Abstract
Metal ferrites like nickel ferrite, copper ferrite, zinc ferrite, cobalt ferrite, and other ferrites are highly abundant and used as catalysts in various transformations. However, CaFe2O4 is the most abundant alkali metal ferrite, which is eco-friendly and non-toxic. CaFe2O4 is a superparamagnetic material that can be easily recovered from the reaction media due to its magnetic properties. Therefore, it has been widely used in numerous applications, such as dye degradation, removal of heavy metal ions, transesterification reaction, gas sensing, photocatalytic water splitting, drug delivery, etc. In this review, (1) magnetic properties and crystal structure of spinal CaFe2O4 are discussed, (2) potential applications of CaFe2O4 are discussed, and (3) various synthesis techniques of CaFe2O4 are demonstrated. CaFe2O4 shows photocatalytic properties due to its narrow band gap (1.9 eV), abundant functional group, and high surface area. It is found that CaFe2O4 possesses a remarkable potential for energy and environmental remediation. In this review, we have added the photocatalytic mechanism of various pollutants. At last, future perspectives are given for developing novel, sustainable CaFe2O4 or CaFe2O4-based nanocomposite.
Keywords
Introduction
Metal ferrite nanoparticles (MFNPs) have been in focus because of their wide range of applications in various disciplines, such as high-density magnetic recording, microwave devices, ceramic coating, and environmental protection.1–5 Metal oxides having a general formula of AB2O4 with a spinel structure are defined as spinel ferrite nanoparticles (SFNPs). In AB2O4, A represents metal cations (Cu, Zn, Mn, Ca, Mg, Ba) present at tetrahedral sites, whereas B is present at octahedral sites, with iron cation being the most common. It should be noted that the metal cations at both locations are tetrahedrally and octahedrally coupled to oxygen atoms, correspondingly. Most of the MFNPs that have been produced display ferromagnetic, ferrimagnetic, and superparamagnetic characteristics. Special attention is being paid to superparamagnetic MFNPs, which have a significant role in wastewater treatment and other applications. 6 Spinels are excellent photocatalysts, and their discovery has sparked new interest in conserving the environment 7 and transforming solar energy into usable forms. 8 At temperatures underneath the Neel temperature (180 K), the oxide was discovered to be superparamagnetic 9 with an insulator characteristic. 10
Calcium ferrite (CF) is one of the most important members of the spinel family because of its abundant nature and eco-friendly behavior. CFNPs contain superparamagnetic behavior and work as excellent catalysts for numerous applications. For the removal of dyes from the aqua matrix, the adsorption performance of magnetic CFNPs has been investigated since CFNPs are believed to be more benign than other ferrites due to their environmental friendliness and non-toxic behavior. CF has grown in popularity because it contains photoelectrochemical characteristics. For the first time, the photoelectrochemical aspects of CFNPs are examined to evaluate their photocatalytic activity. 11 Photocatalysis is effective at mineralizing organic contaminants (dyes, drugs, pesticides, and so on).12,13 CF has specific properties, such as excellent thermal and chemical stability, optical characteristics, and superparamagnetic properties, making it a significant spinel ferrite. CFNPs might also be a contender for large-scale use as an adsorbent because of their great mechanical and chemical stability as well as ease of magnetic separation due to their superparamagnetic activity. Furthermore, calcium is abundant in nature and non-toxic, which is why it is grabbing the attention of researchers. 14 CFs are employed in ceramics, photocatalysis, and supraparamagnetic applications. 15
CF is a p-type semiconductor that has approximately 1.9 eV band gap. Since superparamagnetic CFNPs do not coagulate without an external magnetic field, they may diffuse in water. They did, however, become magnetic particles with an external magnetic field and can be readily separated.16,17 The band gap values of CF may absorb visible light, but they are also predicted to effectively create hydroxyl radicals. Because of this property, it is sensitive to visible light. With the n-type semiconductors, CFNPs form numerous composites, forming p-n heterojunctions. 18 The formation of p-n type heterojunction enhances the photocatalytic activity of the nanocomposites and makes them more applicable for charge carrier separation. 19
A broad range of synthesis techniques has been described, such as combustion, precipitation, thermal decomposition, solvothermal, sonochemical, sol-gel, hydrothermal, green synthesis, and microwave-assisted methods, which have all been utilized to synthesize CFNPs of various sizes as well as morphologies.20–22 The porosity, as well as the morphology, of photocatalysts is influenced by the procedures of synthesis as well as the different temperatures of calcination. The photocatalytic activity and magnetic behavior of CFNPs depend on the size as well as the method of fabrication. As a result, the photocatalytic efficacy of CFNPs produced using various reaction parameters and techniques varies. Nevertheless, the biosynthetic procedure for producing the CF photocatalyst would be preferable. Hen eggshells and iron rust were used for the green synthesis of CFNPs followed by the milling method. 23 Additionally, in particular, CF synthesized at 800°C degrades a wide range of organic pollutants, including methylene orange, tetracycline hydrochloride, rhodamine B, and humic acid. It gives stable performance for eight cycles, including without leaching of iron particles, and with sunlight and natural water, it shows good degradation efficiency. 24 Additionally, this approach provides several benefits, including a higher yield of homogenous size and morphology and a shorter reaction time under mild reaction conditions. Despite this, the majority of CFNPs produced had ferromagnetic, ferrimagnetic, as well as superparamagnetic characteristics.6,25 CFNPs might also be a possible contender as an adsorbent for large-scale use due to their outstanding chemical and mechanical stability, as well as their simple magnetic separation due to their superparamagnetic activity. The adsorption ability of adsorbents is regulated by their characteristics, such as specific surface area, presence of functional groups, and pore size distribution. 26 The crystalline structure of CF promotes greater effectiveness by providing additional active surface area.
There are several reviews on NiFe2O4, ZnFe2O4, and CuFe2O4.2,6,27 However, to the best of our knowledge, there is no review based on CF. Although there are several studies on this topic due to its unique physicochemical characteristics. So, this review includes a brief study of CFNPs and their numerous binary and ternary nanocomposites. In addition to that, the magnetic properties, synthetic procedure, photocatalytic behavior, and adsorption activity have been discussed. Various modification techniques are used to enhance the properties of CFNPs. The application of CF-based composites in multiple areas, such as water decontamination, energy production, transesterification reaction, and medical application, is explained. In conclusion, we have added future perspectives for the effective development of CF-based composites for young and dynamic researchers.
Magnetic properties and structure of CaFe2O4
The electromagnetic properties and structure of CFNPs depend entirely on the size and method of fabrication. 28 CF has an unusual crystal structure that includes FeO6 octahedra that share both corners and edges, as well as zigzag Fe chains that are arranged in a beehive tube network. The nominal CF was found to have a long-range antiferromagnetic spin ordering at Néel temperature TN = 180 K. 29 CFNPs do show superparamagnetic behavior at 300 K. For the cubic spinel phase, saturation magnetization rises with particle size, which is another property of superparamagnetic behavior. On increasing the size of particles, an increase in saturation magnetization has been obtained, which indicates the superparamagnetic characteristic of CFNPs. 30 The anomalous magnetic structure of CF was the topic of controversy in the initial studies, but it is still not fully considered. 28 Since Stock et al.'s neutron diffraction experiments on CF single crystals, there has been an increased focus on the area.31,32
The Fe3+ ions in the CaFe2O4 structure are crystallographically separated into two locations, Fe(1) and Fe(2), each accompanied in octahedral coordination by six oxygen atoms, forming zigzag chains parallel to the b-axis. As seen in Figure 1, FeO6 octahedral within the same chain share edges while corners link adjacent chains. The magnetic coupling between spins occurs, as in numerous oxides, through the oxygen-mediated superexchange, whose strength and sign are determined by the Fe-O-Fe bond angles. Inter-chain antiferromagnetic interactions with corner-sharing Fe(1)O6 and Fe(2)O6 octahedra with bond angles of around 120° were discovered. Relatively weak couplings, J1, and J2 occur within edge-sharing FeO6 octahedral inside the similar zigzag chain, which is joined by angles of around 100°C (Figure 2). Das et al. 29 recently proposed that CF's magnetic structure may be seen as an armchair-like structure sloping downward along the a-axis. Nevertheless, there is not a consensus on how to evaluate magnetic susceptibility data in the scientific literature. Oxygen seems to have a significant effect on the magnetic characteristics of CF.
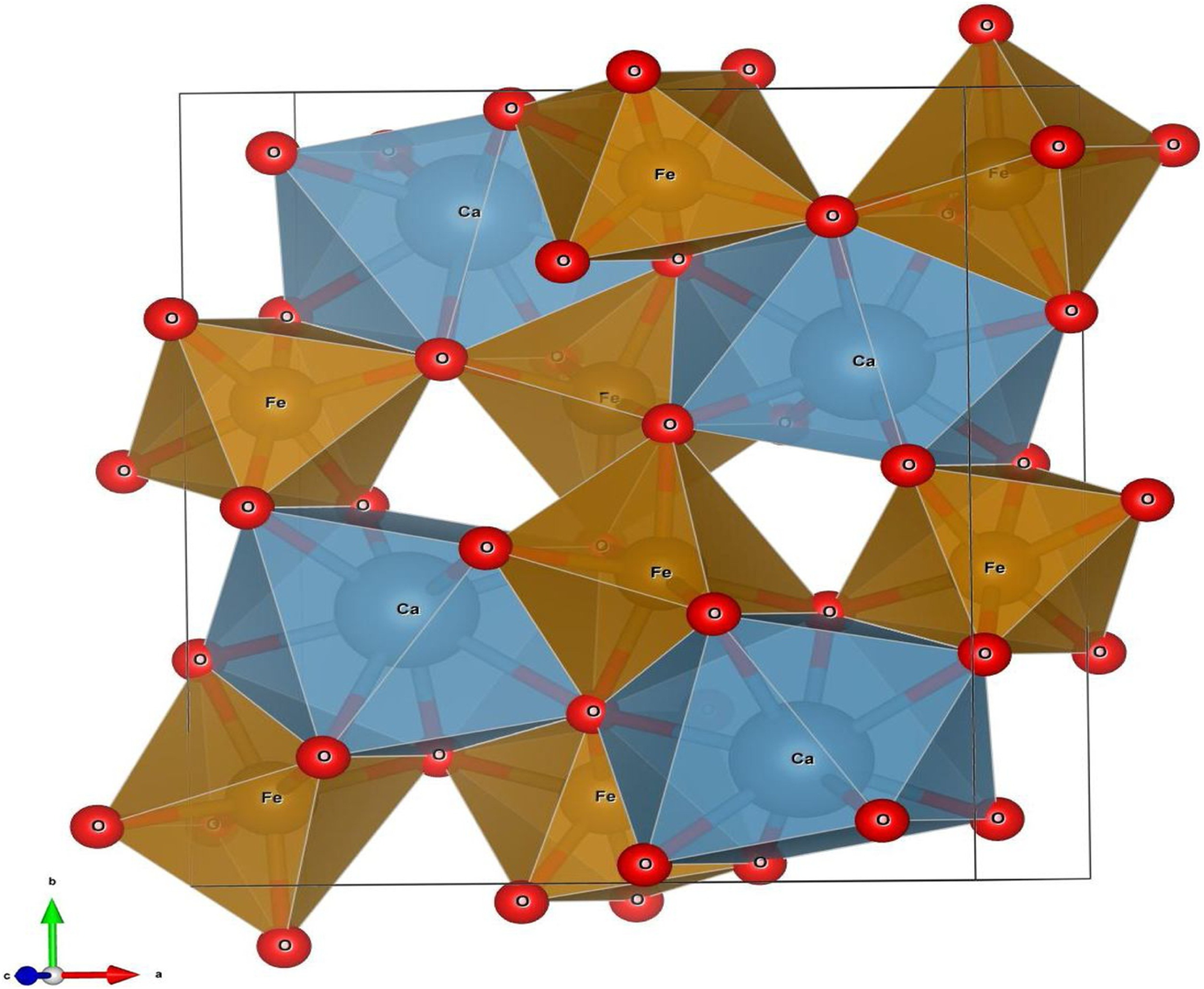
Structure of CaFe2O4 spinel. 33
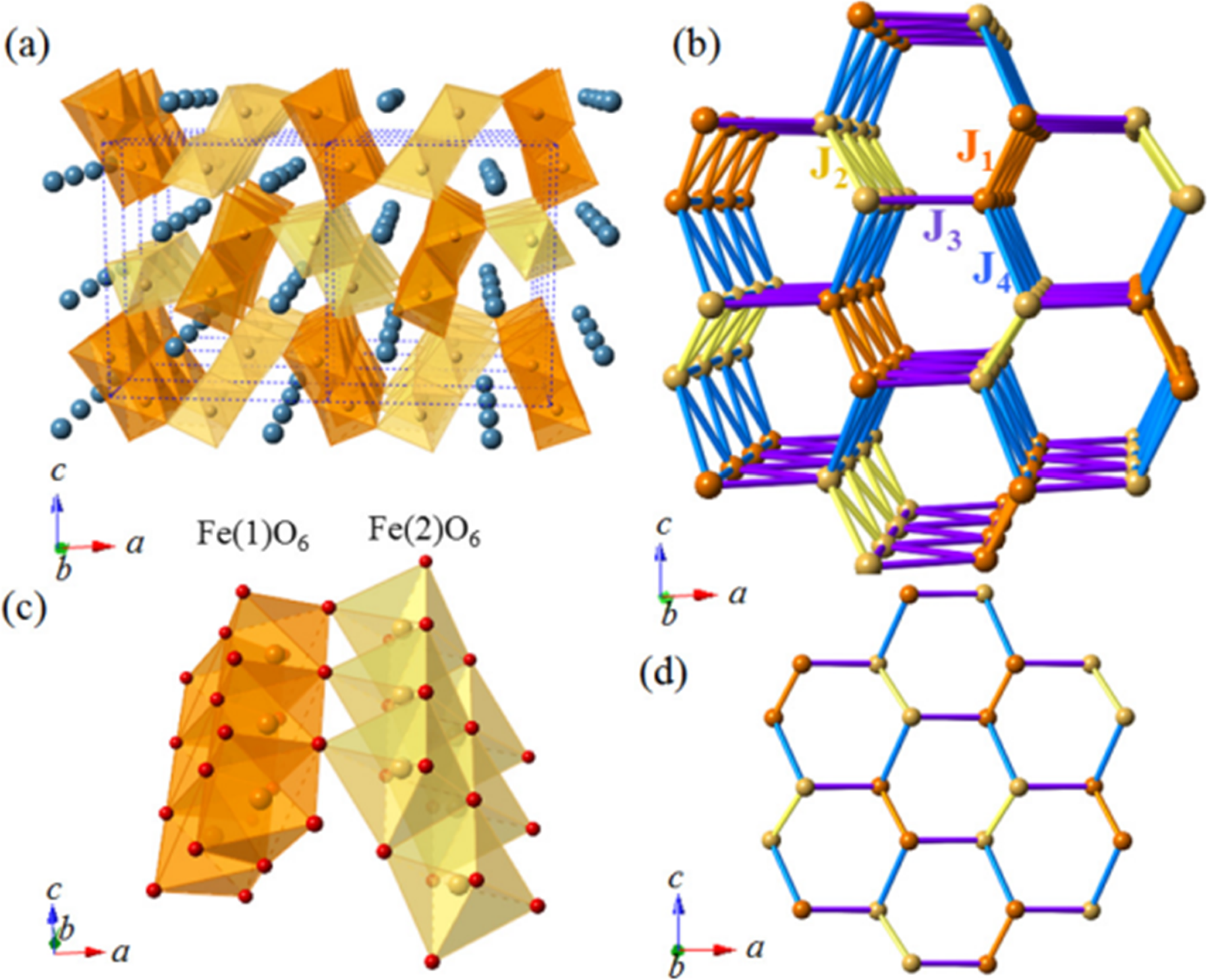
Crystal structure of CaFe2O4 (a) shows the edge- and corner-sharing FeO6 octahedra, with Ca atoms (colored blue online) occupying the interstitial sites. The two sets of octahedra (colored orange and yellow online) are centered around Fe(1) and Fe(2) atoms, respectively. (b) shows Fe-zigzag chains running along the b axis to form hexagonal tunnels arranged in a distorted honeycomb lattice. (c) shows a zoomed-in view of the Fe(1)O6 and Fe(2)O6 octahedra in chain along the b direction, with the oxygen atoms depicted as small spheres. (d) shows a projection of (b) onto the ac plane. The exchange couplings J1,J2,J3, and J4 are labeled in (b). The complete bond lengths and bond angles. 29
Photocatalytic mechanism of CaFe2O4
The principal purposes of photocatalytic activity are to use solar energy for the production of power and the protection of the environment.35,36 In the photocatalysis process, an electron is transferred from the valence band to the conduction band in this condition, generating photo-induced holes in the valence band. Photo-induced electrons are readily absorbed in the conduction band by dissolved oxygen molecules, resulting in the formation of a reactive superoxide anionic free radical. It then interacts with the hydrogen ion to produce reactive hydroxyl free radicals.
Furthermore, the hole reacts with H2O to produce the hydroxyl radical. The contaminant ions or molecules are close to the created radicals due to the strong adsorption capacity of CFNPs, and they have been readily destroyed by the produced hydroxyl radicals. The products of degradation have been exfoliated from the active surface regions of nanoparticles. Furthermore, the degradation of organic molecules occurs, producing water molecules and harmless carbon dioxide 37 (Figure 3).
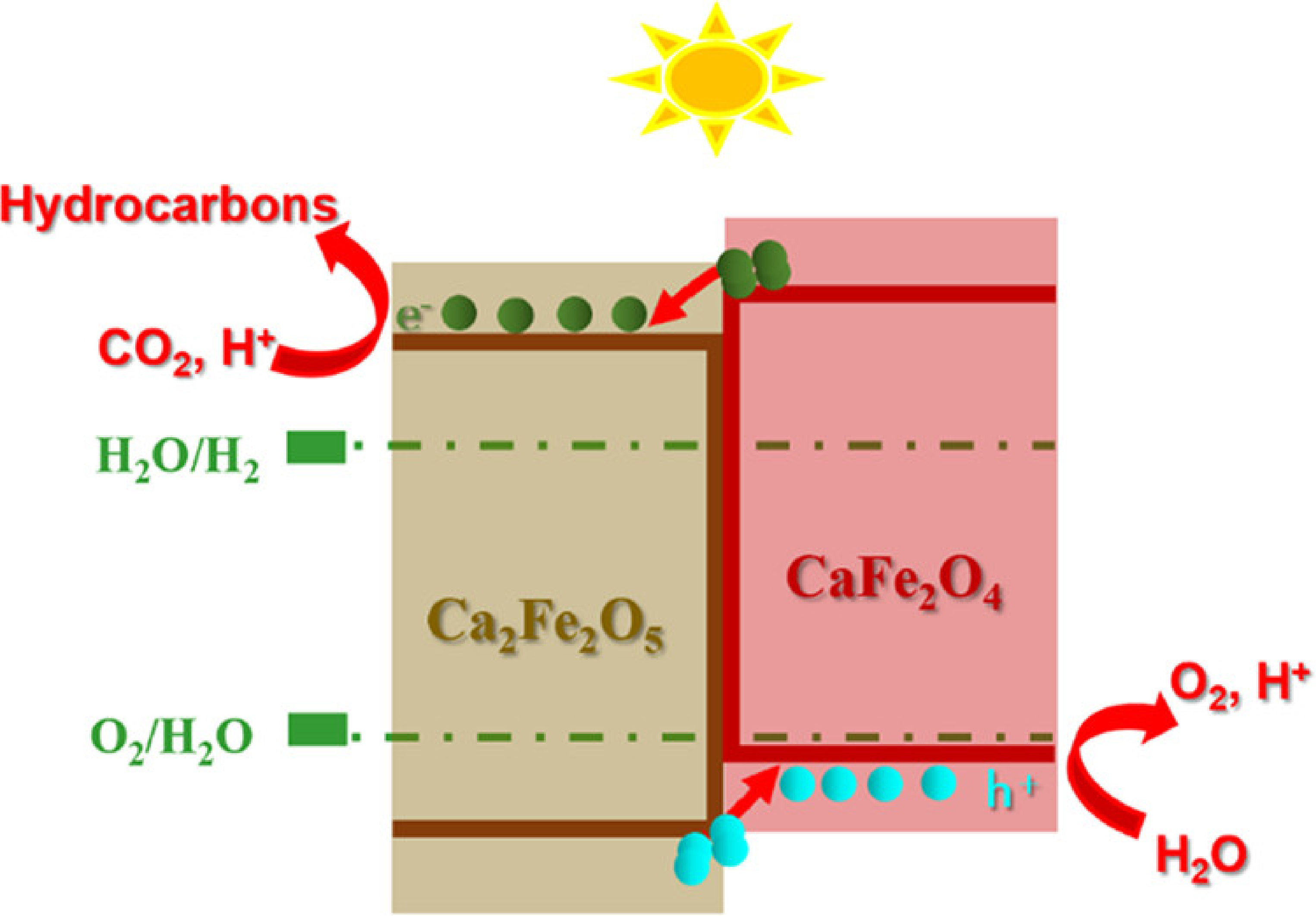
Free radical photocatalytic mechanism of CF. 38
Synthesis of CaFe2O4 nanoparticles and their composites
Sol-gel method
For the preparation of nanostructured metal oxides, the sol-gel process is an excellent wet method. The sol-gel method combines a metal precursor and citric acid to form a gel solution. The metal precursor is mixed in water and ethanol at pH 9 until the gel solution forms, and then the gel is dried at 450–800°C. 39 CFNPs synthesized by this method exhibit wonderful photocatalytic and optical activities. In general, photocatalyst performance is greatly influenced by its optical activities. As a result, UV–vis DRS was used to study the optical absorption characteristics of the as-synthesized catalysts. All of the samples responded well to UV and visible light. CaFe2O4-800 had the maximum absorption capability of light in the range of 300−520 nm, suggesting a higher possible photocatalytic activity. 23 It is one of the simplest, most cost-effective, and most significant methods to synthesize CFNPs with excellent properties. Figure 4 depicts the synthetic route of CFNPs.
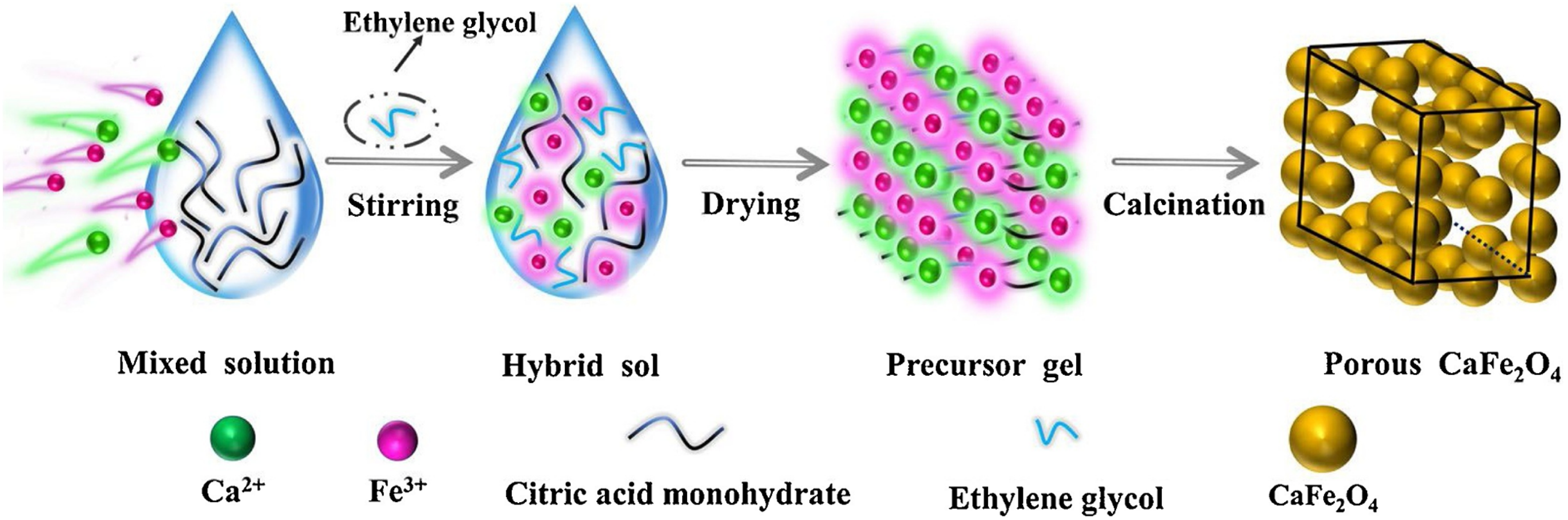
Schematic illustration of the formation procedure of CaFe2O4. 24
Molten salt synthesis
The molten salt synthesis process offers distinct benefits in catalyst development, particularly for materials that need higher temperatures during synthesis. 40 This approach may readily change samples with particular morphologies, like rods, wires, plates, and flowers, as well as reduce the liquid phase formation temperature by using inorganic salts with lower melting point.41,42 This is the shape modification process of CF aggregated particles (APs) to CF nanorods (NRs). Due to the abundance of grain boundaries, which inhibit photogenerated charge transfer and function as charge recombination sites, CFAPs have poor photocatalytic activity. The molten salt-assisted alteration may quickly modify the morphology of CF NRs and reduce the creation of grain boundaries, allowing photogenerated charges to be separated and transferred for extremely effective photocatalytic degradation. 43 Figure 5 depicts the synthetic process of CF APs and CF NRs 43
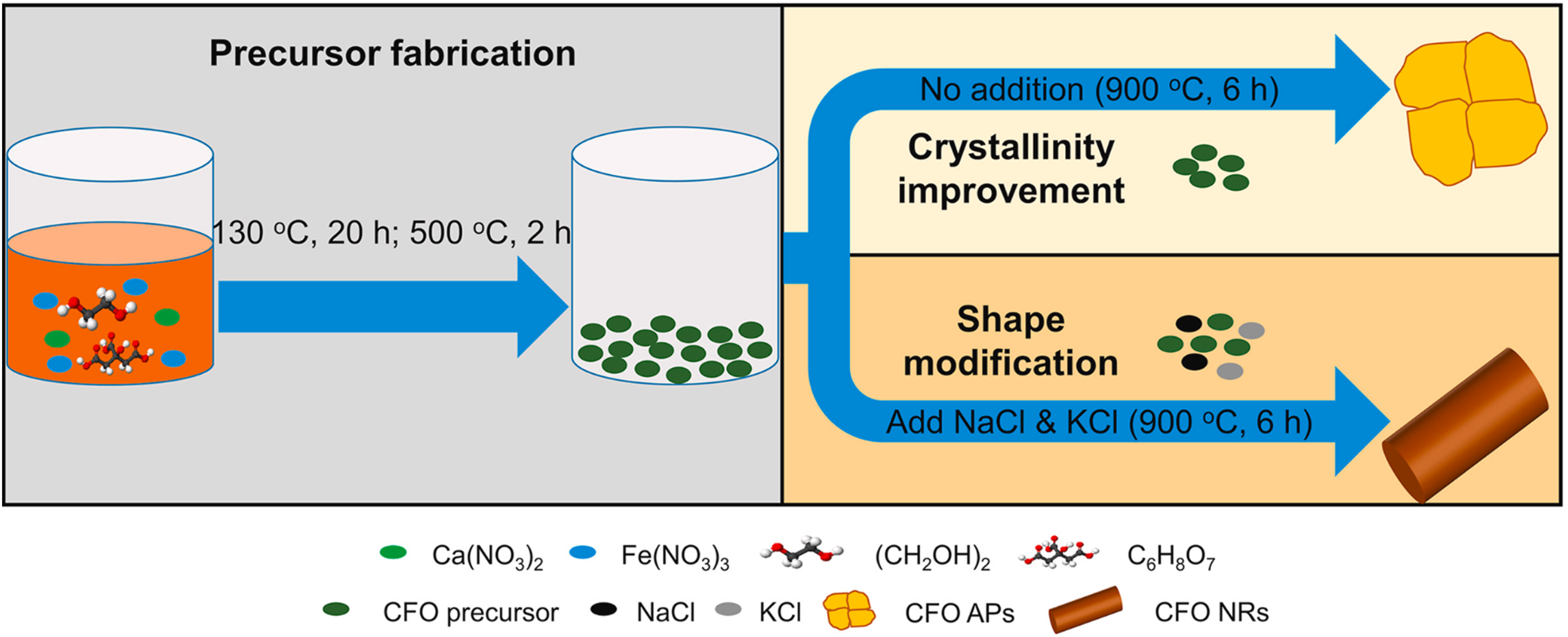
Schematic diagram of the synthesis process of CFAPs and CF NRs. 43
Solution combustion method
In this method, lemon juice is used as fuel. The starting materials, calcium nitrate and iron nitrate, were dissolved in lemon juice for 20 min using a magnetic stirrer to make a homogenous solution. This homogenous solution is further preheated at 500°C in a muffle furnace. At high temperatures, some gases are released, and converted into a brown-colored nanomaterial with a porous structure. This porous nanomaterial was calcined for 16 h at 800°C to form CFNPs. Figure 6 shows the flowchart of the synthesis of CFNPs using the solution combustion method. This method is also included as a green synthesis method because of the use of lemon juice. 44
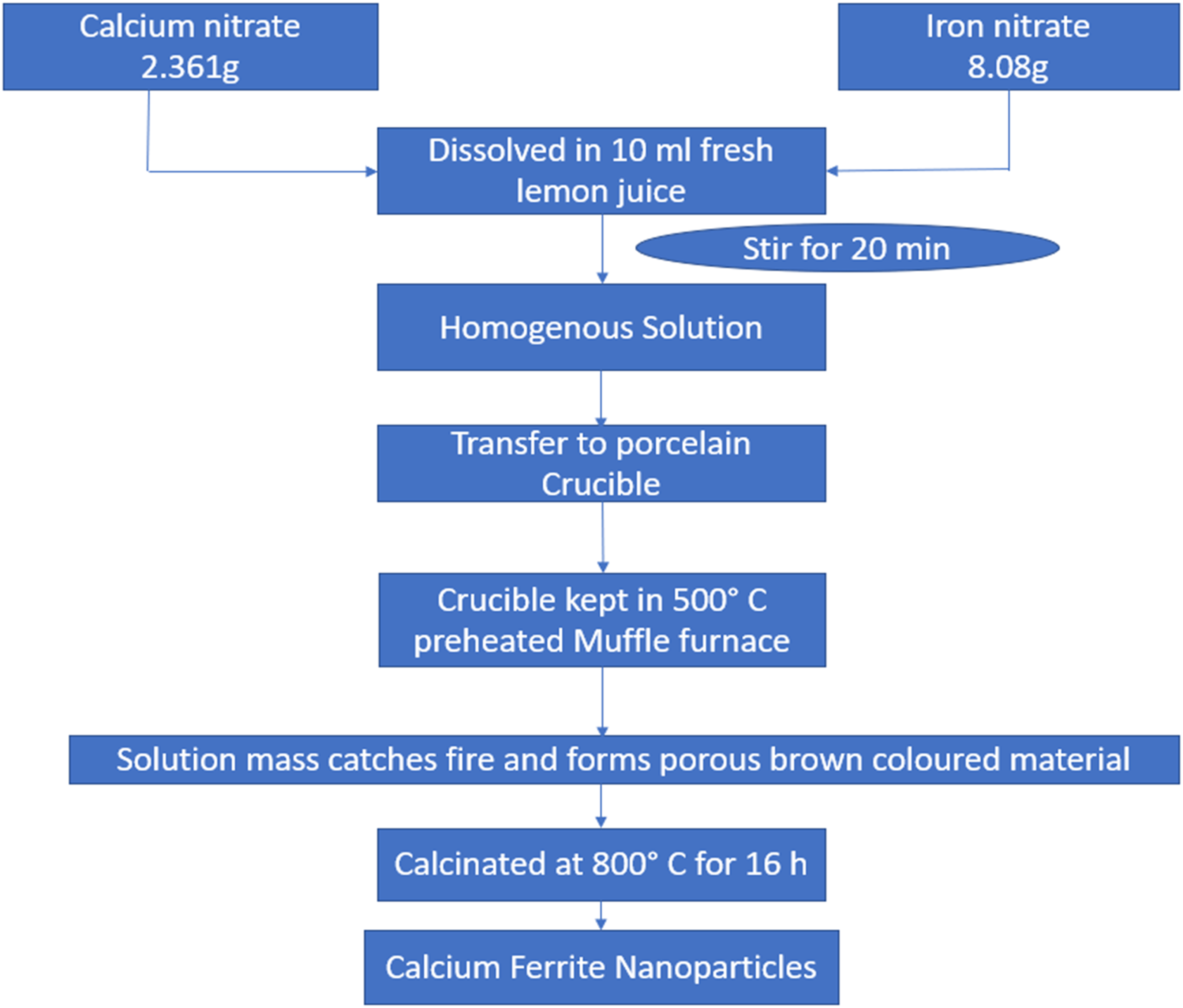
Schematic presentation of synthesis of CFNPs via solution combustion method. 44
Facile precipitation method
By using facile precipitation method, CF-CeO2 NCs was fabricated. CFNPs was formed further using the same chemical procedure as of CF-CeO2. In this process, water was used as a solvent with some green and biocompatible agents such as glucose, starch, and polyvinyl pyrrolidone. For example, 0.2 g calcium nitrate and 0.68 g iron nitrate were dissolved in 200 ml of distilled water. Further, to obtain approximately 10 pH of the solution, 1 M NaOH (10 ml) was added to the previous solution, which resulted in a brown precipitate. Using centrifugation, it was rinsed with distilled water and then calcined at 80–500°C to obtain black CFNPs. Later on, for the synthesis of CF-CeO2, 0.1 g of CFNP was dissolved in 200 ml distilled water then 0.4 g of ceric sulfate was incorporated into the solution. After this, NaOH was slowly added to the solution to reach pH 10 and stir for 30 min. Figure 7 shows the synthesis of CF-CeO2 NCs followed by the precipitation method. 45
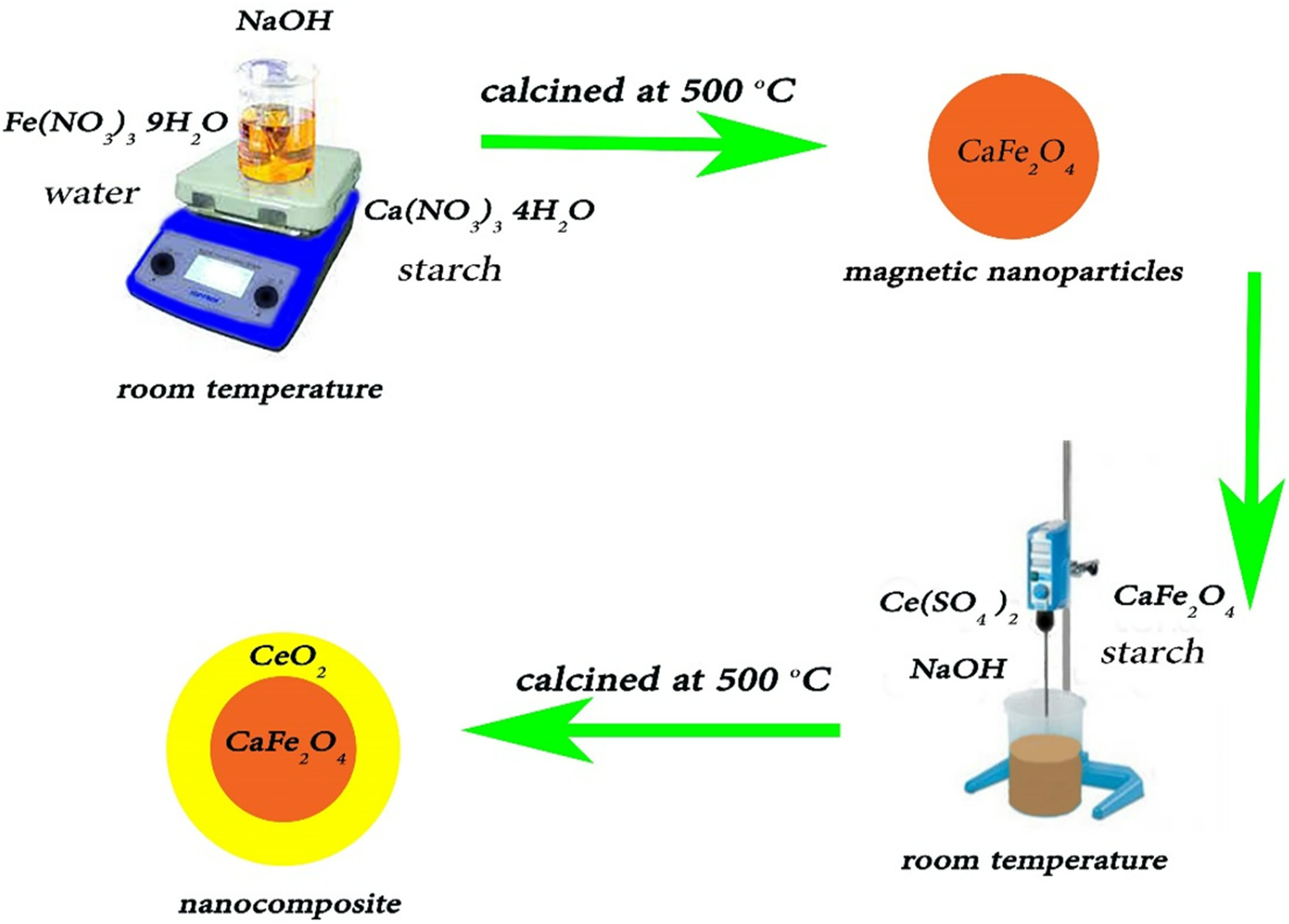
Synthesis of CF-CeO2 NCs via precipitation method. 45
Simple two-step process
It is a simple chemical process by which CF-gwt-ignore-inputC3N4 NCs are synthesized. In this process, melamine was used as a precursor for g-C3N4, as well as calcium nitrate and iron nitrate in ethanol for CF particles. In the first step, CFNPs and g-C3N4 were fabricated separately using the precursor demonstrated in Figure 8. Further, for the synthesis of CF-gC3N4, 0.5 g of g-C3N4 was taken with different amounts of CF (20, 30, and 40%) and dissolved in 40 ml of ethanol under 30 min of ultrasonication. The as-prepared solution was kept for 12 h with stirring and then transferred into an autoclave and kept for 6 h at 120°C. After centrifugation of the obtained mixture, it is dried at 70°C in an oven, which results in a pale brown catalyst. As prepared catalysts have different amounts of CF, this results in different photocatalytic activities of NCs. 46
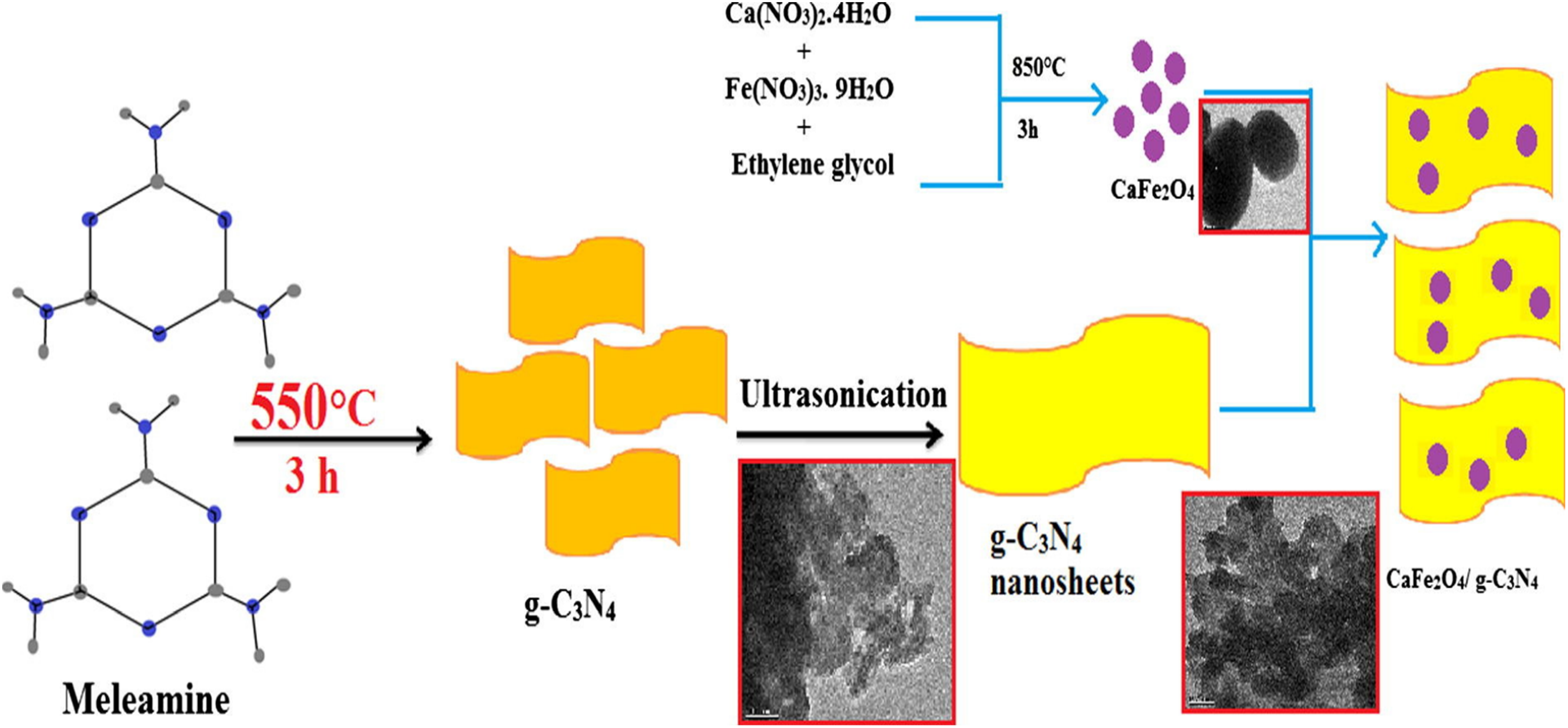
Schematic representation for the synthesis of CaFe2O4/g-C3N4 NCs by two-step process. 46
CaFe2O4 nanocomposites and their application
As CF is a p-type semiconductor, which has superparamagnetic behavior, it forms various composites with n-type semiconductors followed by p-n heterojunction. These composites have different properties and applications, which are based on their methods of preparation. CFNPs and their NCs have wide applications in various fields like decontamination of wastewater, energy production, gas sensing, transesterification, and many more. A brief explanation of the application parts of CFNPs and NCs has been discussed below.
Decontamination of wastewater
For wastewater remediation, a variety of water purification methods, such as flotation, coagulation, electrochemical, bioremediation, ultrafiltration, electrodialysis, and adsorption methods, are available.47–52 However, these systems are expensive to implement and also have boundaries on their wastewater purification efficiency. As a result, novel and affordable treatment techniques for wastewater and industrial effluents are expected to emerge. Semiconductor photocatalysis is regarded as an innovative technique to overcome the current drawbacks because it employs a photocatalyst for effluent treatment with solar energy. By absorbing solar energy, photocatalytic materials may conduct reductive and oxidative elimination of inorganic and organic contaminants. Semiconductor nanoparticles and nanocomposites are employed in the treatment of wastewater.
CFNPs and nanocomposites have large active surface areas, surface charges, distinctive morphology, and adjustable optical properties, so they are widely used in the treatment of wastewater. Numerous self-assembled nanocomposite materials have recently been widely researched because they have some specific physical and chemical characteristics. Because of these characteristics, they apply to various environmental applications, such as the removal of poisonous dyes, medicinal chemicals, and heavy metals from aqueous environments.53,54 In the contaminated water, many effluents, such as dyes, drugs, and metal ions, are present, which are effectively eliminated by the use of CFNPs and NCs. In the treatment of wastewater, CFNPs containing superparamagnetic properties are incredibly significant. CFNPs and CFNCs are applicable for both adsorptions as well as photocatalysis methods.
Dye degradation
The world is cautious about the potentially harmful effects of dyes on the environment, which threaten the ecology. 55 Over 100 tonnes of dyes, mainly from the textile sector, are dumped into the aquatic habitat yearly. 56 Most synthetic dyes have carcinogenic, genotoxic, non-biodegradable, and non-photodegradable characteristics. Such colors in wastewater produce a terrible odor and harm human, aquatic, and environmental health. 57 The usage of dyes is harmful because they are mutagenic, carcinogenic, or teratogenic, and, as a result, they may affect numerous marine living species (for example, fish). Furthermore, dyes can potentially cause major health consequences in humans, including harm to the brain, kidneys, liver, reproductive system, and central nervous system. In addition to other effects, mutagenesis, carcinogenesis, chromosomal fractures, and respiratory issues are all possible side effects of dye poisoning. As a result, dye degradation is one of the major issues to date. CFNPs and NCs are suitable candidates in this field and have given a marvelous performance in the degradation of dyes. CFNPs and NCs can degrade cationic and anionic dyes, but it has been observed that most of the dyes degraded by CFNPs are cationic dyes.
Degradation of cationic dyes
The degradation of various cationic dyes like Rhodamine B (Rh B), Basic Blue 41 (BB 41), Brilliant Green (BG), and Methylene Blue (MB) has been discussed using numerous NCs of CF and NPs. Most NCs give results under radiation (visible light, LED light, sunlight, microwave irradiation); others offer excellent performance in the absence of light, which completely depends on their preparation methods. Methylene blue is one of the most degraded dyes using different CFNCs. CF-ZnO (60 mg/l) is able to degrade 97.5% Rh B under visible light. The major role in the degradation of Rh B has been played by OH radicals. ZnO is an n-type semiconductor that forms p-n heterojunction with CFNPs to enhance photocatalytic performance. 58 CFNPs fabricated by the sol-gel method processed at 800°C calcination temperature give the photocatalytic degradation of Rh B under irradiation of LED light. 24
The photoelectrochemical oxidation of BB 41 has occurred using CF under sunlight, followed by the generation of an OH radical. The photocatalytic process was carried out in a Pyrex reactor linked to a thermostatic water bath (25°C). Connect to that aquatic system to reduce vortex phenomena and sedimentation at pH = 6.4. 100 mg of CF has been magnetically suspended (200/min) in a 20 ppm solution of BB 41 (100 ml). The reactor was allowed access to sunlight (980 Wm2), and using a conventional light meter (Testo 450), the flux was measured. 11
A chemical precipitation process was used to effectively produce CFNPs, which were then used in the persulfate (PS)-induced sono-assisted degradation of BG dye from the water phase. At a dye concentration of 50 mg/L, a PS dose of 200 mg/L, a catalyst dosage of 0.5 g/L, and a 15-min ultrasonic (US) irradiation duration, up to 99.9% degradation of BG dye, was obtained. Nevertheless, when CF was added to the US system, the degradation efficiency was increased to 89%, but when only US was used, the BG degradation was only 40.74%. 59
The composite catalyst formed by CF and alpha Fe2O3 with a molar ratio of 1:2 (CF12) shows the best activity in the degradation of MB. In just 180 min, 95.6% degradation efficiency has been achieved with the composite CF12 under sunlight. The composite's improved photocatalytic behavior can be attributed to the heterojunction among both alpha-Fe2O3 and CF, which reduces photogenerated holes and electron recombination. 60 It has been observed that the heterostructures of CF/g-C3N4 had better visible-light photocatalytic activity for the breakdown of MB when exposed to ultraviolet light. The degradation efficiency of 94% was observed with light irradiation within 120 min. Improved photocatalytic activity results from the synergistic impact of g-C3N4, CF, as well as other heterostructures with close interfacial contact. 46 The CF composites with carbon decoration have an improved activity of photocatalysis towards the removal of MB in the aqua matrix under the irradiation of visible light. 58 wt.% C/CF was the optimum photocatalyst, with a rate constant of degradation of 0.0058 min−1, and it was approximately 4.8 times greater than pure CFNPs (0.0012 min−1). 61 CF nanorods synthesized using molten salt-assisted shape modification show great efficiency toward the photocatalytic degradation of MB. 43 The composite p-CF/n-Ag3VO4 gives 85.4% photocatalytic degradation of MB when the p-CF/n-Ag3VO4 and 2.0 wt.% amount of doped p-CF have been ball milled till the 12 h. 62 CF@Bi2O3 is an efficient photocatalyst for the degradation of MB under visible light. 63 The photocatalyst MgFe2O4-CF fabricated by the recyclization of electric arc furnace (EAF) dust gives 45% photocatalytic degradation of MB in 240 min under visible light irradiation. 64 p-CF/n-ZnO photocatalyst fabricated by the ball milling method gives 73.4% photocatalytic degradation of MB with 1.0 wt.% amount of doped p-CF. 65 Ceramic powder of CF with the doping of Ti and La metals shows the degradation of MB under visible light. 66 Up to 97%, MB was degraded using CdO/CF NCs in 160 min in the presence of an OH radical source (ammonium hydroxide). Solvents also affect the photocatalytic efficiency, and it has been observed that photocatalytic efficiency decreased when the solvent was changed (benzoquinone, methanol, isopropanol). The catalyst shows outstanding performance till the sixth run. 36 CF fabricated using the sol-gel process shows 90% degradation of crystal violet within 10 min under microwave irradiation. A total of 90% of crystal violet was eliminated in 8 min when the concentration was less than 100 mg/l. 67
Degradation of anionic dyes
Anionic dyes like Reactive Orange 12 (RO12), Congo red, methyl red, methyl orange, and Eriochrome Black T (EBT) are effectively removed from the water matrix using CFNCs. For the removal of RO12 from an aqueous medium, CFNPs were utilized as adsorbents. The surface area of CFNPs is 230 m2/g, and the average pore diameter is around 2.5 nm. At a pH of 2.0 and an uptake capacity of 276.92 mg/g, the RO12 elimination efficiency peaked at 77%. When the adsorbent dosage was increased from 0.125 to 1.0 g/L, the percentage of dye removal increased by 24.54–95.67%; however, further increases in adsorbent dose did not significantly improve dye adsorption efficiency. At larger doses of CFNPs, a rapid rise in absorption efficiency on a rising dosage of adsorbent relates to the increased surface area with more adsorption sites. With an increment in the dose of adsorbent from 0.125 to 1.5 g/L, the capacity of adsorption (mg/g) declines from 157.06 to 52.14 mg/g. This might be attributed to (a) at larger doses, the number of unsaturated active sites, as well as (b) on the surface of the adsorbent, the number of overlapping active adsorption sites, leading to higher doses at lower adsorption capacity. At smaller concentrations of dye, there were unsaturated active sites rather than higher removal efficiencies, whereas, at increasing concentrations of dye, all active sites were saturated as well as adsorption efficiency approached equilibrium. 68
***CFNPs give the adsorption of Congo red with 74.5% efficiency of elimination in 120 min, whereas CF-NGO nanocomposites adsorb 88.0% Congo Red within the same time. 69 CF fabricated at a specific calcination temperature (800°C) gives the degradation of methylene orange (MO). 23 of MO (with an initial concentration of 98.37 mg/l) was degraded in 24 min with 99.88% elimination efficiency in the presence of CF-MnFe2O4 magnetic NCs with 0.58 g/l dose of catalyst 70 98.92% MO were eliminated by CF-ZrO2 magnetic NCs in 25 min at 30°C temperature with 0.54 g/l catalyst dose and 22 mg/l concentration of dye. 71 The NCs of CF/CaTiO3 shows the photocatalytic degradation of methyl orange with 89% removal efficiency with 0.2 g/l amount of catalyst In comparison, 97% removal efficiency was obtained for the degradation of methyl red with a similar catalyst amount. 72 CF-MnFe2O4 NCs degrade EBT coupled with ultrasonic irradiation. The efficiency of degradation obtained was approximately 88%. 73 Numerous azo dyes like Acid Blue, Acid Black, and Acid Violet were degraded by various CFNPs and NCs. CF-CeO2 shows degradation towards azo dyes such as Acid Blue, Acid Black, and Acid Violet under microwave irradiation. The catalyst was fabricated by the facile precipitation method. The degradation of Acid Blue, Acid Violet, and Acid Black occurred between 90, 80, and 60 min. 45 CF-ZnO NCs fabricated by simple hydrothermal method give degradation of several toxic azo dyes like Acid Violet, Acid Brown, and Acid Black under UV light. It provides 96, 98, and 99.99% degradation of Acid Violet, Acid Brown, and Acid Black, respectively, within 60 min. 74 CA-Ag-CF NCs were fabricated by electron spinning using cellulose acetate (CA) nanofibers and NCs with CF and Ag. These NCs were used for the photocatalytic degradation of specific azo dyes such as Acid Violet 7, Acid Brown 14, Acid Blue 92, and Acid Red 151. The efficiency of degradation was obtained at about 100, 80, 70, and 50% for Acid Blue 92, Acid Violet 7, Acid Brown 14, and Acid Red 151, respectively. The contact time for degradation was 100 min under visible light. 75 CFNPs fabricated by solution combustion method followed by green synthesis are efficient photocatalysts for the degradation of Evans blue. The catalyst gives the best efficiency for five runs. 44 Table 1 represents the summary of the degradation of cationic and anionic dyes using various CFNPs and NCs.
Summary of degradation of cationic and anionic dyes using various CF, CFNPs, and CFNCs.
Removal of heavy metals ions
Contamination of water by heavy metals is caused by a variety of human activities, including the combustion of fossil fuels, mining, vehicle exhaust gases, agriculture, and the disposal of liquid and solid wastes. Due to volcanoes, erosion, thermal spring activity, infiltration, and other factors, heavy metals may also be found in nature. This water poisoning poses a danger to living things, as most of the heavy metals are hazardous to aquatic environments and human beings. Heavy metals such as lead, chromium, mercury, and nickel have the worst environmental impact. Moreover, these metals are poisonous, non-biodegradable, and typically resistant to traditional elimination procedures, even at relatively low concentrations. 76 These heavy metal ions may cause numerous severe human diseases like kidney pathology, respiratory problems, cancer, and neurological disorders. Cadmium metal ions cause high blood pressure and many issues related to the kidneys. 77 In the same way, lead is responsible for decreased fertility and neurological disorders. Chromium ions can cause skin lesions, breathing problems, and carcinogenic behavior. 78 Therefore, degrading these metal ions is one of the most challenging tasks. In this way, CFNPs and NCs play a significant role in degrading these metal ions.
Degradation of Cr(VI) ion
Chromium is a dangerous element that is primarily found in the hexavalent and trivalent valences of Cr(Vl) and Cr(III) and is released during industrial processes such as electroplating, leather tanning, and coloration.
79
It is poisonous to humans and has been associated with carcinogenic and mutagenic consequences.
80
The best way to get rid of Cr(VI) is to convert it to less toxic Cr(III). The photocatalytic mechanism is considered one of the best methods to chemically reduce heavy metal ions due to its exceptional features like recyclability and low cost The most effective way to reduce Cr(VI) is to convert it to the least harmful Cr(III).
81
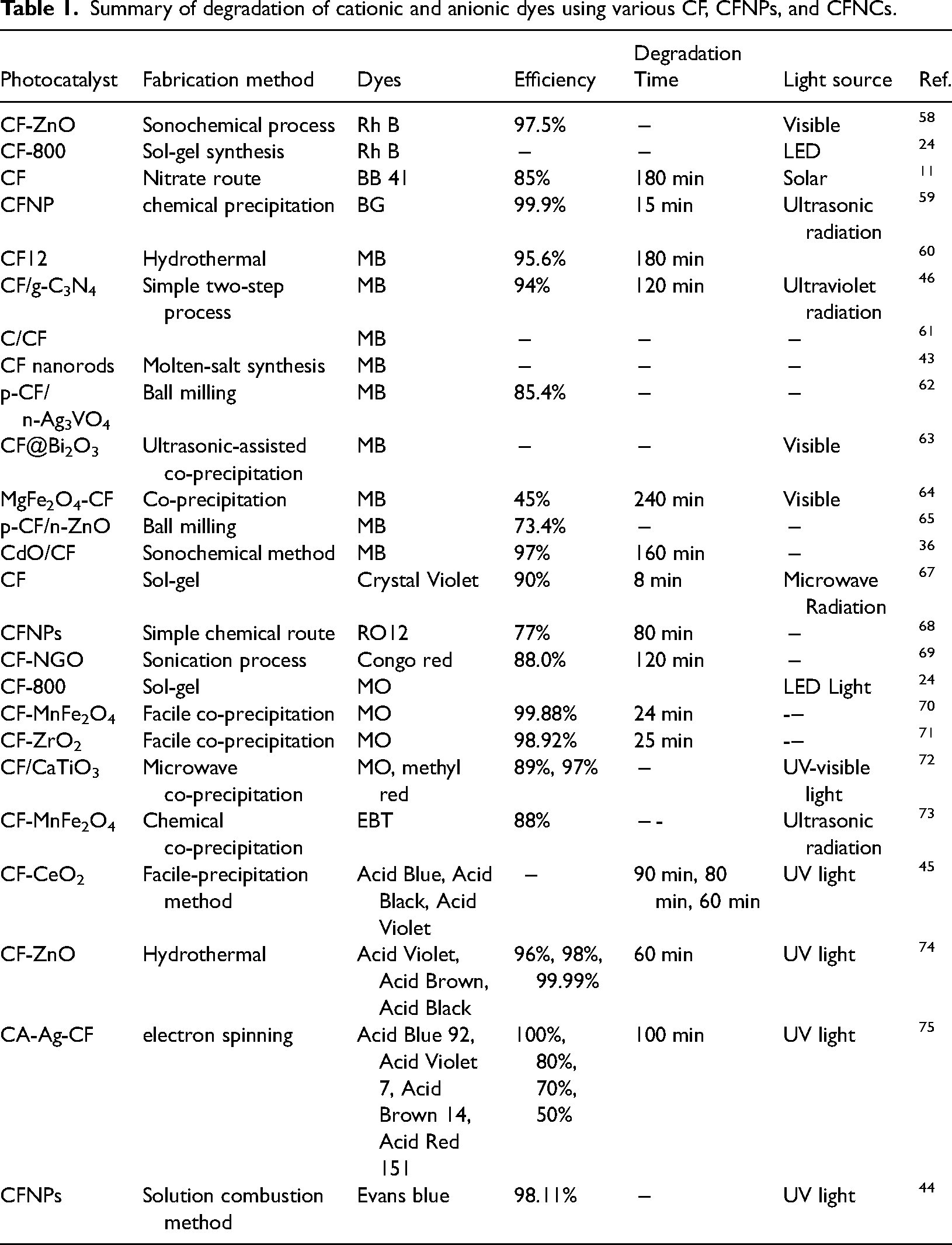
CFNPs (size 34 nm) fabricated by the nitrate route significantly reduce Cr(VI). According to the literature, CF is an n-type semiconductor with a negative VSCE flat band potential (Figure 9), which is convenient for exchanging electrons with chromate species because of its cathodic behavior. In the presence of oxalic acid, it shows more reactivity and is also more stable towards photocorrosion of NPs. Irradiation of visible light increases the reduction of Cr(VI) on CFNPs. Under ideal circumstances, including the initial concentration of 30 ppm, catalyst dose of 0.6 g/l, and pH 3, elimination of 86% of HCrO4− has been accomplished under illumination (16 mW cm−2) whereas, under solar light (98 mW cm−2), the photoactivity achieved a maximum of 100%.
33
It has been observed that the photocatalytic reduction of Cr(VI) is carried out in the presence of oxalic acid by mixing CF into the solution of Cr(VI). The CF is resistant to photocorrosion with oxalic acid, which absorbs photo holes and improves photoactivity by separating electron/hole pairs. The reduction efficiency has been calculated using equation (1).
33
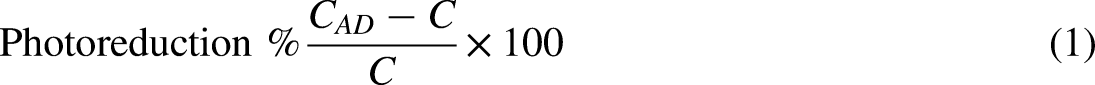
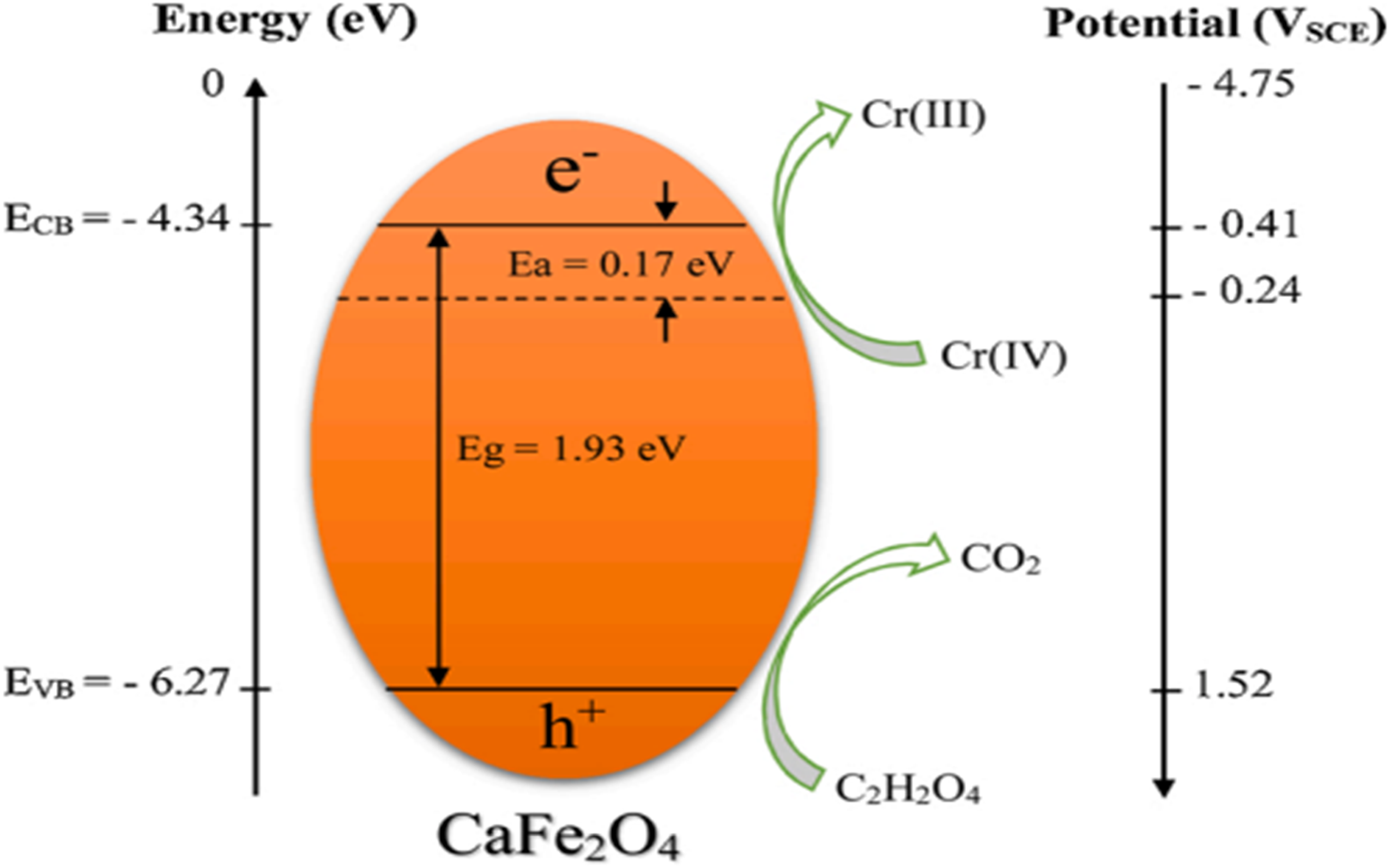
Reduction of Cr(VI) to Cr(III) using CF prepared using oxalic acid. 33
Here, C = Cr(VI) concentration after illumination at the time (t)
CAD = initial concentration of Cr(VI) after adsorption
Adsorption of Pb(II)
Lead is the most toxic metal for water contamination mostly found as Pb(II). Effluent wastes mainly release Pb(II) ions from mining, smelting, galvanization, and battery production. It has harmful effects on children's as well as adults’ health and causes renal damage, high blood pressure, neurological disorder, stomach pains, and headaches. 82 The magnetic NCs of N-doped graphene oxide (NGO) with CFNPs were fabricated by the sonication method for the effective removal of Pb(II) ions. The surface area of graphene oxide (GO) is increased by N-doping. Furthermore, the combination of NGOs and CFNPs enhances the photocatalytic and adsorptive characteristics of NCs because NGOs prevent the aggregation of CFNPs by preventing the recombination of the e− h+ pair. For Pb(II) ions, a 0.01 g dose of catalyst CF-NGO in a 1:2 ratio gives 84.7% elimination. For CF-NGO, the equilibrium was obtained in 60 min because of the significant interaction between the metal ions and the adsorbent. 88.6% removal was obtained in 120 min with an optimum dose of 1.0 g/l of catalyst. The order of removal of metal ions using CF-NGO is as follows: Pb(II) > Cd(II) > Zn(II) > Ni(II). 69
Antibacterial performance
Bacteria are one of the major pollutants in water pollution and cause numerous bacterial diseases. CFNPs and NCs play a significant role in the removal of bacteria from the water matrix. The NCs of CdO/CF prevent the potential growth of bacteria. 36 98% B. subtilis and 99.9% E. coli bacteria were removed by CF-ZnO NCs under visible light. 58 Further, it has been observed that the NCs of CF-Bi2O3 prevented the growth of E. coli and B. subtilis. 63
Removal of antibiotics
Many antibiotics emitted by individuals, industries, and hospitals end up in sewage systems and municipal plants. Chronic toxicity, a barrier to photosynthesis in aquatic systems, and antibiotic gene resistance in microorganisms are only a few of the negative impacts of widespread antibiotic usage that have become a worldwide concern. Removing antibiotics from wastewater is essential since they may include trace amounts of organic pollutants. Antibiotic residues have been found in water sources and the environment worldwide. 83 Oral medications, which are around 90% expelled from the human body and hence end up in marine systems, are a common source of pharmaceuticals and illegal drugs as pollutants in aquatic habitats. 84 To resolve the drug removal problem, some CFNCs have been fabricated, which are discussed here. The effective removal of tetracycline (TC), tetracycline hydrochloride (TCH), cefazoline (CFZ), and metronidazole (MTZ) has been done using CFNCs.
In the past several decades, personal care and pharmaceutical items discharged into environmental water resources have become a severe health hazard. Antibiotics are unusual among medications because they solely act on disease-causing bacteria and have no impact on animal or human tissues. Tetracycline (TC) drugs are commonly prescribed antibiotics that are well known for their inexpensive cost of manufacture and excellent activity. Despite their significant consumption in human and veterinary medicine, TC compounds may constitute a substantial hazard to the environment and health. They must be eliminated from the ecosystem at allowed concentrations. 85 Because TC molecules are relatively persistent in the environment, total removal by the standard activated sludge technique is difficult. The hydrophilic character of TC molecules, as well as their structure containing carbonyl and amino groups, make them ideal for adsorption. Due to the van der Waals force of attraction, TC molecules are attracted to the active sites of the adsorbent surface. For the removal of the TC drug, a CF-ZrO-MNC (calcium ferrite-zirconium oxide magnetic nanocomposite) was created. 86 It has been observed that the TC adsorption on the surface of the catalyst CF-ZrO-MNC entirely depends on the pH. At pH 6.0, CF-ZrO-MNC removed more than 65% of the TC, but the effectiveness dropped further at pH 6.0. Because there are enough H+ ions available at pH 2.0–4.0, both CF-ZrO-MNC and TC get protonated, resulting in electrostatic repulsion between the active sites of protonated CF-ZrO-MNC and cationic TC, confirming the detrimental adsorption process of TC. When pH exceeds 8.0, electrostatic repulsion occurs owing to the abundance of OH ions in the aqueous medium, causing deprotonation of CF-ZrO-MNC and TC. Nevertheless, at pH 5.0–7.0, TC is accessible in its zwitterion form, including positively charged groups with strong electrostatic attraction to the CF-ZrO-negatively charged MNC's active surface sites. 86
To investigate the reusability and stability of CF-ZrO-MNC, the adsorption-desorption of TC was carried out with CF-ZrO-MNC for five successive runs. At 6.0 pH of solution within 60 min, when the concentration of TC was 40.0 mg/l, the removal efficiency of TC was 98.14%. 86 The concomitant removal of tetracycline hydrochloride (TCH) coupled with the CF/Bi2O3 composite has been studied. Within 30 min, the maximum efficiency of removal of TCH was 97.8% using a 24.6 kV peak pulse voltage, an initial pH of 3, a 50 Hz pulse frequency, and an airflow rate of 2 l/min. For the removal of TCH in the water system by pulsed discharge plasma on the water surface (WSPDP) + CF/Bi2O3, the prominent reactive radicals were OH radical O2 and h+. O3 and H2O2 were indeed implicated in the synergistic system's TCH degradation, as well as the incorporation of CF/Bi2O3 may considerably enhance O3 and H2O2 usage in the solution 87 (Figure 10).
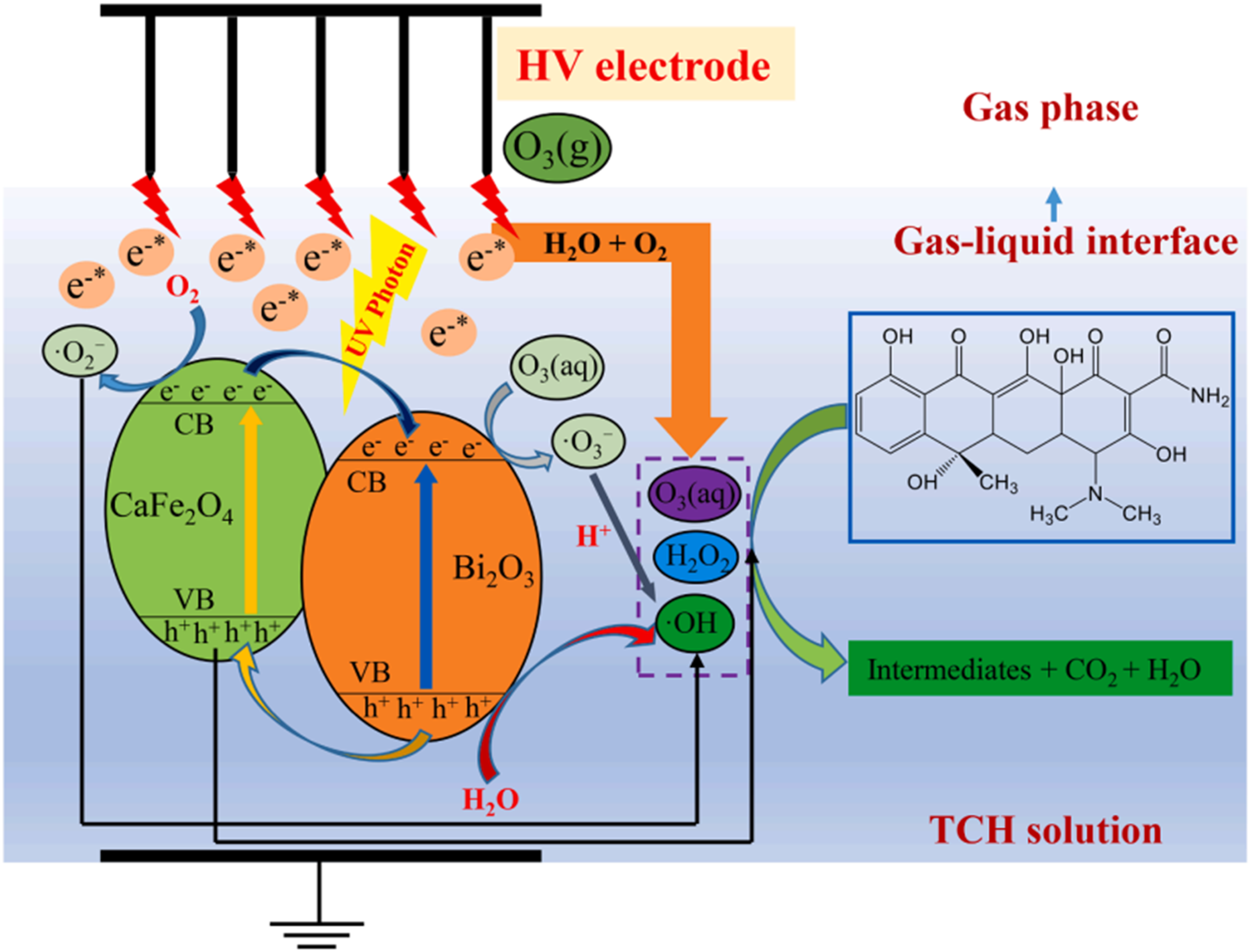
Schematic illustration of TCH degradation mechanism in the WSPDP + CF/Bi2O3 system. 88
A new catalyst filter was fabricated for the photocatalytic degradation of TC. The hydrothermal method was used to deposit CF and LaFeO3 perovskites on polyurethane filters (CF/PU and LF/PU). The removal efficiency of TC has been obtained at 80% for CF/PU, whereas LF/PU shows 94% removal of TC under visible light, followed by photo Fenton decomposition. The catalyst offers a 76% degradation performance for seawater, and a 71% removal of TC from lake water has been observed. 89 The CF/ZnCoO4 composite was fabricated by a two-step synthesis method and is an efficient catalyst for the degradation of TC. Additionally, the catalyst shows good degradation efficiency towards chlortetracycline as well as oxytetracycline. It gives an 88% removal of TC within 100 min of light irradiation (Table 2). 90
Various CF, CFNPs, and CFNCs with their fabrication methods, efficiency, and removal time for the removal of antibiotics.
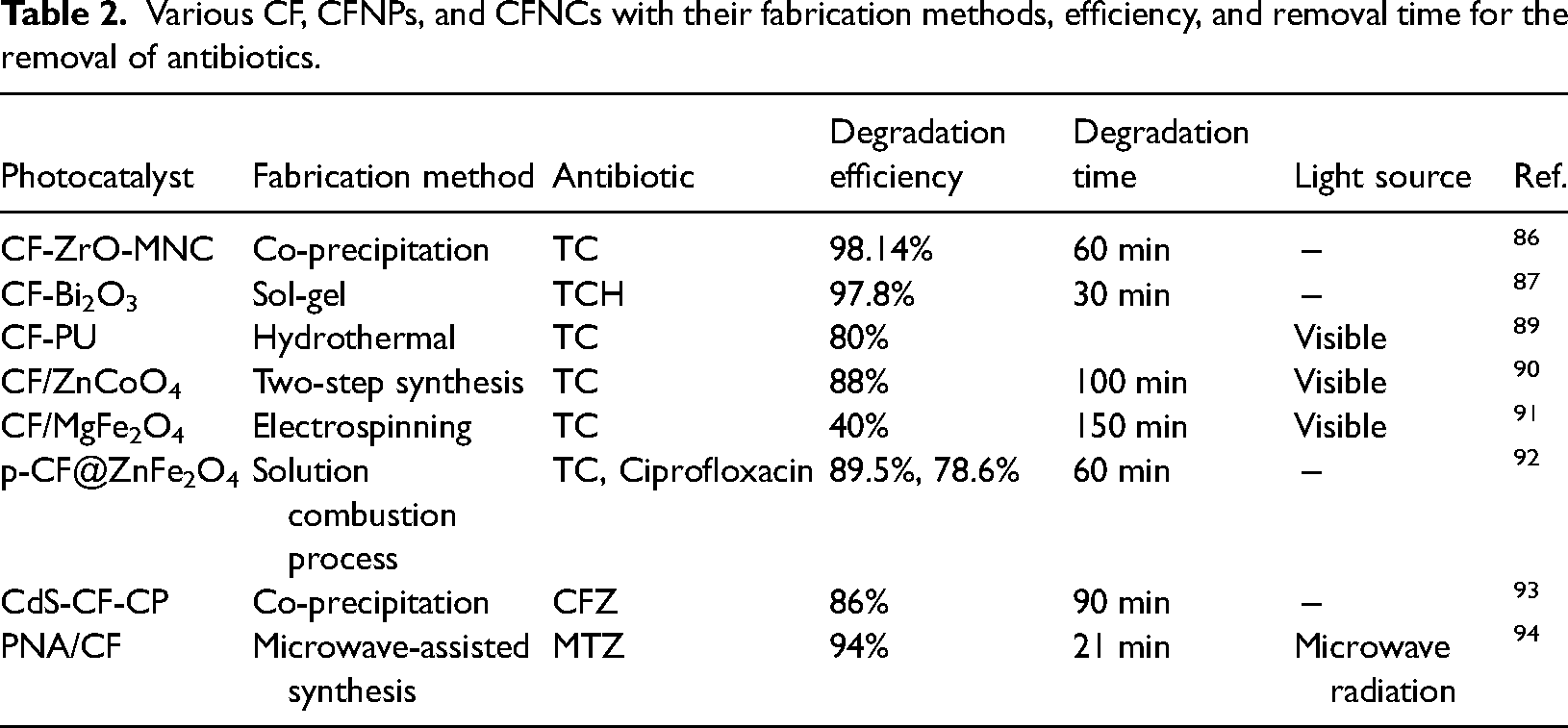
CF/MgFe2O4 nanowires were fabricated using an electrospinning method and were used for the degradation of TC. After 150 min of reaction time, 40% removal efficiency was observed under visible light. 91 The P-CF@ZnFe2O4 photocatalyst was fabricated by a simple solution combustion process, where the catalyst with 40% CF loading at the surface of ZnFe2O4 gives the best degradation results for the degradation of antibiotics. The heterostructure of the catalyst improves the photocatalytic activity. When the antibiotic solution concentration was 20 ppm, it gave 89.5% degradation of TC and 78.6% degradation of Ciprofloxacin in 1 h. 92 The CdS-CF composite with clinoptilolite zeolite support (CdS-CF-CP) performed well as a photocatalyst for the degradation of the antibiotic CFZ. 86% removal of CFZ was obtained within 90 min, at pH 7, with a 0.6 g/l dose of catalyst, and the initial concentration of CFZ was 15 mg/l. 93 Microwave-assisted methods were used to fabricate poly (1-napthylamine) PNA/CF nanohybrids. This nanohybrid is highly efficient for the degradation of the drug MTZ. 94% degradation of MTZ has been obtained within 21 min with 40-PNA/CF under microwave irradiation. 94
Transesterification reaction
Biodiesel has received a lot of interest as an ecologically benign and potentially renewable alternative to diesel fuel derived from petroleum. Traditionally, biodiesel production at a commercial level has been achieved by transesterifying acids that contain triglyceride components with methanol or ethanol in the presence of a liquid base catalyst. Moreover, biodiesel production using homogeneous base catalysts causes high costs, environmental issues, as well as isolation problems. Therefore, heterogeneous catalysts were used, which make biodiesel production more convenient due to their easy isolation, reusability of the catalyst, green nature, and low production cost by using waste cooking oils and low-cost feedstock. 95 The heterogeneous catalyst CF-Ca2Fe2O5, fabricated by co-precipitation and calcination, shows significant results in biodiesel production from jatropha oil as well as soybean oil. The catalyst shows the best results with soybean oil. With a 1:15 oil:methanol molar ratio and a 4 wt.% catalyst, an 85.4% biodiesel yield was obtained within 30 min at 373 K. In the H2 atmosphere, the reduction of the catalyst is obtained in the CF-Ca2Fe2O5-Fe3O4-Fe crystal phase. With soybean oil and jatropha oil, this reduced catalyst yields 83.5 and 78.2% biodiesel, respectively.
The catalyst showed significant results for three runs, and biodiesel yield depended on the reaction time and temperature. The magnetic behavior of the catalyst makes easier separation of the catalyst (Figure 11). 96 The fundamental force of the Ca2Fe2O5 derived from waste materials was practically identical to that of the similar structure developed from high-purity reagents. In the transesterification reaction, the Ca2Fe2O5 perovskite showed excellent catalytic activity, and an 80% conversion yield was obtained in 10 h using a 1:3 oil-to-ethanol molar ratio. 97

Image of magnetic CaFe2O4–Ca2Fe2O5–Fe–Fe3O4 catalyst separated from biodiesel products by a magnet. 96
Hydrogen evolution reaction
For the production of hydrogen, photocatalytic water splitting is a potential technique for generating renewable energy while simultaneously lowering the cost of manufacturing. This is because the excitation smoothly attains the dissociates and interface, resulting in minimal recombination. 98 Only water will be released during the combustion process of hydrogen gas (H2), which is a by-product of the energy conversion process. Because of this, hydrogen has been identified as a viable contender for renewable energy to alleviate the energy problem created by the depletion of fossil fuel supplies as well as the resulting negative environmental consequences. Electrolysis, thermochemical conversion of biomass, plasma arc decomposition, biofuel reforming, thermochemical water splitting, and gasification are just a few of the techniques employed for hydrogen gas production.88,99
CF is the ternary oxide discovered for the first time for water splitting as a photocathode. They became the first to demonstrate the utilization of CF by pellet sintering at extreme temperatures (1200°C) as a photocathode in the hydrogen evolution reaction (HER). 100 Ida et al. 101 built a simultaneous cell for water splitting in basic conditions, employing a photoanode of a TiO2 electrode as well as a photocathode of a (hk0)-oriented CF film plated on a Pt substrate. The bulk heterojunction photocatalyst MgFe2O4/CF NPs is highly efficient photocatalyst for hydrogen evolution and oxidative degradation of isopropyl alcohol. 102 Nano-configured particulate equipment has been created for photocatalytic applications using cost-effective solution-based techniques. As a result, nanoparticle junctions of CF/PbBi2Nb2O9, 103 CdS/AgGaS2, 104 WO3/W/PbBi2Nb2O9, 105 and TiO2/CdS 106 have been created, demonstrating improved photocatalytic activity for water splitting and organic compound degradation when compared to materials without a junction structure. Under visible light (>420 nm), the material demonstrated remarkable photocatalytic activity similar to oxide photocatalysts for hydrogen evolution from water, isopropyl alcohol degradation, and photocurrent production. A simple polymer complex (PC) approach has been used to create this adequately structured material. Metal ferrites with a spinel structure, such as p-type CF (CF) 100 and n-type MgFe2O4 (MF), 107 were chosen as the starting materials. The most significant magnetic materials are spinel ferrites, which also have intriguing photocatalytic capabilities to evolve oxygen and hydrogen from water. 107
Based on the absorption coefficient of CF, a self-sufficiency assessment is carried out when CF is employed to absorb sunlight. For fuel converters or electricity, an excellent solar energy system must operate without any external bias potentials or biasing components. 108 The synthesis of fuels via photoelectrochemical converters (PECs) is the most convenient way to store energy currently available. It enables the storage of solar energy as chemical bonds, which is beneficial. The voltage necessary for fuel synthesis is given by the sun's absorbed photons. However, because of loss of voltage owing to resistive and charge transport, electrode overpotentials, and other factors, semiconductors that absorb sunlight must yield photovoltages that are larger than the redox processes involved in the production of fuel. Solar energy has sparked a particular interest in the production of electric power and hydrogen from water, both of which may be used to power devices. A manufactured photosynthesis system containing semiconductor electrodes is required for this. Light breaking down water molecules into oxygen and hydrogen allows them to function.
For these semiconductors to be suitable for use in the system of solar water splitting, they should possess many specific features. As a result of its characteristics for water splitting, the CF semiconductor is one of the world's most extensively researched ternary oxide photocatalysts (2H2O → 2H2 + O2).109,110 In experiments, the energy band gap of CF is between 1.85 and 1.9 eV. The energetic position of the valence band (VB) and conduction band (CB) edges is appropriate for the splitting of water: Water molecules were oxidized by holes from VB (promoting the production of O2) and reduced by electrons from CB (promoting the production of H2). 111 Furthermore, it is composed of low-cost components like calcium and iron. With these characteristics, this chemical has been investigated for its ability to induce the splitting of water when exposed to solar light. When n-type TiO2 electrodes and p-type CF are connected under light, oxygen and hydrogen are generated. Photoelectrochemical converters that are self-sufficient (that is, they operate in the absence of any kind of external bias potential) must produce adequate photovoltage for water splitting [V = 1.23 V] to function correctly. Without this, the photoelectrochemical converters will require external potential and cease to be self-sufficient. Despite this, the photovoltaic voltage V must be greater than 1.6 V when energy loss is considered. The valence band (VB) and conduction band (CB) edges should be below the O2/H2O (5.73 eV from the vacuum zero energy) and above the H2/H2O (4.5 eV from the vacuum zero energy) redox potentials for the system to be self-sufficient. 112
On the other hand, CF has been investigated for its magnetic characteristics, which is a distinct perspective. Doping is an alternate method of enhancing the efficiency of PECs. The CF's photocurrent density may be empirically increased by doping it with Na, Ag, and Mg under a variety of light sources; because of the doping process, the band gap and electrical constitution of the semiconductor were changed.110,113 CF nanofilms (thickness = 100 nm) were fabricated by pulsed laser deposition method (PLD) and were used to produce H2 gas by splitting water. It gives significant production of H2 gas on the photoelectrode of CF in the absence of a sacrificial agent and external voltage. 114 CdS-CF fabricated by thermal deposition was utilized for H2 evolution. After 3 h of visible light illumination, the photocatalytic H2 evolution quantity on the optimized CS/CFO composite from water with ethanol was up to 2200 mol, almost double that of the pure CdS. 115
Oxygen evolution reaction
Electrolysis of alkaline water has recently gained much interest since alkaline circumstances enable perovskites to be used as electrocatalysts. In this respect, research over the last decade has focused on developing highly active electrocatalysts made of nonprecious and plentiful materials. The slow anodic oxygen evolution reaction (OER) (4OH− → O2 + 2H2O + 4e−) is the fundamental problem these days for the splitting of alkaline water. 116 As a result, cost-effective electrocatalysts with strong activity in OER are highly coveted. As iron is one of the most abundant metals in nature, electrocatalysts based on iron (Fe) have lately attracted a lot of attention in the search for alternative catalysts for OER. Fe also has some benefits, including low toxicity, low cost, and environmental friendliness. Although simple oxides of Fe have modest activity in OER on their own, combining them with different metals may significantly improve the performance of OER. 117 Because of their excellent electrochemical activity and ease of synthesis, many perovskite-type oxides and spinels based on Fe have piqued interest. Yamada et al. 101 determined that the performance of OER in Fe-based perovskites is CaFeO3 > SrFeO3 > LaFeO3 > YFeO3. 118
Researchers are focused on bimetallic oxides of Ca and Fe due to the abundant nature of Ca (fifth Clarke number), the inexpensive cost, and eco-friendly behavior, all of which make them very appealing in practical application terms. The orthorhombic CF was chosen as a potential OER electrocatalyst CF is a p-type oxide semiconductor used in the solar splitting of water as a photocathode. The efficiency of oxygen reduction reactions is very high. 119 It has a reasonably high faradaic efficiency and has been used in the solar splitting of water as a photocathode. There has been no earlier publication that we are aware of that uses CF in the electrochemical OER in splitting water as an anode catalyst. Using comparisons with comparable Fe-based bimetallic oxide analogs, the electrocatalytic activity of OER of CF has been examined in an alkaline medium. It explains key aspects of boosting the activity of OER of CF by analyzing the activity of OER of CF in an alkaline medium.
In terms of environmental friendliness, cost efficiency, and simplicity of synthesis, the assessment of the activities of OER specifically revealed that CF is a good option as an inherently active OER electrocatalyst Furthermore, the performance of OER for long-time endurance is a vital consideration for practical application. First, it must be shown that CF accurately catalyzed OER catalysis with its original composition and structure throughout the early steps of the measurement of OER. 120
Gas sensing
Poisonous gas sensing has always been a top issue in pollution management, industry, the defense sector, and medical diagnosis. Gas sensing is also necessary for the daily life of human beings, not only beneficial to these industries. 121 Researchers are looking for sensing devices with exceptional selectivity, excellent sensitivity, recovery speed, rapid response, and porous structure. 122 Therefore, CFNCs were employed as cost-effective gas sensors with excellent efficiency, for poisonous gases like HCHO, isoprene, etc.
Formaldehyde (HCHO) is a toxic gas that is dangerous to human health. Researchers have looked at the gas-sensing capabilities of the CF to HCHO conversion. The CF-based sensor has an increased gas response (16.50), a rapid response-recovery time (153, and 54 s), excellent moisture resistance, and long-term stability, among other characteristics. This one-of-a-kind CF nanocube has the potential to be exploited as a promising HCHO gas detection material. When inhaled in high concentrations, formaldehyde may cause cancer and congenital disabilities in humans. Even at low concentrations, HCHO shows teratogenic and carcinogenic behavior and may cause significant health issues in humans. Indoor ornamental elements progressively emit formaldehyde, which accumulates in the space. As a result, creating sensitive formaldehyde sensors is critical for preserving the indoor environment and human health. 123
The isoprene molecule has wide applications in the commercial synthesis of rubber. While its uses have been thoroughly researched, there has been little study on the safe usage of this molecule. The scarcity of safety information has been a source of concern, and this research focuses on assessing the potentially harmful features of isoprene as a result. 124 Isoprene is a hazardous volatile organic molecule that is poisonous to human health and should be avoided. The development of gas-sensing materials for the detection and analysis of isoprene has a significant amount of potential use. The hydrothermal technique has been used to produce CF, ZnFe2O4 (ZF), and CF/ZF composites, among other materials. The CF/ZF-50% mixture, in a particular material, has a porous structure like walnut and demonstrates outstanding gas sensing capabilities to isoprene, along with a strong response to 30 ppm isoprene (S = 19.50), a quick reaction-recovery time (72–35 s), as well as a lower processing temperature (200°C). Apart from that, the CF/ZF-50%-based sensor has high selectivity, long-term stability, excellent moisture resistance, and a low level of detection (LOD) of 0.12 ppb (Figure 12). More in-depth investigation reveals that such exceptional gas sensing characteristics of CF/ZF-50% could be contributed to the improved performance of electron-hole (e−-h+) separation rate, the high content of OV, the reduction of charge transfer resistance, the narrowed band gap, the large BET surface area, as well as the establishment of CF/ZF p-n heterojunctions. These findings suggest that using suitable CF in conjunction with ZF to generate CF/ZF is a potential technique for developing a better isoprene gas sensor for various applications. 125 To monitor ethanol gas at room temperature, gas sensors based on p-type metal oxide semiconductor CaFe2O4 nanopowder have been developed and evaluated. 126 CaFe2O4 and PANI nanocomposites were fabricated and used as LPG sensors. 127
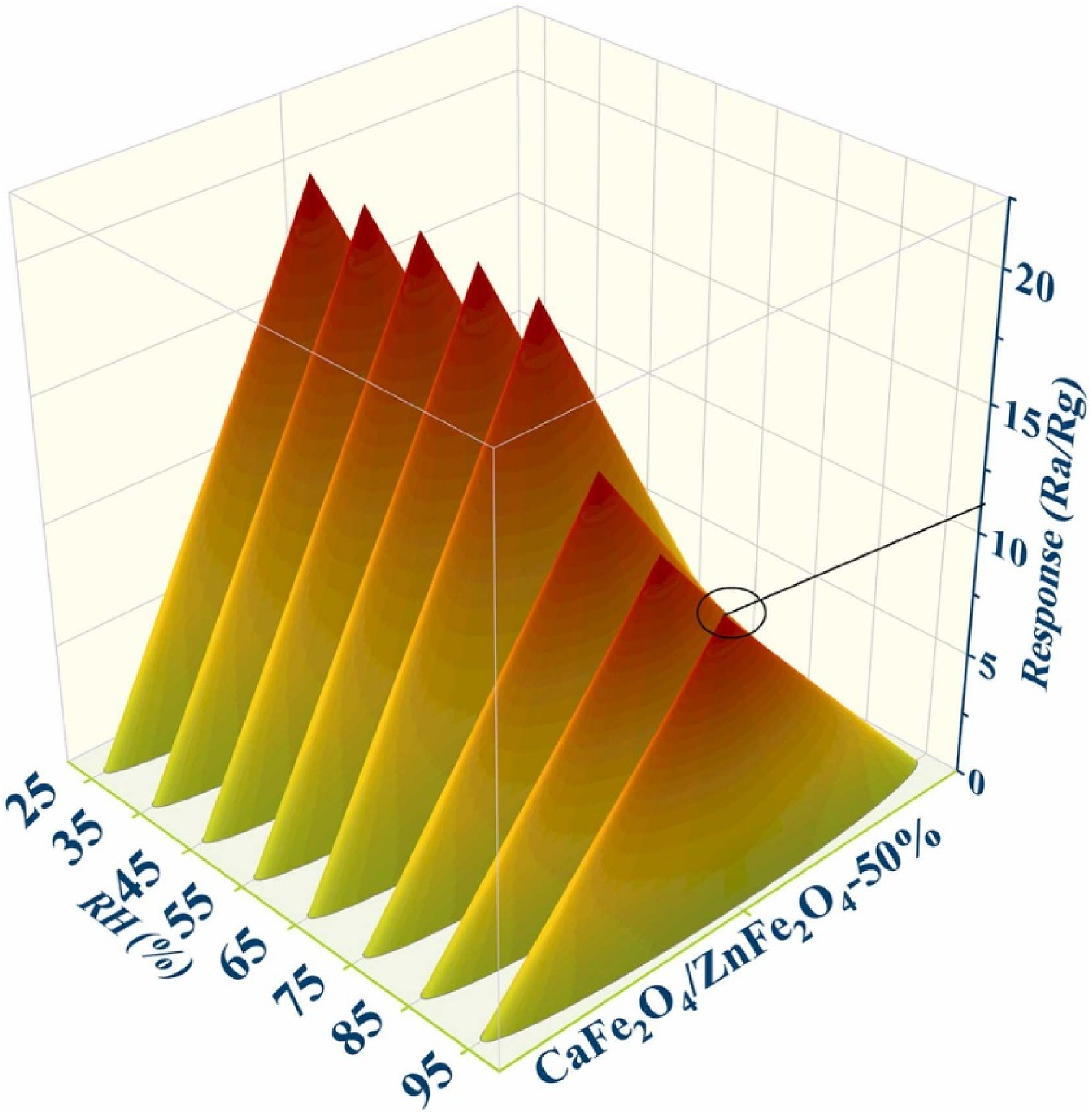
The gas responses of CaFe2O4/ZnFe2O4-50%-based sensor (working at 200°C) to 30 ppm isoprene at different relative humidity (25, 35, 45, 55, 65, 75, 85, and 95% RH), and the environment temperature is 25°C in the test chamber. 125
Medical application
CFNPs and NCs are excellent in medicinal applications as anticancer agents and drug delivery. CF is a suitable carrier for drug delivery systems due to its biocompatible as well as environmentally friendly behavior. 128 Thermal treatment processes were utilized to fabricate CF@PVA (calcium ferrite/polyvinyl alcohol) at different temperatures. These are the significant nanocarriers for the delivery of the drug curcumin as well as in the therapy of breast cancer. It has been observed by the drug delivery system of curcumin that these nanocarriers show pH-dependent behavior. The best performance was regarded in the case of nanocarriers fabricated at a temperature of 823 K.. 129 CFNPs fabricated by the sol-gel method were used for the drug delivery system. 130 Ionic gelation desolvation-fabricated casein-CF nanohybrids exhibit anticancer activity against breast and ovarian cancer. These nanohybrids were suitable carriers for drug delivery systems—89.54% encapsulation of hesperidin optimized by Taguchi in the carrier. The use of superparamagnetic CFNPs increased magnetically induced drug delivery and drug encapsulation. The carrier has a characteristic of the stimuli-responsive release of the drug, with a more incredible release under acidic pH (5.4 and 1.2) and good stability at physiological pH (7.4), which is favorable for anticancer applications. 131 The BSA-CF (bovine serum albumin) nanohybrid fabricated by the sol-gel method was used in the controlled drug delivery system of hesperidin and eugenol. 85.58 and 62.94% encapsulation efficiency were optimized for eugenol and hesperidin, respectively. It shows a magnetic field and pH-dependent drug delivery system for hydrophobic anticancer drugs. The nanocarrier showed pH-dependent drug release, great release at acidic pH, and excellent stability at standard pH, indicating that it might be used for cancer-site-specific drug delivery. In the presence of an external magnetic field, superparamagnetic CFNPs allowed for successful drug loading and faster drug release. 132 CFNPs produced via the auto-combustion process were effective carriers for drug delivery. 133 Casein-CF nanocarrier was obtained by the hybridization of protein carrier casein and superparamagnetic CF and was used in the delivery of curcumin anticancer drug. 134 HAP-CF (hydroxyapatite) is manufactured using a simple method to administer amoxicillin. For regulated and targeted drug delivery systems, HAP-CF may be considered a stimuli-responsive and biocompatible carrier. 135 In the fabrication of derivatives of spiro-oxindole by one-pot synthesis, a three-component reaction of CH-acids (1,3-dicarbonyl compounds), malononitrile, and isatin gives a significant yield in the presence of CF@MgAl-LDH. 136 CF-Chitosan was used in ampicillin's effective drug delivery system. 9 CF-PVP (polyvinylpyrrolidone) nanocubes fabricated by thermal deposition and exchange of ligands were used for the pH-dependent delivery of doxorubicin. 137
Comparison of CF with other metal ferrites
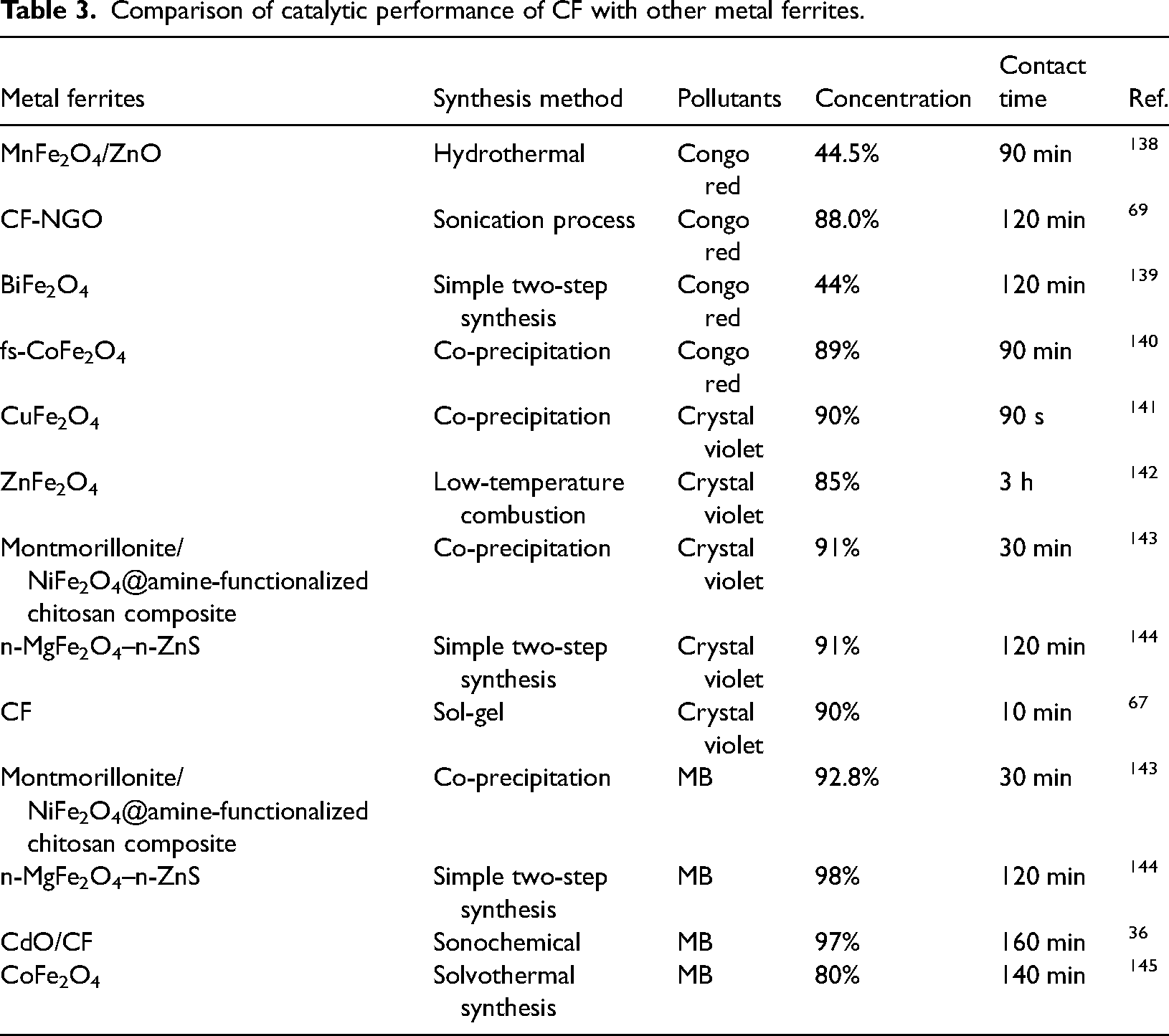
CF and other metal ferrites are widely used in various applications due to their specific characteristics and properties explained in the literature. However, the catalytic performance is highly dependent on the fabrication method; therefore, if the same ferrite is fabricated followed by different methods, the catalytic activity of the ferrite will be different. Comparing CF with other metal ferrites, it has been found that they have similar applications, like water decontamination. Moreover, CF has excellent activity compared to other metal ferrites, which can be explained by the data in Table 3.
Comparison of catalytic performance of CF with other metal ferrites.
Practical implications and cost benefits
For the practical application of any material, it should be stable, reusable, and cost-effective, with easy fabrication and excellent applicability. CFNPs and NCs have tremendous applications due to their specific characteristics explained in the literature. Meanwhile, the stability and reusability of these NCs make them practically applicable and economically feasible. Since both the metal ions Ca and Fe are abundant in nature, these metal precursors are readily available and affordable. Due to the magnetic behavior of these, they are accessible to separate without using expensive equipment. Therefore, these NCs are practically applicable at the commercial level.
Reusability of CFNPs and NCs
Stability and reusability are the crucial factors of a catalyst for practical application, which makes the catalyst significant for cost-effective and commercial application. CFNPs and NCs are easy to separate after use because of the magnetic behavior of iron and can be reused for the cycle. But in the case of CFNPs and NCs, there is a huge possibility of iron leaching, which causes a significant decrease in the catalytic activity. Therefore, it is necessary to check out the stability of the catalyst. To check the stability and leaching of the catalyst, some characterization techniques (XRD, FTIR, SEM-EDS) should be done. Instead, most CFNPs and NCs are stable and reusable for consecutive cycles. For example, CF-800 is stable and performs well for eight consecutive cycles, and no iron leaching was obtained. 24 CF12 gives 93.5% degradation of MB after five cycles, making it an excellent catalyst 60 . CF-Bi2O3 was reusable for six cycles with a slight decline in catalyst efficiency (first cycle: 95.7% and sixth cycle: 94.3%). 63 MgFe2O4-CF was reusable for up to three cycles with similar catalytic performance. 64 CF-NGO gives excellent performance for six cycles with a little decrement in performance. 69
Conclusion and future perspectives
Compared to other spinel ferrites, CF has gotten more attention because of its biocompatibility, superparamagnetic behavior, and abundant nature. This is the first review that deals with the recent advancement of CFNPs and their binary and ternary NCs in different areas. The search has found that the significant application of CF is based on its photocatalytic activity. This photocatalytic mechanism mainly involves the formation of radicals such as hydroxyl radicals and superoxide radicals. The spinel structure, magnetic behavior, and applications of CFNPs and NCs are explained in detail in this study. Various techniques, such as sol-gel, co-precipitation, molten salt, and solution combustion method, are used to effectively synthesize CF-based composite. CFNPs and NCs were effectively used in degrading dyes, pharmaceuticals, and the removal of heavy metals. Various other applications of CF-based composite for antibacterial performance, transesterification reaction, gas sensing, dye detection, drug delivery of some anticancer drugs, and H2 and O2 production by water splitting are also discussed here. Although so much development, there is a gap exists, which is as follows:
Important characteristics for enhancing catalytic capabilities include nanoparticle crystallinity, size, and accessibility of the pollutant surface. Regarding this, tiny NPs with good crystallinity are very advantageous due to their large surface area, which encourages strong catalytic activity. High-temperature manufacturing is required to create nanoparticles with enhanced crystallinity. Therefore, it is crucial to optimize crystallinity without changing the size of the particle. There are many green sources of calcium development, like egg shells, animal bones, and others. Green synthesis is a cost-effective technique that also helps in the utilization of waste products. So, further research should be carried out in the direction of green synthesis of CF, which is beneficial for our environment as well. Double doping of anion and cation on the CF surface may enhance photocatalytic efficiency due to a reduction in the recombination of electrons and holes .
146
CFNPs are an application in the field of energy storage (Li-ion and Na-ion batteries) that is less studied. Transesterification and water splitting for green and clean energy production should be explored. The large-scale production of CFNPs for various pollutants is not yet discovered. So large-scale production is necessary for energy and environmental applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
