Abstract
Levosimendan (Levo) is an inodilator improving cardiac output (CO) and reducing afterload in heart failure. Previously, we reported that Levo improved CO but not blood pressure (BP) in a rodent model of verapamil poisoning. We theorised that Levo-induced vasodilation should not influence BP to a similar degree in metoprolol poisoning. Aim: To assess the effect of Levo on haemodynamics in a rodent model of metoprolol poisoning. Method: Anaesthetized male Wistar rats were infused metoprolol continuously. When the BP dropped to 50% of baseline (time 0) rats received 1 of the 4 treatments: (a) control (0.9% saline bolus + infusion); (b) Levo-
Introduction
The two most commonly encountered classes of drug poisoning resulting in clinically significant myocardial toxicity and heart failure are the beta-receptor antagonists (BBs) and the calcium channel blocking agents. Levosimendan (Levo) is a novel cardiac inotropic agent that exerts its effects by sensitizing troponin-C to calcium, thus increasing myocardial contractility without increasing intracellular calcium and adenosine triphosphate concentrations or myocardial oxygen demand.1,2 It also possesses mild inhibitory actions on phosphodiesterase-III and relaxes peripheral vascular smooth muscle by agonism of vascular smooth muscle voltage-gated potassium channels. 3 As a result, Levo increases cardiac output (CO) and reduces afterload. These effects may be beneficial in nontoxicologic forms of heart failure where reduction of afterload may reduce the degree of cardiac work in a pathologically compromised myocardium. We have previously reported on the effects of this agent in a rodent model of verapamil poisoning. 4 Levo infusion produced a moderate but significant improvement in CO in verapamil-poisoned rats but did not result in a concomitant rise in systemic blood pressure (BP), suggesting that its vasodilatory properties may antagonize any benefits seen in the improvement in CO. 4 –6 Similarly, Kurola et al. reported an improvement in CO and survival, but not BP, in a swine model of verapamil poisoning following administration of a single bolus dose of Levo. 7
To date, there is only one study reporting the effects of Levo in experimental BB poisoning. Leppikangas et al. observed that a single bolus injection of Levo at a dose of 1.2 mg, approximately 50 μg/kg, improved cardiac function and BP in a swine model of propranolol toxicity. 8 The degree of propranolol toxicity induced in this study was moderate and there may have been some mismatch in the severity of poisoning between the no treatment control group and the Levo group prior to the initiation of treatment. 9 Additionally, Levo was only administered as a single bolus dose rather than as an infusion as is commonly the case, and the positive control treatment arm used dobutamine, a relatively weak catecholaminergic inotrope. As a result of this preliminary work, we undertook a study to examine the effects of Levo in BB poisoning, utilising our previously reported rat model of cardiovascular drug poisoning. 4,6 We chose metoprolol for this series of experiments and aimed to assess whether there was any difference in CO or BP in animals receiving Levo infusion with and without a loading dose.
Method
This was a randomized open-label study utilising male adult albino Wistar rats. Blinding was not performed as a single investigator performed the experiments once the surgery was completed. The method was similar to that described previously. 4
Animals
Consent was obtained from the Monash University Animal Ethics Committee. Male adult albino inbred Wistar rats (13–14 weeks old and 350–450 g), from the Monash University Animal House breeding colony, were used to avoid problems associated with the oestrus cycle of the female rat.
Preexperiment procedures
Animals were anaesthetized initially with isoflurane (Aerrane, Baxter Health Care, Toongabbie, New South Wales, Australia) in an enclosed Perspex anaesthesia box. Once unconscious, 80 mg/kg intraperitoneal pentobarbital, a long-acting barbiturate agent, was administered (Ilium Pentobarbitone, 60 mg/ml, Troy Laboratories, Smithfield, New South Wales, Australia). Animals were placed on a heating pad and under a heating lamp to maintain a body temperature between 36 and 38°C. The trachea was cannulated and the animals were ventilated (10 ml/kg and 55 breaths/min) using an Ugo Basile model 7025 Ventilator (Comerio, Varese, Italy). The left carotid artery was cannulated and a MLT1402 T-type Ultra Fast Thermocouple probe (AD Instruments, Castle Hill, Australia) was passed through the cannula for aortic blood temperature measurement for CO estimation by the thermodilution technique. The left and right jugular veins were cannulated with double-lumen polyethylene catheters (Intramedic, North Rocks, Australia) for infusion of drugs and fluids. The left femoral artery was cannulated with single-lumen polyethylene tubing (BD Diagnostics, Sparks, Maryland, USA) for arterial BP monitoring via an ADInstruments MLT844 physiological pressure transducer and ML110 bridge amp (ADIstrusments, Castle Hill, Australia). A rectal temperature probe was inserted for monitoring the tissue temperature. Cutaneous electrocardiogram (ECG) electrodes were placed for single lead recording of heart rate (HR) and rhythm. Continuous data collection for ECG, arterial BP, rectal and core temperature was performed using a PowerLab 4/30 data acquisition system and Chart Version 5.0 software (ADInstruments, Castle Hill, Australia). CO was estimated sequentially with the PowerLab cardiac output system and Cardiac Output Module Software (ADInstrument, Castle Hill, Australia). Briefly, 100 μl of cold Hartmanns solution was injected via the left internal jugular vein and a thermodilution curve was recorded from the carotid thermistor probe on PowerLab Chart Software. CO was calculated using the area under the curve by Cardiac Output Module found in PowerLab Chart Software. CO is reported in ml/min/100 g. Average CO for healthy rats is reported as 20–25 ml/min/100 g. 10
Once the surgery was completed, animals were left undisturbed for 30 min for equilibration. Prior to the commencement of experiment, the baseline HR, mean arterial BP (MAP), systolic BP (SBP) and CO were recorded. Haemodynamic parameters were then recorded at peak toxicity (time 0) and every 10 min during treatment for 1 h or until death. Two or sometimes three syringe pumps were used for drug infusion during the experiment (Graseby 3100 (Graseby Ltd, Hertfordshire, UK); Terumo model STO 523 (Terumo Corp, Tokyo, Japan)). Animals were euthanized at the end of the experiment.
Experimental protocol
There were 10 animals per study group. Animals were administered metoprolol (Sigma Chemicals, MO, USA) made up to a concentration of 10 mg/ml in 0.9% saline by continuous infusion of 24 mg/kg/h until MAP fell to 50% of baseline (time 0). The metoprolol infusion was then reduced to 12 mg/kg/h to maintain toxicity. Once toxicity was established, HR, MAP, SBP and CO were recorded and animals were randomly assigned to one of four treatment groups (10 rats in each group).
The treatment groups were:
Control. Rats received normal saline (0.9% saline, Baxter Health Care, Toongabbie, New South Wales, Australia) loading dose of 10 ml/kg infused over 10 min followed by 0.9% saline infusion of 10 ml/kg/h for 60 min.
Levo-L (Levo + loading dose). Rats received Levo (Simdax, Orion Corporation, Finland) 36 μg/kg in 10 ml/kg of 0.9% saline as loading dose, infused over 10 min followed by 0.6 μg/kg/min as an infusion dose in 10 ml/kg/h 0.9% saline for 60 min.
Levo-I (Levo infusion only without loading dose). Rats received Levo 0.6 μg/kg/min as an infusion with 10 ml/kg of 0.9% saline as loading dose, infused over 10 min, which was then continued at 10 ml/kg/h for 60 min.
Epi (Epinephrine infusion). Rats received 0.5 μg/kg/min of epinephrine (1 mg/ml, AstraZeneca Pty Ltd, Australia) as a continuous infusion with 10 ml/kg loading dose of 0.9% saline followed by 10 ml/kg/h over 60 min.
Treatment lasted a total of 70 min. Ten minutes for loading dose of 0.9% saline followed by 60 min for maintenance infusion. HR, CO, SBP and MAP were measured every 10 min during the course of the treatment. All animals received a 10 ml/kg loading dose of fluid and 10 ml/kg/h as an infusion with their respective treatments.
Statistical method
Data were plotted graphically as mean ± SEM for the variables every 10 min until the end of the 70-min treatment protocol or death. Results are expressed as mean ± SEM and were considered statistically significant at p < 0.05 (2 tailed). Continuous haemodynamic measures were examined using one-way analysis of variance (ANOVA) and Neuman-Keuls posttest comparing each treatment group for normally distributed data or Kruskal-Wallis posttest for nonparametric data. Differences in survival are reported using Kaplan-Meier survival curve analysis. The survival curve of each treatment group was compared to control using the logrank test. Sample size was based upon the results seen in our previous studies with verapamil poisoning and Levo, where 10 animals per groups were sufficient to detect a 25% increase in CO from peak toxicity between control and treatment groups. 4,6 Statistical analyses were carried out using GraphPad Prism 4.0 and InStat 3.01 statistical software (GraphPad Software, Inc) as well as MedCalc statistical software for survival analysis (Version 11.6.1.0), Mariakirke, Belgium.
Results
Prior to the administration of metoprolol, baseline MAP, CO and HR were found to be similar in all groups (Table 1), and there was also no significant difference in peak toxicity haemodynamic parameters (time 0) in all groups (Table 2). Animal weights were also comparable between groups.
Summary of baseline haemodynamic parameters (n = 10 per group)

Epi: epinephrine; Levo-I: levosimendan infusion only without loading dose; Levo-L: levosimendan + loading dose.
Summary of peak toxicity (t = 0) haemodynamic parameters (n = 10 per group)

Epi: epinephrine; Levo-I: levosimendan infusion only without loading dose; Levo-L: levosimendan + loading dose.
Survival
The Levo-L treatment group showed a statistically significant greater survival proportion at 70 min compared to the control (50% vs. 20%, p = 0.04, χ 2 = 4.3). There were no other differences in survival between the treatment groups (Figure 1).
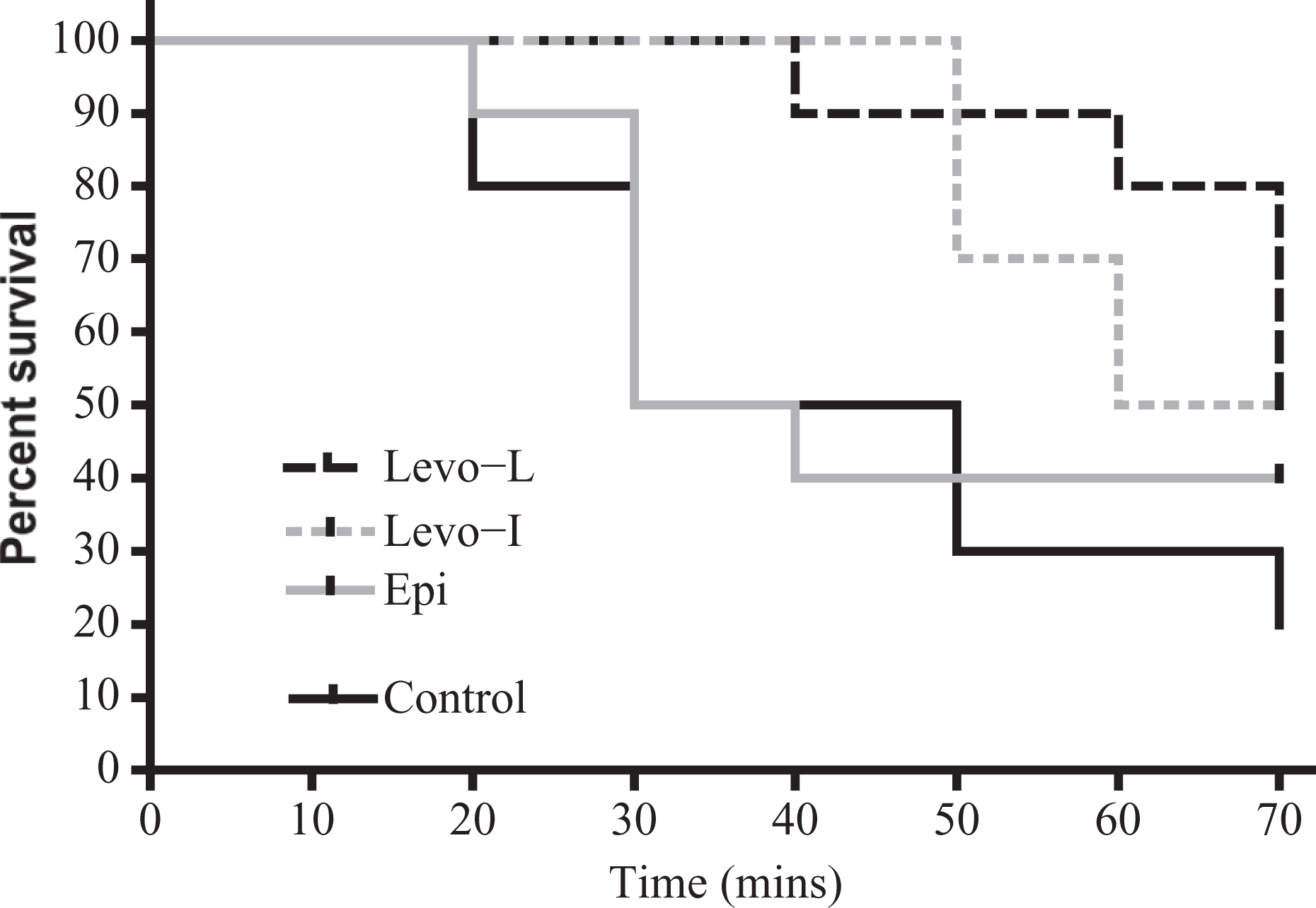
Kaplan-Meier survival curve of animals over the time course of the experiment (n = 10 per group). There was a significant difference in survival between Levo-L and control over the time course of the experiment (50% vs. 20% at 70 min, p = 0.04, χ 2 = 4.3). There were no other differences in survival between treatment groups and control. Levo-L: levosimendan with loading dose; Levo-I: levosimendan infusion without loading dose; Epi: epinephrine.
Cardiac output
Levo produced a statistically significant increase in CO in the Levo-L group (p = 0.022, 95% CI: 1.473–17.465) and Levo-I arms (p = 0.042, 95% CI: 4.243–20.032) at t = 10 min only. Mean CO remained higher than that of control for both Levo treatment groups at all other time points during treatment but this did not reach significance. Epinephrine did not produce a significant increase in CO at any time point compared to control (Figure 2).
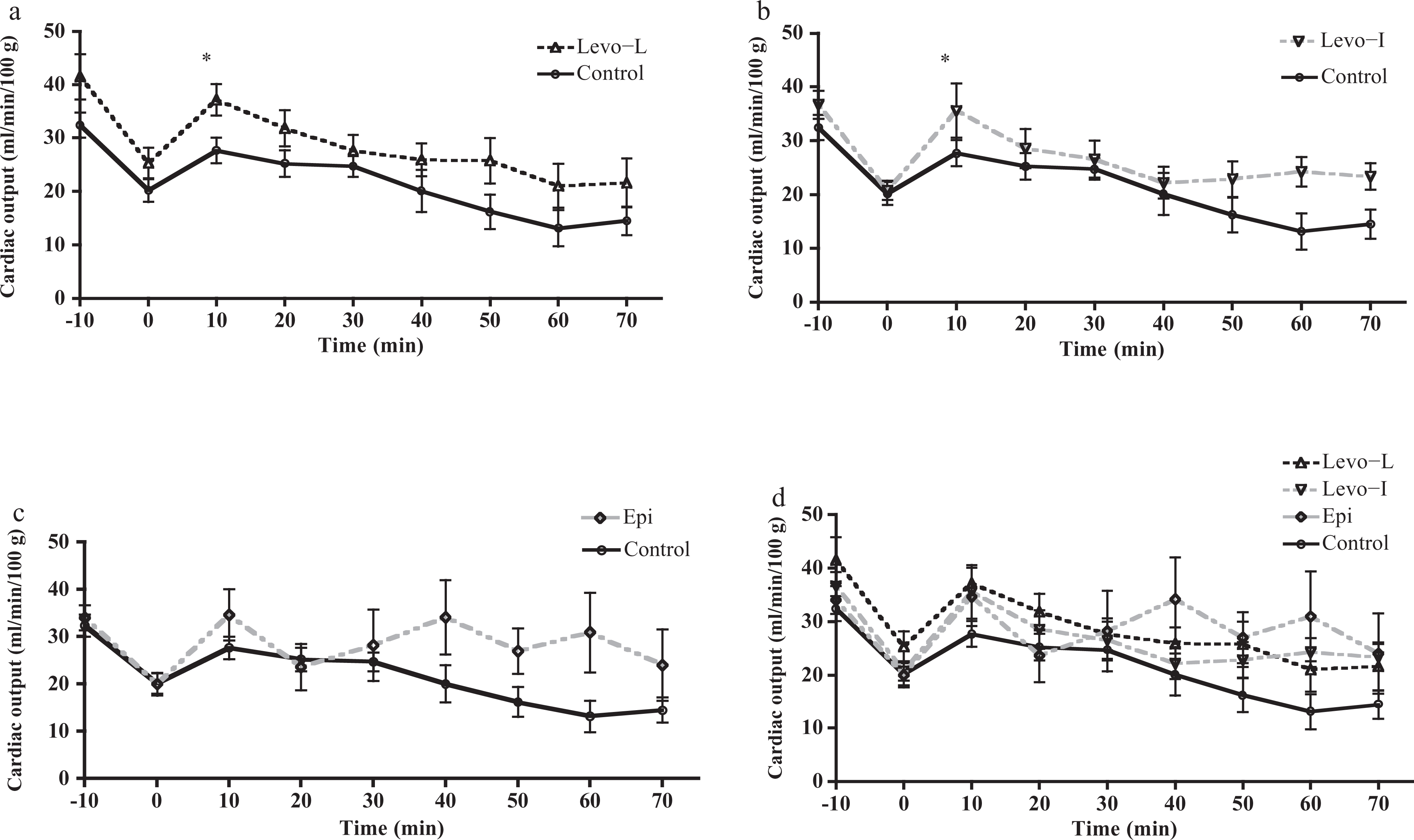
Cardiac output (CO) versus time for the three treatment groups compared to control (n = 10 per group). Panel (a) shows Levo-L: levosimendan with a loading dose. CO significantly greater at t = 10 time point (p = 0.022, 95% CI: 1.473–17.465). Panel (b) shows Levo-I: Levosimendan without loading dose. CO significantly greater at t = 10 time point (p = 0.042, 95% CI: 4.243–20.032). Panel (c) shows Epi: epinephrine versus control. No significant differences in CO at any time point. Panel (d) shows all treatment groups.
Mean arterial pressure
BP did not increase in either Levo treatment arm. However, there was a trend for BP to be lower in the Levo-L group compared to the control group but this was not statistically significant. Epinephrine infusion was associated with a statistically significant increase in MAP at all time points compared to control and Levo-L groups. There was also a significant increase in MAP between epinephrine and Levo-I group from t = 30 min to t = 70 min (Figure 3). The observed response of SBP in all treatments was similar to that seen with MAP (data not shown).
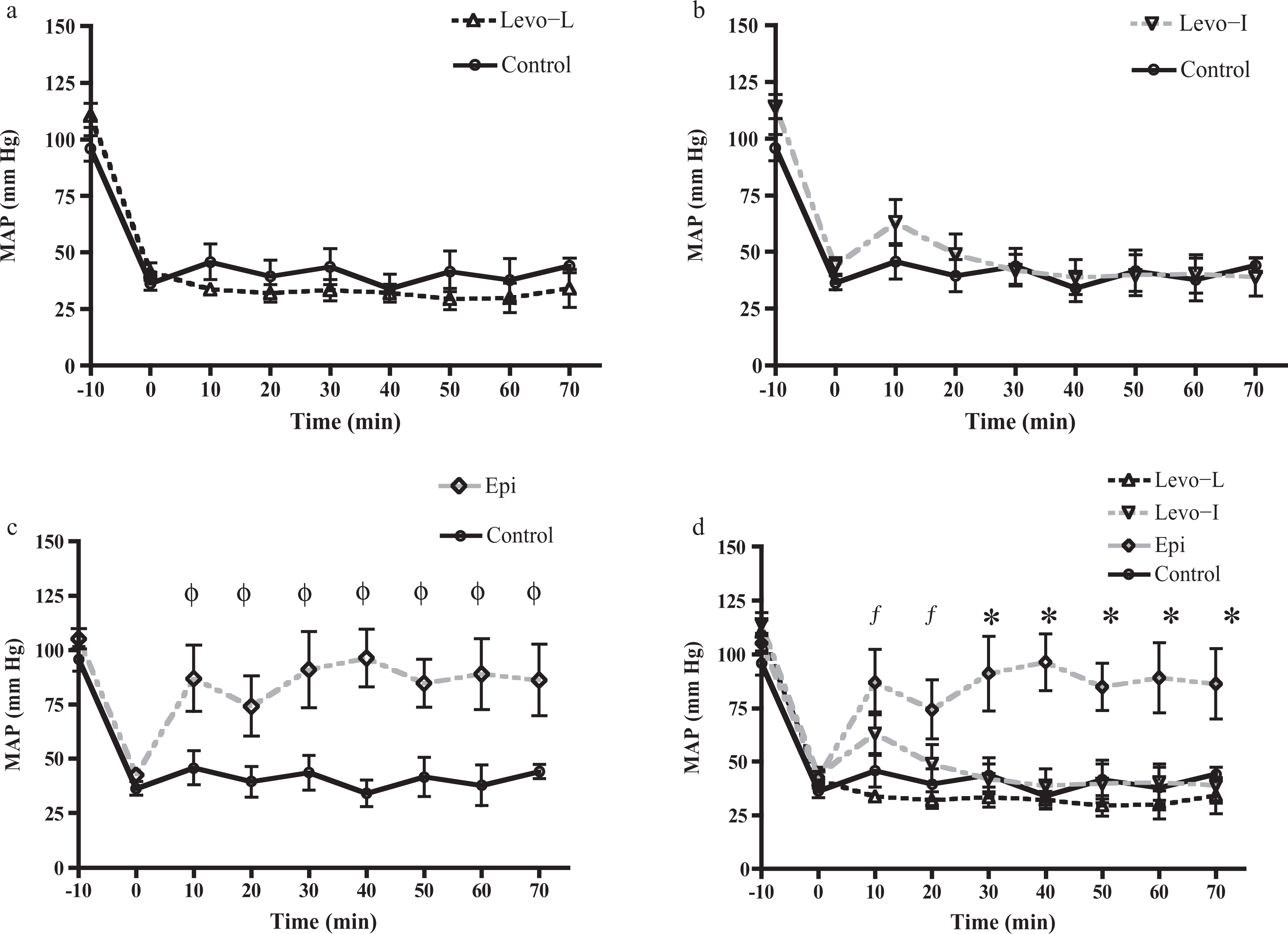
Mean arterial blood pressure (MAP) versus time for the three treatment groups and control (n = 10 per group). Panel (a) shows Levo-L: levosimendan with a loading dose. Panel (b) shows Levo-I: levosimendan without loading dose. MAP was no different from control at any time point for either levosimendan treatment group. Panel (c) shows epinephrine versus control. MAP was significantly greater than control at every time point. Panel (d) is a comparison of all treatment groups in one figure. Epi produced a significant increase in blood pressure compared to control and Levo-L groups at t = 10 and 20 min (f). (p < 0.01 at t = 10 and p < 0.05 at t = 20 min). *Epi produced a significant increase in MAP compared to all other treatment groups from t = 30 onward (t = 30: p < 0.01, t = 40: p < 0.05, t = 50: p < 0.01, t + 60: p < 0.05, t = 70: p < 0.05). Analysis of variance (ANOVA) with Neuman-Keuls post-test.
Heart rate
There was no difference in mean HR at any individual time point for any of the treatment groups compared to control (Figure 4). Dysrrhythmias noted during the metoprolol infusion were sinus and junctional bradycardia both seen as preterminal events in animals that did not survive to the end of the 70-min protocol.
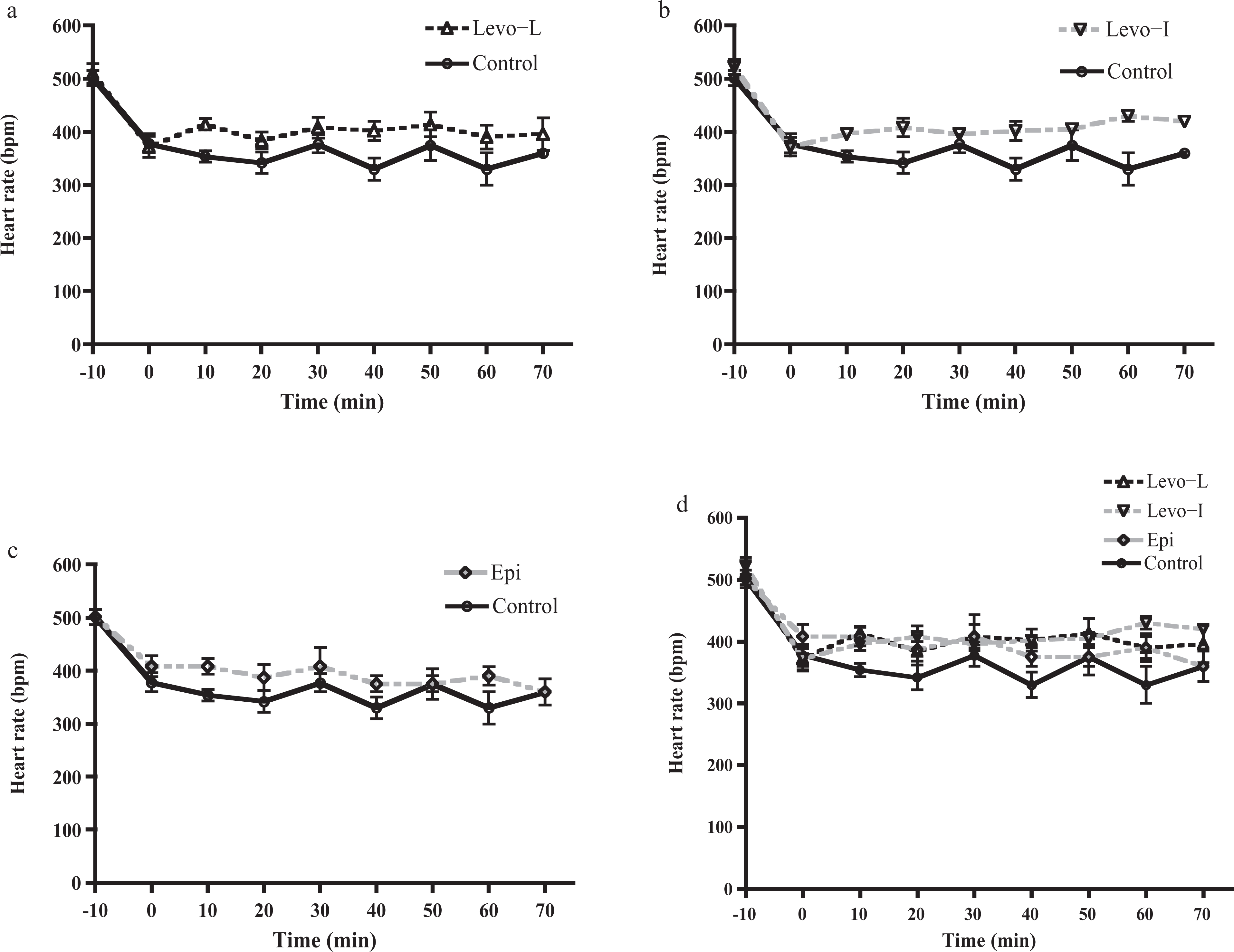
Heart rate (HR) versus time for the three treatment groups compared to control (n = 10 per group). Panel (a) shows Levo-L: levosimendan with a loading dose. Panel (b) shows Levo-I: levosimendan without loading dose. Panel (c) shows Epi: epinephrine versus control. Panel (d) compares all treatment groups in one figure. There were no differences in HR from control at any time point for any treatment group.
Discussion
BB poisoning has the potential to cause significant haemodynamic compromise resulting from depression of myocardial contractility and associated bradycardia. In severe cases of poisoning, treatment with beta-receptor agonists such as epinephrine and isoproterenol, may be relatively ineffective due to the degree of beta-receptor antagonism that may occur in the presence of high blood concentrations of receptor antagonists. Atypical inotropic agents such as high-dose insulin euglycemia therapy and high-dose glucagon therapy may improve myocardial function without the need to directly activate beta-adrenergic receptors. 11 –16 Levo is a positive inotropic agent with its mechanism of action being the sensitization of troponin-C to calcium, resulting in improved contractility without any increase in myocardial oxygen demand. Levo is also a vascular smooth muscle voltage-gated potassium channel agonist, reducing afterload and peripheral vascular resistance. 17 These two characteristics place this agent in the class of drugs known as ‘inodilators.’ Levo is currently used for the treatment of decompensated congestive heart failure, acute myocardial infarction and postcardiac bypass surgery. 3,18 Several studies have demonstrated the benefits of Levo in patients after cardiopulmonary bypass to enhance cardiac performance postsurgery. 19 There are very few clinical reports on Levo use in cardiovascular drug poisoning. Osthoff et al. report a case of verapamil poisoning unresponsive to standard measures where a gradual recovery was observed after the commencement of a Levo infusion. 20 Similarly, Varpula et al. describe two cases of severe verapamil poisoning on multiple inotropic therapies where a slow improvement was observed once a Levo infusion had been commenced. 21 In both reports, the response to Levo was clouded by the fact that other drugs had been used for a prolonged period and the haemodynamic response was gradual rather than immediate after the institution of Levo.
Animal studies describing the use of Levo as a single or adjuvant agent in severe verapamil and propranolol poisoning report varied haemodynamic results with this agent. Previously, we have reported that Levo produced a moderate improvement in CO in a rodent model of severe verapamil poisoning without any improvement in BP. 4 –6 It was theorised that the vasodilatory effect of Levo may enhance the peripheral vasodilation produced by verapamil, thus negating any increase in CO. Similarly, Kurola et al. reported moderate increase in CO and no improvement in BP in a porcine model of verapamil poisoning. 7 Finally, Abraham et al. reported that Levo did not improve survival in verapamil-poisoned rodents. 22 In the only study to date examining the effect of Levo in beta-blocker poisoning, Leppikangas et al. reported that a single bolus injection at a dose of around 50 μg/kg improved the survival, CO and BP in propranolol-toxic swine and was superior to dobutamine infusion in all measures. 8 Although swine were used in this model, the degree of cardiac toxicity induced in animals was not dissimilar to our study, with a target CO and BP at peak toxicity around 40–50% of that seen prior to the induction of poisoning. This study was critiqued by Lugassy et al. who observed that there were inequities in the baseline BP of the treatment groups with only the control group achieving significant beta-blockade as evidenced by a fall in BP and HR. 9 Also, the choice of dobutamine as the positive control treatment was unusual as this is not the catecholamine agent of choice for most physicians treating severe BB or calcium channel antagonist poisoning. Levo has also been shown to be superior to dobutamine in the clinical setting of chronic low-output heart failure. 23 Finally, Leppikangas et al. administered Levo as a single loading dose and did not proceed with an infusion.
In our model of metoprolol poisoning, we found that treatment with Levo infusion with and without a loading dose resulted in a moderate but nonsustained improvement in CO and no improvement in BP, at the dose studied. Interestingly, the improvements in CO were less than those observed in our previous studies examining the effect of Levo in verapamil-poisoned rodents, utilising comparable doses of Levo. 4 –6 We observed a similar lack of improvement in BP with the administration of Levo in metoprolol poisoning that was also present in our previous study of this inotrope in verapamil poisoning. Unlike Leppikangas, we elected to use epinephrine as our positive control catecholamine infusion. Epinephrine is a more potent direct beta-receptor agonist than dobutamine and also has significant alpha-adrenergic receptor agonist properties. Interestingly, in our study, epinephrine produced significantly higher BP than Levo in surviving animals without an appreciable increase in CO compared to saline-treated control animals, suggesting a primary vasopressor response rather than myocardial stimulation. This may indicate that the degree of metoprolol-induced beta-receptor antagonism was greater than that which could be overcome with the dose of epinephrine infused in this study. We did not use an escalating dose of epinephrine, and it is possible that increasing doses may have overcome the beta-receptor antagonism induced by metoprolol.
There are a number of limitations in our study. It was conducted in anaesthetized rats with intravenous administration of metoprolol, and this does not necessarily reflect the human clinical overdose setting. Notably, animals did not develop bradycardia during metoprolol infusion despite a drop in BP of 50% from the baseline at the time of initiation of treatment. Bradycardia is a significant early feature of human beta-blocker poisoning but only developed in rats as a preterminal finding. We observed a similar HR response to verapamil in our previous experiments with Levo. 4 –6 However, the degree of hypotension and fall in CO in this study suggests that the severity of poisoning was significant prior to treatment. Nevertheless, Levo has improved CO in both human and animal models of nontoxicological severe heart failure. 24 ,25 This was a nonblinded experimental study. Since the investigator knew the treatment groups being administered in every experiment, observer bias may have been introduced into data collection. However, in this study the objective haemodynamic data were collected, which gave little opportunity to make subjective interpretations of responses to the various therapies.
We used a similar infusion dose of Levo as in our previous studies of verapamil poisoning. The degree of improvement in CO at each individual time point with this infusion was less than that observed following verapamil poisoning in a similar model. As a result, the lack of statistical significance between the Levo treatment arms and control at individual time points may be a reflection of inadequate sample size or a poor response to this dose of drug in this particular model of poisoning. We also did not examine any surrogate markers of haemodynamic compromise such as development or resolution of metabolic acidaemia or changes in blood lactate concentration to Levo or epinephrine. These also may indicate the efficacy or lack thereof, for the therapies investigated.
The optimal dose of Levo in the treatment of drug-induced heart failure is uncertain. A higher dose may potentially result in greater increases in CO, but conversely, it may also produce more significant hypotension due to the drug’s vasodilatory effects. 8 In future, we aim to assess higher doses of Levo, with and without loading doses, with poisoning from other beta-receptor agonists, such as propranolol. It is also possible that the degree of vasodilatory effect of Levo may vary across species and that the rat does not parallel the human response with similar drug doses.
Finally, pentobarbital is a long-acting barbiturate animal anaesthetic, with minimal effects on haemodynamics in cardiovascular animal research. In high doses, it may result in myocardial depression and lower the BP in animals due to central inhibition of the vasomotor centre. The doses used in this study were similar to those cited in previous research examining cardiovascular physiology in rodents. 26
Conclusion
We observed only a mild improvement in CO with Levo infusion with and without a loading dose in a model of severe metoprolol poisoning when compared to saline control-treated animals. As observed previously by us in verapamil poisoning, there was no significant improvement in BP with Levo administration despite moderate improvement in CO. Epinephrine increased BP. This was most likely the result of its vasoconstrictive properties rather than a direct effect on myocardial contractility. Further study of dosing regimens of Levo are required before it can be considered for routine use in human drug-induced cardiac decompensation. Care should also be exercised if considering the use of this drug in human poisoning-related heart failure, given the potential for exacerbation of hypotension from increased peripheral vasodilation.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
