Abstract
Keywords
Independent studies have shown that expressions of fear augment sensory vigilance and are associated with enhanced behavioral and neural markers of attention and perceptual processing (e.g., Anderson et al., 2003; Phelps et al., 2006; Pourtois et al., 2005). Converging evidence comes from research on the effects of body odors (axillar sweat) emitted when a person is experiencing fear (fear chemosignals). Fear chemosignals alter the sensory systems of a receiver’s face, activating the relevant fear-related muscle (
We begin with a brief overview of the research on fear chemosignals and how they induce vigilance, modulating receivers’ facial sensory system to increase sensory acquisition. We then introduce inattentional blindness as a phenomenon and examine whether the state induced by fear chemosignals reduces this phenomenon. We then merge the research on two differing perspectives to examine this issue further. In the closing discussion, we draw out the potential ramifications of our research for people performing continued-vigilance tasks, ranging from air traffic controllers to drivers.
Fear Chemosignals and Their Functions
Emotion-related facial expressions have rarely been examined as moderating an experiencer’s exposure to environmental stimulation. The existing studies in this research field have shown that fear facial expressions strongly impact experiencers’ sensory acquisition (Lee et al., 2013; Susskind et al., 2008). Fear is a high-vigilance emotional state (LeDoux & Pine, 2016). While experiencing fear, individuals tend to activate the medial frontalis muscle in the forehead, causing their eye and nasal apertures to widen. This, in turn, leads to an expanded visual field, which facilitates eye movements and results in higher volumes of air intake. This state is commonly referred to as a state of
Significantly, fear chemosignals induce the same biomechanical changes in receivers’ faces, also modulating sensory acquisition. It has repeatedly been shown that fear chemosignals, measured by facial electromyography (fEMG), activate the medial frontalis (e.g., de Groot et al., 2012, 2014b, 2015; Gomes et al., 2020, 2023; Kamiloğlu et al., 2018), increasing the number of fixations, increasing nasal-inspiratory volume, enhancing perception, and accelerating eye movements during target localization (de Groot et al., 2012). All this occurs without the receiver’s awareness and does not give rise to an emotional state of fear but mainly enhanced scanning of the environment. The differential processing of environmental events has been demonstrated in research (e.g., de Groot et al., 2012, 2017; Gomes & Semin, 2021), reaffirming this subtle preemotion stage of information processing (Lee et al., 2013). Some have argued that fear activates a sensory-search process that has become evolutionarily consolidated. It, therefore, has acquired a highly adaptive value over the evolutionary course of history across animal species and is conserved in mammalian brains (LeDoux, 2012).
Inattentional Blindness
The phenomenon referred to as
Statement of Relevance
Psychological scientists have documented the failure to notice unexpected objects or events when attention is focused elsewhere. This is known as
Selective attention can filter irrelevant information, and participants often do not perceive the objects that do not receive attention. More importantly, unexpected but distinctive objects do not automatically attract attention (see Simons & Chabris, 1999). The particular tasks that are designed to tap inattentional blindness require focused attention. While the participant is performing a primary task (e.g., counting the number of times the basketball changes hands), an unexpected event occurs (e.g., a gorilla passes by). For individuals who do not have a primary task, the unexpected event is self-evident and likely to be identified. However, for those who performed the primary task, the unexpected event is usually not detected (see Simons, 2000). The function of attention in this dynamic-empirical context is to act as a filter for irrelevant events that are not perceived. One’s active goals influence our attention and determine what we attend to in the real world. Such selective attention has a striking influence on what we see, and the phenomenon of inattentional blindness is a dramatic example (Most et al., 2005). Even strong stimuli are not attended to consciously (Mack & Rock, 1998).
Study and Design
Although the research on inattentional blindness identifies task characteristics and situated constraints responsible for the phenomenon, it has not been concerned with processes that can likely reduce inattentional blindness. This was the object of the current set of studies. The main experiment was designed to test whether the increased vigilance and sensory acquisition triggered by fear chemosignals enhance the detection of unexpected events, thereby reducing inattentional blindness. To examine this idea experimentally, we adapted Simons and Chabris’s (1999) classic gorilla study to a virtual-reality scenario. As the original study is well known, we created an aquarium environment virtual-reality scenario (see Fig. 1) in which the critical agents were two schools of fish. Early on, in the aquarium scenario, morsels of food started coming from the top of the aquarium. The participant’s task was to count how many morsels of food one of the two schools of fish consumed. While this morsel-consumption event was occurring, a shark proceeded from one side of the aquarium to the other. After a while, a dolphin entered the scene and swam across the aquarium (the order of shark and dolphin appearances was also randomized). The participant’s task was to count the number of morsels the bluefish consumed as accurately as possible (primary task). The shark and the dolphin constituted the unexpected critical events.

Aquarium with fish and shark.
The experiment had a between-participants factor: Participants were randomly exposed either to fear chemosignals or to rest body odors. Independently, a further group of participants joined the baseline study, where no instruction to count bluefish consuming morsels was given and where they were not exposed to any body odor. This baseline study was conducted to identify how often the dolphin and shark would be spotted without a specific task for the participants and to establish a reference point for the experimental study. We could thus say how much inattentional blindness was observed in the experimental study in the fear-chemosignal and rest-body-odor conditions.
The general hypothesis was that in the fear-chemosignal condition, participants would see unexpected stimuli more frequently than in the rest-body-odor condition. In the baseline study, we expected participants to spot the dolphin and the shark more frequently than in the two body-odor conditions of the main experiment, as no fish-counting task was given to the participants. However, the difference between the detection rates in the baseline study and the fear-chemosignal condition will be shorter than in the rest-body-odor condition. These predictions were based on the argument that fear chemosignals induce a state of heightened vigilance and increased sensory acquisition because of the opening of the eyes, raising of brows, and enhanced perception, as has been demonstrated repeatedly in earlier research.
The main experiment was preregistered on the Open Science Framework (https://osf.io/kfbq2). The raw data and statistical analysis for both the baseline study and the main experiment are available here: https://osf.io/me9hv.
The Baseline Study
Participants
Eighty-four female students from ISPA – Instituto Universitário in Lisbon, Portugal, gave informed consent and participated voluntarily in the study. Eight participants were excluded because they had uncorrected visual impairments or because they misunderstood the task. Thus, 76 participants aged 18 to 56 years (
Procedure
The experiment was conducted in a stand-alone manner. After giving informed consent, participants filled in a short sociodemographic questionnaire (e.g., age, visual problems, attention-related disorders) to confirm whether they were eligible to participate. Eligible participants performed the task. They were asked to wear a virtual-reality headset (Pico Neo 3 Pro Eye, Pico Technology, St Neots, United Kingdom) and place their chin on a chin rest. Then they received verbal instruction informing them that they could start the task.
The experimental task consisted of an aquarium scenario involving two types of fish with distinct colors (i.e., blue and orange) passing by. At a particular point in the scenario (approximately 2 s after the start of the virtual reality), morsels of food started coming from the top of the aquarium and were grabbed by 30% of the blue and orange fish. In total, 120 fish passed by per minute. Participants received no specific instructions for the task. They were just informed that they would observe an underwater scenario. The scenario lasted 2 min.
While this morsel-consumption event proceeded, a shark proceeded from one side of the aquarium to the other (the side was randomized between participants). After a while, a dolphin entered the scene and swam across the aquarium (the starting side was randomized between participants). The order of shark and dolphin appearances was randomized. They appeared in the aquarium in a 10-s window around the task’s midpoint. Importantly, the shark and dolphin were equated for size and luminosity. Moreover, in a pilot experiment (
At the end of the experimental task, participants were instructed, in a randomized order, to state whether they saw a shark, a dolphin, and an octopus during the task. The octopus question was a control question to exclude participants who reported seeing an octopus that was never there.
Baseline results
The percentage of detected stimuli was computed to examine the detection level when participants did not allocate their attention to a distractor task. In this baseline study, 152 stimuli were displayed (i.e., a shark and a dolphin were presented to each of the 76 participants). In total, the participants detected 88 stimuli (36 dolphins and 52 sharks), or 57.8% of the stimuli (see Fig. 2).
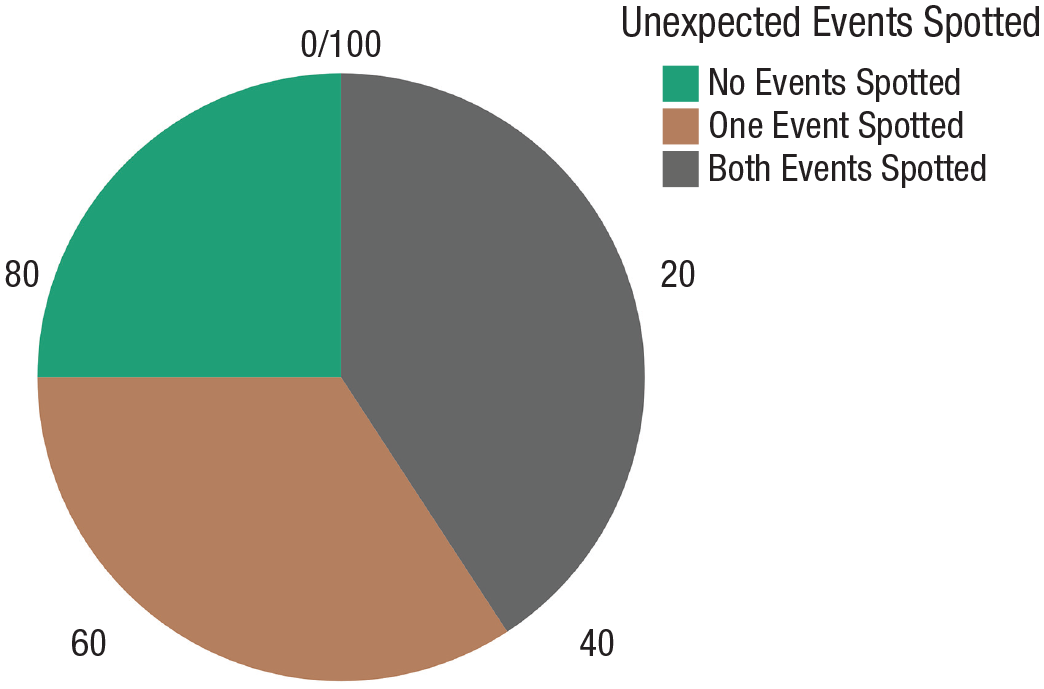
Percentage of participants who detected both, one, or neither of the unexpected events (i.e., shark and dolphin passing by) in the baseline study.
The Experiment
Participants
Two hundred and eighty-eight university students from ISPA gave informed consent and participated voluntarily in the study. However, 32 participants were excluded from the final sample because of a software problem in recording the data; failing the control question (see Procedure section); or having reported diagnosed attentional problems, noncorrected vision impairments, or anxiety-related disorders. Hence, 256 participants, aged 18 to 40 years (
In line with previous research on human emotion-related chemosignals (e.g., de Groot et al., 2015; Gomes et al., 2023; Kamiloğlu et al., 2018), all the participants were heterosexual, Caucasian, right-handed, nonsmoking women. During data collection, participants reported no neurological or psychiatric disorders, chronic respiratory diseases, allergic reactions, illnesses, or colds. Females were recruited because they have been reported to be more sensitive toward emotional signals (Pause et al., 2020) and have a superior sense of smell compared to male participants (e.g., Brand & Millot, 2001). Moreover, earlier research showed that women perceive male sweat differently as a function of their and donors’ sexual orientation (e.g., Martins et al., 2005). Thus, only heterosexual women were included as receivers in the present study.
The procedure was approved by the host institution’s ethics committee and was conducted following the Declaration of Helsinki guidelines and the American Psychological Association standards.
Materials
Body odors collected in two distinct emotion conditions (i.e., fear and rest states) were used in the present study (i.e., olfactory stimuli). The sweat samples were collected at ISPA following the procedures commonly reported in the literature (e.g., de Groot et al., 2015; Gomes et al., 2023; Kamiloğlu et al., 2018).
Twenty-three heterosexual, nonsmoking, Caucasian Portuguese males, aged between 18 and 36 (
Fear and rest states were induced in sweat donors by exposing them to two sets of film clips that had been used with the same aim in previously published research (Gomes et al., 2020, 2023; Silva et al., 2020). 1 Nonwoven absorbent pads (70% viscose, 30% polyester; Wells, Sonae SA, Portugal) were placed in participants’ armpits to collect their sweat during the two emotion-inducing sessions.
In line with previous studies (e.g., de Groot et al., 2015; Gomes et al., 2023; Kamiloğlu et al., 2018), the emotion manipulation in each session was confirmed by quantifying the sweat produced by subtracting the pads’ initial weights from the pads’ weight after the sweat collection, and by asking participants to rate their feelings on sliders ranging from 0 to 100 (i.e., to what extent they felt angry, fearful, disgusted, sad, surprised, neutral, calm, happy, and amused while watching the film clips; for these data, please see the Supplemental Material).
Moreover, as in earlier research (e.g., de Groot et al., 2015; Gomes et al., 2023; Kamiloğlu et al., 2018), donors followed a strict protocol involving several dietary, hygienic, and social restrictions on the 2 days preceding the sampling session to prevent sweat contamination (for details of the sweat collection, see Gomes et al., 2020).
After finishing the sweat collection, each pad was cut into eight pieces. Then, to reduce the effects of possible interindividual differences among donors, four pad pieces from four different donors (randomly selected) were combined to create what we termed a “super-donor.” These super-donors were the olfactory stimuli presented to the receiver participants. Notably, the same combination of donors was used to create fear and the rest super-donors. The olfactory stimuli were stored in amber vials at −80 ºC until an hour before the experimental session. Each super-donor was used four times, being conserved again at −80 ºC after each experimental session.
Procedure
The experiment was conducted in a stand-alone work area. As in previous research (e.g., de Groot et al., 2015; Gomes et al., 2023; Kamiloğlu et al., 2018), the experimenter was a female to avoid mood alterations in the receiver participants (who were all female) from the presence of a male (Jacob et al., 2001).
The super-donor samples were removed from the freezer 1 hour before the data-collection session started. A numerical code identified the emotional condition of each super-donor so that neither the experimenter nor the participant would be aware of the emotional condition in each data-collection session (i.e., a double-blind experiment).
Similar to the procedure in the baseline study, participants gave informed consent and then filled in a sociodemographic questionnaire (e.g., age, sexual orientation) to confirm whether they were eligible to participate. Eligible participants were escorted to the experimental session and randomly assigned to one of the body-odor conditions (i.e., fear chemosignals or rest body odors). They were asked to wear a Pico Neo 3 Pro Eye virtual-reality headset (Pico Technology); they adjusted it to fit, positioned their chin on the chin rest, and listened to the instructions for the experimental task. A vial containing the correct body odor assigned to that session was placed 2 cm away from the participants’ nostrils (see Fig. 3) and was opened by the experimenter. Participants received verbal instructions to start the task. The procedure was the same as in the baseline study; participants were instructed to count how many morsels of food the bluefish consumed.

The experimental setup. Participants were asked to wear a virtual-reality headset and place their chin on a chin rest. A metal claw held the vial containing the super-donor sample 2 cm away from the participants’ noses.
As in the baseline study, at the end of the experimental task participants were instructed first to report how many bluefish they saw snapping up the food morsels and then, in a randomized order, to state whether they saw a shark, a dolphin, and an octopus during the task. Once again, the octopus question was a control question to exclude participants who reported seeing the octopus who was never there. A video of the task can be seen at https://osf.io/me9hv. The experimental procedure took approximately 15 min, and participants received monetary compensation (€5) or course credits.
Statistical analysis
To assess whether participants were similarly engaged in the experimental task in both body-odor conditions, and to compensate for the fact that the data were not normally distributed, we used a Mann-Whitney test. Body-odor condition was a factor, and the number of bluefish that participants saw grabbing the food was the dependent variable.
To examine whether fear chemosignals (vs. rest body odors) increase the detection of the task-nonrelevant stimuli, we used a Poisson regression (i.e., a generalized linear model with a Poisson distribution and a log link). Body-odor condition was a factor, and the number of detected task-nonrelevant stimuli (i.e., 0, 1, or 2 stimuli detected) was the dependent variable. All statistical analyses were performed using JASP (version 0.17.1.0; JASP Team, 2023).
Results
A Mann-Whitney test was performed to evaluate whether the reported number of bluefish eating the food differed as a function of body-odor condition. The results indicate that both participants in the fear-chemosignal condition (
Concerning the detection of the task-nonrelevant stimuli, the Poisson regression model indicates that including the body-odor condition significantly improves the model fit, χ2(1) = 6.10,
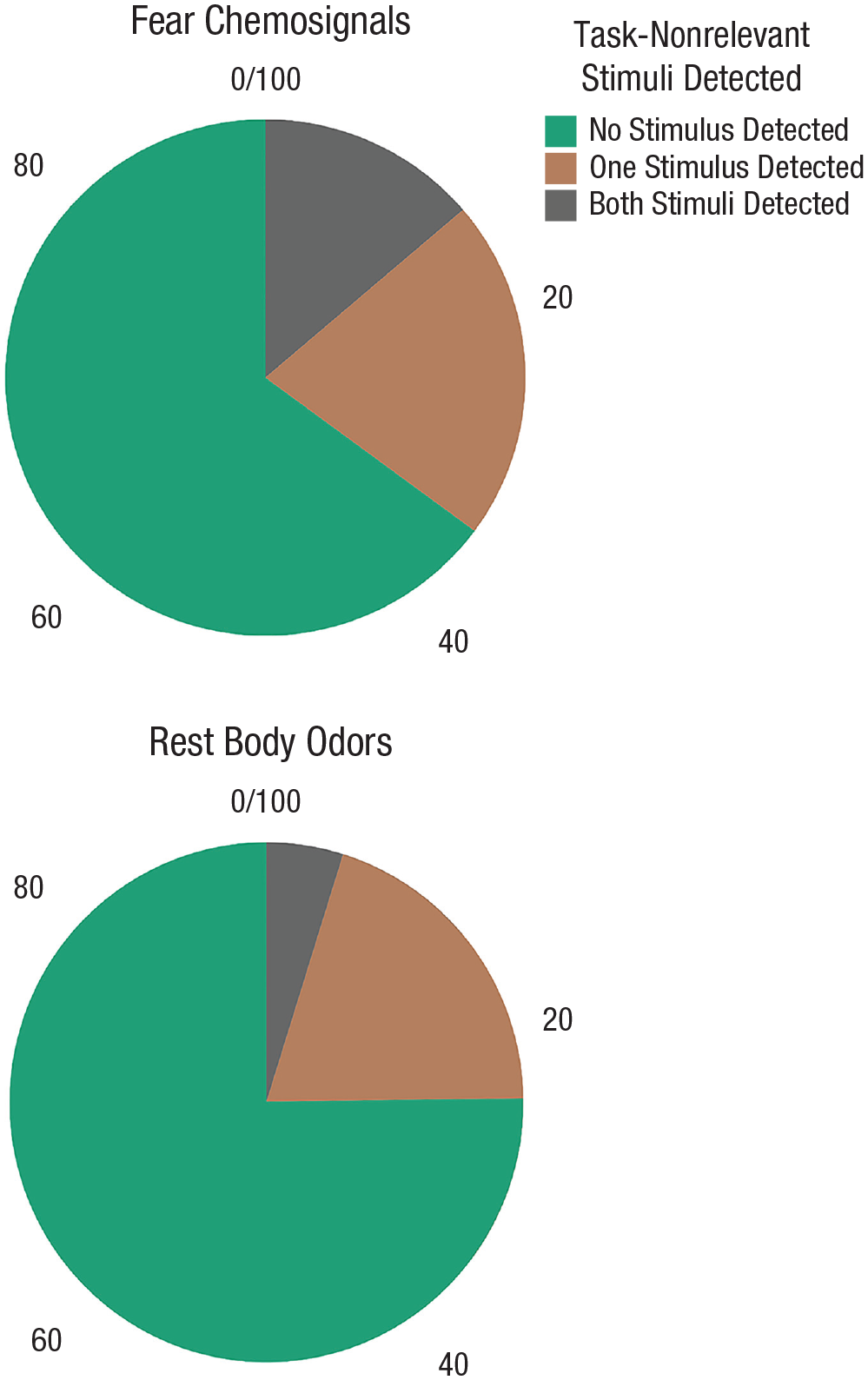
Percentage of participants per body-odor condition who detected both, one, or neither of the task-nonrelevant stimuli in the main experiment.
Notably, the introduction of the counting task resulted in inattentional blindness, significantly reducing the detection of stimuli irrelevant to the task (overall, only 19.7% of the stimuli were detected) when compared to the detection rate in the baseline study (where 57.9% of the stimuli were detected). However, as expected, our results showed that inattentional blindness was significantly more evident in the rest-body-odor condition (only 14.8% of the stimuli were detected). By contrast, exposure to fear chemosignals reduced it significantly; only 24.4% of the stimuli were detected successfully.
Discussion and Conclusions
Fear chemosignals significantly increase the detection of unexpected stimuli relative to the rest-body-odor condition. Compared to the rest condition, the increase in detection in the fear-chemosignal condition is about 10%, which, although a small number, would be an incredibly significant reduction when translated to real-life contexts, as we argue below. These findings suggest that vigilance and increased sensory acquisition activated by fear chemosignals (e.g., de Groot et al., 2012, 2014b; Gomes et al., 2020, 2023; Gomes & Semin, 2021) enhance the detection of unexpected objects in a dynamic physical environment. Moreover, these findings suggest that the unexpected stimuli detected need not be valence-consistent with the emotion induction for the sweat collection, as is often assumed (i.e., fear chemosignals enhance the perception of fear, but not other emotion-related facial expressions; Kamiloğlu et al., 2018). Arguably, at least one task-nonrelevant stimulus (i.e., the dolphin) was neither negative nor threat- or fear-related. Hence, our findings seem to suggest that the activated state of sensory acquisition with the chemosignals does not need to converge with the stimuli used (i.e., negative) to produce the chemosignals. This means that the state induced in the receiver of fear chemosignals is likely to be a state of general vigilance rather than one framed by the emotion-inducing properties (fear) of the stimuli used during sweat production. The resulting sweat (chemosignals) activates a generic state of sensory acquisition. This argument remains speculative, and further research is needed employing stimuli that pose distinct threat or fear levels. However, this finding opens a broader way of looking into how chemosignals function on their receivers and invites an examination of the implications for other chemosignals, such as happiness or disgust (see de Groot et al., 2012, 2015).
One limitation of our study is that we did not examine participants’ eye movements. This prevented us from addressing the potential oculomotor changes induced by fear chemosignals and their relationship with the observed reduction in inattentional blindness. For future research to delve into this phenomenon, it would be useful to incorporate eye-tracking measures. This will help elucidate the underlying processes driving the observed effects of fear chemosignals in enhancing attention.
We conducted this research by bringing together two separate areas of study that had not been studied before. Our findings suggest that fear chemosignals substantially reduce inattentional blindness, opening the door for research with a broad range of practical implications. This is particularly relevant for tasks that demand high levels of sustained vigilance, such as air-traffic control, driving, and reviewing radiology exams, to name a few (e.g., Blachman & Proschan, 1959; Williams et al., 2021). Decreasing the issue of inattentional blindness in these tasks, even by a small percentage, would significantly enhance the preservation of human lives and resources.
However, investigating the practical implications of fear chemosignals in enhancing vigilance, particularly over extended durations, should also examine potential negative outcomes. For instance, studies on loneliness have posited that it heightens vigilance, which could, when experienced for more extended periods, lead to issues like cardiac problems (see Hawkley & Cacioppo, 2010). Therefore, whether intensifying vigilance through prolonged exposure to fear chemosignals might yield comparable adverse effects is a question that future researchers should address, focusing on the practical impacts of this olfactory stimulus in improving human attention.
An unexamined and important question is how long the effectiveness of fear chemosignals lasts. To our knowledge, there is no systematic analysis of the duration of the efficacy of emotion-induced chemosignals. There are also no studies documenting how prolonged exposure to fear chemosignals affects the state of sensory acquisition. One can speculate that extended exposure effects on sensory acquisition are either temporary or that sensory acquisition becomes habituated. These are important questions from the current research that should be addressed.
A related concern is the generalizability of our findings across situations that could induce inattentional blindness. The question is whether the type of experimental ecology and the body-odor manipulation we have used generalize to other situated ecologies (e.g., driving behavior or air-traffic-control contexts). This will remain an open question because the parameters of different situations may activate attentional resources differently. However, fear chemosignals may always enhance sensory acquisition unless the situated ecology itself activates a ceiling effect in terms of sensory acquisition. The other parameter is culture: Does the fear-chemosignals effect we have noted in this experiment have the same influence across different cultural samples? Research has shown that fear chemosignals have comparable effects in several populations, such as Dutch (e.g., de Groot et al., 2012), Chinese (de Groot et al., 2018), and Portuguese samples (Gomes et al., 2020). Although the cultural background of participants in these experiments appears not to be problematic, the question of participant sex remains another potential concern regarding generalizability. We know, for instance, that male and female samples do not always respond to emotion-related body odors in the same way (e.g., de Groot et al., 2014a). Generally, females are more sensitive to the differential effects of emotion-related body odors (e.g., Pause et al., 2010). This is another possible limiting condition that must be considered.
A further important issue is emotion-induced sweat production. This is a very time-consuming and expensive process, and a synthetic reproduction of fear sweat would be the obvious solution. For this, a careful chemical analysis of the composition of the chemical compounds of fear-induced sweat would be necessary should one wish to reap the applied benefits of the current research. A significant challenge is that emotion chemosignals (e.g., fear) often include hundreds, if not thousands, of molecules. If it were possible to analyze the chemical compounds of, for instance, fear chemosignals, then such a synthetic compound would save an incredible amount of time and money. It would potentially free researchers from the limitations of taking samples from parts of the body over time (currently researchers typically collect odors on pads under the armpits). Such synthetic solutions would cut the time of sweat collection considerably and would be economically viable. Of course, researchers would have to demonstrate that synthetically produced fear sweat has the same properties as naturally produced fear sweat.
Supplemental Material
sj-docx-1-pss-10.1177_09567976231213572 – Supplemental material for Investigating Inattentional Blindness Through the Lens of Fear Chemosignals
Supplemental material, sj-docx-1-pss-10.1177_09567976231213572 for Investigating Inattentional Blindness Through the Lens of Fear Chemosignals by Gün R. Semin, Michael DePhillips and Nuno Gomes in Psychological Science
Footnotes
Acknowledgements
The authors would like to thank Andreia Ribeiro, Andreia Santiago, Ana Luís, Carina Parshotam, and Mariana Brazão for their data collection assistance and Miguel Benrós for technical support.
Transparency
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
