Abstract
Electron microscopy was used to examine changes in the subcellular organelles of the rat kidney at different time intervals after a single exposure to cyclophosphamide (CP). The morphological changes were studied at different time points (6 hrs, 16 hrs and 24 hrs) after a single-dose administration of CP. Six rats were killed at each time intervals after the administration of CP. Saline-treated rats served as controls. CP administration resulted in alterations in various subcellular organelles including peroxisomes, lysosomes, mitochondria, and the endoplasmic reticulum (ER) of the renal tubular epithelium as well as damage to the glomerulus. The basement membrane of the glomerulus was thickened. Many podocytes were destroyed. The nucleoplasm of the endothelial cell showed fewer granularities. The tubules were distorted and the brush border was destroyed. Two striking features in the renal tubular cells are increase in number and size of the peroxisomes (peroxisome proliferation) and decrease in the number of lysosomes. The mitochondria were elongated and the number was increased in the tubules of CP-treated rats. The ER was dilated. Cell necrosis was also seen. This study is an evidence of changes in morphology of rat kidney after induction of renal damage by a single dose of CP. Since transmission electron microscopy is the highest magnification tool at present, it can be useful in estimating the degree of injury and outcome of alternative treatment strategies in the management of CP-induced renal damage after establishing a scoring system.
Introduction
Cyclophosphamide (CP) is a drug with a wide spectrum of clinical uses and it has been proved to be effective in the treatment of cancer and non-malignant disease states such as rheumatoid arthritis. CP is widely used in the treatment of solid tumors and B cell malignant disease, such as lymphoma, 1 myeloma, 2 chronic lymphocytic leukemia, 3 acute myeloid leukemia, 4 breast cancer, 5 cervical cancer, 6 lung cancer, 7 ovarian cancer, 8 endometrial cancer, 9 and Waldenstrom’s macroglobulinemia. 10
However, this drug may induce acute inflammation of the urinary bladder (cystitis), renal damage, and liver damage, thereby limiting the therapeutic use of the drug. Besides, hemorrhagic cystitis (HC), kidney damage is a major dose-limiting side effect of CP. Studies have shown that CP can be nephrotoxic, both in humans and animal models. CP can result in glomerular dysfunction and tubular dysfunction.11–17 The mechanism of urotoxicity of CP has been well-studied. The mechanism of renal toxicity of CP is not clear although oxidative stress and nitrosative stress have been reported.18–20
In the present study, the effects of CP on subcellular organelles of the renal tubules and consequence of these effects are discussed.
Materials and methods
Chemicals
CP (endoxan) was purchased from Zydus Biogen, Ahmedabad, India. Toluidine blue, uranyl acetate, propylene oxide, sodium citrate, and lead nitrate and all other chemicals and reagents used were of analytical grade and were purchased from Sisco Research Laboratories (SRL) Pvt. Ltd., Mumbai, India.
Animals and treatments
Adult male Wistar rats (200–250 g) were used for the experiments. The study was approved by the animal ethics Committee for the Purpose of Control and Supervision of Experimentation on Animals (CPCSEA), Government of India. The guidelines were followed. The rats were maintained in a controlled environment at 20°C and 40%–50% humidity, with 12 hrs of light per 24-hr period. Rodent diet and tap water were consumed by rats ad libitum.
Experimental design
The rats were divided into 2 groups and were treated as follows:
Group I (control): The rats in this group ( Group II: The rats (
Six rats were killed at each time interval of 6 hrs, 16 hrs, and 24 hrs after the administration of CP. The kidneys were removed, blotted dry, and processed for electron microscopic studies.
Scanning electron microscopy of kidney epithelium
The kidneys were fixed in 3% glutaraldehyde and washed in 0.1 M sodium cacodylate buffer pH 7.4, post-fixed by 1% osmium tetraoxide and washed in buffer, and dehydrated in increasing concentrations of alcohol. The tissues were washed with propylene oxide and embedded in epoxy-resin embedding medium. Semi-thin sections approximately 1 μm thickness were cut with a glass knife on Leica ultracut (UCT) ultramicrotome. Sections were stained with toluidine blue. Ultrathin (below 100 nm) sections were collected on copper grids, stained with uranyl acetate and Reynold’s solution (sodium citrate and lead nitrate), and examined with transmission electron microscope (Philips 201C by Netherland) and photographed. Both the glomeruli and tubules were examined.
Results
The glomerulus of the control rats appeared normal (Figure 1a). The proximal convoluted tubule showed normal brush border. The cells lining the tubule had a number of mitochondria (normal feature) and lysosomes (Figure 1b). Distal convoluted tubule showed numerous mitochondria (Figure 1c).

Electron microscopic appearance of glomerulus and tubules of control rats. (a) Glomerulus of control rat showing normal structure, ×1500. (b) Proximal tubule showing numerous mitochondria and lysosomes, ×1500. (c) Distal tubules showing numerous mitochondria, ×3000.
Treatment with CP resulted in remarkable damage to cells of the glomerulus and the tubules. Six hours after treatment with CP, destruction of the podocytes was observed (Figure 2a). In the convoluted tubule, increase in the number of mitochondria and lysosomes was observed (Figure 2b).
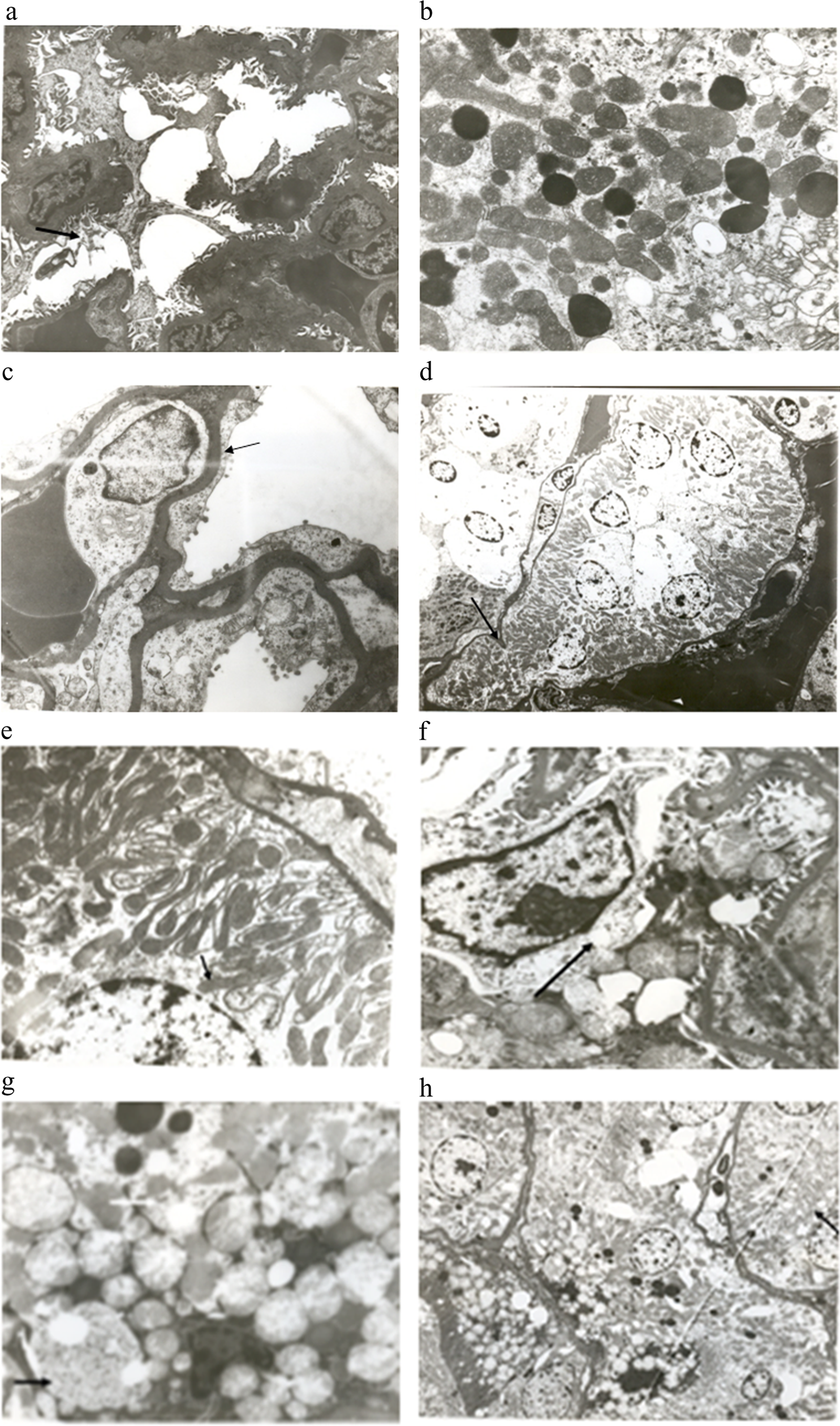
Electron microscopic appearance of glomerulus and tubules of cyclophosphamide treated rats. (a) Shows destruction of podocytes (arrow), ×3000. (b) Shows increased number of mitochondria and lysosomes in the tubules, ×10,000. (c) Thickening of the basement membrane (arrow) and nucleoplasm of endothelial cell showing less granularity, ×7000. (d) Shows distortion of tubules (arrow) and renal tubular necrosis, ×1500. (e) Elongated mitochondria (arrow) in the renal tubules, ×7000. (f) Shows vacuolization of mesangial cell, ×10,000. (g) Shows increase in the number and size of peroxisomes (black arrow), polymorphic mitochondria, round vacuoles filled with opaque substance (white arrow), ×10,000. (h) Dilated endoplasmic reticulum and cytoplasmic vacuolization, ×2000.
Sixteen hours after treatment with CP, the basement membrane was thickened. Many more podocytes were destroyed. The nucleoplasm of the endothelial cell showed fewer granularities (Figure 2c). Distortion of the tubule was also seen. The brush border was destroyed and lysosomes had disappeared. Cell necrosis was also seen (Figure 2d). The mitochondria in the proximal convoluted tubules (PCT) were elongated (Figure 2e).
Twenty-four hours after treatment with CP, in the glomerulus, there was vacuolization of cytoplasm of the mesangial cells (Figure 2f). In the tubules, many peroxisomes were seen and they were enlarged. Polymorphic mitochondria were increased in number. There were very few lysosomes. Round vacuoles filled with homogenous opaque substance were also observed (Figure 2g). The endoplasmic reticulum (ER) was dilated. Cytoplasmic vacuolization was also seen in the tubule (Figure 2h).
In our previous study, we have reported the effect of CP on the protein content and the activities of the lysosomal enzymes of the rat kidney. 21 The protein content of the CP-treated rat kidney was increased by 110% at 16 hrs and by 171% at 24 hrs as compared with control. We observed significant decreases in the activities of all the lysosomal enzymes studied in the kidneys of CP-treated rats at 16 hrs and 24 hrs as compared with control. The activity of N-acetylglucosaminidase was decreased by 46% at 16 hrs and by 50% at 24 hrs after treatment with CP as compared with control. With respect to glucuronidase, the activity was decreased by 31% at 6 hrs, by 47% at 16 hrs and by 35% at 24 hrs after treatment with CP. The activity of acid phosphatase was decreased by 30% at 16 hrs and 24 hrs after treatment with CP.
Discussion
The present study addresses the ultrastructural changes in the rat kidney after the administration of CP. We analyzed the endothelial cell and the mesangial cell of the glomerulus, the peroxisomes, lysosomes, mitochondria, and ER of the tubules. Three striking findings in the present study are (1) increase in number and size of peroxisomes (peroxisomal proliferation), (2) decrease in the number of lysosomes, and (3) glomerular changes. Peroxisome proliferation, decrease in the number of lysosomes, and glomerular damage have not been previously reported, although a study done 4 decades ago reports increase in size of peroxisomes after the administration of single dose of CP. 22 Lavin and Koss 22 have demonstrated changes such as swelling of peroxisomes, dilatation of ER, and cell necrosis of the tubules in the rat kidney after the administration of a single dose of CP. However, they did not observe any glomerular abnormalities, decrease in the number of lysosomes, and peroxisome proliferation that were observed in our study. 22 Our study is the first to demonstrate peroxisomal proliferation and decrease in the number of lysosomes and glomerular changes in the kidneys of rats after CP administration.
Initially, it was assumed that the main function of peroxisomes was the decomposition of H2O2, a reactive oxygen species (ROS) generated by different peroxisomal oxidases (mainly flavoproteins) via catalase, the classical peroxisomal marker enzyme. However, it is now clear that peroxisomes are involved in generation of ROS. The main metabolic processes contributing to the generation of H2O2 in peroxisomes are the β-oxidation of fatty acids, the enzymatic reactions of the flavin oxidases, and the disproportionation of superoxide radicals. 23
Xenobiotics that induce peroxisome proliferation have been shown to increase the expression of peroxisomal β-oxidation enzymes several fold (10–20-fold). 24 These include di (2-ethylhexyl) phthalatephthalate ester plasticizers, PAHs and oil derivatives, PCBs, certain pesticides, and ciprofibrate. Such conditions are considered to generate peroxisome-induced oxidative stress, which may overwhelm the antioxidant capacity and lead to cellular injury. In rat and mouse liver in vivo and in isolated cultured hepatocytes, peroxisome proliferators induce disproportionate increases in the activity of enzymes that generate and degrade hydrogen peroxide, oxidize DNA, lipids and other molecules. Reddy and co-workers originally proposed that fatty acyl-CoA oxidase in the peroxisome is the enzyme responsible for oxidative stress by peroxisome proliferators.25,26 Recent evidence indicates that persistent peroxisome proliferation leads to the formation of 8-hydroxyguanosine in rat liver DNA, which supports the role for oxidative stress. 27
In the liver, xenobiotics cause peroxisome proliferation and sustained a peroxisome proliferator activated receptor (PPAR) α-dependent activation of the H2O2-generating enzymes ACOX-1 and CYP4A and a decline in the activities of H2O2-degrading catalase and glutathione peroxidase.28,29 These changes result in the accumulation of the oxidative stress marker lipofuscin and the formation of 8-hydroxy-2′-deoxyguanosine DNA adducts in liver cells.30,31 Our study is the first one to show peroxisomal proliferation in the kidney after the administration of CP.
Another significant finding is the disappearance of lysosomes and extensive cytoplasmic vacuolization containing opaque substance in the renal tubular cells. Lysosomes which are major machinery for recycling intra- and extracellular material have long been regarded as “suicide bags.” Lysosomes contain several digestive enzymes including proteases that digest several extracellular proteins as well as cytoplasmic organelles and cytosolic proteins including glycoproteins. Lysosomal dysfunction (decrease in the activities of the lysosomal enzymes) can result in lack of digestion of proteins and other macromolecules that are regularly degraded by the lysosomes thereby resulting in the accumulation of undigested proteins and other macromeolecules within the cell. Accordingly, in a recent study, we have demonstrated increase in protein content and decrease in the activities of the lysosomal enzymes in the kidneys of CP-treated rats.
21
We have demonstrated increase in protein content in the kidney by 110%, 24 hrs after treatment with CP as compared with control. We have also demonstrated significant decrease in the activities of the lysosomal enzymes, namely,
Lysosomal dysfunction can result in lack of digestion of proteins that are regularly degraded by the lysosomes thereby resulting in increased half-life of proteins. This results in the accumulation of abnormal amount of proteins within the cell. Increases in protein content alone are being used as marker for hypertrophy, although increase in the ratio of protein to DNA is considered a better marker. 32 CP-induced renal epithelial cell damage is associated with increase in total protein content of the kidney as well as a decrease in the activity of lysosomal enzymes suggesting accumulation of proteins secondary to lysosomal dysfunction. Thus, lysosomal dysfunction may contribute at least in part to CP-induced renal damage.
In the present study, besides increase in the number and size of peroxisomes and decrease in the number of lysosomes, mitochondrial damage was also observed. The mitochondria were polymorphic and elongated. Mitochondria are the energy source of the cell and therefore damage to mitochondria can deplete the ATP of cell, resulting in necrosis. 33 It is noteworthy to mention that 16 hrs after the administration of CP, we observed cell necrosis of the renal tubular cells.
Peroxisomes and mitochondria cooperate in the maintenance of several metabolic pathways including the complete oxidative degradation of fatty acids. In some cases, these two subcellular organelles share the same functions such as β-oxidation of long-chain fatty acids or ketogenesis. 34 Recently, direct contacts between peroxisomes and mitochondria by means of cargo selective transport by vesicular carriers were described in mammalian cells. 35 Like peroxisomes, the mitochondria have a key role in both the production and scavenging of ROS. The mitochondrial electron transport chain is a major site of free radical generation with a rate of cellular output comparable to or even higher than peroxisomal ROS production. Not surprisingly, abnormalities in peroxisomal metabolic machinery may lead to mitochondrial malfunction, which in turn aggravates cellular damage.
ER is normally composed of tubules, vesicles, and sacs forming lamella-like shape. 36 Crucial pathways that are related to carbohydrate metabolism, biotransformation, steroid metabolism, and protein processing function in the ER with integration to other organelles.37,38 Dilated ER is considered a representative of damaged cells. 36 When we examined ERs of the rats treated with CP, we found large dilatations and vacuolizations in ERs in many areas.
The results of the present study show that CP targets mainly the peroxisomes, lysosomes, mitochondria, and ER of the renal tubules. This study is an evidence of changes in morphology of rat kidney after induction of renal damage by a single dose of CP. Since transmission electron microscopy is the highest magnification tool at present, it can be useful in estimating the degree of injury and outcome of alternative treatment strategies in the management of CP-induced renal damage after establishing a scoring system.
At present, we are investigating the activities of the different ROS-generating and ROS-degrading enzymes in the kidneys of CP-treated rats.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
