Abstract
Background: Nonsteroidal anti-inflammatory drugs may cause severe injury to all parts of the gastrointestinal tract. It has been hypothesised that probiotic bacteria might reduce this adverse effect. The aim of this study was to perform a morphometric evaluation of the gastrointestinal tract in experimental pigs treated using a 10-day high-dose of indomethacin with or without Escherichia coli Nissle 1917 (EcN). Methods: Twenty-four healthy mature pigs were included: Group A (controls; 6 animals), Group B (EcN; n = 6), Group C (indomethacin; n = 6) and Group D (EcN & indomethacin; n = 6). EcN (3.5 × 1010 live bacteria/day for 14 days) and/or indomethacin (15 mg/kg/day for 10 days) were administered. Specimens of the stomach, small and large bowel were routinely processed for microscopic examination. The height of glandular mucosa, height and width of interfoveolar spaces and villi and basement size of epithelial cells were evaluated. Results: Different effects of indomethacin and EcN on particular parts of the gastrointestinal tract were shown. The indomethacin and probiotics group demonstrated a significantly lower height of cryptal mucosa and colonocytes and widening of the basement size of colonocytes compared to controls (p = 0.004; p < 0.001; p = 0.025). The height of cryptal mucosa was significantly higher in the EcN group compared to controls (p = 0.001). Conclusions: Indomethacin alone induced marked adaptation of the gastric mucosa. EcN alone provided a significant favourable trophic effect on the colonic mucosa. However, indomethacin and probiotics administered together comprise the worst impact on all porcine stomach, small and large bowel.
Introduction
Nonsteroidal anti-inflammatory drugs (NSAIDs) represent the group of most commonly used drugs worldwide. NSAIDs may cause severe injury to all parts of the gastrointestinal tract, both in humans and in an experimental setting.1–5 Gastric damage by NSAIDs is primarily a consequence of inhibition of cyclooxygenase-1 (COX-1) in the upper GI tract. COX-1 inhibition reduces mucosal generation of protective prostaglandins such as prostaglandin E2. While inhibition of COX-1 is the major mechanism by which NSAIDs produce gastric injury, mediators besides prostaglandins and nitric oxide may also be involved. 2 The pathogenesis of NSAID-induced entero- and colopathy is more multifactorial and complex than formerly assumed but has still not been fully understood. A combination of local and systemic effects plays an important role in pathogenesis. NSAID-induced entero- and colopathy is a stepwise process involving direct mucosal toxicity, mitochondrial damage, breakdown of intercellular integrity, enterohepatic recirculation and neutrophil activation by luminal contents including bacteria. 5 Aspirin and some NSAIDs, including indomethacin, influence intestinal bacteria.6–10 Metronidazol can reduce indomethacin-induced intestinal toxicity. 11
Probiotic bacteria are live microorganisms that when administered in adequate amounts confer a health benefit on the host. 12 Probiotics likely function through enhancement of the barrier function of the gut, immunomodulation and competitive adherence to the mucus and epithelium. 13 Furthermore, probiotic bacteria might exert a systemic anti-inflammatory effect 14 and modulate apoptosis. 15 Probiotics have been suggested for amelioration or prevention of various diseases including antibiotic-associated diarrhoea, irritable bowel syndrome and inflammatory bowel disease. 16
It has been hypothesised that probiotic bacteria might reduce the adverse effects of NSAIDs on the gastrointestinal tract. However, initial studies provided controversial results, both with ameliorating and deteriorating outcomes.17–20
The aim of this project was to perform a morphometric study of the gastrointestinal tract in experimental pigs treated using short-term high-dose indomethacin with or without probiotic bacteria Escherichia coli Nissle 1917 (EcN).
Methods
Animals
Twenty-four healthy young mature (4–5 months old) female pigs (Sus scrofa f. domestica, hybrids of Czech White and Landrace breeds) weighing 33.0 ± 1.7 kg, were included in our study. The animals were divided into four groups: Group A (controls; 6 animals), Group B (probiotics alone; n = 6), Group C (indomethacin alone; n = 6) and Group D (indomethacin & probiotics; n = 6). All animals were fed twice a day (standard assorted A1 food of equal amounts).
Drug and probiotic bacteria administration
EcN (3.5 × 1010 live bacteria/day for 14 days) and/or indomethacin (Indomethacin suppositories, Berlin-Chemie, Germany; 15 mg/kg/day for 10 days) were administrated as one-shot dietary bolus to hungry pigs. EcN was provided by the Institute of Microbiology, Academy of Sciences of the Czech Republic.
Autopsy
Twenty-four hours after the last drug and/or probiotic bacteria administration (Groups B to D) or after 14 days of stabling (Group A), the pigs were killed (after 24 hours of fasting) by means of pharmacological euthanasia (i.v. administration of embutramide, mebezonium iodide and tetracaine hydrochloride—T61, Intervet International BV, Boxmeer, the Netherlands; dose of 2 mL per kg) and exsanguinated. Immediate autopsy was performed and specimens for structural and morphometric analysis were taken.
Histological examination
Only samples of macroscopically normal appearance were extirpated. Specimens of the stomach, jejunum, ileum and colon were routinely processed for microscopical examination. The samples were fixed with 10% neutral buffered formalin for 24 hours and subsequently embedded into paraffin. Five-micrometers-thick tissue sections of each part were cut and stained with hematoxylin eosin for morphometric evaluation and Gram staining to exclude bacterial invasion.
Measurement of morphometric parameters
Stained samples were evaluated using a BX-51 microscope (Olympus Optical Co, Tokyo, Japan) and computer image analysis ImagePro 5.1 (Media Cybernetics, Bethesda, MD, USA). Ten microscopic fields at a 100-, 200- and 800-fold original magnification were taken from each pig. The following parameters were evaluated:
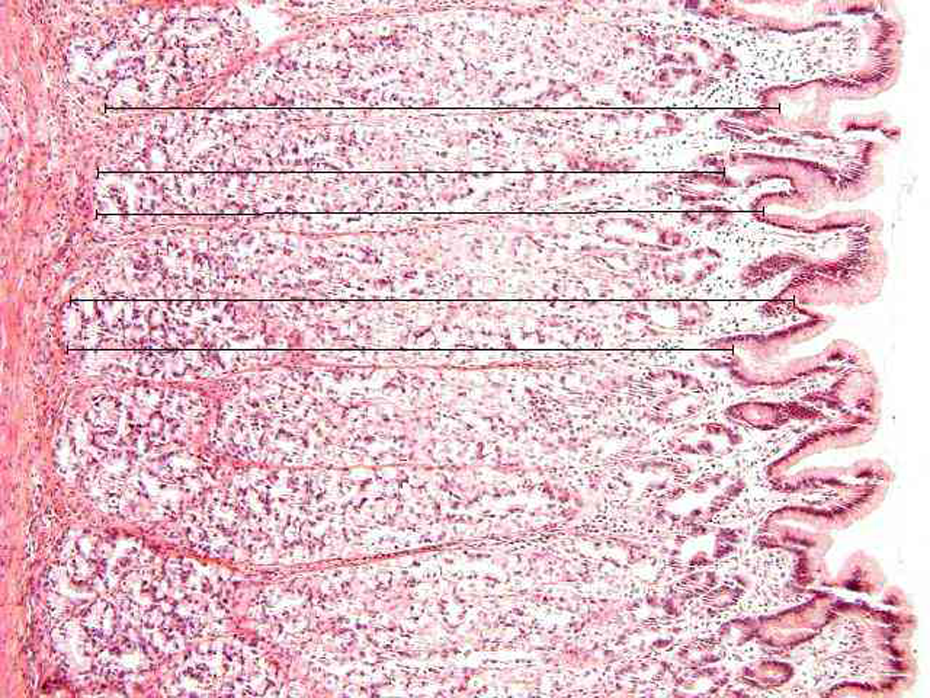
In the stomach, the height of glandular mucosa at 100-fold original magnification (see Figure 1), height and width of interfoveolar spaces at 200-fold original magnification (Figure 2) and length of the basement membrane of 10 epithelial cells together with height of epithelial cells localised at the base of the interfoveolar space were assessed at 800-fold original magnification (Figure 3). In the jejunum and ileum, the height of cryptal mucosa at 100-fold original magnification (Figure 4), height and width of villi at 200-fold original magnification (Figure 5) and length of the lamina basalis of 10 epithelial cells together with height of enterocytes localised at the base of villi were measured at 800-fold original magnification (Figure 6). In the colon, the height of cryptal mucosa was evaluated at 100-fold original magnification (Figure 7). The length of the basement membrane of 10 epithelial cells together with height of colonocytes localised at the lumen space were measured at 800-fold original magnification (Figure 8).
A sample of the porcine stomach at 100-fold original magnification. The distance between foveolar bottom and the bottom of crypts leading vertically towards the muscularis mucosae was measured for evaluation of the height of glandular mucosa in the stomach. Five measurements per microscopic field were performed. Hematoxylin-eosin staining.

The porcine stomach at 200-fold original magnification. The width of the interfoveolar space at its base and height of the space represented by the axis line between the centre of the width line and the top of the interfoveolar space were measured. Two measurements per microscopic field were performed. Hematoxylin-eosin staining.

The length of the basement membrane of 10 epithelial cells localised at the base of the interfoveolar space and the height of the 10 cells were evaluated in the porcine stomach at 800-fold original magnification. Hematoxylin-eosin staining.

A sample of the control porcine ileum at 100-fold original magnification. For evaluation of the height of cryptal mucosa in the ileum and jejunum, only microphotographs of longitudinal crypt sections were taken. The height of cryptal mucosa was determined as the distance between the villus base and the end of Lieberkühn crypts leading vertically from the villus base towards the muscularis mucosae. Five measurements per microscopic field were performed. Hematoxylin-eosin staining.

The porcine ileum at 200-fold original magnification. The width of a villus was measured at its base, while its length was represented by the axis distance between the centre of the width line and the top of the villus. Two measurements per microscopic field were performed. Hematoxylin-eosin staining.
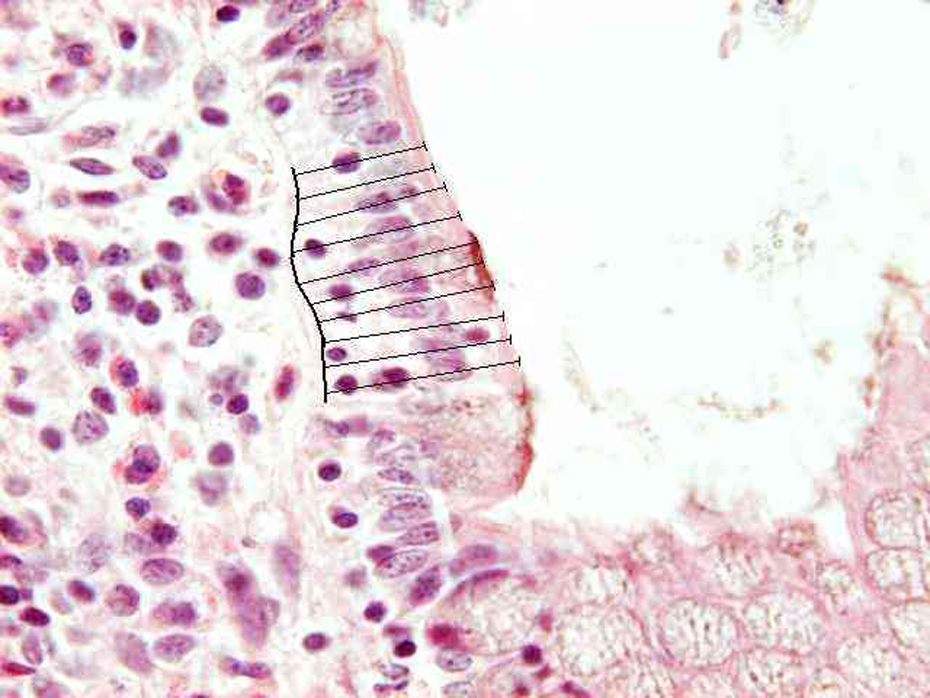
A sample of the porcine ileum at 800-fold original magnification. In the ileum and jejunum, the length of the basement membrane of 10 epithelial cells localised the base of villi and the heights of the 10 enterocytes were measured. Hematoxylin-eosin staining.

The porcine transverse colon at 100-fold original magnification. Microphotographs of longitudinal crypt sections were taken for evaluation of the height of cryptal mucosa in the colon transversum. The height of cryptal mucosa was determined as the distance between the bottom of Lieberkühn crypts and surface leading vertically from the crypts towards the surface. Five measurements per microscopic slide were performed. Hematoxylin-eosin staining.

A sample of the porcine transverse colon at 800-fold original magnification. The length of the basement membrane of 10 epithelial cells localised at the intestine lumen and the heights of the 10 colonocytes were measured. Hematoxylin-eosin staining.
Ethics
The project was approved by the Institutional Review Board of Animal Care Committee of the Institute of Experimental Biopharmaceutics, Academy of Sciences of the Czech Republic, Record Number 1492006. Animals were held and treated in accordance with the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes. 21
Statistical analysis
The results are expressed as mean ± SEM (standard error of the mean). Data were statistically analysed using a Mann-Whitney rank sum test. Statistical software was used for this analysis (SigmaStat version 3.1; Jandel Corporation, Erkrath, Germany). Statistical significance was set at p < 0.05.
Results
Altogether, 18,240 morphometric measurements were accomplished. There was no bacterial contamination in any specimen.
Morphometric analyses of the porcine stomach showed significant difference in both height of epithelial cells and their basement size (expressed as length of the lamina basalis of 10 epithelial cells) between controls and the indomethacin group (p < 0.001). Values of basal size of epithelial cells were significantly higher in the indomethacin and probiotics group compared to the controls (p < 0.001). See Table 1 for details.
Morphometric analysis of the porcine stomach

SEM: standard error of the mean.
Notes: Group A (controls with no treatment; 6 animals); Group B (probiotics alone; n = 6); Group C (indomethacin alone; n = 6); Group D (indomethacin and probiotics; n = 6).
In the jejunum, EcN administration was associated with significantly lower values of height of cryptal mucosa (p < 0.001) and height and width of villi (p = 0.005; p = 0.023) compared to the controls (Table 2).
Morphometric analysis of the porcine jejunum
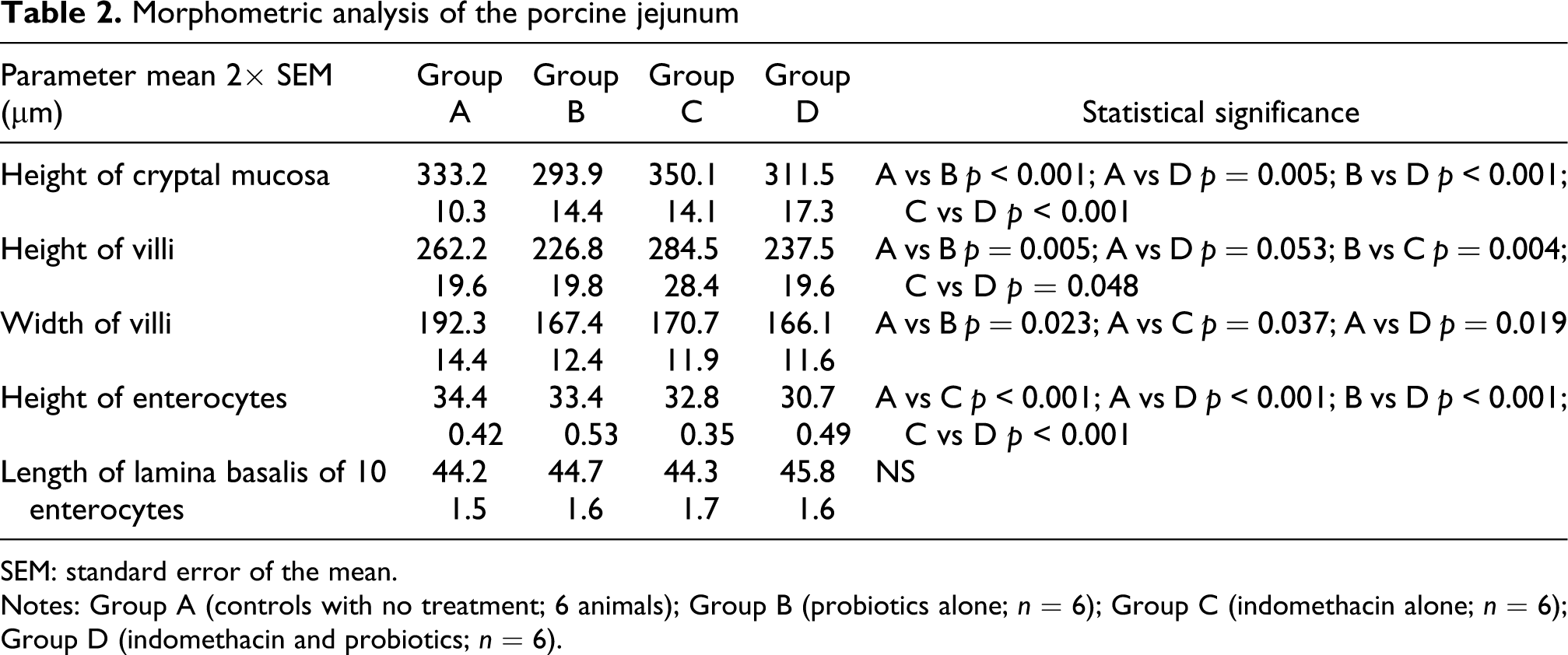
SEM: standard error of the mean.
Notes: Group A (controls with no treatment; 6 animals); Group B (probiotics alone; n = 6); Group C (indomethacin alone; n = 6); Group D (indomethacin and probiotics; n = 6).
Morphometric analyses of the ileum revealed significantly decreased height of enterocytes and widening of their basement size (assessed by length of the basement membrane of 10 epithelial cells) in the indomethacin group and indomethacin and probiotics group compared to controls (p < 0.001). The height of cryptal mucosa was significantly higher in the indomethacin group and indomethacin and probiotics group compared to the controls (p = 0.003; p = 0.044; Table 3).
Morphometric analysis of the porcine ileum

SEM: standard error of the mean.
Notes: Group A (controls with no treatment; 6 animals); Group B (probiotics alone; n = 6); Group C (indomethacin alone; n = 6); Group D (indomethacin and probiotics; n = 6).
In the porcine colon, the indomethacin and probiotics group demonstrated significantly lower height of cryptal mucosa and colonocytes and widening of the basement size colonocytes compared to the controls (p = 0.004; p < 0.001; p = 0.025). The height of cryptal mucosa was significantly higher in the EcN group compared to the controls (p = 0.001; Table 4).
Morphometric analysis of the porcine colon

SEM: standard error of the mean.
Notes: Group A (controls with no treatment; 6 animals); Group B (probiotics alone; n = 6); Group C (indomethacin alone; n = 6); Group D (indomethacin and probiotics; n = 6).
Discussion
The primary objective of this project was to evaluate the effect of indomethacin on morphological changes of the gastrointestinal tract in experimental pigs. We decided to use a short-term high-dose of indomethacin (twice as the highest dose for humans) with or without EcN. Our hypothesis for this study was that probiotic bacteria EcN would protect the porcine stomach and intestine from any harmful effects of indomethacin. Considerably consistent data were achieved in particular groups of animals. Nevertheless, our results must be interpreted with caution. The height of the glandular part in the gastric mucosa was lower in the indomethacin group, which might be interpreted as prompt adaptation changes. The significantly enlarged height of the glandular part and widening of epithelial cells at the basement membrane of the porcine stomach (Group D) is at least partly explained by mucosal oedema and might by assessed as a deteriorating conjunctive effect of indomethacin and EcN administered together. The decreased size of villi in the small intestine in the EcN group might be interpreted as an unfavourable effect of probiotics on the porcine small bowel.
We considered the length of the basement membrane of 10 epithelial cells as the most relevant and reliable marker. Epithelial cells at the lamina basalis decreased, shrank and widened, both in the small and large intestine in the indomethacin and EcN group. These data can be interpreted as a deteriorating impact of combined indomethacin and probiotic bacteria on the porcine intestine. These findings are consistent with the results of our previous study on colicins and microcins in experimental pigs. Indomethacin and EcN administered together comprised the worst impact on bacteriocinogeny in the porcine gastrointestinal tract (compared to indomethacin alone or probiotics alone). 22 EcN alone significantly broadened the width of the glandular part of colonic mucosa. We assess this finding as a positive trophic effect of probiotic bacteria on the porcine large bowel.
We are fully aware of possible limits of our study. Mild edema of the mucosa is difficult to assess accurately. This might influence particular measurements and may make the interpretation of acquired data difficult. We did not evaluate apoptosis. In general, its rate is low in the gastrointestinal tract but might be induced by NSAIDs 23 and modulated by probiotic bacteria. 15 Further markers of mucosal damage/proliferation/apoptosis (proliferating cell nuclear antigen, Ki67, inducible NO synthase, caspase and others) and/or effect on mucins might be considered and addressed in future studies. And last but not least, we did not focus on possible functional changes of the gastrointestinal tract due to action of indomethacin and/or probiotic bacteria.
There are only few data on morphometric analysis in the available literature.24–27 A previously published paper focused on indomethacin-induced injury but not on the synchronous effect of NSAIDs and probiotics. Ettarh et al.24,26 performed morphometric analyses in the indomethacin-treated mouse. They found a significant decrease in villous height, 24 decreased epithelial volume, crypt losses and increased mitotic activity in the non-ulcerated parts of the murine small intestine. 26 Driak et al.28,29 were probably the first to quantify morphometric changes of the rat gastrointestinal tract (after gamma-irradiation). We partly adopted their methods for our experimental setting.
In conclusion, detailed morphometric analysis showed a different effect of indomethacin and probiotic bacteria on particular parts of the porcine gastrointestinal tract. Indomethacin alone induced marked adaptation changes in the gastric mucosa. EcN alone provided a favourable trophic effect on the colonic mucosa. However, indomethacin and probiotics administered together might comprise the worst impact on all porcine stomach, small and large bowel. Further studies are needed to clarify this phenomenon. This should be helpful in the context of possible interaction of probiotics or EcN and intestinal transport of model drugs, xenobiotics and/or function tests.
Footnotes
Acknowledgement
The authors would like to thank Professor Helena Tlaskalova-Hogenova, MD, DSc (Institute of Microbiology, Academy of Sciences of the Czech Republic) for her kind provision of Escherichia coli Nissle 1917 for all experiments.
The study was supported by research project MZO 00179906 from the Ministry of Health of the Czech Republic and by research grant GAČR 305/080535, Czech Republic.
The authors disclose no conflicts of interest.
