Abstract
The renin-angiotensin system (RAS) subserves vital physiological functions and also implicated in certain pathological states. Modulation of this system has been proposed in recent studies to be a promising strategy in treating liver fibrosis. We investigated the effect of the pharmacologic inhibition of RAS with angiotensin-converting enzyme inhibitor or angiotensin receptor blocker in CCl4-induced liver injury with a view to ascertaining the chemopreventive benefit. Fifty-six Wistar albino rats were divided into eight experimental groups of seven rats/group. Groups 1–4 received normal saline (10 ml/kg), enalapril (0.6 mg/kg), losartan (1.4 mg/kg) and CCl4 (80 mg/kg), respectively. Groups 5–8 were pretreated with enalapril (0.3 mg/kg), enalapril (0.6 mg/kg), losartan (0.7 mg/kg) and losartan (1.4 mg/kg) 1 hour before CCl4 administration. Experiment lasted 11 days and dosing was via oral route. Rats were killed 24 hours after the last treatment. Serum activities of alkaline phosphatase, aspartate and alanine aminotransferases increased significantly (p < 0.05) by 46.0%, 90.6% and 122.3%, respectively, with severe hepatic centrilobular necrosis, fatty infiltration and increase in liver weight (p < 0.05) in the CCl4-treated rats. Enalapril (0.6 mg/kg) and losartan (1.4 mg/kg) significantly (p < 0.05) increased aspartate aminotransferase activity by 37.0% and 94.7% and produced mild centrilobular and periportal hepatic necrosis, respectively, with enalapril significantly (p < 0.05) increasing liver weight. Serum total cholesterol, triglyceride, albumin and total protein did not change significantly in these rats. Also, glutathione, malondialdehyde and uric acid levels were not significantly altered. Enalapril and losartan failed to attenuate liver injury associated with CCl4 treatment. Although both drugs did not significantly alter serum biochemistry in the CCl4-treated rats, they however produced slight elevations in biomarkers of liver function and appear to worsen liver histopathology. Overall, the chemopreventive benefits of RAS inhibitors in liver disease remain doubtful and should be used with caution during hepatic dysfunction.
Introduction
There is increasing evidence that both the ‘classical’ and the ‘alternative’ renin-angiotensin system (RAS) are upregulated in chronic liver disease,1,2 and that the progression of liver fibrosis may be influenced by a balance between angiotensin-converting enzyme (ACE) and angiotensin-converting enzyme-2 (ACE2) activation. 3 Angiotensin-converting enzyme-2 (ACE2) gene and activity have been reported to be upregulated in experimental model of secondary biliary fibrosis and in humans with hepatitis C.1,2 As fibrosis worsens, the progressive rise in ACE and angiotensin II type 1 (AT1) receptor gene expressions coincide with an increase in ACE2 and mas expression, together with increased plasma levels of both angiotensin-(1-7) [Ang-(1-7)] and angiotensin II (Ang II).2,4
In liver disease, architectural changes to the microscopic structure of the liver lead to capillarization of the hepatic sinusoids, increased extracellular matrix formation and elevated hepatic resistance. The latter impedes liver blood flow and leads to portal hypertension. 5 Stretching of the portal vein (as with increased hepatic resistance to blood flow) and oxidative stress together cause release of vasodilators, including nitric oxide, which induce a number of compensatory mechanisms important for restoring the functional blood volume. These mechanisms are effected via sodium and water preservation and stimulation of the sympathetic nervous system, which together contribute to the development of ascites, oedema, hepatorenal syndrome and a hyperdynamic circulation, all of which are typically seen in patients with advanced liver disease. The RAS is involved with all these processes.5,6
Recently, the manipulation of the RAS with either antagonists of the ‘classical’ pathway, or agonists of the ‘alternative’ pathway was proposed to have potential therapeutic benefits.6,7 The avid interest in RAS-blocking drugs is, in part, related to their relative safety in humans and widespread use in cardiovascular and renal medicine. 6 Several studies have reported the ability of these drugs to limit the progression of pulmonary and renal fibrosis.8,9 Interestingly, beta-blockers, which interact with the RAS by inhibiting renin release, have not been shown to impact on the development or progression of hepatic fibrosis. 6 In this present study, we attempted to investigate the exact involvement of RAS in liver disease by evaluating the effect of RAS inhibitors in experimental liver injury induced by CCl4 and to ascertain whether protection could be obtained either by ACE inhibition or angiotensin II type I receptor antagonism. Experimental model of liver injury with CCl4 injections has revealed many findings, especially mechanisms of liver damage and regeneration. Studies have shown that CCl4 and its metabolites (trichloromethyl [CCl3] and trichloromethylperoxy [OOCCl3] radicals) are capable of inducing peroxidation of cell membrane lipids, regeneration of reactive oxygen free radicals and hepatocellular fatty regeneration with centrilobular necrosis of the liver.10,11 Therefore, CCl4 was the hepatotoxin of choice in our study.
Materials and methods
Chemicals and drugs
Enalapril and losartan were obtained from ATOZ Pharmaceutical Production Limited (India) and Ranbaxy laboratories (India), respectively. Thiobarbituric acid (TBA) was purchased from Sigma Chemical Company (USA). Reduced glutathione (GSH), metaphosphoric acid and trichloroacetic acid (TCA) were purchased from J.I. Baker (USA). Alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total cholesterol (TC), triglyceride (TG) and uric acid (UA) assay kits were obtained from Randox Laboratory (Crumlin, UK), 5′,5′-dithiobis-2-nitrobenzoate (Ellman’s reagent) from Sigma (USA) and sodium hydroxide from Merck (Germany). All other chemicals and reagents used were of analytical grade.
Animals
Albino rats of the Wistar strain were obtained from commercial private colony in Ibadan, Oyo-State, Nigeria. The rats were housed within the experimental animal handling facility of the Department of Pharmacology, Olabisi Onabanjo University, Nigeria, at ambient temperature and humidity with a 12 h light/12 h dark schedule. They were fed with commercially available rat pelleted diet (Bendel Feeds, Edo State, Nigeria) and water ad libitum during period of acclimatization and throughout the period of the experiment. Study was carried out in strict compliance with established guidelines for care and use of laboratory animals in biomedical research.
Experimental design
Fifty-six (56) rats weighing 132.8 ± 1.6 g were divided into eight groups of seven rats per group. Hepatotoxicity was induced by sub-acute administration of CCl4 dissolved in corn oil (0.05 ml CCl4/ml corn oil) following the dosage regimen described by Bruckner et al. 12 Negative control rats in group 1 received normal saline (10 ml/kg) while those in group 2 were treated with CCl4 (80 mg/kg) and served as the positive control. Rats in groups 3 and 4 were treated with enalapril (0.6 mg/kg) and losartan (1.4 mg/kg) respectively. Groups 5, 6, 7 and 8 received enalapril (0.3 mg/kg), enalapril (0.6 mg/kg), losartan (0.7 mg/kg) and losartan (1.4 mg/kg) 1 hour before CCl4 (80 mg/kg) administration respectively. Enalapril and losartan were administered at the therapeutic and twice the therapeutic doses and calculated based on 70 kg body weight for a physiological man (therapeutic doses of enalapril and losartan are 20 mg/70 kg body weight and 50 mg/70 kg body weight, respectively). All treatment was via the oral route. The 11 days treatment started with 5-days administration followed by 2-days rest and then with a continuous administration for 4-days.
Necropsy
The animals were sacrificed by cervical dislocation 24 hours after the last treatment. Blood samples were collected by cardiac puncture into lithium heparin bottles and centrifuged at 4200 rpm at room temperature for 5 minutes to separate plasma. The liver was removed, cleared of adhering tissues and weighed. The weight was recorded in grams and expressed as g/kg body weight. A small portion of the liver was carefully excised, fixed in 10% formaldehyde, dehydrated in graded alcohol and embedded in paraffin. Fine sections were obtained, mounted on glass slides and counter-stained with hematoxyllin and eosin (H&E) for histopathologic examination. The remaining portion of the liver was weighed and homogenized in four volumes of phosphate buffer (0.1 M, pH 7.4). Both plasma and liver homogenate were used for biochemical analysis.
Assessment of Hepatic Function
Liver function was assessed by measuring the activities of AST, ALT and ALP in plasma. AST and ALT activities were determined according to the principle described by Reitman and Frankel 13 while the ALP activity was carried out according to the method described by Roy. 14 To assess the synthetic function of the liver, total protein and albumin concentrations were carried out according to the principle based on Biuret reaction 15 and bromocresol green reaction, 16 respectively.
GSH determination and lipid peroxidation assay
GSH level was estimated at 412 nm following the method of Beutler et al. 17 Lipid peroxidation was estimated spectrophotometrically by the thiobarbituric acid reactive substance (TBARS) method as described by Varshney and Kale 18 and expressed in terms of malondialdehyde (MDA) formed per mg protein.
Uric acid determination
UA was also determined using Randox kit following the principle described by Fossati et al. 19
Cholesterol and triglyceride assay
TC and TG concentrations were estimated following the principle described by Trinder 20 using commercial kits obtained from Randox Laboratories Ltd (Crumlin, UK).
Statistics
Results were expressed as mean ± standard error of mean (SEM). Differences between groups were determined by one-way analysis of variance (ANOVA) using Statistical Package for Social Sciences (SPSS) software for windows. Post hoc testing was performed for inter-group comparisons using the least significant difference (LSD) 21 and p value <0.05 was considered significant.
Results
Liver function
Table 1 shows the result of the effect of enalapril and losartan on normal and CCl4-treated rats. Hepatic damage produced by CCl4 significantly (p < 0.05) led to increases in the activities of AST, ALT and ALP by 90.6%, 122.3% and 46.0% respectively. Enalapril (0.6 mg/kg) and losartan (1.4 mg/kg) when administered individually significantly (p < 0.05) increased AST activity by 37.0% and 94.7%, respectively, in the normal rats. In addition, losartan raised the activity of ALT and ALP by 10.9% and 24.1%, respectively, though not significant (p > 0.05). Enalapril (0.6 mg/kg) produced mild to moderate increases (p > 0.05) in the activity of these enzymes in the CCl4-treated rats. Losartan (1.4 mg/kg), on the other hand, significantly (p < 0.05) enhanced the CCl4-induced increases in AST and ALP activities by 17.7% and 27.3% respectively with only a negligible increase in ALT. Total protein and albumin concentrations were not significantly affected in all the treatment groups (Figure 1).
Effect of enalapril and losartan on liver function in normal and carbon tetrachloride-treated rats

ALT: alanine aminotransferase, AST: aspartate aminotransferase, ALP: alkaline phosphatase, ENL: enalapril, LOS: losartan.
Note: Result expressed as mean ± SEM. Values in parenthesis represent % change; (−) increase; (+) decrease.
a Percentage change relative to control group.
b Percentage change relative to CCl4.
c p < 0.05 when compared with control group
d p < 0.001 when compared with control group.
e p < 0.05 when compared with CCl4 group.
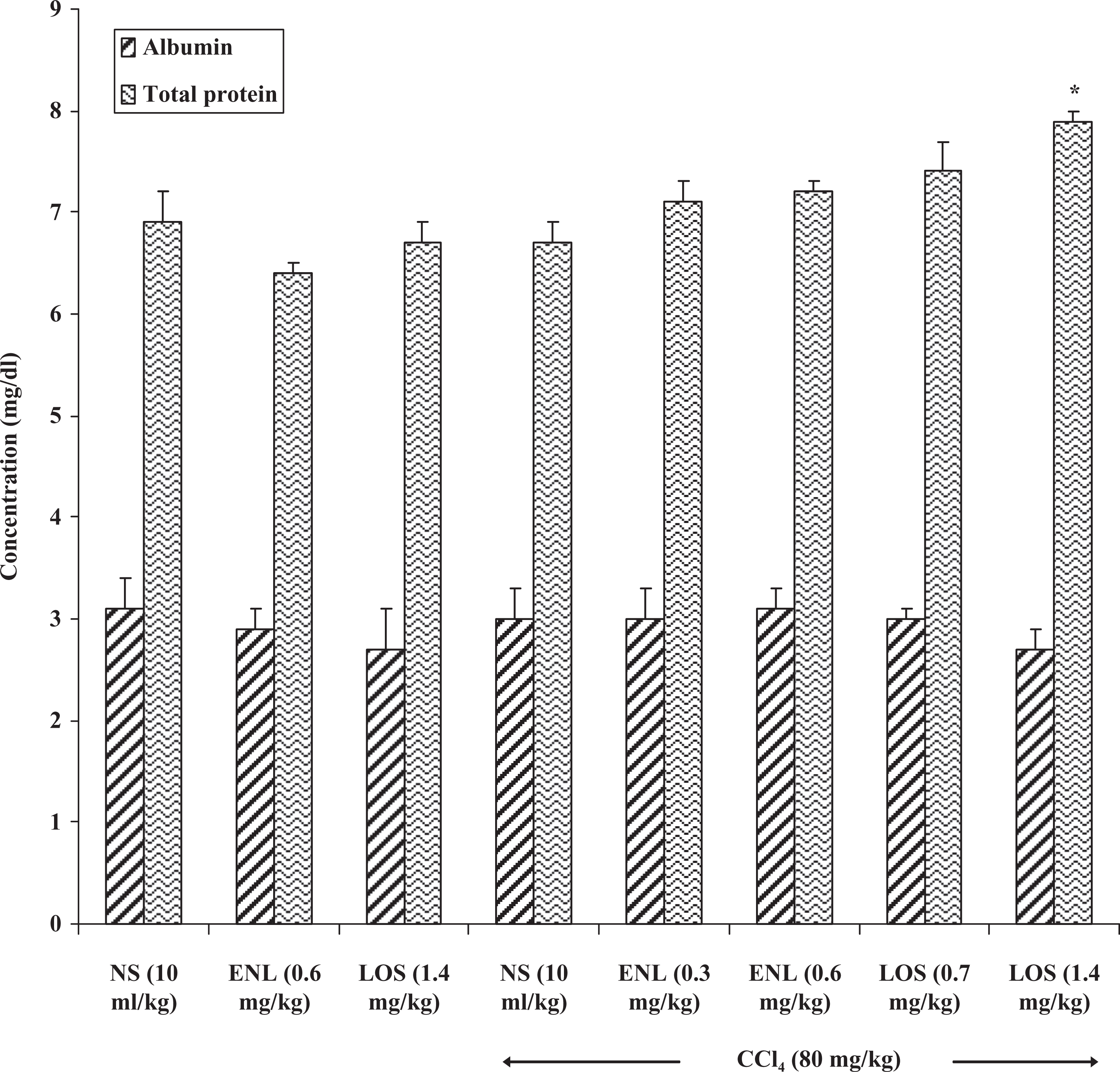
Effect of enalapril and losartan on plasma albumin and total protein levels in normal and carbon tetrachloride-treated rats. Result expressed as: mean ± SEM. *p < 0.05 when compared with CCl4 group. NS: normal saline, ENL: enalapril, LOS: losartan.
GSH and UA
GSH concentration increased by 20.7% in the CCl4-treated rats when compared with control as indicated in Table 2. This value did not change appreciably when these rats were treated with enalapril or losartan prior to CCl4 challenge. Also, enalapril and losartan increased GSH by 10.3% and 24.1%, respectively, in the normal rats. Uric acid concentrations were in the same range in the various treatment groups and were not significantly different from those of the control and CCl4-treated groups (Table 3).
Effect of enalapril and losartan on hepatic reduced glutathione and malondialdehyde levels in normal and carbon tetrachloride treated rats

ENL: enalapril, LOS: losartan, GSH: reduced glutathione, MDA: malondialdehyde.
Note: Result expressed as mean ± SEM. Values in parenthesis represent % change; (−) increase; (+) decrease.
a Percentage change relative to control group.
b Percentage change relative to CCl4.
Effect of enalapril and losartan on uric acid level and liver weight in normal and carbon tetrachloride treated rats

UA: uric acid, ENL: enalapril, LOS: losartan
Note: Result expressed as mean ± SEM. Values in parenthesis represent % change; (−) increase; (+) decrease relative to control group.
a Percentage change relative to control group.
b p < 0.05 when compared with control.
c p < 0.001 when compared with control.
d Percentage change relative to CCl4.
Lipid peroxidation
Table 2 shows the effect of various treatments on lipid peroxidation. MDA level (index of lipid peroxidation) increased by 29.4% following CCl4 administration. Similarly, administration of enalapril and losartan increased MDA concentration by 20.6% and 22.1%, respectively, in the normal rats. MDA values in the CCl4-treated rats were comparable to those in the enalapril plus CCl4 group. However, slight reduction (p > 0.05) was observed in the losartan plus CCl4-treated groups when compared with CCl4 only.
Cholesterol and triglyceride
Results presented in Figure 2 reveal increase in total cholesterol and triglyceride concentrations by 12.5% and 28.1% after CCl4 administration. Enalapril increased TC by 17.2% in the normal rats and 15.9% in the CCl4 -treated rats at 0.6 mg/kg. Also, enalapril increased TG by 19.1% and decreased it by 15.0% at this dose in normal and CCl4 -treated rats respectively. Losartan, on the other hand, produced TC and TG values that were not too different from those of the control and CCl4 -treated rats when administered alone and with CCl4, respectively. These changes were however not significant (p > 0.05).

Effect of enalapril and losartan on plasma total cholesterol and triglyceride concentrations in normal and carbon tetrachloride treated rats. Result expressed as mean ± SEM. Values in parenthesis represent % change; (-) increase, (+) decrease, (a) % change relative to control group. (b) % change relative to CCl4. NS: normal saline, ENL: enalapril, LOS: losartan.
Liver weight
Table 3 shows the effect of various treatments on the rats' liver weight. The liver weight significantly (p < 0.05) increased in the CCl4-treated rats when compared with control. Enalapril (0.6 mg/kg) treatment also significantly (p < 0.05) increased the weight of the liver whereas only very slight change was produced with losartan when compared with control. Both drugs did not reduce the liver weight in the CCl4-treated rats.
Histopathology
CCl4 (80 mg/kg) treatment was characterized by severe hepatic centrilobular necrosis and fatty infiltration in the rats as indicated in Figure 3. Separate treatment with enalapril (0.6 mg/kg) and losartan (1.4 mg/kg) produced mild hepatic centrilobular necrosis, fatty infiltration with few areas of haemorrhage and mild periportal hepatic necrosis respectively in these rats. Pretreatment with enalapril and losartan 1 hour before CCl4 throughout the period of treatment enhanced the morphological damage induced by CCl4. CCl4-treated rats receiving enalapril and enalapril show greater degree of damage when compared with those treated with CCl4 only.

Liver section (×100) of rat treated with [A] normal saline (10 ml/kg) (control). [B] CCl4 (80 mg/kg) showing severe hepatic necrosis (centrilobular) and fatty infiltration. [C] Enalapril (0.6 mg/kg) showing mild hepatic centrilobular necrosis, fatty infiltration with few areas of haemorrhage. [D] Losartan (1.4 mg/kg) showing mild periportal hepatic necrosis. [E] CCl4 + enalapril (0.3 mg/kg) showing severe hepatic centrilobular necrosis, fatty infiltration with portal cellular infiltration by mononuclear cells. [F] CCl4 + enalapril (0.6 mg/kg) showing very severe hepatic necrosis (centrilobular), fatty infiltration. [G] CCl4 + losartan (0.7 mg/kg) showing severe hepatic necrosis (centrilobular), fatty infiltration. [H] CCl4 + losartan (1.4 mg/kg) showing very severe portal and central venous congestion with mild fatty infiltration.
Discussion
The ability of RAS inhibitors to limit the progression of pulmonary and renal fibrosis has been reported.8,9 Strong evidences also abound to suggest that angiotensin II and other key components of the RAS are important mediators in liver fibrosis. 22 There are indications also to suggest that manipulation of the RAS with either antagonists of the‘classical’ pathway, or agonists of the ‘alternative’ pathway could have potential therapeutic benefits. 6 In this present study, we compared the effect of inhibiting ACE using enalapril with blocking angiotensin II type I receptor using losartan in a rat model of CCl4-induced hepatotoxicity in rat.
The acute, subacute and subchronic toxic potency of ingested CCl4 has been well characterized. 12 Similar to previous findings, subacute CCl4 toxicity in this study was characterized by marked increase in the plasma activities of ALP and the aminotransferases (AST and ALT) together with severe hepatic centrilobular necrosis and fatty infiltration. Results from this study show that inhibiting the RAS with once-daily administration of therapeutic and double therapeutic doses of enalapril or losartan did not confer any protective benefit on CCl4-induced liver damage in the rats. Although, RAS inhibitors have been reported to have low incidence of side effects and regarded safe when administered for prolonged periods of time, 6 results from this study appear to suggest the contrary. Our observation supports previous reports of suspected enalapril-induced hepatotoxicity 23 and some cases of marked hepatotoxicity in patients after a short time treatment with losartan.24,25 Enalapril and losartan, when administered separately in this study, produced mild to moderate increases in the marker enzymes of liver function in the CCl4-treated rats, with losartan significantly increasing AST activity at double the therapeutic dose. It thus appears that treatment with RAS inhibitors may exacerbate or predispose to greater hepatic dysfunction during liver disease and losartan appears to possess a greater tendency to produce this effect. The significant increase in AST activity associated with both enalapril and losartan when administered alone coupled with the moderate increase in ALP and ALT activities produced by losartan alone may provide a basis for the exacerbation of hepatic injury. Similarly, the production of mild periportal hepatic necrosis and mild centrilobular hepatic necrosis with fatty infiltration by losartan or enalapril treatments respectively in the normal rats further strengthens this argument. In addition, studies have reported angiotensin converting enzyme inhibitors and possibly angiotensin receptor blockers to be capable of producing greater hypotension in the presence of hepatotoxicity. 26 It is possible therefore that the hypotension secondary to ACE inhibition and/or angiotensin receptor blockers administration may decrease liver perfusion and stimulation, causing secondary hypoxia and exacerbating the hepatotoxicity caused by CCl4. Also, ACE inhibitors and angiotensin receptor blockers may have a direct cytotoxic effect on the liver as previously reported,23,27 and this might have contributed to the enhanced histological damage associated with CCl4-induced hepatic injury. It seems therefore that the inherent risk of hypotension or cytotoxic effects of both drugs may have predisposed to the enhanced histopathological changes observed in this study. Our observations seem to corroborate previous findings with the use of ACE inhibitors in experimental model of liver injury as reported by Ovali et al. 28 However, other conflicting observations exist in the literature.29,30
Furthermore, the moderate increase in total cholesterol and triglyceride levels associated with sub-acute CCl4 treatment in this study was not significantly altered by enalapril and losartan. Although sub-acute treatment with CCl4 significantly elevated marker enzymes (ALP, ALT and AST) of liver function and produced morphological damage, serum albumin and total protein were not significantly altered. In addition, a moderate but non-significant induction of GSH and a slight increase in uric acid level was observed. GSH and uric acid offer protective and anti-oxidant properties for the body homeostasis and detoxification of xenobiotics that underlie body toxicity. 31 The increase in the levels of reduced glutathione and uric acid might be due to the body tolerance or adaptive changes in response to oxidative stress associated with CCl4 toxicity.32,33 This may account for the moderate increase in lipid peroxidation associated with CCl4 toxicity in this study. Enalapril and losartan did not significantly alter these effects in the CCl4-treated rats. Their effects on GSH and MDA levels when administered alone at twice their therapeutic doses were comparable to those of the toxicant.
We therefore conclude that in spite of the reported involvement and the theoretical benefit perceived to be inherent in inhibiting RAS in liver disease, inhibition of the renin-angiotensin system with enalapril and losartan failed to prevent or reduce hepatic damage induced by CCl4. Rather, a predisposition to or exacerbation of liver dysfunction and damage may result.
Footnotes
Acknowledgment
The support of the Head of Chemical Pathology Department, Prof. E.O. Ogunyemi, of the Olabisi Onabanjo University Teaching Hospital, Ogun State, Nigeria, is gratefully acknowledged.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
