Abstract
The present work was to investigate the effects of flavonoid extracts from celery on oxidative stress induced by dichlorvos (DIC) in male Wistar rats maintained on a normal diet. The rats were given DIC through intragastric administration by the dose of 7.2 mg/kg·body weight (bw)/day and additionally added 5% flavonoid extracts to the diet for 4 weeks continuously. The activities of superoxide dismutase (SOD), glutathione peroxidase (GPx), catalase (CAT), glutathione-S-transferase (GST) and the content of malondialdehyde (MDA) in livers of rats were measured at the end of the experiment. Under the influence of DIC, there were significant decrease in the activities of SOD, CAT and GST and significant increase in GPx activity and MDA content. The results also showed that the activities of SOD, GST and CAT in the DIC-treated group declined significantly when compared with the flavonoid extracts group and the DIC + flavonoid extracts group, respectively. With regard to GPx activity and MDA content, significant increase were showed in the DIC-treated group in comparison to those in the flavonoid extracts group and the DIC + flavonoid extracts group, respectively. The observations presented lead us to conclude the harmful effects of DIC during the exposure and the protective role of flavonoids in minimizing these effects.
Introduction
Dichlorvos (DIC), one of the most widely used organophosphorus pesticides, has been used as a crop protectant as well as a general purpose public health insecticide in agriculture and public health programs since 1961. 1 DIC is also one of the most commonly used organophosphorus pesticides in China. Exposure of general public to DIC may occur via air, water or food as it gets readily absorbed through all routes of exposure. 1 Many studies have reported that exposure to low levels of commonly used pesticides could induce oxidative stress as indicated by malondialdehyde (MDA) accumulation and/or glutathione depletion, inhibition or induction of reactive oxygen species (ROS) and quenching endogenous antioxidant enzymes such as superoxide dismutase (SOD), glutathione peroxidase (GPx) and catalase (CAT) in rat tissues. 2–4 Thus, oxidative stress induced by the pesticides in vivo became a field of interest for scientists recently.
Flavonoids, a secondary metabolites from plants, are widespread in plant-derived foods such as vegetables, fruits, seeds and tea and are an integral part of the human diet. Dietary flavonoids were shown to mediate antioxidative, anti-inflammatory, antiproliferative and proapoptotic effects in various cell types. 5 Flavonoids act as antioxidants by chelating redox-active metals and by scavenging free radicals. 6–10 Chelation of both iron and copper by the carbonyl and hydroxyl groups of flavonoids prevents peroxyl radical and lipid peroxidation. 6–8 Flavonoids also function as terminators of free radicals by donation of electrons to form stable products. Flavonoids are very effective scavengers of hydroxyl and peroxyl radicals 6 as well as quenching superoxide radicals and singlet oxygen. 9,10 More recently, it has been shown that at low levels flavonoids provide antioxidant protection, but at high levels they cause DNA damage and apoptosis (cell death). 11
The detrimental health effects of chemicals in foods and/or the environment depend on several factors, one of which is interaction between chemicals in a mixture. Though most studies are conducted with individual chemicals, animals and humans are exposed to multiple chemicals in a complex mixture. Flavonoids in human diets are metabolized by the same pathway as toxic man-made chemicals such as pesticides and other environmental pollutants. Hence a brief investigation for the antioxidative protection of flavonoids against pesticide intoxication in vivo was of much interest.
Celery, which belongs to Umbelliferae family, is widely used as daily vegetable consumption in China. Recently, increasing attention has been paid to the green parts of the plant because of their physiological functionalities, such as lowering cholesterol levels, anti-inflammatory activity, antimicrobial activity and anticancer activity. 12–15 In the authors’ laboratory, the flavonoids content in celery, their metabolism in vivo and their population dietary intake 16,17 have been studied. Therefore, the present study was designed with an aim to carry out an investigation to find out the protective role of the antioxidative components present in celery against the adverse effects of DIC in vivo, based on lipid peroxidation and antioxidant enzyme profiles in rats.
Materials and methods
Chemicals and instruments
The standard kaempferol, quercetin, apigenin and luteolin were obtained from Sigma (St Louis, MO, USA). All other chemicals were of chromatography grade or analytical grade.
Kits for SOD, GPx, MDA, glutathione-S-transferase (GST) and CAT analysis were all purchased from Nanjing Jiancheng Bioengineering Institute (Nanjing, China). DIC (purity >95%) was supplied by Hebei Century Insecticides Limited Company (Hebei, China).
Waters 2690 High-performance liquid chromatography ([HPLC] Waters Corporation, USA), with a Waters 996 diode array detector and an ODS column (SYNERGI POLRR-RP80A, 250 mm × 4.60 mm), Waters Chromatography Workstation, Empower software; 722 spectrophotometer (Shandong Gaomi Analytical Apparatus Corporation, Shandong, China); Allegra™ 64R low-temperature high-speed centrifuge (Beckman, USA); Alpha1-4 vacuum freeze-drying machine (Christ, Germany).
Preparation and HPLC analysis for flavonoid extracts in celery
Fresh celeries were purchased in a local supermarket. First, they were cleaned, chopped into small pieces and mixed and later freeze-dried and were finely powdered and passed through a 40-mesh sieve. Then, samples were extracted using 80% methanol/water (v/v: 80/20) at 60°C twice, mixed carefully and concentrated. The concentrates were pooled and the final concentrate was lyophilized to obtain the dry matter (The celery extracts). The celery extracts were stored at −20°C for further analysis.
Instrument
Apparatus
Identification and quantification of flavonoid aglycons were achieved using the method established by Cao et al. 18 The contents of flavonoid extracts quercetin (Rt = 8.07), luteolin (Rt = 9.62), kaempferol (Rt = 11.87) and apigenin (Rt = 14.18) in celery were 0.03%, 0.92%, 0.46% and 1.71%, respectively (Figure 1).

Representative high-performance liquid chromatography chromatograms of flavonoids in mixed standards (A) and celery extracts (B). Qu: quercetin, Lu: luteolin, Ka: kaempferol, Ap: apigenin.
Animals and treatments
Thirty-two Wistar male 19 rats, weighing 180–220 g, were provided by Vital River Laboratory Animal Technology Limited Company (Beijing, China). They were housed under sterile conditions in wire cages in a room maintained at 22°C with 12-hour light–dark cycles. The rats had free access to food and distilled drinking water during the experiment. They were maintained in accordance with the National Institutes of Health guidelines for care and use of laboratory animals. The study protocol was approved by the animal ethical committee of Harbin Medical University.
After 7 days of acclimation, animals were weighed and randomly allotted to four groups (8 animals in each group) such that all groups had similar average initial body weights, namely control group (Group A), DIC-treated group (Group B), flavonoid extracts group (Group C) and DIC + flavonoid extracts group (Group D). Group A animals received a basal diet, AIN 93M, including cornstarch, casein, maltodextrin, sucrose, soybean oil, fiber, mineral mix, vitamin mix, L-cystine, choline bitartrate and tert-butylhydroquinone. Diets of both groups C and D contained 5% flavonoid extracts. DIC was dissolved in distilled drinking water. Animals of groups B and D were given DIC through intragastric administration (namely by gavage) at a dosage of 7.2 mg/kg body weight·(bw)/day, namely three times of no observed adverse effect level (NOAEL). The NOAEL was obtained after a 2-year study of toxicity and carcinogenicity, which was from 1993 Toxicity Assessment of DIC Residues in food, Chemical Safety Information from Intergovernmental Organizations (INCHEM), the WHO/FAO Joint Meeting on Pesticide Residues (JMPR). Animals were exposed to the regimens once daily for a period of 4 weeks. Every week, we evaluated the weight changes in rats of the four groups. At the end of the treatment period, rats were fasted for 12 hours. Then, the animals were killed with 1% of sodium pentobarbital anesthesia and livers were obtained and stored at –−80°C for further analysis, and all analyses were completed within 3 months of storing samples as the enzymes were stable for this period.
Enzymatic and nonenzymatic antioxidant activities
The tissues were comminuting using liquid nitrogen. The activities of SOD, GPx, CAT and GST and MDA content in liver were analyzed by assay kits. The activity of SOD was determined by the method of xanthine oxidase. GPx was assayed using the method of 5,5′dithiobis-2-nitrobenzoic acid. GST was assayed using the method of 1-chloro-2, 4-dinitrobenzene. CAT activity was measured spectrophotometrically and expressed as U/mg protein, by the reduction rate of hydrogen peroxide. Concentrations of the lipid peroxidation product, MDA, in hepatic homogenates were determined by thiobarbituric acid reactive substance assay based on the reaction of MDA with thiobarbituric acid to produce a complex that can be determined spectrophotometrically. Spectrophotometer wavelength of SOD, GPx, CAT and GST and MDA were 550 nm, 412 nm, 405 nm, 412 nm and 532 nm, respectively. The total protein in the tissue homogenates was determined by the method of Coomassie Brilliant Blue, 20 a method based on the principle that protein is combined with Coomassie brilliant blue G-250 into a protein-pigment conjugate, which is measured at the optical absorbance wavelength of 595 nm and is proportional to protein content. All experiments were fulfilled at room temperature. Other procedures were completed according to the directions of the assay kits.
Statistical analysis
All analyses were performed with the statistics software SPSS version 13.0. Differences among the experimental groups were analyzed using one-way analysis of variance. Significant differences between means were determined by the Student Neuman–Keuls test. Statistical significance was accepted at p < 0.05.
Results
Effects of DIC exposure on the dietary intake, body weight gain and liver weight
The weekly dietary intake of rats in the four groups showed no significant change after weeks 1, 2, 3 and 4, respectively (p > 0.05; Figure 2A). The body weights of rats in four groups were found to increase throughout the 4 weeks, as shown in Figure 2B. The body weight showed no significant change in the DIC-treated group when compared to the other three groups after weeks 1, 2, 3 and 4, respectively (p > 0.05). The body weight in control group, DIC-treated group, flavonoid extracts group and DIC + flavonoid extracts group were increased by 72.1%, 72.6%, 76.7% and 68.4% during the entire 4 weeks of the treatment schedule, respectively. Liver weight of rats in control group, DIC-treated group, flavonoid extracts group and DIC + flavonoid extracts group were 11.61 ± 1.25 g, 10.80 ± 1.05 g, 11.54 ± 1.40 g and 12.01 ± 1.90 g, respectively. And there were no significant differences between control group and every treated group (p > 0.05), respectively.

Effects of dichlorvos (DIC) exposure on the dietary intake (A) and body weight gain (B) of rats (mean ± SD, n = 8). The weekly dietary intake of rats in four groups showed no significant difference compared with each other after weeks 1, 2, 3 and 4. The body weight in the DIC-treated group showed no significant difference compared with the other three groups after weeks 1, 2, 3 and 4.
Enzymatic and nonenzymatic antioxidant activities
Effects of flavonoid extracts on tissue damages induced by DIC were evaluated as the activities of antioxidant enzymes such as SOD, GPx, CAT and GST and content of MDA in liver from control and treated rats.
SOD activity was found to decrease significantly in the liver of the DIC-treated group compared to the other three groups (p < 0.05). SOD activity in the DIC + flavonoid extracts group was increased by 36.8% (p < 0.05), with respect to the DIC-treated group. However, no significant change was observed between the flavonoid extracts group and the DIC + flavonoid extracts group. Meanwhile, the flavonoid extracts group restored the SOD activity to near normal (Figure 3).
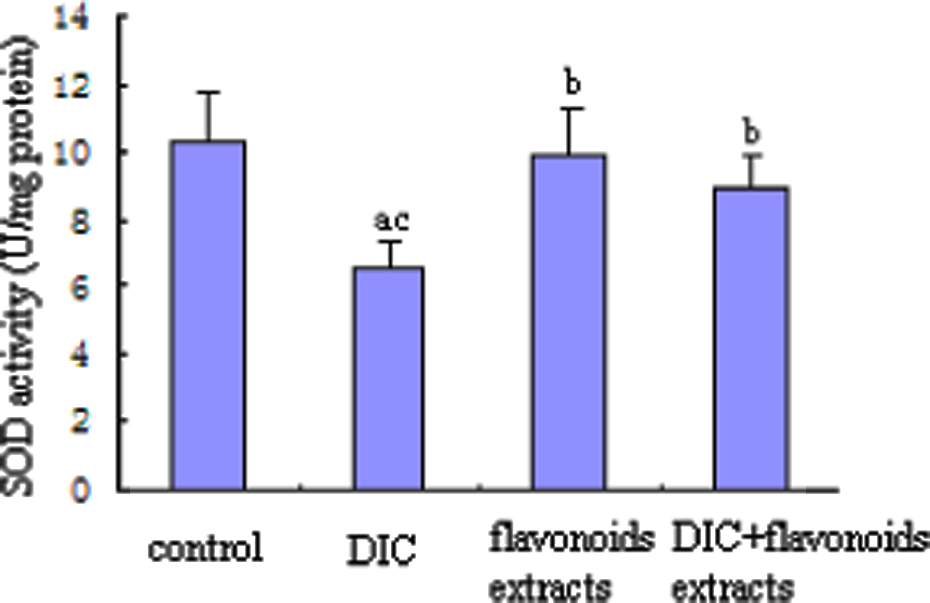
Effect of flavonoid extracts on the liver superoxide dismutase activity in dichlorvos (DIC)-treated rats. The values are expressed as mean ± SD (n = 8). Significantly different from (a) control, (b) DIC and (c) flavonoid extracts (p < 0.05).
GPx activity showed a significant increase in the liver of the DIC-treated group in comparison to those in the other three groups (p < 0.05). GPx activity in the DIC + flavonoid extracts group was decreased by 24.1% (p < 0.05), when compared to the DIC-treated group. No significant change was observed between the flavonoid extracts group and the DIC+ flavonoid extracts group (Figure 4).
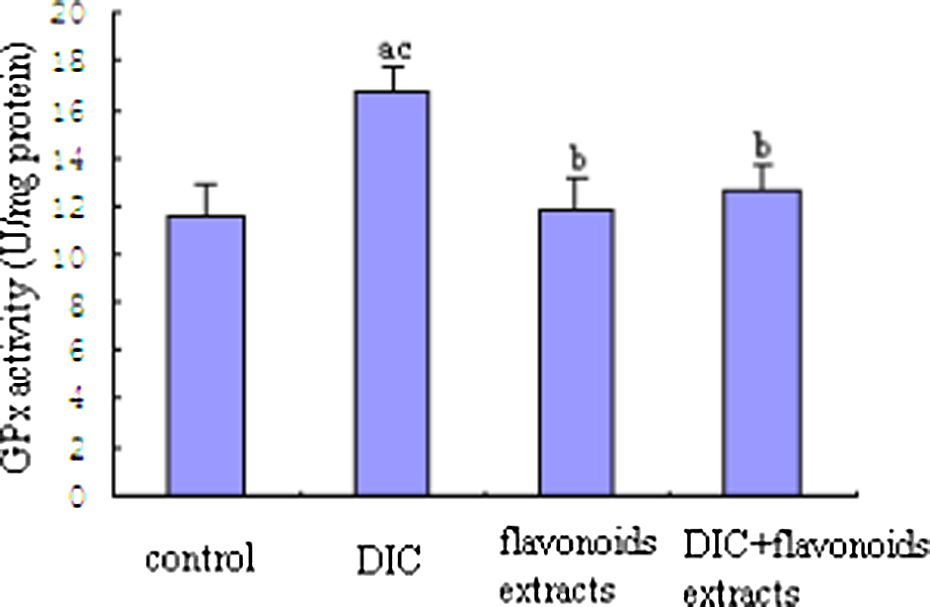
Effect of flavonoid extracts on the liver glutathione peroxidase activity in dichlorvos (DIC)-treated rats. The values are expressed as mean ± SD (n = 8). Significantly different from (a) control, (b) DIC and (c) flavonoid extracts (p < 0.05).
CAT activity was found to decrease significantly in the liver of the DIC-treated group compared to the other three groups (p < 0.05). CAT activity in the DIC + flavonoid extracts group was increased by 69.4% (p < 0.05), with respect to the DIC-treated group. Moreover, significant change was observed between the flavonoid extracts group and the DIC + flavonoid extracts group (p < 0.05) (Figure 5).

Effect of flavonoid extracts on the liver catalase activity in dichlorvos (DIC)-treated rats. The values are expressed as mean ± SD (n = 8). Significantly different from (a) control, (b) DIC and (c) flavonoid extracts (p < 0.05).
A significant decrease in the activity of GST was observed in the DIC-treated group when compared to the other three groups (p < 0.05). GST activity in the DIC + flavonoid extracts group was increased by 17.8% (p < 0.05), in comparison to the DIC-treated group. GST activity in the DIC + flavonoid extracts group showed no significant change in comparison to that in the flavonoid extracts group (Figure 6).

Effect of flavonoid extracts on the liver glutathione-S-transferase activity in dichlorvos (DIC)-treated rats. The values are expressed as mean ± SD (n = 8). Significantly different from (a) control, (b) DIC and (c) flavonoid extracts (p < 0.05).
Content of MDA in the DIC-treated group showed a significant increase in comparison to those in the other three groups (p < 0.05). Content of MDA in the DIC + flavonoid extracts group was decreased by 23.8% (p < 0.05), when compared to the DIC-treated group. No significant change in the content of MDA was observed in the DIC + flavonoid extracts group as compared to the flavonoid extracts group (Figure 7).

Effect of flavonoid extracts on the liver malondialdehyde content in dichlorvos (DIC)-treated rats. The values are expressed as mean ± SD (n = 8). Significantly different from (a) control, (b) DIC and (c) flavonoid extracts (p < 0.05).
Discussion
Organophosphorus pesticides are neurotoxic in nature by acting as inhibitor of neuronal cholinesterase activity. The typical clinical symptoms of acute organophosphate poisoning due to excessive cholinergic effects include bronchospasms, hypersecretion from cholinergic innervated glands along with cardiac disturbances caused by vagotonus and anoxia. There are numerous reported studies suggesting that organophosphorus pesticides could cause lipid peroxidation. 21 Oxidative stress, thus, was presumed to be one of the possible mechanisms involved in the excitotoxic injury. 22 Organophosphorus pesticides may induce oxidative stress, leading to the generation of free radicals and alterations in antioxidant or ROS scavenging enzymes. 23
The antioxidant capacity of flavonoids is determined by their structure. Studies have indicated that the aglycones, including quercetin, luteolin and kaempferol, have greater antioxidant capacity than do the conjugate flavonoids, such as quercetin-3-glucoside, quercitrin and rutin. 24 So we detected and reported flavonoids aglycones in celery following the hydrolysis procedure, and flavonoids extracts from celery were added to the experimental diets because of their greater antioxidant capacity.
SOD, CAT and GPx have been used as markers in detecting the level of oxidative stress in tissues and as major free radical scavengers that protect the biological system from the deleterious effects of harmful toxicants like the pesticides. 25,26 They serve as the primary antioxidant defensive mechanism in the body. Their concentrations are altered rapidly in the body during xenobiotic insults, resulting in their increase or decrease in the body tissues or blood. Lipid peroxidation has also been suggested as one of the major molecular mechanisms in case of pesticide induction and serves as a potent marker of oxidative stress induced by pesticide toxicity. 26 Liver is the main organ where most of the metabolisms take place and also the detoxification of various toxic materials or their metabolites. Conjugation of enzymatic moieties to harmful molecules is a well-known pathway of removal of toxic materials from the system. In this study, we chose SOD, CAT, GPx, GST and MDA as important biomarkers to investigate the protective role of the antioxidative components present in celery extracts against the adverse effects of DIC in vivo, based on lipid peroxidation and antioxidant enzyme profiles in rats.
In the present study, DIC exposure (7.2 mg/kg·bw/day) for 4 weeks showed no obvious detrimental effect on the growth curve of animals, as showed in Figure 2. No significant loss in weight gain may be attributed to the low level of DIC administration in rats and the short-term exposure to DIC. This finding contradicted those reported by Geetu et al. 27 who recorded weight loss following 8 weeks of subchronic DIC exposure (6 mg/kg.bw/day) in rats, which is attributed to impairment of cellular metabolism by the organophosphate compound.
SOD plays an important role in the dismutation of superoxide radicals to form hydrogen peroxide and molecular oxygen and acts as the first line of defense. 28 CAT in turn converts the peroxide into molecular oxygen and water, thus nullifying the pro-oxidative effect of the free radicals. 29 However, the presence of superoxide radicals inhibits the activity of CAT. Singlet oxygen and peroxyl radicals also affect SOD and CAT activities. 30 In some studies 31 , SOD and CAT activities were found to be increased; and in other studies, 32 the activities of these enzymes were found to be decreased. In addition, previous studies have been reported that the modulation of SOD activity by pesticides is variable with low doses of some pesticides not altering, but higher doses increasing the activity in the liver and other tissues. 2,4,33 In the present study, on one hand, the free radical production as an outcome of oxidative stress caused by DIC have reduced the activities of SOD and CAT. On the other hand, a dose of flavonoid extracts to rats have restored their activities, which may be a result of scavenging intracellular free radical oxygen accumulation. These results revealed that flavonoids could prevent oxidative stress induced by DIC through an increase in SOD and CAT.
GPx is an antioxidant seleno-enzyme (found in most tissues) that reduces hydrogen peroxide and alkyl peroxide in tissues and uses glutathione as an essential redox substrate. Previous studies have shown that GPx activity was influenced by pesticide exposure. 34,35 In this study, we found that DIC increased the liver GPx activity in rats, as many researches have reported that low-level organophosphates, including chlorpyrifos-ethyl, appear to increase the activity of GPx in the liver after exposure. 2,36,37 Meanwhile, in our study, flavonoid extracts from celery reduced the elevated GPx activity induced by DIC exposure, due to the greater utilization of glutathione for detoxification of electrophiles and free radicals produced by the flavonoids and their metabolites.
GST is a key enzyme in glutathione-binding reaction. The major function of GST is to catalyze the harmful endogenous substances or foreign electrophilic group and couple with thiol of reduced glutathione, so as to achieve detoxification. Glutathione, a thiol tripeptide, is the most important endogenous antioxidant and is responsible for maintaining the cellular redox state along with smaller thiols such as thioredoxin, glutaredoxin and peroxiredoxin. 38 The major function of glutathione is to scavenge intracellular free radicals and peroxides under normal conditions. The results in the present study also showed that GST activity in DIC-treated rats was decreased because of DIC exposure. Moreover, flavonoid extracts reduced the effect of DIC on liver’s GST activity. These results revealed that DIC-induced oxidative damage can be prevented by flavonoids. However, according to Moskaug et al., 38 in vivo feeding experiments with flavonoid-rich diets do not uniformly induce the enzyme.
In this study, the peroxidation levels in the liver were significantly increased as detected by the high levels of MDA with DIC exposure. Moreover, in our study, low levels of lipid peroxidation in rats treated with the flavonoid extracts and the DIC + flavonoid extracts confirm that damages related to free radical attacks and oxidative stress in systems induced by DIC can be prevented by diet flavonoids.
Taken together, the results of the present study suggest that exposure to DIC could induce oxidative stress. However, dietary supplementation with flavonoid extracts (quercetin, luteolin, kaempferol and apigenin) could improve the harmful effects of DIC by inhibiting the production of free radicals and by altering the enzyme and nonenzyme levels in their bodies to a good extent. These results indicate that flavonoids like apigenin, quercetin, etc. could prevent oxidative stress induced by DIC through a reduction in GPx and MDA and an increase in SOD, CAT and GST. In relation to public health, when there is a possibility of pesticide contamination through our diet, the inclusion of such flavonoids in the diet is strongly suggested.
Footnotes
Financial support from the National Natural Science Foundation of China (81172672) is gratefully acknowledged.
The authors declared no conflicts of interest.
Jie Cao and Xuefei Zhang contributed equally to this work and should be considered as co-first authors.
