Abstract
Vanadium dioxide (VO2) and many binary compounds of vanadium can transform from a semiconducting state to a metallic state under the influence of temperature or light. The transformation is triggered by modifications in the oxide's crystalline structure, often initiated by changes in its light-sensing and electrical attributes. This ability makes them suitable for smart coatings, electronic switches, self-cleaning surfaces, window glass, and many more. These potential applications have prompted tremendous interest in VO2thin films, focusing on their fabrication and application. This article reviews current fabrication methods for VO2thin films, the potential application of their thermochromic and photochromic properties in self-cleaning coatings, the challenges facing their application, and some remedies devised to address the challenges. The various challenges facing its different synthesis and fabrication methods are also highlighted. First, the different transitional phases of VO2and routes to transform from one stage to the other are presented.
Introduction
The emergence of the Industry 4.0 revolution emphasizes the need to use smart materials in many engineering applications. Smart materials are those that manifest self-change once subjected to external stimuli. Vanadium dioxide (VO2) is an inorganic substance that exhibits phase change at very low temperatures. 1 and is thus considered a smart material. The compound has good absorbance in the average infrared spectrum, making it suitable for photocatalytic application. It can continually transform under the influence of temperature and lighting from an insulator or semiconductor to a conductor and vice versa, making it a thermochromic and photochromic smart material. 2 This phase transformation usually occurs with VO2at around 68°C, and in a thin film, this can assist in controlling the sun's radiation. 3 Vanadium itself is polyvalent, existing in several oxidative states. These include V2+, V3+, V4+, V5+,, and more. 4 These vanadium's outstanding properties allow it to exhibit several binary compounds of vanadium and oxygen, such as VO, VO2, V2O3, and V2O5. A reversible semiconductor-to-metal transition, popularly referred to as a metal-to-insulator transition (MIT), often occurs when vanadium oxides express an abrupt change in their intrinsic electrical conductivity under externally induced conditions of stress or temperature. 5 This is why VO2 has been among the most widely recognized oxides in the past six decades for high-sensitivity switching elements and smart devices, including window glass. Besides their catalytic, electronic, and thermochromic applications, self-cleaning and photocatalytic degradation are essential applications of VO2thin films. 6 VO2 has also been doped with other metals or metal composites to improve its self-cleaning functionality.4,7,8 The doping resulted in better optical properties, improved photodegradation characteristics, and excellent hydrophilicity. These made them more suitable for energy saving and smart window uses. Studies on the influence of hydrogenation, 9 optical phase transition properties,10–12 effect on deposition temperatures,13,14 and thermal properties of VO2 thin films15–17 have also been carried out
While VO2 has attracted significant interest as a diverse inorganic material for energy-saving and innovative appliance applications, the experimental construction of pure thermochromic VO2 is very difficult. It is still complicated to prepare a stable thermochromic VO2 structure free of impurities in bulk quantities due to their low redox stability, multivalent character, and many oxidation states. 18 The growing of thermochromic VO2 nanostructures on particular substrates frequently results in structures with confined size and output, suggesting the need for a straightforward and well-ordered large-scale technique for fabricating stable VO2 monocrystals.
However, some authors have researched to mitigate thermochromic deterioration and improve the stability of VO2 thin films.19,20
For more than six decades of existence, research work on VO2has produced only a few review publications. Chain presented a article highlighting the optical characteristics of vanadium oxide coatings, linking them to specimen fashioning and the resulting film structure to better understand film properties. 3 Eyert reviewed the band theory of VO2 and presented the first fundamental calculation to determine the electronic composition of metalliferous rutile and the nonconducting monoclinic (M1) form of VO2. 21 Nag and Haglund reviewed the different synthesis methods of VO2 thin film, emphasizing how VO2 films can be doped to alter their switching characteristics and transition temperatures. 22 Gao et al. offered a fresh look at polymer-aided deposition methods of nanoceramic thermochromic VO2 films and compared them to gas-phase methods. 23 Wang et al. discussed the latest progress in VO2 smart coatings, presenting techniques to ameliorate its thermochromic features. 24 Liu et al. summarized the properties and significant application of VO2, giving insight into future studies and highlighting opportunities and challenges. 25 Ko et al. reported on the most current advances in VO2 technology. Their review discussed the physical sources of optical properties of VO2, developments in switchable color filters, daylight radiative cooling, and highlighted recent discoveries in VO2 photonic applications and more. 26
Currently, the investigation into the properties of self-cleaning films is attracting interest because of their low cost, high durability, and the fact that there is no downtime for maintenance. 7 One form of VO2can be photocatalytic, thus demonstrating its ability to self-clean.27,28 This article presents a detailed review of advances in fabrication methods for VO2 thin films, challenges facing its application, and suggested remedies to address them. First, the different transitional phases of VO2and routes to transform from one stage to the other are presented. Brief historical background of each fabrication technique and current innovations to improve it are reviewed. The potential application of VO2 thin film, challenges facing its applications, and proposed solutions are also examined. The manuscript concludes with the current state of VO2 thermochromism and photochromism properties in self-cleaning technology and suggestions for future research perspectives.
Background of the different transitional phases and phase transformation routes of VO2
Changes from one phase to another in the VO2 binary structure influenced by external stimuli such as magnetic field, strain energy, heat, and surface energy bring about corresponding changes in its physical properties known as a phase transition. This resonates with crystal changes from the low-temperature monoclinic phase to the rutile phase at higher temperatures due to anatomic variance in electron orbitals. It is, therefore, critical to understanding the physical and chemical characteristics of the different VO2 binary phases as it will facilitate the mode of application of each phase. The most common phases recently reported are shown in Figure 1, together with conditions that enable their transformation from one to another. VO2 (M) can either appear as VO2 (M1) or VO2 (M2). Both polymorphs display the desired MIT property. The difference between the two is the space grouping of their unit cell properties. 29 VO2 (R) is the high-temperature rutile phase of VO2, often obtained in the presence of a surfactant. 30 They possess lesser unit cell values than the VO2 (M) polymorphs.
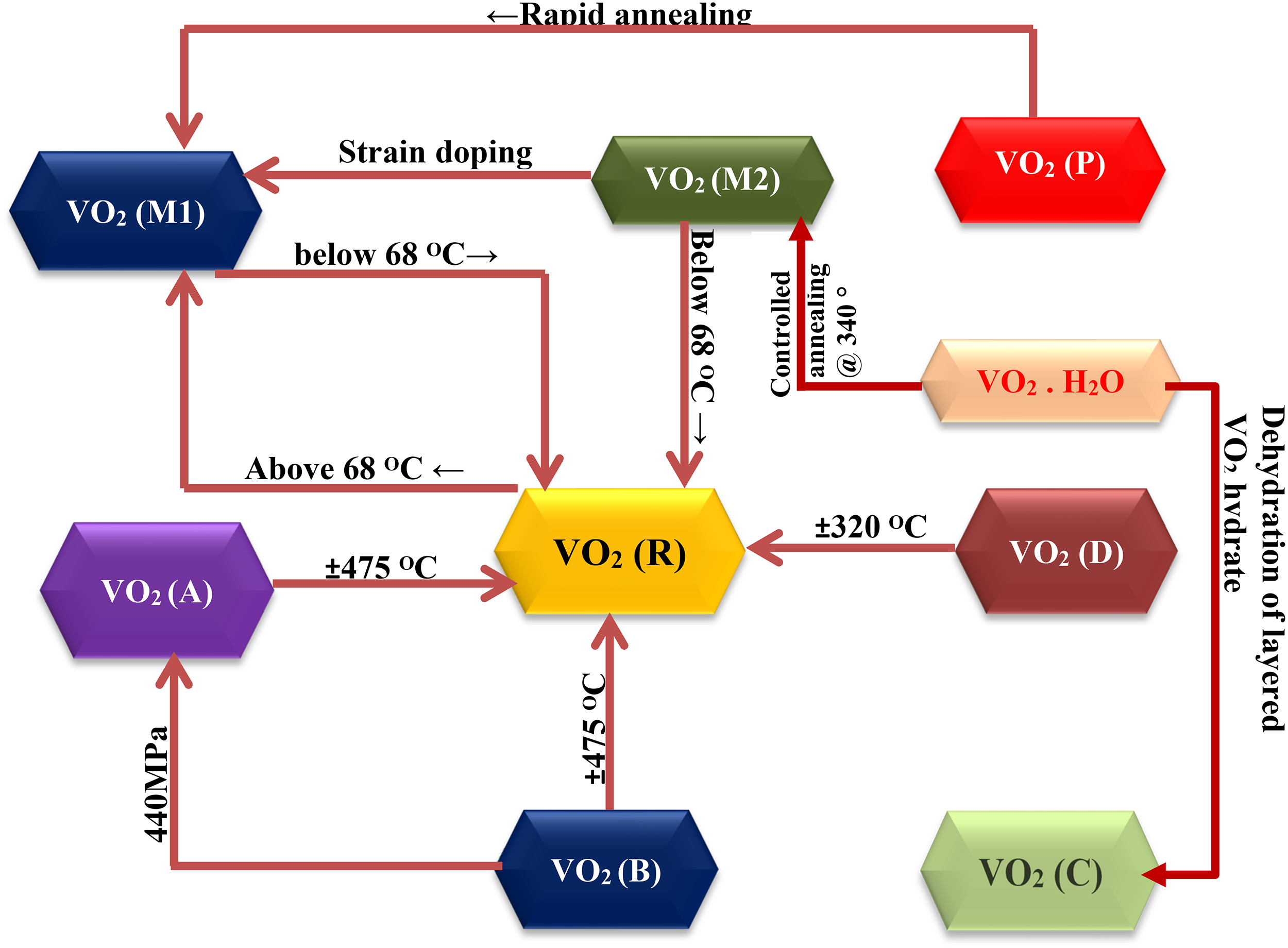
VO2 (A) is another phase of VO2 having unit cell values higher than those of VO2 (R). They display MIT property at a transition temperature of 68°C. VO2 (B) is active between 150 and 260°C. It has found application as a suitable material for the electrode of thermal sensitive batteries due to its layer structure. 33 VO2 (C) can span from a few hours to a few days. It has a format that is made of square pyramids of VO5 binary compounds sharing its four square edges with four close squares of the VO5 pyramid. VO2 (C) was first prepared by Hagrman et al. in a process termed the soft chemical method by dehydrating layered VO2 hydrate (VO2.H2O). 34 VO2 (D) possesses more remarkable magnetic properties and quickly transforms into VO2 (M) at 300°C. Its typical reaction conditions are still under establishment. 35 VO2 (P) was first synthesized by Wu and his collaborators using an elementary chemical reaction path. It quickly transforms into VO2 (M) once under rapid annealing. 36
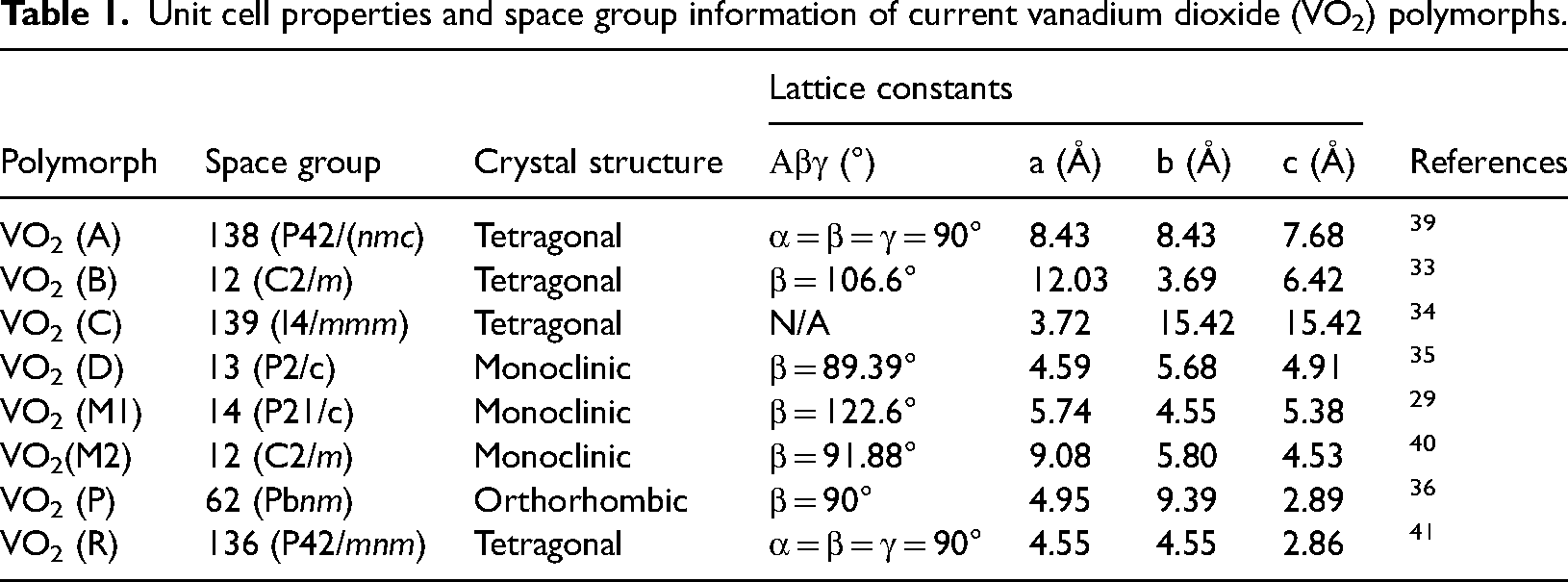
Of all the polymorphs, VO2 (M), VO2 (R), VO2 (A), and VO2 (B) display greater similarity in structures, which is a VO6 octahedron. The six oxygen atoms surround the one vanadium atom at the center. The shapes inform the different spatial groups of VO2 of the octahedron and the different connection models of the atomic coordinates. VO2 (M) contains the transitional and monoclinous phases of VO2(M1) and VO2(M2). After exceeding the MIT temperature, VO2 (M1) transits to VO2 (R). While transiting from the nonconducting to the conducting phase, the six-sided V-O structure changes from a partial six-sided configuration at low to normal octahedron anatomy at high temperatures. The octahedron V-O bond angle also varies from 90°C to between 78 and 99°C. 30 These anatomic changes correspondingly impact the mechanical, magnetic, optical, and electrical properties of VO2 (M1) and VO2 (M2), which may be harnessed for various applications. VO2 (M2) and VO2 (M1) nonconducting phases with the monoclinic structure are identified separately by the alignment of V atoms along the c-coordinate. In VO2 (M1), the pairs of V-V atoms chain along the c-coordinates are curvy, while in VO2 (M2) phase, the V atom chains are either paired and uninclined or are inclined but unpaired along the c-coordinate. VO2 (M2) can be regarded as an intermediary state of VO2 (M1) because it is formed when VO2(M1) is strained along a particular axis or is doped with three positive valence ions. 37 VO2 (A) is made of two blocks of the two-sided octahedral shape formed along the c-coordinate and also possesses MIT property at a higher transition temperature of 162°C. Unlike in VO2 (M), monoclinic VO2 (A) and VO2 (B) do not display visible MIT transition properties that may encourage noticeable reforms in their ocular and electrical stance. The temperature coefficient of resistance of VO2 (B) is outstanding at −7%/K near ambient temperature, making it suitable for application as electrode materials. 38 Below 440MPa, VO2 (B) transforms into VO2 (A). Horrocks et al. demonstrated that in the presence of argon gas, VO2 (A) and VO2 (B) calcined at 475°C could be transformed into VO2(R). Various phases of VO2 may still emerge for thin film and nanostructural applications with the advancement of material manufacturing technology. Table 1 lists crystallographic information on different VO2 polymorphs, including their space data.
Unit cell properties and space group information of current vanadium dioxide (VO2) polymorphs.
Methods to synthesize and deposit VO2thin films
In this section, the various VO2 thin-film fabrication methods are described. It has been reported that the morphology of VO2 thin films depends on the synthesis and deposition method. 42 Here, the merits and demerits of each approach relative to the surface texture and possible influence on self-cleaning ability are reviewed
Sol–gel
Sol–gel is one of the wet chemical synthesis procedures that involves dissolving atomic particles of a precursor (generally a metal alkoxide) into water or alcohol and then heating and stirring it to form a gel. The prepared precursor solution may be hydrolyzed or condensed, aged and dried, or heat treated. The metal alkoxide activity, the water-to-alkoxide ratio, solution pH, redox conditions, solvent properties, and additives affect the hydrolysis and condensation reactions in sol–gel synthesis. The initial precursor state will significantly impact the prepared films’ chemical, physical, and stoichiometric properties. Catalysts are often employed to regulate hydrolysis and condensation in sol–gel processes. Figure 2 shows the sol–gel process and possible deposition routes following the gel formation. The first sol–gel synthesis of VO2dates back to 1983 when Greenberg used Vanadium(V) oxytripropoxide as a precursor to synthesize VO2. 43 Table 2 summarizes recent efforts in advancing the fabrication of VO2 thin films using the sol–gel process.
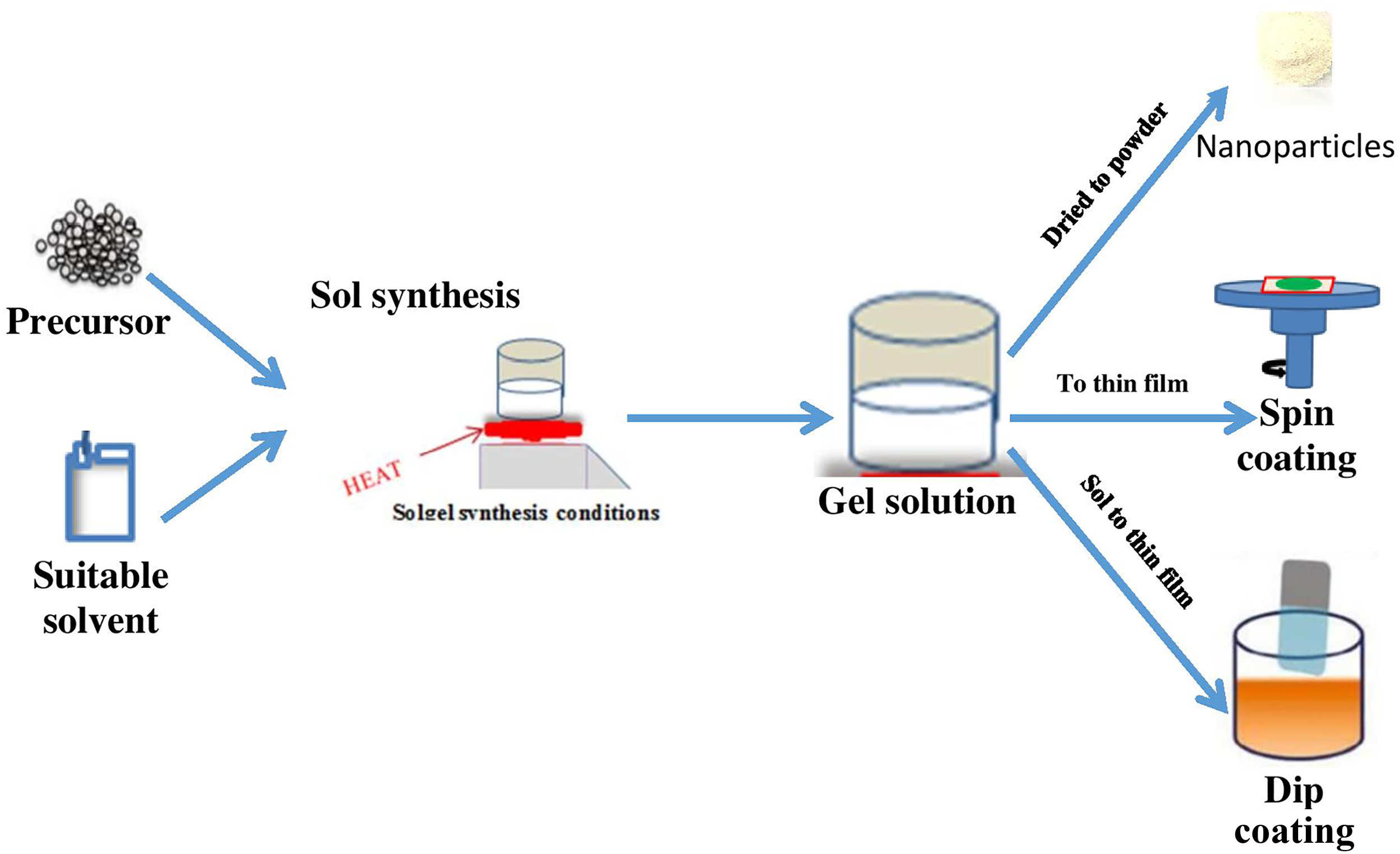
Illustration of the sol–gel process.
Key outcomes of some advances in the sol–gel processing of vanadium dioxide (VO2)thin films.

Many researchers are interested in sol–gel technology because it is a cheap, uncomplicated, and low-temperature process. Key advantages and disadvantages of the sol–gel process are presented in Table 3, together with those of the other deposition methods.
The advantages and disadvantages of the various vanadium dioxide (VO2) thin-film deposition methods, some applications, and references.

The dip-coating method
Dip-coating is a common technique for applying a thin, homogenous coating on substrates of different geometries. It involves immersing the substrate in a gel of the material, usually less dense, to allow excess material to flow back into the gel solution while the substrate is lifted out, as shown in Figure 3. A nip roller squeezes the surplus material off the substrate surface to create a smooth thin film. 48
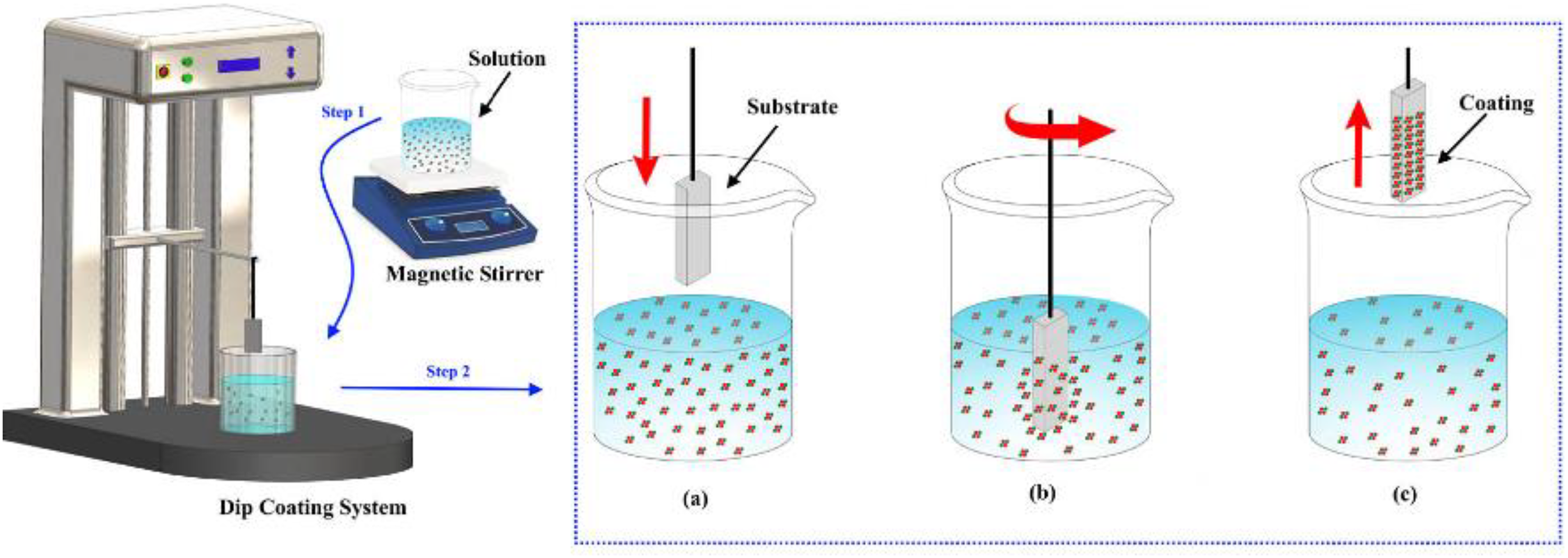
Annotated depiction of the dip-coating technique adapted from Taşdemir et al. 77
The dip-coating technique is handy for parts with complex geometries and curved surfaces, which other spraying methods cannot coat. Although automatic machines are the most convenient, hand dipping works well and speeds up the bonding of the coating onto the substrate. 78 Before dipping, the coating solution is first synthesized, just as in the sol–gel process. The substrate is then immersed consistently in the coating solution.
Usually, the substrate is left in the solution for a short period and then pulled out vertically at a steady speed. 79 As the substrate is lifted from the solution, the material hangs onto it, forming a film. Research works to enhance the deposition of VO2 thin film using the dip-coating procedure are being carried out using different precursor types. Costals et al. used vanadium tetrakis (t-butoxide) as a precursor to synthesize and dip coat VO2 thin films on glass slides. The coated film exhibited alternate semiconductor-to-metal transition around 67°C. 53 The method was also employed to doped VO2thin films with molybdenum and coated on a glass substrate. The thin film was heat-treated in a low-pressure carbon monoxide atmosphere to investigate the impact of the dopants on the semiconductor-to-metal phase change transition temperature. The dopant lowered the transition temperature and improved optical reflectance. 80 Recently, Outón et al. dip-coated VO2 thin films by hydrolyzing vanadyl acetylacetonate in a polymer-aided deposition process to study the impact of process parameters on their use in smart window glasses. The thermochromic characteristic of the VO2 thin films was investigated by altering reduction temperatures and drying conditions. The optical stability of the film relative to its thermochromic action at the microlevel in the material could be aligned to macro changes in its structure. 49
The dip-coating approach allows the film to be deposited on both substrate surfaces, easy doping of different metal oxides on each other, excellent layer uniformity, and the capacity to coat large substrate surfaces. It has the disadvantage of being a lengthy procedure and allows delicate parts to slip and drop off the conveyor, creating inconsistency in film thickness.
Atomic layer deposition
With atomic layer deposition (ALD), thin and precisely patterned films can be created on various surfaces. This deposition technology is well progressed. It addresses the pressing requirement in industries for better-quality thin films with thicknesses stretching from less than a nanometer to a few tens over a tangible substrate surface. Because of the inherent self-limiting capability of the ALD technique, submicron proportionate surfaces, including rounded solids and nanostructured porous materials, can be coated to conformity. 81 These capabilities have made the ALD one of the most preferred thin-film deposition methods.
In the ALD operation, two precursors or more are pumped changeably into a chamber holding a substrate at a set temperature and pressure. This allows materials to be deposited layer by layer on the substrate's surface. 50 In each ALD deposition cycle, the first precursor is exposed in the reactor chamber to create the primary layer on the substrate surface. Excess of the first precursor and all the by-products are then purged out. The second precursor is exposed in the chamber. After which excess of the second precursor and any products are also purged out. The procedure keeps recurring until the desired film thickness is obtained. 51
The ALD's capability to fabricate complex and three-dimensional nanoscale materials has attracted great interest in using the technique to develop thin films. Several factors have been considered for the ALD of VO2 thin films from using novel precursors to considering several parameters governing the process. Willinger et al. used the ALD as a new technique to coat VO2 films as sensing layers on multi-walled carbon nanotube using vanadium n-propoxide as a precursor and acetic acid at 200°C. A consistent and well-controlled coating of vanadium oxide on the inside and outside walls of the carbon nanotubes (CNTs) was achieved.52,82 The technology provided a new method for fabricating non-sticky resilient composite electrode fabric with considerable thickness, controlled porosity, increased electrical conductivity, and cyclic stability in supercapacitors. Blanquart et al. employed the ALD to fabricate VO2 thin films from vanadyl acetylacetonate precursor. They studied the impact of process parameters and post-deposition calcination on the film structure. They reported that it was feasible to preferentially generate either VO2 or V2O5 by changing the environment in which the films were annealed. 83 Prasadam et al. demonstrated the highly conformal nature of the ALD by creating vanadium oxide films on CNTs. Figure 4 shows images from a scanning electron microscope (SEM) of the CNT layers before and after the ALD of a conformal vanadium oxide film with a thickness of 15 nm. The SEM examination of the cross-section demonstrated the conformal character of ALD deposits. The vanadium oxide film coated on the CNT and the silicon substrates had the same thickness. 84

Example of scanning electron microscope (SEM) images for carbon nanotubes (CNTs) prior (upper row) and after (lower row) the atomic layer deposition (ALD) of vanadium dioxide (VO2) film. 84
VO2thin films grown by ALD have also been adapted to address some critical issues, such as complacent absorption and durable instability accompanying the extensively employed spiro-OMeTAD and MoOx hole transport and barrier layers in lucid and n–i–p self-assembled solar cells for two-end sequential devices. Raiford and collaborators presented an alternate hole contact bilayer of a 30 nm thick spiro-Tetra(N,N-di-p-toly)amino-9,9-spirobifluorene (TTB) undoped layer and 9 nm of air-stable vanadium oxide produced through ALD, using vanadium oxytriisopropoxide as the precursor and water as an oxygen source. This led to optimized optical functionality of the perovskite monolithic cell on a heterostructured silicon substrate. 54 Yang et al. used the ALD process to address the limiting effect of power conversion efficiency caused by excessive carrier recombination losses at the contact points in crystalline silicon solar cells. 55 Costals et al. published results of vanadium oxide films produced by ALD, performing as a hole-selective junction for n-type transparent silicon solar cells without needing an extra stabilizing sheet. The obtained both-sided contact solar cells had more than an 18% efficiency increase. This revealed the potential of transition metal oxides deposited using the ALD process. 53 Figure 5(a) shows a scanning transmission electron microscopic image of the deposited vanadium oxide thin film on a crystalline silicon substrate, (d) and (e) revealing the interlayer between Indium tin oxide and Al2O3 solar cell structures, while Figure 5(b), (c) and (f) depicts XPS analysis for vanadium 2p3/2, silicon 2p orbits, and carrier life span against charge density, respectively. Due to its atomic-level dominance in film thickness and high conformance on nanostructured surfaces, the ALD technique deposits various metal-based compounds. ALD methods that extend beyond binary material deposition to permit the synthesis of alloyed, doped, ternary, or quaternary materials are becoming more popular. 85
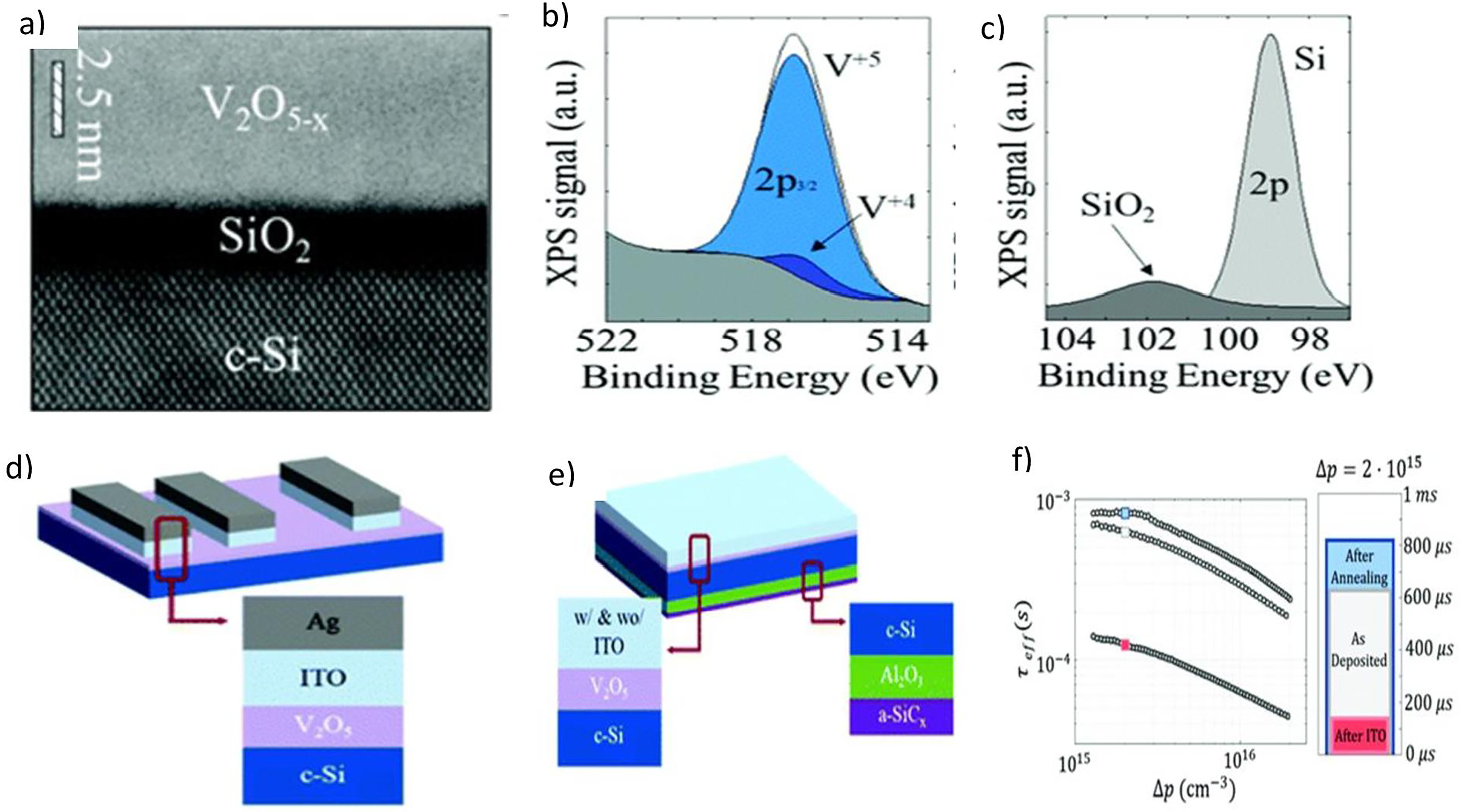
Atomic layer deposition (ALD) deposited vanadium oxides film adapted from Adamatzky and Schubert. 85 ; (a) STEM images of vanadium oxide on bare silicon with the interlayer region, (b) XPS for vanadium 2p3/2 orbitals, (c) XPS for silicon 2p orbits, (d) schematic of film structure with transfer length, (e) quasi state film structure with photoconductance measurement technique, and (f) a plot of effective carrier life span against excess charge carrier density.
The main merits of the ALD process—which at the moment is an excellent deposition method compared to other deposition techniques—is that it can produce pin-hole-free high-density ultra-thin films with exceptional film thickness conformity. The method's drawbacks include significant material and energy waste, toxic nanoparticle emissions, and prolonged deposition times. 56 Comparatively, VO2thin films generated by the ALD process are well suited for microelectronics, holograms, planar lenses, rectifiers, and wave plates.
Pulse laser deposition
Pulsed laser deposition (PLD) is a fast-growing and widely used growth process for thin films and submicron structures, primarily in research settings but with commercial applications. 86 It has drawn significant interest in the past decade due to its capability to manufacture stoichiometric films of complicated substances, such as superconductors with accurate crystal structures. With the PLD technique, a high-energy laser source is pointed at a target surface in a highly vacuumed chamber. The target surface is rubbed to create a plasma plume. The plasma plume contains the target molecules, which are then coated on the substrate surface to obtain a thin film. Using a high-energy laser, rapid melting and evaporation of a target cause all the elements in the target to evaporate simultaneously. This approach can make multicomponent coatings with stoichiometry proportionate to target substances quickly. 87 Figure 6 shows a schematic depiction of the most recent PLD process used to create VO2 thin films on Silicon wafers to study their electrical and optical properties at different temperatures. 88
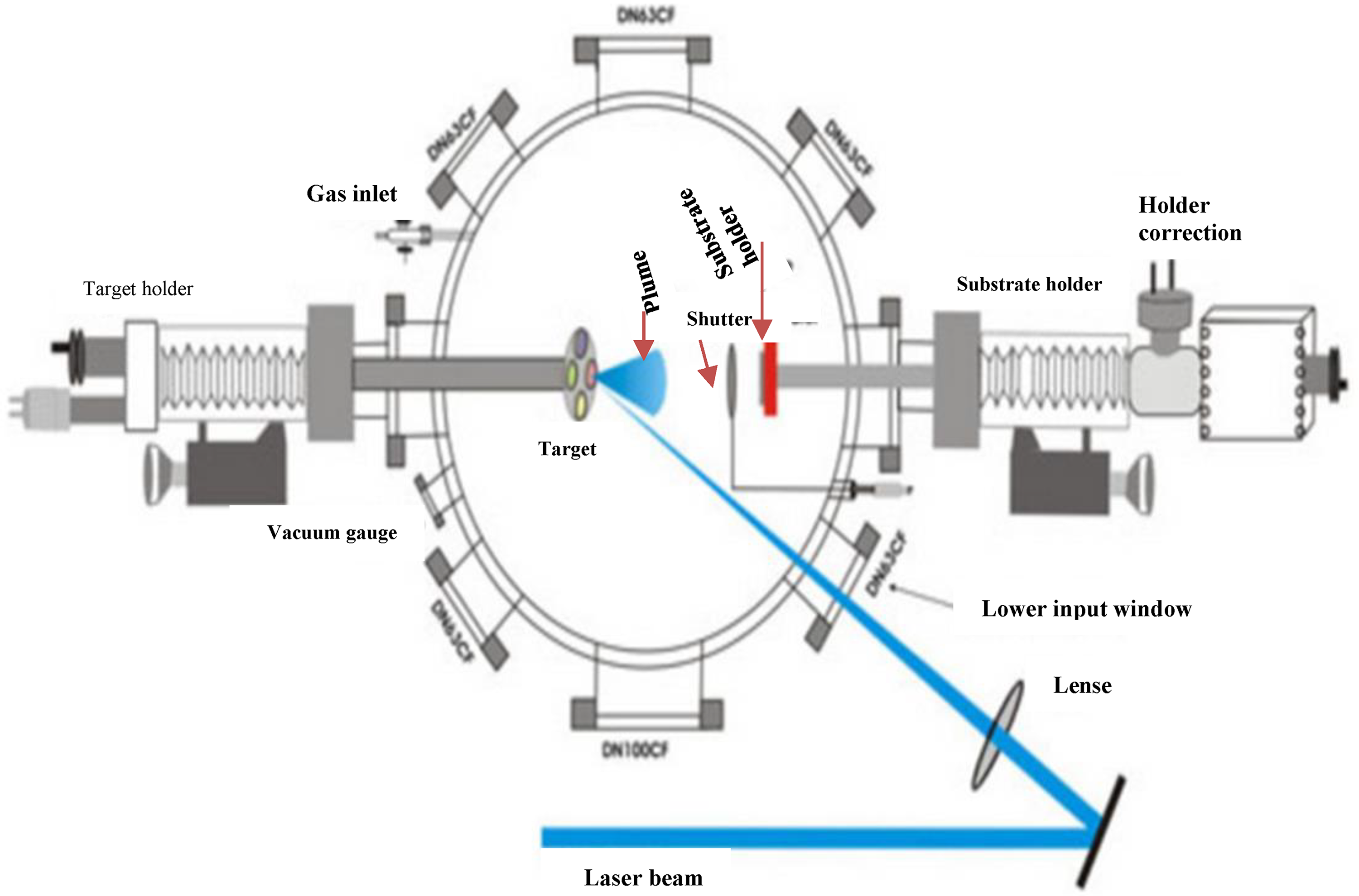
Illustration of the pulsed laser process used to deposit vanadium dioxide (VO2) thin films on silicon substrate reproduced from Ahmed et al. 88 .
It is now possible to control PLD-generated thin film's properties to meet particular requirements for a specific application by adjusting deposition parameters such as laser flux, background gas, substrate temperature, and distance from the target. 89 Several kinds of materials are now being deposited by PLD, including ferromagnetic materials. 90 , chalcogenides, 91 metals, 92 oxides of metals, 93 and laser host materials, like garnets, which are often trichotomous or quaternion in composition.
Lee et al. established the possibility of reducing the order of magnitude of resistivity of VO2hin films by oxidation in a well-controlled environment using the PLD technique. 94 Shibuya and Sawa employed the PLD method to explore the influence of substrate temperature and oxygen pressure on the optical and anatomical properties of VO2 thin films. Their findings created a phase diagram for oxygen pressure against the inverse of substrate temperature for vanadium oxide films, which now serve as a reference for optimizing the conditions for VO2 film formation on silicon substrates. 95 The impact of gamma rays on PLD-grown VO2 thin films to understand the implications of gamma rays on the properties of the thin films on a silicon substrate studied by Madiba et al. Substantial gamma irradiations were focused on the thin film in amounts similar to those experienced by space mission jets. The gamma rays created disorders in the delicate film structures pushing its orientation to lower contact angles. Tetra hertz transmission evaluations demonstrated the preservation of VO2's integrated design after gamma irradiation; gamma-ray exposure caused a reduction in coating resistivity and transition temperature. 96 The findings encourage the possibility for VO2 films to be used as a shielding layer for spacecraft.
PLD is one of the most reliable methods for depositing single-phase VO2 thin films without thermodynamic instability issues. Though this is possible, the process is affected by parameters such as target-substrate distance, oxygen flow rate, laser energy, substrate temperature, and more. Bukhari et al. looked into the impacts of oxygen flow rate on the quality of VO2 thin films under constant pressure. 97 The study established the effect of the gas flow rate on the MIT transition property of VO2 thin film using the PLD process. In the experiments, the resistivity variance between the metallic and insulator phases, the temperature span of the transition, and the width of lagging were all seen to be changing by controlling the flow rate. The results of this research set a benchmark for producing thin VO2 films that are of high quality. Figure 7(a) to (g) shows the morphologies and structures of the obtained VO2 thin films under different oxygen flow rates, (r) depicts XRD, (s) XPS scan, while (t) and (u) are the HR-XPS of V2p and 01s regions, respectively of VO2 thin film from PLD method.
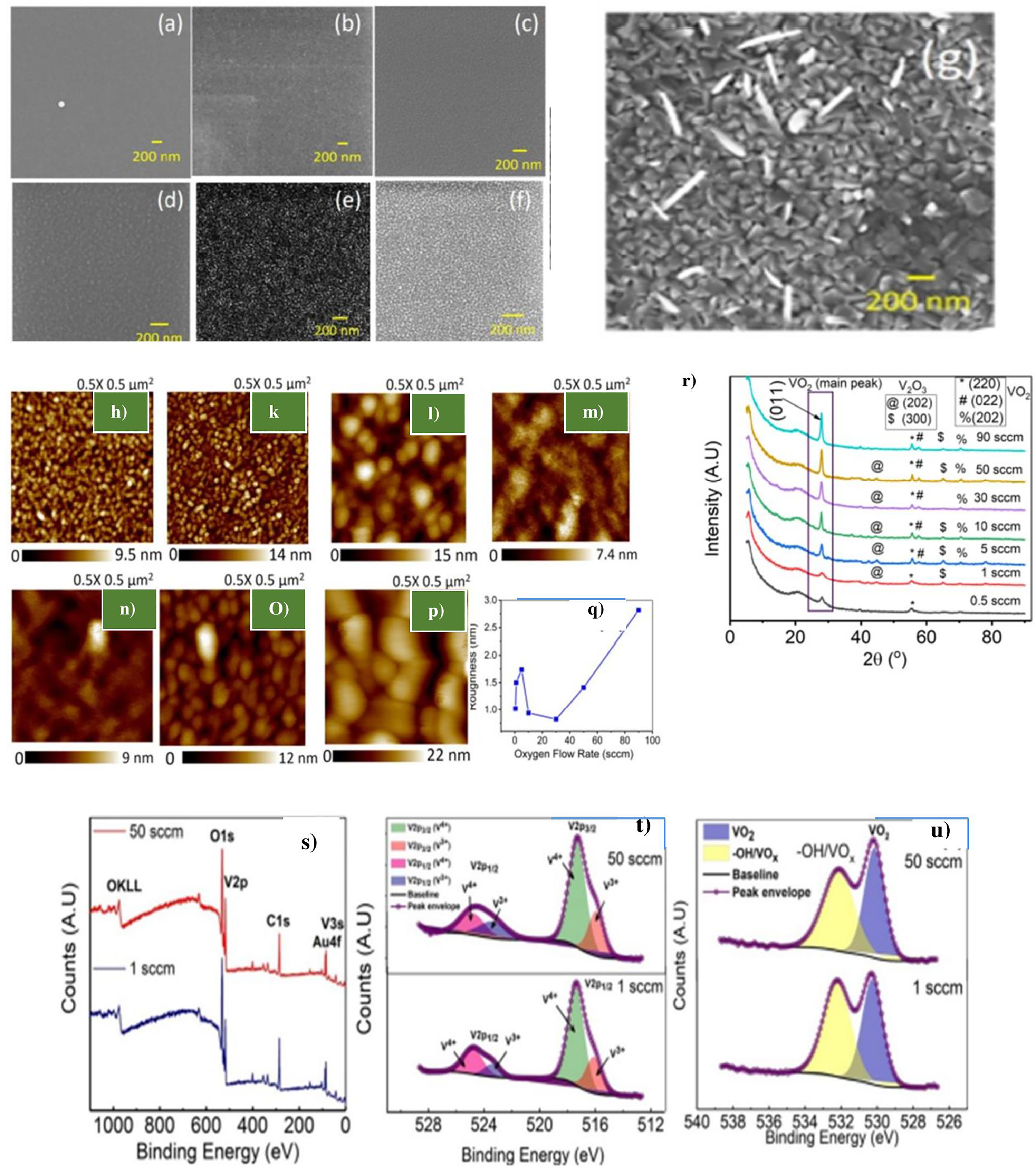
Influence of oxygen flow rate on the thermochromic property of pulsed laser deposition (PLD)-generated vanadium dioxide (VO2) thin films reproduced from Bukhari et al. 97 (a) to (g) SEM images at 0.5, 1, 10, 30, 50, and 90 sccm gas flow rate, respectively; (h) to (p) VO2 thin film's roughness at 0.5, 1, 10,30,50, and 90 sccm gas flow rate; (q) plot of average surface roughness versus flow rate; (r) XRD plot of the VO2 thin film at 0.5, 1, 10,30,50, and 90 sccm gas flow rate; (s) XPS scan at 1 and 50 sccm gas flow rate; (t) HR-XPS image showing VO2 thin film at V2p region and several oxidation states; and (u) HR-XPS spectra of VO2 thin film in O1s state at 1 and 50 sccm flow rate.
The application of VO2 thin films in metamaterials and smart window glass has also been made possible with the PLD method. Bhardwaj et al. grew absolute VO2 (M1) phase thin films on silicon and silicon dioxide substrate in the presence of heat. Despite their lower heat loss, the thin-film transition rate increased resistance and improved reflectivity by 60%, indicating their potential for smart window use. 98 Although limited in terms of surface-covered area, PLD as a thin-film synthesis method continues to pique researchers’ interest due to several key advantages. The PLD method makes it simple to make multicomponent films with the desired stoichiometric ratio. It has a high deposition rate, a short test period, and requires a low substrate temperature.
The advantages of the PLD procedure include adaptability to create consistent films from multi-target with multilayers. There are no restrictions on the types of PLD targets that can be used, making it easy to create a wide range of thin-film materials and heterojunction films. All PLD plasma-generated films have lower porosity, increased adherence to the substrate, improved morphology and chemical composition uniformity, and controlled degree of phase, crystallinity, and thickness of deposited coatings. The whole setup is very affordable, nonpolluting, and easy to manage. However, the process is sluggish compared to other deposition methods. In addition, it is not yet possible to use the PLD for vast area deposition. Because of the high cost and large deposition volume of laser equipment for film preparation, the PLD is best suited for areas like optical technology, sensor technology, microelectronics, and new material development. 57
The spin coating method
Spin coating is a thin-film coating technique that uses a spinning process to produce uniformly adherent films on flat surfaces. A spin coater is the name of the device used to spin a coat. Danglad-Flores et al. were among the first researchers to utilize and expand on this method. 58 The centrifugal force produced by the spinning object serves as the basis for how the spin coater functions. A small amount of the coating material is carefully placed onto the center of a flat substrate while either motionless or rotating at a plodding speed. The flat substrate then spins from a slow to a fast rate through the machine’s spinning action along with the material that has been applied. The high-speed spinning of the flat substrate caused the capillary action and centrifugal force to drag the viscous solution, forcing it to spread into an even film. All moisture evaporates during the spreading process, leaving the inhaled material on the glass surface as a thin film. Figure 8 shows the spin coating procedure can be broken down into five stages. Post-spin coating procedures such as drying and annealing may follow the deposition phase.
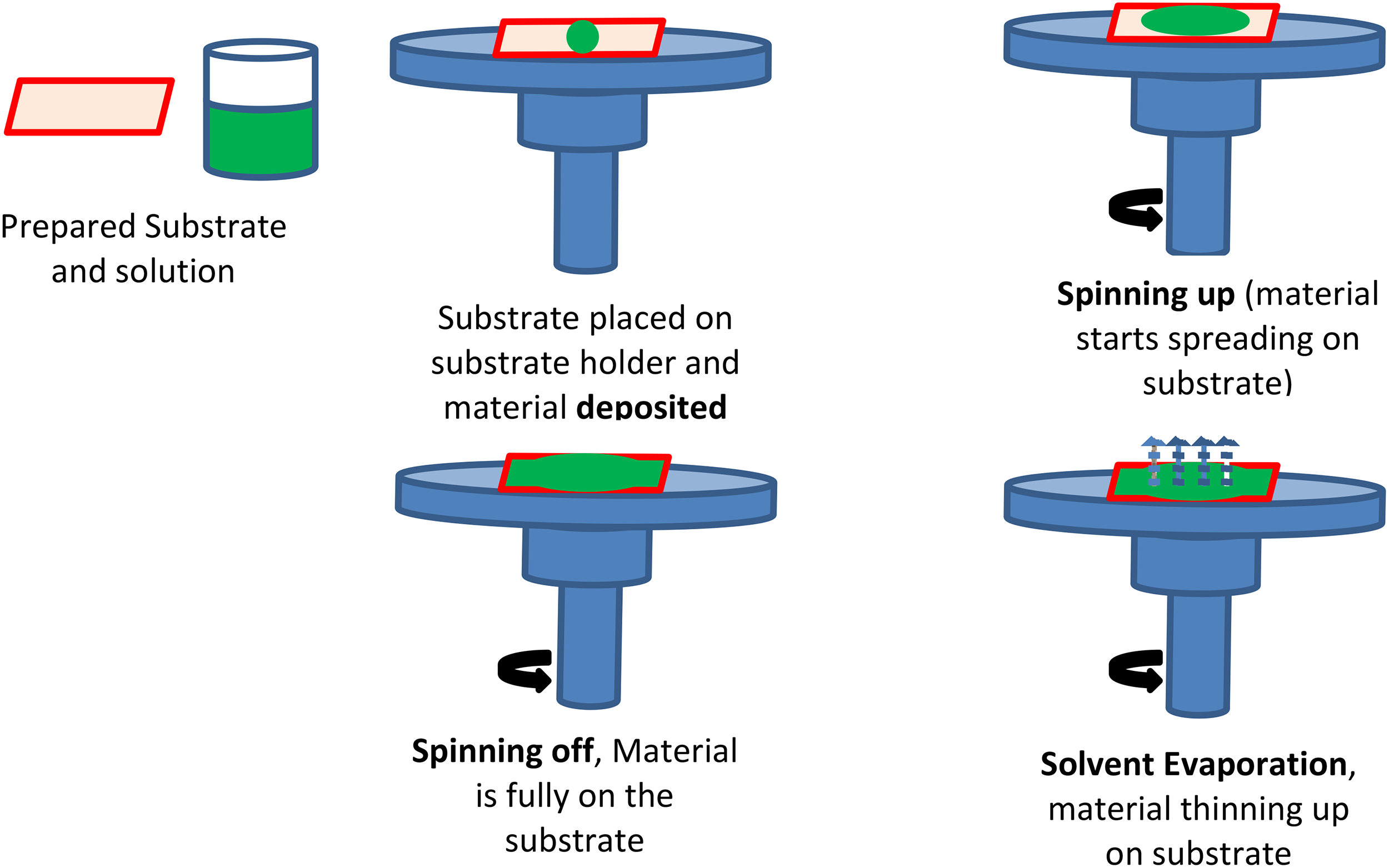
Principal stages of the spin coating process. 99
Van Zele et al. used the spin coating method to create thermochromic VO2 thin films. They analyzed the relationship between these thermochromic characteristics and coating thickness to create an ideal thermochromic film that balances a high degree of optical transparency and strong thermochromic switching capabilities. 59 The definition of IR switching was determined to be the difference in transmittance between 25°C and 100°C, taken at wavelength λ = 2000 nm, as shown in equation (1)
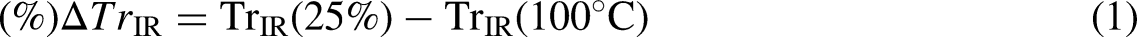
Equation (2) was used to evaluate the rate of solar modulation (Tmsol)
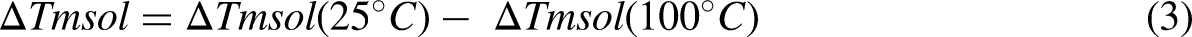


where
Other VO2 thin-film fabrication techniques
One most common VO2 deposition methods are the magnetron sputtering method. With magnetron sputtering, a gaseous plasma is created and contained within the area where the substance to be deposited—known as the “target”—is located. Highly energized ions hit down the target's surface in the plasma, and the released atoms pass through the vacuum conditions and deposit onto a substrate to form a thin layer. The earliest magnetron-sputtering experiments were conducted in the late 1930s, while the term magnetron first appeared in print in 1921. 66 There are numerous source configurations and control options for magnetron sputtering. Successful magnetron sputter deposition methods investigated recently include high-power impulse magnetron sputtering (HiPIMS), pulsed direct current (DC) sputtering, reactive gas sputtering, radio frequency (RF) magnetron sputtering, DC magnetron sputtering, and facing target sputtering. DC sputtering and RF magnetron sputtering are the most often used in the deposition of VO2 thin films. 67 Some innovations have been made to improve these popular methods by making them dual ion assisted or RF-inverted cylindrical magnetron sputtering, producing high-quality VO2 thin films. 103 Recently research interest has been picked in using the HiPIMS sputtering technique to deposit VO2 thin films.68,104 In reality, the production of high-quality VO2 films requires high temperatures exceeding 400°C, thereby restricting their use on a broad scale and their compatibility with substrates that are sensitive to temperature. Due to the increased ionization level, high plasma density, and consequently high ion flux of the HiPIMS technique toward the substrate, high crystalline thermochromic VO2 thin films can be deposited at lower temperatures. 69
Another approach for synthesizing crystalline compounds from a hot aqueous solution at high vapor pressure is the hydrothermal method. In this method, crystals are formed in an autoclave that contains both materials and water. Since the starting materials for hydrothermal synthesis are high-purity precursors, the powdered materials have higher chemical purity. 70 Gao et al. used hydrothermal synthesis to prepare well-crystallized, highly oriented, and free-standing VO2 (B) thin films. 71 The thermal properties of the produced VO2 (B) thin films were examined to increase their suitability for Li-ion batteries. Another work on hydrothermal synthesis of VO2 thin films was carried out by Xygkis et al. In the experiment, a set quantity of VO2 grains was distributed in differing amounts of polyvinylpyrrolidone, resulting in hybrid materials with different VO2/polyvinylpyrrolidone concentrations when dropped on amalgamated silica glass substrates. Varying the molar ratio of VO2/polyvinylpyrrolidone influenced a shift in the films’ critical transition temperatures, affirming its suitability for energy-saving surfaces. 105
The ratio of the reducing agents is critical in hydrothermal synthesis as it influences the outcome morphology of the thin films. Considering the film's high sensitivity to temperature changes, the hydrothermal synthesis reaction is seemingly governed by thermodynamics and agitation. 72 This technique allows high-vapor pressure materials to grow near or at the material's melting point. 106 Crystals of high quality can be synthesized using this method. Its simplicity makes it an excellent candidate for industrial application.
Photosensitive sol–gel is another innovation recently introduced by Wu et al. to fabricate VO2 thin films at a microscale. The new microfabrication method involved the exposition of coated films’ solution prepared from photosensitive precursors to ultraviolet irradiation, as shown in Figure 9. Then after, the patterned films are heat treated in the presence of nitrogen at 550°C for an hour. It is a prospective substitute technique for VO2 thin-film-based optics, electronics, and magnetic devices. It has the benefit of producing a variety of pleasing patterns, is inexpensive, and is straightforward. 73 Also successfully used in the deposition of VO2 thin films are methods such as electron beam deposition,74,107 multilevel absorption, 75 thermal oxidation,76,108 and variable emittance. 109
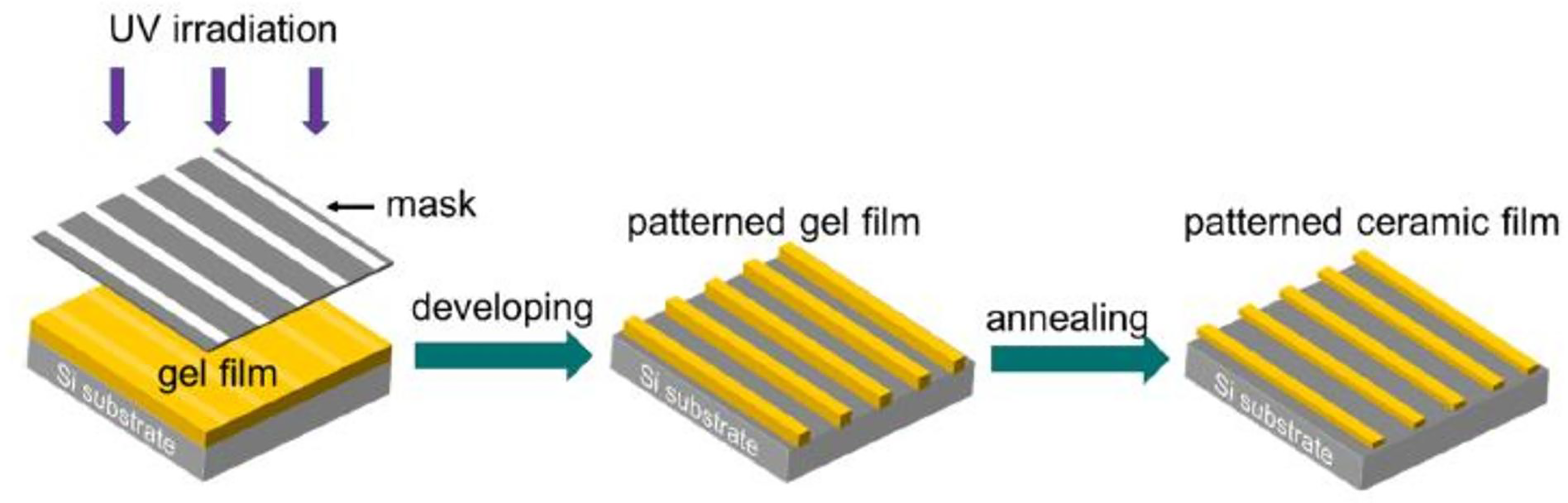
Fabrication of patterned-vanadium dioxide (VO2) thin films using the photosensitive sol–gel method. 73
By way of electrochemical or photochemical electron transfer reactions through optical excitation of electron–hole pairs, vanadium can be changed from one valence metal state to another. For this reason, it can now address a range of construction needs, including store window displays, tomographic screens, conversion of solar energy, and concealment. 110
An oblique angle deposition technique has also been developed to create VO2 thin films with different porosities and microstructures, referred to as sculptured thin films. Porosity and microstructure-controlled thin films with increased optical constants and unique properties are helpful in several applications, such as wrinkled filters, sensors, and phase retarders. 25 Sun et al. prepared VO2 sculptured thin film by reducing V2O5 through thermal evaporation and annealing. In examining how oblique incidence angles affect its potential for application, they found that VO2-sculptured thin films are more practical for use as smart windows than uniform VO2 films. 111
Possible application of VO2 thin films
Thin films of VO2 and many of its composites have found application in microelectronics, solar cells, self-cleaning, temperature control, switches, memory devices, actuators, and smart windows manufacturing. Monoclinic VO2 has received much attention for applications not only as smart solar control coatings, with the potential to reduce the need for heating and air conditioning loads within building infrastructure, but also as self-cleaning surfaces. Its phase transition characteristics also involve changes in electrical conductivity. The monoclinic phase semiconductors with a distinct band gap (∼0.7 eV), and the rutile phase shows semi-metallic properties. These characteristic features make VO2 a unique material for thermochromic applications. 112 VO2 thermochromic coatings on composite structures have been successfully applied as antifogging, energy-saving, and self-cleaning coatings on buildings, window glass, and car bodies. 113 These multifunctional characteristics can be integrated into the different layers of the film and make it applicable to headway communication, defense, and space networks. 114 The thermochromic property of VO2 has also made it suitable for thermoregulating applications such as heat sensors and s solar energy control surfaces. These properties applied on textile surfaces have shown better durability, great super-hydrophobicity, and self-cleaning. 115 The ability of VO2 thin films to modify solar energy helps to optimize the optical performance of other self-cleaning catalysts such as TiO2, giving it the capability of withstanding extreme weather changes and multifunction advantages in self-cleaning, especially the TiO2–VO2 composites. 116 Another exciting application of thermochromic VO2 property is in the antibacterial enhancement of paints when double-doped with ZnO and WO3. Prommin and his collaborators demonstrated that white color paint with ZnO and WO3 co-doped VO2 nano-pigment exhibited self-cleaning properties and almost a 100% antibacterial success under UV irradiation. 117
The reversible photochromic property of VO2 enhances the hydrophilic and photocatalytic properties of surfaces, making it suitable for developing environmentally friendly smart windows. Though no literature is currently available, the photochromic VO2 property can be used in designing and regulating the coloration degree of surfaces exposed to sunlight. 4
Flexible VO2 films could be used in contemporary, flexible electronics because of their unique phase-transition property. However, flexible substrates must be used to generate flexible VO2 films for practical applications. To fabricate thin films of VO2 that are flexible, Kim et al. adopted a transfer method whereby graphene was produced on Copper foils to support already created radiofrequency magnetron-sputtered VO2 films. Copper sheets were then etched, and a graphene-VO2 hybrid was applied to the polyethylene terephthalate film. The resulting flexible VO2 films bonded to the glass using appropriate adhesives. The strategy was innovative but complex, raising production costs. 118 Figure 10(a) shows the detailed steps of the transfer technique and (b) the anatomy of the produced graphene-reinforced VO2 film on polyethylene terephthalate.
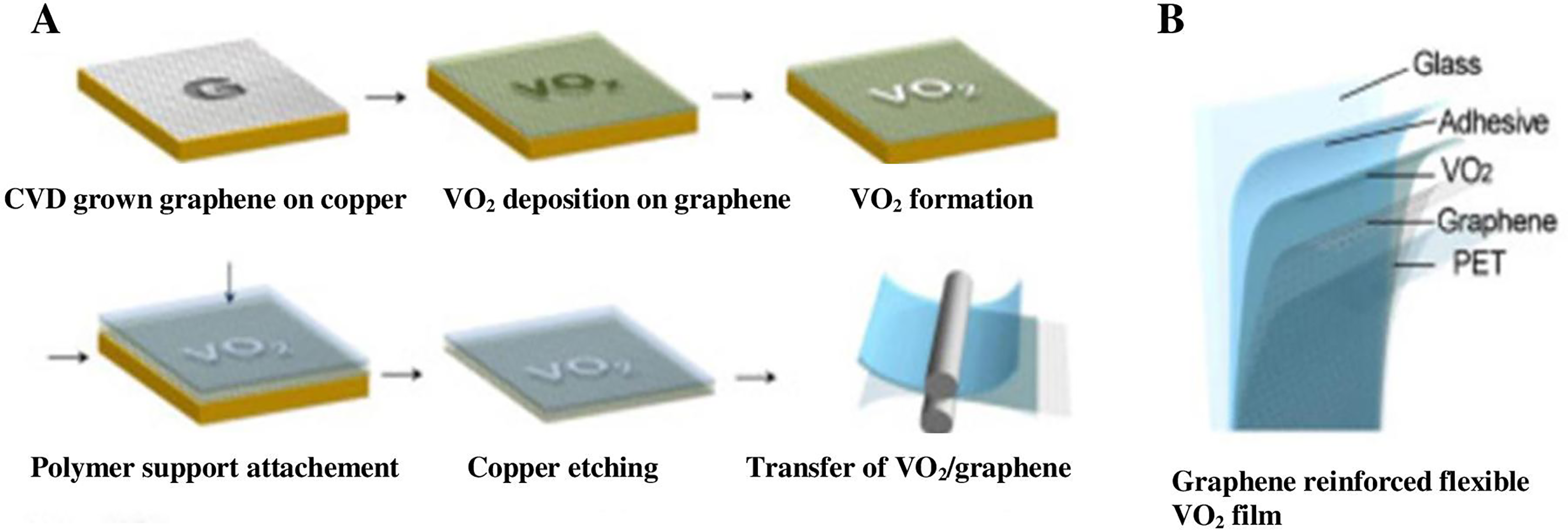
(a) Steps in the creation of graphene-reinforced flexible vanadium dioxide (VO2) film and (b) different layers of the graphene-reinforced VO2 coating on polyethylene terephthalate substrate adapted from Cao et al. 119
VO2 has also been investigated for metasurface device applications such as biosensing, 120 planar lenses, 121 absorbers surfaces,122,123 radiative thermal rectifiers, 124 holograms, 125 wave plates, 126 and more. In the past, adjustable metasurfaces were made to control only a single variable, such as phase or amplitude. However, by adjusting the device temperature, it is now possible to create devices that can change both amplitude and phase at a specific wavelength using VO2
Thermochromic property of VO2 and self-cleaning
Thermochromic coatings based on VO2 have been studied extensively for over four decades, with today being a pivotal time for VO2 applications. 24 Changes from the monoclinic phase at low temperatures to the rutile phase at higher temperatures result in structural modifications. In contrast to the monoclinic phase, which is semiconducting and transmissive, the rutile is metallic and reflective in the infrared spectrum. The optical characteristics of the VO2 in the visible part of the spectrum remain unchanged in both the rutile and monoclinic phases. 127 Lowering the transition temperature for VO2from 68°C to more closely to ambient temperature makes it suitable as a self-cleaning coating. 128 The drop in transition temperature can be achieved by doping the VO2lattice with metal ions. It has been established that at the low transition temperature, the absorption band edge of VO2 films shifts into the UV region of the spectrum, leaving films transparent in the visible region and improving solar absorption and photocatalytic activity. 129 Based on its exceptional thermochromism, self-cleaning characteristics and photocatalytic features are combined into VO2 material to degrade pollutants in the presence of UV irradiation. 20
For thermochromic VO2to be effective in self-cleaning coatings, issues of transition efficiency maximization, hysteresis reduction, and infrared reflectivity must be considered appropriately. 130 The Ti4+ ion could dope thermochromic VO2 because it has a comparable ionic radius and electronic structure to the V4+ ion. 131 In awareness of this characteristic, Chen et al. investigated the effect of Ti doping on VO2 polymer composites by evaluating the optical characteristics of films made of pure VO2 and films with various Ti doping concentrations. Their analysis showed that flexible VO2 films with Ti-doping had better optical elements desired for good self-cleaning functioning, as shown in Figure 11(a) to (d). 132 The considerably smaller particle sizes were thought to be responsible for improving the optical characteristics of doped VO2 films.
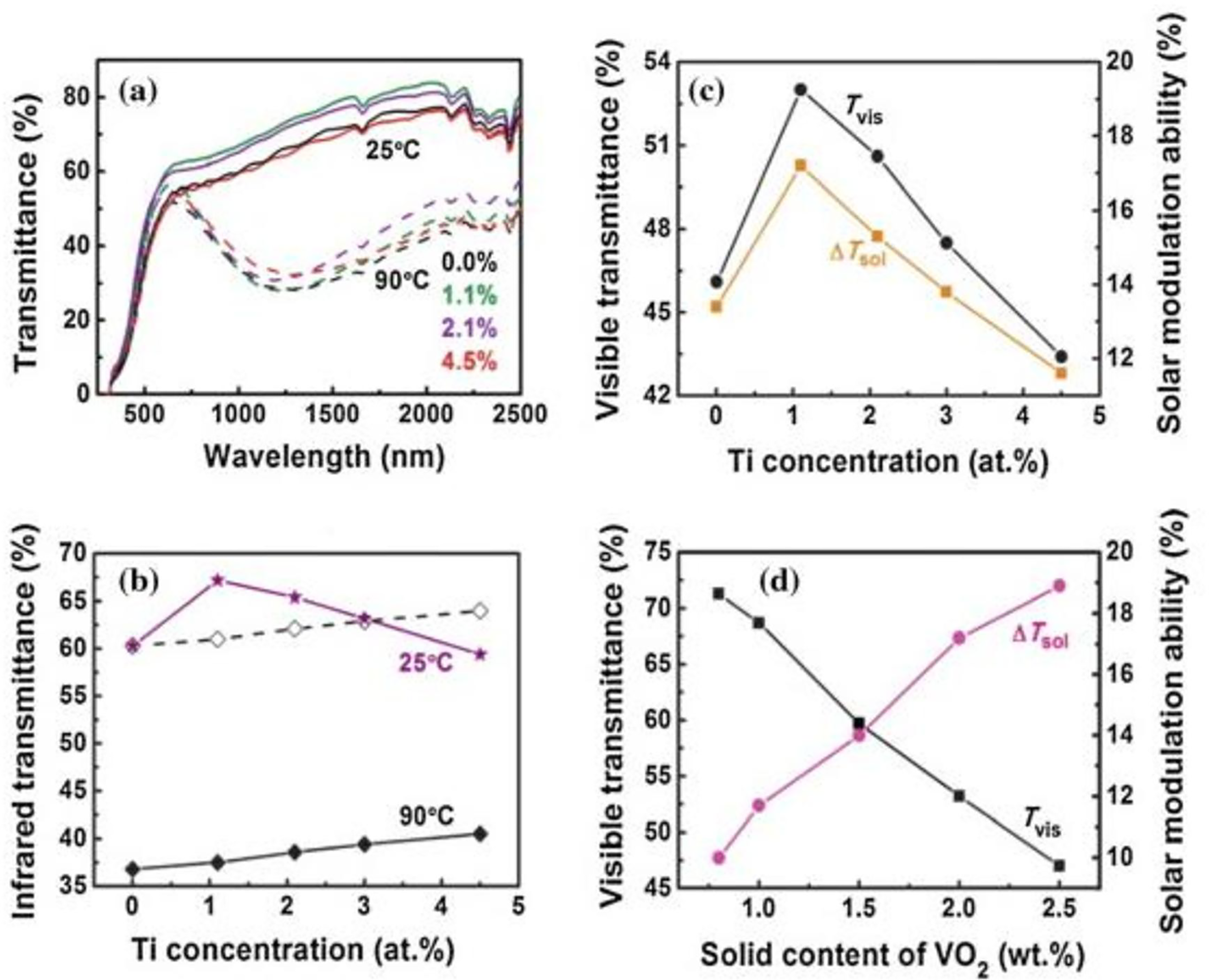
The differences in optical characteristics of pure vanadium dioxide (VO2) and Ti-doped VO2 films. (a) The plot of transmittance against wavelength for 25°C and 90°C deposition conditions, (b) Transmittance of films near-infrared spectrum. (c) The film's ability to transmit and modulate visible light. (d) The 1.1% Ti-doped VO2 film's ability to modulate the sun’s light and its ability to send visible light at different VO2 concentrations. 132
Thermochromic VO2 films are desired in self-cleaning applications, but equity between their luminous transmittance and solar modulation needs to be achieved. In self-cleaning, thin films with high luminous transmittance are preferred because they allow enough solar rays to reach the solar cells beneath the coatings, thus leading to increased power conversion efficiency.VO2 alone has not been proven to deliver this desired characteristic. About 7%, 50%, and 43% of solar energy are included in the solar spectrum's ultraviolet, visible, and infrared portions, correspondingly. 133 Increasing the disparity between the semiconducting and metallic states in the visible light spectrum makes it possible to significantly boost the corresponding solar modulation ability of VO2-based self-cleaning coatings. This indicates that the luminous transmittance of the VO2 smart layer in its semiconducting form should be higher than that in the visible light area, which should be maintained at least 50%. According to several studies, combining VO2's solar transmittance with specific materials produces strong reflection in the visible light area at various temperatures. 134 Utilizing SiO2, TiO2, and ZrO2, among other materials, enhanced VO2 luminous transmittance. 135 The optimized VO2/TiO2 structure showed a tremendous improvement in the luminous transmittance of VO2, making it appropriate for use in self-cleaning systems. These composite films demonstrated self-cleaning qualities with a low contact angle and breakdown of organic pollutants due to crystallized anatase TiO2. 116
Optimization is needed on the refractive index (n) and film thickness (d) for a maximum integrated luminous transmittance. A study by Xu et al. showed that the superior value of n = 2.2 is obtained at a thickness of 50 nm for VO2. 136 Before a straightforward and economical solution can be created, more studies will be needed to determine how well VO2-based self-cleaning ability can balance their luminous transmittances and solar modulation capacity. The best structure at the moment, taking advantage of the thermochromic property of VO2, striking a parity amid its solar modulation and luminous transmittance, and simultaneously delivering good self-cleaning characteristics is the proposed Cr2O3/VO2/SiO2 by Chang et al. 20 The bottom Cr2O3 layer improves VO2 crystallinity and the luminous transmittance of the structure. The top SiO2 layer enhances resistance against environmental hazards, serves as a reflection-reducing layer, and provides an excellent self-cleaning capability. To prevent VO2 from environmental deterioration when used as self-cleaning overlays, some chemically resilient oxide coatings, like WO3, Al2O3, and CeO2, were explored but posed a challenge whereby some of these substances have the propensity to impair VO2's optical characteristics.137,138
Photochromic property of VO2 and self-cleaning
Light-induced reversible color shift is known as photochromism. The photochromism of VO2 is equally an extensive area of research interest. It is expressed by VO2s’ ability to display quick coloring and bleaching capabilities under sunlight or UV irradiation. It is induced by modifying the valency of vanadium by photolysis or an electrochemical electron transport reaction that occurs when a light-excited electron–hole pair is present. Once stimulated by light, VO2 automatically starts modulating the transmittance of light in the natural environment. 139 Poor transmittance may be due to high absorption or high reflection of the solar rays. It is beneficial for self-cleaning due to increased absorption in the overlay films since high solar absorption leads to more photocatalytic activity.
Photochrominisn of VO2 in isolation has not been openly reported to contribute enormously to the self-cleaning of surfaces because single-phase VO2 photocatalysts are uncommon. 140 Minimal research has recently been conducted to investigate VO2's ability to do self-cleaning.27,141 It has been shown to degrade methylene blue, toluidine blue O, RhB aqueous solution, and naproxen. Figure 12(a) to (d) shows the self-cleaning ability of VO2 of different thicknesses using MB as a pollutant. The graph clearly shows that the degradation rate was fastest for VO2 film of 300 nm thickness. The photochromic property of VO2 produces photo-generated holes that aid in creating oxygen vacancies, which adsorb water molecules and encourage hydrophilic action, which photocatalytically destroys organic pollutants. The electron–hole pairs are consumed when the breakdown is complete, resulting in more oxygen vacancies. This is because good photochromic VO2 in high-intensity light permits visible light transmission through coatings of lower thickness. 142 Shinen et al. reported that thin films with a high thickness absorb more light reducing the film’s transmittance due to increased surface roughness. This led to increased absorbance and self-cleaning activities. 143 Work to improve photochromism and self-cleaning of VO2 by combining it with other materials is already promising. 144
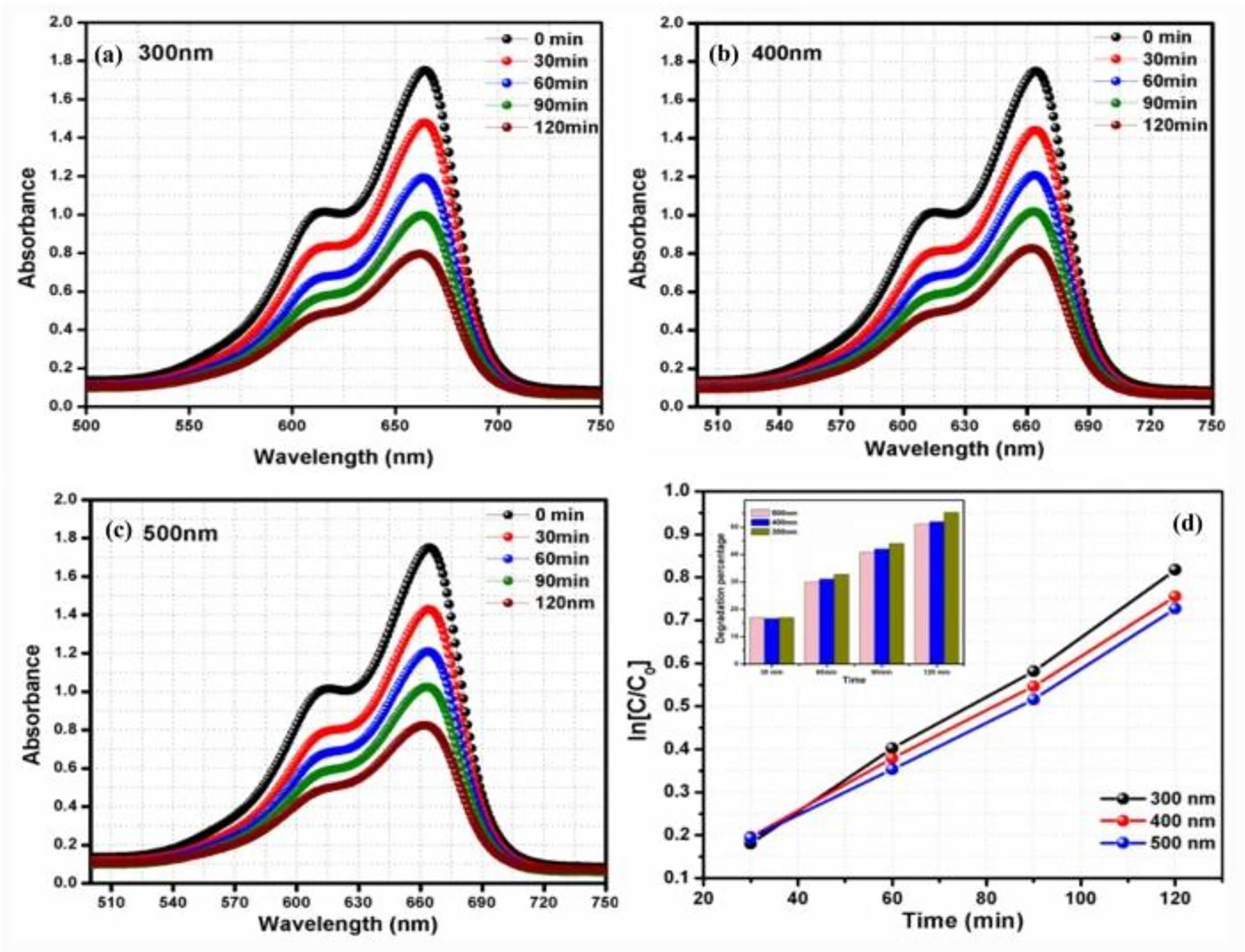
Photocatalytic degradation of methylene blue for different thicknesses of vanadium dioxide (VO2) thin film; (a) 300nm, (b) 400nm, (c) 500 nm, and (d) rate constant for each thickness. Reproduced with permission from Zhang et al. 142
Challenges facing the application of VO2 as self-cleaning thin films and possible remedies
The semiconducting and metallic phases of VO2 exhibit strong absorption in the visible region, demonstrating high photocatalytic and self-cleaning potential. However, the VO2 films with the best solar modulation are translucent, limiting their practical application in energy-efficient casements. Due to the release of V-ion from VO2-rooted thermochromic coatings as they age, many crystalline VO2 nanoparticles on layers turn out to be hazardous. According to a study conducted by Wu et al., as VO2 nanoparticles age, more V ions are produced from them than from the original films, resulting in high bacterial toxicity. The degradation reaction leading to the release of V-ions that form toxic bacteria may be characterized in two steps, as illustrated in equations (6) and (7).
145


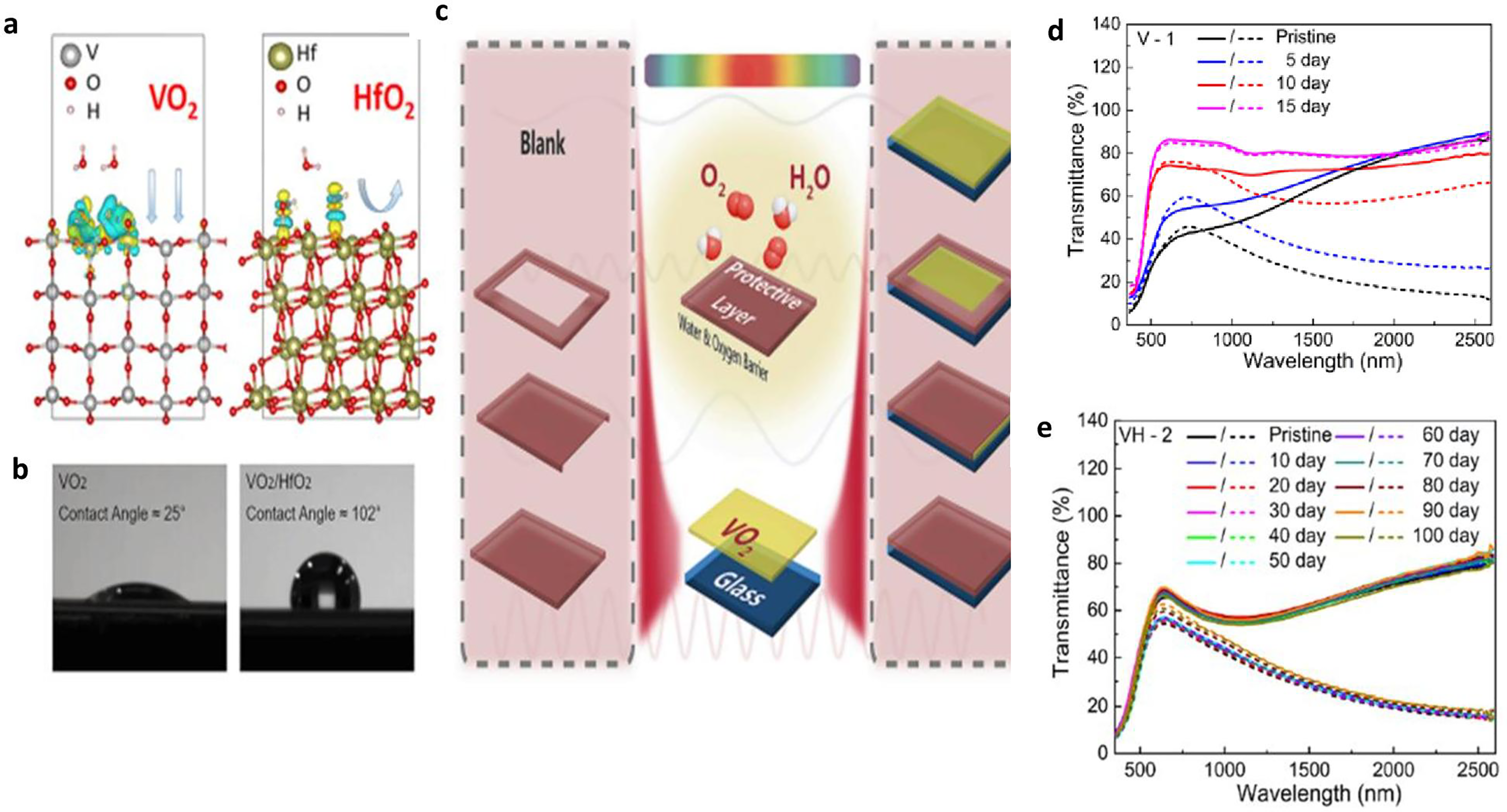
Influence of HfO2 layer on the deterioration of vanadium dioxide (VO2) films in the presence of moisture reproduced from Chang et al. 19 (a) Relationship of water molecules, VO2, and hafnium dioxide surfaces. (b) A single layer of VO2 that was hydrophilic changed to a hydrophobic surface when HfO2 was added. (c) The different samples of the composite film with HfO2 protection layer encapsulated. (d) The transmission spectrum of a plain VO2 sample reveals a quick decline in thermochromic ability. (e) The transmittance spectra of the VO2 sample with a complete HfO2 layer encapsulation exhibit strong stability.
Figure 13(b) shows that the VO2 surface exhibited significant hydrophilicity and wettability because the unsaturated surface oxygen in VO2 formed hydrogen bonds with the water hydrogen cations. Figures 13 (d) and (e) show the result of the deterioration process of HfO2 encapsulation. The relative solar modulation capabilities and optical variance at 2500 nm, which are crucial indices to indicate VO2's phase transition feature, were computed together with the periodic transmittance measurements to assess the stability of VO2 films. The life span of the fabricated VO2/HfO2 composite film was estimated using the Hallberg and Peck acceleration factor defined by equation (8).
147
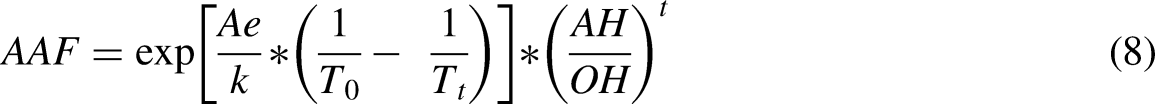
Depositing crystalline VO2 without a buffer layer necessitates soaring temperatures to achieve better lattice anatomy and stoichiometric on flexible substrates. Therefore, doping VO2 with buffer materials such as Cr2O3, ZnO, V2O3, and SnO2; can reduce the temperature at which VO2 films can be deposited. Hu et al. carried out such a study and, as shown in Figure 14(a) to (d), observed a distinct decrease in the width of temperature hysteresis at the highest Ti dopant concentration. 148
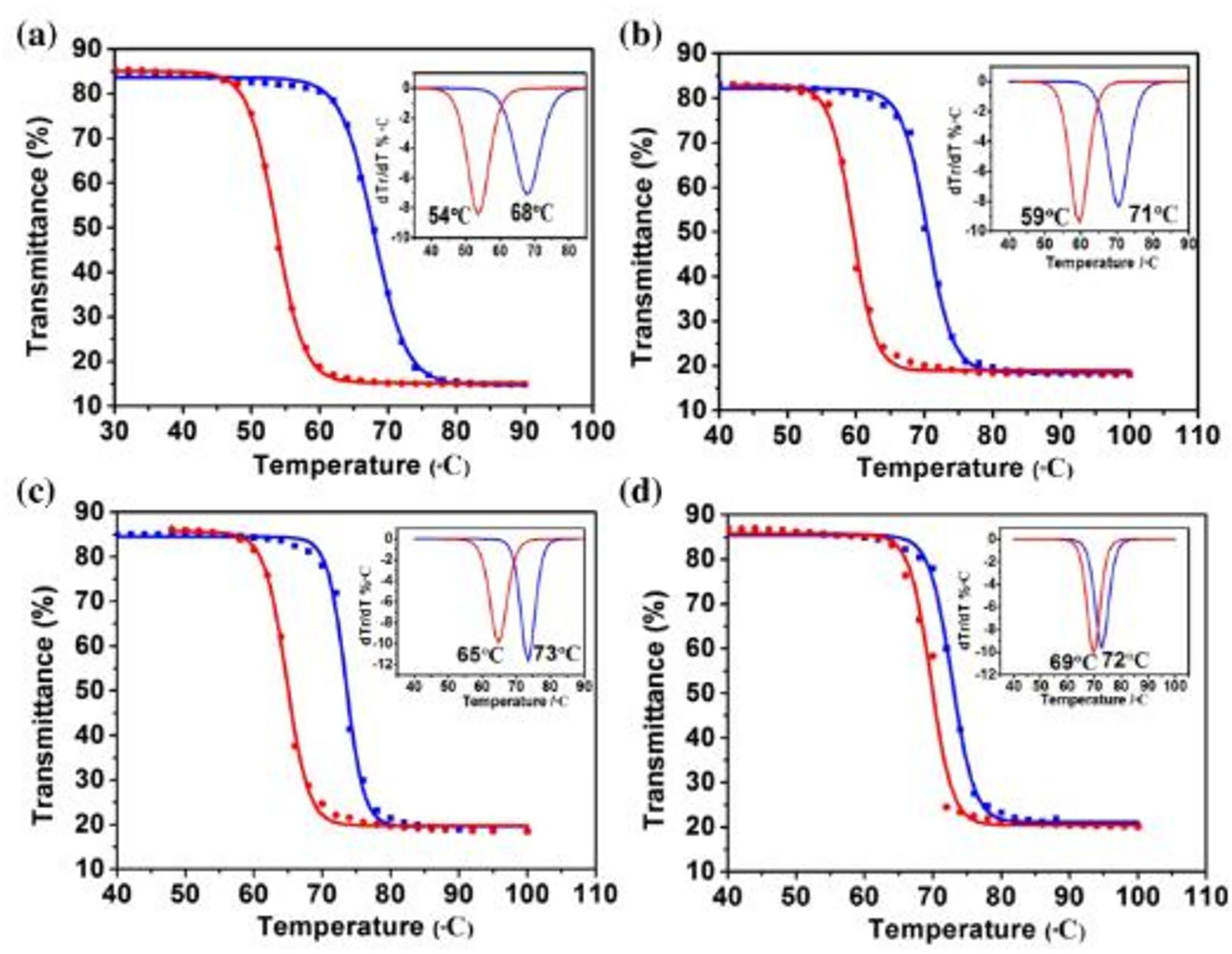
Hysteresis loops of vanadium dioxide (VO2) and Ti-doped VO2 transmittances versus temperature at a fixed wavelength for (a) pure VO2, then VO2 doped with (b) 1.2, (c) 2.0, and (d) 2.8 percentage concentrations of Ti, respectively. 132
The main challenges facing the application of VO2 thin films and proposed remedies to make them suitable for industrial applications in self-cleaning and smart windows are presented in Table 4.
Some challenges facing vanadium dioxide (VO2) thin-film application and proposed remedies.

Conclusion
This article has reviewed the different methods of fabricating VO2 thin films. The different transitional phases of VO2 and the different routes of obtaining one phase from the other were examined. Advances in the various techniques to fabricate VO2 thin films and the challenges facing each method were also reviewed, followed by suggestions that have been made to address those challenges. The article looked at the different sectors for possible application of the different phases of VO2 films based on the individual properties of each transitional phase. The review concludes with efforts and the feasibility of harnessing the thermochromic and photochromic characteristics of VO2 in better self-cleaning technology. It is observed that much research is needed to understand how the photochromism of VO2 can be harnessed for self-cleaning action. This research suggests that the stability issue may be solved by doping VO2 with other metal oxides and producing multilayered thin films with sandwich architectures made of various polymers and metal oxides. This will assist in preparing VO2 thin films and their composite for photocatalytic and self-cleaning surfaces.
Future research outlook
Most VO2 thin-film deposition techniques are only viable at the laboratory level. The chemical vapor deposition method is the only technology currently appropriate for industrial processing. However, it has a high operational cost. The creation of straightforward but effective scale-up techniques for synthesis and film deposition will be crucial in the future. The production of VO2 thin films on an industrial scale is also considered viable using a two-step process. Future studies should be concerned about how colors like yellow, brown, and deep blue will improve the self-cleaning viability of photochromic VO2. Although adding titanium, magnesium, and fluorine has improved its optical and self-cleaning qualities, there is still a need to develop methods of managing VO2.
Once exposed to moist acidic environments, VO2's thermochromic characteristics appear to deteriorate, making its commercial applications, like self-cleaning film, difficult. More research will be needed to get around this restriction.
There has not been much research done to prove how photochromism works or how useful it is for self-cleaning. More theoretical research should be conducted to comprehend further the connection between photocatalytic activities and VO2 color changes caused by visible light exposure.
Footnotes
Acknowledgments
The authors acknowledge support from the National Research Foundation of South Africa, The University Research Council of the University of Johannesburg, South Africa, and the Global Excellence Stature award.
Author’s contribution
All authors contributed to the study's conception and design. Mr Valantine Takwa Lukong performed the originator, data collection, and first draft. Preliminary review and editing was done by Dr Ukoba Kingsley and Prof. Tien-Chien Jen carried out final editing and source of funding. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
