Abstract
Considering the potential adverse effects of selenium and iodine deficiencies, and frequency of intensive but improper use of insecticides, this study was designed to evaluate the effects of a pyrethroid insecticide, fenvalerate, on the oxidant/antioxidant status of liver using a rat model of iodine and/or selenium deficiency. The study was conducted on eight groups of 3-week old Wistar rats. Iodine and/or selenium deficiency was introduced by feeding the animals with a diet containing <0.005 mg selenium/kg and/or supplying with 1% sodium perchlorate containing drinking water for a period of 7 weeks. Fenvalerate exposure (100 mg/kg/d, i.p., for the last 7 days) in normal rats increased hepatic glutathione peroxidase activity and lipid peroxidation, decreased glutathione content, but did not change the activities of catalase or any of the superoxide dismutase forms; in iodine-deficient animals caused only the elevation of lipid peroxidation; in selenium-deficient animals and in combined iodine/selenium deficiency decreased glutathione peroxidase, increased catalase activities and lipid peroxidation, and decreased all the forms of superoxide dismutase activity only in combined deficiency. These results suggested that fenvalerate is an oxidant stress inducer in rat liver, and its potential effects on pro-oxidant/antioxidant balance may also be important for human populations, particularly with iodine and/or selenium deficiencies.
Keywords
Introduction
Pyrethroid insecticides are synthetically derived chemical forms of naturally occurring pyrethrins that have higher insecticidal activities and lower mammalian toxicities. They are intensively used worldwide for more than 30 years, accounting for almost 30% of the overall insecticide market. 1 Photostability, high efficacy at low concentrations, ease of disintegration and limited soil persistence are the other advantages of their use.2–4 Pyrethroids, including fenvalerate ((RS)-alpha-cyano-3-phenoxybenzyl (RS)-2-(4-chlorophenyl)-3-methylbutyrate), are potent neurotoxicants in vertebrates and invertebrates acting directly on the axons by interference with the sodium channel.5,6 They have recently been linked to endocrine disruption,7–9 and induction of hepatotoxicity was reported in several studies.10,11 Involvement of reactive oxygen species (ROS) in the toxicity of various pesticides has been suggested, including organochlorine12,13 and organophosphate derivatives,14–16 but the available data on pyrethroids are not comprehensive and rather controversial.17–22 Furthermore, the effects of pyrethroids on pro-oxidant/antioxidant balance in trace element deficiency states have not been studied. However, in a previous study, we have shown that fenvalerate induces marked alterations in thyroid status in both normal and iodine- and/or selenium-deficient rats. 23
Selenium is an integral component of several major metabolic pathways including antioxidant defense systems and thyroid hormone metabolism. It is an essential constituent of a number of enzymes that have mainly antioxidant functions24,25 and thus involves in the regulation of cellular redox equilibrium. 26 Therefore, selenium-deficient animals may be more susceptible to oxidative stress. Selenium is also required for appropriate synthesis, activation and metabolism of thyroid hormones, because iodothyronine 5’-deiodinases (DIOs) are selenoenzymes that catalyze the metabolic conversion of thyroxine (T4) to the major biologically active hormone, 3,5,3’-tri-iodothyronine (T3).25,27 Selenium deficiency impairs T4 conversion to T3, and selenium supplementation decreases plasma T4 levels, increases DIOs activity and improves the conversion of T4 to T3.28,29 Iodine is an essential component of both T3 and T4. Iodine deficiency is a recognized nutritional risk factor for thyroid dysfunction (hypothyroidism) and impaired mental and physical development, and severe iodine deficiency is the main cause of endemic goiter and cretinism in several geographic parts of the world.30,31 Combined iodine and selenium deficiency exacerbates hypothyroidism and is the cause of myxoedematous cretinism. 32 However, both selenium and iodine are inadequately available for man and livestock in many parts of the world. 33
Considering the potential adverse effects of iodine and selenium deficiencies, and taking into account the frequency of intensive but improper use of insecticides, this study was designed to evaluate the effects of the widely used pyrethroid insecticide, fenvalerate, on the hepatic pro-oxidant/antioxidant status on a rat model of iodine and/or selenium deficiency.
Material and methods
Chemicals
Technical grade fenvalerate was from Koruma Tarım (İzmit, Turkey) and had a purity of 92%. All other chemicals and reagents used were obtained from Sigma (St Louis, Missouri, USA) or Merck (Darmstadt, Germany). Selenium-deficient diet (<0.005 mg selenium/kg) was supplied by SAFE (Augy, France).
Animals, diet, and treatment
Three-week-old male Wistar rats were obtained from Hacettepe University Experimental Animal Laboratory. The animals were divided randomly in eight groups of eight each, and each group was housed in plastic cages with stainless-steel grid tops. The cages were maintained in a room with controlled temperature (23°C), humidity (50%), and a 12-hour light-dark cycle. Feeding period was 7 weeks, and animals were allowed free access to diet and water. Body weights (bw) were monitored weekly.
Experimental groups
(1) Control group (C) was fed with regular diet (~0.15 mg/kg selenium) and drinking water. (2) Control-fenvalerate group (CF) was fed with regular diet and drinking water and treated with 100 mg/kg/d, i.p., fenvalerate (~1/3 LD50) during the last week of the feeding period. (3) Iodine-deficient group (ID) was fed with the same regular diet and received 1% sodium perchlorate-containing drinking water to introduce iodine deficiency as described earlier. 34 (4) Iodine-deficient-fenvalerate group (IDF) was fed with regular diet, received 1% sodium perchlorate-containing drinking water, and treated with 100 mg/kg/d, i.p., fenvalerate during the last week of the feeding period. (5) Selenium-deficient group (SeD) was fed with selenium-deficient diet containing ≤0.005 mg/kg selenium and received normal drinking water. (6) Selenium deficient-fenvalerate group (SeDF) was fed with selenium-deficient diet, received drinking water, and was treated with 100 mg/kg/d, i.p., fenvalerate during the last week of the feeding period. (7) Iodine and selenium-deficient group (ISeD) received selenium-deficient diet and 1% sodium perchlorate-containing drinking water. (8) Iodine and selenium deficient-fenvalerate group (ISeDF) was fed with selenium-deficient diet, received 1% sodium perchlorate-containing drinking water, and was treated with 100 mg/kg/d, i.p., fenvalerate during the last week of the feeding period (Table 1 ).
Experimental groups, diet and treatment

Abbreviations: C: Control, CF: Control-fenvalerate group, IDF: Iodine-deficient-fenvalerate group, SeD: Selenium-deficient group, SeDF: Selenium deficient-fenvalerate group, ISeDF: selenium deficient-fenvalerate group.
a Feeding period: 7 weeks.
b Length of fenvalerate treatment: 7 days (last week of feeding period); route: i.p.
The animals were handled humanely and with regard for alleviation of suffering, and the study was approved by the Hacettepe University Ethical Committee.
Twenty-four hours after the last dose of fenvalerate, animals were weighed and sacrificed by decapitation under thiopental anesthesia. Livers were removed, immediately frozen, and kept at –80°C until analysis.
Preparation of tissue homogenates
Hepatic tissue homogenates were prepared in a volume of ice-cold buffer containing tris (10 mM), diethylenetriaminepentaacetic acid (1 mM), and phenylmethanesulphonyl fluoride (1 mM; adjusted to pH 7.4) using a Teflon pestle homogenizer to obtain 10% (w/v) whole homogenate. After centrifugation of the homogenates at 1500 × g, 4°C, for 10 min, thiobarbituric acid reactive substances (TBARs) concentrations were measured in the supernatant. The rest of the supernatants were recentifugated at 9500 × g, 4°C for 20 min, and the antioxidant enzyme activities (glutathione peroxidase [GPx1], catalase [CAT], superoxide dismutases [SODTotal, SODCu,Zn, SODMn]) were determined in the supernatant. For the measurement of the glutathione (GSH) levels, the tissue homogenate was diluted with metaphosphoric acid (6%), centrifuged at 1500 × g, 4°C, for 10 min, and supernatant was used for the total GSH determination.
Measured parameters and methods
Antioxidant enzyme activities
The activity of CAT was measured spectrophotometrically at 240 nm as the decomposition of H2O2. 35 One unit of CAT activity was defined as the amount of enzyme required to decompose 1 μmol of H2O2 in 1 min.
The activity of GPx1 was determined spectrophotometrically using t-butyl hydroperoxide as the substrate and measuring the decrease in NADPH concentration at 340 nm which is proportional to the enzyme. 36 One unit of GPx1 activity was defined as the amount of GPx1 required to oxidize 1 μmol of NADPH per minute.
The activities of SODtotal, SODMn, and SODCu,Zn were assessed by monitoring auto-oxidation of pyrogallol at 420 nm. 37 One unit of SOD activity was defined as the amount of enzyme required to inhibit the rate of pyrogallol auto-oxidation by 50%. The specific activity for all enzymes was expressed in units per mg protein for tissues. The protein content of the samples was determined by the method of Lowry. 38
Lipid peroxidation and total glutathione levels
Lipid peroxidation (LP) levels were quantified measuring TBARs concentrations by a spectrofluorometric assay described by Richard et al., 39 and the level of TBARs was expressed as nanomoles of malondialdehyde per g tissue. The total GSH content in liver tissue was determined spectrofotometrically by the method of Akerboom and Sies 40 and the results were given as micromoles of GSH per g tissue.
Statistical analysis
Experimental data were analyzed with one-way analysis of variance (ANOVA) followed by the Student’s t-test using SPSS Software version 12.0 (SPSS Inc., Chicago, Illinois, USA). The p values <0.05 were considered significant. All results were expressed as mean ± SEM.
Results
Throughout the experiments, all animals appeared to remain healthy. There was no mortality, and no clinical signs of fenvalerate poisoning were evident in the treatment groups. Significant differences in the food intake were not observed between the groups. However, at the end of the 7 weeks, body weights of the fenvalerate-receiving groups (CF, IDF, SeDF, ISeDF), SeD and ISeD groups were significantly lower than that of control group, as well as their respective controls, that is CF < C; IDF < ID; SeDF < SeD; ISeDF < ISeD (Figure 1 ).
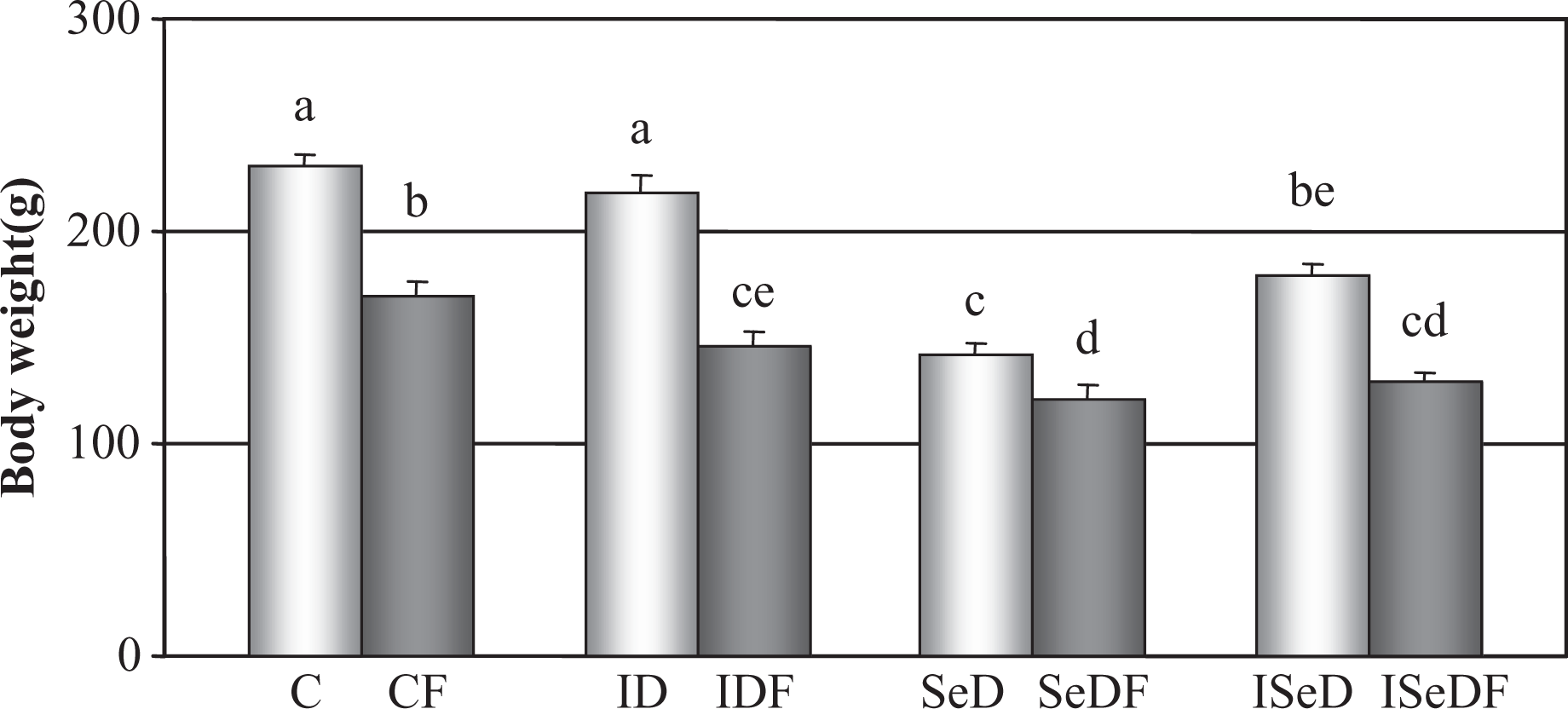
Body weights of rats in the study groups. Bars show the body weights of the rats at the end of the 7-week feeding period.
As shown in Figure 2 , a significant increase in GPx1 activity (~50%) and a significant decrease in CAT activity (~30%) were observed in iodine-deficient rats (p < 0.05). Selenium deficiency and combined iodine/selenium deficiency caused significant reductions in the activities of both enzymes (~95% in GPx1 and ~40% in CAT, p < 0.05). There were no alterations in any other parameters measured in iodine-deficient animals, whereas significant decreases in GSH content (~50%), and increases in TBARs levels (~70%-80%, p < 0.05) were observed in both SeD and ISeD groups (Figures 2 and 3 ).

Hepatic GPx1 and CAT activities in the study groups.

Hepatic TBARs levels and total GSH contents in the study groups.
Effects of fenvalerate on normal rats: Fenvalerate exposure in normal rats caused significant increases in GPx1 activity (~20%) and TBARS levels (~40%) and decreased GSH content (~35%) of the hepatic tissue (p < 0.05; Figures 2 and 3) but did not change the activities of CAT or any of the SOD forms (p > 0.05; Figure 2 and 4 ).

Hepatic SOD activities in the study groups.
Effects of fenvalerate on deficient animals: In fenvalerate-treated iodine-deficient animals (IDF), except for a significant elevation of TBARs (~30%, p<0.05), no alterations were observed in the other parameters compared to ID rats.
In selenium-deficient animals (SeDF) and in animals with combined iodine/selenium deficiency (ISeDF), fenvalerate exposure decreased GPx1 activity and increased TBARs concentrations compared to their respective controls (SeD and ISeD; Figures 2 and 3). Activities of CAT increased significantly compared to their respective controls (SeD and ISeD) but did not differ than the normal control value. GSH content did not change in SeDF group compared to C group or SeD but decreased significantly in ISeDF compared to both C and ISeD groups (Figures 2 and 3).
All three forms of SOD activities were found to be altered only in ISeDF group, with a significant decrease (~35%–50%, p < 0.05) compared to the respective control, ISeD group. Fenvalerate exposure in selenium deficiency caused a decrease only in the activity of SODCu,Zn compared to SeD rats (Figure 4).
Discussion
Oxidative stress is defined as an imbalance of cellular pro-oxidants and antioxidants, plays an important role in the modulation of several important physiological functions, but also accounts for changes that can be detrimental to the cells. 41 Currently, there is a growing interest to the environmental chemicals that can cause oxidative stress particularly in the endocrine system, 42 besides the association of oxidative stress with degenerative diseases including cancer. 43 The results of the present study indicates that the widely used pyrethroid insecticide, fenvalerate, causes a pro-oxidant/antioxidant imbalance in hepatic tissues of rats as shown by elevated TBARs concentration, decreased total GSH content, and increased GPx1 activity. The reduction in total GSH content of the liver might be the result of its participation in terminating free radical reactions initiated by fenvalerate. The increased activity of GPx1 was possibly as an adaptive response to the oxidant stress. In fact, the selenoenzyme GPx1 is known to play an important role in the defense mechanisms of mammals against oxidative damage by catalyzing the reduction of H2O2 and a large variety of hydroperoxides with GSH as the hydrogen donor, thereby involving in the regulation of the cellular redox status. 44 Thus, the significant alterations observed in the hepatic antioxidant defense system and augmentation of oxidative stress parameter TBARS indicate the induction of oxidative stress by fenvalerate exposure in rats.
The involvement of ROS in the toxicity of various classes of pesticides has been previously reported.12–16,45 Although the induction of oxidative stress and alteration of cellular antioxidant system by some pyrethroid insecticides have been shown previously, the study designs of each were different with respect to the dose, route, and length of insecticide treatment and measured parameters, and their results were rather controversial.17–22,46,47 In agreement with our findings, Kale et al. 19 reported that a single oral dose of fenvalerate (as low as 0.001% LD50) causes significant elevations in LP and decreases in GSH content of rat liver as well as kidney and heart tissues. However, in contrast to our findings, they observed increases in hepatic CAT and SOD activities. The results of Prasanthi et al. 22 were also in the same line as our results as they observed increased hepatic TBARs level and GPx1 activity and a significant decrease of GSH content in rats fed with 250, 500, or 1000 ppm fenvalerate-containing diet for 28 days. However, they observed decreases in the activities of SOD and CAT in contrast to both ours and the findings of Kale et al. 19
In the present study, we induced iodine deficiency by supplying animals 1% sodium perchloride containing drinking water. Perchlorate is known to effectively block the uptake of iodine by the thyroid gland,48,49 by competing with iodide at the sodium-iodide symporter (NIS), the transport system that transports iodine into the thyroid.50,51 As we have shown earlier, 23 iodine deficiency and resulting hypothyroidism produced by perchloride treatment was evident by higher thyroid-stimulating hormone (TSH) and lower plasma TT4 and TT3 levels along with the increased thyroid weights in rats. The results presented herein showed that iodine deficiency altered only GPx1 and CAT activities in rat liver and did not cause an elevation of LP. These results are in the same line as those of our previous study designed similarly, but, with 5 weeks of treatment. 34 However, probably due the shorter feeding period, the previous study did not reveal an alteration in GPx1 activity in ID rats. Variations in thyroid hormone levels are among the main physiological modulators of in vivo cellular oxidative stress. Hypermetabolic state in hyperthyroidism is associated with increases in free radical production and enhanced LP, while hypometabolic state in hypothyroidism is generally associated with a decrease in free radical production.52,53 Das and Chainy 54 demonstrated that mitochondrial antioxidant defense system in rat liver is considerably influenced by the thyroid states of the body and the activity of CAT is significantly reduced in hypothyroid rats as we observed. Thus, our results in ID rats were in agreement with those of published data. On the other hand, in the present study, fenvalerate exposure induced significant level of LP in hypothyroid rats while was not effective in altering the antioxidant status significantly.
In selenium-deficient rats, due to the depletion of selenium reservoirs, a marked prooxidant/antioxidant imbalance was evident by highly reduced GSH content and CAT activity, elevated TBARs levels, and substantial losses of GPx1 activity. In fact, selenium is an integral component at the catalytic sites of the GPx1, and selenium deficiency causes a profound reduction in GPx1 activity in several tissues, particularly in liver, and results in increased oxidative stress.27,55 Therefore, oxidative insult potential of fenvalerate resulted in further depression of GPx1 activity, enhancement of LP and activity of CAT in SeDF group compared to SeD rats.
With regard to the alterations of TBARs levels and GPx1 and CAT activities, the same trend as selenium deficiency was observed in combined iodine/selenium deficiency. However, the activities of SODtotal, SODMn, and SODCu,Zn were significantly high in ISeD group, while there was no change in SeD rats. Accordingly, fenvalerate exposure in ISeD animals aggravated the already present prooxidant/antioxidant imbalance and thus markedly enhanced TBARs concentrations, significantly reduced GSH content, and antioxidant enzyme activities compared to ISeD group, indicating the higher susceptibility of combined iodine/selenium deficiency state to the oxidative insult of fenvalerate.
In conclusion, our findings showed that fenvalerate has a potential to induce significant hepatic oxidative stress in selenium- and/or iodine-deficient rats, as well as healthy controls. Consequences of pro-oxidant/antioxidant imbalance induced by fenvalerate exposure in rats may also be critically important for human populations with iodine- or selenium-deficiencies, particularly with combined iodine/selenium deficiency. The effects of fenvalerate shown herein might also be related to its thyroid-modifying potential that we reported previously. 23 However, for elaborating and better understanding the insights of the fenvalerate action, further studies would certainly be needed.
Footnotes
This study was supported by Hacettepe University Research Fund, 01.01.301.001.
