope (Nikon Microphot-FX, Tokyo, Japan) were used for axon number counting. 22,23 Two-dimensional dissector technique were performed to obtain an estimation of total axon number in an unbiased manner from nerve cross-section. In this application, the area of the unbiased counting frame was 1600 µm2. A counting frame was placed on to a monitor and the sampled area was chosen by a systematic uniform random manner via dial indicator-controlled specimen stage. Meander sampling of each sectioned nerve profiles was done in 70 × 70 µm2 step size in a systematic-random manner. This ensures that all locations within a nerve cross-section were equally represented and that all axon profiles were sampled with an equal probability regardless of shape, size, orientation, and location. 24,25 Another stereological workstation that was composed of a CCD digital camera, image capture card (Flash Point 3D, Integral Technologies, Indianapolis, Indiana, USA), personal computer, and computer-controlled motorized specimen stage (Prior Scientific, Cambridge, UK), a microcator (Heidenhein Traunreut, Germany) and a light microscope (Leica, Wetzlar, Germany) were used for stereological analyses of myelin thickness, axon cross-sectional area, as well as nerve fiber and axon diameters. A software program (CAST-GRID®-Computer Assisted Stereological Toolbox-Olympus, Copenhagen, Denmark) was used to control, measure, record stereological data, and capture digital images of the sections. This system reproduced microscopic images (obtained through a ×60, NA 1.40) on the computer monitor at a final magnification of 3209 that allowed accurate recognition and quantification of myelinated nerve fibers.
A two-dimensional isotropic uniform random nucleator was used for the estimation of axon area, nerve fiber and axon diameters, and thickness of myelin sheet using an oil objective (×60, NA 1.40) at a final magnification of ×3209. Meander sampling of each sectioned nerve profiles was done over successive, systemic-random steps of 70 × 70 µm2 step sizes.
Biochemical analyses
The excised tissue samples were weighed and then immediately stored at −30°C. The right sciatic and muscle tissues were perfused with 1.15% ice-cold KCl, minced, and homogenized in five volumes (w/v) of the same solution. Assays were performed on the supernatant of the homogenate that was prepared at 14,000 r/min for 30 min at +4°C. The protein concentration of the tissue was measured by the method of Lowry et al. 26 Activities of CAT were determined by measuring a decrease in hydrogen peroxide concentration at 230 nm by the method of Beutler. 27 Activity of SOD was measured according to the method described by Fridovich. 28 Level of lipid peroxidation in the tissue samples was expressed as MDA. It was measured according to the procedure of Ohkawa et al. 29 The activity of GSH-Px assay was based on the method of Paglia and Valentine. 30 Levels of NO were determined with Griess reaction. 31 Levels of serum PON1 were measured spectrophotometrically by the modified Eckerson et al. method. 32
Statistical analyses
The data was analyzed using Statistical Package for the Social Sciences version 11.5 (SPSS 11.5 for Windows, Chicago, Illinois, USA). The variables between the groups were tested by Mann–Whitney U test. Correlations between variables were performed with the Spearman correlation test. A value of p < 0.05 indicates a significant difference.
Results
Stereological analyses and histological findings
The quantitative results of the sciatic nerves including mean axon area, axon numbers, nerve fiber diameters, axon diameters, and thickness of myelin sheets in the HCQ and the control groups are shown in Table 1. The difference in the total number of myelinated axons and axon area between the control group and the HCQ groups were not statistically significant (p > 0.05, Table 1). The nerve fiber diameter and the thickness of the myelin sheet were significantly lower in the HCQ group than in the control group (p = 0.026, Table 1). The axon diameter was significantly decreased in the HCQ group than in the control group (p = 0.03, Table 1). The electron microscopic images of the myelinated fibers are shown in the figures. In order to show myelin degeneration in selected sections, electron microscopic images were obtained by electron microscopy (Figures 1 and 2).

Electron microscopic appearance in hydroxychloroquine-administered group. Myelin degenerations are seen as swelling in myelin shealting and shrinkage in axons (arrow).

Electron microscopic appearance in the control group. Regular form and thickness of myelin shealting (arrow head).
Comparison of the results of stereological analyses of nerve fiber diameter, axon diameter, myelin thickness, axon number, and axon cross-section area measurements between control and HCQ groups in the sciatic nerve (mean ± SD)
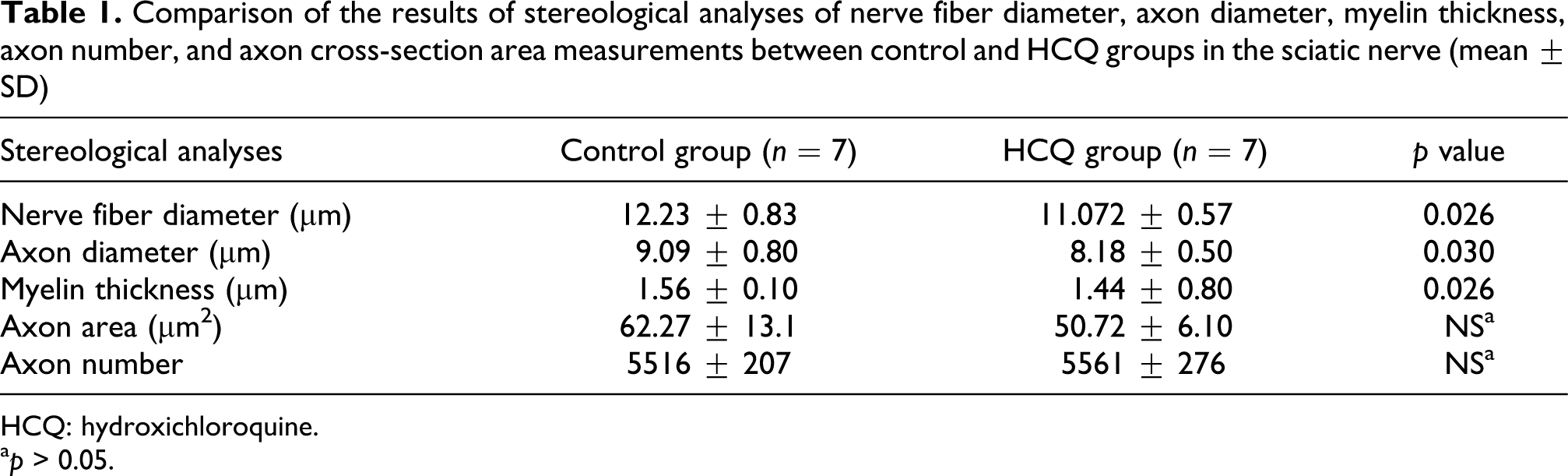
HCQ: hydroxichloroquine.
a p > 0.05.
Biochemical results
Biochemical results are shown in the Tables 2 and 3. The MDA levels in sciatic (24.0 ± 4.2 and 16.0 ± 2.8 nmol/g protein, p = 0.001) and muscle tissues (46.2 ± 8.1 and 27.2 ± 4.8 nmol/g protein, p = 0.002) were significantly higher in the HCQ group than in the control group. Similarly, the NO levels in the sciatic (0.14 ± 0.02 and 10.0 ± 0.02 µmol/g protein, p = 0.004) and the muscle tissues (0.24 ± 0.04 and 0.16 ± 0.03 nmol/g protein, p = 0.001) were significantly higher in the HCQ group than in the control group. The activities of SOD and GSH-Px in the sciatic nerves were significantly higher in the HCQ group than in the control group (activities of SOD 77.7 ± 10.3 U/g protein vs. 61.3 ± 8.2 U/g protein, p = 0.006; activities of GSH-Px 8.95 ± 1.05 U/g protein vs. 7.33 ± 0.87 U/g protein, p = 0.009). The activities of SOD and GSH-Px in the muscle tissue were significantly higher in the HCQ group than in the control group (activities of SOD 0.56 ± 0.01 U/g protein vs. 0.42 ± 0.07 U/g protein, p = 0.024; activities of GSH-Px 13.3 ± 1.7 U/g protein vs. 10.5 ± 1.3 U/g protein, p = 0.005). The activity of CAT in the sciatic nerves was not statistically different between the HCQ group and the control group (0.29 ± 0.06 U/g protein vs. 0.33 ± 0.06 U/g protein, p > 0.05). The activities of CAT and PON1 in the muscle tissue were significantly lower in the HCQ group than in the control group (activities of CAT: 0.41 ± 0.04 U/g protein vs. 0.51 ± 0.06 U/g protein, p = 0.002; activities of PON1: 0.14 ± 0.012 U/g protein vs. 0.18 ± 0.016 U/g protein, p = 0.001). The activities of PON1 in the sciatic nerve were significantly decreased in the HCQ group than in the control group (0.13 ± 0.01 U/g protein vs. 0.15 ± 0.01 U/g protein, p = 0.049). There was a positive correlation between the level of MDA and the activities of SOD in the sciatic nerve (p = 0.005, r = 0.71) and muscle tissue (p = 0.001, r = 0.78) in the HCQ group. There were negative correlations between the levels of MDA and the activities of CAT and PONI (p = 0.001, r = −0.79; p = 0.015, r = −0.63, respectively). There were negative correlations between the level of NO and the activities of PON1 in the sciatic nerve (p = 0.012, r = −0.65) and muscle tissues (p = 0.001, r = −0.78).
Oxidant/antioxidant parameters in sciatic nerve of rats in control and HCQ groups (mean ± SD)

HCQ: hydroxichloroquine; SOD: superoxide dismutase; CAT: catalase; GSH-Px: glutathione peroxidase; NO: nitric oxide; MDA: malondialdehyde; PON1: paraoxanase.
a p > 0.05.
Oxidant/antioxidant parameters between control and HCQ groups in muscle tissues of rats (mean ± SD)
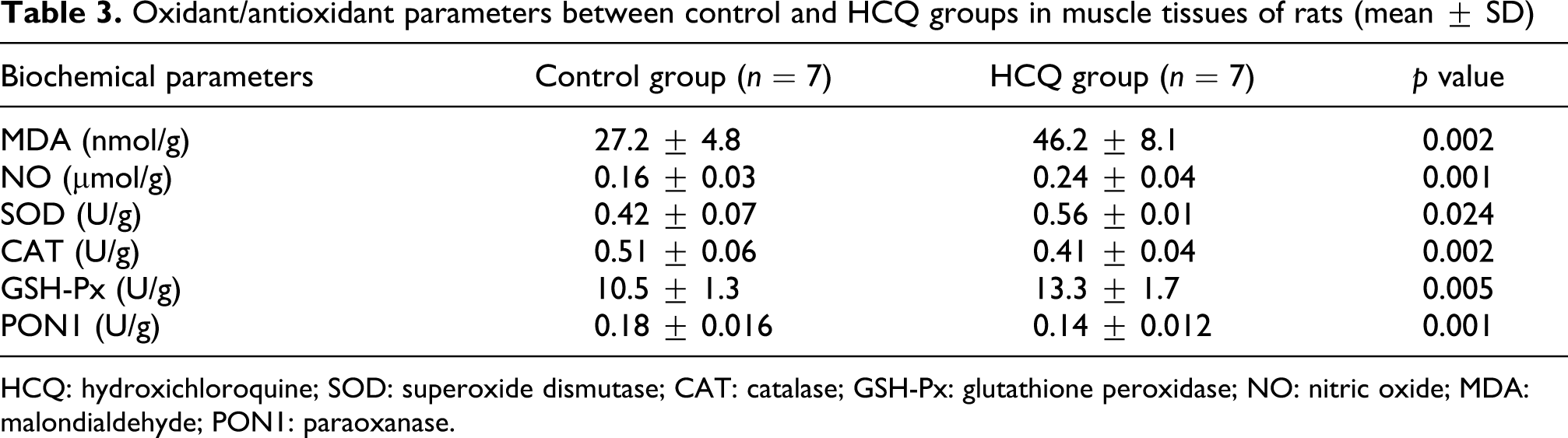
HCQ: hydroxichloroquine; SOD: superoxide dismutase; CAT: catalase; GSH-Px: glutathione peroxidase; NO: nitric oxide; MDA: malondialdehyde; PON1: paraoxanase.
Discussion
Based on our results, we postulate that HCQ may cause oxidative tissue damage by increasing lipid peroxidation in sciatic nerve and muscle tissue and may alter the activities of antioxidant enzymes. The increased levels of oxidative metabolites such as NO and MDA, the changed activities of the antioxidative enzymes, and the alteration of histopathological features of the sciatic nerves support our hypothesis.
HCQ has been synthesized as a modification of CQ phosphate. It has been shown that CQ is more effective and toxic than HCQ on a milligram basis. 3 There are side effects to both drugs including ocular, dermatological, hematological, and neuromuscular abnormalities. 1,2,5,8 Both CQ and HCQ can lead to damage of nerve fibers and heart and skeletal muscles. Respiratory failure related to HCQ-induced myopathy was reported in a patient with rheumatoid arthritis. 6 Stein et al. 5 emphasized that if a patient develops proximal myopathy, cardiomyopathy, or neuropathy after starting HCQ, the patient should be evaluated for possible HCQ neuromyotoxicity. The pathogenesis of HCQ-induced neuromyotoxicity is not exactly clear. We postulated that oxidative stress might play an important role in the pathogenesis of HCQ-induced neuromyotoxicity. Therefore, we investigated the oxidant/antioxidant parameters of sciatic and muscle tissues in HCQ-administered rats. An imbalance between oxidants and antioxidants in favor of the oxidants, which may cause tissue damage, is termed oxidative stress. 15,16 Oxidative stress is an underlying pathogenesis of several toxicities induced by drugs, such as methotrexate and CQ.14–18 Free radicals are involved in the peroxidative damage to cells; the main free radicals occurring in various conditions are superoxide, hydroxyl, peroxyl radicals, and NO. ROS may play a role in oxidative damage of cell structures such as DNA, proteins, and lipids. The tissue damage resulting from these radicals may be quantitatively determined by measurement of the level of MDA, which is an oxidative metabolite and an indicator of lipid peroxidation. In addition, antioxidant enzymes can play an important role in the protective mechanisms against oxidative stress. 16,33 Both MDA and NO are oxidant parameters, 34,35 and in our study, HCQ led to an increase in MDA levels, which was a reliable marker of lipid peroxidation in the sciatic nerve and muscle tissue of the rats. This finding showed that orally administered HCQ at a dose of 160 mg/kg may lead to oxidative stress in sciatic nerve and muscle tissues of rats.
NO is a short-lived radical that is generated during the conversion of
To defend against ROS, cells contain complex cellular defense mechanisms to reduce the risk of oxidative injury. Primary antioxidant enzymes include SOD, GSH-Px, and CAT, which convert free radicals or reactive oxygen intermediates to nonradical products 34 –36 and are responsible for removing superoxide radicals, hydrogen peroxide, and hydroperoxides. In sciatic nerve and muscle tissues, HCQ treatment causes a dramatic increase in the activities of SOD and GSH-Px. An SOD, which converts superoxide anion to hydrogen peroxide, is an important part of primary antioxidant defense, protecting the cell against toxic effect of superoxide radicals. The increased enzyme activity of SOD and GSH-Px in the HCQ treatment group may have been an adaptive response to the elevated oxidative stress. Similar results for the positive correlation between activity of SOD and level of MDA was demonstrated in a previous study. 37 These findings demonstrate increased ROS in the sciatic and muscle tissues. The increased activity of SOD may be an indicative of increased production of superoxide anion in the nerve and muscle tissues of HCQ-administered rats. GSH-Px, an antioxidant enzyme, can eliminate hydrogen peroxide and lipid hydroperoxides using reduced glutathione as a reactive hydrogen radical donor. Increased activity of GSH-Px enzyme may reflect increased production of hydrogen peroxide, which could be considered a protective response of living bodies against increased oxidative stress caused by ROS. 34 –36 In the muscle tissues of rats, HCQ treatment led to a significant decrease in the activity of CAT. This may be related to a decrease in CAT production and/or increased catabolism due to HCQ toxicity. However, the HCQ treatment did not lead to dramatic decreases in the activity of CAT in the sciatic nerve. In our study, there was a negative correlation between level of MDA and activity of CAT in muscle tissues. 38 Reduced antioxidant activities of CAT enzyme may lead to an increase in lipid peroxidation products. In the muscle and sciatic nerve tissues of rats, the HCQ treatment caused a significant decrease in the activity of PON1 in our study, in which there was a negative correlation between level of MDA and activity of PON1. In addition, there was a negative correlation between levels of NO and activities of CAT in both muscle and nerve tissues. 39 A PON1 is a serum enzyme that prevents oxidation of low-density lipoprotein by hydrolyzing lipid peroxides; it has an effect on hydrogen peroxide as well. PON1 has been thought to possess peroxidase-like activities, as it reduces hydroperoxides and hydroxyl radicals. As a result, activity of PON1 is reduced in high oxidative stress conditions. 40 Therefore, we suggest that the decreased activities of PON1 of the sciatic nerves and the muscle tissues of the rats might be related to HCQ-induced oxidative stress.
Estes et al. 41 reported morphologic changes in human peripheral nerves due to CQ toxicity. However, HCQ-related morphological alteration of the sciatic nerve of rats has not been studied previously. A morphometric analysis of peripheral nerves is an important measure, providing more objective and reproducible data than the conventional qualitative/visual examination methods. Stereological methods are commonly used in research regarding nerve injuries, repair, and regeneration. 10,11 Quantitative features of nerve fibers, including axon number, axonal area, and myelin thickness, are usually parameters for determining nerve injury types. Using the systematic random sampling approach, stereological analysis techniques can provide accurate and reliable estimates of morphological data. 22 –25 As such, in this study; we performed a stereological morphometric analysis of the sciatic nerves obtained from the HCQ-administered rats. Mean nerve fiber diameter, axon diameter, and myelin sheet thickness in the sciatic nerve were lower in HCQ-administered rats than in the control rats. However, no significant differences were found between the groups in the mean axon area or the axon numbers. These findings showed negative effects of long-term HCQ administration on sciatic nerve myelin sheet and axonal size.
In conclusion, our data indicates that oxidative stress may play an important role in HCQ-induced neuromyotoxicity; therefore, modulation of oxidative stress by antioxidants may be useful in its prevention and/or reduction. Further studies on this topic are warranted for better insight into HCQ-induced neuromyotoxicity.
