Abstract
Water hyacinth is an invasive alien plant with several impacts on the environment, economy and society. The plant’s high degree of proliferation makes its mitigation difficult and sometimes complex. However, existing evidence suggests that water hyacinth is a sustainable substrate for biogas production. Using the pretreatment processes for the optimisation conditions for biogas production from water hyacinth, this study analysed the effects of moisture content (60%, 70% and 75%) on Trichoderma atroviride pretreatment of water hyacinth and the impact of the pretreatment on biogas production. Anaerobic digestion of the water hyacinth process was performed at 35°C for 35 days. The modified Gompertz model was used to analyse and predict the appropriate kinetic variables of the digestion process. Biogas yields from untreated, pretreated-60%, pretreated-70% and pretreated-75% were optimal at 135, 210, 217 and 223.4 mL/g of volatile solids (VS). These results suggest the pretreatment of water hyacinth enhanced the degradability of water hyacinth by breaking down the cell wall structure and facilitating its use by microorganisms. Furthermore, the results also confirmed that the higher the moisture content, the easier the biodegradation rate and, consequently, the higher the biogas yield. The model predicted maximum methane production potential ranging from 91.84 to 201 mL/g VS, and the maximum methane yield rate was within 10.12–15.12 mL/day. The lag phase varied between 2.46 and 6.94 days. The percentage error between experimental and model outcomes for untreated, pretreated-60%, pretreated-70% and pretreated-75% are 17.96%, 16.67%, 14.20% and 4.68%, respectively, while the coefficients of determination of the model varied between 0.905 and 0.975, demonstrating significant reliability on attained factors.
Introduction
The demand for cost-effective and sustainable mitigation measures of invasive alien species has increased in recent years due to the threats they pose to the pillars of sustainability, namely the environment, social development, and the economy. In South Africa, water hyacinth is listed as a Category 1 weed in the National Environmental Management Biodiversity Act (10/2004): Alien and Invasive Species Regulations, 2014, which demands its mitigation. Water hyacinth, scientifically referred to as Eichhornia crassipes, has recently gained attention due to its potential financial and environmental benefits in recovering its valuable resource. Ilo et al., 1 for example, observed that this aquatic plant has been utilised in phytoremediation, animal feed production, as biofertiliser, biopolymers and bioenergy. Bioenergy is an essential contributor to world energy security; it significantly reduces dependency on fossil fuels, thus, mitigating the devastating effects of greenhouse gas emissions. 2 The valorisation of water hyacinth for bioenergy production is considered promising because of its availability, high biomass yield and does not contend with arable land with food crops. 3
The conversion routes of E. crassipes into bioenergy are through a thermochemical process such as pyrolysis4,5 or gasification 6 and a biochemical process such as anaerobic digestion (AD). 7 While pyrolysis is deemed one of the most suitable and sustainable thermal conversion processes in transforming biomass to bioenergy because of its simple operation and low investment costs, 4 AD has also received attention because of its adaptation to a broad spectrum of feedstocks, nutrient recovery, and net energy output. 8 AD is the decomposition of organic materials by microorganisms in the absence of oxygen, producing methane-rich gas called biogas and improved digestate.9,10 The produced methane is converted for various energy needs; electricity, transportation fuel, cooking and heating. The potential of E. crassipes for AD has been reported in the literature.7,11 The process comprises four stages: hydrolysis, acidogenesis, acetogenesis and methanogenesis. 8 In most cases, hydrolysis is deemed the slowest or rate-limiting stage because of biomass's lignocellulosic content, which impedes microbial hydrolysis of the feedstock, thereby significantly limiting the large-scale biogas production.12,13
Although limited hydrolysis decreases the hydrolysates produced for further breakdown, the pretreatment process influences the hydrolytic stage. Pretreatment reduces the structural barrier of the lignocellulosic components, which primarily comprise three polymers bound by many inter-polymer interactions, namely lignin, cellulose and hemicellulose. It delignifies the biomass, increases the surface area for easier degradation by the microorganisms and decreases the rate of crystallinity and polymerisation. Amongst the pretreatment methods, biological pretreatment is considered a sustainable method for improving the biodegradability of lignocellulose materials.4,15 This is because its use is cost-effective, requires low energy and does not produce inhibitors. 16 Fungal pretreatment, particularly white-rot fungi, is mainly used in the pretreatment of lignocellulosic biomass for biogas production because they mineralise lignin. 17 Empirical evidence suggests a promising biogas yield after fungal pretreatment of lignocellulose biomass such as rice straw, corn stover and forest residues.18–21 Li et al. 19 disclosed the highest methane yield of 61.9% during the AD of fungal pretreatment of corn straw, while Mustafa et al. 21 reported a 120% and 78.3% increase in methane yield during P. ostreatus and T. ressei pretreatment of rice straw, compared to the untreated substrate.
Therefore, fungal pretreatment of E. crassipes is crucial for optimal biogas yield. Trichoderma atroviride, a white-rot fungus, is considered in this study because it is known as a producer of a complex array of cellulose-degrading enzymes such as β-glucosidase. 22 As much as fungal pretreatment is sustainable and cost-effective for optimal biogas yield, it is considered time-consuming. However, considerations such as moisture content (MC), temperature, nutrient supplements, and culture duration are imperative for fungal growth and metabolite formation and, afterwards, higher lignin degradation. Improving the fundamental parameters is vital to enhancing the techno-economic performance of the T. atroviride pretreatment of water hyacinth.
MCs contribute significantly during fungal pretreatment, affecting fungal growth and activities. 23 They increase the efficacy of the pretreatment and, subsequently, biogas yield.15,24,25 Enormous free water decreases the interparticle spaces and reduces the accessible oxygen for the fungal growth, 20 while a low MC deactivates the fungi. Lalak et al. 15 reported 65% MCs as optimal for hemicellulose and lignin removal during the Flammulina velutipes fungal pretreatment of tall wheat grass, Shi et al. 26 observed 75% MC as the optimal lignin degradation in cotton stalks using Phanerochaete chrysosporium pretreatment and Meehnian et al. 27 reported the optimal lignin degradation in cotton stalks using Daedalea flavida at 75% MC. Mutschlechner et al., 25 on the other hand, considered 70% as the optimal moisture level during a biological pretreatment of bio-waste with Trichoderma viride. However, according to Nadir et al., 23 optimal MC is dependent on the fungal strain and the substrate utilised. Thus, it is essential to determine the optimal MCs in T. atroviride fungal pretreatment in order to reduce the pretreatment time, degrade lignin and, therefore, disintegrate the complex lignocellulosic structure to increase biogas yield.
In this paper, the following are the objectives: (a) assess the effect of MC during pretreatment of water hyacinth with T. atroviride on the biodegradation process; (b) determine the effect of T. atroviride in improving the biogas and methane yield for greater productivity and cost-effectiveness; (c) investigate the experimental kinetics in the context of ‘goodness of fit’ with the modified Gompertz model. The novelty of the study is that no known studies, to the best of the authors’ knowledge, have investigated and systematically analysed the effect of T. atroviride on the AD of water hyacinth for optimal biogas yield and, more specifically, the MC. We argue that the kinetic constants obtained from these models can be used to explain the effects of T. atroviride pretreatment on biogas yield.
Materials and methods
Substrate and inoculum
Water hyacinth was collected from a dam located at Ixopo (30°8′53″S 30°4′35″E) in KwaZulu-Natal Province, South Africa. The stem and leaves of the plant were considered at the laboratory, while the roots were discarded because, according to the study of Xu, 28 the water hyacinth leaves and stems have high degradation rate than the root. The high fibre content of the root is considered to require more residence time during fungal pretreatment. The materials were cleaned and oven-dried at 105 °C for 24 hours to a constant weight. Subsequently, they were ground in a slow-speed granulator (GSL 180, Zerma). The dried water hyacinth particles were stored at ambient temperature for further use. Fresh cow dung, collected from KwaUmgeni Livestock (29°29′11″S 30°26′13″E), Pietermaritzburg, in KwaZulu-Natal Province of South Africa, was sieved through a 1 mm to remove particles and grit and diluted with deionised water (1:1, w/v) to form a slurry. The slurry, hereby called cow dung inoculum, was stored in a Duran 1 L bottle and kept in a water bath at 34 ± 2°C with a thermostat for 15 days for acclimation. The application of a long start-up phase is for the adaption of the microbial community to improve reaction rates. The volatile organic matter in the slurry is expected to be degraded. The substrate and inoculum were characterised and presented as illustrated in Table 1.
Preliminary characterisation of the substrate and inoculum.
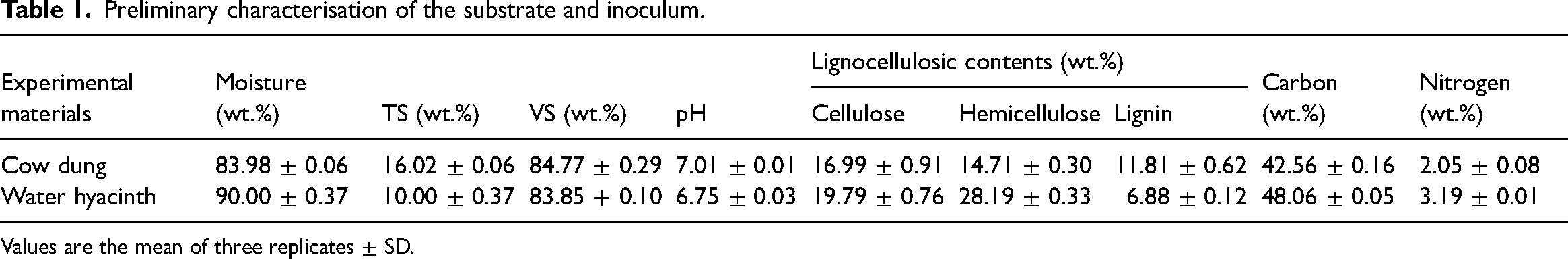
Values are the mean of three replicates ± SD.
Pretreatment process
Fungal strain and inoculation
White-rot fungus, T. atroviride (PPRI 4705), as shown in Figure 1, was procured from Agricultural Research Council (ARC), Pretoria, South Africa, on potato dextrose agar (PDA) plates. Inoculation cultures were activated by cutting out a 1 cm2 culture piece and placing it on PDA plates. The PDA plates were prepared with 5 g PDA (Sigma-Aldrich), 5 g bacteriological agar (BioLab), and 500 mL distilled water. The cultures were incubated for 7 days at 25°C. Subsequently, three plugs of active mycelia from the plates were homogenised with 70 mL of sterile deionised water containing 0.1% of Tween 80 (Sigma-Aldrich) and utilised as the fungal culture inoculum.
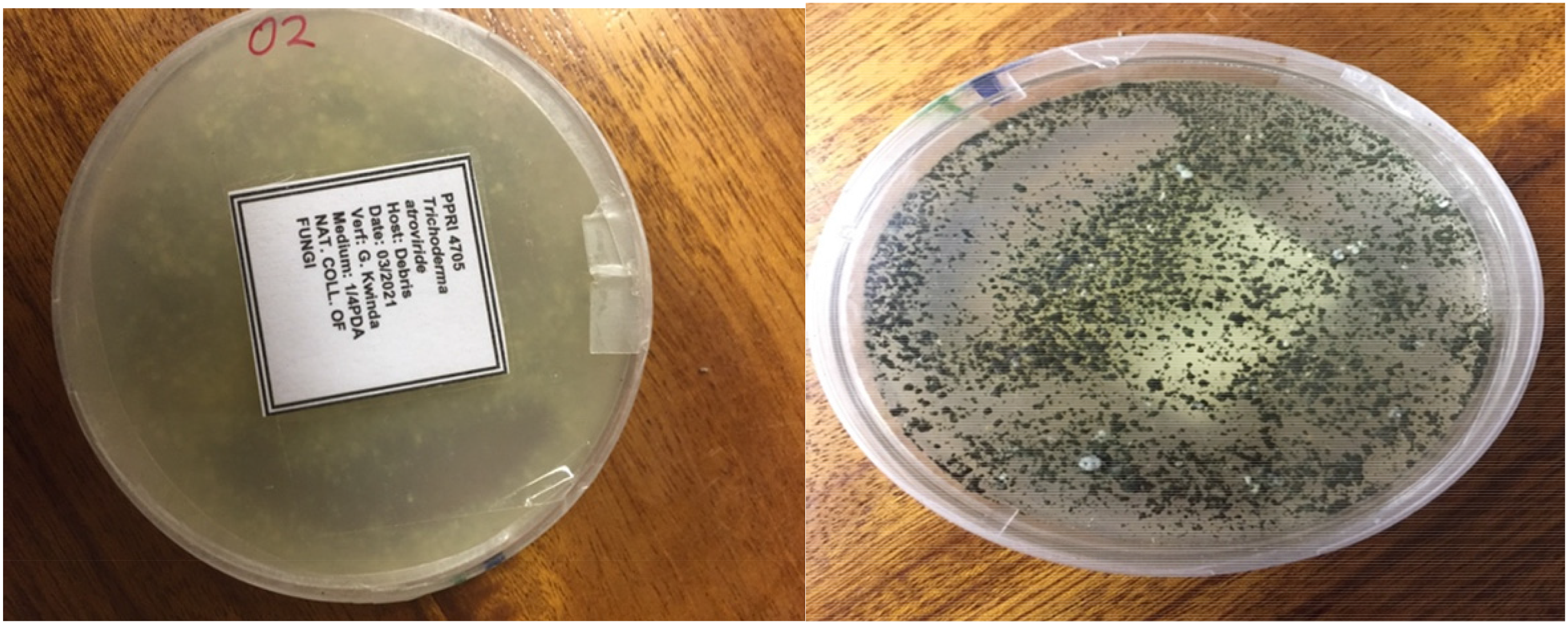
T. atroviride fungal strain.
Pretreatment of water hyacinth
Fungal pretreatment was achieved according to García-Torreiro et al. 29 and Mustafa et al. 21 All experiments were performed in triplicate for validation. Water hyacinth substrates of 16 g were put in nine 250 mL Erlenmeyer flasks. In order to ascertain the optimal MC for lignin degradation during fungal pretreatment of water hyacinth, a laboratory scale experiment at different MCs was used. The MCs ranges (60, 70 and 75% [w/v]) were chosen based on previous literature.15,25–27 The optimal humidity content was explored by varying the added sterilised deionised water to achieve a controlled environment, obtaining the desired MC. The flasks were covered with cotton stoppers and aluminium foil to reduce moisture loss and autoclaved (SLE-FA Series, Equitron) at 121°C for 15 minutes. The flasks were left to cool, and each flask was inoculated aseptically with 16 mL of the fungal culture inoculum and maintained statically in an incubator (IncoTherm; Labotec) at 30°C for 15 days. The choice of 15 days was based on previous studies as a moderately short incubation time considerably favours delignification.30,31 Three additional autoclaved flasks containing samples without fungal culture inoculum served as the control. The pretreated substrates were identified as P-60%, P-70% and P-75%.
Experimental setup
The batch test used Erlenmeyer flasks (1000 mL), as shown in Figure 2. Cow dung inoculum was added to three flasks containing pretreated water hyacinth (10 g) at 60%, 70% and 75% initial MCs to make an inoculum/water hyacinth ratio of 2.5 (v/w) according to Syaichurrozi et al., 32 and Syaichurrozi. 33 The samples were coded with unique identification numbers as P-60%, P-70% and P-75%. A flask contained untreated water hyacinth (10 g) with inoculum (25 g), and another had only inoculum (25 g), which served as a control experiment to establish the background gas productivity. Each flask was set up in triplicate and filled to a working volume of 700 mL using distilled water. The headspace of each flask was flushed with nitrogen gas (N2) for 5 minutes to maintain an anaerobic environment before they were sealed and placed in a thermostatically controlled water bath (35°C). The water bath was lined with insulating material to prevent excessive heat losses and create a dark environment for the flasks. The dark environment was to avert photosynthetic organisms’ growth. Also, AD in dark environments is considered more effective than in light environments. According to Hajji et al., 34 unlighted digesters generate high biogas and methane yield compared to lighted digesters. Each flask was fixed to a eudiometer (300 mL capacity) containing a confining liquid and connected to a 1000- mL collection bottle (Duran). The confining liquid [30 mL of sulphuric acid (H2SO4), distilled water, 200 g of sodium sulphate decahydrate (Na2SO4·10H20), few drops of methyl orange solution] was prepared according to Mursecet al., 35 Hughes et al. 36 to avert the solubility of carbon dioxide (CO2). The produced biogas displaced the confining liquid from the eudiometer; the volume of liquid displaced is equal to the volume of biogas produced. 37 The quantity of the confining liquid is regulated after each reading. Mixing was done manually twice daily to avert the surface crust's development and ensure the microorganisms and substrate homogeneity. The test was continued until the methane production rate became low.

Experimental setup.
Analytical parameters
The physicochemical properties of the substrates before and after digestion were determined. Total solids (TS) and volatile solids (VS) were determined according to Standard Methods.
38
Lignocellulose contents of the substrate and inoculum were determined using the Chesson method as described by Wardani et al.
39
The pH of the sludge was measured with a microprocessor pH metre (pH 212, Hanna Instruments), while the electrical conductivity (EC) was measured with a conductivity metre (ES 215, Hanna Instruments). The carbon (C) and nitrogen (N) elements were established with Flash2000 Organic Elemental Analyser (ThermoFisher Scientific). The volume of the biogas from each flask was measured every 24 hours using the liquid displacement method. The biogas yields were attuned to standard conditions (0°C, 1 atm) using Equation (1)
1
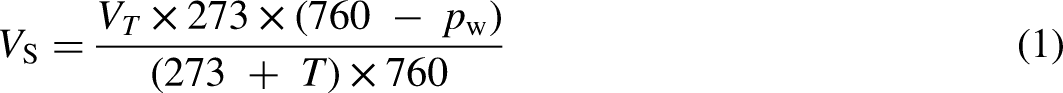
Kinetic and statistical study
The assessment of parameters required for optimal biogas yield was estimated using the Modified Gompertz equation.40,41 As presented in Equation (2), the equation assumes that the microbial activity in the anaerobic digesters is directly related to the biogas production rate.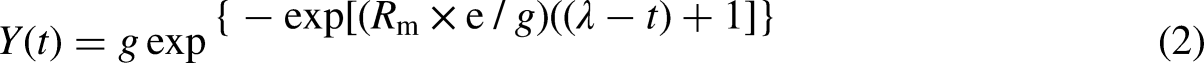
Results and discussion
Substrate and inoculum characteristics
The rate and composition of biogas yield depend on the substrate's characteristics. Therefore, the composition, such as moisture, TS, VS, pH, lignocellulosic, carbon and nitrogen contents of water hyacinth before pretreatment and cow dung, were examined (Table 1). The MCs of water hyacinth and cow dung were 90 wt.% and 84 wt.%, respectively. The outcome is comparable to Barua et al., 42 which reported the MC of water hyacinth and cow dung inoculum as 90% and 80%, respectively. The TS and VS of water hyacinth are 11.02 wt.% and 85.91 wt.%, while cow dung is 17.22 and 84.77 wt.%. The high VS of water hyacinth signifies its suitability for the AD process. Mathew et al. 43 observed a slightly similar trend and reported TS and VS of 11.4 wt.% and 86.2 wt.% for water hyacinth and 18.9 wt.% and 74.6 wt.% for cow dung, respectively. The pH of a feedstock is crucial in achieving process balance and adequate biogas yield; low pH hinders the metabolism of the methanogenic community and, subsequently, methane yield, while a high pH causes imbalance as a result of the expeditious change of ammonia nitrogen to free nitrogen. The pH values of water hyacinth and inoculum were within the reported optimal pH range of 6.8–7.4. 44
The lignocellulosic content of water hyacinth comprised 19.79 wt.% cellulose, 28.19 wt.% hemicelluloses and 6.88 wt.% lignin, which is somewhat different to the outcome of Varanasi et al. 45 who reported 24.8 wt.% of cellulose, 30 wt.% of hemicellulose and 5.6 wt.% of lignin of water hyacinth. Cellulose and hemicellulose are the key carbon source of anaerobic microorganisms 15 ; therefore, water hyacinth with 47.98% of holocellulose (cellulose and hemicellulose) indicates its potential for methane production. However, lignin inhibits the breakdown of the holocellulose content to monomeric sugars, which are needed to convert substrates effectively to biofuels. The lignin content of 6.88 wt.% implies a possibility of inhibition during the biodegradation process. The carbon and nitrogen contents of 48.06 wt.% and 3.19 wt.%, respectively, are similar to the 46.52 wt.% and 3.0 wt.% reported by Hu et al. 46 and different to 31.5 wt.% and 2.80 wt.% described by Varanasi et al. 45 The cow dung’s 16.99 wt.% cellulose, 14.71 wt.% hemicelluloses, 42.56 wt.% carbon and 2.05 wt.% nitrogen contents are similar to the 15.31 wt.% cellulose, 14.05 wt.% hemicellulose, 31.43 wt.% carbon and 2.21 wt.% nitrogen specified by Li et al. 47
The difference in this study’s substrate and inoculum characteristics and other studies is ascribed to the different geographical sites and experimental settings. These physicochemical compositions create a reference point for the suitability of biomass conversion. According to Nwokolo et al., 48 the compositions influence the degradation kinetics of the substrate, biogas yield and methane content. The characterisation, therefore, identified the recalcitrance nature of water hyacinth and the need for pretreatment. Nevertheless, the results from this study signify that water hyacinth is appropriate for AD and has a high biogas yield potential.
Effect of different moisture contents on the biodegradation process
The influent and effluent characteristics of the AD of pretreated and untreated E. crassipes are presented in Table 2.
Influent and effluent sludge characteristics.
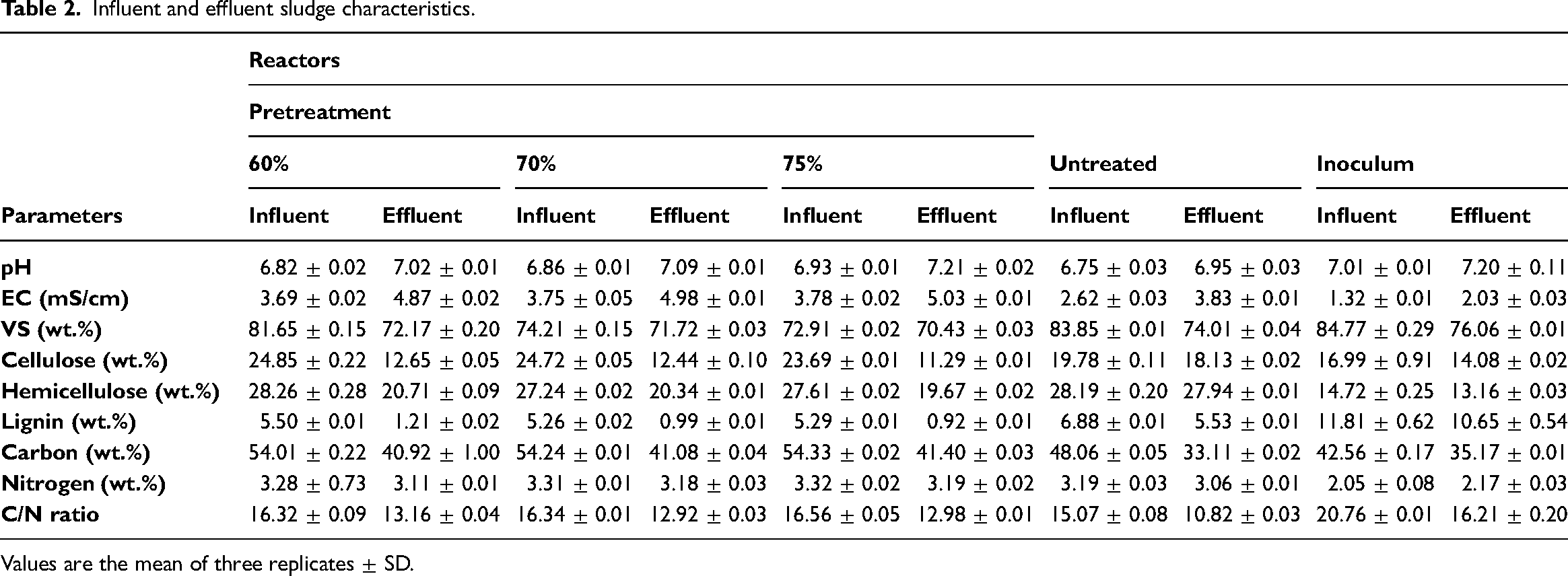
Values are the mean of three replicates ± SD.
AD involves reactions executed by various microorganisms; hydrolysis, acidogenesis, acetogenesis and methanogenesis. 49 As a result of these microbial consortia and their enzymatic activities, pH is considered to have an essential impact on the digestion process.49,50 Ali and Sun 14 investigated the influence of physicochemical and fungal pretreatment on park wastes for biogas production under mesophilic conditions. They reported an increase in the pH of untreated (7.1–7.5) and pretreated (7.5–7.8) substrates. The present study indicated the pH of the influent for P-60%, P-70% and P-75% as 6.82, 6.86 and 6.93, respectively, while untreated and inoculum were 6.75 and 7.02 (Table 2). However, at the end of the fermentation process, a slight increase in pH was noted at 7.02, 7.09 and 7.21 for P-60%, P-70% and P-75%, while untreated and inoculum were 6.95 and 7.20, respectively. The increase in pH indicates steady biogas generation, as postulated by Alfa et al. 51 It also signifies the end of volatile fatty acids’ (VFAs’) metabolism. Change in pH is dependent on the progress of the substrate's degradation; the generation of VFAs during acidogenesis reduces pH. Over time, the VFA is degraded into hydrogen and acetate. The acetate is transformed into methane during methanogenesis.
EC, which is described as the ability of a solution to conduct electrical current, is in direct proportion to ion concentration. 52 This parameter has been used for AD process monitoring; Robles et al. 52 measured the methane production using the EC, while Aceves-Lara et al. 53 used EC for measuring VFA, alkalinity and bicarbonate concentrations during the anaerobic fermentation process. In this study, an increase in EC was observed; while the EC for P-60%, P-70% and P-75% increased from 3.69, 3.75 and 3.78 to 4.87, 4.98 and 5.03 mS/cm, the untreated substrate and cow dung inoculum increased slightly from 2.62 to 3.83 and 1.32 to 2.03 mS/cm, respectively (Table 2). This increase was expected due to the rise in the pH of the effluents, which implies the development of bicarbonates. Furthermore, the high MC caused a higher degree of dilution. Similarly, Kassongo et al. 54 observed a 25% increase in EC in AD of unamended grape marc and cheese whey under mesophilic temperature and Ali and Sun 14 reported the EC of the untreated feedstocks to vary from 2.6 to 3.7 dS/m, while pretreated substrate increased to 4.6 dS/m at the end of the digestion. The result suggests that T. atroviride introduced ion concentrations and improved nutrient accessibility at good MCs. Achieving increased conductivity is a substantial economic and environmental reflection as the methane produced is sustainable, and no external materials are added. 54
The lignocellulosic content of the influent and effluent sludge is presented in Table 2, and it can be noted that all of their component masses decreased. The untreated sludge decreased by 8.34, 0.87 and 19.62%, while the inoculum decreased by 17.13, 10.60 and 9.82% for cellulose, hemicellulose and lignin, respectively. However, fungal pretreatment improved the depolymerisation and solubilisation of WH's hemicellulose and lignin contents, thus increasing the accessibility of the cellulose to microorganisms or enzymes. While P-60% had 49.10, 26.71 and 78% decreases in cellulose, hemicellulose and lignin contents, respectively, P-70% reduced the respective components to 49.84, 25.33 and 81.18% content, and P-75% resulted in a high decrease in cellulose, hemicellulose and lignin (52.34, 28.75 and 82.61%, respectively). Among the three pretreatment options, 75% of MCs of the pretreatment efficiently degraded the lignocellulose matrix and altered its constituents. Several studies also reported a decrease in the lignocellulosic content as a result of pretreatment.15,18,55 Lalak et al. 15 studied the effect of three MCs (45, 65, 75%) during Flammulina velutipes pretreatment on AD of Tall Wheat Grass (Agropyron elongatum) at 37°C and reported the highest lignin degradation at 65% MCs. Zhao et al. 20 stated a high lignin degradation of 20% in Ceriporiopsis subvermispora pretreated yard trimmings at 60% MC. A valid explanation for the differences in MCs is probably associated with the water-holding capability of the different biomass.
Similar to the carbon, the nitrogen content of the influent from different MCs of the pretreated and untreated reduced considerably from the effluent. This resulted in a reduction in carbon to nitrogen ratio (C/N), which according to Kassongo et al., 54 is fundamental for the growth and functioning of the microorganisms. The percentage decrease of the C/N ratio for P-60%, P-70% and P-75% were 19.36, 20.93 and 21.62%, while the untreated and cow dung inoculum were 28.20 and 21.92%, respectively (Table 2). These decreases in the C/N ratio result from the degradation of organic matter and the build-up of inorganic nitrogen sources. The optimal C/N is considered at 20–30 for balanced nutrition for the microorganisms and improved biogas yield. 56 A high C/N ratio limits nitrogen needed for microbial growth, while a low C/N impedes biogas yield as a result of ammonia inhibition. However, the C/N ratios of both pretreated and untreated substrates are below this range, while the cow dung inoculum is adequate. The C/N ratio is considered to rely on substrate and inoculum characteristics; Kwietniewska and Tys 57 reported the C/N ratio of freshwater microalgae as 10.2 and terrestrial plants as 36. Co-digestion of substrates is therefore recommended for future analysis as it efficiently balances the substrate carbon and nitrogen and consequently improves biogas yield.
Generally, the result of this study implies that MC substantially impacts the biodegradation process. From the analysed process factors, the adjustment of the MC to a level of 75% optimised the delignification of the water hyacinth by T. atroviride, which may have enhanced the availability of holocellulose to enzymes and anaerobic microorganisms. This outcome was similar to Shi et al., 26 which obtained a 27.6% delignification at 75% MC in the fungal pretreatment of corn stalk and the outcome was insignificantly different from the 80% MC. However, a higher MC during pretreatment is reported to decrease the porosity of the substrate and restrain oxygen transfer. According to Kainthola et al., 17 adequate MCs improve delignification as a result of the absorption of water molecules by solids, which results in softened substrates, decreased cohesive internal forces, enlarged crystalline matrix of cellulose and increased enzyme accessibility.
Effect of T. atroviride pretreatment on biogas and methane production
The optimisation conditions for biogas and methane generation from water hyacinth were explored through the pretreatment process. Methane contents are the principal pointer of biogas quality; higher methane content is directly proportional to good quality biogas. Biogas compositions such as carbon dioxide and methane contents were measured daily for 35 days. The daily and cumulative biogas and methane production values are depicted in Figure 3 (a–d) and Supplementary material: Table S1. Biogas and methane production from untreated water hyacinth displayed a more prolonged early phase of up to 3 days due to the abridged accessibility of the substrate by the microorganisms as a result of its lignin component. Thereafter biogas and methane production rapidly increased to a maximum yield of 135 mL/g VS and 91.84 mL/g VS, respectively, at days 20 and 22. The yield decreased afterwards to 87 mL/g VS for biogas and 63 mL/g VS for methane on day 32. Biogas and methane yield from the untreated water hyacinth remained relatively stable until day 35. Biogas production from inoculum started on the 2nd day and increased gradually till the 20th day. The inoculum’s maximum biogas and methane yields were 63.5 mL/g VS and 40.19 mL/g VS, respectively. The biogas yield for inoculum and untreated cow dung were remarkably lower than the pretreated substrates. The biogas and methane yield from water hyacinth pretreated with T. atroviride began on day 2 due to the exponential growth of microbes in the reactor. This implies that pretreatment quickens biogas generation and decreases the lag phase. The maximum biogas yield of 210.3 mL/g VS and methane yield of 169.76 mL/g VS were noted for P-60% on days 23 and 22, respectively. Biogas yield of 217 mL/g VS and methane yield of 180.91 mL/g VS were observed for P-70% at day 22, while biogas and methane yield of 223.4 mL/g VS and 200 mL/g VS, respectively, were noted for P-75%, at day 23 (Figure 3(a) and (c)).
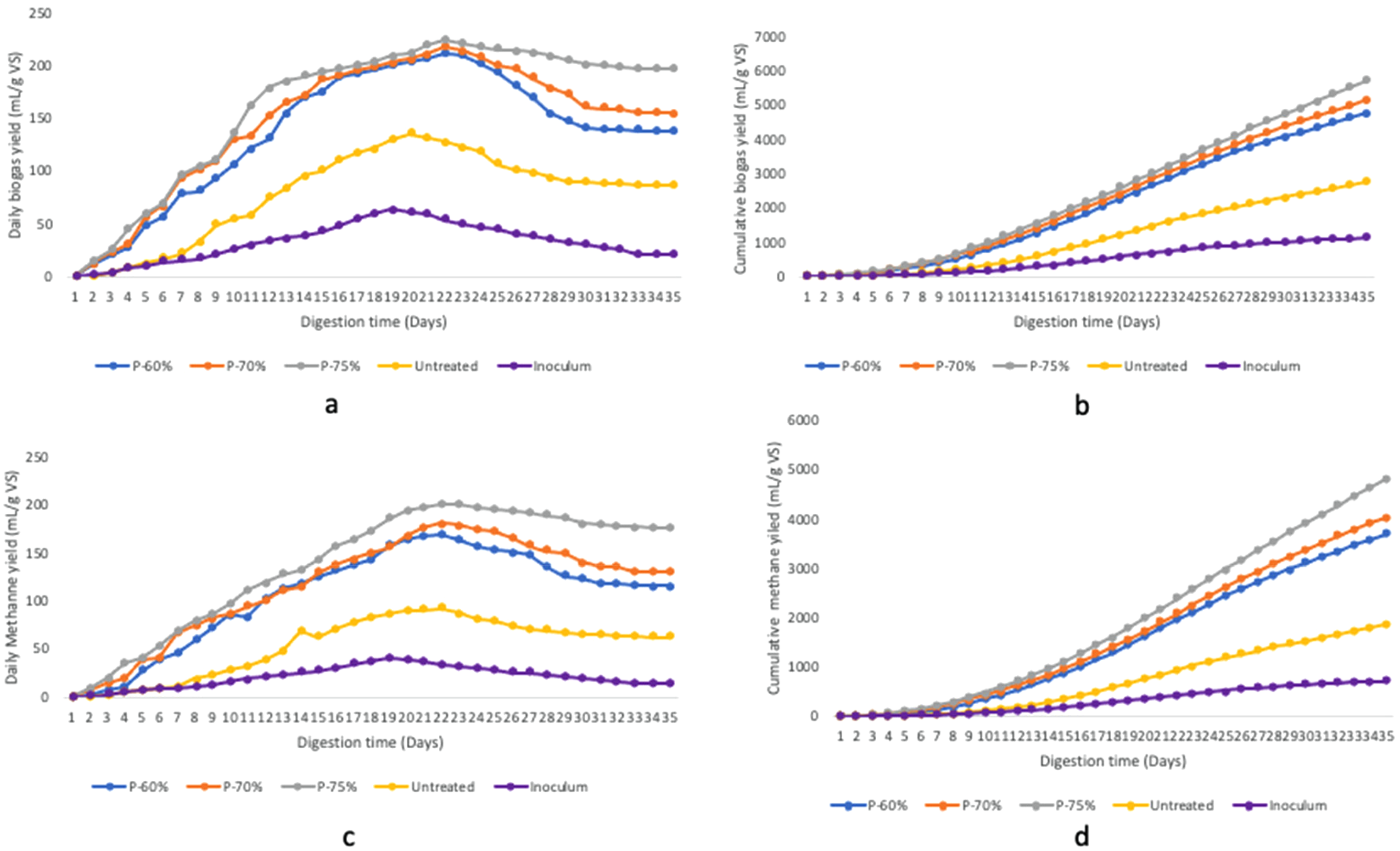
Daily and cumulative yield of biogas (a and b) and methane (c and d) from anaerobic digestion of pretreated and untreated water hyacinth.
The average percentage of methane content for inoculum, untreated, P-60%, P-70% and P-75% were 64, 65, 73, 76 and 82%, respectively, while the average percentage of their respective carbon dioxide content were 36, 35, 27, 24 and 18%. For biogas and methane yield, considerable impacts of the differences in MC during pretreatment were observed. The results indicate that the low methane yield of P-60% compared to other pretreated samples was linked to inadequate water levels affecting nutrient accessibility and cell metabolism. T. atroviride fungal pretreatment of water hyacinth with 75% moisture enhanced the degradability of water hyacinth by breaking down the cell wall structure, facilitated its use by microorganisms, and consequently yielded a higher percentage of biogas. This is similar to the study of Mustafa et al., 21 who noted a maximum average methane yield of 263 L/kg VS (120% higher than untreated) and 214 L/kg VS (78.3% higher than untreated) during digestion of P. ostreatus and T. reesei pretreated rice straw under mesophilic temperature, respectively, at 75% MC. It is interesting to note that statistical analysis showed no significant difference (p-value of 0.209) in the biogas production across the pretreated tests (P-60%, P-70% and P-75%); however, there was a significant difference across the methane yields (p-value of 0.058).
Figure 3(b and d) depicts cumulative biogas and methane production. The cumulative biogas yield for untreated, inoculum, P-60%, P-70% and P-75% are 2744.1, 1131.2, 4763.2, 5153.6 and 5714.8 mL/g VS, respectively. Compared to the untreated substrate, the yield significantly increased with increasing MC, resulting in a 73.58% improvement in P-60%, 87.81% in P-70% and 108.25% in P-75%. The cumulative methane yield followed a similar pattern as the cumulative biogas yield. The outcome observed were 1855.04 mL/g VS for untreated, 719 mL/g VS – cow dung inoculum, 3704.04 mL/g VS – P-60%, 4043.38 mL/g VS – P-70% and 4817.13 mL/g VS – P-75%. These outcomes imply that the T. atroviride pretreatment at diverse MCs efficiently enhances biogas and methane yield. This impact is probably due to the water molecule playing a crucial part in the fungal treatment at the different MCs. Bound water can form hydrogen bonds with cellulose, causing the crystalline cellulose structure to swell and increasing enzyme availability. 15
Kinetic simulation of biogas production
The reliability of the model outcomes from the modified Gompertz model was assessed by plotting the predicted cumulative methane values, presented in Supplementary material: Table S2, against the experimental values, as shown in Figure 4(a–d). Pretreatment improved the methane yield; the higher the MC, the easier the rate of biodegradation and, consequently, the higher the methane yield. The graphs confirmed that the modified Gompertz model depicts cumulative methane yield as a function of retention time. The outcomes of the kinetic simulation are presented in Table 3. The result shows that maximum methane potential increased with a high MC of pretreatment. P-75% had the highest maximum methane potential of 192.50 mL/g VS, possibly because the pretreatment made the substrate more degradable. However, Suksong, Wongfaed 58 reported a higher predicted methane yield. T. reesei and P. ostreatus fungal pretreated oil palm empty fruit bunches at 80% MCs and 85% MCs, respectively, were 315.2 and 263.4 mL CH4/g. The lag phase gives the time required by anaerobic microbes to adjust in the slurry before biogas production. A high lag phase means the microbes need a more extended period to generate biogas and vice versa. The shortest lag phase by P-75% was 2.46 days, while the longest lag phase was untreated for 6.94 days. Kainthola et al. 18 achieved a lower lag phase (2.14–3.59 days) for fungal pretreatment of rice straw. Therefore, the long lag phase for the untreated water hyacinth is due to the inability of the microbes to degrade lignin content. This validates that fungal pretreatment reduced the lag phase of the AD process.
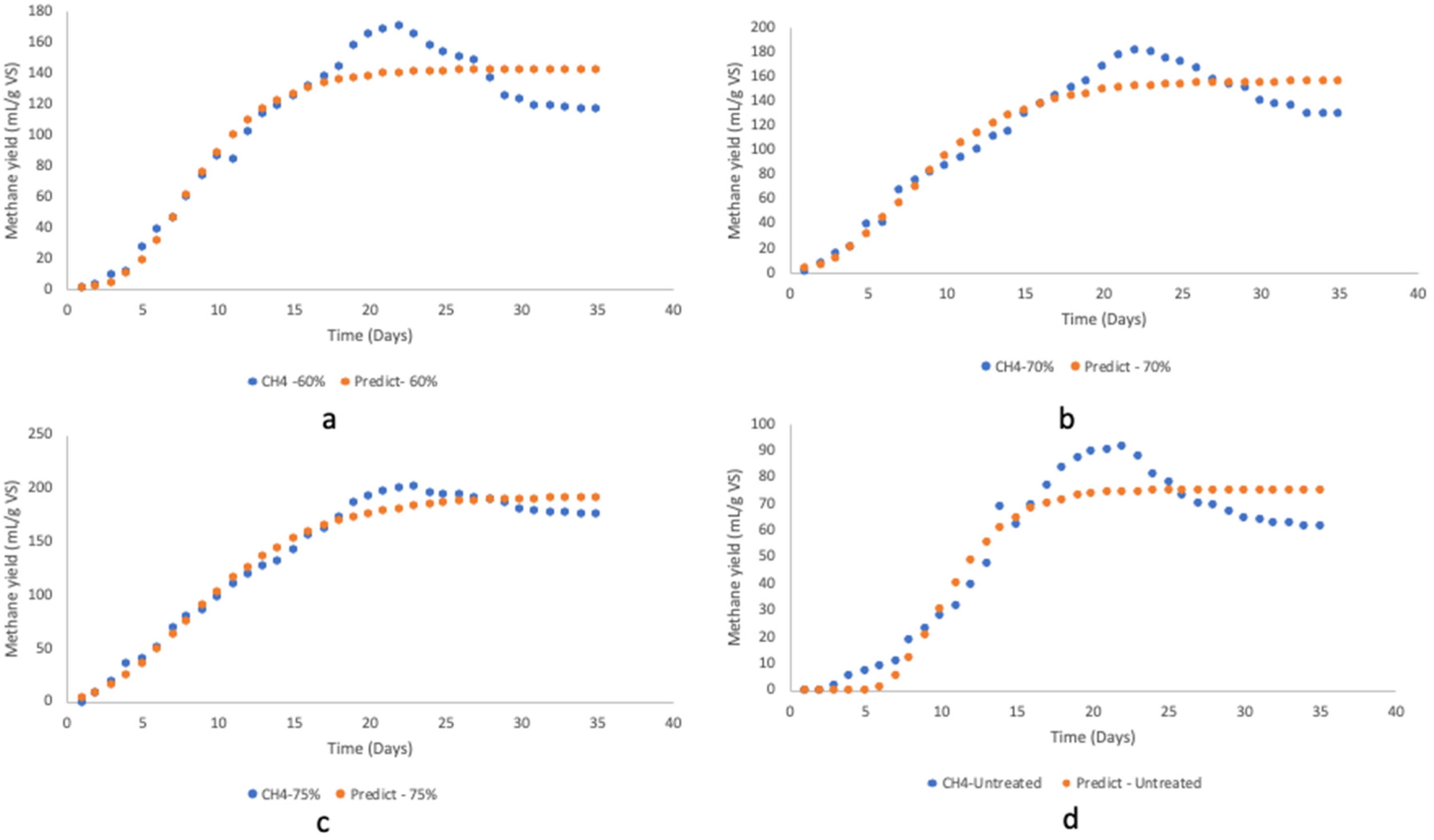
Modified Gompertz model fitting for T. atroviride pretreated and untreated water hyacinth (a) P-60%, (b) P-70%, (c) P-75% and (d) untreated.
Results of the kinetic study – modified Gompertz model.

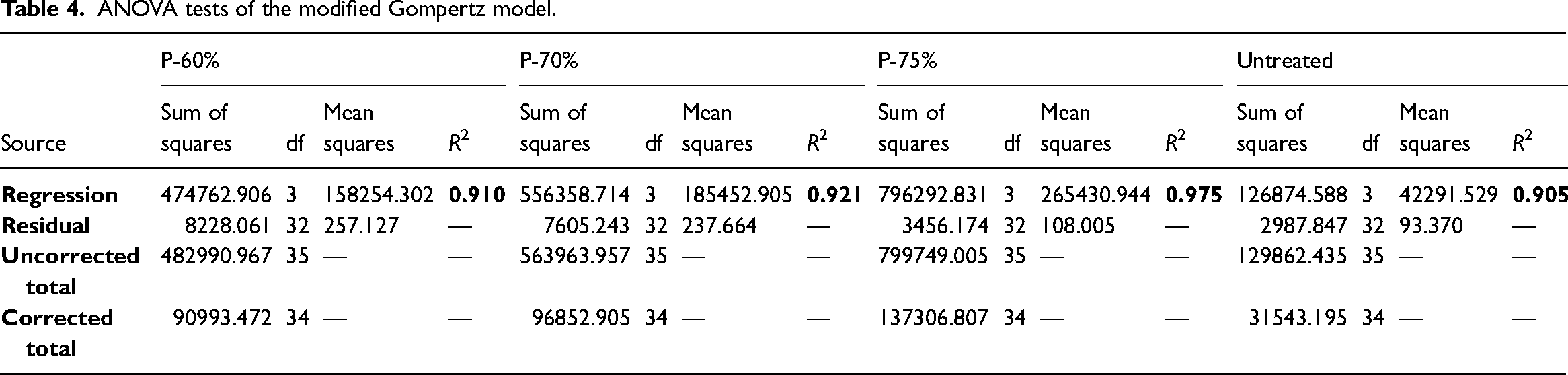
The model’s performance followed the trend P-75% > P-70% > P-60% > untreated. Untreated water hyacinth had a higher percentage error of 17.96%, while P-75% had the lowest error of 4.68%. According to Kıvak, 59 the percentage error between experimental and predicted outcomes ought to be less than 20% for best fitting. This study's average fitting error between experimental and predicted treatment types was 13.37%. Furthermore, the goodness of fit showed R2 values of 0.905, 0.910, 0.921 and 0.975 for untreated, P-60%, P-70%, and P-75%, respectively (Table 4). According to Barua et al. 42 and Alfa et al., 51 R2 values close to 1 suggest a good fit, implying a strong correlation between experimental and predicted data by the model. Therefore, the R2 values of >0.90 for both pretreated and untreated water hyacinth imply an efficient methane yield simulation. The outcome of the microbial kinetic factors, Y(t), Rm, implied that the utilisation of T. atroviride fungal pretreatment with AD is a favourable water hyacinth mitigation technique.
ANOVA tests of the modified Gompertz model.
Effect of T. atroviride pretreatment on the structural characteristics of water hyacinth
The FESEM was utilised to measure and examine the untreated water hyacinth and structural changes that had transpired on the T. atroviride fungal pretreated water hyacinth with optimal MCs (P-75%). Figure 5 shows the images of the structural characteristics of P-75% and untreated water hyacinth by FESEM. As shown in Figure 5(a), the image of the untreated water hyacinth revealed a shrivelled and dense surface with visible gorges, which suggests that the fibres were intact. There were no pores on the surface of the untreated water hyacinth. However, T. atroviride fungal pretreatment increased the substrate's surface area and pore sizes by penetrating its structure (Figure 5(b)). The formation of the pores in the cell wall implied lignin degradation, and this agrees with other investigations that consider such microscopic changes in the fibres as lignin disintegration.19,24 The pretreatment by T. atroviride also exposed the secondary rigid walls for microbial accessibility to the cellulose, thereby increasing the utilisation efficacy of water hyacinth and consequently improving the AD process.
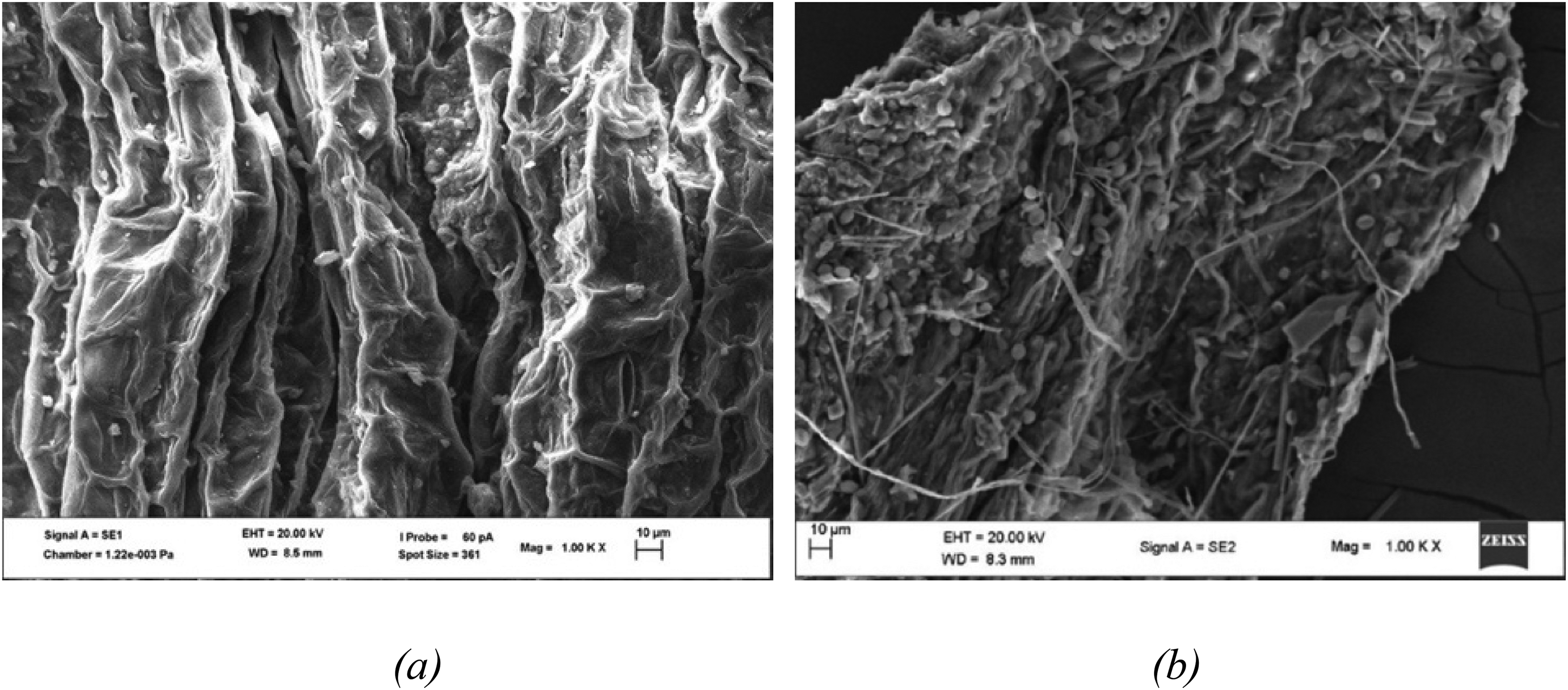
FESEM images of (a) untreated and (b) pretreated water hyacinth with 75% moisture content.
Conclusions
Fungal pretreatment of lignocellulose is considered to be time-consuming, even though they are sustainable and cost-efficient. However, research has shown that understanding the parameters for fungal growth is crucial for optimal yield. Mesophilic batch anaerobic digesters were utilised to explore the effects of different MCs during T. atroviride fungal pretreatment on the anaerobic biodegradation process and biogas and methane yields. The MCs substantially affected the T. atroviride pretreatment of water hyacinth by causing a significant delignification. The optimal degradation of lignin (82.61%) and hemicellulose (28.75%) were attained at a MC of 75%, resulting in a 108.25% increase in biogas yield compared to untreated. In contrast, degradation of lignin and 78% and 26.71% of hemicellulose were obtained at MCs of 60%, resulting in a 73.58% increase in biogas yield. Also, 81.18% and 25.33% were achieved at MCs of 70%, resulting in an 87.81% increase in biogas yield compared to untreated. However, 75% moisture enhanced the degradability of water hyacinth optimally by breaking down the cell wall structure, facilitated its use by microorganisms, and consequently yielded a higher percentage of biogas. This indicates that the T. atroviride fungal pretreatment is an efficient technique for improving the AD of water hyacinth. The result of the FESEM image validates the effectiveness of T. atroviride fungal pretreatment in lignin degradation. Furthermore, modified Gompertz indicated R2 (0.975) of P-75% to be more fit compared to other MCs and less fit for the untreated. The study is deemed an initial step in developing approaches to further impact hydrolysis and eventually enhance the methane yield. It offers potential solutions in biorefineries for greater output, substrate efficacy and viable operational processes. The finding indicates an essential prospect for considering water hyacinth as a valuable resource material beneficial to low carbon and resilient climate economy. It is anticipated that the study's outcome will contribute to policy declaration and scholarly considerations that would give rise to more research, especially on the techno-economic performance of the T. atroviride pretreatment of water hyacinth and financing towards upscaling biogas production.
Contributions
Obianuju Patience Ilo – Conceptualisation, formal analysis and investigation, and writing – original draft. S’phumelele Lucky Nkomo, Ntandoyenkosi Malusi Mkhize, and Mulala Danny Simatele – writing, review and editing, supervision. Onisimo Mutanga – funding acquisition, supervision. All authors read and approved the final manuscript.
Supplemental Material
sj-docx-1-eae-10.1177_0958305X221141397 - Supplemental material for The effects of Trichoderma atroviride pretreatment on the biogas production from anaerobic digestion of water hyacinth
Supplemental material, sj-docx-1-eae-10.1177_0958305X221141397 for The effects of Trichoderma atroviride pretreatment on the biogas production from anaerobic digestion of water hyacinth by Obianuju Patience Ilo and S’phumelele Lucky Nkomo, Ntandoyenkosi Malusi Mkhize, Onisimo Mutanga, Mulala Danny Simatele in Energy & Environment
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of South Africa (NRF) Research Chair initiative in Land Use Planning and Management, (grant number 84157).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
