Abstract
This study examines the effects of vitamin E and an extremey low frequency electromagnetic field (ELF-EMF) and their combination in different time intervals of exposure of vitamin E (tocopherol) on the AT478 murine squamous cell carcinoma line. This study provides insight into the influence of correlations between ELF-EMF and vitamin E supplementation on antioxidant enzyme activity in malignant cells in vitro. Following vitamin E treatment, activity of the antioxidant enzymes is increased in an exposure-dependent manner compared with the untreated group. Application of ELF-EMF alone or with vitamin E increases both superoxide dismutase isoenzymes and glutathione peroxidase activities in comparison to the control group. The results suggest that ELF-EMF alters antioxidative activities of vitamin E in AT478 tumor cells. This study confirms the role of vitamin E in decreasing susceptibility to lipid peroxidation in AT478 tumor cells.
Keywords
Recent in vivo and in vitro studies showed that physical factors such as 60-Hz electric and magnetic fields can affect cell metabolism.
1
It is believed that exposure to extremely low frequency electromagnetic fields (ELF-EMF, frequencies less than 200-300 Hz) can alter the transcription and translation of genes such as
Reactive oxygen species (ROS) are emerging as critical signaling molecules. Free radicals are chemical species containing 1 or more unpaired electrons. 4 These ROS are known to cause DNA damage and lipid peroxidation/oxidation in the membranes. 5,6 The unpaired electrons of oxygen react to form partially reduced highly reactive species that are classified as ROS, including superoxide (ċO2 -), hydrogen peroxide (H2O2), hydroxyl radical, and peroxynitrite. Various enzyme systems produce ROS, including the mitochondrial electron transport chain, cytochrome P450, lipooxygenase, cyclooxygenase, the NADPH oxidase complex, xanthine oxidase, and peroxisomes. 7,8 Mitochondrial oxygen metabolism, the dominant source of superoxide radicals, results from incomplete coupling of electrons and H+ with oxygen in the electron transport chain. According to the work of James et al, 9 ROS may have at least 3 basal functions in tumor cells metabolism. The first perspective is the traditional view of ROS generating oxidative stress as a precursor to tissue injury, which involves complex interaction of free radical production, detoxification, and repair of radical damage. The second perspective views ROS as crucial messengers of signal transduction that play an essential role in tumor cells proliferation or in the maintenance of genomic instability, which in turn facilitates growth. 10,11 The third, evolving, perspective is the view of ROS as secondary “death markers” for cells that are in the process of committing to apoptotic or necrotic pathways following a toxic insult. 12
To protect against the adverse effects of the ROS, cells posses a complex machinery of antioxidant compounds and enzymes, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GSH-Px). The activities of these enzymes have been shown to be regulated by nutrients 5 and hormones. 13
Antioxidants may act as free radical scavengers, and it is therefore likely that dietary antioxidants such as vitamin C, vitamin E, and β-carotene are important in inhibiting cancer development in vivo. 14,15 Lipid peroxidation has been shown to cause a profound alteration in structural integrity and functions of cell membrane. Increasing levels of malondialdehyde (MDA) as a marker of lipid peroxidation may indicate a role of free radicals in pathogenesis of precancerous condition lesion and cancer. 16 Examination of certain malignant cell types showed decreased susceptibility to lipid peroxidation compared with their nonmalignant counterparts. 17,18 Vitamin E is an essential fat-soluble vitamin that functions, at least in part, as a lipid-soluble antioxidant. This activity is one of the recognized or postulated vitamin E biological activities, such as various antioxidant functions (inhibition of lipid peroxidation or inhibition of nitric acid species formation), cholesterol and thromboxane suppressive actions, and proapoptotic activity. 19
Free radicals can generate lipid peroxidation in an organism, and MDA is one of the final products of polyunsaturated fatty acid peroxidation in cells. An increase in free radicals causes overproduction of MDA. The MDA level is a commonly known as a marker of oxidative stress and antioxidant status in patients with cancer. 20
Exposure to ELF-EMFs is generally believed to be innocuous to humans because of the low level energy provided by these fields, which is of a magnitude well below that required to affect the metabolic rate of the human body. 21-23 However, an increasing number of studies have reported that ELF-EMF exposure is capable of eliciting in vivo and in vitro biological effects. 24-25 ELF-EMF exposure has been shown to increase oxidative stress in some models like chick embryos, 26 mammalian cultured cells, 27 human erythrocytes, 28 and tumor cells. 29 The increased oxidative-stress involves oxidative DNA damage and lipid peroxidation 30 and may cause a number of systemic disturbances. 25
Few studies have demonstrated that both ELF-EMF and antioxidant supplementation may influence antioxidant enzyme activities on normal and tumor cells in vitro. Our previous studies demonstrated that both ELF-EMF and melatonin may alter antioxidative enzyme activities and lipid peroxidation in 3T3-L1 preadipocytes 1 and in AT478 murine squamous carcinoma cells. 31
The aim of present study was to examine the effects of ELF-EMF and antioxidant supplementation (vitamin E) on antioxidant enzymes activity and level of lipid peroxidation in AT478 murine squamous cell carcinoma culture in vitro.
Methods
Cell Culture
AT478 is a well-differentiated murine squamous cell carcinoma line, derived from spontaneous tumors of the cervix (vaginal mucosa) originally arising from a C3H mouse inbred colony. Squamous cell carcinoma is a cell line culture that has the ability to grow in the flat form of multilayered epithelial colonies (megacolonies) in continuous contact with basal layer and other cells. 32 Briefly, about 4 × 104 cells in 50 μL of Dulbecco’s modified Eagle’s Medium (Sigma, St Louis, Mo), supplemented with 10% fetal bovine serum (Gibco, North Andover, Mass), were deposited as single drops in culture bottles (in each flask) and left for 24 hours for attachment. After 1 day, single nonattached cells were removed by rinsing with medium, and fresh growth medium was added. Cells were incubated for 48 to 72 hours in an atmosphere of 37°C air and 6% CO2. After this time, megacolonies reached diameters of about 0.8 to 1 cm and contained about 4 × 105 cells.
Vitamin E and Magnetic Field Exposure
Vitamin E
After exposition of the cultures cells to the ELF-EMF (16 minutes), antioxidative enzyme activities of manganese (Mn) SOD, copper/zinc (Cu/Zn) SOD, and GSH-Px and levels of MDA with or without vitamin E were measured. According to the treatment, groups were assigned as follows: control group without vitamin E and ELF-EMF, and other groups treated solely with vitamin E or both ELF-EMF and vitamin E exposition. Medium was collected after 24 or 72 hours of incubation. Five repeats of each test were performed.
Enzymes
MnSOD and Cu/ZnSOD (EC 1.15.1.1) isoenzyme activities were estimated according to the Oyanagui method 34 and expressed in nitrite units per milliliter (NU/mL). The activity of GSH-Px (EC 1.11.1.19) was measured following the method of Paglia and Valentine, 35 using enzymatic conjunction with glutathione reductase and expressed in micromoles of NADPH2/mL/min. Levels of MDA were estimated after calorimetric method and presented in micromoles of MDA per milliliter of medium, using reaction with thiobarbituric acid and calculated by spectrophotometry (515 nm absorbance; 522 nm emission). 36
Statistics
All results are presented as mean ± SEM. To compare results, Student’s
Results
Effects of vitamin E supplementation on MnSOD and Cu/ZnSOD as well as GSH-Px activity and MDA levels were studied as a function of time in AT478 C3H cell culture medium. All analyzed enzyme activities significantly increased compared with the untreated control group (Tables 1 and 2).
Antioxidative Enzyme Activities and Malondialdehyde Levels After 24 Hours of Incubation in AT478 Media Cells
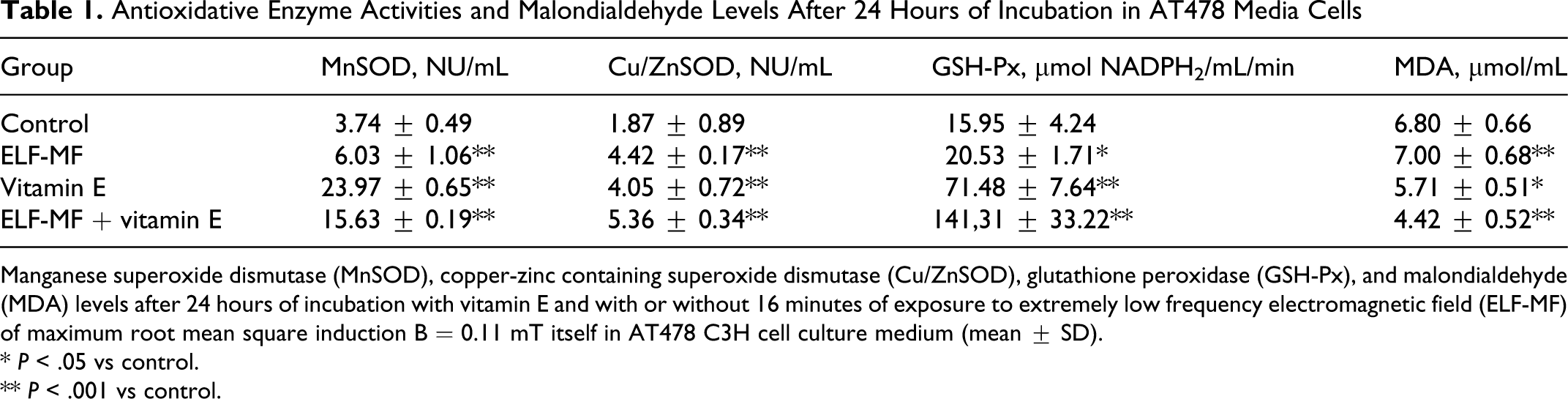
Manganese superoxide dismutase (MnSOD), copper-zinc containing superoxide dismutase (Cu/ZnSOD), glutathione peroxidase (GSH-Px), and malondialdehyde (MDA) levels after 24 hours of incubation with vitamin E and with or without 16 minutes of exposure to extremely low frequency electromagnetic field (ELF-MF) of maximum root mean square induction B = 0.11 mT itself in AT478 C3H cell culture medium (mean ± SD).
*
**
Antioxidative Enzyme Activities After 72 Hours of Incubation in AT478 Media Cells
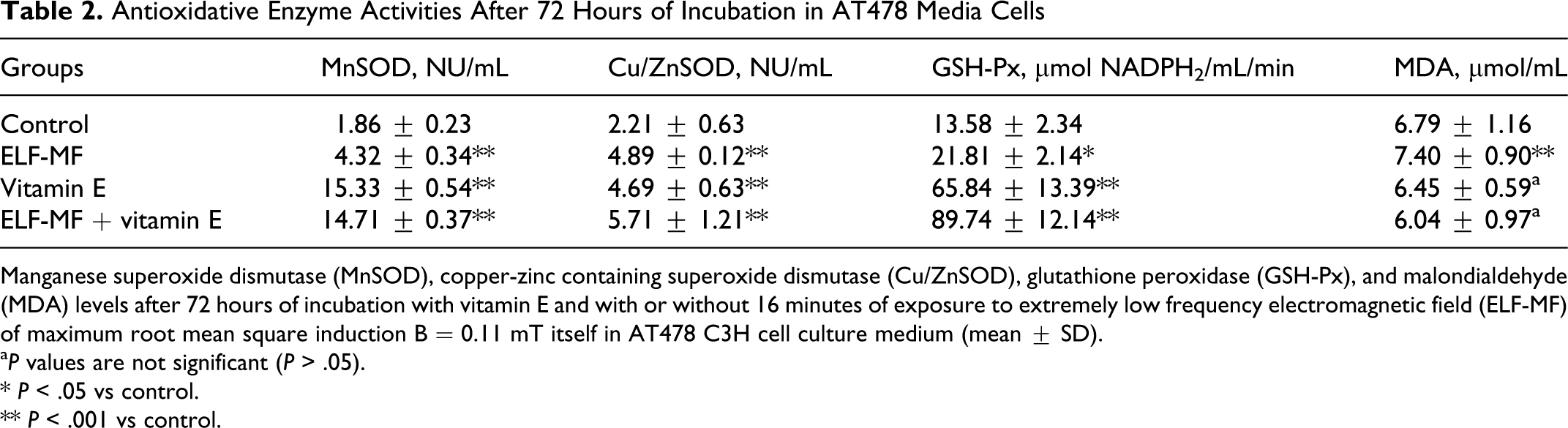
Manganese superoxide dismutase (MnSOD), copper-zinc containing superoxide dismutase (Cu/ZnSOD), glutathione peroxidase (GSH-Px), and malondialdehyde (MDA) levels after 72 hours of incubation with vitamin E and with or without 16 minutes of exposure to extremely low frequency electromagnetic field (ELF-MF) of maximum root mean square induction B = 0.11 mT itself in AT478 C3H cell culture medium (mean ± SD).
a
*
**
In the group with both vitamin E and ELF-EMF exposure for 24 hours, a marked increase in Cu/ZnSOD activity was observed in comparison with the group treated only with vitamin E for 24 hours and 72 hours (5.36 ± 0.34 vs 4.05 ± 0.72 NU/mL (24 hours) and 4.69 ± 0.63 NU/mL (72 hours), respectively;
We also demonstrated that the level of MDA was significantly lower in the group stimulated with both ELF-EMF and vitamin E compared with the control group but only after 24 hours of exposure (4.42 ± 0.52 vs 6.80 ± 0.66 μmol/mL;
After 72 hours of incubation, we observed a higher level of MDA only in the group stimulated with ELF-EMF compared with the control group (7.40 ± 0.90 vs 6.79 ± 1.16 μmol MDA/mL;
The differences in values between all analyzed experimental groups were significant at
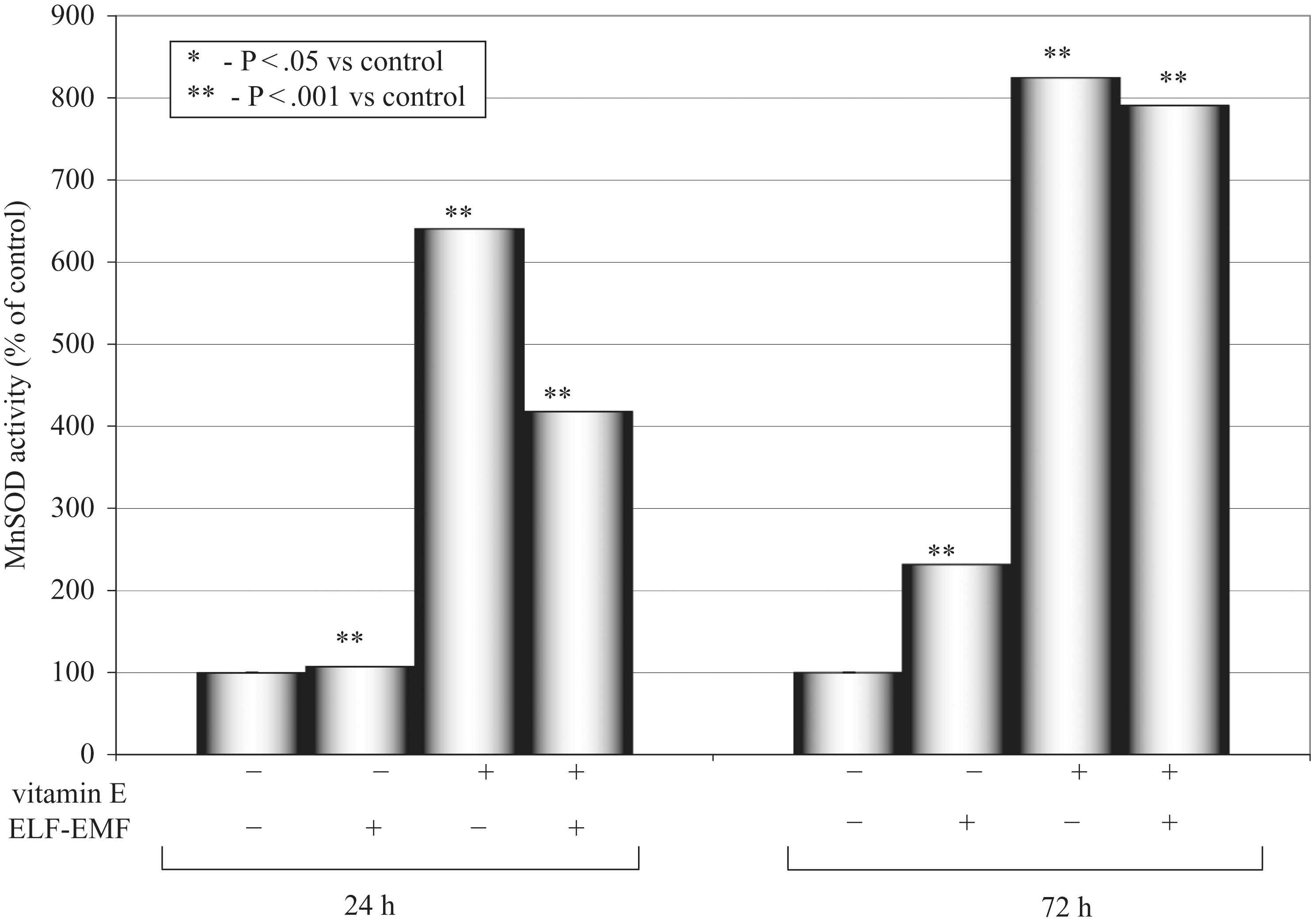
Manganese superoxide dismutase (MnSOD) activity after 24 and 72 hours.
Effects of a high dose of vitamin E and ELF-EMF on Cu/ZnSOD activity as a mean percentage of the control (100%) after 24 hours and 72 hours of incubation time in cultured medium of AT478 murine squamous cell carcinoma are shown in Figure 2.
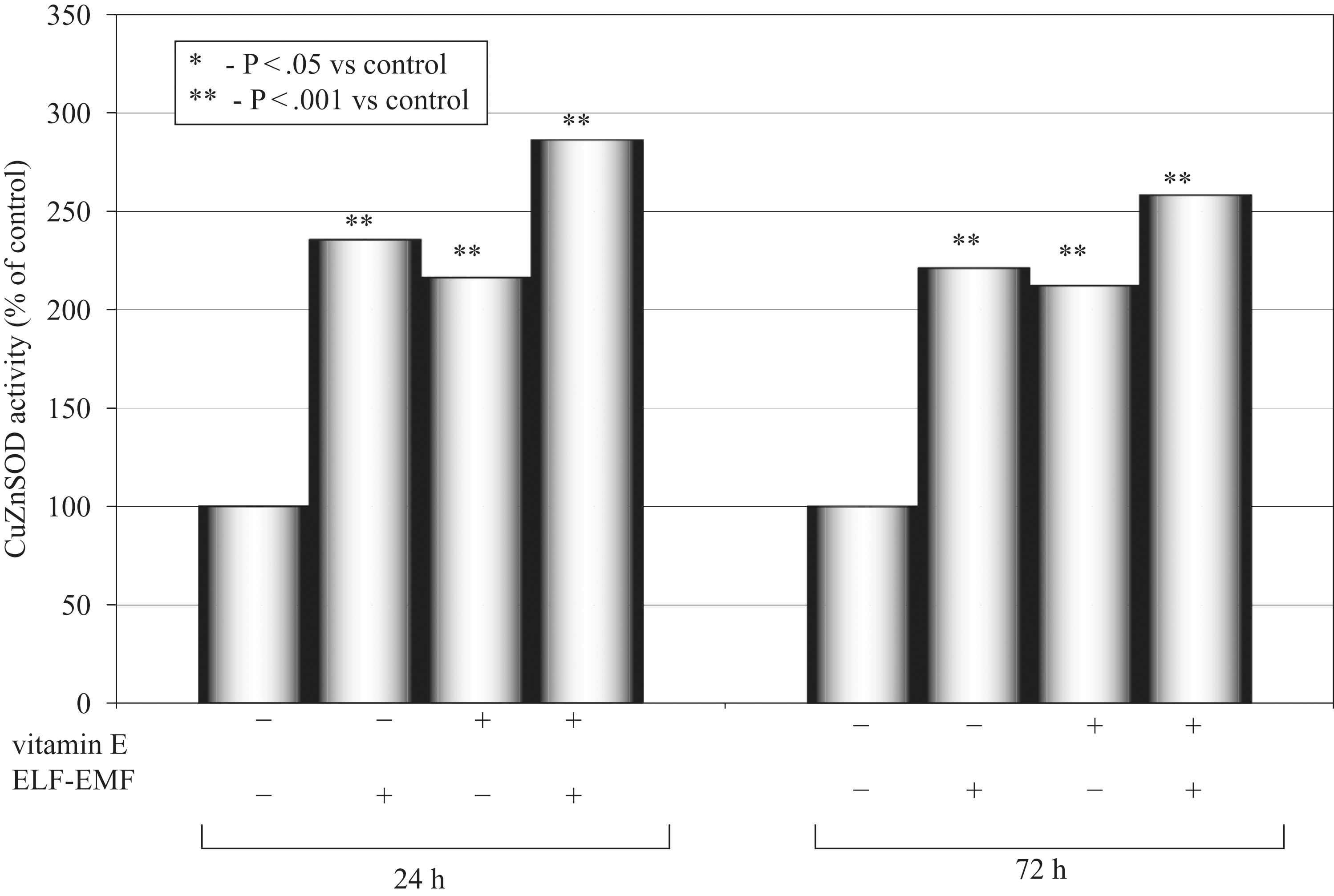
Copper and zinc superoxide dismutase (Cu/ZnSOD) activity after 24 and 72 hours.
Effects of a high dose of vitamin E and ELF-EMF on GSH-Px activity as a mean percentage of the control (100%) after 24 hours and 72 hours of incubation time in cultured medium of AT478 murine squamous cell carcinoma are shown in Figure 3 .
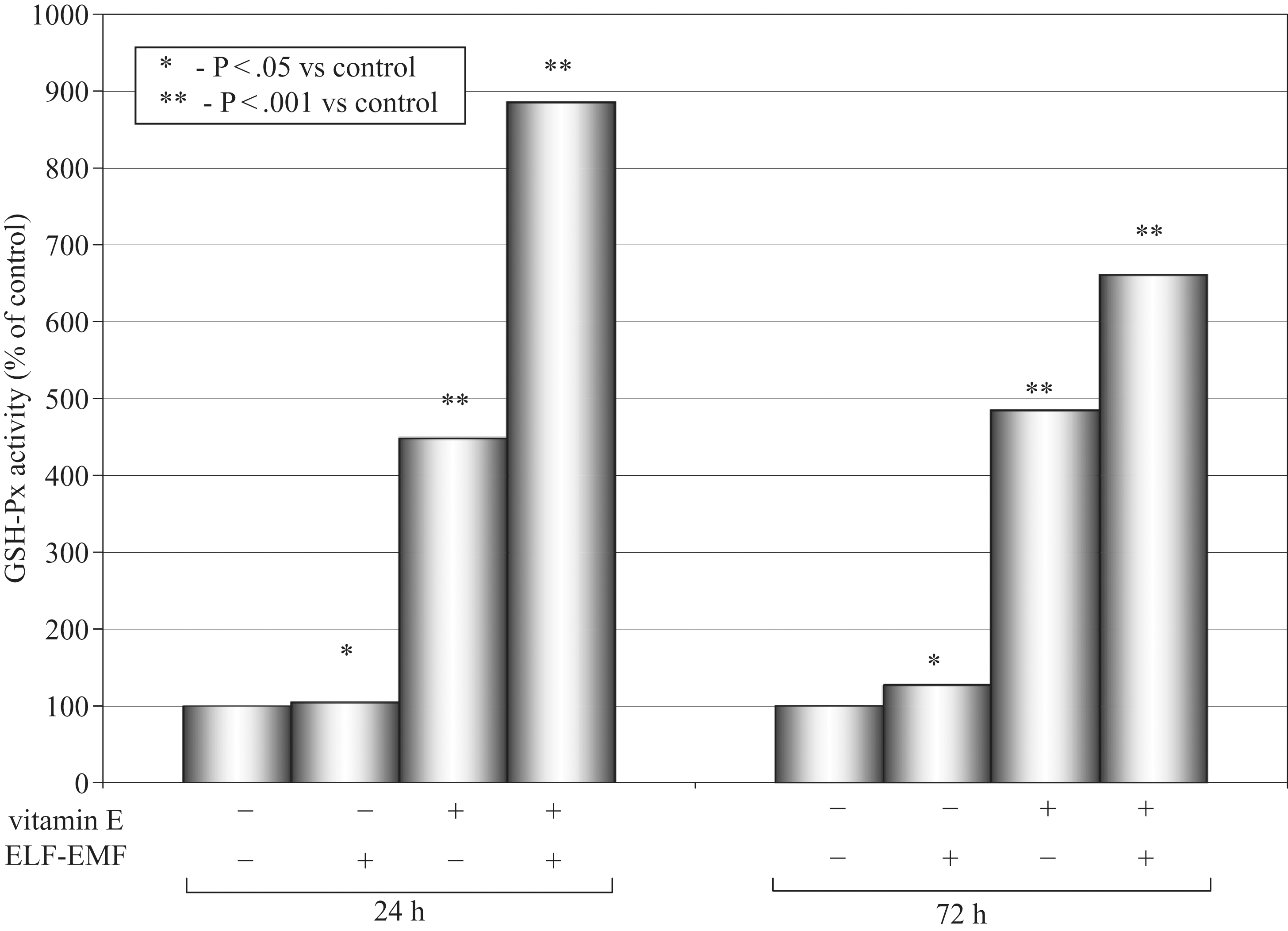
Glutathione peroxidase (GSH-Px) activity and 24 and 72 hours.
Levels of MDA as a mean percentage of the control (100%) after 24 hours and 72 hours of incubation time in cultured medium of AT478 murine squamous cell carcinoma are shown in Figure 4.
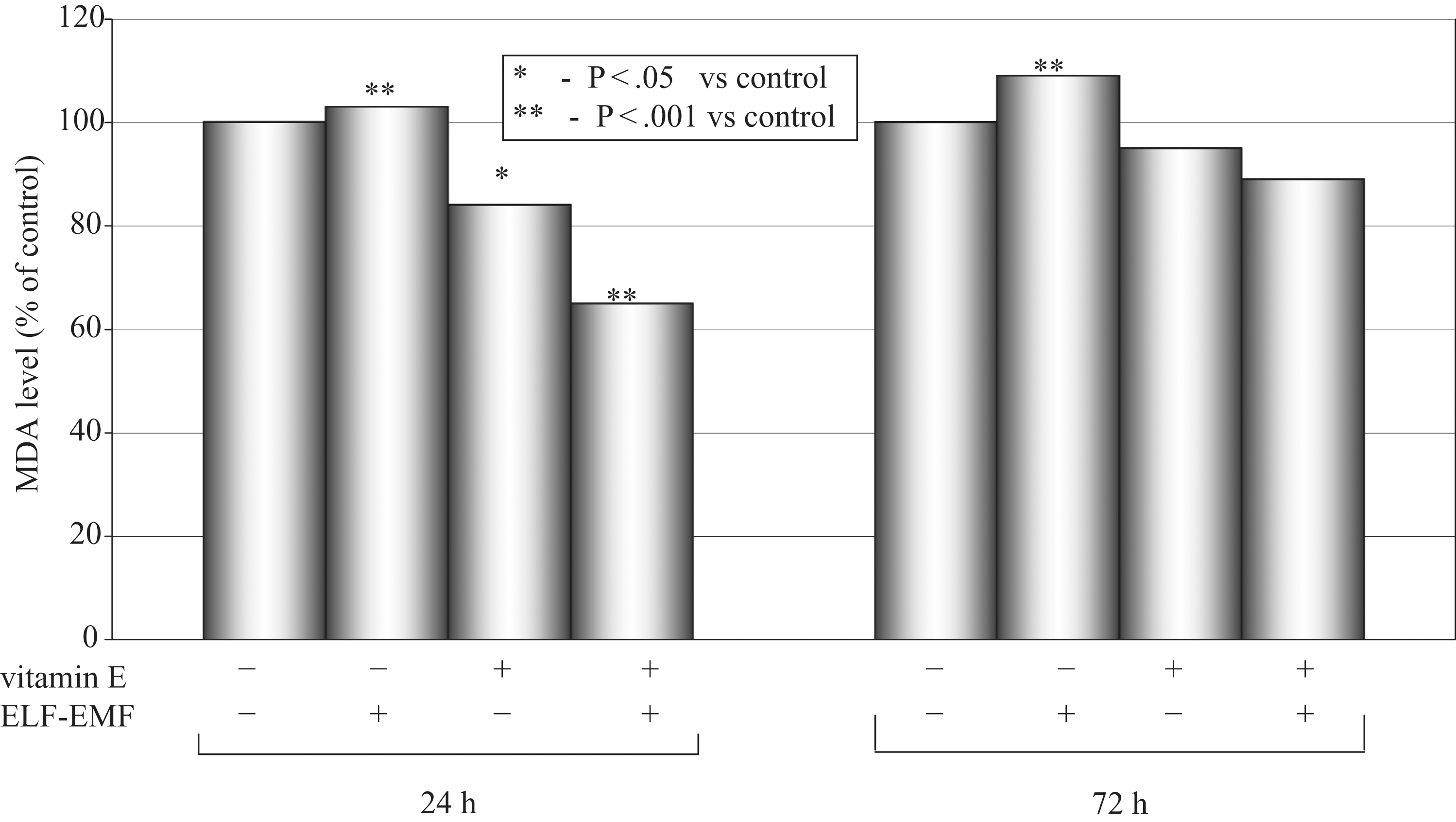
Malondialdehyde (MDA) concentration after 24 and 72 hours.
Discussion
Our data suggest that ELF-EMF plays an important role in affecting the activities of antioxidative enzymes. The present data and those from our previous studies 1,31 also strongly suggest that ELF-EMF may play an important role in regulation of enzyme activity, probably by enhanced ROS production. This agrees with other researchers' results. 28,31,37,38 Many studies have demonstrated biological effects of ELF-EMF with respect to their cytological and biochemical effects, including mutagenicity, clastogenicity, and carcinogenicity of different cells. There is evidence that the outer surface of the cell membrane is the primary locus for ELF-EMF–induced cellular alterations. ELF-EMF modulated proliferation of normal as well as transformed cells in vivo and in vitro. Current clinical and in vitro experiments do not demonstrate that ELF-EMF acts as a cancer initiator. However, it may act as a promoter or may affect tumor progression. Further observations and epidemiological studies of ELF-EMF should be accompanied by laboratory experiments to evaluate its biological and health effects. 38 Whether ELF-EMF exposure is correlated with human cancer is controversial and needs further research. Most studies indicate that ELF-EMF is a nonionizing form of radiation that is too weak to induce thermal effects; therefore, it cannot damage or mutate DNA. 2,39,40 A few studies have been interpreted as indicating either mutagenic or co-mutagenic activity. 41,42 However, the weight of the evidence indicates that ELF-EMF exposure can be an extremely weak initiator, if not a noninitiator, of carcinogenesis. Assuming that ELF-EMF plays a role in carcinogenic process but is not a carcinogenic initiator, it seems that ELF-EMF is likely to be involved in the promotional phase of cancer development. 2
Very few data have been reported on the influence of ELF-EMF exposure on antioxidative enzyme activities in tumor cells in vitro. Increased mitochondrial MnSOD isoenzyme activity after vitamin E treatment suggests that vitamin E increases efficiency of the electron transport chain in the inner mitochondrial membrane and stimulates complex I and IV of the electron transport chain in the mitochondrion. 43,44 This action of vitamin E is expected to increase efficiency of oxidative phosphorylation and adenosine triphosphate production and the high efficiency of ROS elimination by MnSOD itself. 44
In this study, cells subjected only to the action of the magnetic field showed higher oxidative stress in comparison to the control group. The highest MnSOD activity was observed in the supernatants of the cells subjected only to the stimulation with vitamin E, and this effect was exposition time dependent. Activity of MnSOD enzyme was lower in the supernatants of the cells subjected only to stimulation with ELF-EMF and vitamin E, and this effect was more pronounced in the group with 24 hours of stimulation. The activity of the Cu/ZnSOD isoenzyme was highest in the group stimulated with magnetic field and vitamin E in comparison to the control group and the group subjected only to the magnetic field action, and this effect was exposition-time dependent.
SOD converts ċO2 - into H2O2, whereas CAT and peroxidases convert H2O2 into water. Two toxic species, ċO2 - and H2O2, are converted to the harmless product water. An important feature of these enzymes is that they are highly compartmentalized. 45,46 MnSOD is expressed in the mitochondria; Cu/ZnSOD is localized in the cytoplasm. These enzymes reduce oxidative stress in the various parts of the cell. Cu/ZnSOD comprises 90% of total SOD activity in most eukaryotic cells. 46,47 A small fraction of this enzyme has been found in lysosomes, peroxisomes, and nuclei. It has been demonstrated that 2% of Cu/ZnSOD is found in the intermembrane space of mitochondria, 48,49 and this localization was suggested to be important in providing further protection against ROS and preventing superoxide radicals from leaking out of mitochondria. 46 MnSOD is limited to the matrix of mitochondria 50 and exists at much lower concentrations inside the cells than Cu/ZnSOD. Because the mitochondrial respiratory chain is a major site of superoxide generation in most cells, MnSOD plays an important role in maintaining the balance of cellular ROS. 45
Catalase and GSH-Px are able to detoxify hydrogen peroxide to water. SOD and CAT need no cofactors to function, but GSH-Px requires several cofactors and proteins. Two such proteins, glutathione reductase (GR) and glucose-6-phosphate dehydrogenase (G-6-PD), are considered secondary antioxidant enzymes because they do not act on ROS but they enable GSH-Px to function. 51 Functional cooperation between various antioxidative enzymes is presented in Figure 5. In our in vitro model we observed that magnetic field additionally stimulates antioxidative properties of vitamin E by increasing activity of GSH-Px. The highest activity of this enzyme was observed in the group subjected to the action of vitamin E and the magnetic field in comparison to all remaining study groups. Therefore, we can conclude that the cells exposed to the magnetic field and vitamin E may show increased resistance toward exogenic H2O2 and lower oxidative stress and resistance to apoptosis.
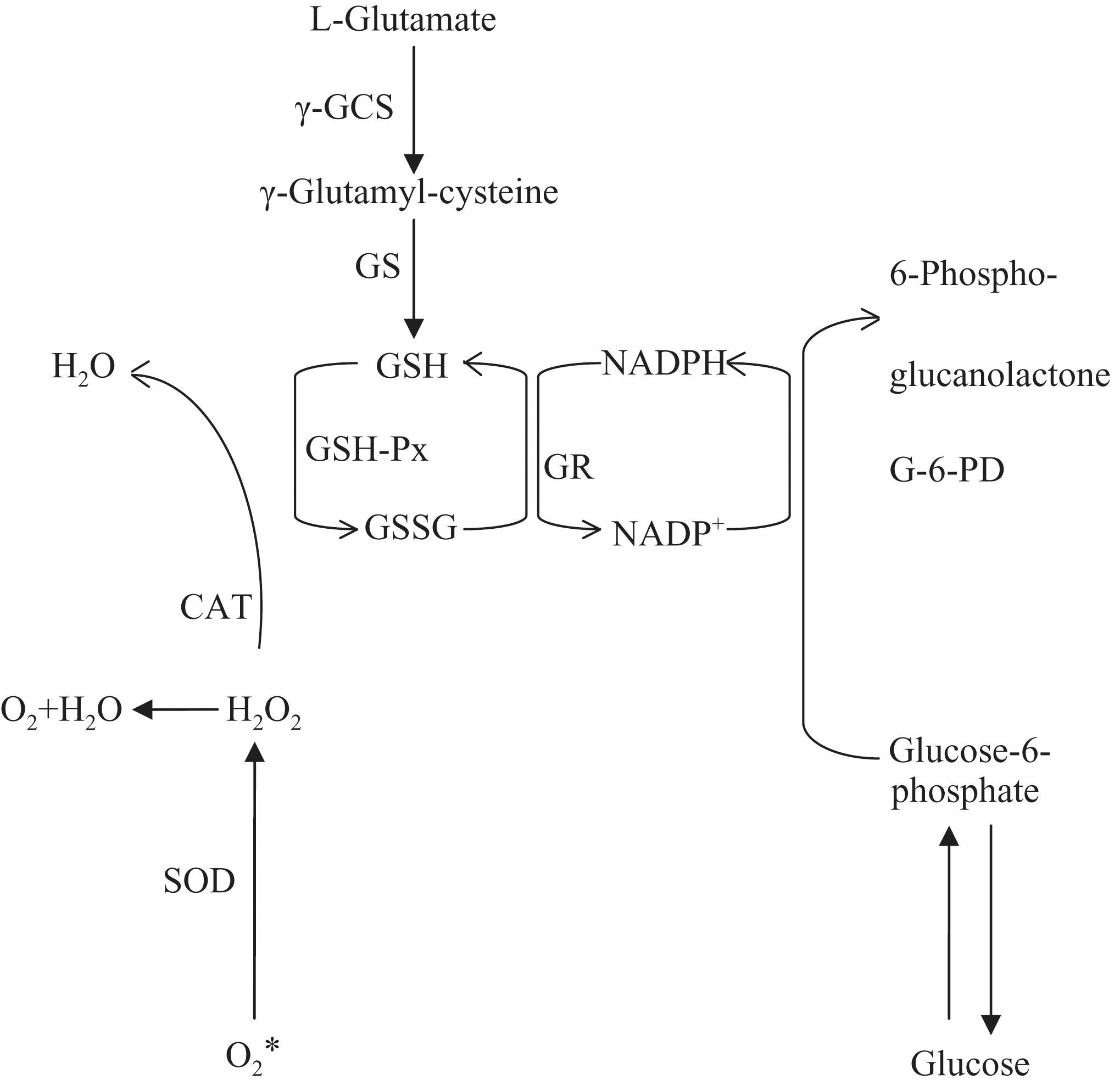
Functionally substrate specific cooperation between various antioxidative enzymes and cofactors. SOD, superoxide dismutase; CAT, catalase; GSH-Px, glutathione peroxidase; GR, glutathione reductase; γ-GCS, γ-glutamylcysteine synthetase; GS, glutamine synthetase; G-6-PD, glucose-6-phosphate dehydrogenase.
The SOD enzymes cooperate with CAT and GSH-Px enzymes. In this pathway, 2 toxic species, ċO2
- and H2O2, are converted to the harmless product water and we observed, as presumed, a highly positive correlation (
Pearson Correlation Coefficients (R) Between All Analyzed Enzyme Activities and Malondialdehyde Levels After 24 Hours of Incubation
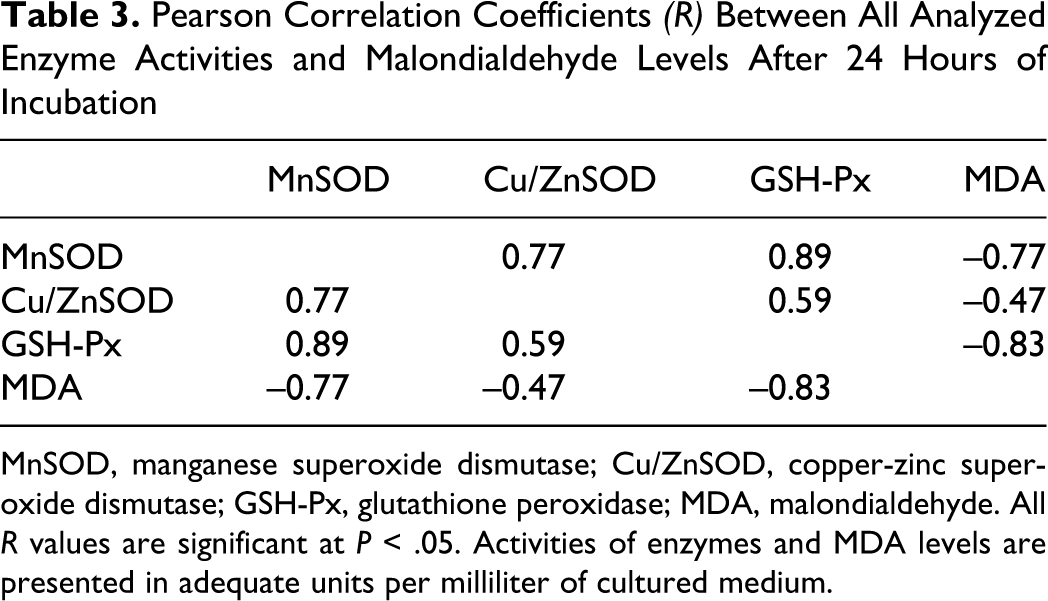
MnSOD, manganese superoxide dismutase; Cu/ZnSOD, copper-zinc superoxide dismutase; GSH-Px, glutathione peroxidase; MDA, malondialdehyde. All
Pearson Correlation Coefficients (R) Between All Analyzed Enzyme Activities and Malondialdehyde Levels After 72 Hours of Incubation
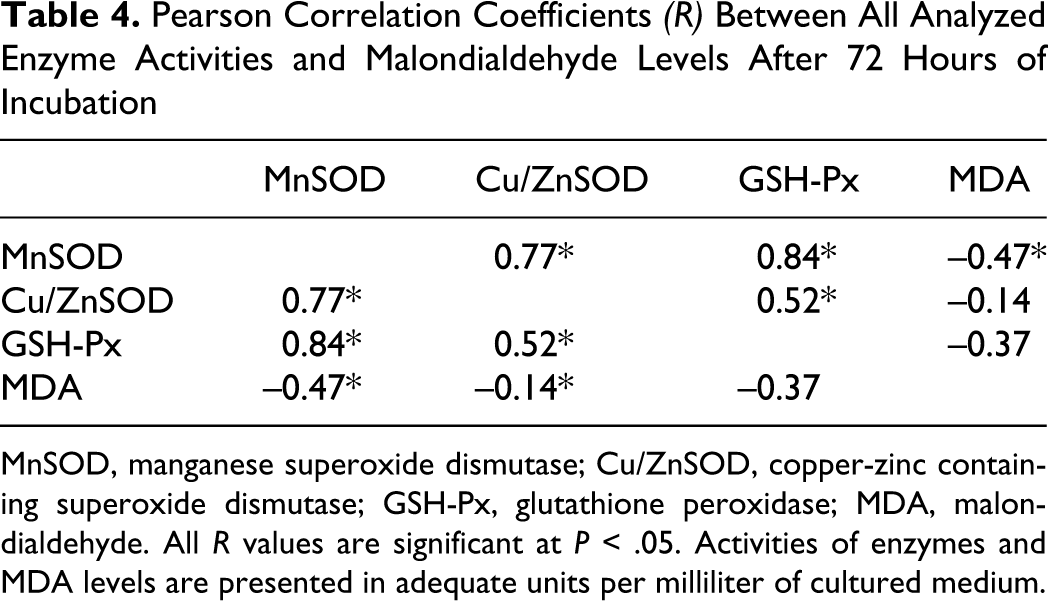
MnSOD, manganese superoxide dismutase; Cu/ZnSOD, copper-zinc containing superoxide dismutase; GSH-Px, glutathione peroxidase; MDA, malondialdehyde. All
In this study we show that stimulation exclusively with a magnetic field causes a small increase in lipid peroxidation (103% in comparison to untreated control;
This creates an opportunity for therapeutic manipulation of the redox states in cancer cells. High levels of intracellular ROS are cytotoxic, whereas ROS at low concentration are necessary for life and are involved in regulation of several key physiological processes such as differentiation, apoptosis, and cell proliferation. Activity of MnSOD was diminished in transformed cells compared with their nonmalignant counterparts. 53 Cancer cells are nearly always low in MnSOD and CAT activity and usually low in Cu/ZnSOD activity. 54,55 If antioxidant enzymes are important in cancer, then normalization of the level of these enzymes should result in reversion of at least part of the cancer phenotype. Some data have shown 51,54,55 that elevation of SOD levels by exposure to a superoxide generator like ELF-EMF or manipulation of cancer cells with low molecular weight antioxidants can inhibit cancer cell growth. From a therapeutic point of view, cancer cells exposed to ELF-EMF generate high levels of hydrogen peroxide, which can be inhibited by drugs such as 1,3-bis (2-chloroethyl)-1-nitrosouera (BCNU), a clinically used anticancer drug that causes alkylation and also inhibits GR activity. Inhibition of GR prevents conversion of GSH disulfide back to GSH and thus interferes with the removal of peroxides by the GSH-Px pathway. In the present study, the highest level of GSH-Px activity was observed in the group stimulated with both ELF-EMF and vitamin E. This experimental group should be less susceptible to hydrogen peroxide levels. Additional inhibition of GR activity in a group stimulated with ELF-EMF and vitamin E is a promising therapeutic possibility to achieve growth inhibition of cancer cells in vitro. If GR is inhibited, cells cannot remove hydrogen peroxide. 56 It has been reported that H2O2 induces apoptosis of a wide range of tumor cells in vitro via activation of the caspase cascade. 57 Hydrogen peroxide readily crosses cellular membranes and causes oxidative damage to DNA, protein, and lipids by direct oxidation or via the transition metal-driven Haber Weiss reaction to the extremely reactive hydroxyl radical. 58
Unfortunately, high levels of GSH-Px in cancer cells lead to cancer cell survival via effective removal of hydrogen peroxide. Increased activity of detoxifying pathways like GSH-Px can make tumor cells less susceptible to chemotherapy-mediated damage. 59,60 Earlier work demonstrated that the addition of radical scavengers and compounds with peroxidase activity can reduce the cytotoxic effect of doxorubicin chemotherapy in vitro. 60,61
Current in vitro data have shown that a number of cancer cell lines contain elevated levels of MnSOD and decreased levels of CAT, and this change in steady-state levels of hydrogen peroxide correlates with increased metastasis (increased expression of matrix metalloproteinase enzymes), proliferation, and resistance to apoptosis. 62-64 Interestingly, MnSOD-dependent increases in matrix metalloproteinase expression can be reversed by the H2O2-detoxifying enzymes CAT and GSH-Px. 65
Increased activity of SOD isoenzymes in tumor metastasis cells may lead to increased resistance to ROS-mediated damage by immune cells penetrating the bloodstream. 66 We showed that ELF-EMF changes antioxidative properties of vitamin E in AT478 cell culture. Stimulation with a magnetic field or vitamin E alters the activities of antioxidative enzymes. Because stimulation with both factors changed the activities of antioxidative enzymes in the culture, further studies should be conducted to determine the influence of these factors on proliferation processes and chemosensitivity of the cancer cells to commonly used cytostatics causing ROS-mediated damage.
