Abstract
Keywords
Case
A 33-year-old male presented to the emergency department (ED) with a variety of symptoms including polydipsia, polyuria, nausea, bouts of blurry vision, headaches increasing in pain and frequency and malaise. During the initial triage interview the patient spoke clearly and understood and verbalized appropriate answers to questions. He did not complain of immediate pain or headache and his gait and affect were normal. A rapid finger stick blood glucose measurement returned a ‘critical high’ reading (BG >500 mg/dl). Blood was drawn and sent to the lab, and he was immediately escorted back to the ED treatment area. Initial blood glucose was 1082 mg/dl (60.1 mmol/L). Additional blood was drawn and revealed an arterial blood pH of 7.37 with corresponding blood glucose of 1166 mg/dl (64.8 mmol/L). Despite an impressively high blood glucose level, the patient was fully coherent, alert and oriented. He was noted to be extremely hyperglycemic and dehydrated (see Table 1).
Patient information

The patient explained that he had become extremely thirsty over the past few weeks coupled with an increased urinary output. Four days prior to his presentation he felt increasingly nauseous, feverish and had increasing bouts of blurry vision. Upon hearing the concern surrounding his high blood glucose, he admitted to recently completing a course of anabolic steroids, which included injections of a bovine growth hormone and testosterone, for recreational bodybuilding (see Table 2). His self-prescribed regimen began approximately 3.5 months prior, with discontinuation of injections 3 weeks before presentation. The patient was worried about his current symptoms and appeared honest about his recreational self-medication. He denied use of alcohol and tobacco. Recreational drug abuse was ruled out by a rapid urine toxicology screen using a Bio-Rad TOX/see® rapid urine test kit.
Self-prescribed anabolic steroid regimen

a Sustanon 250 contains: testosterone propionate, 30 mg; testosterone phenylpropionate, 60 mg; testosterone isocaproate, 60 mg; and testosterone decanoate, 100 mg, diluted in benzyl alcohol 10% (v/v).
b The trenbolone acetate implants were crushed, dissolved in an oil base, and ‘filtered’ for injection. Individual and cumulative dose unknown. Unknown if Finaplix-H or -S was used. Finaplix-H contains 200 mg, Finaplix-S 140 mg.
This patient had no family history of diabetes and had a non-contributory past medical history. Despite the patient’s lucid behavior at presentation, his blood glucose increased from 1082 (60.1 mmol/L) to 1166 mg/dl (64.8 mmol/L) and serum osmolality increased from 302 to 307 mOsm/kg during the short interval while bedded in the ED. The patient’s serum also tested positive for acetone. The patient’s complaints combined with these laboratory values indicated a deteriorating condition, and changes in mental status were likely to soon follow.
Initial treatment consisted of intravenous fluids, a bolus of 10 units of regular insulin, and an insulin drip managed per local protocol. Serum potassium was supplemented on an as-needed basis during the insulin therapy. Blood glucose slowly decreased to near-nominal levels over 6 hours with provided therapy (see Figure 1). The patient stabilized over the first 24 hours and was diagnosed with new onset diabetes. He was discharged after 2 days in the hospital and was provided education on how to manage his blood glucose. He was also instructed to follow up with his primary health care provider and was discouraged from using further anabolic steroids, growth hormones or other illicit and non-prescribed medications.
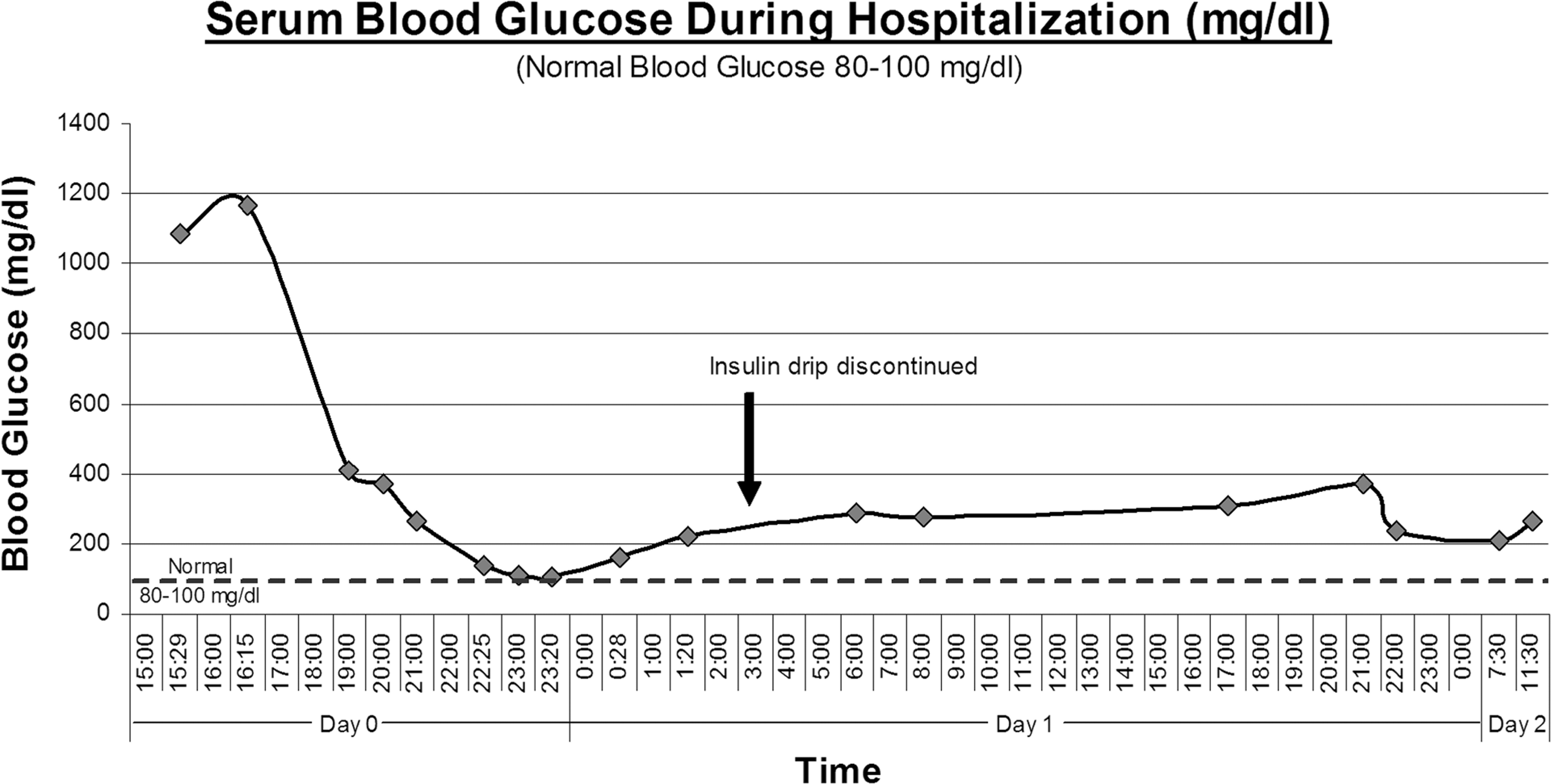
Serum blood glucose.
Discussion
Anabolic androgenic steroids (AASs) promote muscle growth by stimulating cell proliferation and increasing the metabolic processes involving protein synthesis through several mechanisms.1–4 Discussions regarding growth hormone use in athletes state possible increases in skeletal muscle mass and strength with an associated reduction in body fat. It is believed that athletes use supratherapeutic doses of growth hormones and other anabolic agents, possibly up to 10-fold of normal, to achieve desired increases in performance.5,6 These performance enhancing agents are unregulated, lack clinical and safety data, and users select various drugs and dosing regimens based on recommendations provided by underground literature, training colleagues and availability of drug.
Discussions with regards to the adverse effects of AASs cover a wide range of systemic effects and end-organ damage ranging from dermatologic maladies to sudden cardiac death. It has been demonstrated that AAS use can affect glucose homeostasis through increasing cellular insulin resistance and reducing glucose uptake.5–9 Drug-induced cases of diabetes and hyperosmolar hyperglycemic syndrome (HHS) have been reported in the medical literature due to prescription medications such as the growth hormone somatostatin and glucocorticoids.10–12 There is little clinical or safety data available on the effects of the bovine growth hormone, trenbolone acetate, in humans aside from anecdotal data and Internet information sharing.
Trenbolone acetate is a veterinary product and licensed as a growth promoter for farm animals in the United States and several other meat-exporting countries. It is produced as a solid dosage form intended for use as a sub-dermal ear implant in approved feed animals. Trenbolone acetate is easy to procure and is sold as an over-the-counter product in animal feed stores under the brand name Finaplix®. Finaplix-H® used in heifers contains 200 mg of trenbolone acetate per implant, Finaplix-S® for steers contains 140 mg/impant. 13 Significant increases in animal muscle meat have been associated with the use of trenbolone implants; however due to safety concerns, trenbolone is banned in feed animals in some European countries.14,15 Crushing implants is discouraged and has been noted to increase multiple adverse effects in cattle. 16 Trenbolone acetate is not indicated for use in humans. Despite this fact, information on trenbolone acetate ‘for injection’ can be found on various bodybuilder and steroid Internet websites. There are various crude ‘recipes' and ‘kits' which instruct users on methods to convert the trenbolone pellets into an injectable liquid form at home. There are also many underground laboratories that produce and sell injectable versions of this drug for athletes.
The side effects of prescribed, therapeutic human growth hormone use are well documented. In patients who developed acromegaly, from excessive growth hormone, up to 40% are diagnosed with diabetes due to induced insulin resistance. 6 A study by Bramnert et al. demonstrated that low dose growth hormone administered to growth hormone deficient adults showed a trend toward insulin resistance and glucose intolerance at 1 week and 6 months. 17 Moller and Jorgensen postulate that excess growth hormone secretion in previously diagnosed diabetic patients may play a role in the infamous ‘dawn phenomenon,’ increasing insulin requirements in the early morning hours. 18 Use of growth hormone at therapeutic and supratherapeutic doses has been reported to induce glucose intolerance and increased insulin resistance; concurrent use of other anabolic agents may increase these undesired effects.5,19,20 A controlled study by Cohen and Hickman found that the degree of glucose intolerance in steroid-using subjects was higher than the non-users. Cohen and Hickman also postulated that there may be a subset of the population that may have a predisposition for developing diabetes after AAS use. 7 The use of growth hormone was not mentioned in their study population.
A case report by Young et al., describes diabetes induced by AAS, including growth hormone, in an athlete. Young’s patient presented to the ED with a blood glucose of 543.6 mg/dl (30.2 mmol/L) and abdominal pain and was diagnosed with diabetes due to growth hormone misuse. He had used anabolic steroids for years and stopped injecting insulin after a series of hypoglycemic episodes. After halting insulin use, the patient developed and was diagnosed with diabetes. With treatment, the diabetes appeared to resolve within 6 weeks. 8 Insulin is a widely used/abused medication by AAS users because of the known hyperglycemic effects of steroids. Insulin is also considered an anabolic hormone as it increases glucose uptake and increases cellular metabolism assisting in muscle growth.21,22 In contrast, the current patient did not use insulin to augment his AAS regimen and his AAS use was more impulsive versus continuous. He used a combination of an unknown amount of unregulated trenbolone acetate and testosterone products. The patient in Young’s case likely used human growth hormone, as the injected amounts were measured in international units (IUs); no discussion documents the product in their report. The products the current patient used were not available for inspection or purity testing.
It is worthwhile to mention other likely AAS-induced side effects that were noted during this case. This patient’s triglycerides were high with a low measured HDL (see Table 1). Multiple reports describe anomalies in lipid profiles combined with coronary artery narrowing in AAS users. The patient’s hemoglobin A(1c) suggests that blood glucose was uncontrolled during the previous 90 days, which is coincidental to the initiation of his AAS regimen. Increases in human HA(1c) levels have not previously been correlated with veterinary growth hormone use. As the maximal rate of change of HA(1c) may vary and there is no baseline measurement of the patient’s HA(1c), the question remains if the AAS use is fully responsible for the new onset diabetes or did the drugs exacerbate an underlying condition.
Confounding issues that arise with this case include the unknown amount of trenbolone acetate the patient was injecting. He received the vial from a friend who had produced the injectable form by crushing and dissolving solid trenbolone acetate implants. The process used, type of implants and amount of implants dissolved into solution are all unknown. Injectable testosterone, (Sustanon 250®), was delivered from an overseas company through a mail order Internet website. There is potential for these injectable products, procured from questionable sources, to contain contaminants, other drugs or toxins, which could be an unknown cause of the presenting symptoms. However, as it is known that growth hormone and somatostatin have both caused diabetes, the potential for an ultra concentrated dose of trenbolone acetate, intended for use in cattle, may alone be the culprit in this case of new onset diabetes.5,6,8,12
It is difficult to imply a direct causal relationship between the combination growth hormone/testosterone and diabetes. However the correlation of timing and the known risks associated with AAS abuse, specifically growth hormone, make this association worthy of scrutiny. The paucity of information on AAS-induced diabetes may be due to factors such as under reporting or denial of steroid use when patients present to health care providers for diabetic symptoms. Many AAS users self-manage adverse effects avoiding their health care providers all together. To our knowledge this is the first known report with regards to human abuse of trenbolone acetate, a ‘commonly’ used agent among body builders. Further surveillance and research are warranted on the correlation of diabetes, HA(1c), and trenbolone acetate abuse in humans.
Follow up
Follow up with the patient, 3 months after his ED visit, showed persistence of the diabetic condition with an improved control of his blood glucose. His daily average blood glucose was controlled between 100 and 110 mg/dl and a recent HA(1c) measured 7.2%. Three months after discharge, the patient no longer required multiple daily, sliding scale insulin injections. His only requirement included a daily subcutaneous dose of 35 units of long-acting glargine insulin (Lantus®) with no other insulins or oral medications. He stated that he still worked out but denied further use of AAS or bovine growth hormone.
Conclusion
We report a case of a young bodybuilder with no significant past medical history who was diagnosed with new onset diabetes associated with supratherapeutic injections of the bovine growth hormone, trenbolone acetate and testosterone. AAS abuse has the potential to induce or exacerbate diabetic conditions due to decreased glucose tolerance and increased insulin resistance. The effects and side effects of athletic misadventures with AAS may never be fully elucidated; however, one would expect that athletes abusing growth hormone, especially a growth hormone designed for cattle, may express some features of acromegaly and develop diabetes.
Footnotes
All authors contributed significantly to this report and its revision. This information has not been presented prior to this submission.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
None of the authors report any conflict of interest.
