Abstract
There are no controlled human studies to determine the efficacy of benzodiazepines or antipsychotic medications for prevention or treatment of acute cocaine toxicity. The only available controlled data are from animal models and these studies have reported inconsistent benefits. The objective of this study was to quantify the reported efficacy of benzodiazepines and antipsychotic medication for the prevention of mortality due to cocaine poisoning. We conducted a systematic review to identify English language articles describing experiments that compared a benzodiazepine or antipsychotic medication to placebo for the prevention of acute cocaine toxicity in an animal model. We then used these articles in a meta-analysis with a random-effects model to quantify the absolute risk reduction observed in these experiments. We found 10 articles evaluating antipsychotic medications and 15 articles evaluating benzodiazepines. Antipsychotic medications reduced the risk of death by 27% (95% CI, 15.2%–38.7%) compared to placebo and benzodiazepines reduced the risk of death by 52% (42.8%–60.7%) compared to placebo. Both treatments showed evidence of a dose-response effect, and no experiment found a statistically significant increase in risk of death. We conclude that both benzodiazepines and antipsychotic medications are effective for the prevention of lethality from cocaine toxicity in animal models.
Keywords
Introduction
In 2004, it was estimated that 34 million people in the United States had used cocaine at least one time. 1 In 2005, there were 448,481 cocaine-related visits to US Emergency Departments (EDs). 2 Cocaine intoxication leads to numerous physiologic abnormalities and can lead to death by various mechanisms including, among others, cardiac ischemia, cardiac dysrrhythmias, hyperthermia and seizures. While cocaine has some peripheral effects (including increased release of catecholamines and sodium channel antagonism), the majority of the toxic effects are mediated by central nervous system (CNS) neurotransmitters such as dopamine and serotonin.3–6 Increased release of these neurotransmitters results in CNS stimulation and activation of the sympathetic autonomic system. Mouse studies suggest that cocaine acts at the dopamine transporter, muscarinic, and sigma receptors to produce lethality. 6 The majority of therapies for cocaine intoxication have been directed at reversing the CNS effects.
Historically, benzodiazepines have been the first-line treatment for acute cocaine intoxication. These agents are recommended for the treatment of tachycardia, hypertension, agitation, and seizures associated with cocaine. Benzodiazepines are gamma amino butyric acid (GABA) agonists, which have marked sedative-hypnotic, anti-anxiolytic, and anticonvulsant effects.3–6 The CNS depressive effects produce physiological antagonism of the CNS stimulatory effects of cocaine. Benzodiazepines reduce the mortality of acute cocaine poisoning in animal models,7–20 but the degree of this effect is unclear.
Typical antipsychotic medications are characterized by their anti-dopaminergic activity in the CNS while newer antipsychotic medications have increased affinity for serotonin receptor subtypes and have been labeled as atypical antipsychotics. 21 Importantly, the atypical antipsychotic medications bind at several sites relevant to cocaine toxicity. Olanzapine has a high affinity for the M1 muscarinic receptor, and ziprasidone inhibits the dopamine transporter. 22 This suggests that the atypical antipsychotic medications may be useful therapies for preventing or treating cocaine toxicity. Animal studies of antipsychotic medications for cocaine toxicity have produced mixed results. While some studies have reported a protective effect, others have found no benefit. Some have suggested that antipsychotic medications, especially phenothiazines and butyrophenones, should be avoided in acute cocaine poisoning due to the possibility of lowering the seizure threshold and worsening the epileptogenic effects of cocaine, prolonging the QTc interval and precipitating dysrrhythmias, exacerbating tachycardia, and hindering the ability to dissipate heat.
It is unlikely that human trials will ever measure the efficacy of these 2 treatments. Furthermore, most animal studies are relatively small and not powered for precise measurement of the effect on lethality. The objective of this study was to quantify the efficacy of benzodiazepines and antipsychotic medication for the prevention of mortality due to cocaine poisoning in animal models using a meta-analysis of reported studies.
Methods
Inclusion criteria
As our primary objective was to evaluate the efficacy of benzodiazepines and antipsychotic medications for prevention of cocaine toxicity, we sought studies that compared either of these medications to placebo in an animal model that included mortality as an outcome.
Literature searches
We conducted a PubMed search of English articles on May 25, 2010, to identify articles describing studies of antipsychotic medications using the following search terms: haloperidol or ziprasidone or olanzapine or chlorpromazine [MeSH term, substance name or text word] and cocaine [MeSH term] and animals [MeSH term]. Similarly, for the benzodiazepines we conduced PubMed search of English articles using the following search terms: benzodiazepine [MeSH Term] AND cocaine [MeSH Term] AND animals [MeSH Term].
The titles and abstracts were reviewed to identify studies that reported the use of the medications for prevention of lethality from cocaine poisoning. The articles that met inclusion criteria were reviewed and we abstracted the following information: specific medication, medication dose, cocaine dose, animal type, number in each treatment group, and outcomes in each treatment group. When outcomes were reported as percentages with a range of sample sizes, we assumed the smallest sample size that could yield that proportion. For example, if the authors reported that 6–8 animals per group were used and the proportion surviving was 66%, we assumed that there were 6 animals in the group and 4 survived.
We performed a meta-analyses to determine the absolute risk difference of survival (% surviving in treatment group minus % surviving in placebo) with 95% CI for antipsychotic medications vs placebo and benzodiazepine vs placebo. The analysis was performed in using Comprehensive Meta-Analysis version 2.2.048 software (Biostat Inc., Englewood, NJ, USA) using random effects models. If a manuscript reported several experiments with different doses of either cocaine or treatment medication, we included each experiment separately in our model. Our primary analysis plan included an overall evaluation by class (benzodiazepines and antipsychotic medications). When available, we reported results where antipsychotic medications and benzodiazepines were compared directly. We also evaluated medication-specific effects and dose-response effects. Our analysis of dose response was limited to medications where more than 2 doses were compared in a similar model (i.e. rodent or large animal).
Results
Search results
The antipsychotic medications search identified 399 articles and our benzodiazepine search identified 387 articles (two were excluded because they were not English). After review of the titles and abstracts, 10 articles compared an antipsychotic medication to placebo and 15 articles reported a study comparing a benzodiazepine to placebo in the setting of acute cocaine poisoning with survival as an outcome.
Characteristics of studies
The antipsychotic medications studied included ziprasidone (n = 2, 0.4–4 mg/kg),15,23 haloperidol (n = 4, 0.3–30 mg/kg),16,18,24,25 chlorpromazine (n = 4, 2–20 mg/kg),17,19,20,26 and olanzapine (n = 1, 1 mg/kg). 27 The study animals were: rats (n = 4), mice (n = 5), dogs (n = 1), and monkeys (n = 1). Benzodiazepines studied included diazepam (n = 11, 0.5–10 mg/kg),7,8,10–13,15,17–20 clonazepam (n = 1, 0.3–3 mg/kg), 8 Ro 15–4513 (n = 1, 5–15 mg/kg), 9 LA-VI (n = 1, 1–5 mg/kg), 13 LA-XIV (n = 1, 0.1–0.5 mg/kg), 13 and midazolam (n = 1, 0.32–3.2 mg/kg). 14 The study animals were: rats (n = 7), mice (n = 4), dogs (n = 1), and monkeys (n = 1).
Both antipsychotic medications and benzodiazepines improved survival from cocaine poisoning (Figures 1 and 2). Antipsychotic medications increased survival by 27.0% (95% CI, 15.2%–38.7%) while benzodiazepines increased survival by 51.7% (42.8%–60.7%). No experiments reported a statistically increased risk of mortality with any antipsychotic medication or benzodiazepine treatment.

Forrest plot of experiments comparing survival of animals treated with antipsychotic medications or placebo.
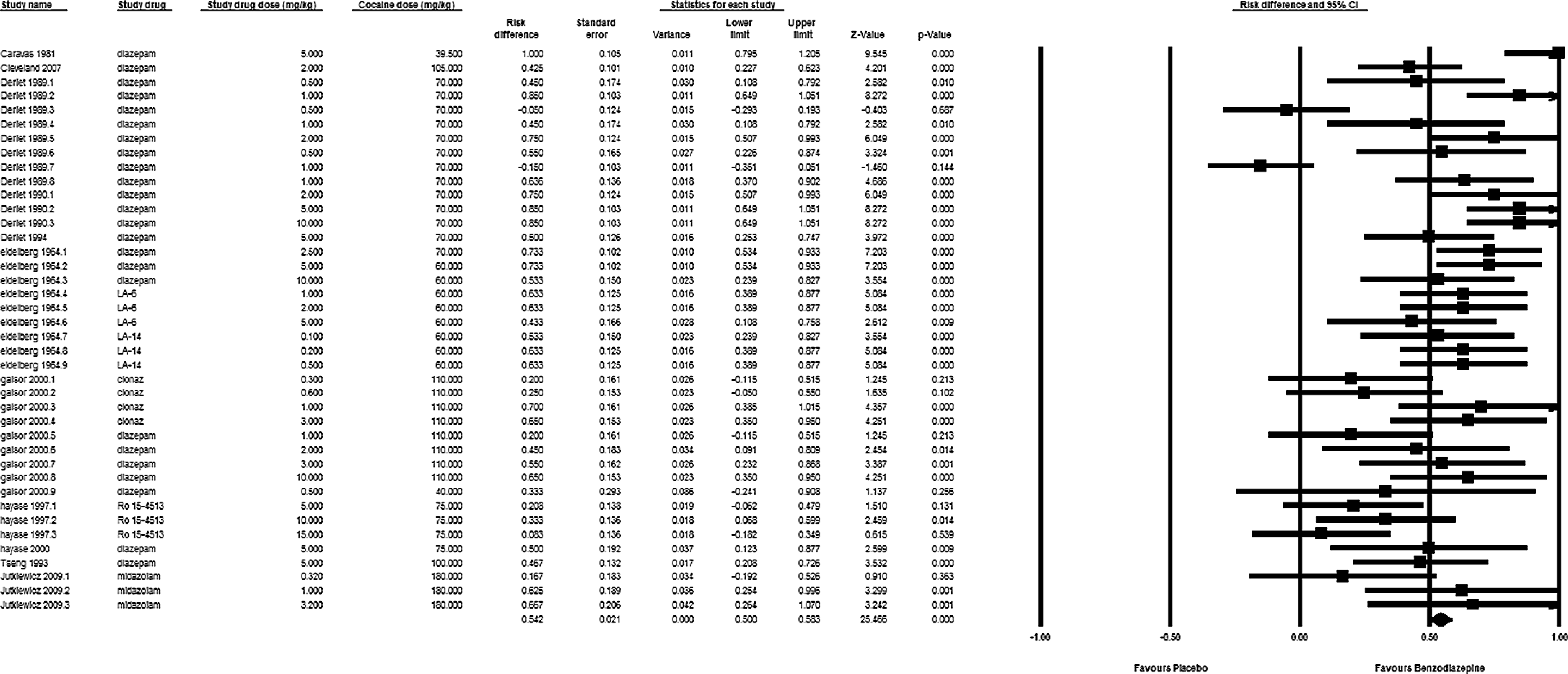
Forrest plot of experiments comparing survival of animals treated with benzodiazepines or placebo.
Five studies directly compared antipsychotic medications and benzodiazepines.15,17–20 Catravas found both chlorpromazine (12 mg/kg IV) and diazepam (3–5 mg/kg iv) prevented the death following an “LD100” of IV cocaine in a dog model. Guinn reported that chlorpromazine (10 mg/kg) prevented death in 3/3 primates treated with a lethal dose of IV cocaine (25.5 mg/kg, 0/3 control animals survived), while 2/3 of diazepam (0.5 mg/kg iv)-treated animals survived at that dose. Smith evaluated diazepam (5 mg/kg IV) and chlorpromazine (20 mg/kg IV) for prevention of lethality from an LD 50 of IV cocaine (14 mg/kg). Six of ten animals survived with both treatments. Derlet demonstrated dramatically improved survival with diazepam treatment (9/10 at 2 mg/kg and 10/10 at 5 and 10 mg/kg) but only 30%–40% survival with haloperidol at doses of 5–20 mg/kg. Finally, Cleveland reported similar survival following pretreatment with 4 mg/kg ziprasidone and 2 mg/kg diazepam (65% vs 70%, respectively).
Dose effects
Seventeen rodent experiments from 4 studies compared multiple doses of diazepam. The doses ranged from 0.5 to 20 mg. Three studies showed a clear dose-response effect (better survival with higher doses).8,12,18 One of these studies evaluated also varied in the timing of diazepam relative to cocaine dosing (i.e. 30 minutes pre-cocaine, 8 minutes pre-cocaine, and 3 minutes post-cocaine). 12 The dose-response effect was evident under all experimental conditions. The one study that did not find a clear dose-response effect reported survival in 10/10 animals treated with 2.5 and 10 mg/kg but only 8/10 animals treated with 20 mg/kg survived. 13 One study compared three doses of midazolam and found a clear dose-response effect between 0.32 and 3.2 mg/kg. 14
One study compared two doses of ziprasidone (0.4 and 4 mg/kg), 23 and one study compared two doses of chlorpromazine (5 and 20 mg/kg) in rodents. 26 Both studies found a better effect at the higher dose. Four studies compared multiple doses of haloperidol (0.5–20 mg/kg). One study found a modest effect (4/8 survival at 1 and 5 mg/kg and 7/7 survival at 10 and 20 kg) 16 while the other studies did not find a dose-response effect.18,24,25
Discussion
Antipsychotic medications and benzodiazepines are effective for the prevention of cocaine toxicity in animal models. Compared to placebo, benzodiazepines increased survival more than antipsychotic medications. We found no studies that reported a statistically increased rate of death for either treatment.
The magnitude of the risk difference for each medication compared to placebo suggests that benzodiazepines are more effective than antipsychotic medications. However, this conclusion must be tempered. First, very few studies directly compared the two treatments. It is possible that the higher survival rate in the benzodiazepine studies may be due to different experimental conditions rather than differences in efficacy. Second, while there is little variation in the mechanisms of action among the benzodiazepines, the antipsychotic medications have variable pharmacology. Our meta-analysis provides a single effect measure for antipsychotics that may not be generalizable to each individual medication. However, it is important to note that no study reported increased mortality following antipsychotic medication pretreatment.
While the results of animal studies cannot be directly applied to treatment of human disease, animal models often provide the only practical way that therapies can be studied in a well controlled manner. Acute cocaine toxicity is one example of a condition where controlled human studies are not feasible. Severe toxicity is rare, requires immediate treatment, and renders patients unable to provide informed consent. The only human evidence for treatment efficacy in severe toxicity is descriptive rather than comparative, and no report describes more than three cases from a single center. The heterogeneity of cases is extreme. Reports have described cases of severe toxicity presenting as cardiac arrest, while others were initially stable and only developed symptoms once they arrived to health care. In fact, clinicians are often treating patients with undifferentiated agitation where a history of cocaine use may be concealed or otherwise unavailable. This spectrum of disease further complicates the planning of clinical trials as it increases the sample size required to detect a difference in the efficacy of treatments. Overall, these factors make a meaningful human study of treatments for severe cocaine toxicity very unlikely.
While quality human studies evaluating therapies for acute cocaine toxicity are unlikely, clinicians still have to make treatment decisions using the best available evidence. Standard toxicology and emergency medicine texts recommend benzodiazepines as first-line treatment for acute cocaine toxicity, and our findings support these recommendations. However, many experts recommend against the use of antipsychotic medications for treatment of cocaine-induced delirium. They suggest that these medications will increase toxicity by increasing seizures, prolongation of the QT interval, and decreased heat dissipation. Our results suggest that antipsychotic medications are effective for the prevention of cocaine toxicity in animal models and we found no evidence that these medications increase the toxicity of cocaine.
There are several limitations to our study. First, we only reviewed published manuscripts. Thus, our findings are susceptible to publication bias if studies that found these treatments were ineffective are less likely to be published than positive studies. A second limitation is that there were multiple doses of benzodiazepines and antipsychotic medications used in many of the included papers. If the published manuscripts included multiple experiments using less effective (i.e. lower) doses, the magnitude of the estimated risk reduction would be biased toward the null. As our calculated risk reductions for both therapies were statistically significant, we believe our conclusions are valid and that it is likely that the true benefit of these treatments (when administered in the optimum dose) is greater than our estimates of the risk reductions. We also limited our findings to paper written in English and to a single database (PubMed). While the limitation to a single language often biases the results in human studies, that this restriction should not bias our results. We believe this because while there are often substantial differences in populations or clinical circumstances between English speaking and non-English speaking human populations, it seems unlikely that there are substantial differences in animal populations studied by English speaking and non-English speaking researchers. Similarly, it seems unlikely that reports captured in a database other than PubMed would systematically differ from those included in PubMed. Another limitation is that we were not always able to determine the exact sample size when authors reported a percentage surviving (or not surviving) and the sample size was provided as a range for several doses. This increases the imprecision of our estimates. We tried to limit this by assuming the smallest sample size that could produce the percentage reported. If the sample size was actually larger, the impact of the results from the study would be underestimated.
The main implication of our findings is to suggest that antipsychotics may be useful in the treatment of cocaine toxicity. While our work suggests no reason to abandon benzodiazepines as first-line treatment for cocaine intoxication, there are theoretical reasons to suggest that antipsychotic medications may also have a role. Newer antipsychotic medications are potent antagonists of CNS serotonin receptors, and these receptors have been identified as mediating the fatal effects of cocaine. As benzodiazepines work on another family of neurotransmitters, there is a potential that the effects of these two treatments would be additive. There are also times where physicians must treat a patient with agitation before the diagnosis of cocaine intoxication is established. In many institutions, antipsychotic medications are considered the first-line therapy for these cases. Our findings suggest no reason to forgo antipsychotics if there is a possibility of cocaine intoxication as the cause of the agitation.
In conclusion, benzodiazepines and antipsychotic medications attenuate severe cocaine toxicity in animal models. While our comparison was limited by the available studies, benzodiazepines appear to improve survival more than antipsychotics medications. While benzodiazepines should remain the treatment of choice for acute cocaine toxicity, we suggest that antipsychotic medications be investigated further as potential treatments for cocaine intoxication.
Footnotes
Acknowledgements
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute on Drug Abuse or the National Institutes of Health.
The project described was supported by Award Number K08DA020573 from the National Institute on Drug Abuse.
