Abstract
Carotenoid meso-zeaxanthin ((3R, 3′S)-β, β-carotene-3,3′-diol [MZ]) was evaluated for its protective effect against cisplatin-induced nephrotoxicity in Swiss albino mice. Oral administration of MZ was started 5 days prior to cisplatin (16 mg/kg body weight, intraperitoneally as a single dose) injection. Animals in all groups were killed 72 h after cisplatin treatment. In serum, renal function markers like urea and creatinine, which were drastically elevated in cisplatin-treated control animals, were found to be decreased significantly by MZ pretreatment. Cisplatin-induced myelosuppression was also found to be significantly ameliorated by MZ as evident from the increase in white blood cell count, bone marrow cellularity and number of maturing monocytes in MZ-treated animals when compared with cisplatin alone-treated control animals. The levels of antioxidant enzymes—superoxide dismutase, catalase and glutathione peroxidase—as well as the glutathione level in the kidney were decreased after cisplatin treatment. But the levels were markedly increased by the carotenoid treatment in a dose-dependent manner. Addition of MZ was found to inhibit singlet oxygen produced by toluidine blue in vitro. Moreover, administration of MZ to the animals inhibited increased lipid peroxidation, conjugated dienes and hydroperoxides that are formed in the kidney by cisplatin administration. The results of histopathological analysis supported the protective potential of MZ against cisplatin-induced toxicity.
Introduction
Cisplatin (cis-diammine dichloro platinum (II); Figure 1) is one of the potent chemotherapeutic agents having clinical activity against a wide variety of solid tumours. 1 The cytotoxic mode of action of cisplatin is mediated by its interaction with DNA to form intrastrand cross link adducts, which activate several signal transduction pathways like Ataxia Telangiectasia and Rad3-related protein (ATR), p53, p73 and mitogen-activated protein kinase (MAPK) and culminate in the activation of apoptosis. 2 Despite this success, the therapeutic effectiveness of cisplatin is blunted by the development of severe side effects such as nephrotoxicity, myelosuppression, damage to gastrointestinal tract, central nervous system and so on. 3

Structure of cisplatin.
The molecular mechanisms explaining the nephrotoxicity of cisplatin are complex, but it appears that the induction of an oxidative stress within kidney tissue is the decisive factor. Some promising new strategies to reduce the production of reactive oxygen species (ROS) and protect the function of the kidney are now available for the treatment of patients in order to lessen the renal injury associated with cisplatin. However, treatment-related toxicity is still a problem for many patients and more efforts need to go into developing chemoprotective agents that lessen the burden of treatment and importantly are also cost effective.
Meso-zeaxanthin ((3R, 3′S)-β, β-carotene-3,3′-diol [MZ]) is a xanthophyll carotenoid (Figure 2) present in macula lutea of human eye along with zeaxanthin [(3R, 3′R)-β, β-carotene-3, 3′-diol] and lutein [(3R, 3′R, 6′R)-β, ∊-carotene-3, 3′-diol]. Owing to its extended conjugated diene (CD) structure, MZ is believed to be involved in singlet oxygen quenching reactions and found to mitigate age-related macular degeneration and cataract. 4 In addition, MZ contains hydroxyl groups that interact with polar head groups of phospholipid, locking the molecule in a position that spans the membrane. This property allows this molecule to react with free radicals generated in both aqueous medium and membranes, which broadens its antioxidant capacities. 5 In vitro and in vivo studies revealed the antioxidant potential of MZ. 6 It has also been reported that in association with a zeaxanthin-binding protein, the pi-isoform of glutathione-S-transferase, MZ provides better protection against lipid membrane oxidation than zeaxanthin. 7
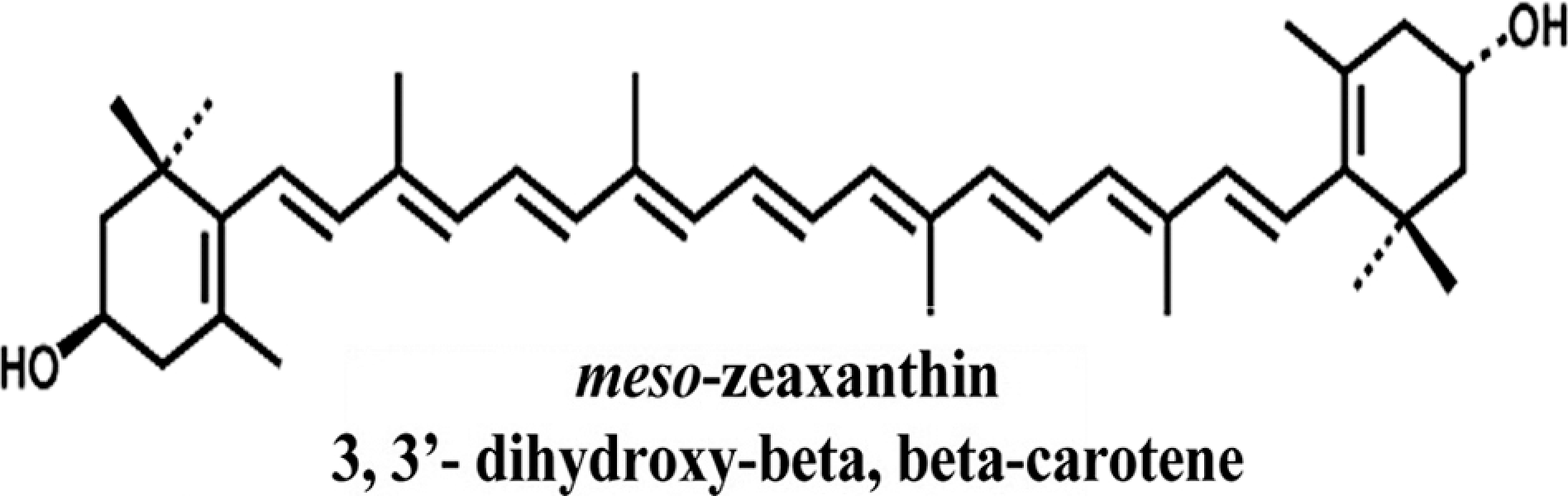
Structure of meso-zeaxanthin.
MZ is found to be present in some foods namely fish skin, shrimp carapace, turtle fat, and so on. Previous studies on this carotenoid showed that it has hepatoprotective, 8 antimutagenic and anticarcinogenic properties. 9 The present study evaluates the chemoprotective potential of carotenoid MZ.
Materials and methods
Chemicals and reagents
Nitro blue tetrazolium (NBT), glutathione (GSH) and 5,5′-dithiobis-(2-nitrobenzoic acid) (DTNB) were purchased from Sisco Research Laboratories Pvt Ltd (Mumbai, India). Cisplatin was obtained from Dabur Pharma (Himachal Pradesh, India). Toluidine blue (TB) was bought from Sigma Aldrich (St. Louis, MO, USA). Thiobarbituric acid was purchased from Himedia Laboratories (Mumbai, India). The kits for estimating urea and creatinine were supplied by Span Diagnostics Ltd (Surat, India). All other chemicals and reagents used were of analytical grade and obtained locally.
Drug
MZ was supplied by Omni Active Health Technologies Pvt. Ltd (Mumbai, India). A 5% suspension of MZ was prepared in sunflower oil.
Animals
Male Swiss albino mice (22.6 ± 1.5 g) were purchased from Small Animal Breeding Station (Mannuthy, Kerala, India) and were housed in well-ventilated cages under controlled conditions of light and humidity and provided with normal mouse chow (Sai Durga Food and Feeds, Bangalore, India) and water ad libitum. All the animal experiments were carried out as per the instructions prescribed by the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forest, Government of India, and implemented through the Institutional Animal Ethical Committee of the Research Centre.
Singlet oxygen quenching of MZ by DMF assay
The ability of MZ to provide photoprotection against singlet oxygen was measured using a previously published method. 10 Briefly, 1 ml of the photosensitiser 5 mM TB in ethanol was illuminated for 5 min in the presence of 0.2 mM 2,5-dimethyl furan (DMF). DMF is very reactive with singlet oxygen, and its rapid photo-oxidation is evidenced by disappearance of its 215 nm absorption peak recorded on a ultraviolet (UV)–visible spectrophotometer. When different volumes of 2 mM MZ stock solution are added to 1 ml of DMF reaction solution, the DMF photo-oxidation rate is reduced. The percentage inhibition of light-induced photo-oxidation of DMF by added carotenoid was then recorded.
Determination of chemoprotective potential of MZ
A total of 30 male Swiss albino mice were divided into 5 groups of 6 animals each and were grouped as follows: group I: normal, group II: Cisplatin alone (Cisplatin control), group III: Cisplatin + sunflower oil (Vehicle control), group IV: Cisplatin + MZ (50 mg/kg body weight [bw]) and group V: Cisplatin + MZ (250 mg/kg bw). These doses were selected because of the previous reports on the hepatoprotective activity of MZ. 8 Animals in groups II–V received a single dose (intraperitoneally) of cisplatin (16 mg/kg bw). Oral administration of MZ was started 5 days prior to cisplatin injection and was continued till the animals were killed by diethyl ether-anaesthesia after 3 days; blood and kidney were collected for various biochemical analyses.
Effect of MZ on haematological profile
Blood was collected by heart puncture and was stored in heparinised vials to analyse various haematological parameters. Haemoglobin was estimated as cyanmethemoglobin formation using Drabkin’s method. 11 Total white blood cells (WBCs) were measured after diluting the blood in Turk’s fluid and counting them using a haemocytometer. 12
Effect of MZ on bone marrow cellularity
Femurs of all animals were taken and bone marrow cells from both femurs were flushed in to phosphate-buffered saline containing 10% goat serum to determine bone marrow cellularity. 13
Effect of MZ on α-esterase activity
Bone marrow cells from the above preparation were immediately smeared on a clear glass slide, air dried, stained with α-naphthyl acetate and then counterstained with haematoxylin. 14 α-Esterase-positive cells were counted and expressed as number of positive cells/4000 bone marrow cells.
Effect of MZ on renal function
Serum separated from the blood was used to analyze various kidney function parameters. Serum creatinine level was determined by alkaline picric acid method using a diagnostic kit (Span Diagnostic Pvt Ltd, Ernakulam, Kerala, India). Serum urea was determined by diacetylmonoxime reagent (modified Berthelot methodology) using a diagnostic kit (Span Diagnostic Pvt Ltd).
Effect of MZ on oxidative stress in kidney
Kidneys were quickly removed and washed with ice-cold normal saline, and homogenates (10% w/v) were prepared in 0.1 M Tris HCl. Lipid peroxidation (LPO) in the homogenate was estimated by thiobarbituric acid method. 15 Formation of CDs and tissue hydroperoxides (HPs) in the homogenate were determined by the modified method of John and Steven. 16
Effect on antioxidant enzyme profile of the kidney
Kidney homogenate (10%) was centrifuged at 1000 rpm for 30 min at 4°C and was used for the analyses of antioxidant enzymes. Superoxide dismutase (SOD) activity in the homogenate was measured by NBT reduction method. 17 Catalase (CAT) activity was estimated by measuring the rate of decomposition of hydrogen peroxide at 240 nm. 18 Assay of glutathione peroxidase (GPx) was carried out based on the oxidation of GSH in the presence of H2O2, 19 and the level of GSH in the tissue homogenate was analysed based on its reaction with DTNB. 20 Estimation of the total protein was carried out by the method of Lowry et al. 21
Histopathological analysis
A small portion of kidney was taken for histopathological analysis. Immediately after removal, kidney was washed in phosphate-buffered solution, fixed in 10% formaldehyde solution and then embedded in wax. Sections (4 µm) were taken and stained with haematoxylin–eosin.
Statistical analysis
The values were expressed as mean ± SD. The mean values were statistically analyzed using one-way analysis of variance using graph pad Instat 3 software (GraphPad Software, Inc. La Jolla, USA) followed by appropriate post hoc test (Dunnett multiple comparison test). Significant levels for cisplatin control group were determined by comparison with normal group; whereas significant levels for MZ-treated and vehicle control groups were determined by comparison with cisplatin control group. A p < 0.05 was considered statistically significant.
Results
Singlet oxygen quenching ability of MZ was investigated by UV-absorption spectroscopy using DMF as the reporter compound. When different concentrations of MZ (10, 20, 30, 40, 50 and 60 µg/ml) were added to DMF reaction solution, the DMF photo-oxidation rate was reduced in a concentration-dependent manner. The concentration needed for 50% inhibition (IC50) of singlet oxygen was found to be 38 µg/ml (Figure 3).
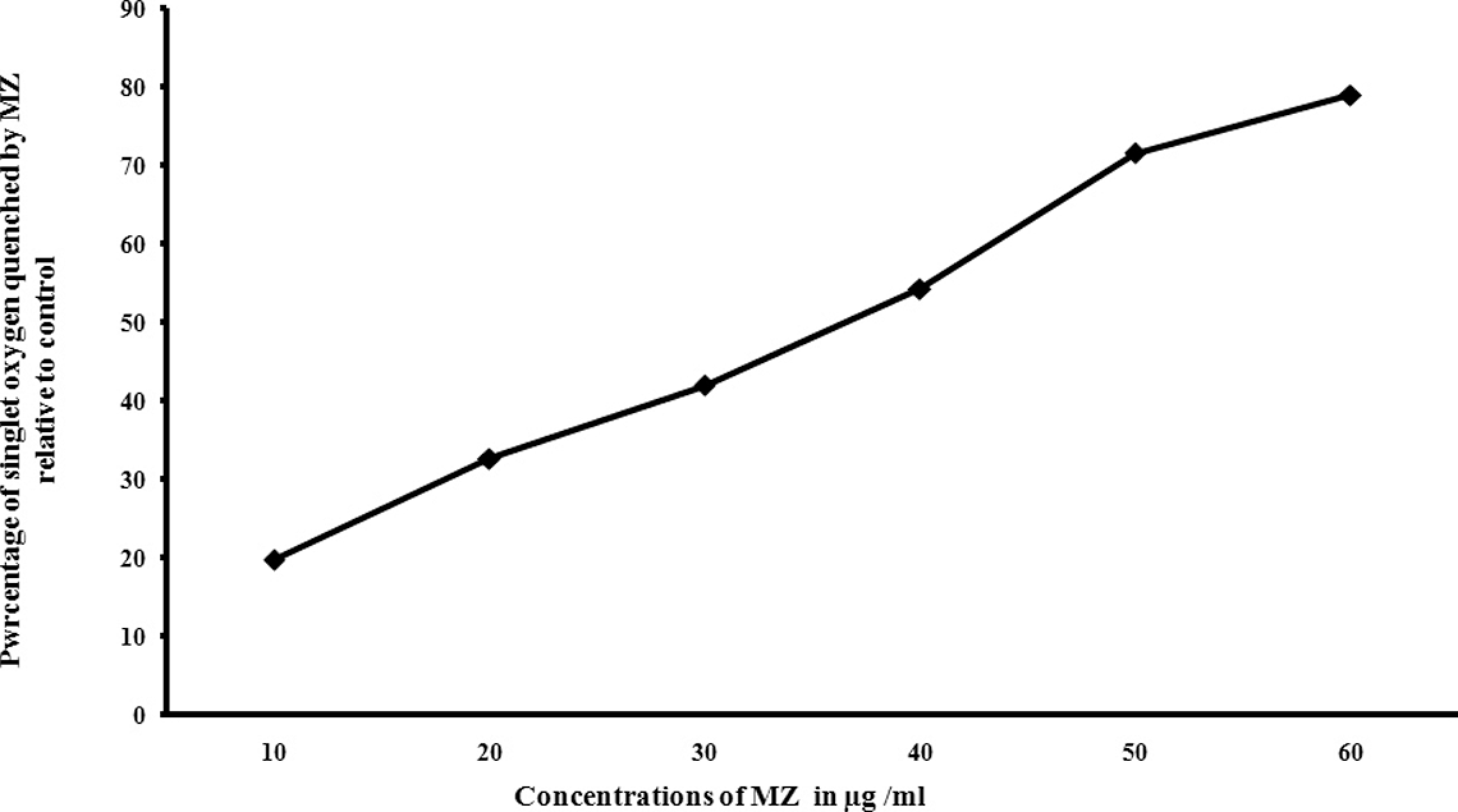
Measurement of singlet oxygen quenching ability of MZ by DMF assay. Different concentrations of MZ was added to 1 ml of 0.2 mM DMF reaction solution in ethanol, and the absorption spectra at 215 nm were recorded on a spectrophotometer after solution was illuminated for 5 min. The difference in spectra between DMF and illuminated DMF solution were used as control. The difference in spectra between illuminated carotenoid solution and illuminated DMF solution were used to calculate the relative percentage of singlet oxygen quenched by MZ. MZ: meso-zeaxanthin; DMF: 2,5-dimethyl furan.
Total WBC count, haemoglobin content, bone marrow cellularity and α-esterase cells were considerably decreased after cisplatin challenge in both vehicle and cisplatin control groups. Treatment with MZ significantly increased these levels in the drug-treated groups in a dose-dependent manner (Tables 1 and 2).
Effect of MZ on haematological parameters during cisplatin-induced nephrotoxicity a

MZ: meso-zeaxanthin; WBC: white blood cell.
a Values are expressed as mean ± SD; n = 6. Values were statistically analysed using one-way analysis of variance followed by Dunnett multiple comparison test.
b p < 0.001 is significantly different from normal group.
c p > 0.05 is significantly different from cisplatin control groups.
d p < 0.001 is significantly different from cisplatin control groups.
Effect of MZ on bone marrow cellularity and α-esterase-positive cells during cisplatin-induced nephrotoxicity a
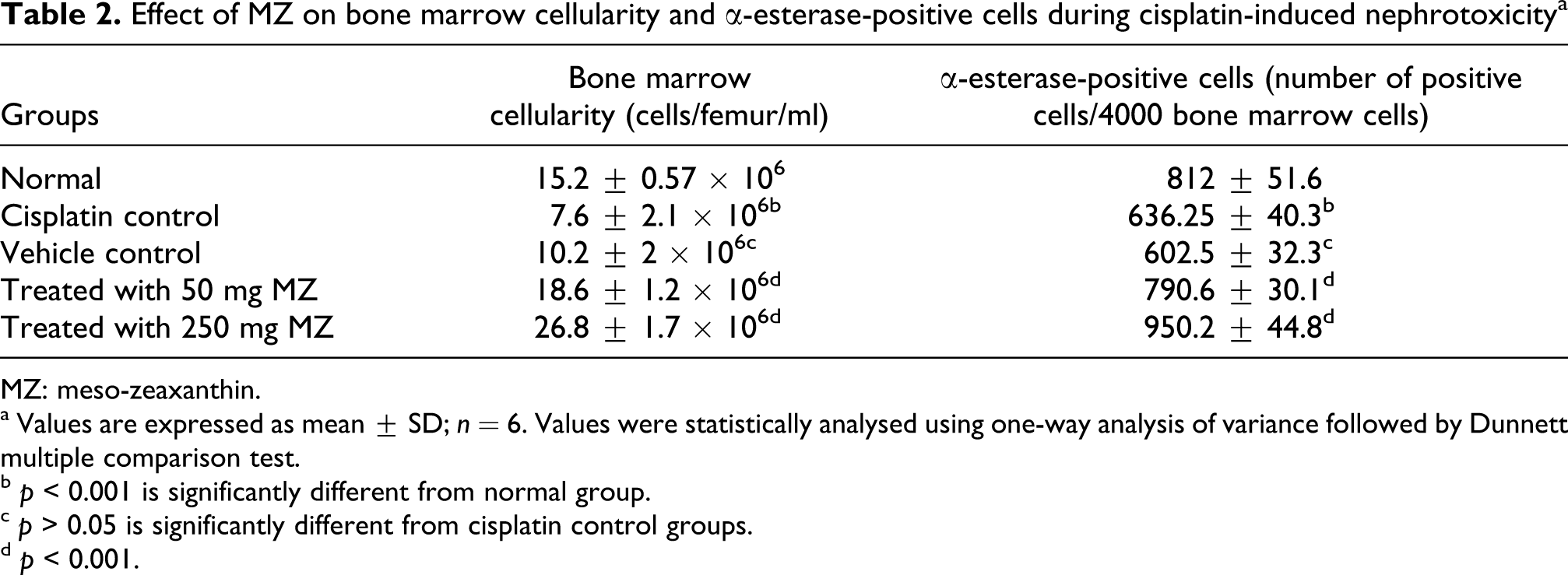
MZ: meso-zeaxanthin.
a Values are expressed as mean ± SD; n = 6. Values were statistically analysed using one-way analysis of variance followed by Dunnett multiple comparison test.
b p < 0.001 is significantly different from normal group.
c p > 0.05 is significantly different from cisplatin control groups.
d p < 0.001.
Renal function markers like urea and creatinine were found to be markedly elevated in both cisplatin control (i.e., 154.36 ± 2.8 and 2.7 ± 0.04, respectively) and in vehicle control (150.4 ± 0.57 and 2.58 ± 0.9, respectively) animals when compared with that of normal animals (37.2 ± 2.09 and 0.75 ± 0.04, respectively), indicating renal damage. These increased levels were decreased significantly by MZ treatment. In the 250 mg/kg bw drug pretreated group, level of urea was 50.3 ± 3.04 and level of creatinine was 0.9 ± 0.04 (Figure 4).

Effect of MZ on serum parameters during cisplatin-induced nephrotoxicity. Animals were pretreated with MZ at different doses 5 days prior to cisplatin (16 mg/kg body weight ) injection and were killed 72 h after cisplatin treatment, and parameters like (a) serum urea and (b) serum creatinine were analysed. Values are expressed as mean ± SD; n = 6. Values were statistically analysed using one-way analysis of variance followed by Dunnett multiple comparison test. a: p < 0.001 is significantly different from normal group; b: p < 0.001 is significantly different from cisplatin control groups; ns: p > 0.05 is significantly different from cisplatin control groups. MZ: meso-zeaxanthin.
The levels of SOD, CAT, GPx and GSH were decreased in the control group of animals. MZ administration significantly enhanced these antioxidant systems in the kidney (Table 3). LPO, HP and CD, the markers of tissue damage, were high in cisplatin-treated control group when compared with those of normal. Treatment with MZ significantly reduced these elevated levels to normal levels (Table 4).
Effect of MZ on antioxidant enzymes and GSH in the cisplatin-treated kidney a

MZ: meso-zeaxanthin; GSH: glutathione; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase.
a Values are expressed as mean ± SD; n = 6. Values were statistically analysed using one-way analysis of variance followed by Dunnett multiple comparison test.
b p < 0.05 is significantly different from normal group.
c p < 0.01 is significantly different from normal group.
d p < 0.001 is significantly different from normal group.
e p > 0.05 is significantly different from cisplatin control groups.
f p < 0.001 is significantly different from cisplatin control groups.
Effect of MZ on oxidative stress markers in cisplatin-treated kidney a

MZ: meso-zeaxanthin; LPO: lipid peroxidation
aValues are expressed as mean ± SD; n = 6. Values were statistically analysed using one-way analysis of variance followed by Dunnett multiple comparison test.
b p < 0.001 is significantly different from normal group.
c p > 0.05 is significantly different from cisplatin control groups.
d p < 0.001 is significantly different from cisplatin control groups.
e p < 0.01 is significantly different from cisplatin control groups.
Histopathology of kidney of the control group animals showed many atrophied glomeruli, Bowman’s capsule appear widened, renal tubules showed atrophy in many areas and lumen of many of them contained hyalinised material; interstitial tissue also showed scattered lymphocytes and plasma cells. Such changes were found to be minimal in the MZ-treated animals, indicating its protective role (Figure 5).
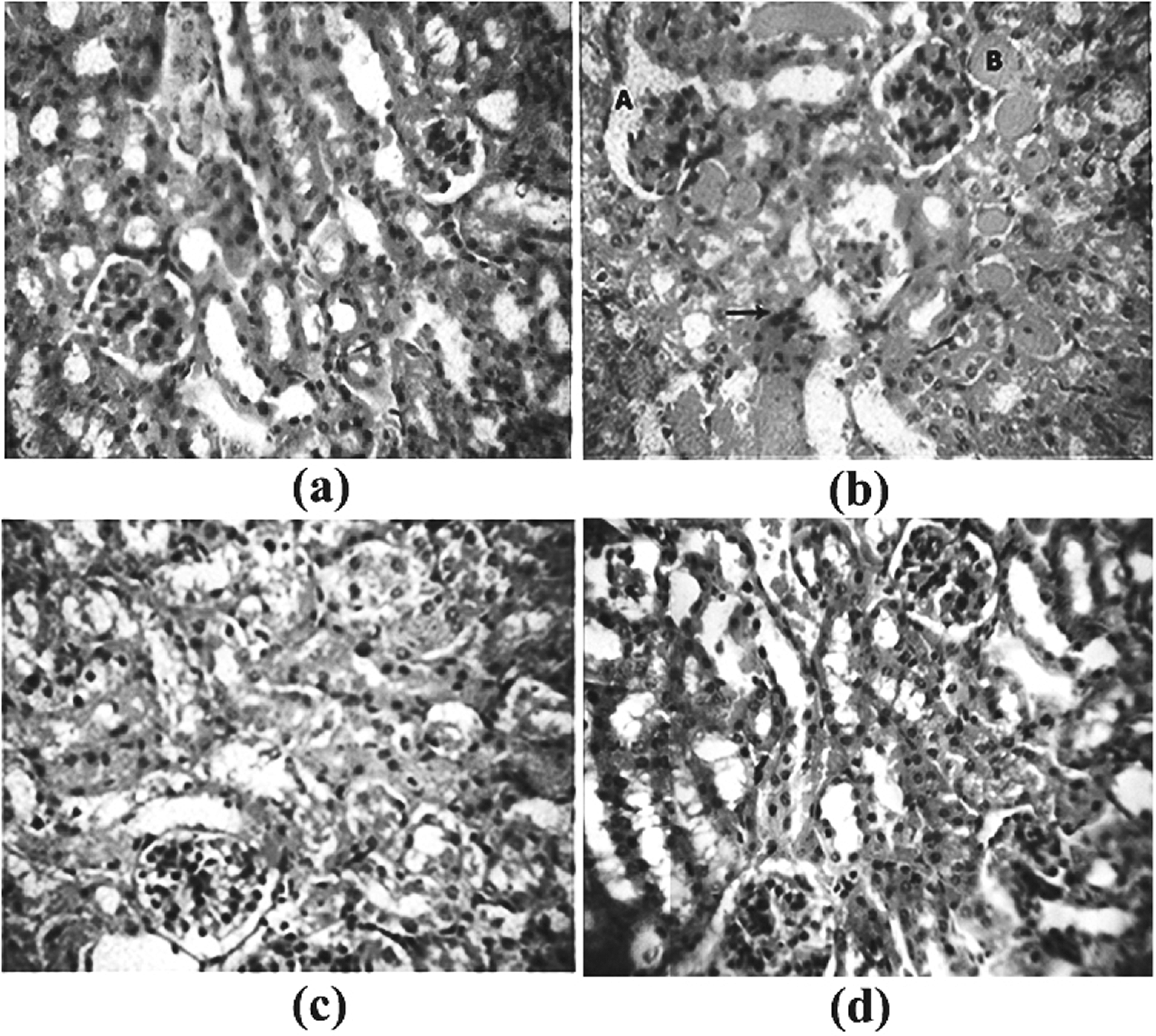
Histopathological analysis of cisplatin-treated kidney. (a) Normal histology of a kidney. (b) Cisplatin-alone control; ‘A’ indicates widened and atrophied glomerulus and Bowman’s capsule; ‘B’ indicates hyalinization of renal tubules, indicating infiltration of plasma cells in the interstitial tissue. (c) Cisplatin + 50 mg/kg body weight MZ-treated histology. (d) Cisplatin + 250 mg/kg body weight MZ-treated histology (haematoxylin–eosin staining, ×400). MZ: meso-zeaxanthin.
Discussion
Cisplatin continues to be a mainstay chemotherapeutic agent, but dose-limiting nephrotoxicity remains a major side effect. Platinum compounds are believed to mediate their cytotoxic effects through their interaction with DNA. In an aqueous environment, the chloride ligands of cisplatin are replaced by water molecules, generating a positively charged electrophile. This electrophile reacts with nucleophilic sites on intracellular macromolecules like DNA to form inter- and intra-strand crosslinks, thereby arresting DNA synthesis and replication in rapidly proliferating cells and in turn induces apoptosis.
Many chemotherapeutic agents including cisplatin cause myelosuppression. This could be understood from the decrease in the levels of total WBC count, haemoglobin content, bone marrow cellularity and α-esterase-positive cells of the cisplatin-challenged control group animals. But administration of MZ significantly reversed the myelosuppression produced by cisplatin in a dose-dependent manner, indicating the immunomodulatory potential of carotenoid MZ.
Cisplatin induces the generation of various ROS through inactivation of cellular antioxidant system, disruption of mitochondrial respiratory chain or interaction with microsomal cytochrome P450. 22 Highly potent ROS appears to target multiple cellular components, such as lipids, proteins and DNA and activate multiple signalling pathways and thereby implicated in the pathogenesis of acute cisplatin-induced renal injury. 23 Antioxidant enzymes and GSH are among the endogenous system that are available for the removal or detoxification of these free radicals and their products. The antioxidant status in the kidney tissue of mice was found to be decreased after cisplatin administration and was significantly improved by the MZ treatment in a dose-dependent manner. Various oxidative stress markers in the kidney, such as LPO, CD and HP, which were markedly elevated by cisplatin-challenge, were decreased to normal levels by pretreatment with MZ.
Our previous studies on MZ revealed that this carotenoid has good antioxidant activity both in vitro and in vivo. Antioxidant potential of MZ was found to be significantly higher than that of ascorbic acid and trolox. 6 In the present study, we evaluated the singlet oxygen quenching potential of MZ, and its IC50 was found to be 38 µg/ml. Similar studies using electron paramagnetic resonance (EPR) spectra have also been reported. 24
There are no reports on the occurrence of MZ naturally in blood; most MZ is probably endogenously synthesised from lutein. However, MZ is bioavailable when taken as a supplement and is found to effectively increase the macular pigment levels. 25 Toxicity studies on this compound have shown that it is nontoxic. 26
MZ satisfies most of the properties of a good chemoprotector pertaining to its nontoxicity profile, acceptable route of administration (oral), antioxidant status and immunomodulatory effects. So a combination therapy of cisplatin with MZ will have a profound beneficial effect in the treatment of cancer. Further studies on its molecular mechanism are required.
Footnotes
Acknowledgements
The authors thank M/s. Omni Active Health Technologies Pvt Ltd, for providing funds to carry out this work, and Dr Jayant Deshpande and Mr Abhijit Bhattacharya for their interest in this work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
