Abstract
The consumption of a high-fat diet (HFD) is considered a risk factor for obesity development. Nonetheless, a causal role of dietary fat has never been documented, because of inadequate animal models. In our study, one group of rats was fed with standard rat diet, while other group of rats fed with high-fat diet for 4 weeks. After 4 weeks of feeding, the hemodynamic parameters in the rats fed with HFD were significantly increased as compared with control rats. Rats fed with HFD had elevated levels of serum lipids, insulin, leptin, glucose and apolipoprotein B. Lipid peroxides and caspase-3 levels were increased while serum apolipoprotein A1 and antioxidant enzymes levels in heart tissues were decreased in HFD-induced obesity in rats as compared to normal healthy control rats fed on standard rat pellet diet. This model of diet-induced obesity will be a useful tool for studying the mechanisms by which dietary fat induces the obesity in humans.
Introduction
Obesity has become a global health epidemic which accompanied with dyslipidemia, diabetes, insulin resistance, hypertension, stroke and arteriosclerosis. The prevalence of obesity in the United States has increased; presently, one-third of American adults are considered obese. Among other factors, obesity is an important contributor to essential hypertension in humans. Data from the Framingham Heart Study suggest that 78% of essential hypertension in men and 65% of essential hypertension in women can be directly attributed to obesity. 1 However, the mechanism that links obesity with high blood pressure and altered renal function has not been fully elucidated.
Despite considerable efforts by scientists and health care professionals to understand and successfully treat obesity, its incidence continues to rise and the obesity-related health costs are staggering. 2 The trend toward greater obesity in the young raises concern because infant or childhood obesity alone significantly increases susceptibility to adult chronic diseases, including cardiovascular disease, hypertension and diabetes. 3 Although, it is clear that genetic factors contribute to the propensity of an individual to become obese, the striking increase in overweight that occurs as previously underdeveloped countries modernize and the continued growth in the numbers of obese individuals in developed countries indicate an important role for environmental factors as well. The consumption of a high-energy density, high-fat diet (HFD) 4 is thought to be one of the main factors.
The epidemiological studies have showed a significant positive correlation between mean dietary fat intake and the incidence of obesity and its related complications and risk factors. 5,6 Furthermore, as the mean amount of fat in the diet increases, the incidence of obesity also increases. 7 The association of dietary fat with the incidence of obesity is exaggerated in some genetically isolated populations, such as the Pima Indians. 8 Recent studies have shown that consumption of dietary fats promote hypothalamic resistance to the main anorexigenic hormones, leptin and insulin, leading to the progressive loss of the balance between food intake and thermogenesis and, therefore, resulting in body mass gain. 9,10
Although diets high in carbohydrates may also predispose an individual to obesity, our focus was on the effects of diet high in fat because of the epidemiological evidence implicating fat in both obesity and public health concerns. What is not clear is whether or how dietary fat per se is the predisposing factor, in particular because the incidence of obesity has tended to increase in the face of a slightly decreased mean fat intake in the United States. 11
A number of studies have demonstrated that antioxidants may act as a regulator of obesity in mice or rats with HFD. Obesity has been shown to be one of the conditions that decrease antioxidant capacity. 12 Obesity seems to decrease antioxidant defense by lowering the levels of antioxidant enzymes (catalase, glutathione peroxidase [GPx] and glutathione reductase [GR]). 13
More specifically, we described a model that controls key variables to better reach unambiguous conclusions regarding the role of dietary fat in the etiology of obesity. Identifying the regulatory processes that mediate HFD-induced obesity is of fundamental importance and will require a well-established and controlled animal model.
The experiments described in this report were to establish such a model and to determine whether the rats fed with the HFD share important characteristics with obese humans. 4 The purpose of the present study was to document the development of animal model and to partially characterize the model with respect to some factors likely to be involved in the cardiovascular changes associated with obesity.
Materials and methods
Animals
The study was approved by the Institutional Animal Ethics Committee (IAEC) of Hamdard University, New Delhi, which is registered with Committee for the Purpose of Control & Supervision of Experiments on Animals (CPCSEA), Government of India, India (Registration No. 173/CPCSEA, 28 Jan 2000). Wistar male albino rats, weighing 150−200 g, were procured from the Central Animal House Facility, Hamdard University, New Delhi, and acclimatized under standard laboratory conditions at 25 ± 2°C, and relative humidity (50% ± 15%) and normal. The animals were kept in polypropylene cages under standard laboratory conditions (12 hour light and 12 hour dark: day: night cycle) and had a free access to tap water ad libitum.
Experimental design
After acclimatization, all the animals were randomly divided into two groups of eight animals each and were treated as follows: Group I (normal healthy control) – rats fed with normal rat pellet diet (12.5% lipids, 62.3% carbohydrate and 24.3% protein; Amrut rat feed, Mfd by: Pranav Agro Industries Ltd, Maharashtra, India) for 28 days; Group II (pathogenic control) – rats fed with HFD (42% lipids, 36% carbohydrate and 22% protein) for 28 days. HFD was purchased from National Centre for Laboratory Animal Sciences (NCLAS), National Institute of Nutrition (NIN), Hyderabad, Andhra Pradesh, India. All other chemicals used were of analytical grade. Double distilled water was used for all biochemical assays.
Body weight gain, food and water intake measurement
Body weight was measured weekly, body weight change measured from difference between final body weight and initial body weight. Food intake was estimated by subtracting the amount of food left in the cages from the total amount of food provided to each rat. 14
Measurement of hemodynamic parameters
Hemodynamic parameters (systolic, diastolic, mean arterial blood pressure, heart rate) were measured by Non-Invasive Blood Pressure Recorder using rat tail-cuff method (Kent Scientific Corporation, Torrington, Connecticut, USA).
Measurement of serum parameters
Blood was collected from the retro-orbital plexus of both the groups of overnight fasted rats using microcapillary tubes on 29th day. Serum was separated by centrifugation (4000 rpm, 10 min) and transferred to Eppendorf tubes. After serum collection, all animals were killed by cervical dislocation; hearts and visceral fat pad weights (mesenteric, perirenal and epididymal fats) were dissected out and weighed. The concentrations of lactate dehydrogenase (LDH; Reckon Diagnostics Pvt Ltd, Baroda, Gujarat, India), glucose, total cholesterol (TC), triglycerides (TGs; all the three from Span Diagnostics Ltd, Surat, Gujarat, India) and high-density lipoprotein-cholesterol (HDL-C; Reckon Diagnostics Pvt Ltd, Baroda, Gujarat, India) in serum were measured with commercial kits. The concentrations of insulin, leptin and apolipoproteins-A1 and B in the serum were measured, respectively, with rat insulin ELISA kit (Alpco Diagnostics, Salem, USA), rat leptin ELISA kit (BioVendor, Czech Republic) and apo-A1 and B, immunoturbidimetric immunoaasay kit (Randox Laboratories Ltd, Antrim, UK).
Determination of antioxidant enzymes in heart tissue
All antioxidant enzyme activities were determined after six rat hearts tissues homogenized with phosphate buffer saline at a pH of 7·4. Glutathione (GSH), 15 glutathione peroxidase (GPx), 16 glutathione reductase (GR), 17 glutathione-S-transferase (GST), 18 superoxide dismutase (SOD) 19 and catalase 20 activities were estimated, respectively.
Determination of lipid peroxidation, sodium potassium ATPase and caspase-3 activity in heart tissue
Lipid peroxidation was determined with spectrophotometric measurement of the amount of malondialdehyde equivalents with thiobarbituric acid and was expressed as thiobarbituric acid-reactive substances (TBARS; nmol malondialdehyde/mg protein) 21 and sodium potassium ATPase activity was measured in heart tissue by method of Bonting. 22
The reaction mixture consisted of 100 µL tissue homogenate incubated in a medium containing Tris-HCl (75 mM, pH-7.5), NaCl (600 mM), KCl (50 mM), MgCl2 (5 mM), EDTA (1 mM) and ATP (3 mM) for 30 min at 30°C. The reaction was arrested by the addition of trichloro acetic acid (TCA). Na+/K+ ATPase activity is expressed as µmoles of phosphorous liberated/min per mg of protein.
Caspase-3 activity was measured using Caspase-3/CPP32 Colorimetric Assay Kit (BioVision, Mountain View, California, USA). A total of 50 µL supernatant from homogenized tissue with cooled lysis buffer was taken from each sample and 50 μL of 2X Reaction Buffer (containing 10 mM Dithiothreitol [DTT]) was added to each sample. Then, 5 μL of the 4 mM DEVD-pNA substrate (200 μM final conc.) was added and incubated at 37°C for 1−2 hours to allow a dissociation of p-nitroanilide (pNA) from the conjugate DEVD-pNA. CPP-32 activity was measured spectrophotometrically at 405 nm using a 100-μL micro quartz cuvet (Sigma, St. Louis, MO, USA) or sample was diluted to 1 mL with Dilution Buffer. Caspase-3 activity was calculated as nmole/hour per mg protein. 23
Histopathological studies
The studied animals were killed and two rat hearts tissue samples were collected, fixed in 10% formalin buffered solution, cut into 5-µm sections and stained with haematoxylin/eosin. The sections of heart tissues were studied to determine the level of tissue damage by HFD.
Statistical analysis
Statistical analysis was carried out using Graphpad Prism 3.0 (Graphpad software, San Diego, California, USA). All of the data were expressed as mean ± SEM. Statistical analysis was performed by Dunnett’s t-test and p < 0.05 were considered to be significant.
Results
Results of our research work show that HFD-induced obesity results in an increase in systolic (from 127.83 ± 6.86 to 165.83 ± 10.33 mmHg; p < 0.01), diastolic (from 96 ± 4.65 to 116.66 ± 5.7 mmHg; p < 0.05) and mean arterial blood pressures (BP) (from 106.16 ± 5.00 to 132.66 ± 7.1 mmHg; p < 0.01) as well as increase in heart rate (from 421.83 ± 15.77 to 687 ± 67.83 bpm) in HFD-treated rats as compared to normal healthy control rats as recorded by Non-Invasive Blood Pressure recorder using rat tail cuff method (Table 1).
Effect of high-fat diet on hemodynamic changes in albino rats by non-invasive bp (NIBP) recorder using tail-cuff method a

Abbreviations: BP: blood pressures, HFD: high-fat diet.
a All values were expressed as mean ± SEM for eight rats in each group.
b p < 0.05 as compared to normal healthy control group (group I).
c p < 0.01 as compared to normal healthy control group (group I).
HFD group (group II), shows significant (p < 0.01) increase in body weight gain than those of the normal healthy control group (group I), after 4 week of treatment. Food intake in HFD group was higher than that of normal healthy control group (Table 2).
Effect of high-fat diet on food, water intake, body weight gain, heart weight and visceral fat pad weights in albino rats a

Abbreviation: HFD: high-fat diet.
a All values were expressed as mean ± SEM for six rats in each group.
b p < 0.05 as compared to normal healthy control group (group I).
c p < 0.01 as compared to normal healthy control group (group I).
Serum TC, TGs, low-density lipoprotein-cholesterol (LDL-C), very low-density lipoprotein cholesterol (VLDL-C), atherogenic indexes (TC/HDL-C and LDL-C/HDL-C) and apolipoprotein-B levels in HFD group (group II) were significantly (p < 0.01) increased as compared to the normal healthy control group (group I), while HDL-C and apolipoprotein-A1 levels were significantly reduced in group II as compared to group I (Tables 3 and 4). Serum insulin, glucose and LDH levels were significantly (p < 0.01) increased in HFD-treated group as compared to the normal healthy control group. Serum leptin level in HFD-treated group was significantly (p < 0.05) increased as compared those in normal healthy control group.
Effect of high-fat diet on serum total cholesterol (TC), high-density lipoprotein-cholesterol (HDL-C), low-density lipoprotein-cholesterol (LDL-C), very low-density lipoprotein-cholesterol (VLDL-C) and triglycerides (TGs) levels in albino rats a
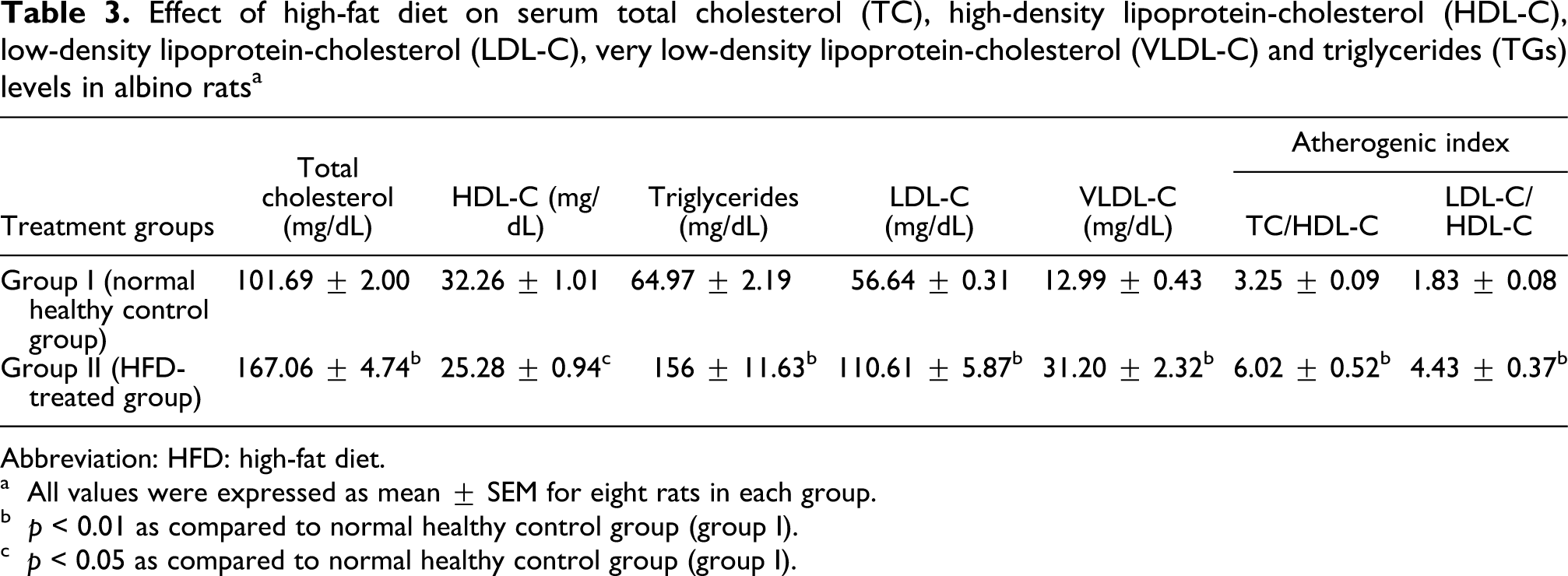
Abbreviation: HFD: high-fat diet.
a All values were expressed as mean ± SEM for eight rats in each group.
b p < 0.01 as compared to normal healthy control group (group I).
c p < 0.05 as compared to normal healthy control group (group I).
Effect of high-fat diet on serum leptin, insulin, glucose, apolipoprotein-a1 and b (Apo-A1 and B) and lactate dehydrogenase (LDH) levels in albino rats a

Abbreviation: HFD: high-fat diet.
a All values were expressed as mean ± SEM for eight rats in each group.
b p < 0.05 as compared to normal healthy control group (group I).
c p < 0.01 as compared to normal healthy control group (group I).
HFD group shows a significant reduction in the activities of antioxidant enzymes in the cardiac tissue of rats with obesity induced by HFD. The glutathione-dependent antioxidant enzymes (GPx, GR, GST), catalase and superoxide dismutase activities were significantly (p < 0.01) reduced in the HFD group when compared with normal healthy control group (Tables 5 and 6). Lipid peroxide (TBARS) levels were significantly (p < 0.01) increased in HFD-fed rats as compared to the normal healthy control rats. Sodium potassium ATPase activity was reduced significantly (p < 0.01) in the rats fed with HFD as compared to the normal healthy control rats. The mean caspase-3 levels were significantly (p < 0.01) increased by 3.4-folds in HFD-fed rats as compared to normal healthy control rats (Table 6).
Effect of high-fat diet on cardiac tissue glutathione, glutathione peroxidase (GPx), glutathione reductase (GR) and glutathione-S-transferase (GST) levels in albino rats a

Abbreviation: CDNB: 1-Chloro-2, 4-dinitrobenzene; HFD: high-fat diet; NADPH: Nicotinamide Adenine Dinucleotide Phosphate.
a All values were expressed as mean ± SEM for six rats in each group.
b p < 0.01 as compared to normal healthy control group (group I).
Effect of high-fat diet on cardiac tissue catalase (CAT), superoxide dismutase (SOD), lipid peroxides (LPO), sodium potassium ATPase (Na+-K+ ATPase) and caspase-3 levels in albino rats a

Abbreviations: HFD: high-fat diet, TBARS: thiobarbituric acid-reactive substances, MDA: malondialdehyde.
a All values were expressed as mean ± SEM for six rats in each group.
b p < 0.01 as compared to normal healthy control group (group I).
Heart and visceral fat pad weights (mesenteric, perirenal and epididymal fats) were significantly (p < 0.05) increased in HFD-fed rats as compared to the normal healthy control rats (Table 2).
Histopathological observation of rat’s heart tissue of HFD group showed deposition of fat globules in myocardial cells with marked fatty changes in portal region than in peripheral region as compared to normal healthy control rat’s heart tissue which showed normal architecture with regular morphology of myocardial cells and no fatty changes (Figure 1A and B).
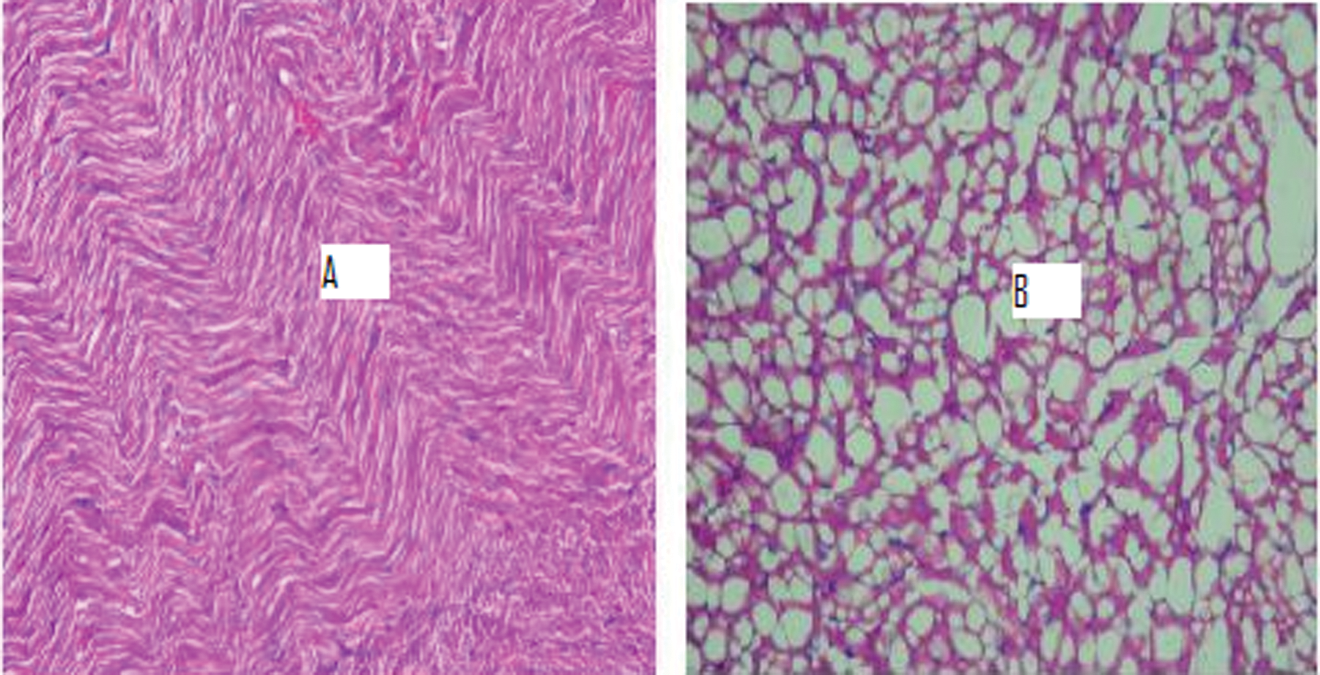
(A and B) Photomicrograph of heart tissue (n = 2) of normal healthy control rats showed normal architecture with regular morphology of myocardial cells while high-fat diet-fed rats showed deposition of fat globules in myocardial cells with marked fatty changes in portal region than in peripheral region. (Magnification ×400 and scale bar: 100 µm).
Discussion
A model of diet-induced obesity in rats is well controlled and shares many features with human obesity. Dietary fat is one of the most important environmental factors associated with the incidence of cardiovascular diseases (CVD); high cholesterol and saturated fat diets have been shown to promote atherosclerosis. 24 A rodent model of obesity based on the intake of HFD is advantageous in studying obesity-related cardiovascular abnormalities. 25 In the present study, body weight gain was higher in rats fed with HFD as compared to the normal healthy control rats. Matsuo et al 26 reported that body weight gain was greater in beef tallow diet group than in the other dietary groups.
Results of our research work show that HFD-induced obesity in rats resulted in a significant increase in systolic, diastolic and mean arterial blood pressures as well as increase in heart rate in HFD-treated rats as compared to normal healthy control rats. This suggests that a short period of high fat diet intake may increase Ca2+ channel numbers or alter channel regulation, leading to increased transmembrane Ca2+ influx. Elevation in Ca2+current density is associated with significantly elevated blood pressure. 27 The results of our research work showing increase in systolic BP (from 127.83 ± 6.96 mmHg to 165.83 ± 10.33 mmHg) and diastolic BP (from 96 ± 4.65 mmHg to 116.66 ± 7.1 mm Hg) corroborates with the study of Aubin et al. 28 They reported that in rats fed a HFD, systolic BP (171 ± 7 mmHg) and diastolic BP (109 ± 3 mmHg) were increased as compared to a standard diet-fed rats (systolic BP, 134 ± 8 mmHg; diastolic BP, 96 ± 5 mmHg).
In the present study, heart and visceral fat pad weights were significantly affected by HFD in HFD group. The visceral fat pads (i.e. mesenteric, perirenal and epididymal fat) of HFD rats weighed 50% more than those of normal healthy control rats as feeding of HFD in rats increases body weight, adiposity and visceral fat deposition. 29 The present study was designed to establish whether alterations due to HFD in rats would result in different levels of dyslipidemia and oxidative stress. HFD is a good source for the induction of obesity in normal healthy rats. Many studies indicated that obesity is induced by a high-energy diet containing 40% beef tallow. 30,31 We found that HFD for a period of 4 weeks produced significant increase in the serum TC, TGs and LDL-C levels in HFD-induced obesity in rats. Serum HDL-C levels were decreased in the HFD group rats as compared to the normal healthy control group rats. Our study results are supported by the work of Lavie and Milani, 32 which indicated that obesity adversely affects plasma lipids, especially by increasing TC and decreasing the levels of HDL-C. The HFD might lead to an increase in the synthesis of phospholipids and cholesterol esters in rats. 33 Hyson et al 34 indicated that the blood levels of LDL-C and its oxidation are related to cardiovascular risk.
Another early detectable parameter in our model is lipid peroxidation. Free radicals are known to be involved in a variety of human pathologies, including atherosclerosis, 35 obesity 36 and hypertension. 37 A potential mechanism for the generation of free radicals may be the activation of β-adrenergic receptors reported for obesity-prone rats. 38 This could increase lipolysis to yield free fatty acids that are able to uncouple the mitochondrial phosphorylation and further generate free radicals. 39
In animal and human studies, obesity is associated with a decrease in tissue or plasma antioxidant capacity. 40 GSH constitutes the first line of defense against free radicals and is also responsible for the maintenance of protein thiols and acts as a substrate for GPx and GST. 41 The present data indicate that GSH content was depleted in the rats with obesity induced by a HFD. Enzymatic antioxidants, such as superoxide dismutase, catalase or GPx, GR and GST can scavenge reactive oxygen species and free radicals or prevent their formation. 42 The results of present study showed that antioxidant enzyme activities (SOD, catalase, GSH, GPx, GR and GST) in the HFD group (i.e. group II) were significantly reduced as compared to the normal healthy control group (i.e. group I). HFD-induced obesity is associated with insulin resistance, cardiac hypertrophy and myocardial apoptosis. Caspase-3 is a key enzyme involved in the caspase-dependant apoptotic pathway. It has been reported by Fang et al 43 that HFD feeding triggered elevated myocardial apoptosis, as assessed by elevated caspase-3 activity. In the present study, caspase-3 activity in heart tissue increased in HFD-fed rats, which corroborates with the findings of Fang et al. 43
Apolipoprotein A1 (Apo-A1) is associated with HDL, having several antiatherogenic properties. Apolipoprotein B (Apo-B) is associated with low-density lipoprotein, intermediate-density lipoprotein (IDL), very low-density lipoprotein (VLDL) and chylomicrons. 44 Serum Apo-B levels were significantly increased in HFD rats as compared to the normal healthy control rats while serum Apo-A1 levels significantly decreased in HFD rats as compared to the normal healthy control rats. Apo-B secretion by the liver is regulated by factors such as rate of cholesterol biosynthesis, availability of triglycerides and cholesterol esters. 45 Measurement of the levels of the important lipoproteins such as Apo-A1 and Apo-B can throw light on lipid metabolism and the effect of various drugs on the same. Additionally, the Apolipoprotein-A1 class of lipoprotein, besides being associated with HDL particle, is known to play important antioxidant and anti-inflammatory roles in atherosclerosis. 46
The obese rats are both hyperleptinemic and hyperinsulinemic, and as occurs in humans, both plasma insulin and leptin concentrations were directly correlated with the degree of adiposity. In all likelihood, the hyperinsulinemia in the HFD-fed rats was a result of insulin resistance, a common feature of human obesity and one that is central to the development of diabetes and cardiovascular disease. Serum insulin level was significantly (p < 0.01) increased in HFD group as compared to the normal control healthy group. This may be due to insulin resistance caused by HFD. Mehta et al 47 indicated that HFD lead to insulin resistance through oxidative stress. In another study, serum insulin levels were higher in the HFD group as compared to the normal control healthy rats. 24 Serum leptin level in the HFD group was significantly (p < 0.05) increased as compared with those in the normal control group. Fried et al 48 indicated that basal levels of leptin are known to be strongly positively correlated with body fat on a HFD. The report indicated that leptin might contribute to hepatic steatosis by promoting insulin resistance and also by altering insulin signaling in hepatocytes, so as to promote increased intracellular fatty acids. 49
HFD damages cell membrane as evident from significant decrease in levels of membrane-bound enzymes like Na+/K+ ATPase as compared to normal control (p < 0.01). Our results are in agreement with the findings of Takeuchi et al 50 who reported the activity of Na+-K+-ATPase in the liver and skeletal muscle was lower in rats fed with the lard diet. The decrease of ATPases could be due to enhanced lipid peroxidation by free radicals. Since this membrane-bound enzymes are ‘SH’ group-containing enzymes, so are they lipid-dependant. In our study, HFD decreases the sodium–potassium ATPase activity in HFD-induced obesity in rats as compared to the normal healthy control rats. Previous investigations have suggested that sodium-potassium–ATPase activity is lower in severely obese patients than in normal controls. 51
In the histology study, heart tissue of HFD-fed rats showed deposition of fat globules in myocardial cells with marked fatty changes in portal region as compared to normal healthy control rat’s heart tissue, which showed normal architecture with regular morphology of myocardial cell.
The results of the present study confirmed that administration of HFD produced significant obesity in rats as indicated by increased body weight gain, hemodynamic changes and an increase in levels of serum lipids, insulin, leptin and Apo-B levels and decrease in serum Apo-A1 and HDL-C levels, cardiac Na+/K+ ATPase activity and antioxidant levels that were further supported by histopathological findings.
Conclusion
In conclusion, we partially characterized a rat model of diet-induced obesity that also develops hypertension. The advantages over other similar animal models are the close similarity to human obesity hypertension, namely activation of rennin angiotensin system (RAS), dyslipidemia and increased oxidative stress. The model may also be useful to study other pathologies associated with obesity, such as atherosclerosis.
Footnotes
The study was supported by major research project grant to Dr. Uma Bhandari by University Grants Commission, New Delhi, India.
