Abstract
Thermal management is a crucial design aspect for the safety and performance of the Lithium-ion battery pack used in electric vehicles. Phase change material (PCM) based battery thermal management is one of the appropriate passive or hybrid thermal management. The present paper highlights the selection of PCM depends on various factors related to application operating conditions like ambient temperature, the heat released by the battery, and the type of battery pack design. In selecting PCM, a thorough understanding of various PCMs requires an overall review of multiple types and properties of organic and inorganic PCM, using phase change equilibrium diagrams to comprehend phase change transformations. Further, the present work summarizes the various studies on PCM durability, different thermal performance enhancement techniques, and methodology for selecting PCM; as a result, assisting the researchers and engineers in choosing the appropriate PCM for the various EV battery thermal management applications in terms of chemical, thermal, and economic aspects
Keywords
Highlights
• Detailed review on physical, chemical and thermal properties of various PCM materials • The aspects of PCM material durability and compatibility are elaborated • To characterize the PCM materials, Phase change equilibrium diagram is a very useful tool • A Novel PCM selection methodology proposed with respect to Electric vehicle application
Introduction
In recent years, the world has been taking measures to reduce pollution by controlling emissions of Greenhouse gases (GHG) by depending mainly on renewable energy sources. To mandate the former initiative, all countries signed a Paris climate agreement that aims to decrease global warming by maintaining temperature rise below 2°C. One of the initiatives countries take to reduce pollution is to deploy electric vehicles (EV) through subsidy schemes. EVs and renewable energy will pave the way for a strong foothold in decreasing pollution.
At present, Lithium-ion batteries (LIB) are found to be a suitable energy source for electric vehicles due to their potential of having a high energy density (Wh/kg) and its extended life span, and also energy on the vehicle accounts for 30%–40% of overall vehicle cost. Therefore, Maximizing the life of the battery and maintaining safety of the battery are the critical issues to be addressed while building an electric vehicle to meet performance. The safety of the battery is ensured by the proper electrical design, structural design, and thermal design around the cells in a pack and mainly it can be achieved by suitable thermal management systems. Since the battery is sensitive to temperature and demands a need of strict thermal control to be in a temperature range between 15°C to 35°C
1
approximately. Operating outside the mentioned range will lead to the following adverse effects. (Figure 1) Temperature effect on lithium-ion battery.
2

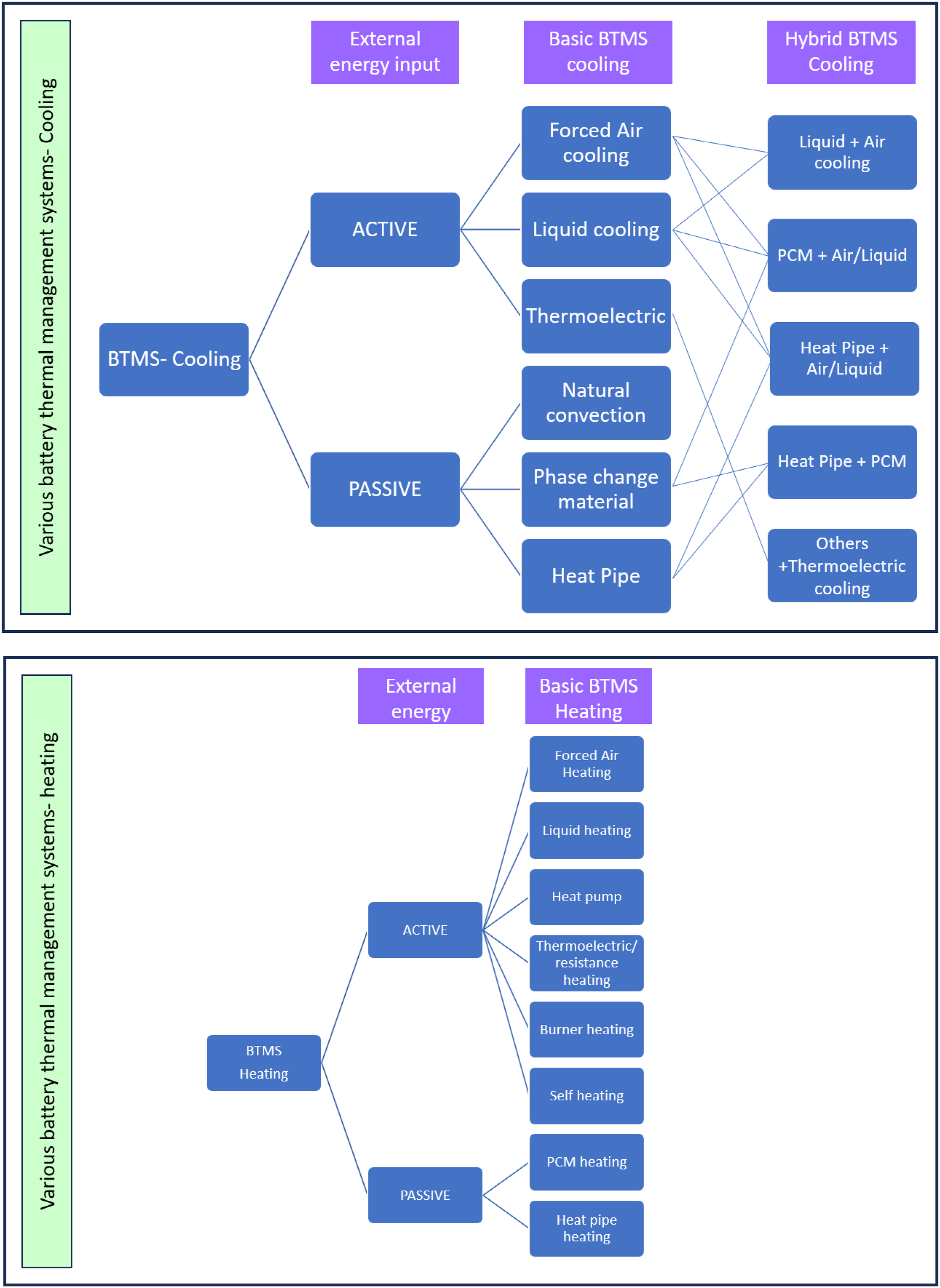
Most safety-related consequences can be controlled by using the proper battery thermal management systems (BTMS). Various thermal management technologies are used globally, as seen in Figure 2. Primarily, classification is based on external power input, namely active BTMS and passive BTMS. In active BTMS, with the help of external power, the cooling operation will be carried out. Whereas, in passive BTMS, without external power, cooling will be done by phase change and heat pipe equipment.
5
PCM-based battery thermal management is passive thermal management systems that are economical and environment-friendly BTMS. Phase change materials (PCM) that absorb or release heat during the phase transition processes is a promising alternative to the active cooling system for battery thermal management. The goals of proper BTMS are like temperature uniformity from cell to cell can be achieved by the PCM/enhanced PCM with Graphite and shows greater efficiency than air cooling, and helps to maintain the optimum temperature range for an individual cell to achieve more significant life. PCM offers the dual quality of buffering and conducting with the simplest design. Thus, in the advancements of Li-ion BTMS, the PCM-based BTMS became a crucial component. Even in environments with high ambient temperatures of 45°C to 50°C, PCM-based thermal management keeps the battery pack's internal temperature well below the prescribed limit. PCM-based BTMS protects the system with high latent heat and thermal conductivity in stressed conditions. For example, at the ambient temperature of 45°C and discharge current rate of 6.67C, the maximum temperature difference between the cells is not increased beyond 0.5°C. In casual operating conditions, the maximum temperature differences are negligible.
6
A PCM based passive thermal management system makes it possible to achieve uniform temperatures under abnormal/stressed and normal conditions. Passive thermal management also provides better safety than the complexity involved in active cooling systems. The enhanced PCM, like the PCM-Graphite matrix can control the thermal runaway by conducting and absorbing the heat within the cells. In Plugin hybrid electric vehicle's PCM based technology for cooling has become an excellent alternative to forced air cooling with compact packs. 7
The PCM shows excellent efficiency even in cold conditions and space applications. The PCM will absorb the excess heat, When the battery module temperature is more than the melting point temperature of the PCM and release it to the atmosphere when the ambient temperature is lesser than the PCM melting point temperature. The structure of the PCM for these applications is very simple, space-saving, and lighter than the active BTMS systems. The PCM-based BTMS are available at cheaper cost and hence they become more attractive in the recent years.
BTMS related review articles.

A few other notable experimental works in the application of PCM thermal management, in particular to the PCM stability are Xiao et al. 13 synthesized a crosslinking polymeric structure having a crosslinking skeleton gives well-defined properties like high latent heat of 98.8 j/g in the absence of leakage and volume expansion. The skeleton structure showed high thermal stability under extremely high-temperature conditions. This polymetric structure will be helpful in the actual application without any comprise on lower thermal conductivity, Li et al. 14 designed a methodology of solvent-evaporation to obtain a new kind of flexible CPCM under room temperature by considering ethylene-vinyl acetate (EVA), cyclohexane as the supporting material and solvent useful for BTMS. This new methodology simplifies the preparation even under harsh environmental conditions. Flexible CPCM-EVA allows a more compact installation, decreasing the contact resistance between the cell surface. CPCM and EVA-PCM show excellent properties at room temperature, like anti-leakage properties and flexibility. In consequence, compared with CPCM, great improvement can be seen in temperature-control performance. Ye et al. 15 proposed a novel CPCM with dual phase change temperature regions. Dual temperature regions will be helpful to provide a suitable optimum working temperature range for the battery, indirectly enhancing the performance by controlling the maximum temperature under harsh working environments. As well as improving the stability of the material. At the same time, Lv et al 16 have made serpentine PCM layers which will leave the provision for secondary cooling by a fan, thus increasing the overall energy density of the module from 107.8 to 121.6 Wh/kg, Lv et al. 17 also proven that with PCM, the life of the module increased by 65.3%. The PCM application to the battery can extend to other energy storage like supercapacitors. Hasan et al. 18 experimentally designed the electrode materials by doping. The co-doped polyaniline chains and nitrogen-doped reduced graphene oxides improve the capacitive performance of the supercapacitors by having unique surface properties, high processability, cycle rate capability, and life cycle. After going through all the works of literature about various properties, PCM materials, and respective advantages and disadvantages, a novel methodology proposed on how to select the suitable PCM material for a given thermal application, which will be helpful for the engineers and researchers in the experimentation field.
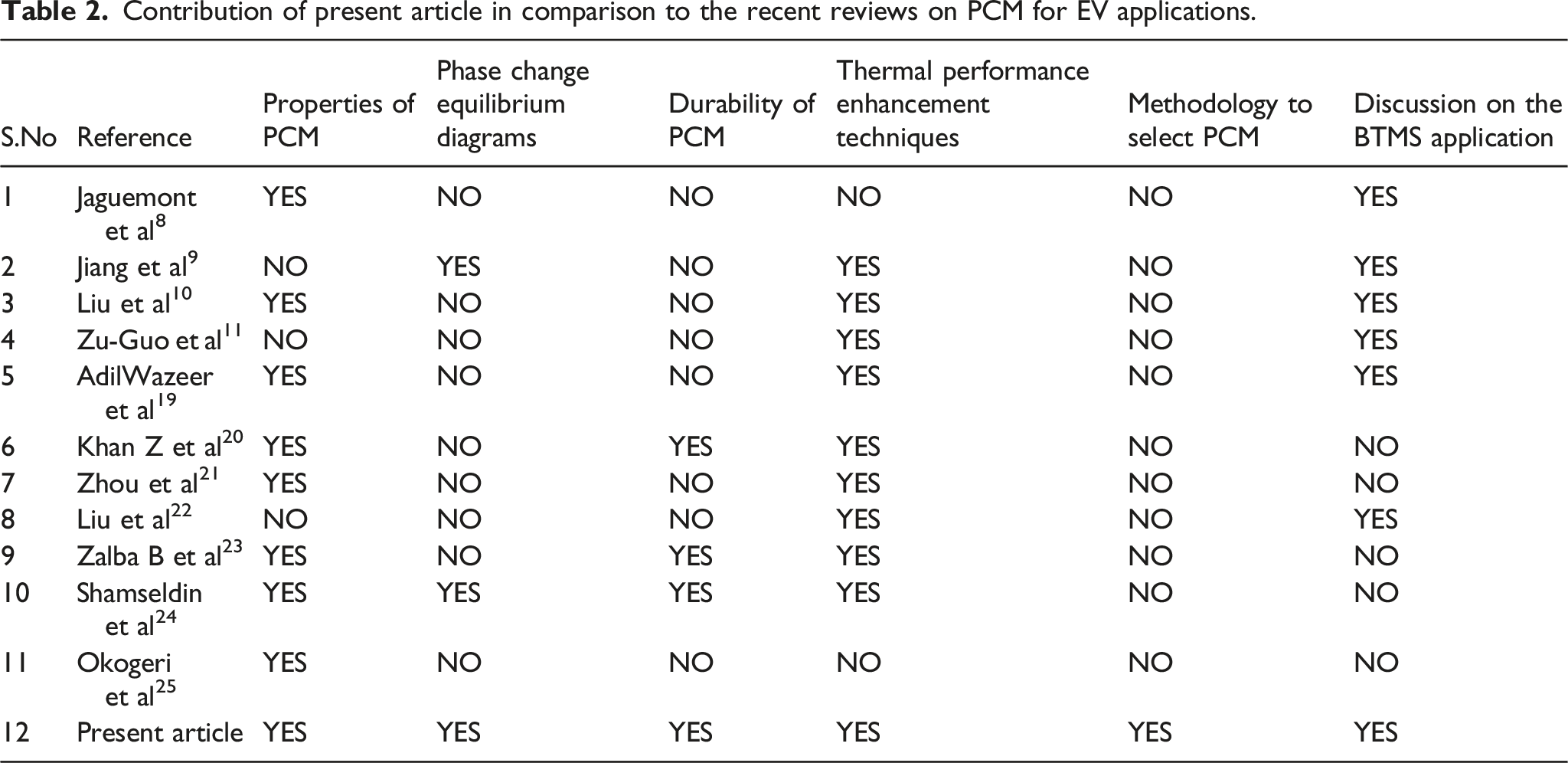
Contribution of present article in comparison to the recent reviews on PCM for EV applications.
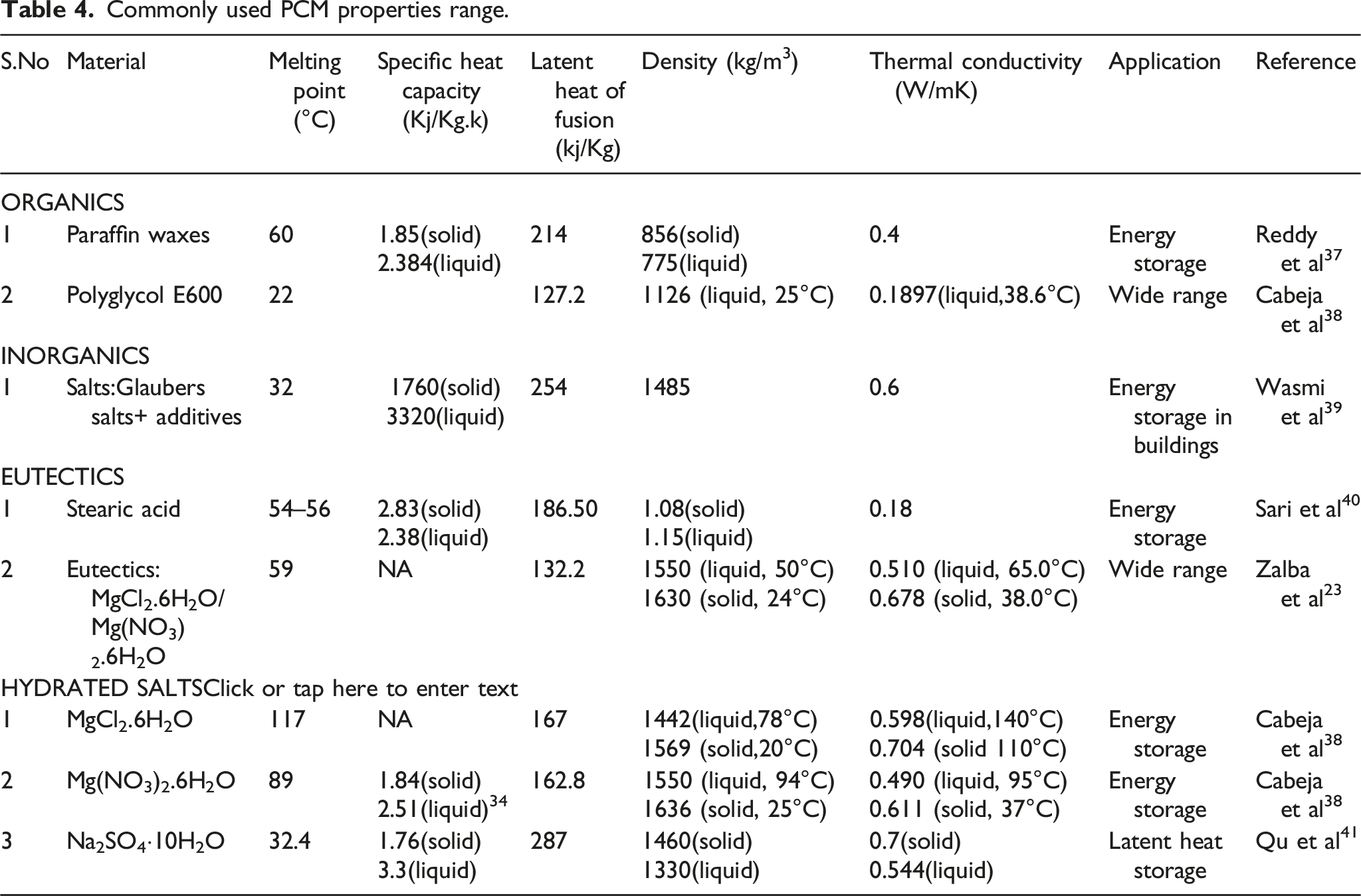
Commonly used PCM properties range.
Detail description of the various works on PCMs as storage material.
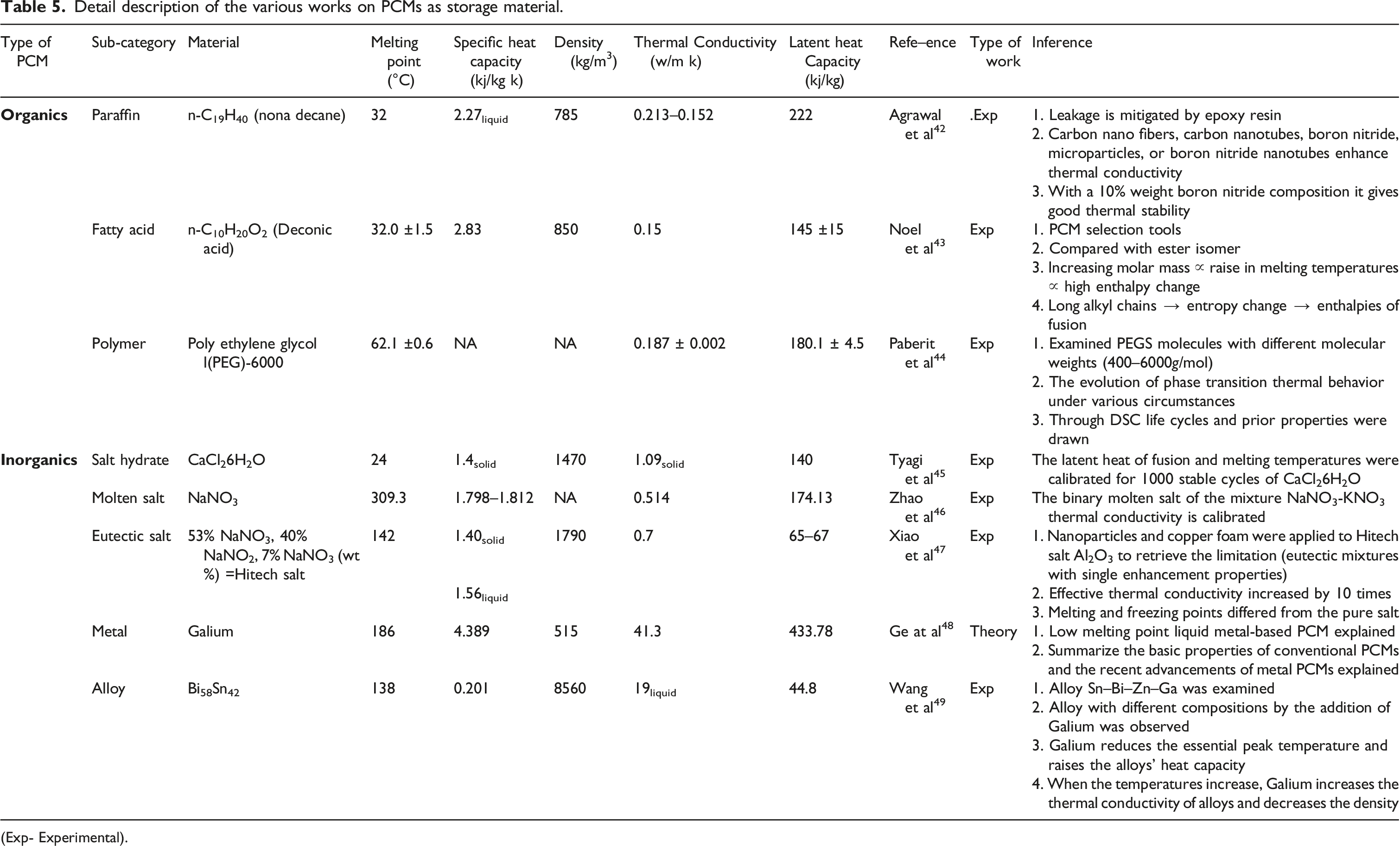
(Exp- Experimental).
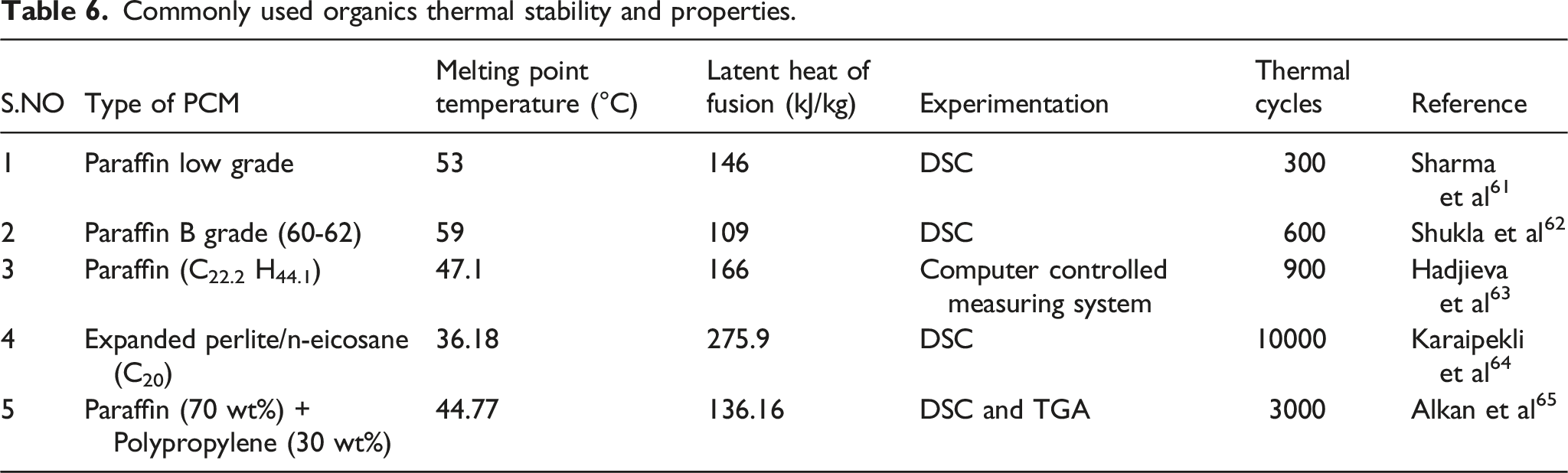
Commonly used organics thermal stability and properties.
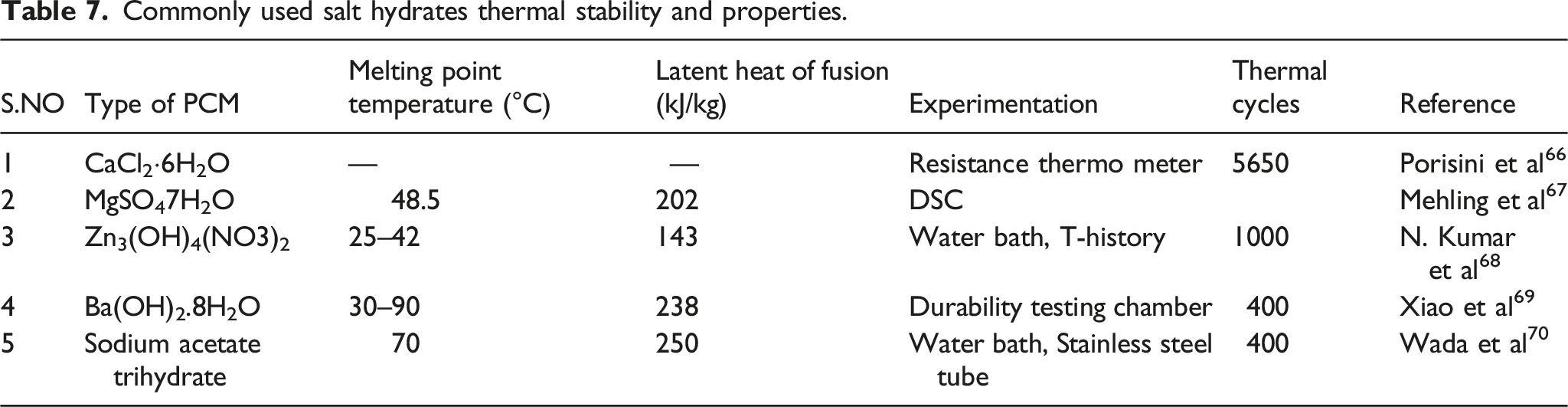
Commonly used salt hydrates thermal stability and properties.
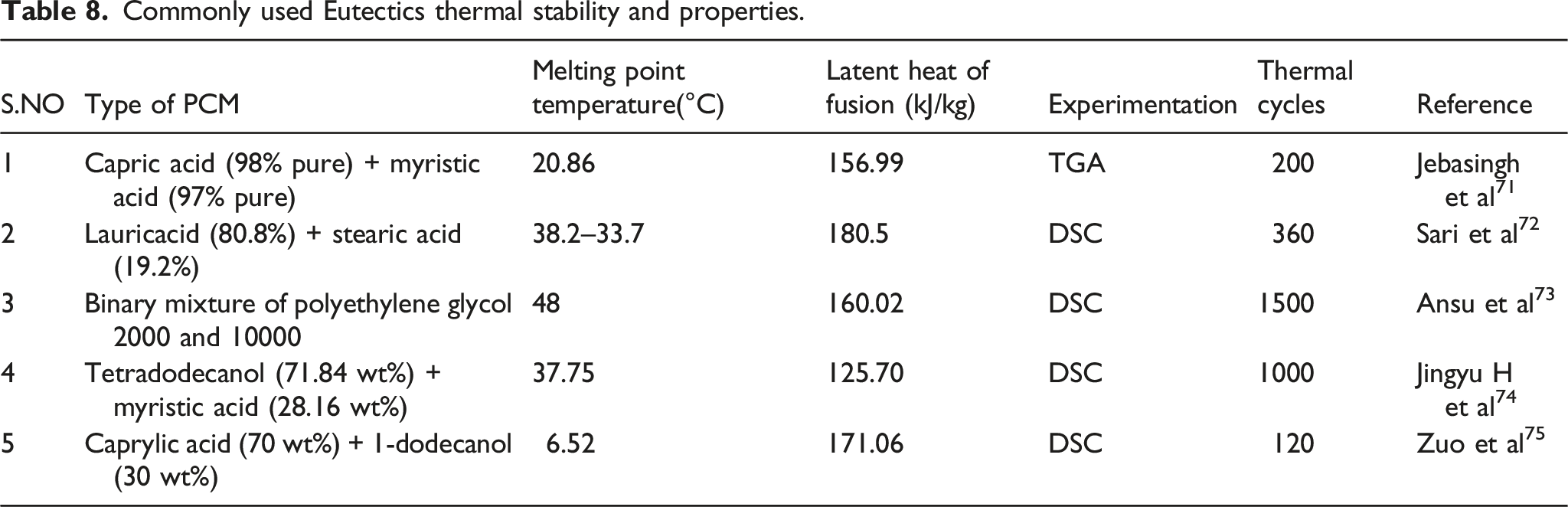
Commonly used Eutectics thermal stability and properties.
Details about phase change materails
The latent heat storage system stores or releases energy with the help of PCM, which can store lots of energy. Their high energy storage potential got activated while phase changing at the isothermal condition. Unlike other materials, PCM undergoes a phase to keep the energy.
Classification of PCM’s
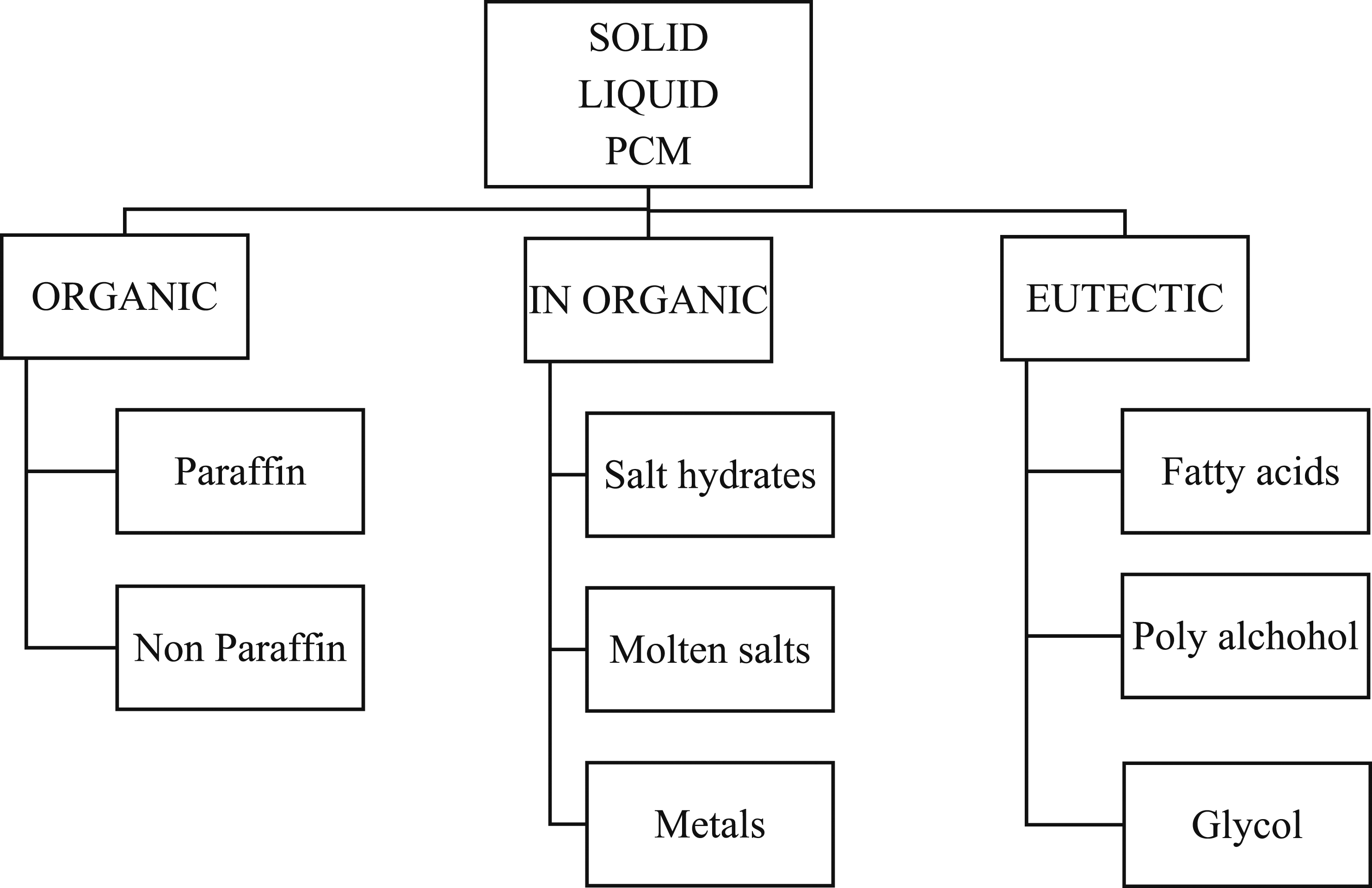
Thermal storage exists in physical and chemical modes. The physical storage is further categorized as sensible heat and latent heat. The PCM stores the energy in latent heat form, maintaining a constant temperature. PCM undergoes different phases as per existing material, i.e., solid to liquid, liquid to liquid, and solid to solid, as shown in Figure 3. Out of these, the latent heat carrying capacity is more than in the solid-to-liquid transition phase; hence the BTMS utilizes the PCM in the solid-to-liquid phase. The solid-to-liquid PCM was classified as per the existing number of carbon atoms as Organic, Inorganic, and Eutectics. The classification details are given in Figure 4. Various phases of PCM and the related mechanisms. Classification of PCM.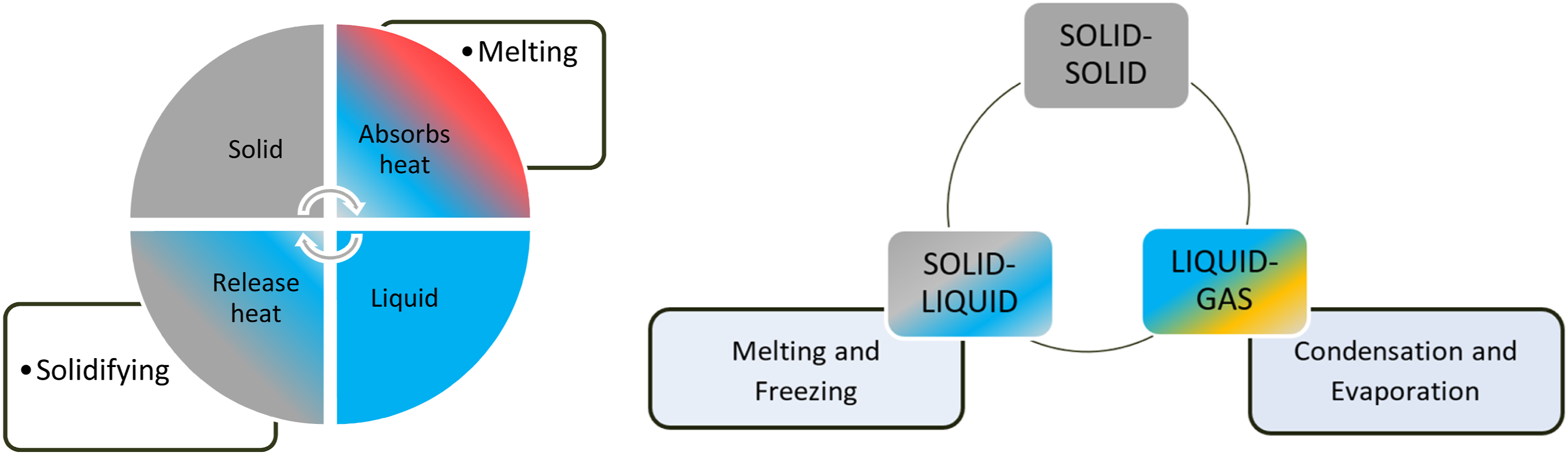
Selection of the material
The selection of PCM related to storage applications, mainly in electric vehicles, is critical. In all aspects of chemical, thermophysical, kinetic, and economic sense, PCM will be selected as per the application demand. To start with, melting temperature is the significant parameter in the selection of PCM, and it is recommended to be within the operating range of Li-ion battery (20°C-60°C), i.e., and often the selected PCM should have a melting temperature below 40°C for Indian Conditions 19 further, PCM should be non-polluted throughout service life, low environmental impact, and must possess recycling capability.
Each type of individual PCM has its own advantage and disadvantage. To evaluate the thermo physical performance of PCM, many calibration techniques are available, namely, the Differential scanning calorimetric method (DSC), Differential thermal analysis (DTA), and T-history method. Thermal conductivity can be calibrated by hot disk apparatus, and thermal stability by Thermo gravimetric analysis (TGA). Fourier can analyze chemical properties transformed by infrared spectroscopy (FT-IR). 21 Calibration of nano-structured PCM can be performed by electrochemical impedance spectroscope (EIS) and X-ray diffractometer (XRD).
Description of PCM
Organics
Organics are alkaline containing carbon-attached hydrogen or C-H bonds. Paraffin and non-paraffin are additional classifications for organic PCM. A straight, saturated hydrocarbon chain with the formula CnH2n+2 makes up the paraffin structure. The properties like melting point (°C) and latent heat of fusion (kJ/Kg) differ with inorganics based on the configuration of carbon with hydrogen ions. Paraffin has many other worthy properties like different melting temperatures, phase transformation temperatures, low vapor pressure, no super cooling, chemically stable, inert to metal containers, and available at reasonable prices. Besides the advantages, paraffin has a few limitations, like low thermal conductivity and the nature of flammability, and shows incompatibility with plastic containers. Non-paraffin organic PCMs have weak flame resistance, poor thermal conductivity, low combustion temperatures, and momentary toxicity.
In organics
Inorganic paraffins are described with the chemical formula CH3 (CH2)2nCOOH (fatty acids) and the basic property of latent heat is comparable with organic paraffins. In comparison to organics, inorganic materials have some advantages, like high heat storage capacity, better thermal conductivity, greater phase change enthalpy, and lower cost compared to organics. 21 The applications of the inorganics are medium to high melting temperature ranges. Demerits of inorganics are highly corrosive in nature, have no specific phase transition due to lack of stability, and behave under cooling. 22 Broadly inorganics are classified as molten salts, salt hydrates, and metals.
Eutectics
Eutectics are the mixture of two or more components having minimum melting point composition. Eutectics tend to freeze to form a nice blend of crystals. Due to this, eutectic is not segregated during melting and freezing. 23 Eutectics exist with the combination of organics and inorganics.
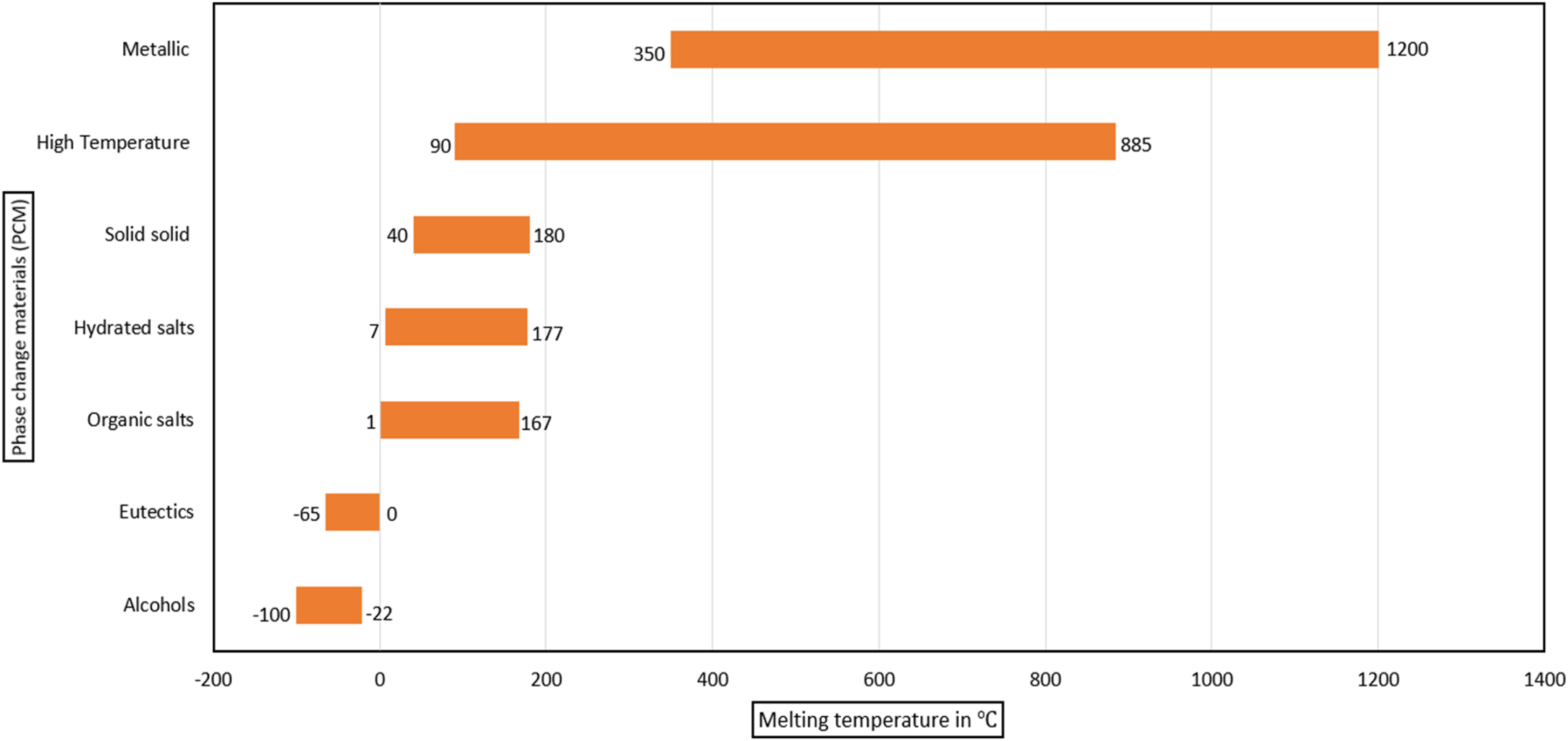
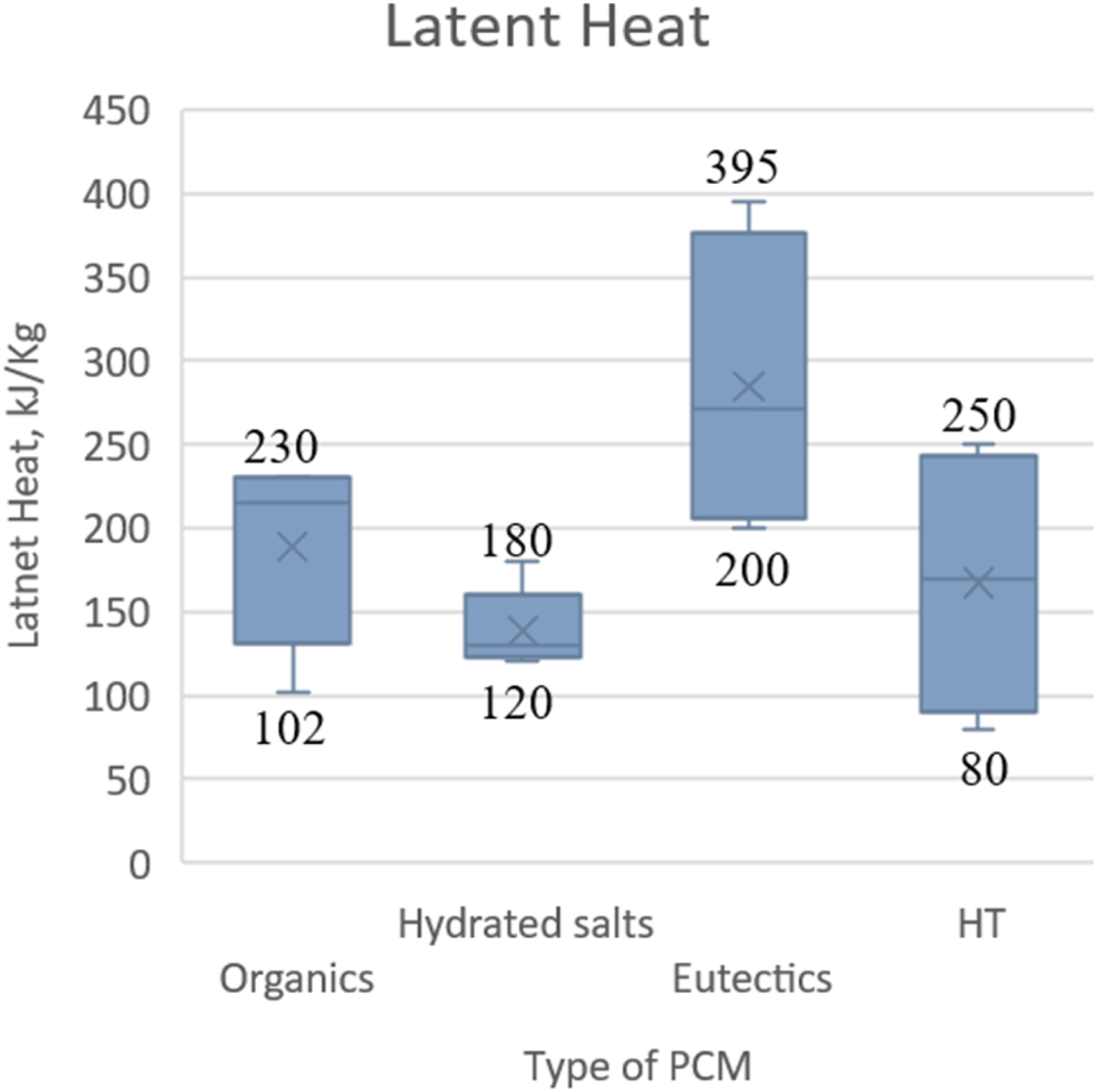
Temperature range of various phase change materials
Even though eutectics have more latent heat values due to their negative melting temperature ranges, they are not suitable PCM for the EV battery application.
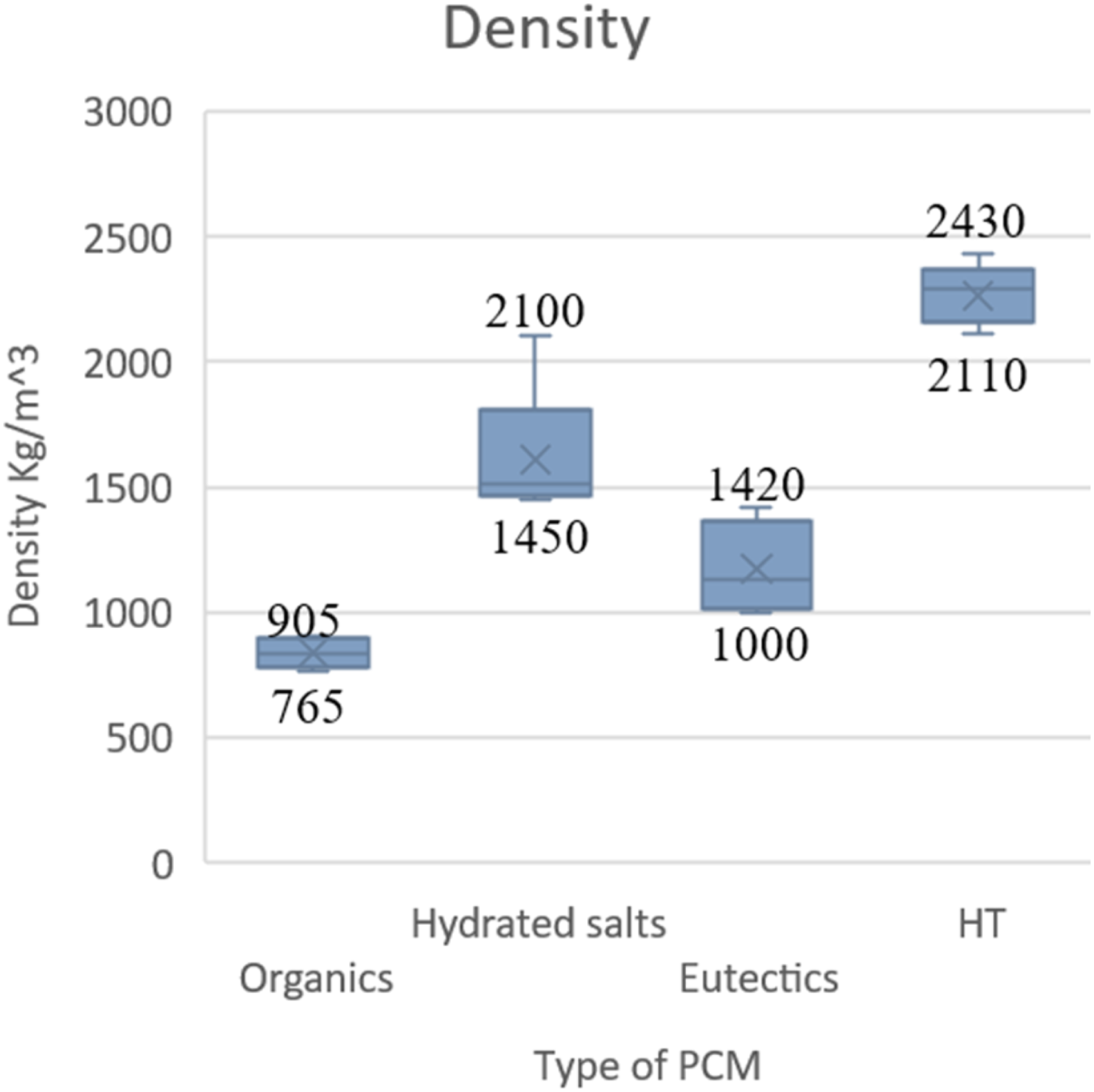
Organics have the lowest density out of all the types of PCM; hydrated salts are a good choice in terms of density.
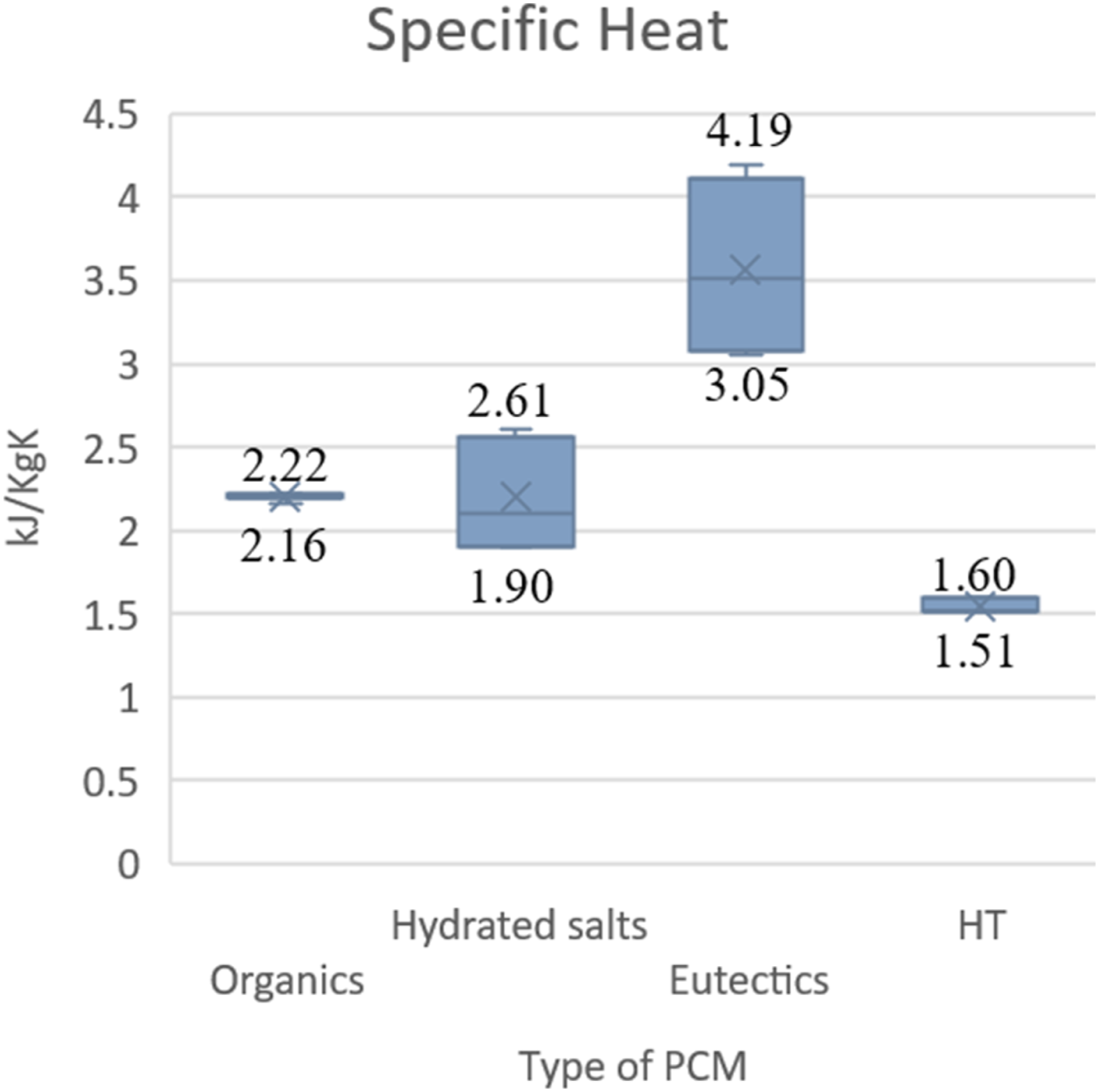
Most of the PCM specific heat will lie between 2–2.5 kJ/kgK except eutectic PCM. Still, eutectics are not suitable for EV battery applications because of the negative melting temperatures.
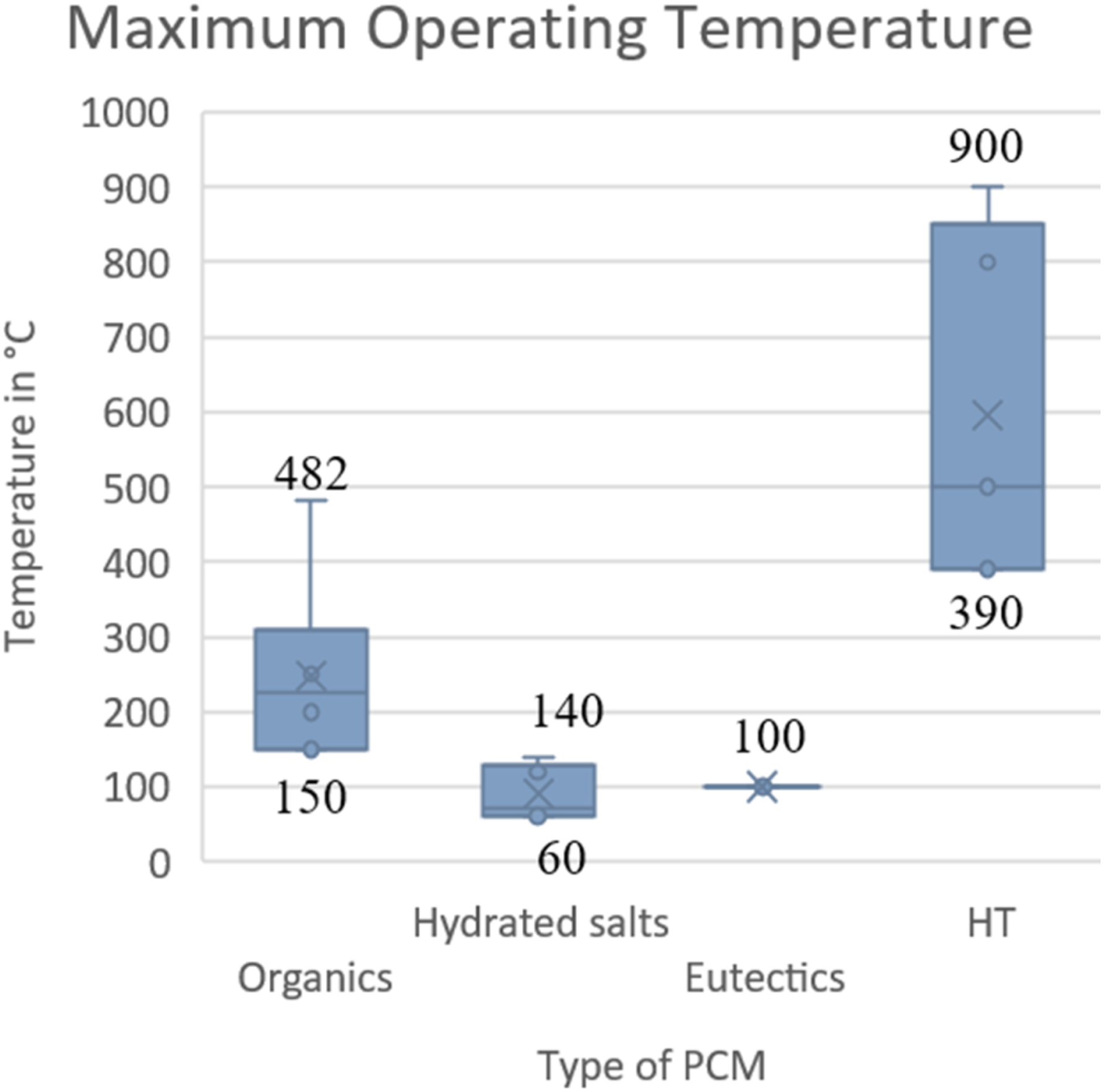
Organics are the second-highest maximum operating temperature materials in the case of PCM. HT PCM has the highest operating temperature, ideal for thermal runaway cases in a battery. Still, the Phase change temperatures of HT would not be a perfect case to use in EV battery applications. (Figures 5–10) Ideal properties of PCM.
20
Operating temperature range of various PCM.
36
The latent heat versus different types of PCM. (Graph reproduced using the literature data).
36
The density versus different types of PCM. (Graph reproduced using the literature data).
36
The specific heat versus different types of PCM. (Graph reproduced using the literature data).
36
Maximum operating temperature versus different types of PCM. (Graph reproduced using the literature data).
36

Brief description of phase equilibrium diagrams to characterize different PCM
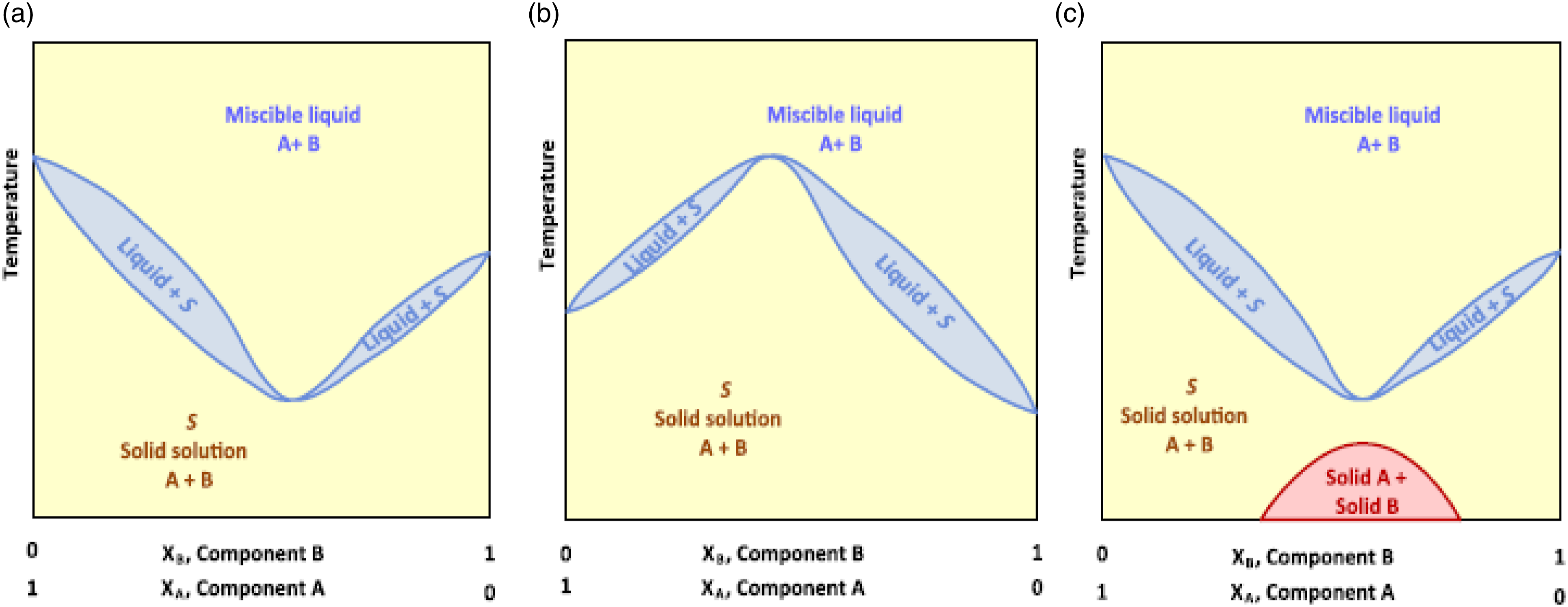
Material mixtures can exist with many components. The basic mixture starts with one individual pure component, showing its phase change at a particular temperature. A binary mixture is the simplest multicomponent mixture have desirable phase equilibrium characteristics. Based on the observed desirable changes in the PCM, there were nine different types of phase change equilibrium systems were illustrated and described below in Figures 11–13. Each system is miscible in the liquid phase and the condensed systems are assumed at constant pressure condition by neglecting the vapor phase. (a) Congruent isomorphous lowest melting temperature, (b) congruent Isomorphous highest melting temperature, (c) Partially congruent isomorphous lowest melting temperature.
51
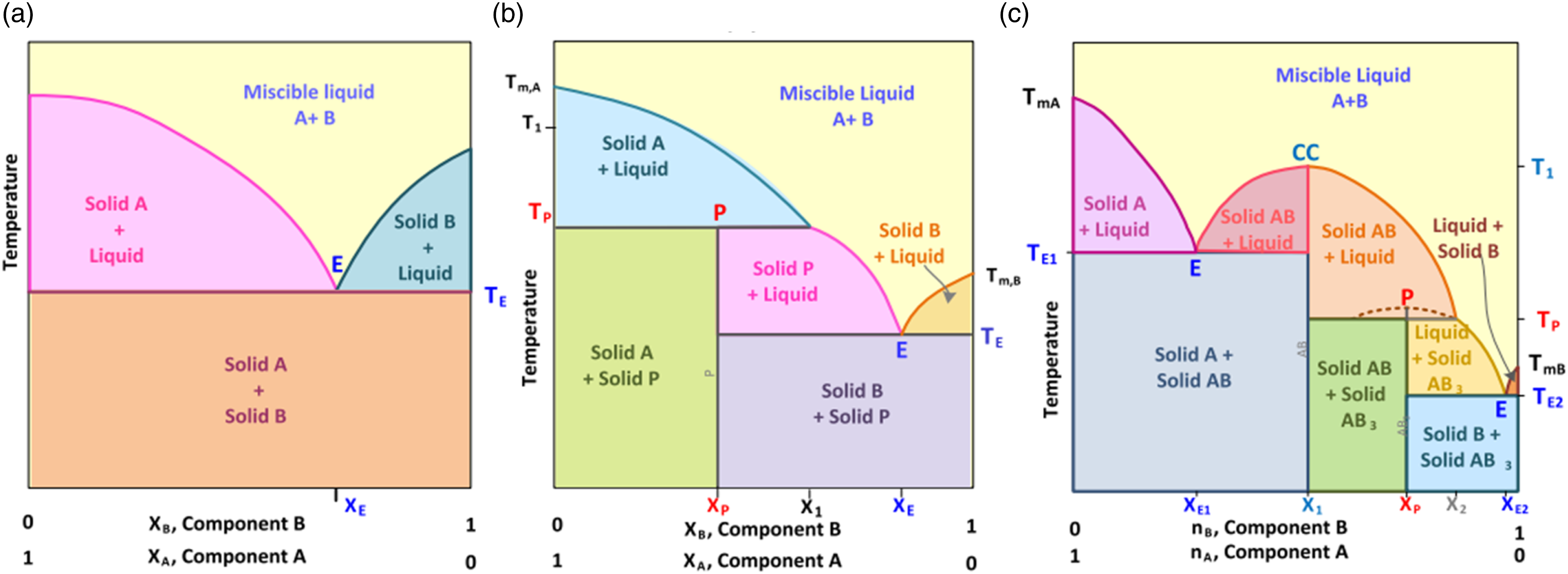
(a) Partially existed isomorphous eutectic (b) partially existed isomorphous peritectic (c) partially existed isomorphous congruent melting compound (additionally with eutectic and peritectic contents).
51
(a) Non-isomorphous eutectic (b) non-isomorphous peritectic, (c) non-isomorphous congruent melting compound formation (additionally with eutectic and peritectic contents).
45
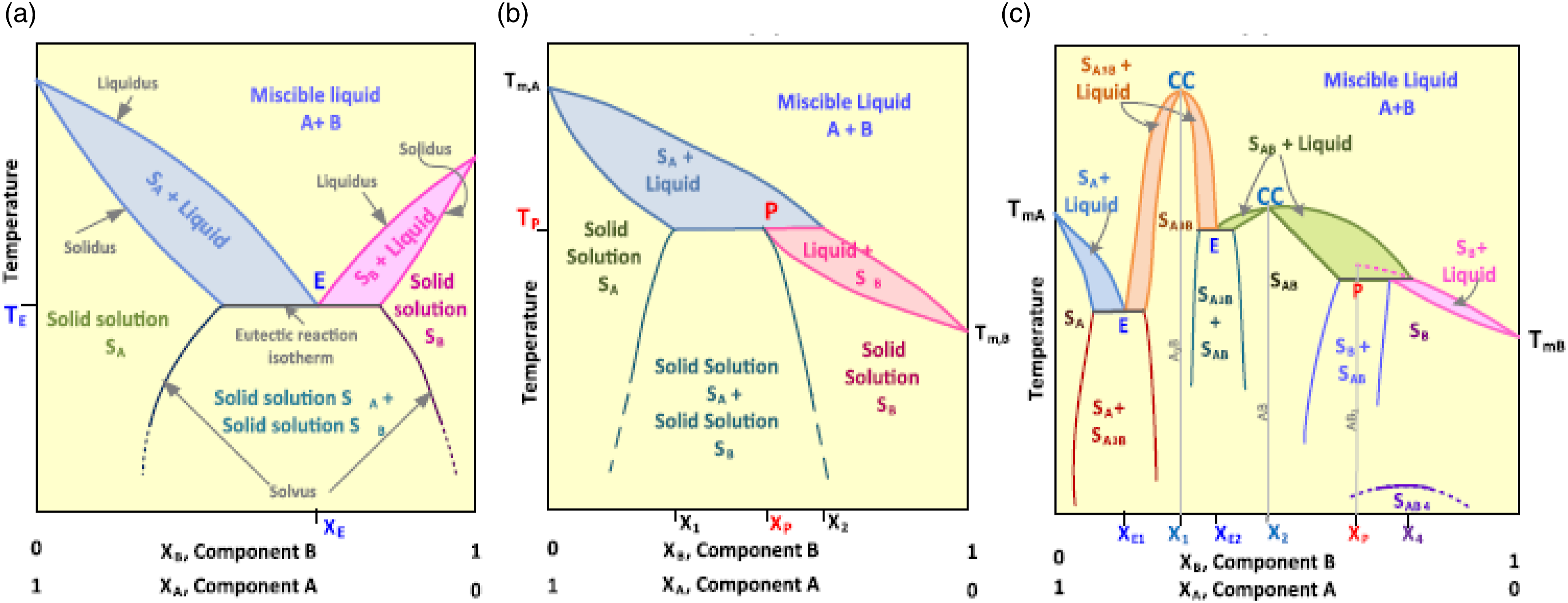
Solid solutions, eutectics, and compounds are the three different types of phase variations that exist in binary solid mixtures. Solid solutions are described as single-phase solutions made of more than single component, generally called solute and solvent, these components made homogeneous composition. Eutectics are homogeneous mixtures of different compositions which melts or solidifies at lower temperature than one of the components melting points. Compound solutions are mixtures formed with fixed stoichiometric proportioned atoms of more than one element. 50
Figure 11 describes the phase equilibrium of the miscible solution formed with components A and B in solid and liquid phases. These all are characterized by the tangential contacts between liquid and solid curves at congruent melting points. Liquidus curves represent the temperatures at which different alloy components begin to freeze while cooling and finish melting. In contrast, the solidus curves represent the temperature points at which the alloy components of various proportions begin to freeze when exposed to a cooling medium, and initiate melting when subjected to a heating medium. 52 The definition of congruent is solid phase and liquid phase existing in equilibrium with the same composition and, ideally, having a one precise melting point with no phase separation. The term isomorphous indicates that all components present in A and B are completely mutually miscible in all proportions. Figure 11(a) and (b) show congruent isomorphous melting for solid solutions at their lowest and highest melting temperatures. Adding one element lowers the melting point temperature of another element in the isomorphous congruent minimum melting instance.
In the isomorphous congruent maximum melting case, the addition of one element will increase another element's melting point temperature. Figure 11(c) describes the isomorphous congruent melting partially where the miscibility gap exists below the melting point temperature within the miscible solid solution of A and B which is very similar to the (a) in ideal conditions.
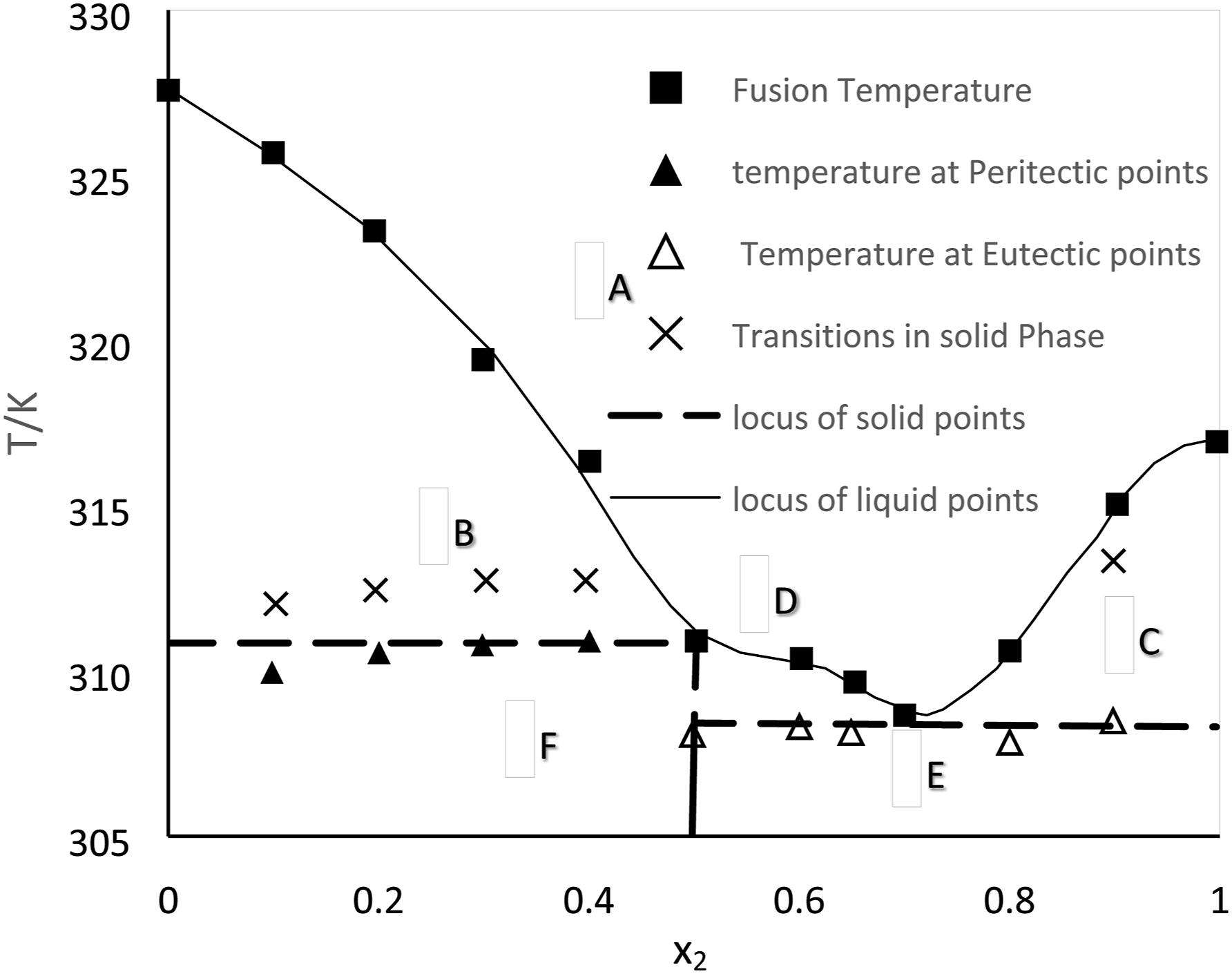
Figure 12 displays the Compound mixtures of eutectic and peritectic compounds are formed due to their reversible nature, which prevents phase separation. As a result, when these mixtures solidify, two distinct solid phases with different compositions emerge from the liquid phase. Eutectics does not accurately match congruent melting systems even though it exhibits similarity with them through the presence of sharp melting points. PCM exhibits perfect behavior in eutectic conditions. This solid composition will show phase separation in case one solid exposes to supercooling. Eutectics can occur in partially isomorphous or non-isomorphous systems, but not in fully isomorphous systems. Figure 12(a) shows two solid solutions combined composition is equal to the liquid phase composition at eutectic isotherm, indicating that sharp point with E. Figure 12(b) shows the solid solution of formed peritectic is the subset of eutectics. Peritectic exhibit incongruent melting by changing a solid and a liquid into a solid. Peritectic reactions can experience coring, which hinders them from reaching the complete stage of phase transformation. Coring reduces the effective freezing temperature and hinders the attainment of equilibrium, leading to significant phase separation rather than a sharp, centered point. Figure 12(c) represents a compound mixture of components A and B. Components are partially isomorphically mixed and melted. As a result, congruently, eutectics and peritectic are used to create compound formulations with solid solutions (SA,SB), compounds (SAB, SAB + Liquid), eutectics(E), peritectic(P).
Figure 13 is representing the non- isomorphous eutectics, peritectic and compounds mixtures. In this system even if peritectic exists, there will always be at least one eutectic. Figure 13(a) represents a simple eutectic structure formed by the solid components A and B. Very sharp unique melting point(eutectic) created with two pure components in the manner of non-isomorphous system due to those two solid components are complete immiscible with each other. The existence of a eutectic point in a non-isomorphous system is always conceivable due to the reason that a component's adoption lowers another component's melting temperature. Similarly Figure 13(b) depicts the peritectic formation from the two solid components (A, B). Along with eutectic, a different substance (P) form. Figure 13(c) represents the creation of a straightforward congruent melting compound from two solid components (A, B).
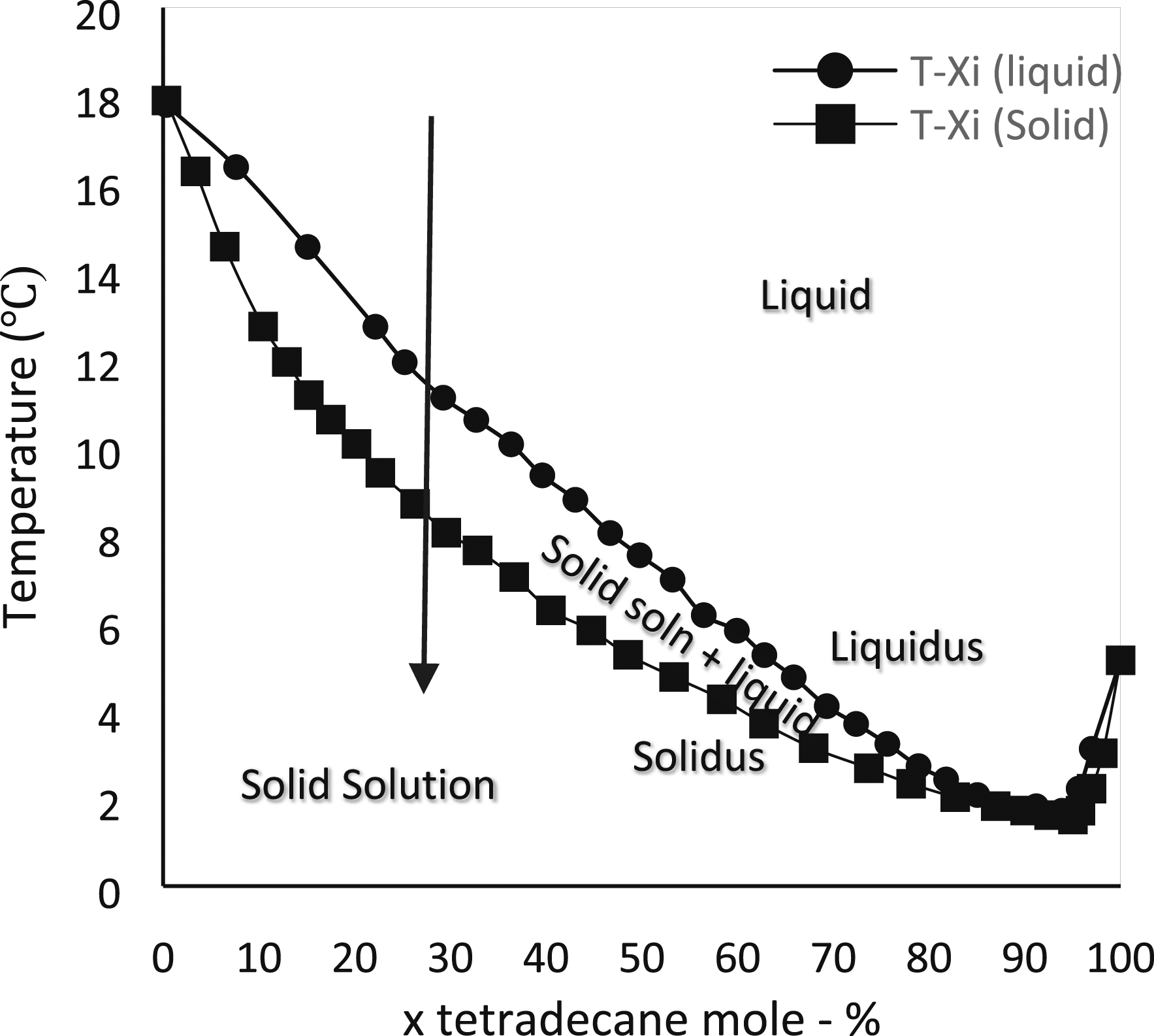
In the field of PCM-based BTMS, various researchers used different kinds of PCMs to achieve lesser temperatures in the battery pack. The base PCM for many of the experiments is paraffin due to its advantages shown in Table 2. In order to study the thermal behavior of lithium-titanate (LTO) battery cells during a high C-rate discharging operation, Behi et al.
80
employed paraffin as PCM and a heat pipe cooling system. The basic phase shift transition diagram of the binary combination of paraffins C14H30 and C16H34 is shown in Figure 15. Pseudo-phase diagrams of binary wax blends for Bees wax and paraffin.
55
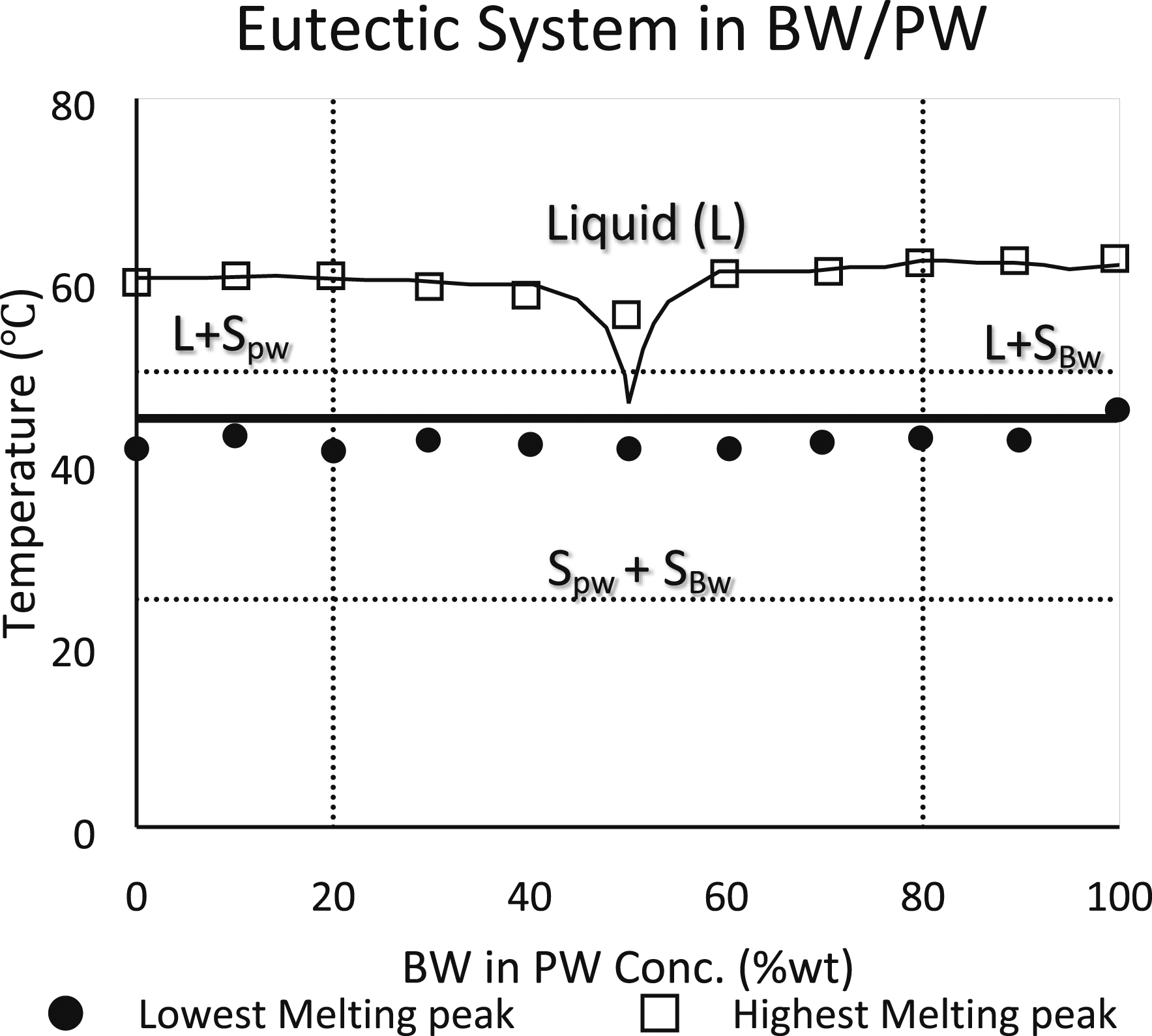
Putra et al.
55
used beeswax to determine the cooling system’s effectiveness at the temperature range of 25°C–55°C in the application of EVs. Figure 14 shows the basic eutectic blend of beeswax and paraffin wax. In order to attain the ideal temperature range with the aid of bionic liquid channels, Yang et al.
56
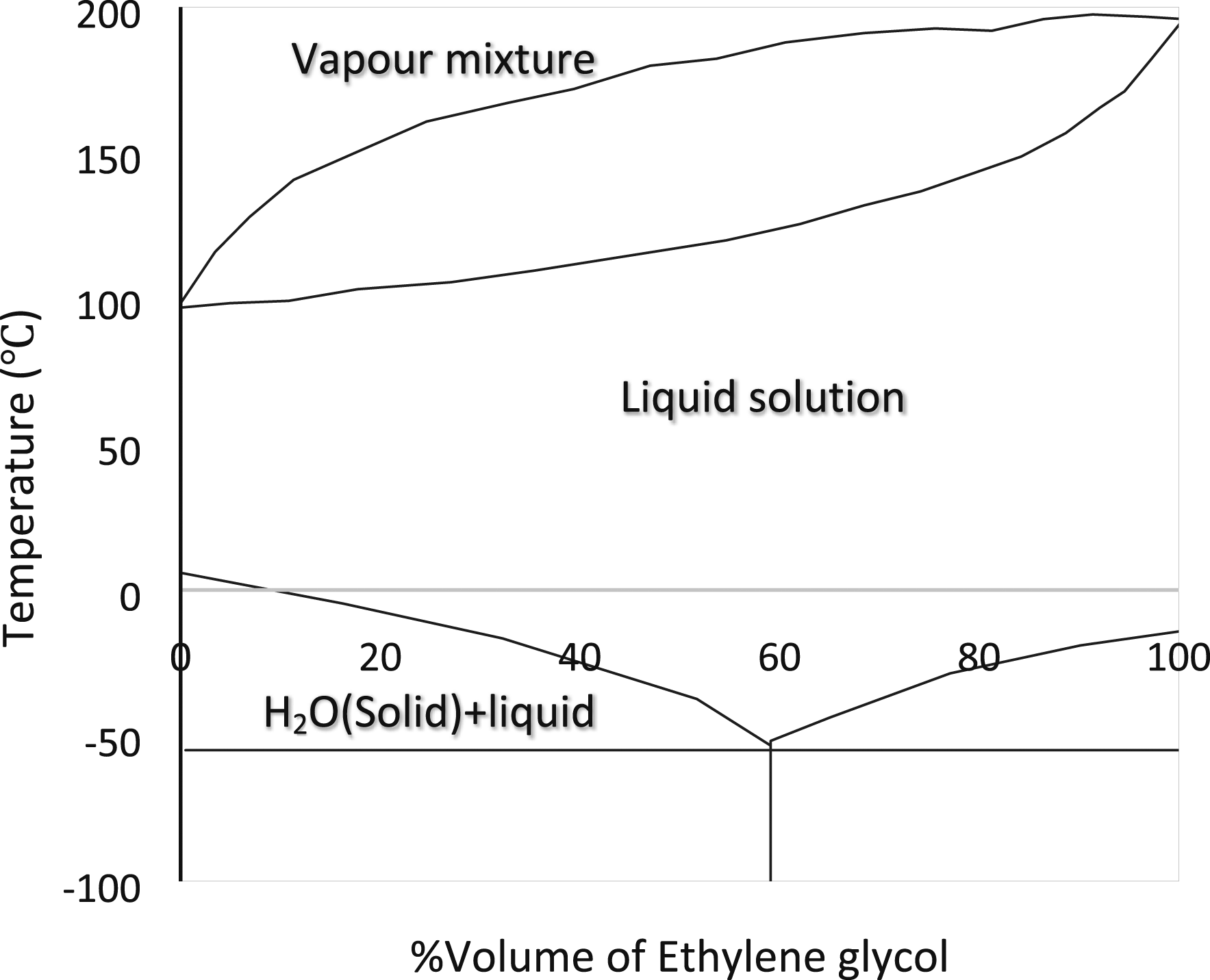
used polyethylene glycol-1000 (PEG) as PCM and 16% ethylene glycol solution as a coolant in a PCM-based BTMS. Figure 16 displays the diagram of the volume% of the pure ethylene glycol phase transition. In order to attain the battery module's ideal temperature range, Sihang Hu et al.
81
reported the passive thermal management work on a battery module using a composite PCM combination (Lauric acid + expand graphite + graphene). Costa et al.
57
gave a clear explanation of the binary mixtures at the solid-liquid equilibrium of the fatty acids and triacylglycerols. Figure 17 shows the reaction between lauric and myristic acids, which led to the formation of a non-isomorphous congruent melting compound with six distinct locations. The phase diagram of the mixture is thus divided into six regions: region A, where the liquid phase is located above the locus of the liquidus point; region B, where the solid myristic acid coexists with the liquid phase; region C, where the solid lauric acid coexists with the liquid phase; region D, where a pure solid combination of the compound (myristic acid+ lauric acid) coexists with the liquid mixture; and regions E and F, where the pure solid lauric acid and myristic acid respectively. C14H30 and C16H34 binary mixture liquid –solid phase diagram.
80
Phase diagram for ethylene glycol water solutions at 100 kPa.
51
Phase diagram of lauric + myristic acids.
54
The corrosive nature of the salt hydrate PCM might cause other materials to lose their structural integrity and shorten the battery pack's lifespan. 10 As a result, salt hydrates are rarely used in the BTMS application.
Examples: phase equilibrium diagrams of inorganic salt hydrates systems.
Miscellaneous properties of the PCM
Selection of the material provides a brief description of the factors to be taken into account when choosing a PCM, and it highlights the key factors that are necessary to comprehend PCM fully. This discussion concerns thermal stability, including robustness and compatibility with the containers.
Thermal stability of PCM
The material's durability is determined by how well it holds together after repetitive use and thermal cycling in storage application intended for melting and freezing. After long-run thermal cycles, the PCM should be chemically, thermally, and physically stable. The prior required properties for BTMS application are melting point temperature and latent heat of fusion. As a result, the initial stability test of PCMs should be carried out in accordance with their real usage. PCM’s thermal stability calibrating thermostatic chamber/bath, constant temperature oven, electric hot plate, and DSC, TGA.
Calibration of thermal stability of PCM
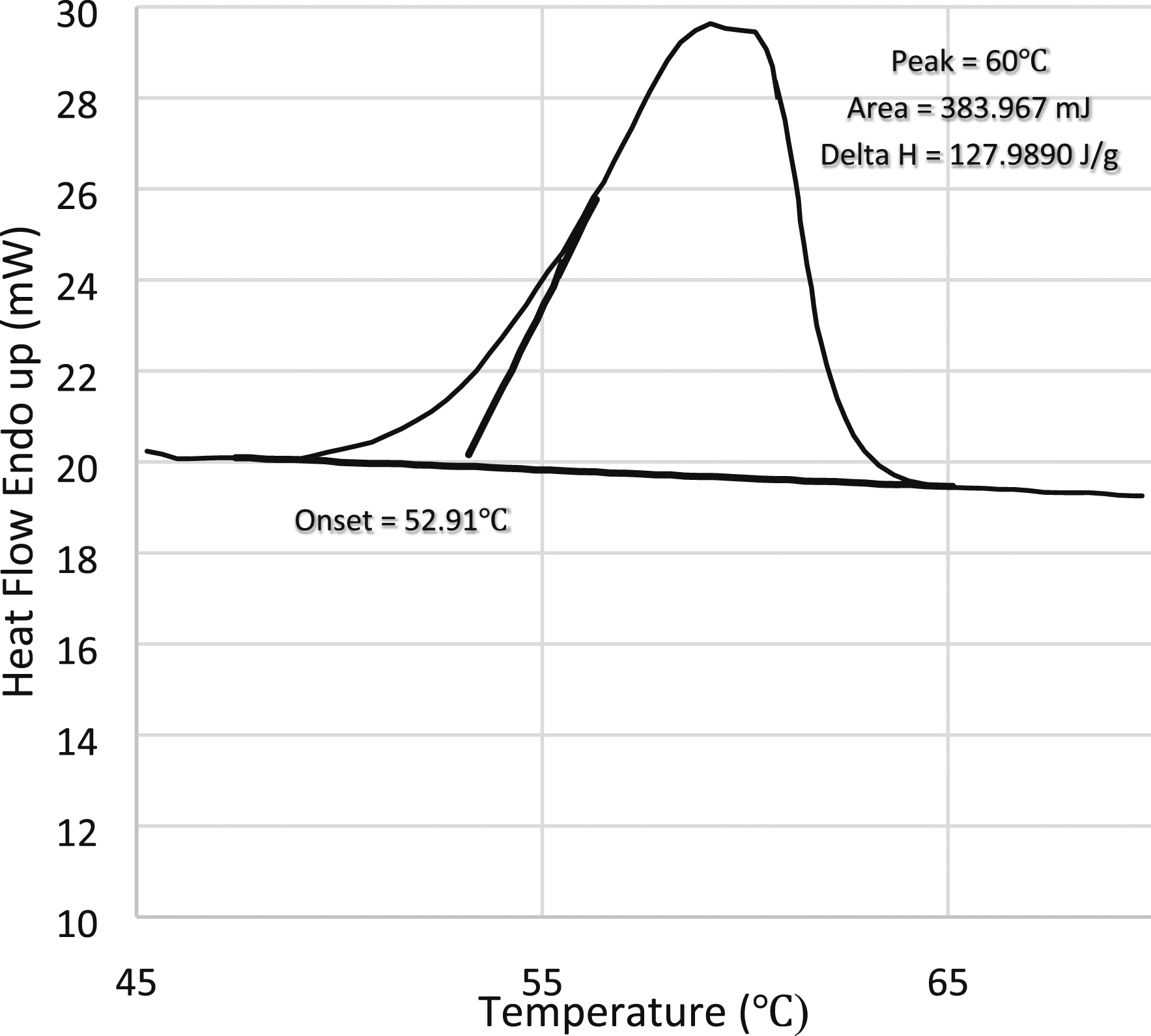
DSC is the widely used equipment for the application of PCM-based BTMS, on the basis of thermo analytical principal DSC works. A PCM sample and reference are considered, throughout the experiment and are necessary to keep a constant temperature. Using DSC analysis, heat is applied to the PCM sample and reference substance to keep their respective temperatures constant, after a certain time the PCM sample undergoes phase transitions and it requires sufficient heat to maintain a constant temperature with the reference. The requirement of heat for the PCM sample based on process is exothermic or endothermic. By measuring the difference in heat flow between the PCM sample and the reference, the DSC will determine if there are heat transactions of absorbing or releasing. A curve known as the DSC curve is a plot between heat flow and temperature. The area under the peak of that curve represents the latent heat of fusion. Drawing onset linen with a curve fitting to the raising part of the peak results in the phase transition temperature and phase transition range is the region between the curve's onset and peak temperature 60
To calibrate the change of latent heat fusion and temperature of phase transition after every specified cycle, i.e., 500 or 1000 cycles, a relative percentage difference (RPD) is to be noted.
61
The RPD of any property i of the PCM at any number of cycles n and the 0th cycle may be defined as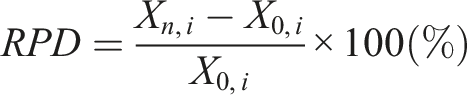
Where Xn,i = onset and peak temperatures and the latent heat of the PCM after n cycles
X0,i = onset and peak temperatures of the PCM at 0th cycle.
Various works of literature explained the thermal stability of PCM in detail. Many of the sources are from experimental results. Some of the regularly used PCMs are shown in the below tables.
PCM compatibility with container
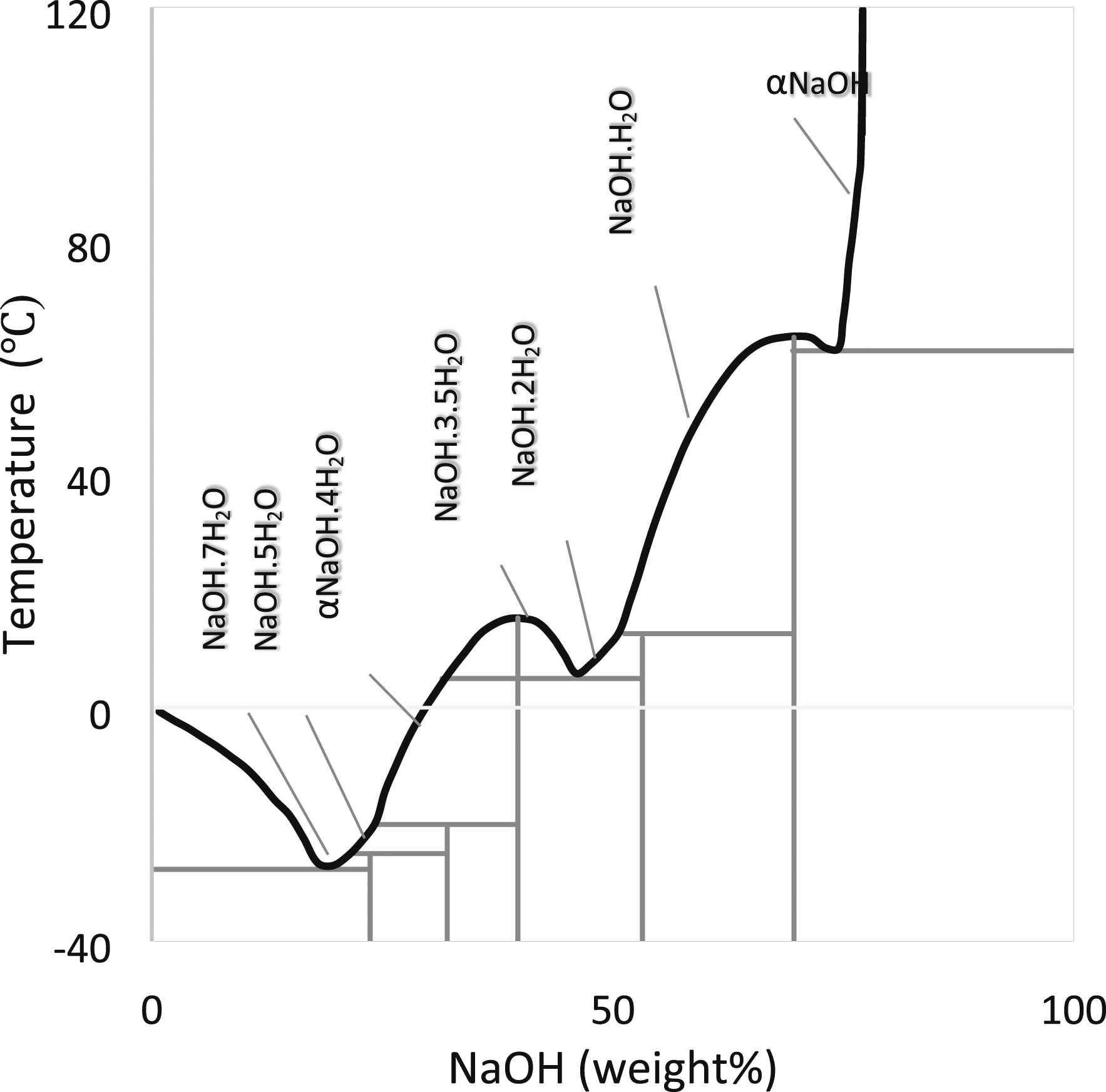
For the long-term stability of PCM, compatibility with adjacent support structures is important. Hence, corrosion is not present in the container while it is in contact with PCM, and the progress of the chemical reaction also slows down. Castellon et al.[77] experimentally shown the behavior of plastics with PCMs. Plastics considered in this work are HDPE, EGYPTENE, HD6070UA, LDPE RIBLENE FM34, and Exxon Mobil PP 7011L1, PCM as commercial-grade organic kinds of paraffin are RT20, RT25, and RT27, inorganic salt hydrate. Plastics were immersed in PCM-contained beakers and performed thermal cycles by keeping them in the oven. After 400 days, Different tests measured material compatibility and stability and concluded that HDPE showed better results and that paraffin is more compatible than salt hydrate. Gerard Ferrer et al.[78]explained the corrosion effect on metal containers using PCM in energy storage applications. In this work, five different metals, namely aluminum, copper, carbon steel, stainless steel 304, and stainless steel 316 were selected with four different PCM arrangements of two eutectic mixtures Capric acid (73.5%) + myristic acid (26.5%), Capric acid (75.2%) + palmitic acid (24.8%), Ester Salt Pure Temp 23, Inorganic Salt SP21E were examined and concluded that Stainless steel 316 and stainless steel 304 showed great corrosion resistance (0-1 mg/cm2yr) and it is compatible any of the studied PCM. Khan et al. [20] gave a detailed review of the performance enhancement of PCMs based on latent heat energy storage in contact with PCMs stability and compatibility. This review says that the long-term productivity of latent heat storage systems is proportionate to Thermo physical stability and compatibility with withholding materials. Khan explained various works done on paraffin and salt hydrates stability and the impact of the compatibility with plastics and other metals. Rawson et al.[79] concerned with finding alloys suitable for energy storage applications. In this view, the CALPHAD (Calculation of phase Diagrams) approach is implemented in the TCAL5 and TCOX8 databases offered by Thermo-Calc Software in order to choose the proper containers for such alloys utilizing automated macro creation. It concludes that databases identified nine container materials compatible with many pure and eutectic mixtures at equilibrium. Also, Thermal properties for hundred eutectic compositions were listed, and this work contributes to finding the compatible phases for identified eutectic compositions and pure mixtures. Nagano et al.[76] considered six different metals Cu, C-steel, Brass, Al, and two different proportions of stainless steel, SUS304, and SUS316, used in the corrosion test. This corrosion test helped determine the compatibility of the salt hydrate PCM mixture of magnesium nitrate hexahydrate (Mg (NO3)2. 6H2O) and additive magnesium chloride hexahydrate (MgCl2.6H2O). (Figures 18–20) The test results showed that SUS316 and Al might be utilized as the storage container material and heat exchangers for this PCM mixture after examining each metal for 90 days in a glass tube at a temperature of 95°C. Figure 21 shows the results of the corrosion test. Gabuler’s salt phase diagram.
58
Phase diagram of sodium hydroxide–water system.
59
DSC measurement of the latent heat of fusion and the melting temperature of paraffin.
60
Magnesium nitrate hexa hydrate with additive magnesium chloride hexahydrate compatibility test with metals surface condition after 90 days observation at the temperature of 95°C.
76
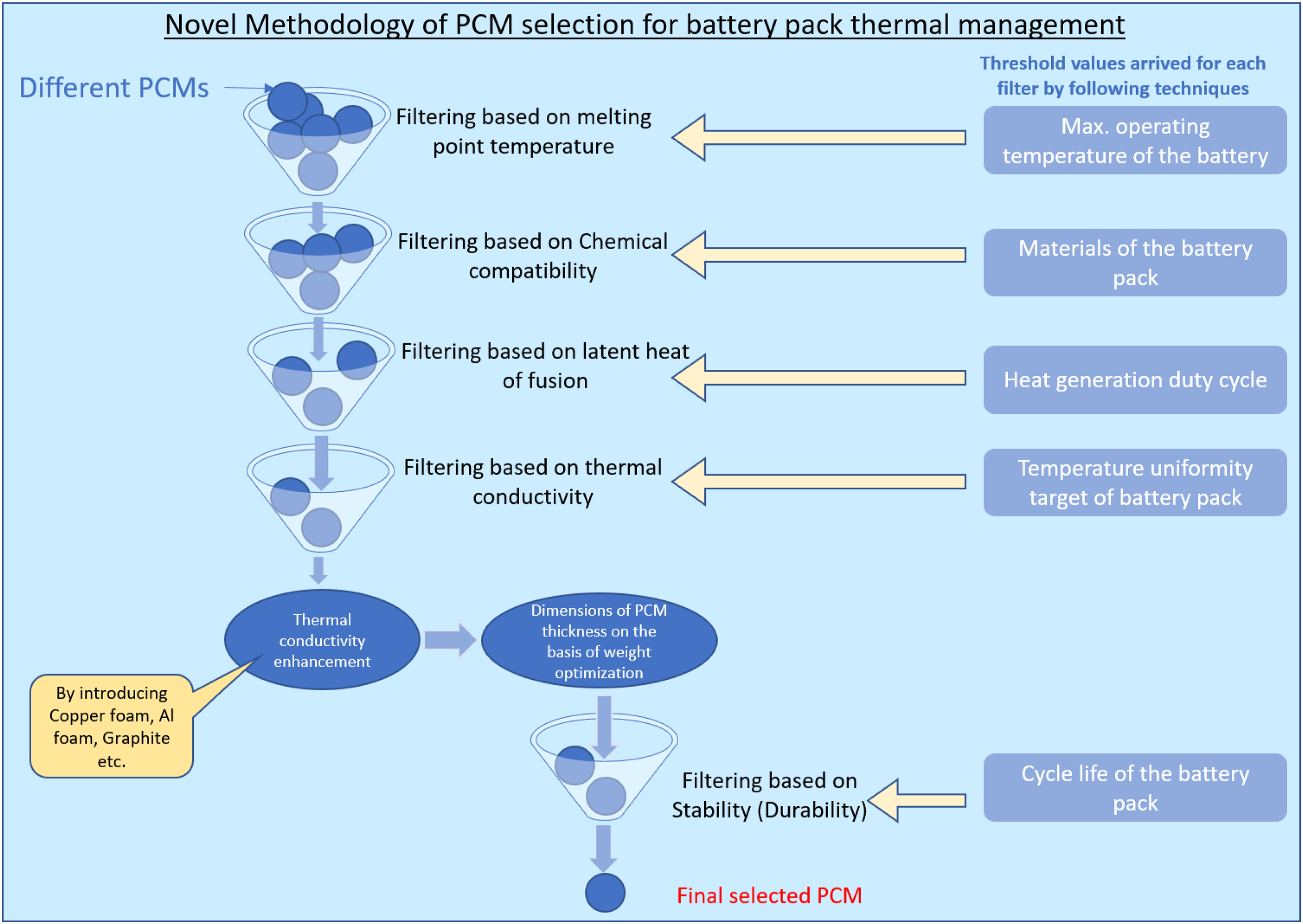
Methodology for selecting suitable PCM for battery thermal management.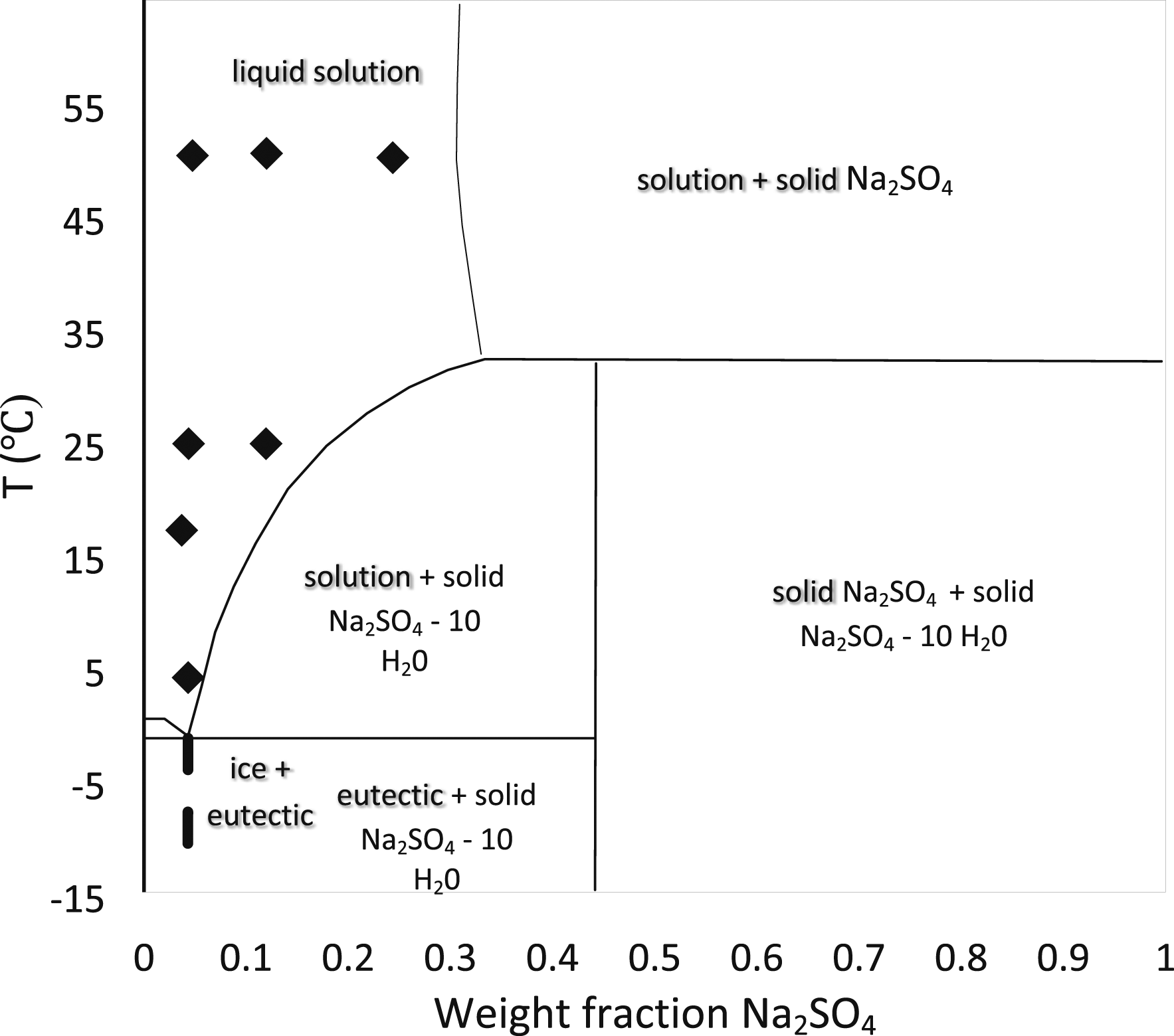

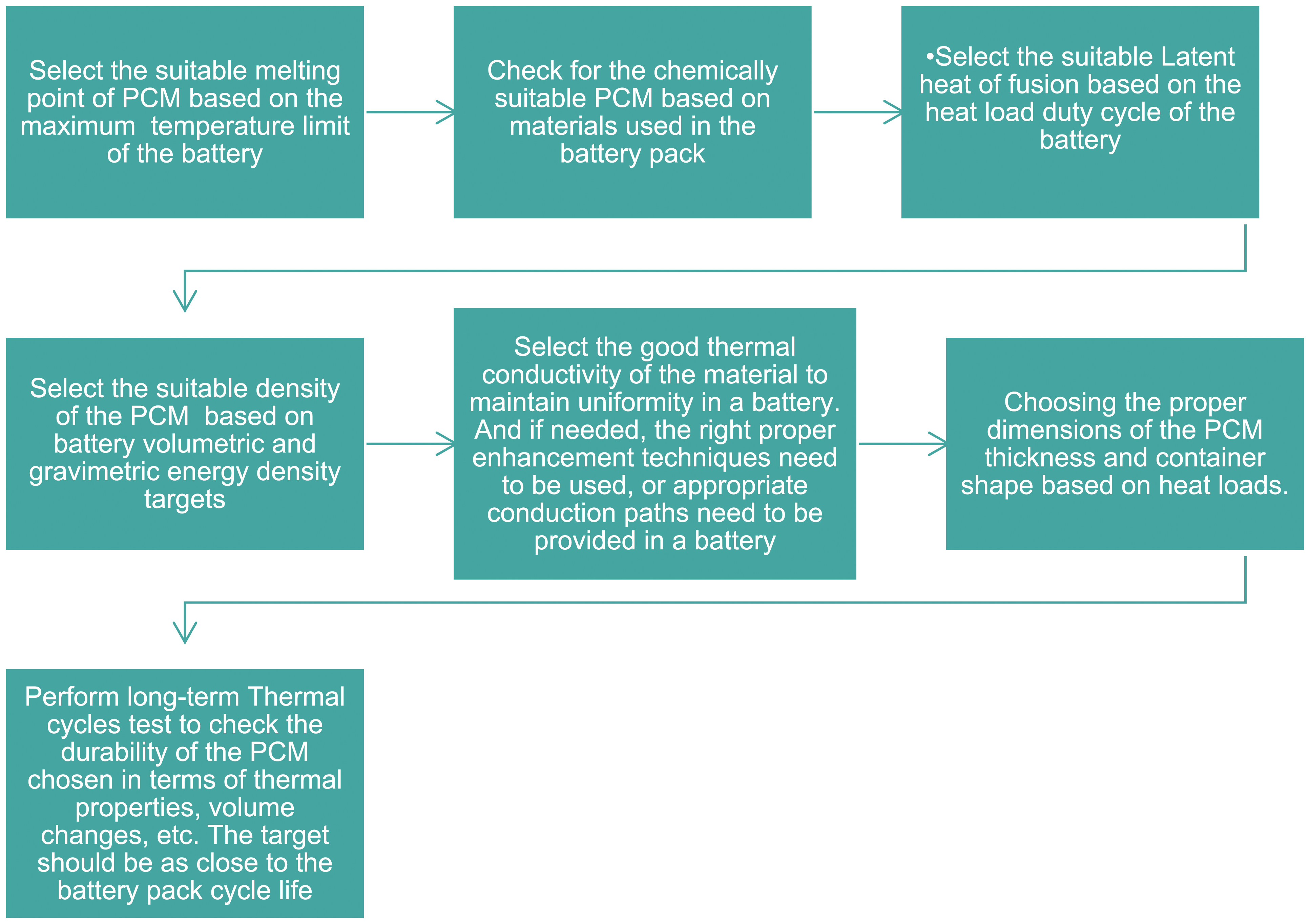
Methodology to select the right PCM and implementation in battery thermal management
The following methods may be used as a guide for applying PCM-based BTMS to choose the PCM for BTMS. Prior concerns such as the needed melting temperature, required density, and latent heat of fusion were considered while choosing the appropriate material. Additionally, performance-improving approaches are taken into account. Many charging and discharging cycles must be carried out after choosing the PCM in order to evaluate the material's durability or life cycle. One of the most important things to think about is if the PCMs are compatible with the containers because many PCMs display corrosion and flammability at higher temperatures. The following technique was developed based on knowledge of numerous literary works (Figure 22).
Conclusion and future scope
Thermal management is crucial and plays a significant role in maintaining the performance and safety of lithium-ion batteries. This work focuses extensively on passive thermal management using phase change materials (PCM). The properties of various PCMs are listed, compared using phase change equilibrium diagrams, and the stability of the PCM is discussed in relation to its lifespan alongside the battery.
Perspectives for selecting the right PCM
• Organic PCM and hydrated salts are the most suitable PCM options as their melting temperatures are closer to the appropriate working temperatures of lithium-ion batteries. • Organic PCM is less corrosive compared to other PCM types. • All PCM materials inherently have low thermal conductivity, which can be mitigated by using thinner layers or by enhancing conductivity through thermal conductivity enhancers. • Composite organic PCMs exhibit higher thermal stability compared to other PCMs. • The durability cycles of the selected PCM should be equivalent to the battery pack's lifespan. • PCM compatibility is crucial because the properties can drastically change when interacting with different materials, even if the individual materials perform well independently.
Future scope in using PCM in (BTMS)
• The durability of PCMs under mechanical vibration and shocks, which are important factors in real-world battery applications, needs to be extensively studied. • Quantitative studies should be conducted to determine the advantages of PCM usage. As PCM adds mass to the battery and the vehicle, its impact on mileage and energy savings should be evaluated for selecting the appropriate PCM. • Numerical simulations are valuable tools for optimizing the design of BTMS and approximating the liquid fraction of PCM over time. This information is crucial for determining the suitability of PCM in a given application, which can be challenging to determine through experimentation alone.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

