Abstract
Summary
The involvement of
With respect to human health, the main focus was on the toxicological effects in rats and mice, conducted by the US National Toxicology Program, the outcome of which played an important role in setting risk assessment parameters for exposure of the fumonisins to humans. The International Agency for Research on Cancer characterized the fumonisins as Group 2B carcinogens, i.e. a possible carcinogen to humans. However, several controversial findings regarding the toxicological effects of the culture material of the fungus and pure FB1 in rats have been reported that should be clarified prior to assessing the risk in humans. The underlying differences between the diets with the high protein levels are likely to sensitize the kidneys to FB1-induced toxic and carcinogenic effects. Several other dietary factors, such as plant extracts (antioxidants) and dietary Fe, could either stimulate or inhibit cancer induction of FB1, which complicates the comparison of toxicological effects of FB1. Cognisance should be taken of the modulating role of dietary constituents as it will determine the outcome of toxicological assays and therefore determine the threshold of an adverse effect in a specific target organ to be used in determining risk assessment parameters.
Other determinants that will impact on risk assessment are thresholds for genotoxic and apparently non-genotoxic carcinogens and the underlying mechanism for cancer induction in the liver and kidneys of rats. Recent developments suggest that thresholds should also apply for genotoxic carcinogens when considering the induction of an adverse biological effect. In this regard, interaction with the DNA is just one event in the multi-step process of cancer development and therefore could not be taken as the basis for applying a no-effect threshold for genotoxins. It would appear that a carcinogen such as FB1, whether it is labeled genotoxic or non-genotoxic per se, exhibits some degree of risk at any level due to additive or synergistic interactions with other xenobiotics and/or dietary constituents. With respect to cancer promotion, it is generally accepted that the disruption of lipid metabolism could explain the toxicological effects in animals. This includes the disruption of sphingolipid, phospholipids and fatty acid (FA) metabolism, which play a major role in the modulation of apoptotic and cell proliferative pathways related to cancer development.
The uncertainty regarding the nephrocarcinogenicity due to a possible FB1/nephropathy interaction raises some doubts about using the outcome of the NTP study in establishing risk assessment parameters in humans. This becomes apparent as a variety of hyperplastic lesions occur in the kidneys as a result of nephropathy, which in the presence of FB1, could play a role in cancer development, especially the occurrence of a subtype referred to as atypical tubule hyperplasia. In the liver, neoplasia also developed from hyperplastic liver foci and nodules, however, with the difference that these lesions are induced by FB1 and not by any dietary-related response. The hepatocarcinogenic properties of the fumonisins, therefore, provide a far better established model to be used in risk assessment for the fumonisins when compared to the nephrocarcinogenicity.
Once the risk parameters of fumonisins have been clarified, regulation in food and the associated risk have to be considered from many perspectives. In developed countries, maize is not a major dietary staple and is used to generate income by exporting the maize to other countries including developing countries. In many of these developing countries, there is a lack of quality control implying that maize highly contaminated with mycotoxins may directly enter the food chain of adults and children. Many of these countries are politically unstable and due to poor socioeconomic status and underdeveloped agricultural practices, control of mycotoxins is difficult or in some cases totally absent. The interaction of politics, economy and technology will eventually determine the impact on health, which will differ between countries. Specific and simple measures should therefore be devised and introduced to reduce the levels of the fumonisins in maize by targeting populations at risk.
Fumonisin B mycotoxicology
FB1 was shown to be responsible for most of the toxicological effects of
The nephrocarcinogenic properties of FB1 could be related to specific dietary interactions such as a high-protein diet selectively sensitizing the kidneys due the chronic progressive nephropathy (CPN). Recently, it was reported that many renal tubule proliferative lesions developed in kidneys of especially male Fischer and Sprague-Dawley rats when chronically fed a high-protein diet, culminating in advanced CPN in older rats. 9 One of these proliferative lesions, atypical tubule hyperplasia (ATH) that develops into adenomas, is regarded as a precursor lesion for kidney carcinogenesis. The use of male Fischer rats receiving a high protein diet in chronic nephrotoxicity and nephrocarcinogenicity models was questioned due to the formation of ATH and adenomas. 10 When considering the toxic and cancer-promoting properties of the fumonisins, specific FB1/high protein diet interactions are likely to prevail whereby ATH could be promoted into adenomas and carcinomas. As the induction of ATH in rats fed a high-protein diet is also age-related, which is of interest as no neoplastic lesions were noticed in the kidneys up to 6 months in any of the FB1-treated rats, while hyperplastic tubule, ATH, adenomas and carcinomas were noticed after 2 years. 6 This would suggest that renal adenomas and carcinomas develop in the presence of advanced CPN, which complicates attempts to mechanistically define the role of FB1. The presence of tubule hyperplasia, ATH and adenomas was also noticed in the females fed the two highest dose levels, although to a far lower extent. 6,8 This is in agreement with the notion that male rats are more sensitive to CPN, which coincided with the higher tumour response when compared to females. However, it should be noted that lower FB1 dose levels were used in the females. The marginal dose response effect regarding the induction of renal tumours by FB1 further suggests a possible interaction with advanced CPN. The NTP study, therefore, is likely to provide additional information on FB/dietary interactions and further elaborate on the cancer-promoting properties of the fumonisins utilizing different experimental protocols. This must be considered against the background that most of the long-term rat studies conducted thus far provide evidence that the fumonisins are liver carcinogens. Of interest is that the NIH 31 diet (20.1% protein) was recently replaced by the NIH 2000 diet containing 14.5% protein to be used in long-term toxicity testing. 11 A similar argument could also be used when considering the hepatocarcinogenicity of FB1 in B6C3F1 female mice. 7 These mice are known to spontaneously develop adenoma and carcinomas over a period of 2 years 12 and the dose-dependent increase in the number of neoplastic lesions can be ascribed to the cancer-promoting properties of FB1 rather than to be considered as a complete hepatocarcinogen.
The liver cancer model utilizing male BD IX rats, therefore, provides a far better model to be utilized in determining risk assessment parameters, as more information is available regarding the underlying mechanisms of cancer induction. In a long-term study in BD IX rats, early neoplastic lesions were already present after 6 months,
13
while the cancer initiating and promoting properties have been well-defined, which proves, beyond any doubt, that FB1 is hepatocarcinogenic. Although the culture material of
In a study with purified FB1 fed to male Sprague Dawley rats, no hepatotoxic changes were observed in the liver of the rats fed a 50 mg FB1/kg diet representing an estimated intake of 4.7 mg FB1/kg bw/day over a period of 4 weeks. 16 Lesions characteristic of those induced by fungal culture material were obtained in the liver at a higher dosage (13.6 mg FB1/kg bw/day) diet with male rats being more sensitive than females. Studies in male Fisher rats at different locations using different diets indicated that very similar toxicological effects were induced in the liver either by incorporating FB1 in the diet or by gavage dosing. 17 –19 Mild changes were observed in the liver at 3.5 mg FB1/kg bw/day dietary level for 21 days, which is in agreement with the long-term study in BD IX rats. 15 In contrast, no hepatotoxic effects were reported in the liver at a dietary intake of up to 5.7 mg FB1/kg bw/day over 90 days, while kidney lesions were noticed at an intake of 0.62 mg FB1/kg bw/day with a NOEL of 0.21 mg FB1/kg bw/day. 20 The latter study is in accordance with the findings of the NTP study where male Fischer rats developed renal adenoma and carcinoma while no toxic lesions were obtained in liver at an exposure level of 6.6 mg FB1/kg bw/day over a period of 2 years. NOEL’s for kidney tumours in male Fischer rats was between FB1 intakes of 0.7 and 2.2 mg FB1/kg bw/day, while for nephrotoxicity it was 0.2 mg FB1/kg bw/day. 7,20 Based on the notion that nephrotoxicity is a prerequisite for renal carcinogenesis, this intake level was considered by the Joint FAO/WHO Expert Committee on Food Additives (JECFA) to obtain the provisional maximum tolerable daily intake (PMTDI) for fumonisins in humans. 21 However, a dose of 0.7 mg FB1/kg bw/day for 2 years induced kidney lesions without the induction of ATH, a precursor lesion of the renal tumours, which raised some doubts about the cell death/compensatory regenerative model for FB1-induced nephrocarcinogensis proposed by Dragan et al. 22 This is further complicated by the induction of proliferative lesions in the kidney due to the development of CPN as discussed above.
The possible role of different dietary constituents on the FB1-induced toxicological effects has been addressed recently.
5
The interaction between FB1 and dietary iron has been investigated, indicating that a protective effect was obtained against cancer promotion, presumably due to the mitogenic properties of iron.
23
In a subsequent long-term study, the role of low levels of dietary iron was re-evaluated in a discontinued FB1 feeding study.
24
A dual role of iron on FB1-induced hepatocarcinogenesis was proposed in that (i) iron overload enhanced the induction of hepatocyte nodules in the presence of FB1 while (ii) after removal of FB1, the continued iron supplementation impaired the progression of the hepatic nodules. The exact mechanism involved is not known, but the differential modulation of FB1 and iron on cell proliferation and oxidative damage could be of importance in altering the growth of preneoplastic lesions. The interaction between antioxidants and FB1-induced hepato- and nephrotoxicity has been investigated by monitoring the protective effect of royal jelly, obtained from worker honey bees, in Sprague-Dawley rats.
25
Co-treatment of the rat with the royal jelly significantly reduced the toxic effects of FB1 in the liver and kidneys in a dose-dependent manner. The protection was associated with a significant increase in the FB1-induced reduction in glutathione peroxidase and superoxide dismutase, while it also counteracted the increase in lipid peroxidation, presumably related to the free oxygen radical scavenging and antioxidant properties of the royal jelly. Garlic and cabbage extracts have been shown to protect against FB-induced developmental toxicity in Sprague-Dawley rats.
26
The extracts significantly reduced the number of skeletal malformations, presumably due to the presence of diallyl sulphide known to protect against oxidative damage.
27
Recent studies showed that herbal extracts countered to hepatotoxic and hepatocarcinogenic properties of the fumonisins in the liver of rats. Extracts of
Cancer initiation by FB 1
Genotoxicity
The genotoxicity of the fumonisins has been debated although it is generally accepted that it lacks DNA interactive reactivity in different short-term genotoxicity assays utilizing bacteria
31
–34
and
Role of oxidative damage
Oxidative DNA damage appears to be one of the major mechanisms proposed for disrupting membrane structures associated with an increased lipid peroxidation,
44
–46
which could explain the genotoxic effects of the FB1 via the formation of hydroxyl and peroxyl radicals. An
Cancer initiation in rat liver
Experimental evidence that FB1 possesses cancer-initiating activity is summarized below: Studies using male BD IX, Fischer 344 and Sprague Dawley rats showed that FB1 exhibits cancer-initiating properties.
17,19,35,51,52
The induction of a hepatotoxic effect, as discussed for the induction of hepatocarcinogenesis, is a prerequisite for cancer initiation. An apparent no-effect threshold related to hepatotoxicity and hepatocyte regeneration exists for cancer initiation, which in turn is determined by the dosage and the duration of exposure. In this regard, a low nontoxic dose effects initiation in the absence of a hepatotoxic effect.
15
The inhibition of hepatocyte proliferation and the induction of apoptosis seem to play a critical role in the genesis and/or the threshold of the initiating event.
17,53
FB1 initiates cancer in rat liver similarly to the genotoxic carcinogens, by inducing “resistant” hepatocytes with a dependence on cell proliferation, although the kinetics differ. Cancer initiation was only effected after prolonged feeding in the diet, and depending on the dosage, could be induced over a 14-day period.
17
Phenotypically, these initiated cells develop into eosinophilic clear cell foci and nodules that stained positively placental g glutamyl-transferase (PGST) and γ-glutamyl-transferase (GGT). The absolute level of events (DNA mutations, etc.), resulting in initiation, occurs at a far slower rate when compared to genotoxic carcinogens. Similarly to genotoxins, cell proliferation associated with hyperplasia failed to enhance, while stimulation of regenerative cell proliferation enhanced the cancer initiating potency of FB1.
19
Irreversibility of cancer initiation by FB1 was demonstrated as a 5-week exposure and resulted in liver adenomas after 1 year in male Fischer 344 rats.
3
Different cancer promoting stimuli, 2-AAF/PH, 2-AAF/CCl4 and PB promote the development of altered preneoplastic hepatic lesions.
35,51
FB1 lacks peroxisome proliferation activity,
54
while promotion of spontaneous-initiated cells in older rats
51
seems not to play a role in FB1-induced carcinogenesis.
The “Effective Dosage Level” for cancer initiation in male Fisher rats fed the AIN 76 diet was determined and interesting aspects regarding exposure levels and initiation were noticed. In this regard, a total dosage of 30 mg FB1/100 g body weight failed to initiate cancer over a 7-day period, whilst the same dosage effected initiation over a period of 21 days. A biphasic effect of FB1 on cell proliferation is proposed in the liver that results in the genesis of altered hepatic lesions, ultimately leading to cancer. A critical balance between compensatory or regenerative cell proliferation, resulted from the hepatotoxicity and the inhibitory effect on cell proliferation, thereof seems to exist, which will eventually determine initiation by FB1 (Figure 1
).
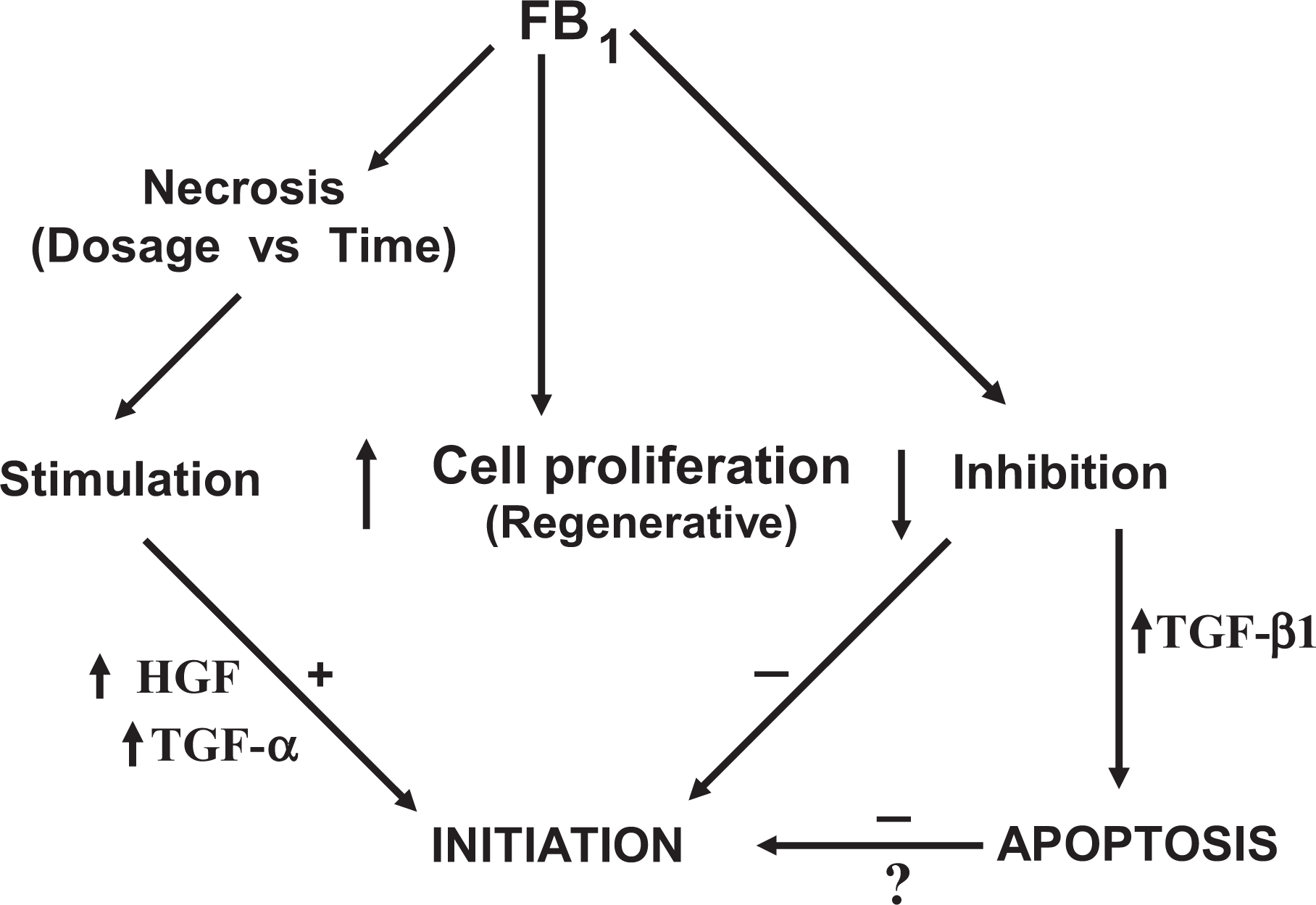
The biphasic response of fumonisin B1 (FB1) on cell proliferation in relation to cancer initiation as a function of the dose used and time of exposure. The over expression of several growth stimulating and inhibitory factors are effected, which are likely to play a determining role in the induction of the altered resistant hepatocytes. 17,53
The cancer-initiating potential of the apparent non-genotoxic FB1 is of interest as it is normally accepted that this stage of cancer induction is associated with a mutation-like event. 55 At present, very little is known about the nature of the initiating step of either genotoxic or non-genotoxic carcinogens, although many other cell parameters such as protein and RNA can be altered independently from changes to the DNA. Apoptosis is also known to play a determining role in the outcome of the initiating event, as initiated cells are more prone to undergo apoptosis. 56 It is known that FB1 induces necrosis as well as apoptosis in the liver that could determine the outcome of cancer initiation. 51,53
Underlying mechanisms during cancer promotion
Numerous studies have been conducted to investigate the cancer-promoting properties of FB1, including
FB1 disrupts lipid metabolism in the cell involving cholesterol, phospholipid, sphingolipid and fatty acid (FA) biosynthesis. The role of sphingolipids in the development of kidney cancer has been reviewed in detail 22,58,66 and will not form part of the current discussions. The role of these changes in the cancer-promoting properties in the liver and kidneys has also been debated as the proposed mechanisms were derived from different schools of thought. Irrespective of the actual mechanism involved, the interaction between the underlying biochemical events involved in determining cell survival has been discussed in detail. 67,68 The key determinants in the survival of early preneoplastic cells are ceramide and arachidonic acid (C20:4n-6), known to be important regulators of cell proliferation and apoptosis. Underlying these mechanisms, FB1 also disrupts the oxidative status of cells that are also important modulators of apoptosis. When considering the liver cancer hypothesis based on the resistant hepatocytes model, the modulation of these events is responsible for the selective proliferation of the resistant hepatocytes. It was shown that FB1 closely mimics events that prevail in these preneoplastic events that are associated with their selective outgrowth and development in neoplasia. Similar events induced in normal cells resulted in the inhibition of growth and the subsequent induction of apoptosis, whereby it creates the selective stimulus during cancer promotion. This hypothesis was studied in detail in the liver, whereas the early events associated with the development of neoplastic lesions in the kidney are not well-characterized. The FA/sphingolipid interactive pathways (Figure 2 ) in relation to oxidative stress reflect alterations related to membranal changes affecting subsequent signal transduction pathways involved in cell survival. However, these pathways are proposed to exist in the liver, the outcome of which will differ when considering the differential effects on normal and pre-neoplastic cells.

The lipid liver cancer model was based on studies regarding the role of lipid metabolism in liver cancer development
69,70
during which different lipid parameters were monitored in hepatocyte nodules over a period of 9 months. In short, the decrease in the PC/PE phospholipid ratio, impairment of the delta 6-desaturase and a decrease in the oxidative status in hepatocyte nodules were identified as the key components responsible for the altered growth characteristics in these lesions. These changes closely mimicked regenerating liver, which further provides proof that they are associated with cell proliferation. Persistent changes in the nodules include an increase in cholesterol and PE, which was associated with an increase in C18:1n-9 and C18:2n-6 while C20:4n-6 decreased in PC and increased in PE, resulting in an increase in C20:4n-6 PC/PE ratio. The end products of both the n-6 and n-3 FA pathways, C20:5n-6 and C22:6n-3 also decreased in PC, resulting in a reduction in the total PUFA and long-chain PUFA levels and oxidative status in the nodules. FB1-induced effects in the liver closely mimic the lipid changes in hepatocyte nodules and are therefore likely to selectively stimulate the growth in hepatocyte nodules, suggesting that similar changes in the normal surrounding tissue resulted in an adverse effect on cell viability. Altered lipid changes induced by FB1 include: Impaired delta 6-desaturase, leading to the accumulation of C18:1n-6, C18:2n-6 and a reduction in LC PUFA levels in the cell.
71
Increased PE resulting in an increased C20:4n-6 PE/PC ratio. Inhibition of ceramide synthase with accumulation of sphinganine and decrease in sphingomyelin. Changes in these parameters differ in the subcellular fractions with the increased PE noticed in the plasma membrane, mitochondria and microsomes, while SM was decreased in mitochondria. The C20:4n-6 PE/PC ratio was altered in all the membranal fractions.
72
Differential effects of these lipid-associated changes on the viability of normal and preneoplastic cells provide the basis for cancer promotion. The association of the lipid changes and the selective induction of growth factors and signaling molecules are likely to inhibit cell proliferation and induce apoptosis in normal cells whilst inducing cell proliferation in preneoplastic cells.
53,73
This inhibitory effect on cell proliferation was demonstrated by FB1 in proliferating hepatocytes in normal liver following partial hepatectomy.
17
In primary hepatocytes, the disruption of prostaglandin metabolism plays a key role in the inhibition of growth stimulatory responses effected by FB1.
74,75
The disruption of sphingolipid, phospholipid and fatty acid metabolism could be related to several downstream events associated with cell proliferation and apoptosis. The FB1-induced overexpression of c myc and TGF-β1 was proposed to be involved in the selective stimulation of the outgrowth of initiated cells resistant to the growth inhibitory effects of TGF-β1 via the regulation of cyclin D1 and retinoblastoma.
76
Studies in rats also showed that FB1 inhibits the proteosomal breakdown of cyclin D1 in neoplastic and preneoplastic cells by activating protein kinase B (Akt), which stimulates the phosphorylation of cyclin D1 via glucacon synthase kinase 3β (GSK-3β). The increased activity of cyclin D kinase and the hyperphosphorilation of retinoblastoma in pre-cancerous liver lesions provide a working model for FB1-induced heptacarcinogensis in rat liver. The activation of Akt is also known to inhibit apoptosis, which will further stimulate the outgrowth of these precursor lesions. Effect of FB1 on cellular oxidative status played an important differential role in the regulation of apoptosis and cell proliferation between normal and neoplastic cells. A recent study showed that FB1 modulated the cellular redox balance in the liver by altering oxidative enzymes and redox-sensitive signaling molecules including the GSH levels, MAPK activation, heat shock proteins, ERK and p38.
29,42
When considering the underlying mechanisms associated with cancer promotion of the fumonisins, the disruption of biochemical pathways associated with different cell growth parameters and aspects regarding the recently derived epigenetic mechanisms of cancer development should also be considered. This theme also links closely to the biological threshold hypothesis discussed above and the current paradigm utilized for risk determination of the fumonisins. Epigenetic mechanisms of cancer development have been recognized for almost 3 decades and are unrelated to genetic variation or mutations generally associated with genotoxic carcinogens.
77
It is currently recognized that epigenetic events are perhaps more common than genetic changes and play an important role in the modulation of functional pathways that are key to neoplastic development. These mechanisms include promoter DNA methylation, histone modification and RNA interference to name a few and provide new opportunities for cancer prevention. It would appear that epigenetic events occur very early in neoplasia and that many tumour-suppressor genes in human neoplasia are inactivated by epigenetic mechanisms.
78,79
Aberrant methylation has been associated with many cancers including colon, oesophageal, liver and lung. The disruption of the folate receptor by FB1 and the subsequent aberrant folate metabolism has been shown to induce neural tube defects (NTD) in mice.
80
FB1 modifies the expression of the folate receptor and folate carrier in HepG2
81
and folate uptake in Caco-2 cells.
82
The disruption of DNA methylation due to folate deficiency and FB1-induced carcinogenesis therefore should be investigated as a possible epigenetic mode of action that should impact on the current risk assessment paradigm for fumonisins.
Interactive mechanistic and biological approaches
Threshold effects related to non-genotoxic and synergistic effects
Separate parameters are used to distinguish between genotoxic and non-genotoxic chemicals when assessing risk to humans.
83
For non-genotoxic carcinogens, a threshold level exists which permits the derivation of a no-observed-effect-level (NOEL). With the introduction of a safety factor, a permissible exposure level is derived below which no relevant human risk is anticipated. For genotoxins, it is generally accepted that a non-threshold level exists although a whole array of threshold effects has been suggested depending on the type of mechanism that prevails.
84
Four basic types of thresholds have been distinguished: a linear non-threshold (LNT) model for genotoxic carcinogens, genotoxic carcinogens where an LNT model is used as a default when the precise nature of the dose response has not been established, genotoxic carcinogens where a practical threshold is likely based on the mechanisms involved and non-genotoxic carcinogens where a perfect threshold exists which is associated with a NOEL as described above.
However, the application of LNT models has been questioned based on mechanistic arguments implying the existence of biologically meaningful threshold dose-response effects for both DNA- and non-DNA-reactive chemicals. It is suggested that a diversity of methods for carcinogenic risk extrapolation to low doses should be applied based on the mode of action.
85
One exception is when the interaction between the carcinogen and cellular target, such as the DNA, represents a single event and in such a case a non-threshold theoretically does exist. However, when a single hit–single target is shown to exist in
When considering the carcinogenic properties of the fumonisins, different scenarios have to be considered regarding threshold effects. It is evident that, although the fumonisins apparently lack direct DNA reactivity in various
Involvement in human diseases
The association between the consumption of
Maize as a risk factor for OC
Maize seed entered Africa during 1500 AD and gradually replaced sorghum and millet as the dominant crop in southern Africa, specifically along the eastern coast in the so-called maize belt, including countries such as South Africa, Zimbabwe, Zambia, Kenya and Ethiopia. 100 A significant correlation exists between the incidence of OC and the maize supply in these regions, while no association was obtained between the supply of sorghum and millet. 101,102 An increase in OC patterns in the former Transkei region of the Eastern Cape Province was first noticed by Rose 103 in 1940 to 1950. The consumption of maize meal and homegrown maize has been identified as a risk factor for the development of OC in case-control studies. 104 –106 Additional risk factors for the development of OC associated with an underlying nutritional deficiency due to the consumption of maize as a major dietary staple have been suggested. These include deficiencies of vitamins, selenium, folate, magnesium and molybdenum. 104,107,108 A specific dietary pattern has been identified in a hospital-based case-control study by principal component analyses, which include a diet of maize, imifino and dry beans as a risk factor for OC development. 106 Two other diet patterns were shown to have a protective effect including dietary pattern 1: sorghum, potted vegetables, fruit, meat and green leafy vegetables and dietary pattern 3: which is mainly wheat based. However, components of the latter two protective diets, specifically wheat, is known not to feature as major dietary staple, while sorghum is not utilized as a dietary staple in the low and high incidence rural districts (Centane, Butterworth, Lusikisiki and Bizana) of the former Transkei region. 109 As the study showed that rural residents were more likely to develop OC, the implication of specific dietary patterns associated with OC is confounded as the study only reflected differences in dietary patterns between the urban and rural populations. This is of particular interest when using a food frequency questionnaire in conducting retrospective nutritional surveys to assess the intake patterns of specific dietary components, which is subjected to recall bias. The identification of specific dietary patterns in a population 106 without quantifying food intake also could provide erroneous information, especially in a population where sorghum and wheat do not form part of a rural diet. 109 This is further emphasized regarding the finding that bean consumption protects against OC development in males but not females, which again could be related to portion sizes consumed. It also contrasted a previous finding 110 that the consumption of beans is associated with OC development.
As the case-control study reflected a high proportion of urban residents (patients [62%] and controls [70%]), extrapolation of findings to OC cancer patterns in rural areas seems inappropriate and findings are subjected to analysis bias and confounders known to exist when conducting hospital-based studies. 111 At present, all the studies conducted in this region have numerous methodological errors due to the type of dietary assessment methods used. The use of culture-specific and validated methods would have been more appropriate in assessing specific dietary patterns associated with OC. 112 The finding that homegrown maize is a risk factor for OC 106 has been reported previously. 104 This strengthened the association of OC with fumonisin contamination of maize and the possible modulating role it could play in the development of the disease. 88,113
Mouldy maize as a risk factor for OC
The consumption of mouldy maize was found not to be a risk factor for OC in Transkei although this finding is clouded by numerous confounding factors.
106
Several aspects should be considered when trying to assess the level of mouldy maize consumption in the Transkeian population. As females prepare the food, including the sorting and washing of the homegrown maize, it is difficult for the males to assess whether they consumed mouldy maize or not. The sorting and washing procedures will also differ from household to household as no standardization has been implemented, which could have a important effect on the level of consumption of the fumonisins. It will therefore be difficult to assess the intake of mouldy maize and any attempt to use visualized
Other diseases
Apart from a possible involvement in OC, fumonisins have also been implicated in the development of liver cancer in studies in China where the levels in maize were monitored over a period of 3 years. 99,116 As in the case of OC, the interaction between mycotoxins including aflatoxin B1 (AFB1), deoxynivalenol and the algal toxins were considered. An interaction between AFB1 and FB1 in the development of liver carcinogenesis in experimental animals has been shown. 118,119 The association between the fumonisins with neural tube defects (NTD) became of interest as these mycotoxins disrupt the folate receptor in cells. 120 –122 The role of sphingolipids and cholesterol, major constituents of lipid rafts associated with the folate receptor, is critical for the early embryonic development. 123 FB1 was shown to induce NTD in mice, presumably via the disruption of sphingolipid metabolism and folate uptake. 80 The induction of NTD was partly prevented by folate supplementation. A recent study showed that fumonisin exposure increases the risk of NTD in humans. 124 The high incidence of NTD closely mimics that of the OC incidence, when considering the incidence levels in Transkei and China. 121
Perspectives on risk assessment of fumonisins as food contaminants
Differences exist in the implementation of risk assessment parameters of toxins and carcinogens between developed and developing countries, especially in remote microenvironments in developing countries where certain foods are used as a sole dietary staple. The health issues associated with these microenvironments are largely ignored when considering global trade between industrialized countries although they could negatively impact on health risk issues regarding food-borne toxins and carcinogens. In South Africa, differences exist in the health risk posed by the fumonisins and are determined by differences in the maize consumption patterns, which vary, not only between the different ethnic groups, but also between rural subsistence farmers from rural areas compared to urbanized populations. An interactive model (Table 1
) has been developed to compare the impact of maize intake and fumonisin contamination levels on the PMTDI.
68,125
With respect to the maize availability to the different population groups, the dietary intake patterns of commercial and homegrown maize vs imported maize need to be considered: Homegrown maize
Interactive association between maize intake, fumonisin B (FB) contamination levels and the resultant PDI (µg FB/kg bw/day) values a
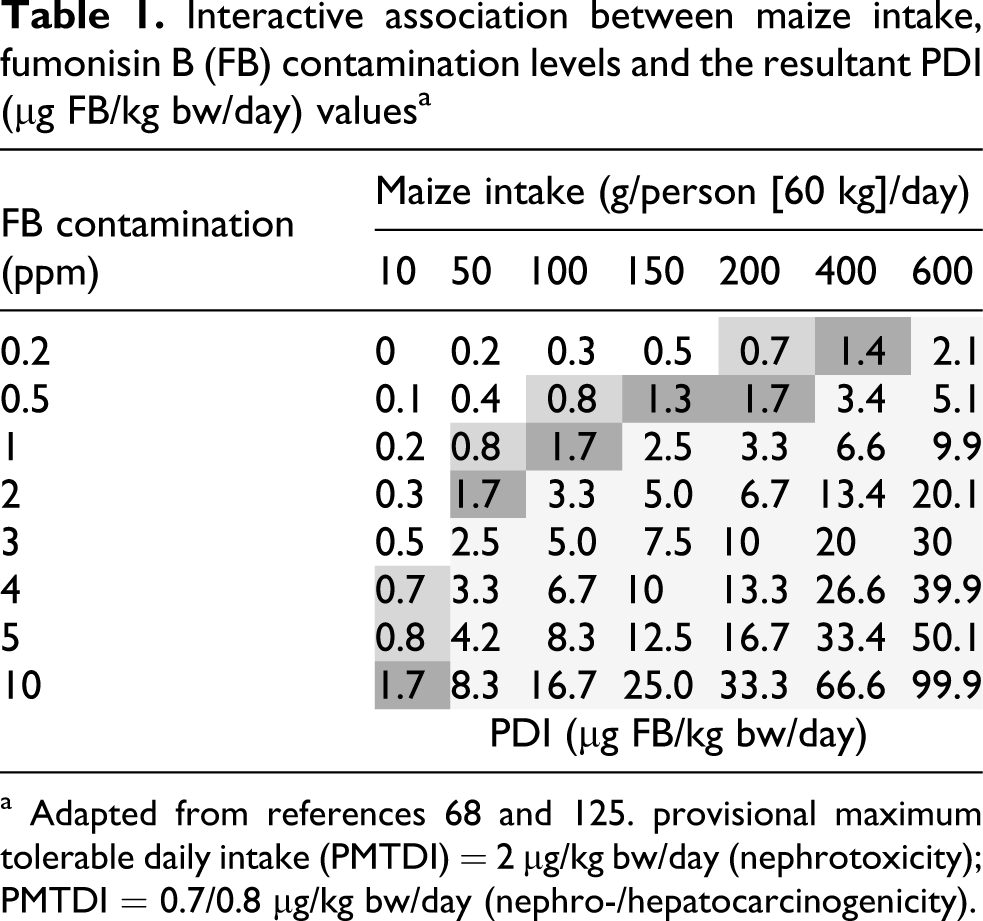
a Adapted from references 68 and 125. provisional maximum tolerable daily intake (PMTDI) = 2 µg/kg bw/day (nephrotoxicity); PMTDI = 0.7/0.8 µg/kg bw/day (nephro-/hepatocarcinogenicity).
Fumonisin contamination of homegrown maize has been linked to the development of OC
86
and more recently to the development of NTD
121
in population groups using maize as a monocereal staple diet. Fumonisin exposure in rural settings in South Africa reaches levels that are far above the PMTDI (2 μg/kg bw/day) level set by JECFA.
126,127
The major determinant, however, appears to be the maize consumption patterns in different so-called “hotspots” of exposure. Most of the epidemiological studies focused on these areas to assess possible interactions of fumonisin exposure to a specific disease pattern in humans. In this context, established risk assessment parameters failed to adhere to the exact principles for which they have been established, i.e. to safeguard humans to the adverse effects of these mycotoxins. Most of the populations in developing countries consuming maize as a monocereal staple diet also lack sufficient levels of micronutrients that may enhance their susceptibility to adverse effects of the fumonisins. These aspects are not included when setting risk assessment parameters and apparent probable daily intake (PDI) levels of the fumonisins in these regions, and values of 5−10 times above the PMTDI are common. It is not known at present what effects the long-term exposure to fumonisins at these levels would have on the health status of specific populations at risk. The need to perform studies to quantify the exposure patterns as well as determining the nutritional status of the people at risk is self-evident. Proper intervention measures to reduce exposure to match the established risk assessment parameters is therefore of critical importance in these regions. The importance of setting new PMTDI levels for fumonisins to safeguard human population groups in developing countries is clearly indicated. A maximum tolerable level (MTL) of 0.2 mg FB/kg maize as suggested by Marasas
128
appears to be remarkably close to a safe contamination level of maize in a South African setting where adults may consume up to 500 g and more maize per day. In children, however, the situation is far worse and intake varies between 169 to 541 g/child/day according to a recent survey in South Africa (http://www.sahealthinfo.org/ nutrition/foodconsumption.htm) resulting in PDI levels (Table 2
) that even further exceed the PMTDI level proposed by JECFA.
21
Commercial vs imported maize
Maize consumption patterns of various population groups in selected countries

PDI: probable daily intake, FB: fumonisins, JECFA 21 : Joint FAO/WHO Expert Committee on Food Additives, GEMS: Global Environment Monitoring System.
The commercial production of white maize, normally used as food, varies between 1.25 and 6.37 million tons per annum in South Africa. 129 A survey of 6 crop years (1989 to 1994) of FB contamination levels in white maize cultivated in the commercial maize production areas in South Africa recorded mean levels of 0.25 to 0.70 mg/kg maize. Imported maize from the United States contained considerably higher FB levels for the 1991 and 1992 crop years and reported to be 1.13 and 1.05 mg FB/kg, respectively (levels reported incorrectly due to a printing error in [ref 129]). If such imported maize enters the human food chain PDI, levels will increase by a factor of 2- to 3-fold as compared to the consumption of South African maize. This is of particular importance during dry seasons when large quantities of maize need to be imported, some of which is channeled into the human food chain. As commercial South African maize contains low levels of FB compared to maize supplied on the international markets, imported maize could exaggerate the risk due to FB intake in the local population. When considering impoverished rural societies depending on maize as a sole dietary staple and a reduced intake of micronutrients, a “catch-22” situation arises, on the one hand with people facing starvation while on the other hand consuming maize containing FB levels that could have adverse effects on their health. Once again, children appear to be the most vulnerable group of the population and PDI values of 3- to 5-fold above those calculated for adults are obtained (Table 2).
Risk assessment parameters for the fumonisins
Different models exist to determine the risk of toxins and carcinogens present in a food crop intended for human consumption. These include the no-observed-effect levels (NOEL)/safety factor approach 137 and computerized models such as the two-dimensional Monte-Carlo simulation 138 proposed recently, the Moolgavkar-Venzon-Knudson (MVK) two-stage, clonal expansion model 139 or dose-response models for tumour incidence. 21 The WHO has adapted the NOEL/Safety factor approach to determine the provisional maximum tolerable daily intake (PMTDI) for natural toxicants. 137 For toxicants, a safety factor of 100 is normally adopted, which consists of a factor 10 for the extrapolation between animal to humans and 10 to account for the difference in sensitivity within the human population with respect to a specific biological adverse effect. The numerical size of the safety factor used may differ depending whether a compound is classified as having toxic effects or upon long-term exposure could result in the induction of a carcinogenic response. In general, larger safety factors are used for carcinogenic compounds depending on the mechanism of cancer induction related to epigenetic (non-genotoxic compounds) or genotoxic mechanisms. 140
A meeting of JECFA on the fumonisin B mycotoxins mainly considered the toxicological effects in experimental animals, possible associations with respect to human diseases as well as food contamination levels in different countries worldwide. 21 A PMTDI level of 2 µg FB/kg body weight/day was calculated based on the NOEL of nephrotoxicity in male Fischer rats using a safety factor of 100. The NOEL for nephrotoxicity was obtained from a long-term chronic-feeding study (2 years) in male Fischer rats by the National Toxicology Program (NTP) in the United States and was equivalent to a dietary level of 5 mg FB1/kg diet resulting in a FB1 intake of 200 µg/kg body weight/day. 6 However, when considering the carcinogenicity of FB1, the NOEL for nephrotoxicity of 200 µg/kg bw/day is below the no-effect threshold for carcinogenicity (700 µg/kg bw/day) for the induction of adenoma and carcinoma in the kidneys. The NOEL for nephrocarcinogenicity and the hepatocarcinogenicity are very similar and vary between FB1 intakes of 700 µg to 800µg/kg bw/day, respectively. A tolerable daily intake (TDI) of 0.8 µg FB/kg bw/day, using the NOEL for hepatocarcinogenicity, and a safety factor of a 1000 has been proposed, 54 which is very similar to the 0.7 µg/kg bw/day using the NOEL for nephrotoxicity and a similar safety factor. The latter approach resulted in a TDI that is in the order of 3 times lower than the PMTDI using the NOEL for nephrotoxicity.
At present, it is not known whether a PMTDI, based purely on the nephrotoxicity whilst ignoring the carcinogenic properties, will safeguard humans from the adverse effects of the fumonisins. Therefore, at present, two safety limits exist i.e. a lower limit 0.7/0.8 µg kg bw/day (lower risk) that focuses on the carcinogenic properties of the fumonisins and a higher limit of 2 µg/kg/day (higher risk) that mainly focused on the nephrotoxicity of the fumonisins and represents a level below the threshold for cancer induction. However, the lower TDI based on carcinogenicity of the fumonisins could have a far more adverse economic impact on international trade and industry, affecting the entire maize industry. 129 In developed countries exporting large quantities of maize, a low TDI could severely affect the maize industry whereas a high TDI may be a health risk for populations with a high maize intake in developing countries importing maize. Different scenarios regarding risk assessment of the fumonisins exist, which could either underestimate the risk posed to human health or overemphasize the risk that would negatively impact on international trade and the maize industry.
Risk paradigms of FB1
FB1 causes diverse toxicological effects in a variety of animal species both experimentally and under natural conditions.
1,2
Recommended maximum residue limits for fumonisins in maize.

Conclusions and recommendations
Several studies exist regarding the possible role of the fumonisins in the development of human diseases of which oesophageal cancer, liver cancer and the development of neural tube defects received the bulk of attention. None of the epidemiological studies conducted thus far could either prove or disprove a causative relationship of the fumonisins with any of the diseases. The numerous determinants hampering studies to evaluate the role in human health include: the use of validated population-specific questionnaires; problems that exist in using food frequency and 24-hour recall questionnaires in populations that are in transition regarding their location, lifestyle and types of food consumed; and the lack of specific methods or biomarkers to monitor exposure at an individual level. Controversies regarding the toxic and carcinogenic properties of the fumonisins also complicate the establishment of risk assessment parameters. These include: the target organ for the toxic and carcinogenic characteristics of the fumonisins; site-specific mechanisms for cancer induction; genotoxic vs non-genotoxic effects; and the existence of a threshold. The approach for fumonisin risk assessment applied by JECFA was that the mycotoxins are of the non-genotoxic type and therefore utilized a theoretical toxic regenerative cell proliferative model for cancer induction supporting the non-linear threshold model for chemical carcinogenesis.
A PMTDI of 2 μg FB/kg bw/day has been derived for the fumonisins using a NOEL for nephrotoxicity (200 µg/kg bw/day) and a safety factor of 100. As this is well below the NOEL for nephrocarcinogenicity (700 µg/kg bw/day), other mechanistic considerations and higher safety factors related to carcinogenicity should also be considered. Further to this debate is the possible interactive role of nephropathy that prevailed in about 90% of the rats due to the high-protein diet used in the chronic study in male Fischer rats. 6
Several aspects regarding the regulation of FB in maize are of importance. Setting tolerance levels of FB in maize could either negatively impact on human health in certain population groups or severely disrupt the maize food chain. The setting of realistic risk assessment parameters in the South African context where neither the health nor the socioeconomic aspects regarding maize production will be compromised requires urgent attention. Aspects regarding international trade and economic realities have also to be taken into account in order to provide realistic control measures. When considering the maximum tolerable levels (MTLs) of fumonisins in maize, the following are of importance: Two types, an ‘economic’ and a ‘scientific or health’ MTLs exist. If the ‘scientific’ MTL is too low, it will destroy the maize industry and people will starve because of the absence of maize rather than become ill because of the consumption of contaminated maize if the ‘economic’ MTL is too high. However, appraisal of the ‘scientific’ MTL makes it abundantly clear that the majority of the South African population, especially children, would be at risk when utilizing maize as a sole dietary staple. Two different scenarios exist with respect to risk of fumonisin exposure in developed vs developing countries. It becomes evident that MTLs that safeguard human health in the one does not necessarily do so in the other. A population that is at the greatest risk with respect to fumonisins in maize is subsistence farmers and children in rural Africa who consume homegrown maize. They have the highest maize intakes and also consume the most highly contaminated maize. Yet, they are unaffected by MTLs set globally to regulate international trade, e.g. a MTL of 1 mg/kg as set by Switzerland will allow a PDI greater than the PMTDI set by JECFA if the intake profiles exceed 100 g/person/day (Table 1). Protection against fumonisin intake and the induction of an adverse effect such as cancer and possibly NTD can be achieved more effectively by other means than MTLs e.g. Effective intervention programmes in reducing fumonisin contamination of maize minimizing FB exposure and the associated risk. Recent studies indicated that applying a simple culturally acceptable intervention method reduces the FB levels on homegrown maize up to 80%.
146
Fortification of maize products with folic acid to counteract the disruption of the folate receptor by fumonisins.
Footnotes
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
