Abstract
The decarbonisation of the heating sector is critical for achieving Net Zero targets, with hydrogen fuel emerging as a promising alternative to natural gas. This study experimentally investigates the operational feasibility, combustion, and emissions characteristics of hydrogen-enriched fuel mixtures in an industrial burner. The PHOTON10 unit is used as an industrial burner. Experiments were conducted using methane (i.e., G20) enriched with hydrogen in different (% vol) quantities to assess flame behaviours like stability and emission profiles. Key challenges identified include flame flashback, thermal retraction, and diminished flame rectification signals, particularly under daylight conditions due to hydrogen’s low visible emissivity. To maintain rated thermal output, hydrogen consumption was increased by up to threefold when operating with 100% hydrogen. Emission analysis revealed significant reductions in CO2, NO, and NOx with increasing hydrogen concentration, while flame temperature decreased correspondingly. All the tested hydrogen-enriched mixtures met regulatory thresholds for CO, CO2, NO, and NOx emissions for industrial applications. The PHOTON10 burner demonstrated compatibility with hydrogen fuels with minimal design modifications, primarily in air intake parameters. In closed-loop operation, reliable flame monitoring was achieved via flue gas temperature and UV flame signal intensity. These findings contribute to the advancement of hydrogen-compatible burner technologies and support the broader transition toward low-carbon industrial heating solutions.
Introduction
Fossil fuels have historically constituted the foundation of global economic development, serving as the primary energy source for industrial expansion, transportation networks, and power generation systems. However, the sustained reliance on these carbon-intensive resources has precipitated profound environmental challenges, including elevated greenhouse gas emissions, deteriorating air quality, and the depletion of finite natural reserves. These impacts have intensified ecological degradation and heightened concerns regarding long-term energy security. 1 Considering these challenges, the industrial sector is increasingly compelled to adopt sustainable and low-emission energy alternatives. Among the emerging solutions, hydrogen has garnered significant attention due to its favourable thermochemical properties and alignment with decarbonization objectives. Supported by technological innovation and progressive policy frameworks, hydrogen is positioned as a key enabler of the energy transition. 2 For instance, the European Union has identified hydrogen as a strategic pillar in its roadmap toward achieving a climate-neutral economy by 2050. 3
Alternative fuels such as hydrogen, and ammonia have created increasing attention for their potential to mitigate environmental impacts and reduce dependence on fossil-based energy sources. 4 Among these, hydrogen is particularly notable due to its high specific energy content and its capacity to produce water as the sole combustion by-product, thereby offering a zero-carbon pathway for energy conversion. 5 Despite these advantages, the widespread adoption of hydrogen faces several technical and economic barriers. These include the elevated costs associated with hydrogen production, the underdeveloped infrastructure for its storage and distribution, and the relatively low efficiency of current conversion technologies.6–8 One promising approach to facilitate the integration of hydrogen into existing energy systems is through its blending with natural gas. This strategy enables a reduction in carbon emissions while leveraging the existing infrastructure for gas distribution and combustion. The concept of hydrogen-enriched fuel mixtures is not novel; it has historical precedence in the use of town gas—a manufactured fuel derived from coal—which was widely distributed in Europe and North America prior to the transition to natural gas. Depending on the production method, town gas typically contained 30–50% hydrogen by volume, thereby demonstrating the long-standing feasibility of hydrogen blending and providing a valuable reference point for contemporary decarbonization initiatives.
Recent advancements in renewable hydrogen production technologies have been closely accompanied by progress in complementary systems, including hydrogen fuel cells,9,10 hydrogen-powered internal combustion engines for transportation applications, 11 and low-emission, fuel-flexible gas turbines for industrial and aerospace sectors. 12 These developments underscore hydrogen’s versatility and its potential to serve as a cornerstone in future low-carbon energy systems. Empirical studies have demonstrated that hydrogen integration can enhance engine performance and reduce carbon dioxide (CO2) emissions. 13 However, significant challenges persist, particularly in the domains of hydrogen production economics, storage logistics, and conversion efficiency. 14 The blending of hydrogen with methane has emerged as a viable strategy to improve combustion efficiency and reduce pollutant emissions, 15 offering a transitional pathway that leverages existing infrastructure while mitigating carbon intensity. Extensive research has explored the adaptation of hydrogen in internal combustion engine applications. Verhelst and Wallner 14 examined the implications of hydrogen’s low energy density, high flame speed, and broad flammability range, highlighting the need for modifications in ignition timing and fuel injection systems. Hydrogen-enriched fuel mixtures exhibit complex interactions with engine performance and emissions. For instance, Liu et al. 16 reported that increasing hydrogen concentration improves brake thermal efficiency (BTE), albeit with a corresponding rise in nitrogen oxides (NOx) emissions. Zhen et al. 1 found that methanol-hydrogen blends enhanced power output and reduced emissions, though benefits were limited to hydrogen concentrations below 10%.
Additional studies have shown that hydrogen combustion reduces hydrocarbon (HC) and carbon monoxide (CO) emissions, though its effects on brake-specific fuel consumption (BSFC) and NOx emissions vary depending on operating conditions and blend ratios.17–19 Safety remains a critical concern in hydrogen integration, particularly regarding the flammability limits and explosive potential of methane-hydrogen mixtures. 20 The transition from natural gas to hydrogen-rich fuels necessitates careful consideration of hydrogen’s distinct physical and chemical properties, including its low ignition energy, high diffusivity, and rapid flame propagation. 5
Fossil fuels have historically underpinned global industrial growth, serving as the dominant energy source for manufacturing, transportation, and power generation. However, sustained reliance on carbon-intensive fuels has contributed to elevated greenhouse gas emissions, declining air quality, and growing concerns over long-term energy security and environmental resilience. These pressures have accelerated the search for low-carbon alternatives within the industrial heating sector. Among the emerging solutions, hydrogen has attracted substantial interest due to its favourable thermochemical properties, capacity for carbon-free combustion, and alignment with national and international decarbonisation strategies. Hydrogen is now recognised as a strategic vector in the transition toward climate neutrality, as reflected in major initiatives such as the European Union’s 2050 net-zero roadmap.
Hydrogen blending into existing natural-gas infrastructure offers a practical pathway for reducing carbon intensity without requiring full-scale equipment replacement. Indeed, the concept has historical precedent in the former “town gas” networks of Europe and North America, where manufactured gas typically contained 30–50% hydrogen.21,22 Modern advances in hydrogen production (supported by parallel developments in fuel cells, hydrogen-internal-combustion engines, and flexible gas-turbine technologies) have renewed interest in hydrogen-natural-gas blends for industrial heating.
Numerous engine-based studies have examined hydrogen addition, reporting complex interactions among flame speed, thermal efficiency, and pollutant formation. While many have shown reductions in CO2, HC, and CO emissions, increases in NOx have also been observed depending on operating conditions, combustion mode, and mixture preparation. Across this literature, NOx behaviour in hydrogen-enriched systems emerges as one of the most contradictory findings. Studies performed in engine or diffusion-dominated environments, such as those by Moreno et al. 23 Liu et al.24,25 and Jeong et al. 26 have commonly reported NOx increases with rising hydrogen fraction due to higher flame temperatures, longer residence times, and the strong activation of thermal (Zeldovich) NO pathways. In contrast, lean premixed and swirl-stabilised combustors frequently demonstrate NOx reductions at elevated hydrogen levels because enhanced mixing suppresses hot spots, shortens high-temperature residence zones, and reduces prompt NO pathways associated with hydrocarbon radicals. These divergent outcomes highlight that NOx formation is highly dependent on equivalence ratio, burner geometry, flow aerodynamics, and the dominant combustion regime is reinforcing the need for system-specific evaluation in industrial premixed burners.
Hydrogen enrichment also influences CO and unburned hydrocarbon (UHC) emissions, with trends that vary across combustion systems. In well-mixed, lean premixed conditions, hydrogen generally reduces CO and UHC emissions by enhancing oxidation completeness, increasing OH radical availability, and reducing ignition delay. Conversely, engine and diffusion-dominated studies have observed CO increases at high hydrogen fractions under fuel-rich or low-temperature conditions where reduced thermal intensity inhibits effective CO oxidation. UHC emissions typically decline with rising hydrogen content due to hydrogen’s lack of carbon-based intermediates and improved flame propagation, although ultra-lean or misfire-prone regimes may produce small increases. These mixed results emphasise the strong dependence of CO and UHC behaviour on hydrogen fraction, equivalence ratio, mixing quality, and burner design.
A growing body of research23–26 has also quantified the influence of hydrogen enrichment on flame stability and flashback in premixed burner systems. Swirl-stabilised flames consistently exhibit improved lean stability and faster ignition with increasing hydrogen fraction but also heightened flashback susceptibility due to the sharp rise in laminar flame speed. Investigations of micro-mix injectors, bluff-body burners, and porous-media burners similarly report enhanced flame anchoring but reduced flashback margin, while larger gas-turbine premixed combustors require modified aerodynamics or flashback arrestors to safely operate at high hydrogen levels. These findings underscore that stability and flashback behaviour are strongly geometry-dependent which is an important consideration for industrial premixed burners such as the PHOTON10 examined in this study.
Hydrogen-rich flames introduce further challenges related to flame detection and monitoring. Owing to their inherently low visible luminosity and weak ionisation signals, hydrogen flames often evade detection by conventional flame-rod rectification systems. Studies such as those by Schefer et al. 27 have shown that hydrogen’s dominant spectral emissions occur in the ultraviolet rather than visible region. Hydrogen also suppresses CH* and C2* chemiluminescence while strengthening OH*-related pathways, making UV- and OH*-based detection significantly more reliable than ionisation-based systems. As industrial burners increasingly adopt hydrogen-rich fuels, these sensing challenges become operationally critical for safety and control.
Porous-media burners (PMBs) have also been shown to substantially reduce CO and NOx emissions compared to conventional free-flame systems. Mohammadi and Ajam demonstrated that introducing a porous matrix enhances internal heat recirculation, increases flame stability, and promotes more complete oxidation, resulting in markedly lower CO and NO emissions in methane-air combustion. Similarly, Ajam and Mohammadi reported that porous-media combustion exhibits near-zero NO formation under stoichiometric conditions and significantly reduced pollutant levels across a wide excess-air range due to extended reaction zones and uniform temperature fields. These findings highlight the potential of porous-media configurations as a low-emission strategy and provide useful context for the broader landscape of industrial hydrogen-compatible combustion technologies.28,29
Hydrogen blending has also been evaluated at industrial scale in boilers, furnaces, and process heaters. Notably, Wang et al. 30 demonstrated that hydrogen-enriched firing in a 108 t/h industrial steam boiler improved combustion efficiency and reduced NOx emissions, while introducing known challenges such as increased flame speed and altered heat-transfer distribution. Other industrial studies report similar trade-offs, highlighting both the decarbonisation potential of hydrogen and the need for design adaptations to manage stability and emissions. However, significantly less work has focused on compact industrial space-heating burners, which operate under different mixing, confinement, and control conditions compared to large boilers or furnaces. The present study helps fill this gap by experimentally characterising hydrogen-enriched combustion in the PHOTON10 premixed industrial heater, a unit representative of widely deployed systems across commercial and industrial buildings.
In addition to these industrially oriented studies, recent investigations of hydrogen-rich swirling flames, hydrogen-rich gas-turbine combustors, and ammonia-reformate hydrogen systems provide further insights into flame structure, emissions, and thermochemical dynamics relevant to premixed burner operation.30–32 These works show that hydrogen increases flame reactivity and alters heat-release patterns, often requiring tailored mixing strategies, revised control algorithms, and advanced sensing technologies. Collectively, the literature establishes that hydrogen enrichment influences flame stability, emissions, flame visibility, and sensing reliability in ways that are highly system specific. This underscores the importance of empirical assessments for industrial space-heating applications such as the PHOTON10 burner analysed in this work.
In line with recent developments in hydrogen combustion research (from 2022 to 2025), this study builds upon the latest findings in hydrogen-rich gas turbines, porous-media combustion, industrial boiler applications, and reformate-based hydrogen systems,26–33 ensuring the present work is situated within the most up-to-date scientific context.
The current study further examines the operational constraints of industrial burners when utilizing hydrogen-rich fuel blends, prior to any modifications to control systems. Particular attention is given to phenomena such as flame instability, flashback, visibility, thermal runaway, and detection. A critical challenge identified is flame rectification, as pure hydrogen flames are nearly invisible under daylight conditions, complicating flame detection and monitoring. Moreover, achieving the rated power output with 100% hydrogen requires significantly increased fuel consumption. Given the limited body of research addressing hydrogen combustion emissions in industrial space heating applications, this study contributes novel insights into the environmental and operational viability of hydrogen as a low-carbon alternative in burner technologies.
Experimental setup
Combustion experiments were conducted using a commercially available PHOTON10 premixed warm air heater, representative of industrial burner systems used in space heating applications. The experimental configurations are shown in Figure 1 (Open Setup) and Figure 2 (Enclosed Setup), designed to facilitate controlled and repeatable testing of hydrogen–methane fuel mixtures across a range of blending ratios. Schematic diagram of the experimental setup for open setup. Schematic diagram of the experimental setup for enclosed setup.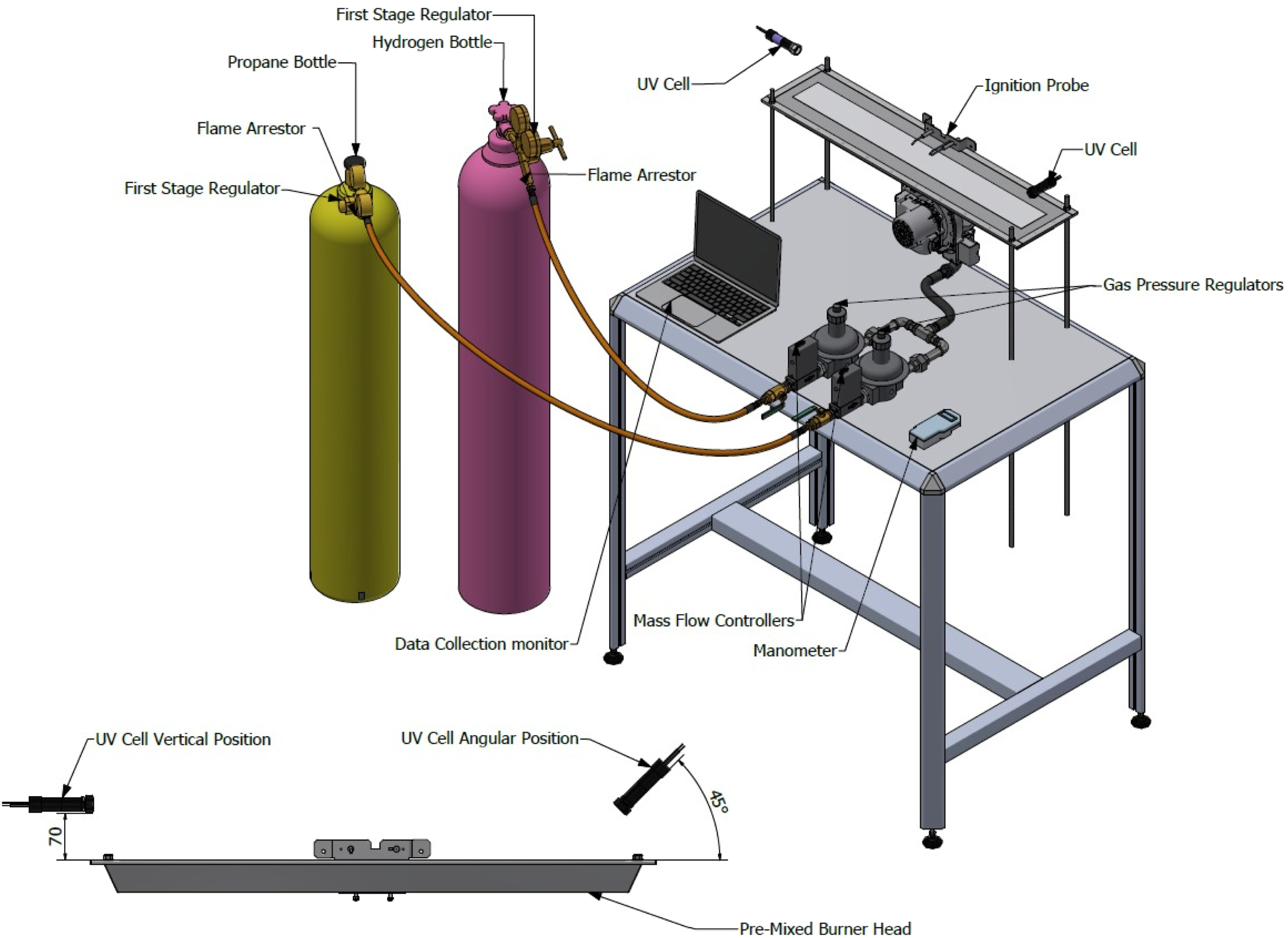
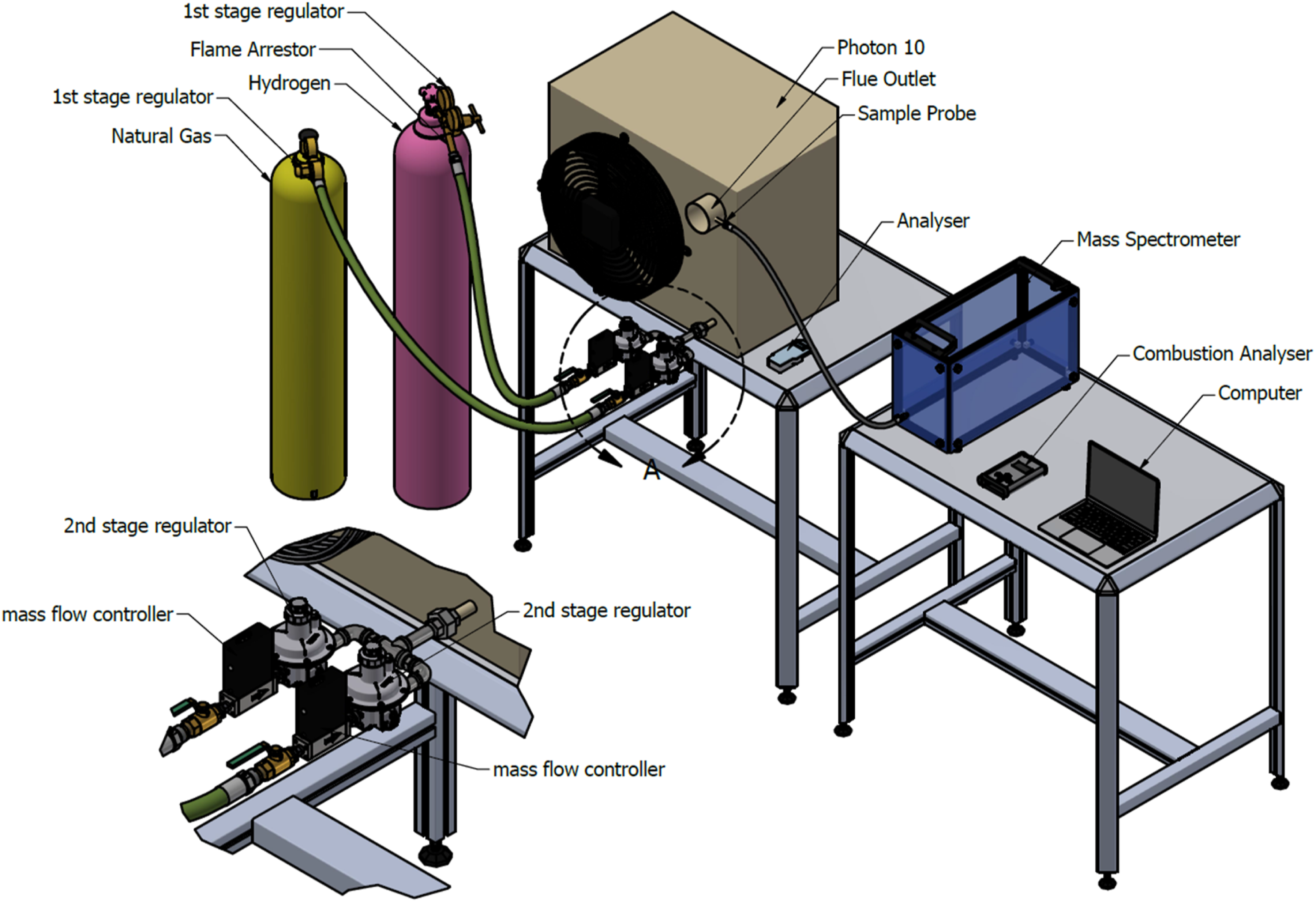
The fuel delivery system comprised two independent gas trains for hydrogen and methane (G20), each equipped with digital volumetric mass flow controllers and dual-stage pressure regulation. The first-stage regulators reduced the cylinder pressure (200 bar) to an intermediate level, while the second-stage regulators further reduced the pressure to 20 mbar, consistent with the operational requirements of the PHOTON10 burner. A flame arrestor was installed downstream of the hydrogen regulator to mitigate the risk of flashback propagation.
Parametric table for the current study.
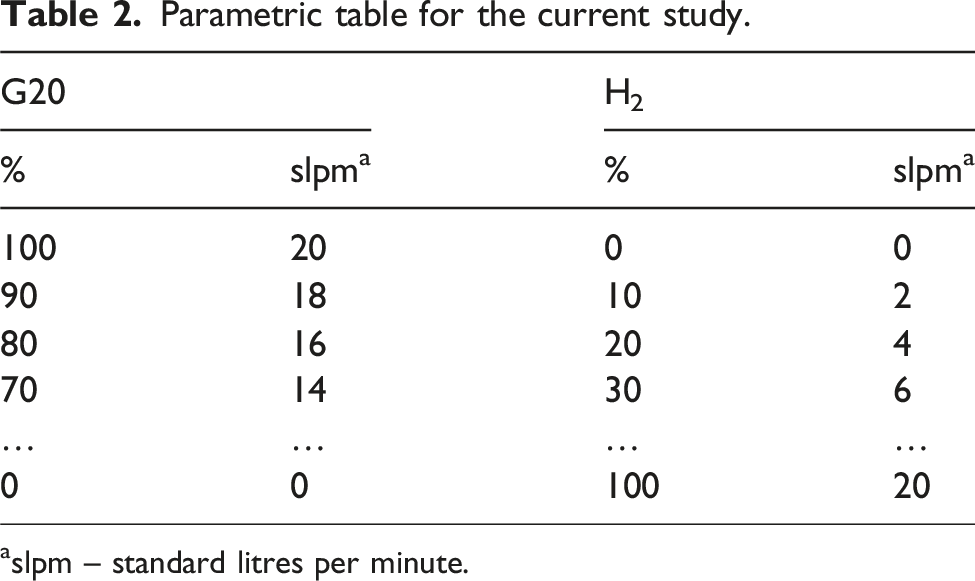
aslpm – standard litres per minute.
Equipment details used for current study.
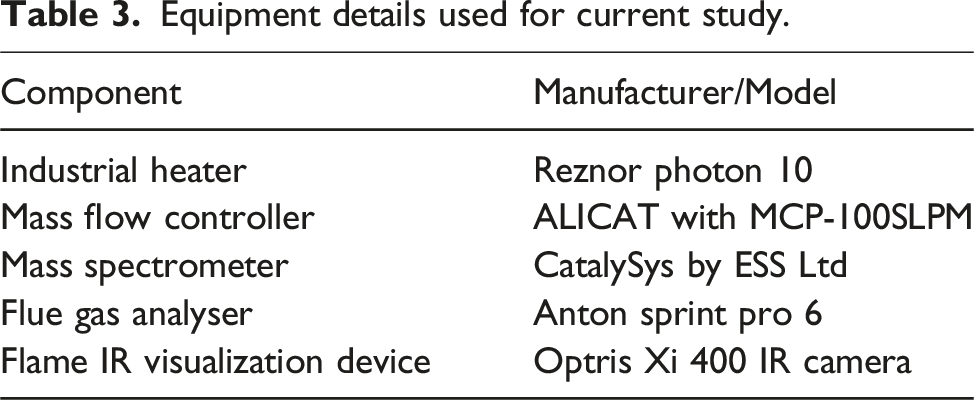
Equipment’s accuracy threshold limits.

Operating conditions.

The PHOTON10 system incorporates multiple safety interlocks and diagnostic feedback mechanisms to monitor ignition success, flame stability, and overall burner status. These features were actively monitored throughout the experimental campaign to ensure safe and reliable operation. A comprehensive summary of the burner’s operating conditions, control parameters, and system specifications is presented in Table 5.
The stoichiometric balance equations with air for methane and hydrogen can be expressed as: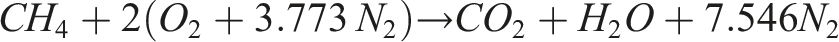
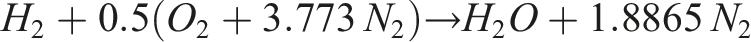
The equivalence ratio (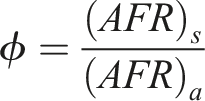
The equivalency ratio (ϕ) is an essential concept in combustion science and engineering that quantifies the ratio of the actual fuel-to-air ratio to the stoichiometric fuel-to-air ratio. 26
It serves as a critical indicator of mixture composition, where values of Effects of ϕ on emission species (Abbass, A. 2025)
1
.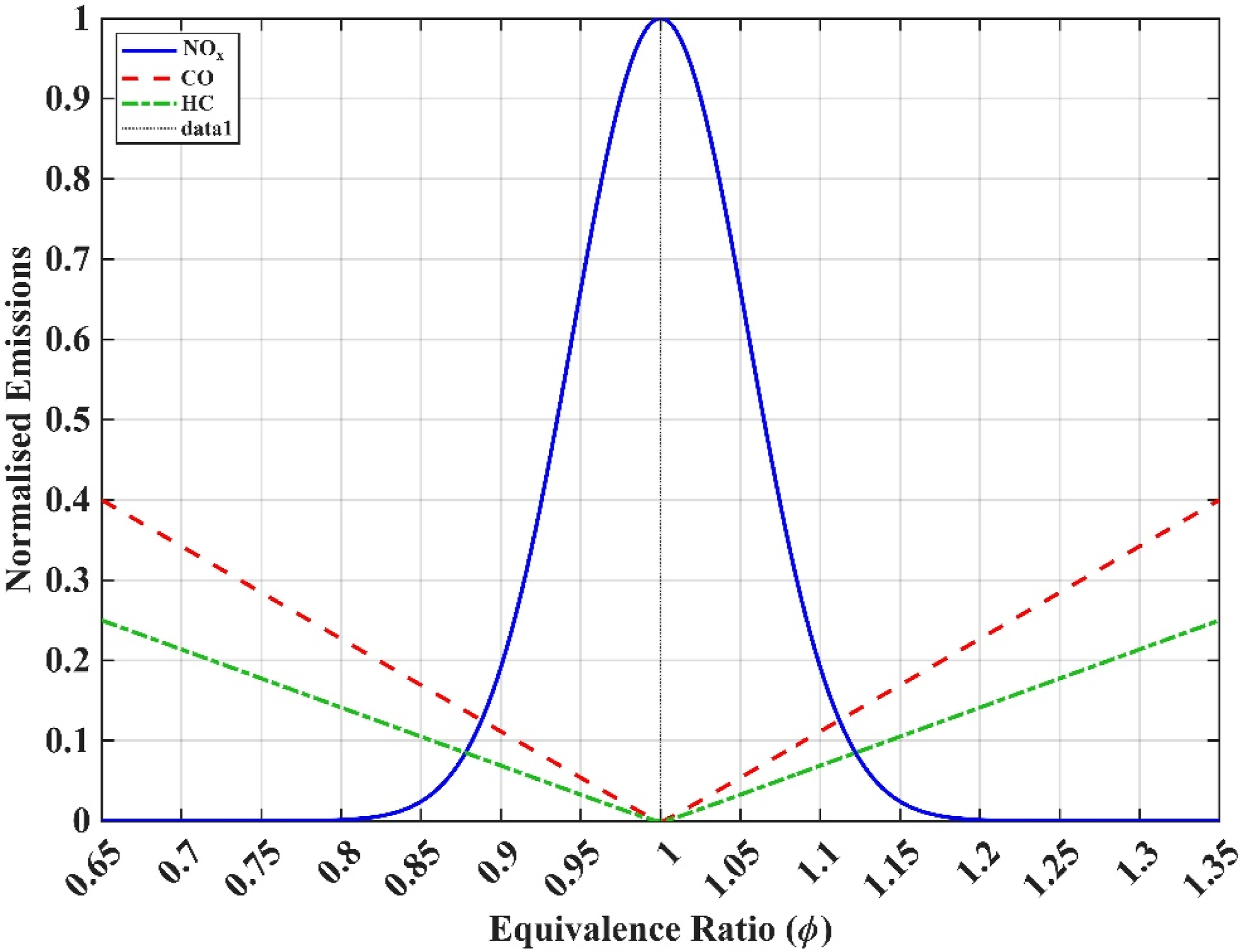
Optimizing the equivalence ratio is therefore essential for achieving a balance between combustion performance and environmental impact. In hydrogen-enriched fuel systems, this optimization becomes increasingly complex due to hydrogen’s unique combustion characteristics, including its high flame speed, broad flammability limits, and low ignition energy.5,43 These properties can amplify both the advantages and risks associated with lean and rich combustion regimes, necessitating precise control strategies to ensure safe and efficient operation.
The natural gas burner head used in this study is a widely utilised component in industrial heating systems, recognized for its robust performance and long-term durability. The burner assembly is fabricated entirely from 304-grade stainless steel, a material selected for its superior corrosion resistance and mechanical strength, rendering it suitable for high-temperature and chemically aggressive environments. Central to the burner’s thermal management strategy is a fibre matting layer composed of FeCrAlM, an advanced iron–chromium–aluminium–molybdenum alloy. This material is engineered to withstand continuous operating temperatures up to 1300°C, thereby ensuring structural integrity and operational reliability under sustained thermal loads. As illustrated in Figure 4, the FeCrAlM matting functions as an effective thermal barrier, mitigating the risk of deformation and mechanical failure due to prolonged exposure to elevated temperatures. The incorporation of FeCrAlM offers significant advantages in terms of thermal protection, contributing to the preservation of burner geometry and performance over extended operational cycles. This is particularly critical in industrial applications where consistent thermal output and system safety are paramount. Recent experimental investigations into hydrogen-enriched natural gas combustion have yielded promising insights into the influence of hydrogen on emission characteristics, combustion dynamics, and surface temperature distributions within the PHOTON10 burner system. These findings not only inform the optimization of the burner under study but also have broader applicability to similar premixed burner configurations. The results contribute to the ongoing development of cleaner and more efficient combustion technologies, particularly in the context of hydrogen integration into existing infrastructure. Premixed burner head (top-view, side-view and bottom-view) used in open setup experiments.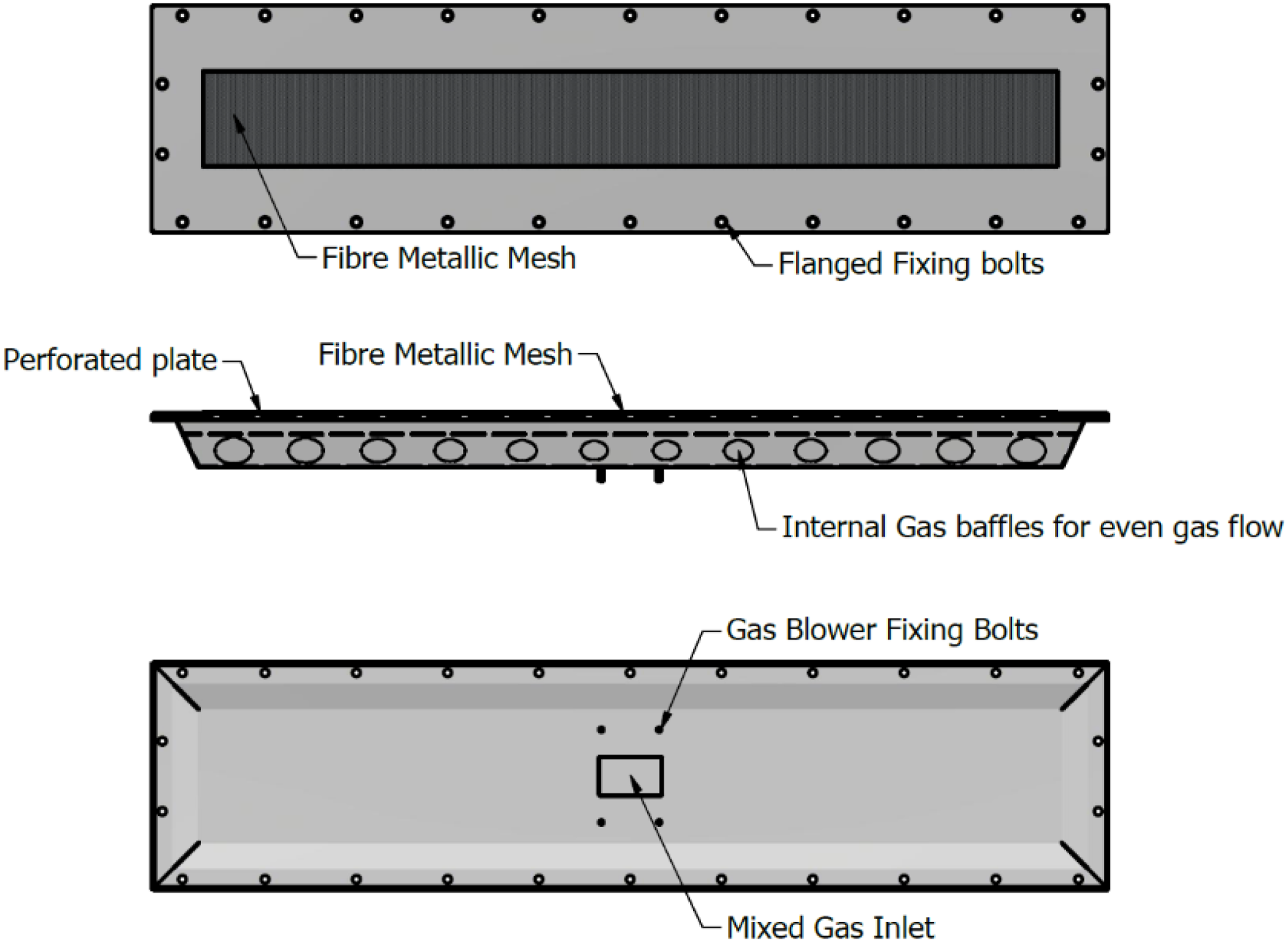
Results and discussions
The results presented in Figure 5 provide critical insights into the operational boundaries of hydrogen-enriched combustion within the PHOTON10 burner system. The flame sustainability data, obtained under open-flame conditions without modification to the burner’s safety control system, reveal a clear trend: increasing hydrogen concentration in the fuel mixture leads to a progressive decline in flame stability. The figure shows that a significant deviation in flame stability at hydrogen concentrations exceeding 50%. Complete flame extinction was observed at hydrogen concentrations above 60%, marking a critical threshold beyond which the burner system, in its current configuration, fails to sustain combustion. Flame sustainability and flame blow-off limits for PHOTON10 in open setup experiments.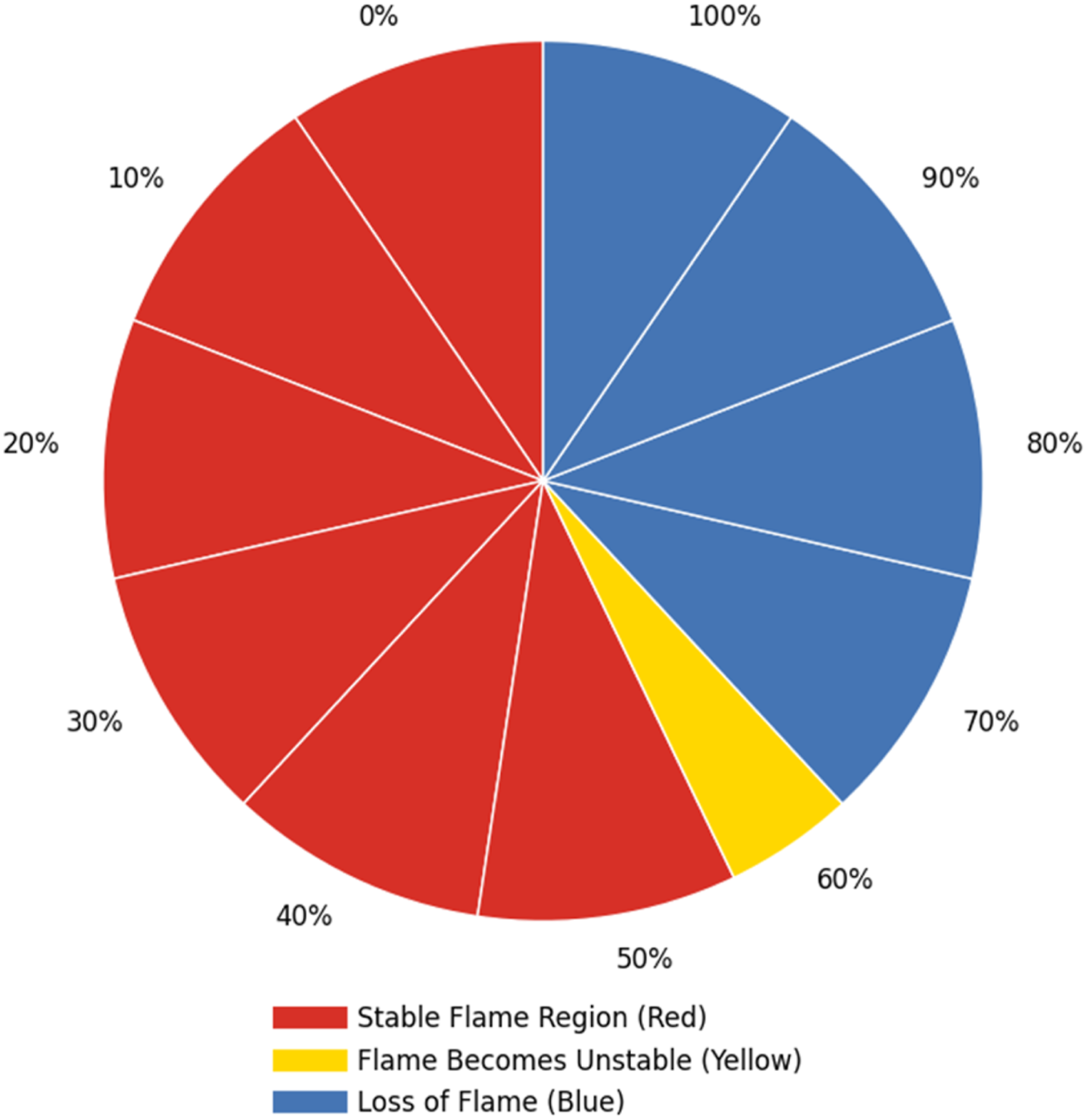
This instability can be attributed to several interrelated factors. First, lean fuel-air mixtures at high hydrogen concentrations may fall below the lower flammability limit, compromising ignition and flame propagation. Second, increased turbulence in the combustion zone can disrupt flame anchoring, particularly in premixed systems. Third, inadequate flame rectification—exacerbated by hydrogen’s low emissivity and near-invisibility in daylight—impairs the burner’s ability to detect and maintain a stable flame. Finally, hydrogen’s high flame speed and altered heat release characteristics introduce dynamic changes to the combustion process that challenge conventional burner designs. These findings underscore the importance of re-evaluating burner control strategies and safety mechanisms when integrating hydrogen into existing natural gas systems. The PHOTON10 burner, while capable of accommodating moderate hydrogen enrichment (up to 50%), requires significant design adaptations to operate safely and efficiently at higher hydrogen concentrations. This result additionally allows researchers to adapt potential modifications that include enhanced flame detection technologies (e.g., UV or IR sensors), optimized air-fuel mixing systems, and advanced control algorithms capable of responding to rapid changes in combustion dynamics. As the heating sector moves toward decarbonization, understanding the combustion limits and safety constraints of hydrogen-enriched systems becomes essential.
Figure 6 presents the measured output current from the flame rectification system (within PHOTON10) during combustion experiments with varying hydrogen concentrations. The graph illustrates a clear downward trend in rectified current as hydrogen content increases, highlighting a critical limitation in the use of conventional flame detection systems for hydrogen-enriched fuel mixtures. The flame rectification system, commonly employed as a flame surveillance/supervision device (FSD) in burner control architectures, operates by detecting the presence of ionized gases within the flame. This ionization enables the conduction of a microampere-level direct current (DC) signal through a sensor rod, which acts as a rectifying element. A minimum current threshold—established at 0.4 μA for PHOTON10—is required to confirm flame presence and permit continued fuel delivery. If the signal falls below this threshold, the system initiates a volatile lockout, terminating fuel flow to prevent unsafe operating conditions. As shown in Figure 6, hydrogen concentrations exceeding 50% result in a marked decline in rectified current, with values approaching or falling below the operational threshold. This degradation is attributed to the inherently low ionization potential of hydrogen flames, which produce insufficient charged species to sustain a measurable rectification signal. At concentrations above 60%, the flame fails to support rectification entirely, rendering the system ineffective for flame detection and compromising burner safety. To address this limitation, the experimental setup was modified to incorporate a UV (ultraviolet) flame detection cell as an alternative FSD. Unlike rectification-based systems, UV sensors detect the ultraviolet radiation emitted by active flames, offering a more robust and reliable method for flame verification in hydrogen-rich environments. This approach is particularly advantageous in industrial applications where flame invisibility and low ionization levels pose significant challenges to conventional detection methods. The integration of UV flame sensing technology represents a critical advancement in burner safety and control for hydrogen-enriched combustion. It enables accurate flame monitoring across a broader range of fuel compositions, thereby supporting the safe deployment of hydrogen as a low-carbon alternative in industrial heating systems. The result from Figure 6 underscore the necessity of adapting burner control systems to accommodate the unique combustion characteristics of hydrogen, particularly in the context of flame detection, stability, and operational reliability. Output current from flame rectification for hydrogen enriched combustion.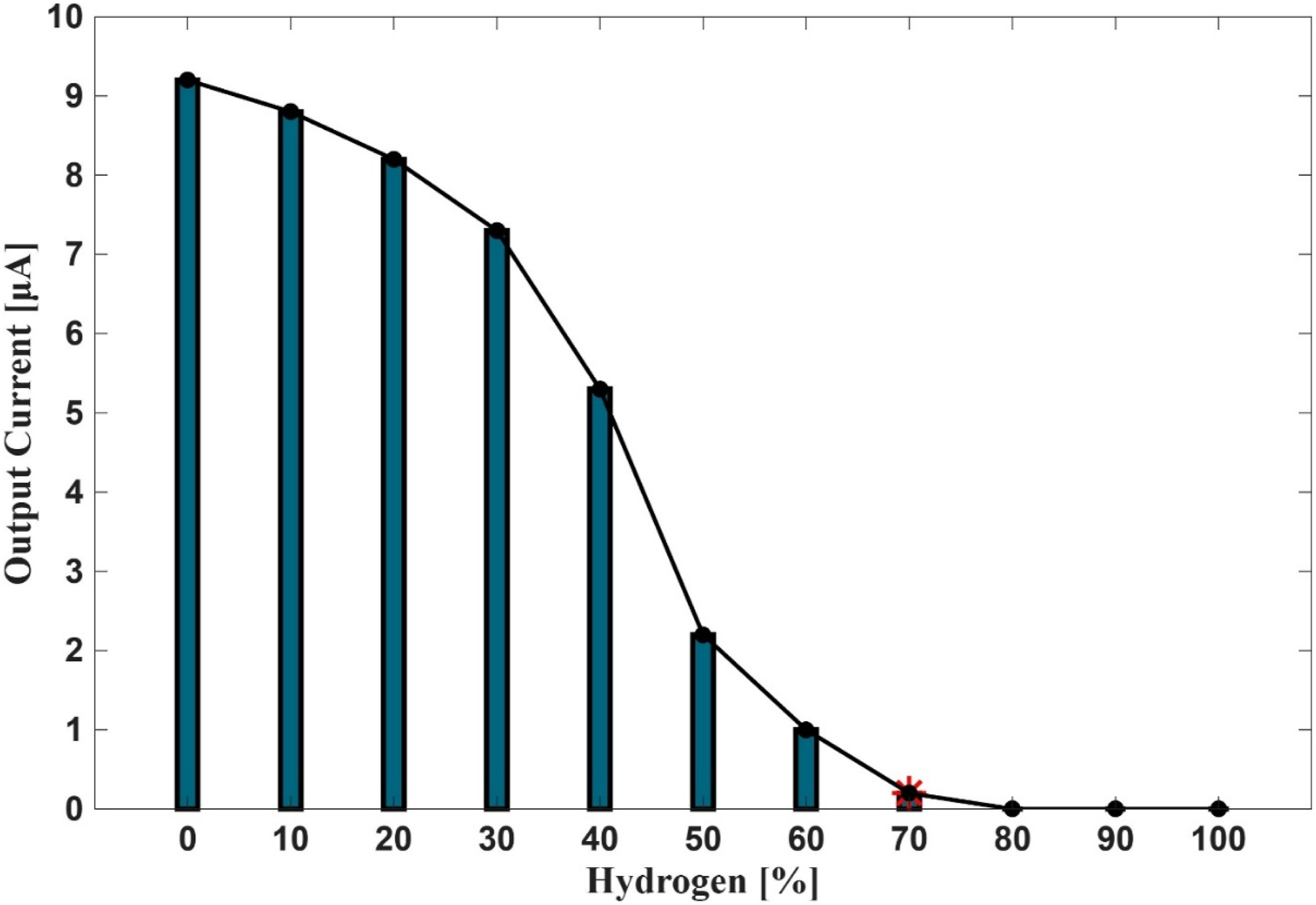
Figure 7 presents a comparative visual analysis of flame appearance across six different fuel compositions ranging from 100% methane (CH4) to 100% hydrogen (H2). The images were captured during open-flame trials using the PHOTON10 burner system, with the objective of evaluating flame visibility and structure under varying hydrogen concentrations. The top row of the figure illustrates flames with lower hydrogen content. The 100% methane flame (top left) exhibits high luminosity and clear visibility under ambient lighting conditions. As hydrogen concentration increases to 20% and 40% (top middle and top right), the flame becomes progressively less luminous, though still discernible. In contrast, the bottom row—representing mixtures with 60%, 80%, and 100% hydrogen—demonstrates a marked reduction in visible radiation. The 80% hydrogen flame (bottom middle) is nearly imperceptible, even against a black steel background designed to enhance contrast. The 100% hydrogen flame (bottom right) is effectively invisible in daylight, confirming previous findings that hydrogen flames emit minimal visible radiation, Schefer, R W, et al,
44
Ellen E. Arens,
45
Zhao, Yan.
46
These observations underscore the limitations of visual flame confirmation in hydrogen-enriched combustion systems. To address this, the experimental setup incorporated a UV flame detection system. For optimal performance, the UV sensor was precisely aligned with the flame and shielded using a quartz window to protect it from direct exposure and thermal damage. This configuration, illustrated schematically in Figure 8, enabled reliable flame detection across all tested hydrogen concentrations. Visualisation of open flame for hydrogen enriched combustion. Flame Surveillance/Supervision Device (FSD) using UV cell for PHOTON10 in open setup experiments.
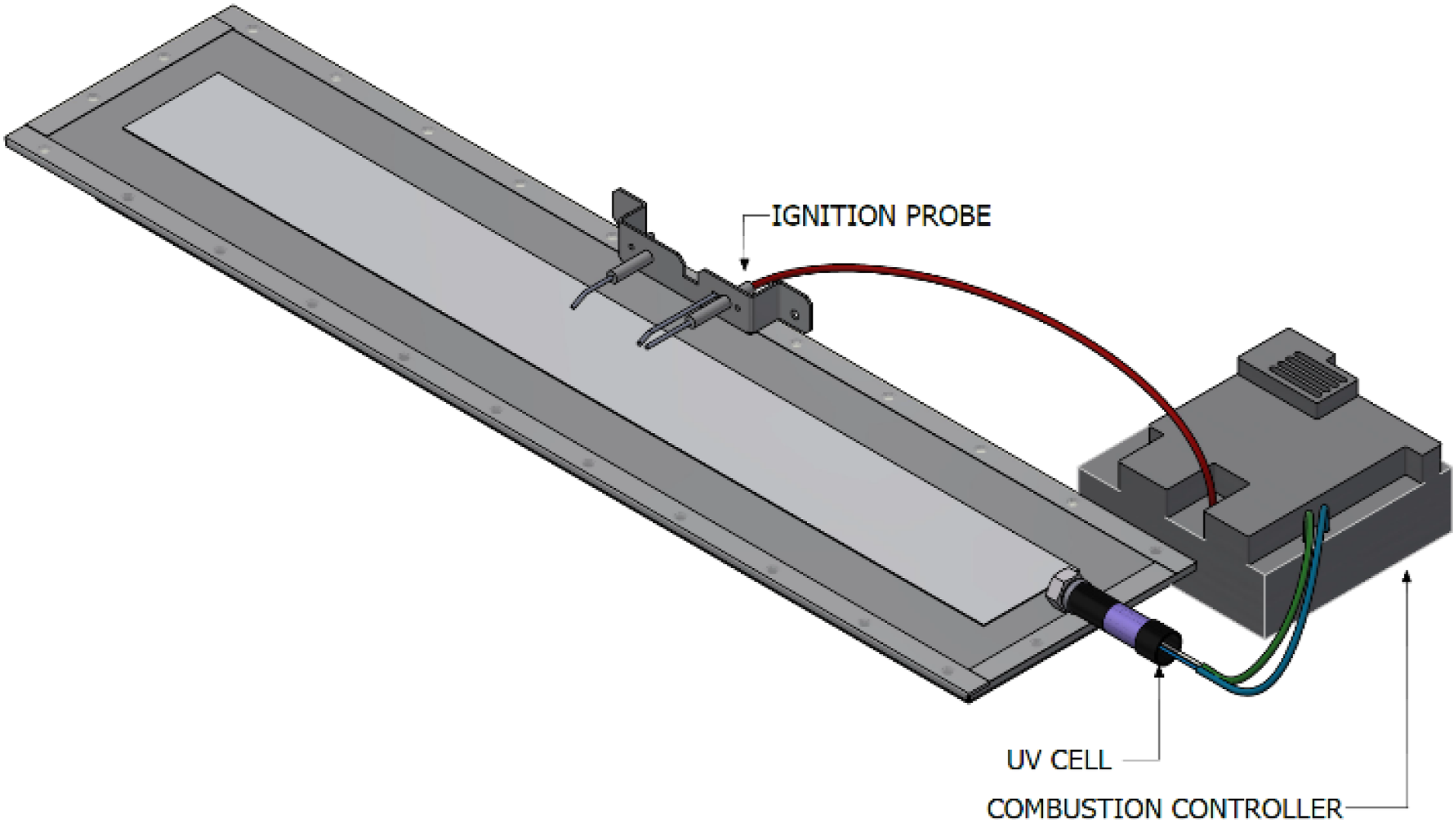
Additionally, an infrared (IR) imaging system was integrated to provide non-intrusive thermal visualisation of the flame. An Optris Xi 12 IR camera was employed to capture real-time thermal profiles during experiments conducted. The IR imagery revealed flame structure, size, and radiative intensity across hydrogen-methane blends ranging from 20% to 60% hydrogen. The mid-wavelength infrared spectrum offered enhanced contrast and detail, allowing for accurate assessment of flame stability and heat distribution—parameters that are not discernible in the visible spectrum. Together, these results highlight the necessity of advanced flame detection technologies in hydrogen combustion systems. The inability to rely on visual cues for flame presence, particularly at high hydrogen concentrations, necessitates the use of UV and IR-based diagnostics to ensure safe and effective burner operation.
Figure 9 presents a series of infrared (IR) images captured during open-flame trials using the PHOTON10 burner system, illustrating flame characteristics across a range of hydrogen-methane fuel mixtures. The top row of images corresponds to lower hydrogen concentrations (0% to 40% H2), while the bottom row represents higher hydrogen content (60% to 100% H2). The IR images reveal that lower hydrogen-enriched mixtures (20–40% H2) produce larger and more thermally intense flames, which are clearly visible in the IR spectrum. These flames exhibit consistent morphology and well-defined thermal gradients, indicative of stable combustion and efficient heat release. In contrast, higher hydrogen-enriched mixtures (≥60% H2) result in smaller, less radiant flames with diminished visibility, even in the infrared range. This reduction in thermal signature is attributed to hydrogen’s lower emissivity and reduced radiative heat release compared to hydrocarbon-dominant mixtures. The 100% hydrogen flame displays minimal IR contrast, reinforcing the challenge of flame detection in hydrogen-rich combustion environments. These findings validate the utility of IR imaging as a diagnostic tool for hydrogen combustion systems, especially in scenarios where flame invisibility poses operational and safety risks. The comparison between Figure 7 and 9 further supports the viability of infrared cameras for visualising hydrogen flames, offering a reliable alternative to conventional visual inspection methods. Additionally, the combustion behaviour of hydrogen differs fundamentally from that of natural gas. Hydrogen ignites at elevated temperatures and possesses a lower dissociation energy, which is endothermic; however, the overall energy release during combustion is exothermically superior. The flame propagation velocity of hydrogen-enriched mixtures increases with hydrogen concentration, particularly under fuel-rich conditions (ϕ > 1). This phenomenon was observed during open-flame experiments, where flame height varied with hydrogen content. The interplay between gas exit velocity and flame propagation rate—higher for hydrogen than methane—directly influences flame geometry and stability. Moreover, flame temperature fluctuations were noted, dependent on the combustion mode (premixed, non-premixed, or diffusion), further emphasising the complexity of hydrogen combustion dynamics. Infrared visualisation of open flame for hydrogen enriched combustion.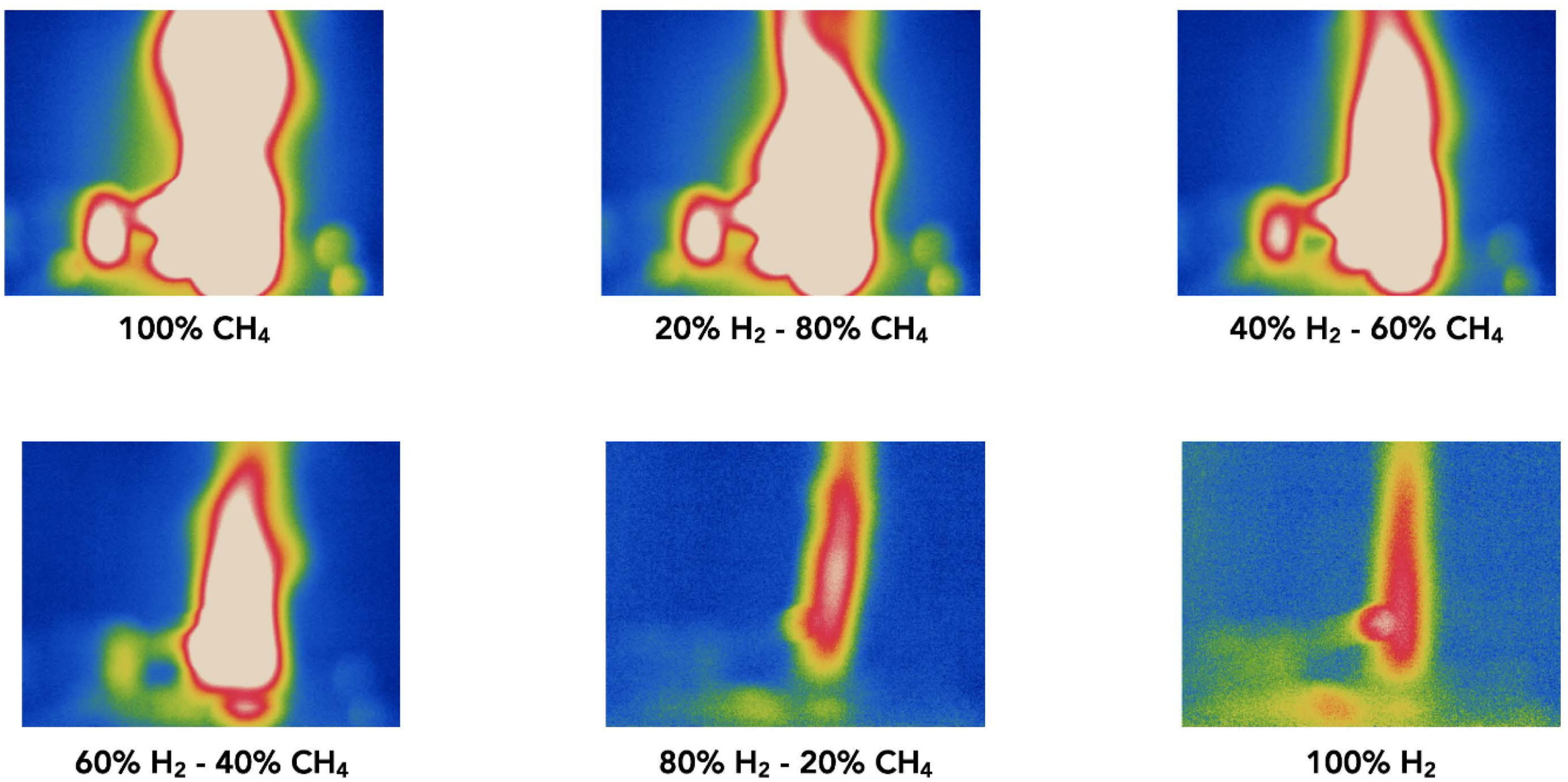
Figure 10 shows the flame temperature profiles measured at two transverse distances—2 cm and 5 cm—from the burner surface across a range of hydrogen concentrations. The data were obtained using Type K thermocouples strategically positioned to capture spatial thermal gradients during combustion. The results reveal a consistent trend: as the hydrogen percentage in the fuel mixture increases, the flame temperature decreases at both measurement points. Additionally, temperatures recorded at 2 cm are uniformly higher than those at 5 cm, indicating a steep thermal gradient and localised heat concentration near the burner exit. These findings are critical for understanding the thermal behaviours of hydrogen-enriched combustion and its implications for burner design and safety. Hydrogen’s lower emissivity and distinct combustion characteristics—such as high flame speed and reduced radiative heat release—contribute to the observed decline in flame temperature with increasing hydrogen content. This reduction in thermal intensity is particularly pronounced beyond 60% hydrogen, aligning with previous observations of diminished flame visibility and radiative output. Flame temperature measured at transverse distances of 2 cm and 5 cm from the burner surface during open setup for hydrogen-enriched conditions.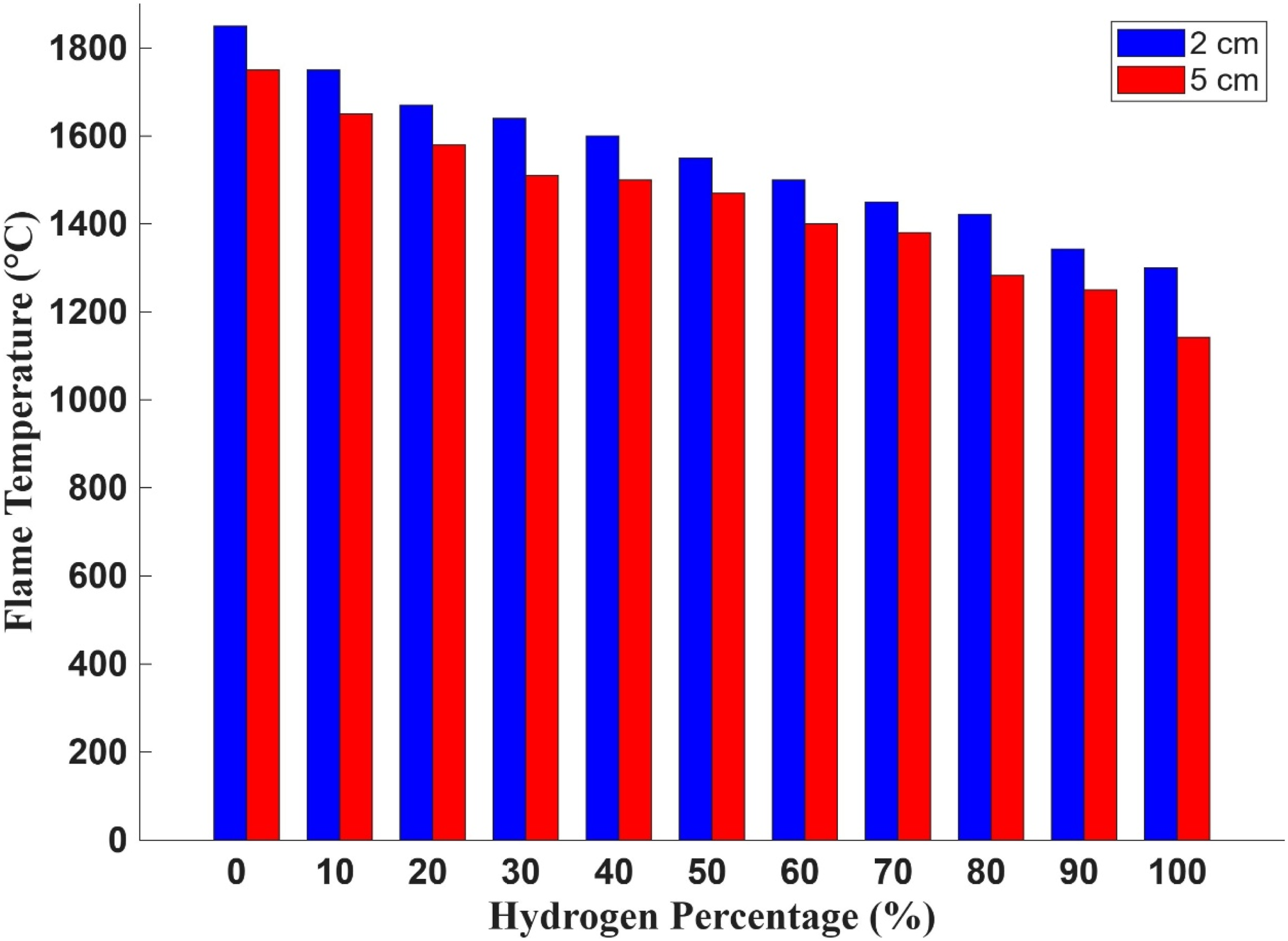
A fundamental consideration in premixed combustion systems is the relationship between the laminar flame speed and the injection velocity of the fuel–air mixture. For stable operation, the injection velocity must exceed the flame propagation speed to prevent flashback, a hazardous condition where the flame travels upstream into the burner’s mixing chamber. Hydrogen’s significantly higher laminar flame speed compared to methane increases the risk of flashback, especially at elevated hydrogen concentrations. This necessitates precise control over fuel injection dynamics and burner geometry to maintain safe operating conditions. The temperature profiling from Figure 10 provides essential insights into the operational limits of the PHOTON10 burner. Understanding how hydrogen blending affects flame temperature and size is crucial for evaluating power output performance and ensuring safe operation. These findings contribute to the broader effort to develop hydrogen-compatible heating systems and support the transition toward low-carbon industrial combustion technologies. Understanding the spatial distribution of flame temperature and the influence of hydrogen on flame propagation is essential for developing safe, efficient, and scalable hydrogen heating technologies.
Figure 11 illustrates the power output performance of the PHOTON10 burner system during open-flame trials across a range of hydrogen-enriched fuel mixtures. The graph presents a clear inverse relationship between hydrogen concentration and burner power output. As the hydrogen percentage in the fuel blend increases from 0% to 100%, the power output decreases progressively, with the most significant reduction observed at full hydrogen combustion. The data indicate that the power output from 100% hydrogen combustion is approximately three times lower than that achieved with 100% methane (natural gas). While hydrogen possesses a high gravimetric energy density, its lower volumetric energy content and reduced radiative heat release result in diminished flame intensity and heat transfer efficiency. These characteristics directly impact the burner’s ability to sustain high thermal output under hydrogen-rich conditions. One can increase hydrogen fuel supply to overcome the shortfall of this power output. The observed trend underscores the importance of evaluating burner performance across different fuel compositions, particularly in the context of transitioning from natural gas to hydrogen. Understanding the power output limitations associated with hydrogen-enriched mixtures is essential for industrial applications where consistent thermal performance is critical. These findings also highlight the need for burner design adaptations, such as increased fuel flow rates, optimised air-fuel mixing, and enhanced heat recovery systems which further compensate for the reduced energy output of hydrogen combustion. Measured thermal power output of the PHOTON10 burner under varying hydrogen-enriched fuel mixtures in open setup experimental.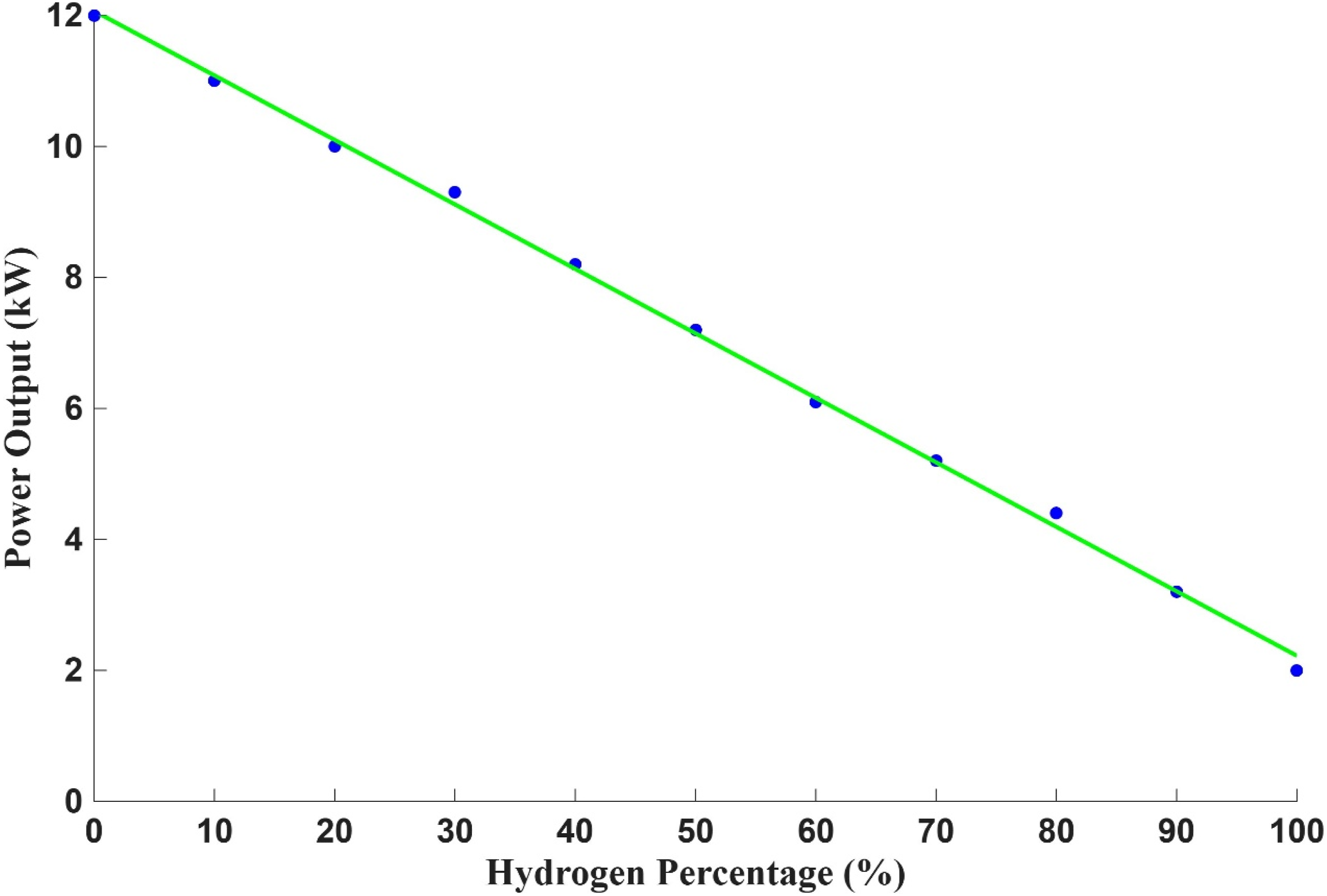
Figure 12 presents a comparative analysis of the power output distribution of the PHOTON10 burner when operating with 100% hydrogen fuel under two distinct fuel intake scenarios. The pie chart illustrates a significant disparity in thermal performance: at the reference flow rate (equivalent to the standard methane intake), the burner achieves only 25% of its rated power output. In contrast, when the hydrogen intake is increased to three times the reference rate (x3 L/min), the burner recovers 75% of its nominal power output. This outcome underscores a critical challenge in hydrogen combustion—its lower volumetric energy density compared to methane necessitates substantially higher fuel flow rates to achieve equivalent thermal energy delivery. These findings have important implications for burner manufacturers and system designers. Maintaining consistent thermal performance across varying fuel compositions requires adaptive fuel delivery mechanisms, including enhanced flow control and pressure regulation systems. The PHOTON10 burner, in its current configuration, demonstrates the capacity to operate with hydrogen, but only under modified intake conditions. Without such adjustments, the system experiences a marked decline in power output, as evidenced by the reference flow scenario. Power distribution at 100% hydrogen to compensate energy outout.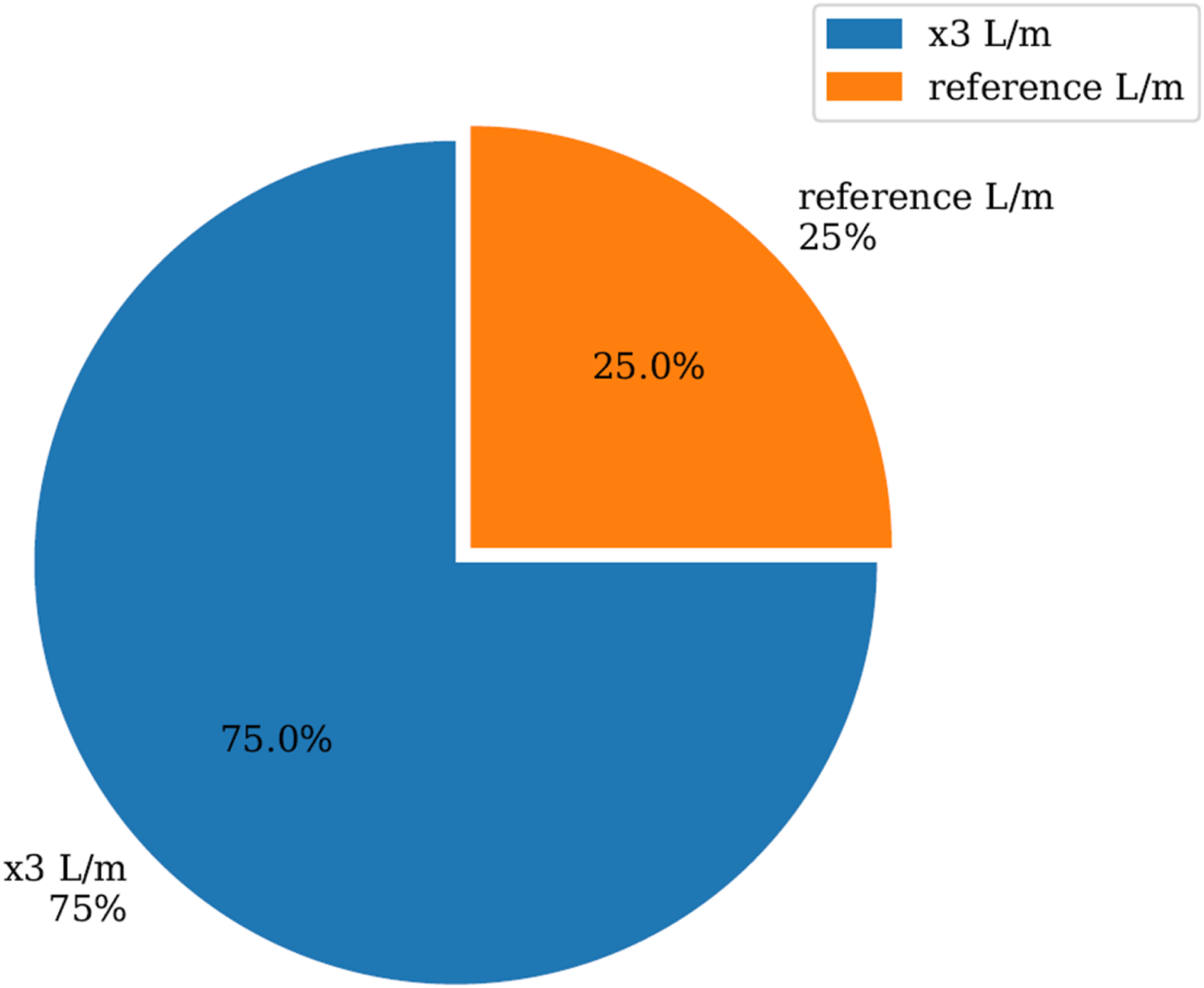
The transition from methane to hydrogen in industrial combustion systems is further complicated by the need to balance efficiency, safety, and emissions. While hydrogen offers significant environmental advantages—such as reduced CO2 and NOx emissions (results discussed in Figure 13–16) —its combustion characteristics differ fundamentally from those of natural gas. Hydrogen’s high flame speed, low emissivity, and reduced radiative heat release affect flame stability, heat transfer, and detection reliability. These factors must be carefully considered in burner design and control strategies. It is also important to note that the results presented in Figure 12 were obtained under open-flame experimental conditions, which may differ from closed-unit performance due to variations in thermal insulation, airflow dynamics, and flue gas recirculation. The subsequent section of this study addresses these variables through closed-unit testing, offering a more comprehensive understanding of hydrogen combustion behaviour under realistic operating conditions. Carbon monoxide CO emission profile of the PHOTON10 burner under enclosed setup with varying hydrogen-enriched fuel mixtures. Carbon dioxide CO2 emission profile of the PHOTON10 burner under enclosed setup with varying hydrogen-enriched fuel mixtures. Nitric oxide NO emission profile of the PHOTON10 burner under enclosed setup with varying hydrogen-enriched fuel mixtures. Nitrogen oxide NOx emission profiles of the PHOTON10 burner under enclosed setup with varying hydrogen-enriched fuel mixtures.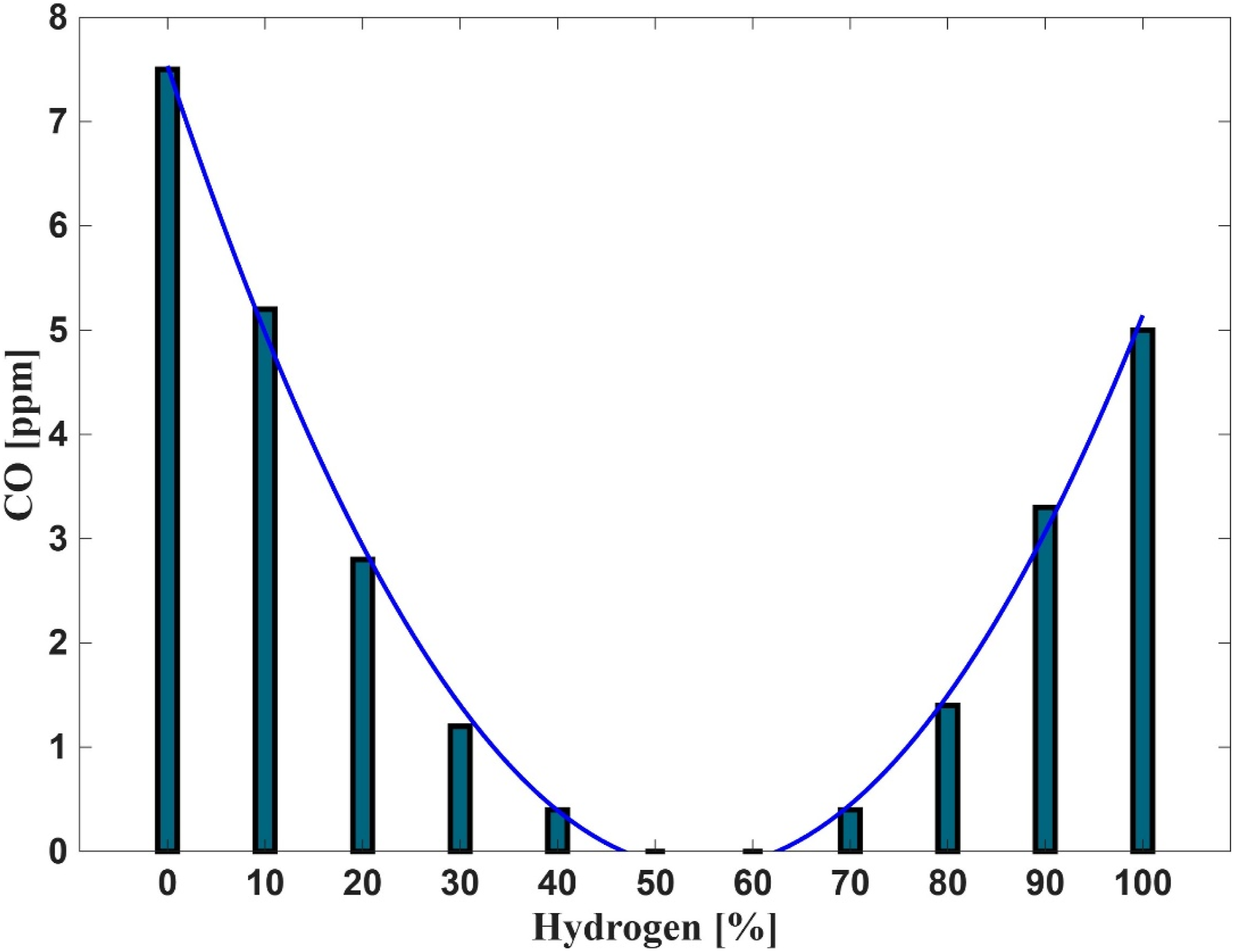
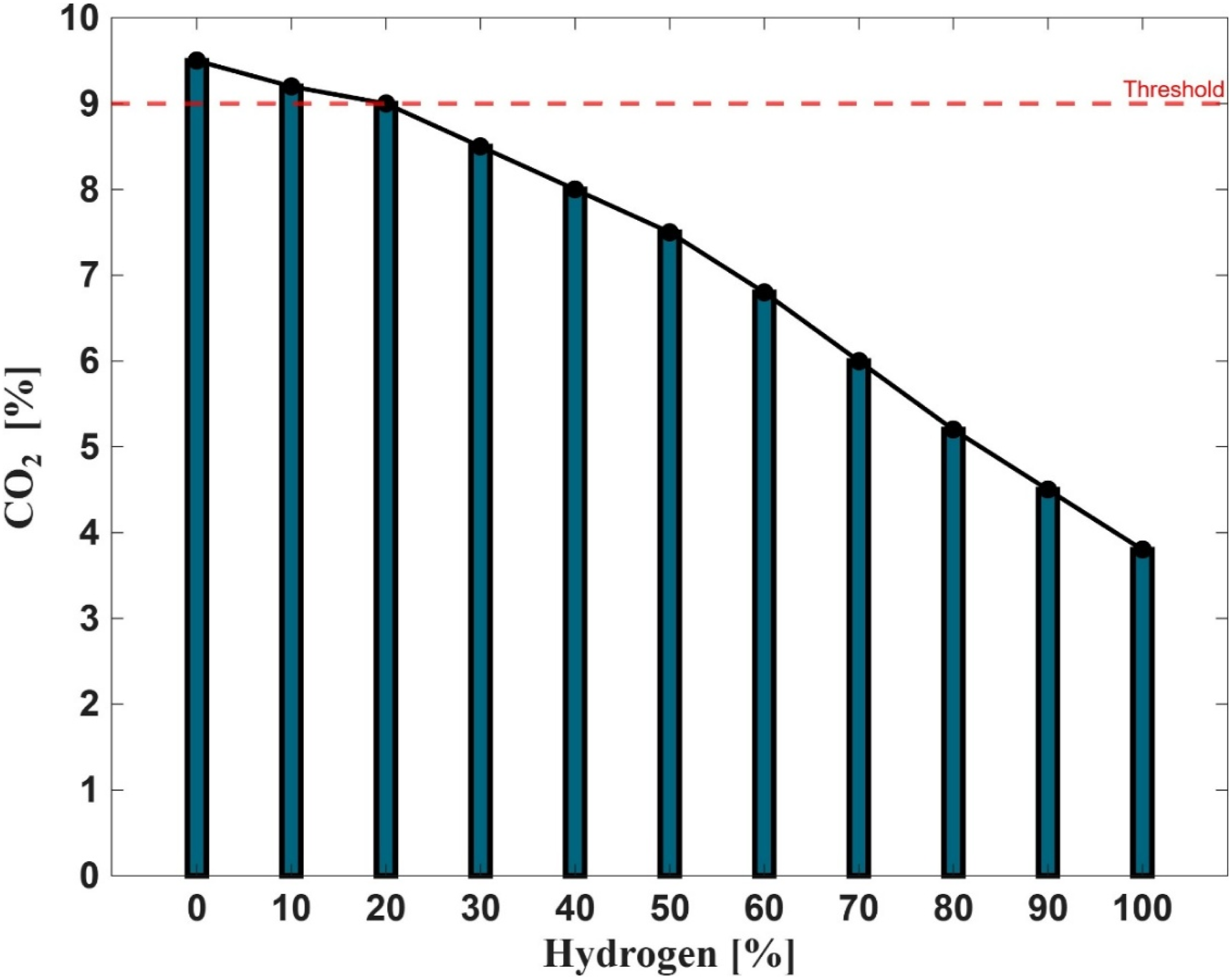
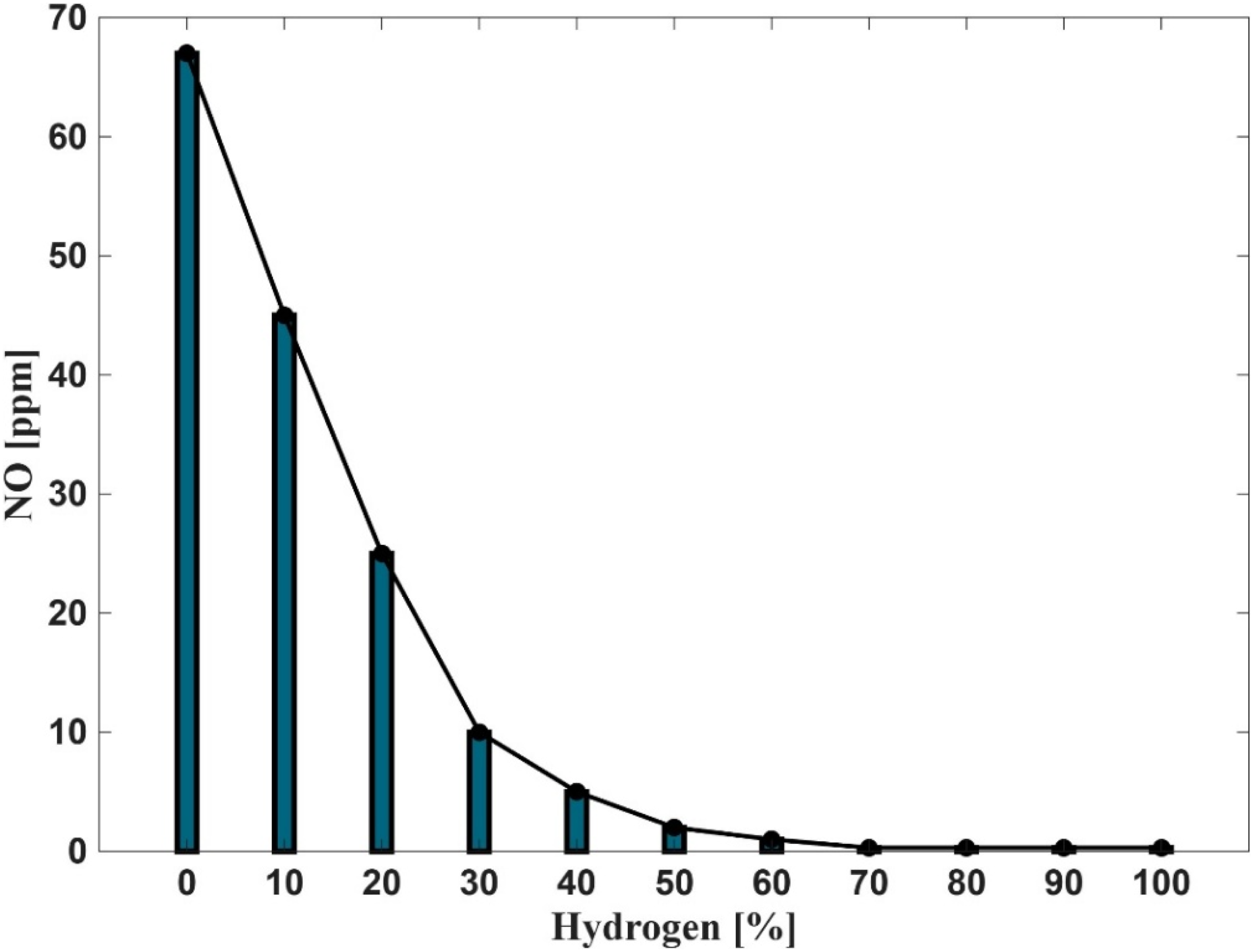
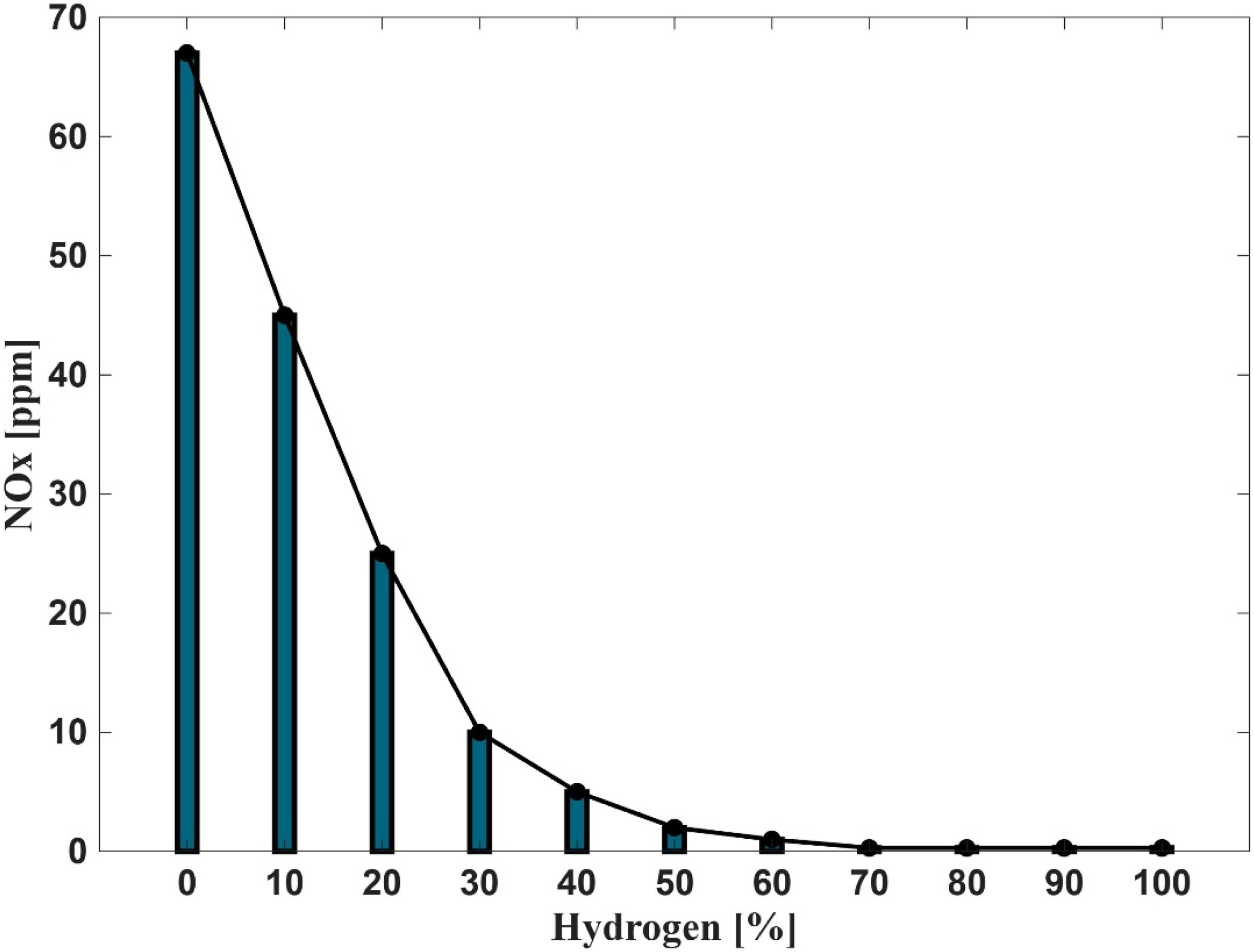
Figure 13 shows the measured carbon monoxide (CO) concentrations during closed-unit experiments using the PHOTON10 burner across a range of hydrogen-enriched fuel mixtures. The data reveal a U-shaped trend in CO emissions. At low hydrogen concentrations (0–20%), CO levels are elevated. As hydrogen content increases to moderate levels (20–80%), CO emissions decline significantly, falling well below the threshold. However, at high hydrogen concentrations (≥80%), CO levels begin to rise again, approaching values like those observed at the lower end of the hydrogen spectrum. This behaviour is closely linked to the thermal and chemical characteristics of hydrogen-enriched combustion. Under fuel-rich conditions (ϕ > 1.65), particularly at 65% hydrogen, CO formation increases due to reduced flame temperatures (see Figure 3 for 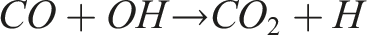
It is important to note that, although the measured CO levels at ∼10% and ∼90% H2 are comparable, the mechanisms differ. At low H2 (CH4-dominant), CO persists where local temperature and OH availability is marginal in lean/near-lean pockets and residence time is insufficient for complete post-flame burnout, consistent with kinetic trends that minimize CO at stoichiometry and increase it away from the optimum. At high H2, the reaction zone becomes more compact and post-flame residence time shortens; any mildly fuel-rich or rapidly cooled regions again limit the temperature-sensitive CO + OH → CO2 + H pathway, yielding residual CO despite a high overall reactivity. Additionally, concentration-based (ppm) reporting can mask mass-normalized differences when fuel composition changes; output-normalized metrics (e.g., ng/J) are preferred because ppm can conflate residence-time and dilution effects in H2-enriched flames.27,47
Figure 14 illustrates the impact of hydrogen-enriched fuel combustion on carbon dioxide (CO2) emissions, based on experimental data obtained from closed-unit trials using the PHOTON10 burner system. The graph presents both measured CO2 concentrations (green bars) and a trend line (blue) across varying hydrogen percentages, with a red dashed horizontal line indicating the reference CO2 level associated with pure methane combustion (∼8.75 %). The results demonstrate a clear inverse relationship between hydrogen concentration and CO2 emissions. As the hydrogen content in the fuel mixture increases, CO2 concentrations consistently decline. Notably, all hydrogen-enriched scenarios tested yielded CO2 levels well below the reference threshold, confirming the effectiveness of hydrogen integration in reducing carbon-based emissions. This trend aligns with broader decarbonisation goals and reinforces hydrogen’s potential as a cleaner alternative to conventional hydrocarbon fuels.48–50
However, this reduction in CO2 is accompanied by a trade-off in carbon monoxide (CO) emissions, as previously discussed in Figure 13. Under fuel-rich conditions and lower flame temperatures, the oxidation of CO to CO2 is inhibited due to reduced availability of hydroxyl (OH) radicals. The reaction: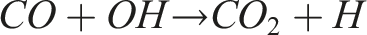
Figures 15 and 16 illustrate the influence of hydrogen enrichment in natural gas on nitric oxide (NO) and Nitrogen oxides (NOx) emissions respectively during combustion. The data reveal a clear inverse relationship between hydrogen concentration in the fuel mixture and NO formation. At 0% hydrogen, NO emissions are approximately 65 ppm, but a sharp decline is observed as hydrogen content increases to 20%, beyond which the reduction continues at a slower rate. When hydrogen exceeds 50%, NO emissions become almost negligible, and at 80–100% hydrogen, they approach zero. This trend underscores the significant role of hydrogen in mitigating NOx emissions.
The observed reduction in NO formation can be explained by the thermal and chemical dynamics governed by the extended Zeldovich mechanism, which primarily controls thermal NO production. The key reactions involved are: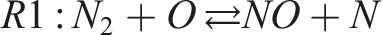
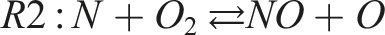
Reaction R1 requires a high activation energy (approximately 319,050 kJ/kmol), making it highly temperature dependent. As hydrogen enrichment increases, the overall flame temperature decreases, especially under fuel-rich conditions, which suppresses the formation of reactive radicals (O, N, OH) essential for NO production. Furthermore, the reduced molar fraction of oxygen in fuel-rich mixtures limits the availability of oxidizing agents, compounding the suppression of NO formation. The duality of NO and NO2 (collectively NOx) is also influenced by these conditions. Lower flame temperatures and reduced oxygen availability hinder the conversion of NO to NO2, resulting in an overall reduction in NOx emissions. These findings confirm that hydrogen enrichment is an effective strategy for reducing NOx emissions in combustion systems.
Figure 17 illustrates the variation of flue gas temperature with increasing hydrogen content in the fuel mixture. The trend shows that at 0% hydrogen, the flue gas temperature is at its highest, indicating efficient combustion of natural gas under baseline conditions. As hydrogen enrichment begins, the temperature initially remains relatively high, suggesting improved combustion stability and completeness at low hydrogen levels. However, beyond a certain threshold (around 30–40% hydrogen), a progressive decline in flue gas temperature is observed, with values dropping below 150°C at 100% hydrogen. This reduction in flue gas temperature at higher hydrogen concentrations can be attributed to several factors: (1) Shift in Combustion Characteristics: Hydrogen has a higher diffusivity and flame speed compared to methane, which can alter flame structure and heat release patterns. At high enrichment levels, the system tends toward fuel-rich conditions, as indicated by elevated equivalence ratios (ϕ), reducing overall combustion efficiency. (2) Lower Heat Release: Although hydrogen has a high heating value per unit mass, its lower volumetric energy density and the reduced oxygen availability in fuel-rich mixtures limit the total heat released during combustion. (3) Impact on NOx Formation: The observed temperature decline correlates with the reduction in NO and NOx emissions discussed in Figures 15 and 16. Lower flame temperatures suppress the formation of reactive radicals (O, N, OH), which are essential for NO formation via the extended Zeldovich mechanism. These findings highlight a trade-off between emission reduction and thermal efficiency. While hydrogen enrichment effectively reduces NOx emissions, excessive hydrogen content can compromise heat recovery and overall system performance. Therefore, optimising hydrogen concentration is essential to balance low emissions, stable combustion, and high thermal efficiency in practical applications. Flue gas temperature profiles of the PHOTON10 burner under enclosed setup with varying hydrogen-enriched fuel mixtures.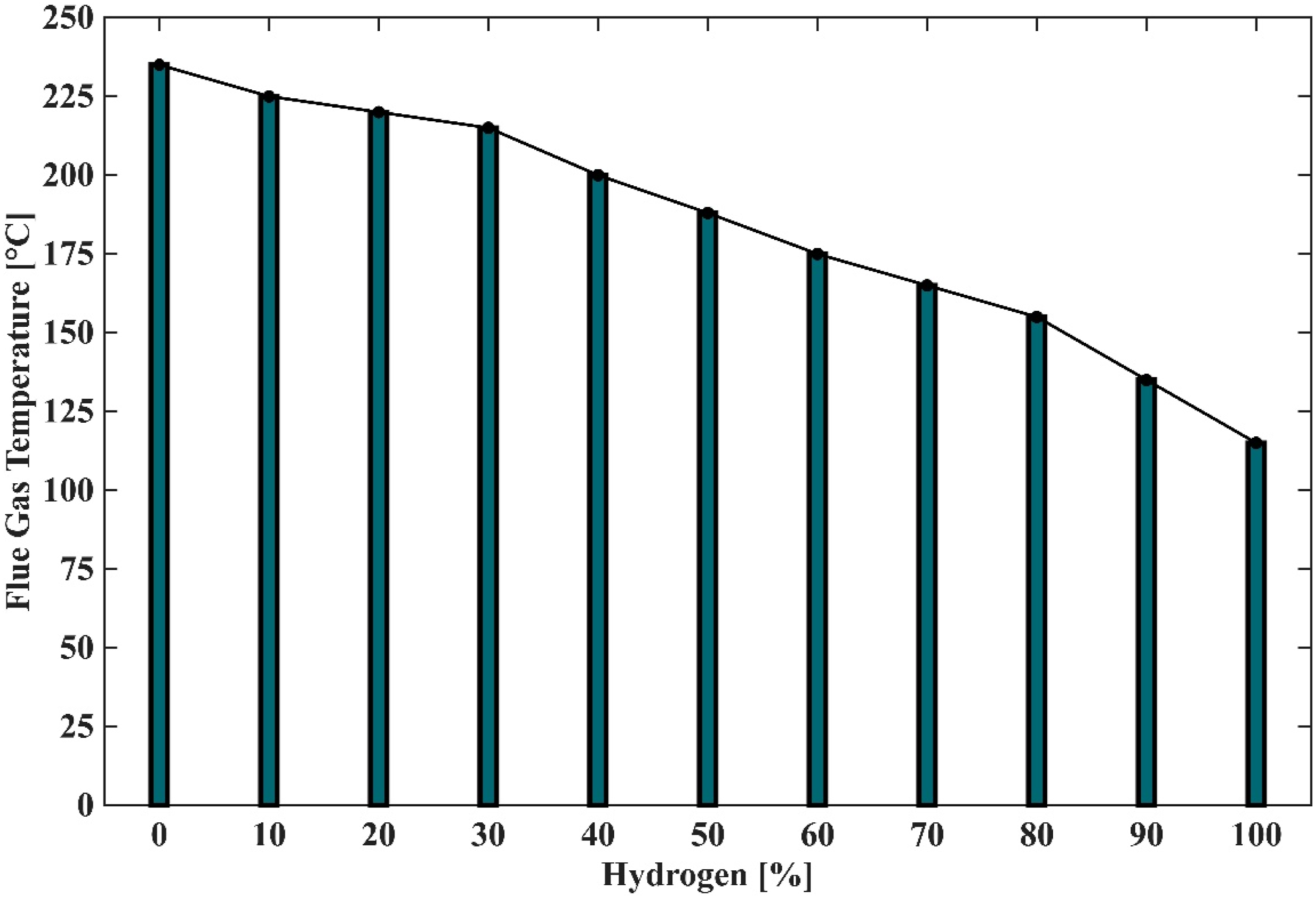
Conclusions
This study has demonstrated both the technical feasibility and the operational boundaries of hydrogen enrichment in a legacy natural-gas burner (PHOTON10) operated without modification to its existing safety-control configuration.
Safe operating window (controls unchanged) - Hydrogen blending up to 50% vol. was achieved with stable flames and reliable operation of the original flame rectification system. Above this threshold, flame instability and intermittent flame detection occurred, attributed to hydrogen’s lower ionization propensity and different combustion kinetics.
Enhanced sensing for hydrogen-rich operation - A UV flame sensor provided robust detection of hydrogen-rich flames where rectification failed, improving reliability for safety-critical use. Because hydrogen-enriched flames exhibit low visible luminosity, IR thermography (Optris Xi 12) was essential for thermal visualization and continuous monitoring.
Flame structure and stability from IR diagnostics (20–60% H2) - IR imaging revealed that 20–40% H2 blends produced larger, hotter, and more stable flames, whereas ∼60% H2 led to smaller, less radiant, and less stable flames. These trends are consistent with hydrogen’s higher flame speed, lower emissivity, and altered heat-release distribution. The results reinforce the necessity of maintaining injection velocity above the local flame speed to avoid flashback—a risk that increases with H2 content. Type-K thermocouple measurements corroborated the thermal fields inferred from IR data.
Thermal performance and power output - While low hydrogen fractions can support complete combustion and higher initial flue-gas temperatures, increasing H2 above ∼40–50% resulted in lower flame and flue-gas temperatures and shorter flames, reflecting reduced radiative output and shifts in combustion regime. In constant-fuel-intake tests, 100% H2 delivered approximately one-third the thermal power of pure methane in the PHOTON10, implying that ∼3× higher volumetric fuel flow is required to match methane’s rated power—an important constraint for fuel delivery hardware and control valves.
Emissions characteristics –
CO2
All hydrogen-enriched cases exhibited CO2 below the ∼8.7 ppm value measured for pure methane, supporting hydrogen’s role in carbon-intensity reduction.
CO
Under fuel-rich operation (e.g., ϕ > 1.65 at ∼65% H2), CO increased from <2 ppm to ∼6 ppm, linked to lower temperatures and reduced OH radical levels that slow the CO + OH → CO2 + H pathway—i.e., incomplete oxidation.
NO/NOx
NO and NOx decreased monotonically with hydrogen fraction; >50% H2 yielded near-negligible NOx, consistent with suppressed Zeldovich thermal NO due to lower flame temperatures and reduced radical pool (O, N, OH).
Practical implications –
Controls and safety
PHOTON10 systems can safely accommodate ≤50% H2 without control redesign, but hydrogen-rich operation should adopt UV-based flame detection and incorporate IR monitoring for diagnostics and condition-based maintenance.
Hardware and fuelling
To maintain nameplate power on high-H2 or 100% H2, systems require larger flow capacity, re-sized nozzles/orifices, and re-tuned control valves to handle the higher volumetric flow demanded by hydrogen’s lower volumetric energy density.
Operations
Avoid sustained fuel-rich conditions at high H2 fractions to limit CO slip; implement
Emissions strategy
Hydrogen blending is a highly effective NOx-mitigation lever; however, ensuring complete oxidation (CO control) and sufficient thermal output requires balanced staging and mixture control.
Limitations and future work –
The current study results are specific to a closed-unit PHOTON10 configuration and may vary with burner geometry, mixing strategy, and scale. The future research should investigate (1) co-optimization of injector design, swirl/mixing, and air–fuel ratio control for higher H2 fractions, (2) advanced sensors (UV, IR, ion-current, OH* chemiluminescence) for multi-modal flame detection and health monitoring; (3) flashback-resistant hardware (flashback arrestors, porous/micro-mixers) and dynamic control to maintain injection velocity margins; and (4) integrated emissions–efficiency trade-off models to navigate NOx, CO, CO2, and power simultaneously.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

