Abstract
Studies have demonstrated that the induction of oxidative stress may be involved in oxidative DNA damage. The present study examined and assessed the hydrogen peroxide (H2O2)-mediated DNA damage in human tumor KB cells and also assessed the ability of Moringa oleifera leaf extracts to inhibit the oxidative damage. H2O2 imposed a stress on the membrane lipids which was quantified by the extent of thiobarbituric acid reactive substances (TBARS) formed. The leaf extracts caused a very significant inhibition of the extent of LPO formation and enhanced the activity of antioxidative enzymes such as superoxide dismutase (SOD) and catalase (CAT) in KB cells. The comet assay was employed to study the DNA damage and its inhibition by the leaf extracts. H2O2 caused a significant increase in the number of cells bearing comets, resulting in significant DNA damage. The leaf extracts significantly reduced the incidence of comets in the oxidant stressed cells. The extent of cytotoxicity of H2O2 in the presence and the absence of leaf extracts studied in KB tumor cells by the MTT assay showed that H2O2 caused a marked decrease in the viability of KB cells where as the leaf extracts effectively increased the viability of assaulted KB cells. The observed cytoprotective activity is probably due to the antioxidant properties of its constituents, mainly phenolics. Total phenolics showed higher correlation with antioxidant activity. The leaf extracts showed higher antioxidant activity than the reference compound. These results suggest that the inhibition by the leaf extracts on oxidative DNA damage could be attributed to their free radical scavenging activities and the effect evidenced in KB cells can be in part correlated to a modulation of redox-sensitive mechanisms.
Introduction
Oxidative stress has been implicated in the pathogenesis of a wide variety of clinical disorders such as neurodegenerative diseases, cancer, cardiovascular disease, atherosclerosis and inflammation. DNA is a crucial target molecule in a cell confronted with direct reactive oxygen species (ROS) insults. Generation of ROS in close proximity to DNA causes various types of DNA damage such as base and sugar damage, strand breaks and DNA-protein crosslinks. 1 In recent years, it has become increasingly clear that ROS, such as O2 •− and H2O2 may act as second messengers and play a role in modulating cellular function. H2O2 produces highly toxic hydroxyl radicals, which cause DNA damage, and contribute to cell death. 2 Membrane lipids present in subcellular organelles are highly susceptible to free radical damage. During lipid peroxidation (LPO), a large number of toxic byproducts are also formed. LPO results in the formation of several toxic byproducts such as 4-hydroxy nonenal and malondialdehyde. They form adducts with DNA and induce mutagenesis, carcinogenicity and apoptosis. 3
DNA damage and inefficient repair may initiate the process of carcinogenesis 4 and modulation of these parameters could provide a potentially effective means of cancer control. It seems important, for this reason, to develop ways to lower the risk and inhibit the effect of ROS-mediated DNA damage. Many substances derived from dietary or medicinal plants are known to be effective and versatile chemopreventive and antitumoral agents in a number of experimental models of carcinogenesis. There is an increasing evidence for an association between a high consumption of fruit and vegetables and reduced risk of oral cancer 5 and prostate carcinoma, the most common tumor in men. 6 Therefore, the purpose of this study was to investigate whether the dietary leaf extract could inhibit the DNA damage and the mechanism against the oxidative assaulted biomolecular targets.
The identification of new antioxidants remains a highly active research area, because these agents may reduce the risk of various chronic diseases caused by ROS. 7 There is an increasing evidence that plant extracts have the potential for scavenging ROS. 8 The growing interest in the substitution of synthetic food antioxidants by natural antioxidants in health implications has hastened the research on vegetable sources and the screening of raw materials for identifying antioxidants. 9 The antioxidants could attenuate this oxidative damage of a tissue indirectly by enhancing natural defenses of cell and/or directly by scavenging the free radical species.
Moringa oleifera is a common tree, native to India and cultivated throughout subtropical areas from West Africa to Fiji. Also known by the names of drumstick plant, kelor tree and horse radish tree, it is a source of food and medicine. The dry leaves are reported to have four times the calcium of milk, thrice the potassium of bananas and seven times the vitamin C of oranges. They contain 17% protein, and are a good source of the B vitamins, and contain two to four times the β-carotene found in carrots. 10 Leaves can be eaten fresh, cooked or stored as dried powder for many months without refrigeration and without loss of nutritional value. Moringa oleifera has a long history in medicine. In recent decades, the extracts of leaves, seeds and roots of Moringa oleifera have been extensively studied for many potential uses including wound healing, 11 anti-tumor, 12 antifertility, 13 hypotensive 14 and analgesic activity. 15 The antioxidant activity and the total phenolic contents of Moringa oleifera leaves in two stages of maturity responsible for preventing the deleterious effects of oxidative stress was disclosed in our recent study. 16 Although many benefits of Moringa oleifera have been claimed, only few authentic studies are available. The aim of the present study was to gather further knowledge regarding the effects of Moringa oleifera extracts on cultured KB cells (known to be a sub line of the ubiquitous KERATIN – forming tumor cell line HeLA) in order to establish new pharmacological possibilities for its application. Therefore, the present investigation was undertaken to evaluate the protective effects of the dietary leaf extracts against oxidative DNA damage induced by H2O2 in KB cells. Cytotoxicity was determined by MTT assay and comet assay was used for the assessment of the DNA damage.
Materials and methods
Chemicals
TBA, H2O2, ethidium bromide and 3-(4, 5-dimethlythiazol-2-yl)-2, 5-di phenyl tetrazolium bromide (MTT) were obtained from Sigma-Aldrich (St. Louis, Missouri, USA). Agarose, low-melting agarose, Dulbecco’s modified essential medium (DMEM) and fetal calf serum (FCS) were obtained from Gibco/BRL life Technologies (Germany). Other chemicals and solvents were purchased from Merck Chemicals, Mumbai, India.
Plant material and preparation of extract
Moringa oleifera leaves were collected from the Horticulture Research Institute, Periyakulam, Tamil Nadu, India, and a voucher specimen was deposited in Botanical Survey of India, Coimbatore, India (BSI/SC/5/25/05-06/Tech-908). The leaves were analyzed at two different stages of growth namely tender and mature in order to study whether a difference in the activity existed in different stages of growth. The air-dried leaves of Moringa oleifera both mature and tender were made into a coarse powder. Five hundred grams of dried powders of leaves were stirred with seven parts of distilled water at 80°C for 2 hours separately and cooled down to room temperature. Each supernatant was recovered after filtration and the solvent was completely removed by rotary vacuum evaporator. The extract was freeze dried and stored in a vacuum desicator for further use. The yield of the extract was 32% with reference to the dry starting material. The concentration of aqueous Moringa oleifera leaf extracts that protected 50% against H2O2-induced cell death was determined in order to establish the cytoprotective potential. Hence, the leaf extracts, both mature and tender leaves, were used at a concentration of 50 mg/mL for the assays.
Phenolic composition and total antioxidant activity
As the leaves when subjected to phytochemical screening revealed the presence of various phytoconstituents, the leaf extracts were then examined for the specific phenolic composition by the method of HPTLC. 17 Quercetin, kaempferol and Moringa oleifera leaf extracts were applied to 10 × 10 cm aluminium-packed HPTLC plates coated with 0.2 mm layers of silica gel G60 F254 plates (Merck). The mobile phase was toluene-dioxane-acetic acid 95:25:4. Compounds were visualized by spraying with Naturstoff A reagent. The amount of the compounds in the samples were calculated by comparing the densitogram peak areas from the samples with those of the standards on the same plate. The total antioxidant activity (AOA) of the leaf extracts was determined by using autoxidation of beta-carotene and linoleic acid coupled reaction method 18 and expressed as percentage of inhibition, relative to control.
Cell culture
KB tumor cell line was obtained from the National Centre for Cell Sciences (NCCS), Pune, India. The cells were maintained in DMEM supplemented with 10% fetal bovine serum (FBS) and antibiotics (100 unit/mL of penicillin G and 100 μg/mL of streptomycin sulfate) and cultured at 37°C in 5% CO2. Confluently grown cells were harvested by trypsinization (0.25%), collected into DMEM containing 10% FBS and held on ice till the assay. Before the assay, the cells were spun down at 2000 rpm for 5 minutes at 4°C in a microfuge and resuspended in Hank’s Balanced Salt Solution (HBSS). The dose of H2O2 as 200 µM was selected based on its effectiveness in inducing DNA damage. H2O2 dissolved in phosphate buffered saline (PBS) was used as positive control.
Single-cell gel electrophoresis (the comet assay) using intact cells
The comet assay also called as the ‘single-cell gel assay’ is a technique to detect DNA damage and repair at the level of single cells. Comet assay was performed in KB cells. 19
The treated cells (KB cells with H2O2 in the presence and the absence of the leaf extracts [20 µL of 50 mg/mL]) were incubated at 37°C for 1 hour. At the end of the incubation period, slides were covered with 0.5% normal melting point agarose (NMA) as the first layer, with a mixture of cell suspension and 0.5% of low-melting point agarose (LMA) as the second layer and finally with 0.5% of LMA (without cell) as the third layer. After solidification, the ‘minigels' were placed in cold working lysing solution and incubated overnight at 4°C. The lysed cells were denatured in the alkaline electrophoresis buffer for 20 minutes and electrophoresed in the same buffer at 25 volts for 20 minutes. The slides were then neutralized in 1 M tris pH 7.5 and stained with ethidium bromide. Scored the slides for the presence of comet tails under oil immersion using a Nikon fluorescent microscope. Comet analysis system was applied to quantify the DNA damage. Triplicate samples were prepared and examined. Images of 100 randomly selected cells from each slide were analyzed. The degree of DNA damage was expressed as the tail moment value. Tail moment = (Tail length × Tail DNA%)/100.
Cell viability
Briefly, KB cells (10 000 cells/well) were cultured in collagen I (50 μg/mL) coated 96-well culture plates, in a total volume of 200 μL DMEM-F12 supplemented with 1% FBS. Oxidation was induced by exposing the cells to 200 µM H2O2 in PBS supplemented with 1%
Lipid peroxide formation
Lipid peroxidation (LPO) has been considered as one of the best known manifestations of oxidant-induced cell injury. Lipid peroxidation was followed by measuring the TBARS formed, 21 with minor modifications and the result expressed as nmole of malondialdehyde (MDA)/mg protein. Equal volumes of leaf extract and 200 µM H2O2 in FBS free DMEM were added to each well and the cell plate was incubated for 24 hours. Briefly, KB cells were lysed using a freezing-thawing method. After lysis, 0.2 mL cell suspension was added to the TBA reagent (1.5 mL of 20% acetic acid, 1.5 mL of 8.1% sodium dodecylsulfate and 1.5 mL of 0.8% TBA). This mixture was incubated at 90°C for 1 hour and then cooled. Four milliliters of a mixture of n-butanol and pyridine (15: 1, v/v) was added, and the whole mixture was centrifuged (15 minutes at 1 500 × g). The absorbance of the upper phase was measured at 532 nm.
Antioxidant enzyme assay
Superoxide dismutase (SOD) activity was assayed according to the method described by Crapo et al. 22 in KB cells, with modifications. Briefly, 20 μL of samples with the same amount of protein were mixed with 870 μL of solution A (50 mmol phosphate buffer [pH 7.8] with 0.1 mol ethylenediaminetetraacetic acid, 0.001 N NaOH with 5 μmol xanthine and 2 μmol cytochrome C) and 20 μL of solution B (50 mmol phosphate buffer [pH 7.8] and 0.2 μmol xanthine oxidase). Enzyme activity in the sample was calculated from a standard curve with the range from 0.05 to 12.5 units/mg protein using SOD enzyme (Sigma). One unit of SOD activity is defined as the enzyme concentration required to inhibit chromogen production by 50% in 5 minutes under the assay conditions.
Catalase (CAT) activity was assayed by the method of Aebi 23 in KB cells with modifications. Briefly, 50 µL of samples with the same amount of protein were mixed with 1 mL of 0.01 mol phosphate buffer (pH 7.0) and H2O2. The mixture was immediately read at 240 nm for 1 minute on a spectrophotometer. Changes in absorbance were taken to be proportional to the breakdown of H2O2. Enzyme activity was expressed as units of CAT/mg protein.
Statistical analysis
All parameters studied were subjected to statistical treatment using Sigma Stat statistical package (Version 3.1). The data were expressed as mean ± SD (n = 6) where ‘n’ represents the no of samples. One-way ANOVA, followed by post-hoc analysis using Fischer’s LSD was adopted to all the parameters under study to test the level of statistical significance. The difference was considered significant if p < 0.05.
Results
Phenolic contents and total antioxidant activity
The leaf extracts examined for the phenolic contents revealed the presence of quercetin and kaempferol as presented in Table 1 . The amount of quercetin and kaempferol observed in mature leaf extract were found to be 795 µg/g and 216 µg/g respectively. Whereas the amount of quercetin and kaempferol observed in tender leaf extract were found to be 436.2 µg/g and 115 µg/g. Among the leaf extracts, the mature leaf extract was found to be the richest source of phenolics than the tender leaf extract. Highest antioxidant activity was observed in mature leaf extract than the tender leaf extract. On estimating the levels of polyphenolic compounds in each fraction using standard quercetin, the aqueous fraction was found to have the higher polyphenolic content. Therefore, this extract was used for the study throughout.
Phenolic contents and Total antioxidant activity in Moringa oleifera leaf extract a
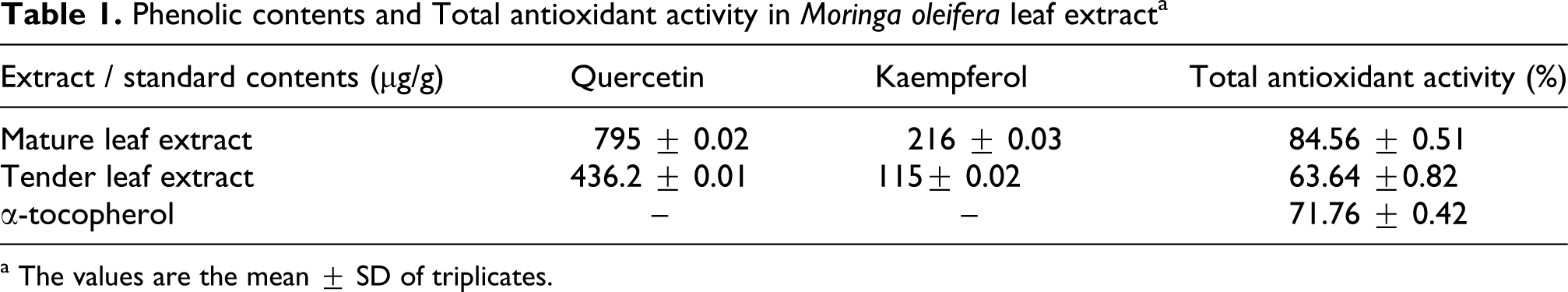
a The values are the mean ± SD of triplicates.
Inhibition of DNA damage in KB cells
The effect of the leaf extracts on DNA damage induced in KB tumor cells by H2O2 was studied using the comet assay. DNA fragmentation analyzed using comet assay is a sensitive method for detecting DNA strand breaks in individual cell and highly efficient in human biomonitoring of natural compound. Quantification of comet data is reported as TDNA (DNA in tail as % of total DNA) and TMOM (tail moment) in Table 2 . The results clearly demonstrate the DNA damage in cells exposed to H2O2. Each cell had the appearance of a comet, with a bright fluorescent head and a tail with an intensity that was related to the amount of damage sustained by the cell. In the H2O2 + leaf extract-treated group, there was a trend of DNA damage reduction in tail comet, which was significant when compared to H2O2 group. The mature and tender leaf extracts of Moringa oleifera significantly reduced the incidence of comets in the oxidant stressed cells.
Comet assay of genomic DNA of KB tumor cells untreated and treated with Moringa oleifera leaf extract

Abbreviations: MLE: mature leaf extract, TLE: tender leaf extract, TDNA: tail DNA, TMOM: tail moment
The values are the mean ± SD of triplicates.
a Significant (p < 0.05) vs untreated control group.
b Significant (p < 0.05) vs to plant control.
c Significant (p < 0.05) vs to H2O2 treated group.
Cell viability
MTT assay, a non-radioactive assay was widely used to quantify cell viability and proliferation. H2O2 caused a marked decrease in the viability of KB cells, whereas mature and tender leaf extracts effectively increased the viability of H2O2-assaulted KB cells. The results summarized in Figure 1 showed that the extracts used at non-toxic concentration in normal cells also exhibited significant inhibitory effect on the proliferation of KB cells.

Effect of Moringa oleifera leaves on viability of KB cells (values are mean ± SD of 3 separate experiments). a p < 0.05 compared to control, b p < 0.05 compared to plant control, c p < 0.05 compared to cells exposed to H2O2 alone.
Effect of leaf extract on lipid per oxidation
An inhibitory effect of leaf extract on H2O2-induced lipid peroxidation was observed through the formation of MDA as shown in Figure 2 . The levels of MDA concentration in H2O2-induced cells were significantly increased compared to control cells. The treatment of cells with the leaf extract significantly reduced the MDA production.

Effect of Moringa oleifera leaves on lipid peroxidation (values are mean ± SD of 3 separate experiments). a p < 0.05 compared to control, b p < 0.05 compared to plant control, c p < 0.05 compared to cells exposed to H2O2 alone.
Effect of leaf extract on antioxidant enzymes
The protective effect of the leaf extract on the levels of the antioxidant enzymes as shown in Figures 3 and 4 reveals that the levels of SOD and CAT in H2O2-induced cells were significantly reduced compared to the control group. However, treatment of the cells with the leaf extract increased the levels of these antioxidant enzymes significantly.

Effect of Moringa oleifera leaves on SOD activity (values are mean ± SD of 3 separate experiments). a p < 0.05 compared to control, b p < 0.05 compared to plant control, c p < 0.05 compared to cells exposed to H2O2 alone.

Effect of Moringa oleifera leaves on CAT activity (values are mean ± SD of 3 separate experiments). a p < 0.05 compared to control, b p < 0.05 compared to plant control, c p < 0.05 compared to cells exposed to H2O2 alone.
Discussion
H2O2 has long been widely used in the pharmaceutical industry as a potent sterilizing agent and it is a well-known bleaching agent. However, it has been found that H2O2 plays an important role in the generation of free radical-induced DNA damage, inducing mutations. 24 H2O2 is one of the major ROS associated with oxidative stress. It readily penetrates into cells and reacts with intracellular metal ions, such as iron or copper, to generate highly reactive hydroxyl radicals that successively attack cellular components including lipids, proteins and DNA to cause a wide variety of oxidative insults. H2O2 has been known to cause DNA damage in the form of single-strand and double-strand breaks. 25
Polyphenols, ascorbates and flavonoids are known to be responsible for antioxidant and free radical scavenging potentials. The total antioxidant activity and the phenolic contents proved the extracts to possess higher values of antioxidant phytochemicals, which suggest that the leaves can be exploited as an important source of natural antioxidants with health-protective potentials. Earlier studies have also shown strong antioxidant and free radical scavenging activities of Moringa oleifera leaves. 26
Oxidative assault to biological system manifests at the cellular level on important biomolecules where the lipid molecules form the primary target and the DNA molecules the ultimate target. Oxidative damage to DNA can result in several consequences including mutagenesis, cancer, age-related disorders and other human pathologies. 27 Foods of plant origin, which are rich in antioxidants, are known to render protection against such diseases. 28 Thus, it is conceivable that antioxidant-rich plant extracts can protect cells against oxidative DNA damage.
Many studies have reported the protection against oxidative DNA damage by herbal extracts and formulations. Oxidative damage mediated as single strand breaks in super coiled PTZ18U plasmid DNA has been reported to be suppressed by 6-gingerol (a phenolic compound in ginger). 29 H2O2-induced damage to super coiled phiX174 DNA was also reported to be reduced by the extracts of Ganoderma lucidum. 30 The phenolics, genistein and resveratrol, inhibited the damage induced by H2O2 in ϕX-174 plasmid DNA. 31 These effects are possibly related to the scavenging activities of the extracts that are rich in Quercetin and kaempferol. 32 Therefore, the extracts of Moringa oleifera leaves were also found to offer very significant protection against oxidative DNA damage under physiological conditions, in this study.
Single-cell gel (SCG) electrophoresis or ‘comet’ assay is a rapid and very sensitive fluorescent microscopic method to examine DNA damage and repair at individual cell level. H2O2 has been shown to affect the DNA directly in terms of strand breaks. DNA fragments produced by single strand breaks manifest themselves as comets upon H2O2 treatment. 33 H2O2-induced DNA damage manifested as comets has been reported to be significantly decreased by many phytochemicals. 34,35 Basal levels of oxidative DNA damage upon treatment with quercetin and caffeic acid documented protection in the comet assay. 36
The results of the present study are also in agreement with the above reports which suggest that the leaf extracts are very effective in counteracting the basal DNA damage. Based on the data obtained from this study, the components present in the Moringa oleifera leaf extracts might be responsible to modulate the hydroxyl radical formation by acting as a direct scavenger. These findings support the use of Moringa oleifera leaves to protect against oxidative DNA damage. It can, thus, be concluded that the consumption of Moringa oleifera leaves can be considered not only to combat pathological conditions but also for promoting general well-being and good health.
Oxidative stress can ultimately lead to cell death including apoptosis. 37 In the present study, the effect of Moringa oleifera leaf extracts on the extent of survival of KB carcinoma cells challenged with H2O2 was followed using the MTT reduction assay. The cell viability decreased significantly with H2O2 treatment but enhanced markedly upon co-treatment with both mature and tender leaves of Moringa oleifera. The MTT assay provides a rapid and versatile method for assessing cell viability. 38 Many herbals and phytochemicals have been reported for their cytoprotective effect using the MTT assay. 39 The methanolic extracts of Areca catechu, Var dulcissima, Paennia suffruticosa, Alpinia officinarum, Glycyrrhiza uralensis and Cinnamomum cassia strongly enhanced the viability against H2O2-induced oxidative damage in V79-4 cells. 40 The MTT assay of Platycodon grandiflorum revealed that it contains a strong polyacetylenic anticancer compound, which exhibited cytotoxicity on three human cancer cell lines (HT-29, Hep G2 and HRT-18). 41 The cytotoxic action of other antioxidants like epigallocatechin gallate on tumor cell lines has also been reported. 42 Genistein, a plant polyphenol, caused human stomach cancer cells to undergo apoptosis. 43
Moringa oleifera leaves also caused a significant reduction in the survival of KB cells, in the absence of H2O2. Wang 44 has reported that the efficiency of compounds to induce apoptosis in cancer cells indicates anticancer activity. In light of all the above reports, it is conceivable that Moringa oleifera leaves may possess anticancer properties. Therefore, the antioxidant effects of herbs on cell viability can be explained by two mechanisms i.e. by direct antioxidant action by scavenging ROS and indirect antioxidant action through the induction of antioxidative enzymes protecting intercellular communications.
LPO has been used as a reliable marker of oxidative stress, both in vitro and in vivo. Several plant extracts have been shown to inhibit LPO as measured by the levels of TBARS. ROS and RNS (reactive nitrogen species) may initiate and propagate lipid peroxidation, of which, the end product MDA possesses genotoxic and mutagenic properties. 45 Lipid peroxidation is a key process in many pathological events and is one of the reactions induced by oxidative stress. Increased MDA accumulation has been noted in response to H2O2 and the cytotoxic effects of H2O2 on KB cells were shown by its strong inhibition of cell growth and MDA formation. These results indicate that the leaf extract is capable of reducing H2O2-induced cytotoxicity and lipid peroxidation. Thus, the prevented lipid peroxidation may explain its cytoprotective property on the cell membrane damage caused by the radicals.
Superoxide dismutase and catalase can act as anticarcinogens and inhibitors at initiation and promotion/transformation stage in carcinogenesis and have a vital antioxidant role in human health, conferred by their scavenging of the free radicals. 46 Treatment with the leaf extract significantly increased the levels of the antioxidant enzymes SOD and CAT. They maintain the defensive mechanism and cellular fluidity evidenced by the increased values of these antioxidant enzymes. The result of this study state that Moringa oleifera leaf extracts can act as effective modulators in reducing the toxicity by enhancing the stimulation of enzymes in the cells under stress.
In conclusion, the results demonstrated that both plasmid and cellular DNA damage caused by H2O2 can be reduced by Moringa oleifera extracts iterates the scavenging capacity of the extracts. The assays on human tumor cell lines demonstrates its antiproliferative activity and thus these findings suggest Moringa oleifera may be useful as a cancer chemopreventive and chemotherapeutic agent and may justify further investigation of other possible beneficial biological properties.
Footnotes
Acknowledgements
I thank Miss Jothipriya and Inbavalli for helping me in this research work and I declare no conflicts.
This research received no specific grant from any funding agency in the public, commercial, or from non-profit sectors.
