Abstract
It is well established that hepatocellular carcinoma (HCC) develops in a multistep process, from chronic hepatitis, cirrhosis to HCC. Adipose tissue is not simply an energy storage organ but also a secretory organ, producing a variety of bioactive molecules known as adipokines, including adiponectin and leptin. Hyaluronic acid (HA) is an extracellular matrix protein, often associated with a variety of human cancers. Our retrospective study determines serum levels of adiponectin, leptin, and HA in HCC of cirrhotic and noncirrhotic patients and compares these levels to patients with cirrhosis and normal subjects. Noncirrhotic HCC (n = 19), cirrhotic HCC (n = 50), cirrhosis (n = 36) patients and twenty one age-, sex-, and body mass index (BMI)-matched normal healthy controls were subjected in the present study. Serum adiponectin, leptin, and HA levels were determined using enzyme-linked immunosorbent assay technique. Levels of serum adiponectin were significantly higher in the cirrhosis and cirrhotic HCC groups than in the normal subjects, whereas serum HA levels were found to significantly increase in all three patients groups. The elevation of serum leptin in our HCC patients, regardless of being cirrhotic or noncirrhotic, but not in the patients with cirrhosis, may shed some light on the significance of serum leptin level in HCC. Further studies are recommended to evaluate the prognostic value of serum leptin level in HCC.
Introduction
Hepatocellular carcinoma (HCC) is a major worldwide health problem and represents the third most common cause of cancer-related death worldwide. 1 There is a considerable geographical variation in the incidence of HCC. 2 In Egypt, up to 90% of HCC cases were attributed to hepatitis C viral (HCV) infection. 3 Approximately 14% of the population in Egypt is infected with HCV and more than 8000 new cases are predicted by 2012. 4 Studies of HCV progression to HCC are expected to provide new insights into the management of this increasingly significant health problem. 5 Over the course of 20 years or more, 10%–30% of HCV carriers develop cirrhosis; patients with cirrhosis have an annual risk of 1%–2% for developing HCC. 6
The relevance of adipose tissue to hepatic diseases is indicated by the higher prevalence of cirrhosis and HCC in obese patients. 7 However, the underlying mechanisms that link obesity to various types of human cancers, including HCC, are poorly understood. One possible hypothesis is deregulated adipokines. The term ‘adipokines’ (adipose tissue cytokines) comprises polypeptide factors which are expressed significantly, although not exclusively, by adipose tissue in a regulated manner. Besides adipocytes, accounting for one third of the cells, resident and infiltrating macrophages and components of the vascular stroma markedly contribute to the production of adipokines. As a result, chronic inflammation in the cirrhotic liver adipose tissue is associated with a modification in the pattern of adipokine secretion. Adiponectin and leptin are the best-studied molecules in this class, but cytokines such as tumor necrosis factor-α (TNF-α) or interleukin-6 (IL-6) and several complement components are also secreted at high levels by the adipose tissue, suggesting a possible link to the immune system. 8,9
Adiponectin is a 244 amino acid protein that plays a pivotal role in metabolic liver diseases. Notably, adiponectin circulates at high concentrations (0.5–30 μg/ml) in plasma under normal physiological conditions. The ease with which the levels of adiponectin can be measured, owing to its high abundance, small diurnal variation and high stability in plasma, has made it an attractive target for measurements in clinical settings. 10 In normal humans and mice, adiponectin expression is restricted to adipose tissue 11 or adipocytic cells within, for example, the liver. 12 Moreover, specific adiponectin receptors that are expressed in the liver and other tissues have been cloned. 13 Although adiponectin has emerged as a valuable biomarker to monitor insulin sensitivity, cardiovascular risk, and inflammation, its significance in the setting of chronic liver diseases still remains unknown. 10
However, carbon tetrachloride-treated mice have been reported to show a dramatic increase in the expression of adiponectin protein as well as mRNA within hepatocytes, possibly implicating adiponectin in contributing to various metabolic abnormalities observed in patients with liver cirrhosis. 12 Hypoadiponectinemia was found in patients with nonalcoholic steatohepatitis (NASH). 14 It has been recently reported that the levels of serum adiponectin in vivo are inversely associated with the risk of endometrial cancer, postmenopausal breast cancer, leukemia, colon cancer, gastric cancer, and prostate cancer. 15 However, there is no sufficient information on the levels of serum adiponectin in liver cancer.
Leptin, a product of the obese gene, is predominant in the adipose tissue but is also found at many other sites. 16 Leptin binds to the leptin receptor (ObR) and exerts its effects in an autocrine or paracrine manner. 17 In addition to its potential association with hematopoiesis, thermogenesis, obesity-related diseases, angiogenesis, 18 and modulation of immune responses, 19 some authors have suggested that leptin is also associated with the initiation and progression of human cancers. 20 It has been shown that leptin has a potential role in the development of HCC. 21 However, the biological and clinical roles that leptin has in HCC are still undetermined. Leptin was also found to promote proliferation, migration, and invasiveness of HCC cells 22 and to increase proliferation and metastatic potential of cholangiocarcinoma cells. 23 Mitogenic action of leptin in HepG2 cells has been recently shown. 24 It is proposed that leptin is a key regulator of the malignant properties of HCC cells through modulation of human telomerase reverse transcriptase, a critical player of oncogenesis. 25 Indeed, the authors reported that as the high expression of leptin and its receptors in HCC liver tissues was not found to be correlated with body mass index (BMI), it could be assumed that the production of leptin in HCC liver is not directly regulated by the adipose tissue deposit but also reflects the intricate interactions taking place into the tumorigenic microenvironment.
Hyaluronan (hyaluronic acid [HA]) is a glycosaminoglycon of the extracellular matrix that is a prominent factor in serum, whenever rapid tissue growth occurs. This takes place particularly during embryogenesis and tumor growth and spread. Clinically, levels of HA on the surface of tumor cells often correlate with cancer aggressiveness. 26 Investigations of various human tumors, such as HCC, 27 several types of lung cancer, 28 and human pancreatic carcimona 29 have demonstrated a high concentration of HA in tumor tissues.
Studies have also shown that the levels of HA are often increased in the serum of patients with various malignant tumors, such as multiple myeloma, 30 breast cancer, 31 bladder cancer, 32 malignant epithelial ovarian tumors, 33 and oral cancer. 34 In addition, it is demonstrated that the level of serum HA is significantly elevated in patients with metastatic disease when compared with sera of patients without metastatic disease, and lower HA concentrations were observed in patients responding to chemotherapy 31 On the other hand, HA has been implicated in regulating tumor malignant behaviors, for example, anchorage-independent growth, tumor cell motility, secretion of matrix metalloproteinases, and tumor angiogenesis. 35
The present study investigates serum levels of adiponectin, leptin, and HA in HCC of cirrhotic and noncirrhotic patients and compares these levels to patients with cirrhosis and normal healthy subjects.
Materials and methods
Patients
This retrospective study was carried out on 69 patients with HCC (male = 43 and female = 26) and 36 patients with liver cirrhosis (male = 23, female = 13, mean age 52.97 ± 1.25 years; range: 40–70 years, BMI: 27.11 ± 1.18 kg/m2) admitted to the Internal Medicine Department of Faculty of Medicine, Ain Shams University, Cairo, Egypt, from January 2008 to February 2009. Subjects were excluded from the study if they were known to have any disease other than liver cancer or cirrhosis. Patients with a history of alcohol abuse, renal insufficiency, proteinuria, suspected infections, clinically overt diabetes mellitus, thyroid dysfunction, or any other endocrine disorder were also excluded from the study. No hormone or thyroid-regulatory medication was administered.
All HCC patients were newly diagnosed and none had received any form of anti-cancer therapy before collection of blood samples for biochemical analysis. Diagnosis of HCC was confirmed by pathology, cytology, imaging (computer tomography and ultrasound), and serum α-fetoprotein (AFP). All patients (n = 105) were infected with HCV. Each patient’s viral infection status was determined by assaying hepatitis B surface antigen (HBsAg), hepatitis B core antigen (anti-HBc), anti-HCV and by polymerase chain reaction for HCV RNA. Cirrhosis was proven through physical examination and imaging. The HCC patients were divided into two groups based on whether their HCC was associated with cirrhosis or not and termed the cirrhotic HCC group (n = 50, male = 31, female = 19; mean age: 58.88 ± 1.37 years, range: 35–76 years, BMI: 30.20 ± 1.21 kg/m2) and noncirrhotic HCC group (n = 19, male = 21, female = 7, mean age: 59.21 ± 2.48 year, range: 35–76 years, BMI: 25.89 ± 1.71 kg/m2), respectively.
A total of 21 healthy control volunteers were matched to the patient population by sex (male = 13, female = 8), mean age (55.7 ± 3.54 year, range: 20–71 years), and BMI (29.1 ± 1.2 kg/m2). None of them had a history of endocrine disorders or hepatic disease; all had completely normal liver function tests, normal ultrasound of the liver and biliary system, and negative serological findings for viral and autoimmune liver disease and diabetes. Body weight and height of the participants were recorded at the time of first examination and BMI was calculated as kilograms per meter squared. Clinical data of the patients and normal controls are given in Table 1. Informed consent was obtained from all participating subjects before the study. The study protocol was approved by the ethics committee of the Faculty of Pharmacy, Cairo University and the National Hepatology and Tropical Medicine Institute, Cairo, Egypt, and conformed to the ethical guidelines of the 1975 Helsinki Declaration.
Blood sampling and biochemical assays
Fasting venous blood samples (∼2 ml) were collected by trained laboratory technicians. A portion of blood was allowed to clot and then centrifuged at 3500g for 5 min to separate the serum used for assessment of aspartate aminotransferase (AST), alanine aminotransferase (ALT), and γ-glutamyltranspeptidase (GGT) activities, total bilirubin, direct bilirubin AFP, viral infection status, and glucose concentrations. Serum aliquots were stored at −80°C until assayed and thawed immediately before the measurements of adiponectin, leptin, and HA levels. Another portion of blood was collected in vacutainer tubes containing citrate to separate plasma used for the assay of albumin and prothrombin time. AST, ALT, GGT activities, total bilirubin, direct bilirubin, albumin, prothrombin time, and glucose concentrations were assayed using Beckman CX4 chemistry analyzer (NY, USA, supplied by the Eastern Co. For Eng. & Trade-Giza, Egypt). AFP and viral status were measured using Abbott, Axyam (USA, Supplied by al kamal company Cairo, Egypt).
Serum level of adiponectin was determined with a commercially available AssayMax Human Adiponectin ELISA kit (Cat. No: EA2500-1, Assaypro, St. Charles, MO, USA) with sensitivity 0.5 ng/ml, and intra-assay and inter-assay variations were 4.1% and 7.2%, respectively. This assay employs a quantitative sandwich enzyme immunoassay technique. Serum level of leptin was measured using the BioSource leptin EASIA kit (Cat. No: KAP2281; BioSource Europe S.A, Nivelles, Belgium). EASIA is a solid-phase enzyme-amplified sensitivity immunoassay performed on microliter plate with a sensitivity of 0.1 ng/ml and intra-assay 3.6%. Serum HA level was determined using an enzyme-linked binding protein assay kit (Corgenix Medical Corporation, Broomfield, CO, USA, Prod. No.029-001) with sensitivity 10 ng/ml, and intra-assay and inter-assay variations were 4.2% and 6.2%, respectively. Levels of adiponectin (μg/ml), leptin (ng/ml), and HA (ng/ml) were calculated by interpolation from a reference curve generated in the same assay with reference standards of known concentrations. All assays were performed in duplicate according to the manufacturer’s instructions.
Statistical analyses
Statistical analysis was performed using the statistical package for social sciences (SPSS, USA). Data are expressed as means ± standard error. The chi-square test was used for the comparisons of proportions. Differences between the groups were assessed by one-way analysis of variance. Duncan’s test was performed for intergroup comparisons. Independent Student’s t test was used to compare means between two groups. Pearson’s correlation coefficient was used to assess possible associations between different parameters. A p < 0.05 was considered significant.
Results
Our healthy normal controls had serum adiponectin, leptin, and HA levels within the expected levels as reported previously for gender- and BMI-specific variations. 10,34,36 Within the patients’ groups, male and female had similar adiponectin and HA levels but significantly different serum leptin (p < 0.001; Table 2).
In comparison with an age-, sex-, and BMI-matched normal healthy control group, serum adiponectin levels in cirrhosis and cirrhotic HCC patients were significantly elevated (p < 0.001; Figure 1A), whereas no change was observed in the noncirrhotic HCC patients. Strikingly, increased serum adiponectin in cirrhotic HCC patient groups (n = 50; 36.68 ± 1.43 μg/ml vs. 11.95 ± 0.38 μg/ml, respectively) was negatively correlated with parameters of hepatic protein synthesis capacity such as albumin (r = −0.857, p < 0.01; Figure 2A) and prothrombin time (r = −0.625, p < 0.01; Figure 2B).
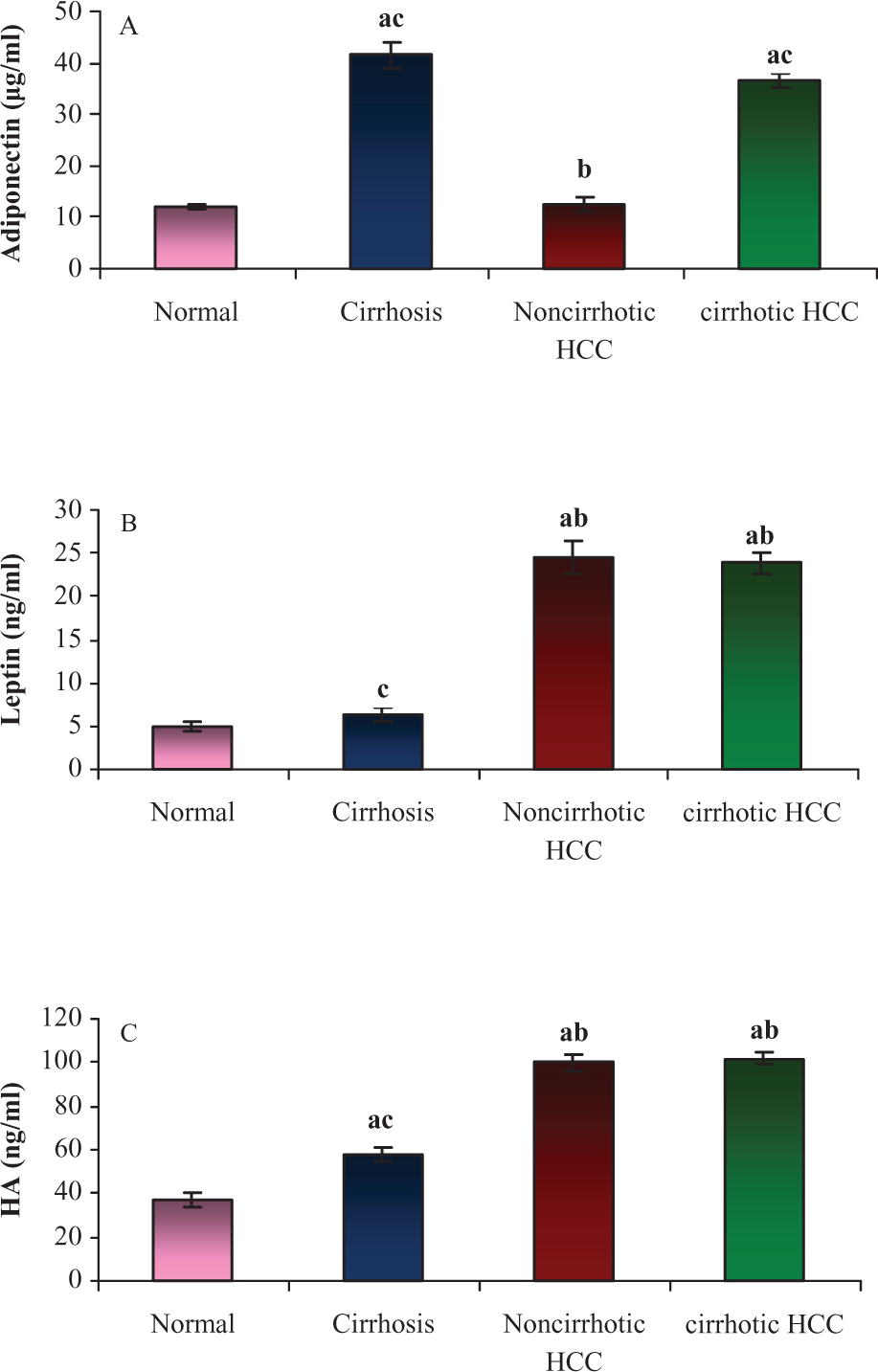
Serum levels of A : adiponectin, B : leptin, and C : HA in patients with cirrhosis, noncirrhotic HCC, cirrhotic HCC, and normal controls. a: Significant difference from normal group. b: Significant difference from cirrhosis group. c: Significant difference from noncirrhotic HCC group. Data represent means ± standard error. Values are statistically significant at p < 0.001. HA: hyaluronic acid; HCC: hepatocellular carcinoma.

Correlation analysis in cirrhotic HCC patients. Serum adiponectin level is negatively correlated with parameters of hepatic protein synthesis capacity such as (A) serum albumin levels and (B) prothrombin time and (C) positively with HA serum levels using Pearson’s correlation coefficient, p < 0.01. HA: hyaluronic acid; HCC: hepatocellular carcinoma.

Correlation analysis between serum AFP and leptin levels in noncirrhotic HCC patients and normal healthy controls (n = 71) using Pearson’s correlation coefficient at p < 0.01. AFP: α-fetoprotein; HCC: hepatocellular carcinoma;

Correlation analysis in noncirrhotic and cirrhotic HCC patients. Serum HA level is negatively correlated with parameters of hepatic protein synthesis capacity such as A: serum albumin levels and B: prothrombin time using Pearson’s correlation coefficient, p < 0.01. HA: hyaluronic acid; HCC: hepatocellular carcinoma.

Correlation analysis in noncirrhotic and cirrhotic HCC patients. Prothrombin time is negatively correlated with A: serum albumin levels and positively correlated with B: serum GGT activity using Pearson’s correlation coefficient, p < 0.01. HCC: hepatocellular carcinoma; GGT: γ-glutamyltranspeptidase.
Clinical details of the cirrhosis, noncirhotic HCC, cirrhotic HCC patients, and the normal controls

AST: aspartate aminotransferase; ALT: alanine aminotransferase; GGT: γ-glutamyltranspeptidase; AFP: α-fetoprotein; HCC: hepatocellular carcinoma; BMI: body mass index.
Data are presented as means ± standard error. Values are statistically significant at p < 0.001. Significance for gender and age is set at p < 0.05.
a Significant difference from normal group.
b Significant difference from cirrhosis group.
c Significant difference from noncirrhotic HCC group.
Levels of serum adiponectin, leptin, and HA of males and females in patients with cirrhosis, noncirrhotic HCC, cirrhotic HCC, and the normal controls

HA: hyaluronic acid; HCC: hepatocellular carcinoma.
Data are presented as means ± standard error.
* Presence of a significant difference between males and females by comparing studied patient groups with age-matched healthy control subjects using independent student’s t tests at p < 0.001.
Alternatively, a significant increase in serum leptin levels was observed in HCC patients whether cirrhotic or noncirrohotic (p < 0.001; Figure 1B), but not in the cirrhosis group when compared with normal healthy controls. A positive correlation (r = 0.54, p < 0.01; Figure 3) was revealed between the serum levels of AFP and leptin in cirrhotic HCC group (n = 50) when the sample size was increased (n = 71) by including the normal healthy population (n = 21).
Elevated serum HA levels were observed in all studied patient groups, with significantly higher levels in the HCC patients (p < 0.001; Figure 1C). The elevated serum HA levels in noncirrhotic and cirrhotic patients were found to be negatively correlated with albumin (r = −0. 70, p < 0.01; Figure 4A) and prothrombin time (r = −0.50, p < 0.01; Figure 4B), whereas a positive correlation was observed with adiponectin (r = 0. 78, p < 0.01; Figure 2C) in cirrhotic HCC patients. Moreover, prothrombin time in the noncirrhotic HCC and cirrhotic HCC patients groups was found to be negatively correlated with serum albumin level (r = −0.85 and −0.79, respectively, p < 0.01) and positively correlated with serum GGT activity (r = 0.65 and 0.56, respectively, p < 0.01), (Figure 5A and B).
Significant increase (p < 0.001) in serum levels of total bilirubin, direct bilirubin, albumin and glucose were detected in cirrhotic and noncirrhotic HCC patients but not in the cirrhosis group when compared with normal healthy controls (p < 0.001; Table 1). On the other hand, an apparent increase in the activity of serum GGT and serum AFP level was observed in liver cirrhosis group when compared with normal healthy controls but failed to reach statistical significance.
Discussion
Adiponectin and leptin are physiologically active polypeptides produced abundantly by adipose tissue, whose multiple functions have started to be understood in the last two decades. As observed in our study, a significant increase in levels of serum adiponectin was associated with cirrhosis and not with HCC.
The pathogenesis of increased level of adiponectin in liver cirrhosis has not been entirely explained. It can be postulated that this increase is due to less clearance of adiponectin in the liver of patients with cirrhosis. In fact, high levels of adiponectin found after bile-duct ligation in mice and in human bile from patients with cholestasis suggest that biliary secretion is involved in adiponectin clearance. 37 Moreover, this increase may be at least in part due to decreased hepatic catabolism as suggested by Tietge et al. 38 This is consistent with a previous study that also linked increased level of adiponectin with liver diseases that have a biliary etiology. 37
It has been recently concluded 10 that adiponectin levels show a two-phase distribution in liver disease. In steatosis and NASH, levels of adiponectin are low, in keeping with a proposed protective effect in these conditions and suggesting elevation of adiponectin as a desirable therapeutic target in nonalcoholic fatty liver disease and in the progression toward cirrhosis. Tietge et al. 38 reported that adiponectin metabolism remains normal in patients with liver disease until cirrhosis is manifested. This was clearly demonstrated among patients of the present study who suffered from liver cirrhosis. Patients with early cirrhosis had normal levels of circulating adiponectin comparable with patients with liver fibrosis and had significantly lower levels of plasma adiponectin than patients with more advanced cirrhosis. This is in harmony with the negative correlations of adiponectin with markers of hepatic protein synthesis, such as albumin (Figure 2A) and coagulation factors such as prothrombin time (Figure 2B) in our cirrhotic HCC patients. These correlations are in agreement with Tietge et al. 38 and demonstrate the link between increased serum adiponectin level, decreased hepatic extraction of adiponectin and deterioration of hepatic function.
In fact, little information is available on the role of adiponectin in liver cancer. Unlike previous cancer studies 15 that reported a decrease in the levels of serum adiponectin, there was no significant change in its level in our noncirrhotic HCC patients. This may be explained on the finding of our study and the previous report of Tietge et al. 37 that adiponectin did not correlate with BMI, which is one of the established parameters influencing adiponectin levels in normal controls, obese patients, patients with diabetes or coronary artery disease. This finding suggests that the physiological adiponectin regulation may be significantly affected in liver diseases and HCC by other parameters. 38 Indeed, this nonsignificant change in level of serum adiponectin may be attributed to small sample size of noncirrhotic HCC patients (n = 19).
In the study of Yoda-Murakami et al., 12 the expression of adiponectin was reported to be increased in mouse livers undergoing CCl4-induced damage. This experiment clearly demonstrates that adiponectin is synthesized by liver cells under appropriate conditions. Considering that adiponectin has high affinity for collagen types I and III, 39 it seems reasonable to consider that circulating adiponectin is captured by collagen types I and III produced by activated liver cells during the early stage of the injury. Subsequent production of proinflammatory cytokines such as IL-6 by surrounding inflammatory cells may trigger the expression of adiponectin in parenchyma cells. This speculation is supported by the fact that the promoter region of the adiponectin gene contains the consensus motif for CCAAT/enhancer binding protein beta (C/EBPβ) (involved in the regulatory expression of many inflammation-associated genes), which is known to be activated by IL-6. The observations that adiponectin attenuates TNF signaling 40 and that it has direct effects on hepatocytes via a specific receptor (AdipoR2) suggest that it might act as an anti-inflammatory protein involved in the repair process of tissue injury. 37
A significant increase in serum glucose level was observed in all HCC patients as compared with patients with only liver cirrhosis and normal healthy controls. Hyperglycemia was reported to promote tumorgenesis by several pathways. The elevated level of serum glucose is advantageous for the increased DNA synthesis of the tumor cells. It provokes release of free radicals which will cause derangement of both the DNA and the enzymes having a role in the repair mechanisms. 41 An apparent increase in serum AFP level that failed to reach statistical significance was observed in the cirrhosis group in our study. This apparent increase in the level of serum AFP in our liver cirrhosis patients is in harmony with previous reports that revealed the elevation of AFP levels in many chronic hepatitis patients without HCC 42 and in some patients with cirrhosis and concomitant high inflammatory activity. 43 Similarly, there are patients with large HCC masses with negative AFP results. 44 This AFP level overlap between HCC and non-HCC that may dampen the detection ability and can be attributed to excessive regeneration after extensive hepatocyte necrosis, and thus, redundant neogenesis may result in a rise in AFP level. 45
The increase in serum leptin levels in our cirrhotic HCC and noncirrhotic HCC patients but not in the cirrhosis group have confirmed findings of other studies that have suggested the association of leptin with the initiation and progression of human cancers. We have also demonstrated that this cannot be attributed to the coinciding cirrhosis as there was no increase in the leptin levels in the serum of patients with just liver cirrhosis. It has been shown that circulating leptin levels are considerably higher in patients with HCC than in normal healthy controls. 46 Leptin has been associated with cancer development both directly and through increased angiogenesis. Leptin acts on endothelial cells 18 and upregulates expression of vascular endothelial growth factor by hepatic stellate cells, and lack of leptin action reduces angiogenesis and formation of preneoplastic foci in experimental steatohepatitis. 9 Moreover, ObR is expressed at higher levels in human HCC, and poorly differentiated HCCs have higher vascularization and ObR expression, showing that leptin/ObR correlate with angiogenesis in HCC in vivo. 22 Leptin was also reported to promote proliferation, migration, and invasiveness of HCC cells 22 and to increase proliferation and metastatic potential of cholangiocarcinoma cells. 23 Leptin’s mitogenic action in HepG2 cells has been recently shown. 24 The positive correlation revealed in cirrhotic HCC group between serum levels of leptin and AFP upon increasing sample size (n = 71) by including the normal healthy population warrants further studies.
On the other hand, the increase in serum HA levels in all our groups of patients studied shows that HA concentrations cannot be used as a specific marker for a particular liver disorder. Increased serum HA levels in both patients with cirrhosis and in patients with a variety of different tumors have been reported previously. The elevated levels of serum HA in various liver diseases characterized by liver fibrosis and cirrhosis can be attributed to decrease in removal of HA from plasma, by a damaged liver and/or increase in hepatic production of HA during liver inflammation and release into circulation. 47 However, HA level was shown to be useful in monitoring the response to interferon treatment in patients with chronic hepatitis C. 48 Serum HA is elevated in patients with alcoholic liver disease and can be used to detect the progression from alcoholic fatty liver to cirrhosis. 49,50
The elevation in serum HA level in cancer patients can be attributed to overproduction due to overexpression of hyaluronan synthases (HAS) in human tumor cells, giving rise to elevated HA production. This mechanism is supported by the study of Kumar et al., 51 which demonstrated reduction in serum HA levels after surgical removal of tumors. By producing antiadhesive HA, HAS can allow tumor cells to release from the primary tumor mass. The interaction of HAS produced HA with receptors such as CD44 or Hyaluronan-mediated motility receptor (RHAMM) promotes the cell changes that allow for the cancer cells to infiltrate the vascular or lymphatic systems. 52 Previous studies have demonstrated that HA plays an important role in the progression of several tumor types and that perturbation of endogenous hyaluronan interactions in malignant mammary carcinoma tumor cells by overexpression of soluble CD44 inhibits growth and induces apoptosis under anchorage-independent conditions. 53 While normal cells, especially epithelia, undergo apoptosis if they become detached from their underlying or pericellular matrices, anchorage-independent growth is a distinguishing characteristic of many tumor cells. 54 It has also been previously shown that the urinary HA levels are 2.5- to 6.5-fold higher in bladder cancer patients and thus serve as a sensitive and specific marker for detecting bladder cancer, regardless of the tumor grade. 55 Higher levels of HA were detected in our cirrhotic and noncirrhotic HCCs than in cirrhosis group. In harmony with this observation, are the observed negative correlations with hepatic protein synthesis in cirrhotic and noncirrhotic HCC patients in our study (Figure 4A and B). Moreover, the positive correlation between levels of serum HA and adiponectin in cirrhotic HCC group (Figure 2C) may be related to their involvement in cirrhosis.
Taken together, based on the findings of our study, it can be concluded that serum levels of adiponectin, leptin, and HA differ regarding their significance in HCC. Increase in serum adiponectin level in our HCC patients can be attributed mainly to cirrhosis, whereas increased HA levels can be attributed to both cirrhosis and cancer. The increase in serum leptin levels in cirrhotic HCC and noncirrhotic HCC patients but not in the cirrhosis group may thus be attributed to HCC. Therefore, these results shed some light on the significance of leptin as a significant parameter in HCC. However, limitations to our study are small sample size and lack of significant correlation between serum leptin and AFP levels. The potential clinical value and possible prognostic impact of leptin serum levels in HCC patients should be evaluated in further prospective studies.
Footnotes
Acknowledgement
We thank Dr Wessam Ahmed, Internal Medicine Department of Faculty of Medicine, Ain Shams University, Cairo, Egypt, for generous and sincere help in collecting the studied samples.
The financial assistance for this work was provided by Faculty of Pharmacy, Cairo University, Cairo, Egypt.
