Abstract
The brain processes short-interval timing but also allows people to project themselves into the past and the future (i.e., mental time travel [MTT]). Beta oscillations index seconds-long-interval timing (i.e., higher beta power is associated with longer durations). Here, we used parietal transcranial alternating current stimulation (tACS) to investigate whether MTT is also supported by parietal beta oscillations and to test the link between MTT and short intervals. Thirty adults performed a novel MTT task while receiving beta and alpha tACS, in addition to no stimulation. Beta tACS corresponded to a temporal underestimation in past but not in future MTT. Furthermore, participants who overestimated seconds-long intervals also overestimated temporal distances in the past-projection MTT condition and showed a stronger effect of beta tACS. These data provide a unique window into temporal perception, showing how beta oscillations may be a common mechanism for short intervals and MTT.
Keywords
Sensing the passage of time is a ubiquitous experience and a building block for many behaviors in daily life, such as speaking, driving a car, and performing physical activities (Paton & Buonomano, 2018). Timing is also critical for motor programming, sensory processing, and most forms of learning. Indeed, we typically combine present information and past experiences to anticipate future events (Paton & Buonomano, 2018). Thus, we can sense and represent the passage of time over a wide range of temporal scales, from a few hundred milliseconds to days or years (Buonomano, 2007). Psychophysical studies show that encoding temporal information in the milliseconds/seconds range is achieved via duration-tuned mechanisms (i.e., changes in neurons’ discharge rate as a function of duration; Cadena-Valencia et al., 2018; Merchant et al., 2013). This is supported by research on endogenous brain oscillations as a mechanism underlying timing networks in the brain. Most studies indicate a supramodal role for beta oscillations (~20 Hz) in the production and perception of temporal intervals (Fujioka et al., 2012; Kononowicz & van Rijn, 2015; Merchant & Bartolo, 2018; Wiener et al., 2018). Beta power indexes temporal durations such that higher power corresponds to longer durations (Bartolo & Merchant, 2015). Studies on both humans and monkeys show that in time-reproduction paradigms, in which participants copy the duration of an event as accurately as possible, trial-by-trial beta power positively correlates with the length of the produced duration (Merchant & Bartolo, 2018; Wiener et al., 2018). In such time-reproduction tasks, beta power is stronger for longer durations, suggesting that it reflects the length of the produced duration (Bartolo et al., 2014; Bartolo & Merchant, 2015; Kononowicz & van Rijn, 2015). Beta power also reflects the subjective length of a duration (Kononowicz & van Rijn, 2015), and beta oscillations may serve as a memory standard to which different durations are compared (Wiener et al., 2018).
Besides sensing the passage of time in the range of milliseconds/seconds range, humans can also mentally project themselves into the past and the future. This ability is termed
Here, we aimed to clarify to what extent MTT relies on beta-based mechanisms similar to those underlying the processing of short durations. To do so, we used transcranial alternating current stimulation (tACS), which allowed us to modulate cortical oscillations while participants performed a novel MTT task. We tested the role of target beta oscillations, which are thought to mainly support seconds-long-interval timing, relative to alpha oscillations, which acted as a control condition because they are thought to support mental imagery more strongly (Bartsch et al., 2015; Xie et al., 2020). In addition to a nonstimulation control condition (sham tACS), beta and alpha tACS were delivered over the posterior parietal cortex, a key area involved in both MTT, short-interval reproduction, and visual mental imagery tasks. To measure participants’ performance in MTT, we developed a new psychophysics task that allowed us to precisely account for self-projection in time. Participants saw face stimuli of different ages, one at a time. Each face was presented with a short phrase describing a particular life event, commonly happening in middle age. Participants performed a two-alternative forced choice: In the past-projection condition, they indicated whether it was likely or unlikely that the person pictured in the stimulus had experienced the life event 10 years previously; in the future-projection condition, they indicated whether it was likely or unlikely that the person pictured in the stimulus would experience the event 10 years in the future. We hypothesized that MTT might rely on parietal beta oscillations because of their supramodal role in the production and perception of short temporal intervals, as opposed to alpha oscillations, which are more relevant for mental imagery. If that was the case, we would expect a stronger modulation of MTT after beta tACS.
Statement of Relevance
We sense the passage of time over a wide range of temporal scales, from seconds to years. Cortical oscillations in the beta band (20 Hz) process seconds-long temporal information. Whether a similar beta-based mechanism is also used for computing longer temporal distances, for instance when we engage in mental time travel (MTT), is currently unknown. By using brain stimulation to exogenously modulate parietal oscillations, we found that beta oscillations are crucial for MTT, too. When we modulated these oscillations, participants underestimated temporal distances in past but not in future MTT. Moreover, participants who overestimated seconds-long intervals also overestimated temporal distances in past MTT and showed a stronger effect of brain stimulation. Our findings show that beta oscillations are a common mechanism for estimating short and longer temporal distances, opening a new window onto the way the brain mentally projects the self in time.
Open Practices Statement
The study reported in this article was preregistered on ClinicalTrials.gov (identifier: NCT04582994; https://clinicaltrials.gov/ct2/show/NCT04582994). Experimental stimuli are available from the Corresponding Author on request. Data have been made publicly available on OSF and can be accessed at https://osf.io/fbdgs/.
Experiment 1
Method
Participants
Thirty stimulation-compatible (Tavakoli & Yun, 2017) participants (age:
Stimuli
We used 30 face identities, 15 women and 15 men, with neutral expressions taken from the Chicago Face Database (Ma et al., 2015). All stimuli in the photos ranged in age between 42 and 50 years. Using FaceApp (https://www.faceapp.com/) and
Experimental procedure
Participants underwent three experimental sessions at least 24 hr apart (Fig. 1a). In each session, they performed the MTT task while receiving continuous tACS at one of two possible frequencies, target beta (22 Hz) and control alpha (10 Hz) oscillations, in addition to a control nonstimulation condition (sham tACS; 4 Hz).
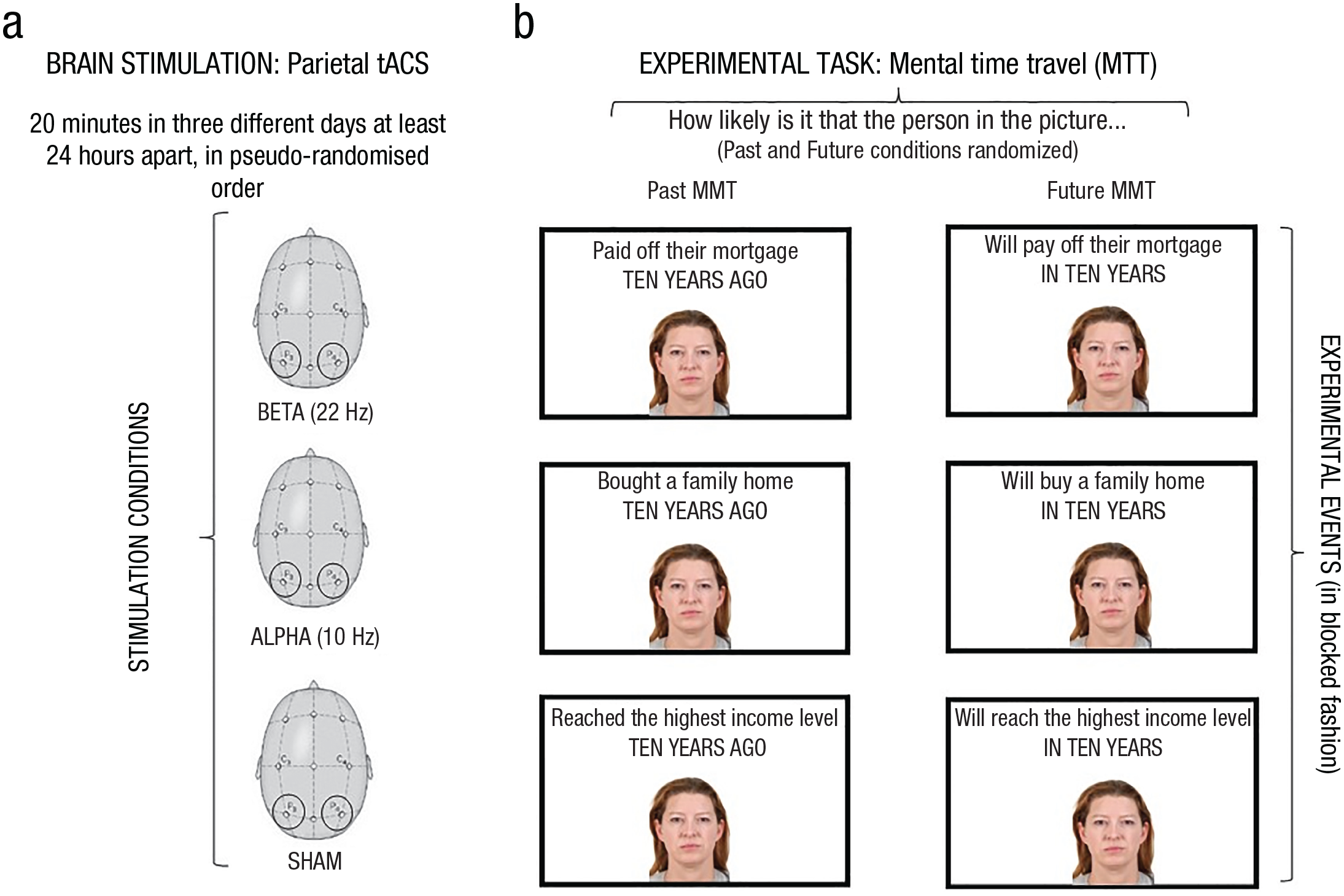
Stimulation conditions (a) and experimental paradigm (b). In (a), circles indicate electrode locations. tACS = transcranial alternating current stimulation.
Transcranial alternating current stimulation
We delivered tACS using a battery-powered direct-current stimulator (neuroConn DC-Stimulator Plus, Ilmenau, Germany) through a pair of rubber electrodes (5 cm × 7 cm) enclosed in saline-soaked sponges and fixed on the head by elastic bands. Based on the standard 10-20 electroencephalography (EEG) procedure, the electrodes were positioned on the target areas corresponding to parietal regions (i.e., P3 and P4). For beta and alpha frequencies, participants were stimulated with an in-phase (0°) alternating-current mode for 20 min for each frequency with a fade in/out period of 20 s and a current strength of 1,500 μA. In the sham condition, the stimulation consisted only of the fade in/out period (40 s in total), but participants were tested in the same setting as in the conditions in which they received the real stimulation. The order of stimulation was pseudorandomized across participants (Fig. 1a). Participants and one of the experimenters were never told whether they received real or sham stimulation. None of the participants was able to discriminate between the sham and an active stimulation condition, as verified on a validated questionnaire that assessed participants’ sensation during the stimulation period (Borghini et al., 2018; see “Supplementary Results” at https://osf.io/24cs7/). Moreover, no participants reported phosphenes during alpha and beta tACS.
MTT task
In the MTT task, participants saw face stimuli of different ages, presented one at a time in the center of a computer screen, each of which was preceded by an interstimulus interval of 500 ms. Each face was presented with a short phrase describing a particular life event, commonly happening in middle age: (a) “to pay off the mortgage,” (b) “to buy a family home,” and (c) “to reach the highest income level.” Participants performed a two-alternative forced-choice task in two conditions: In the past-projection condition, they indicated whether it was likely or unlikely that the person pictured in the stimulus had experienced the presented life event 10 years previously (i.e., “Is it likely/unlikely that they paid off the mortgage ten years ago?” “. . . bought a family home ten years ago?” “. . . reached the highest income level ten years ago?”). In the future-projection condition, participants indicated whether it was likely or unlikely that the person pictured in the stimulus would experience the event 10 years in the future (i.e., “Is it likely/unlikely that they will pay off the mortgage in ten years?” “. . . will buy a family home in ten years?” “. . . will reach the highest income level in ten years?”). Participants responded as quickly and as accurately as possible by pressing either the A or L key on the keyboard (order counterbalanced across participants; Fig. 1b).
The face stimuli and events were presented onscreen for a maximum of 4 s or until participants responded. The three events were administered in three different blocks of 140 trials each. The order of blocks was pseudorandomized across participants and across experimental sessions. Each block consisted of 70 past-projection trials and 70 future-projection trials presented in a randomized order. Thus, a different sample of 10 face identities at all seven age levels were presented in each block, both in the past- and future-projection conditions.
Control task
Our core aim was to test whether projecting ourselves in time may be modulated by beta or alpha oscillations. However, changes in performance of the MTT task may also be observed whether these oscillations simply modulate face processing, which may result in participants misjudging the age of the face stimuli. To rule out this possibility, we also asked participants in each experimental session to perform a control task while they received brain stimulation. In this control task, participants used the numerical keypad on the keyboard to indicate as quickly and as accurately as possible the perceived age of 30 face stimuli presented one at a time. The face stimuli were the same as those used in the MTT task, with a different sample of 10 identities presented at all seven age levels in each experimental session. The order of the MTT task and the control task was counterbalanced across participants.
Results
Data analysis
For each participant, we computed the proportion of “likely” responses as a function of the stimuli’s veridical age, stimulation condition, and time-projection condition. To study the effect of brain stimulation on the MTT task, we used a generalized linear model on the proportion of “likely” responses, with age levels (from youngest to oldest,
Effect of tACS on MTT
As expected, the proportion of “likely” responses changed significantly as a function of the age of the face stimuli, Wald χ2(6,
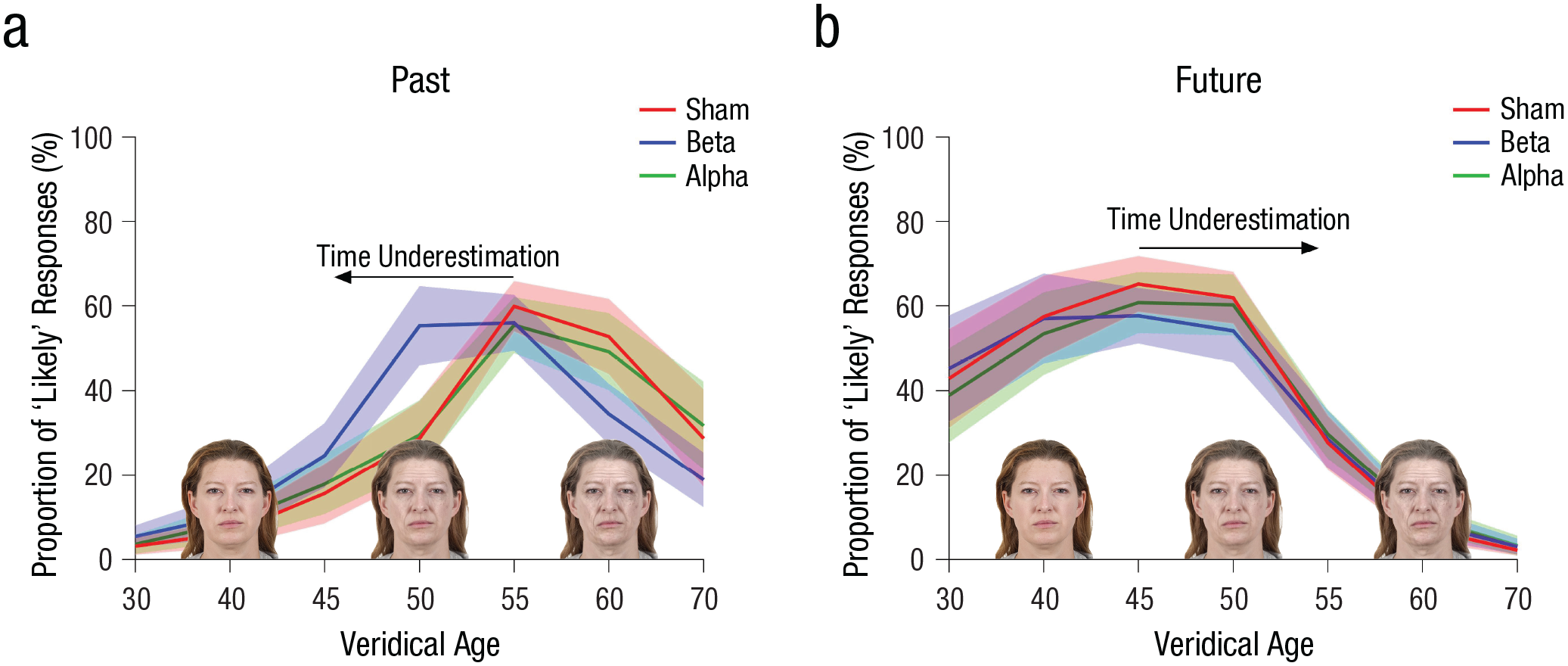
Transcranial alternating current stimulation (tACS) modulation on mental time travel in Experiment 1: proportion of “likely” responses as a function of the face stimuli’s veridical age (from 30 to 70 years) and tACS stimulation condition (beta, alpha, and sham tACS), separately for the past (a) and future (b) conditions. Shaded regions indicate 95% confidence intervals.
To test for the absence of interaction in the future-projection condition, we performed a repeated measures Bayesian analysis of variance (ANOVA) on the proportions of “likely” responses in the future, with type of stimulation (sham, beta, and alpha tACS) and age levels as factors. The Bayesian analysis revealed a Bayes factor (BF) of 276.892 in favor of the null model of no interaction between stimulation condition and age (BF01 = 276.892). In other words, in the future-projection condition, the data were 276.892 times more likely under the null model (i.e., a model not including the effects of the interaction between stimulation condition and age), compared with the alternative model including the interaction.
The pattern of results obtained from the generalized linear model suggests that in the past-projection condition, the peak of the proportion of “likely” responses shifted toward a younger age following brain stimulation at the beta frequency. In other words, beta tACS may have led to an underestimation of temporal distances: On average, participants indicated younger faces as more likely to have experienced an event 10 years in the past. To test this possibility, we fitted a Gaussian curve on the proportions of “likely” responses in the past-projection conditions of sham, alpha, and beta tACS as a function of faces’ veridical age (from 30 to 70 years). The Gaussian function was a good fit to the data (sham tACS condition: mean
Because beta tACS led to an underestimation of time, we examined whether these effects were stronger in participants who overestimated time in the sham tACS condition. To obtain a measure of the effect of beta tACS for each participant, we subtracted the mean of the fitted Gaussian curve in the beta tACS condition from the mean of the fitted Gaussian curve in the sham tACS condition (a greater difference indicated a larger effect of beta tACS). The same procedure was followed for the alpha tACS condition to obtain a measure of the effect of alpha tACS. As expected, the mean in the sham tACS condition significantly and positively correlated with the effect of beta tACS—Pearson’s
Finally, we tested whether age estimation changed following alpha or beta stimulation. An ANOVA with age level and stimulation condition as factors showed only a significant main effect of age level,
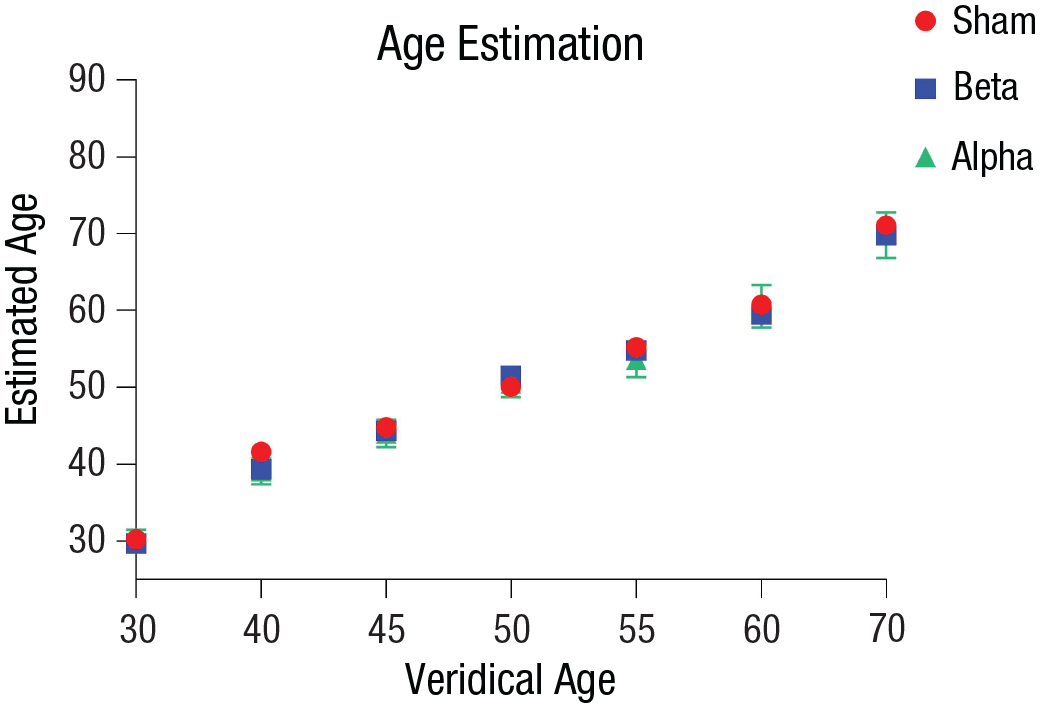
Transcranial alternating current stimulation (tACS) modulation on age estimation in Experiment 1: participants’ age estimation of face stimuli as a function of their veridical age in each tACS stimulation condition (alpha, beta, and sham tACS). Error bars indicate 95% confidence intervals.
Overall, these data are consistent with an underestimation of time in the past MTT during beta tACS, suggesting that beta oscillations are crucial for computing temporal distances during past MTT, in addition to their role in processing short durations. To further test the link between past MTT and time processing, we conducted a second experiment in which the same group of participants performed a time-reproduction task. If processing of seconds-long-interval timing and longer temporal distances are linked, individual differences in past MTT may be more likely correlated with individual variability in time perception. Moreover, the reproduced time intervals may also be more likely to correlate with the effect of beta tACS, similar to the relationship between overestimation of temporal distances in MTT and the beta tACS effect.
Experiment 2
Method
The same group of participants who took part in Experiment 1 was recruited for Experiment 2. For all participants, Experiment 2 was performed during the first experimental session before the stimulation started. Participants performed a time-estimation task that consisted of two phases. In the encoding phase, participants saw a blue square presented in the center of a white screen for a variable duration (1,600, 1,800, 2,000, 2,200, or 2,400 ms). Following an established procedure (Frassinetti et al., 2009; Magnani et al., 2013; Oliveri et al., 2013), we instructed participants to pay attention to the duration of the stimulus without adopting any strategies, such as counting. In the estimation phase, following a 500-ms interstimulus interval, a red square appeared onscreen in the same position as the blue one. Participants pressed the space bar on the keyboard when they judged that the red square had been onscreen for the same length of time as the blue one had been.
Results
To explore how participants performed the time-estimation task, we conducted a one-way ANOVA on the mean reproduced times with intervals (1,600, 1,800, 2,000, 2,200, or 2,400 ms) as within-subjects factors. As expected, there was a main effect of time interval,
For each participant, we also calculated the average estimated time interval and subtracted it from the average veridical time (2,000 ms) to obtain an index of participants’ time overestimation. We then correlated this index with the mean of the Gaussian curves fitted in the past-projection/sham tACS condition. We found that overestimation of time intervals significantly and positively correlated with the mean of the Gaussian function in the past-projection/sham tACS condition. Pearson’s
Next, we tested whether there was a stronger brain-stimulation effect in participants who overestimated time intervals, which would indicate that the processing of short-interval timing was truly linked to processing longer temporal distances in MTT. This would resemble the stronger beta tACS effect found in Experiment 1 in participants who overestimated temporal distances in MTT. Crucially, we found that the effect of beta tACS correlated with the estimated time intervals, such that the larger the effect of beta tACS, the longer the estimated time intervals—Pearson’s
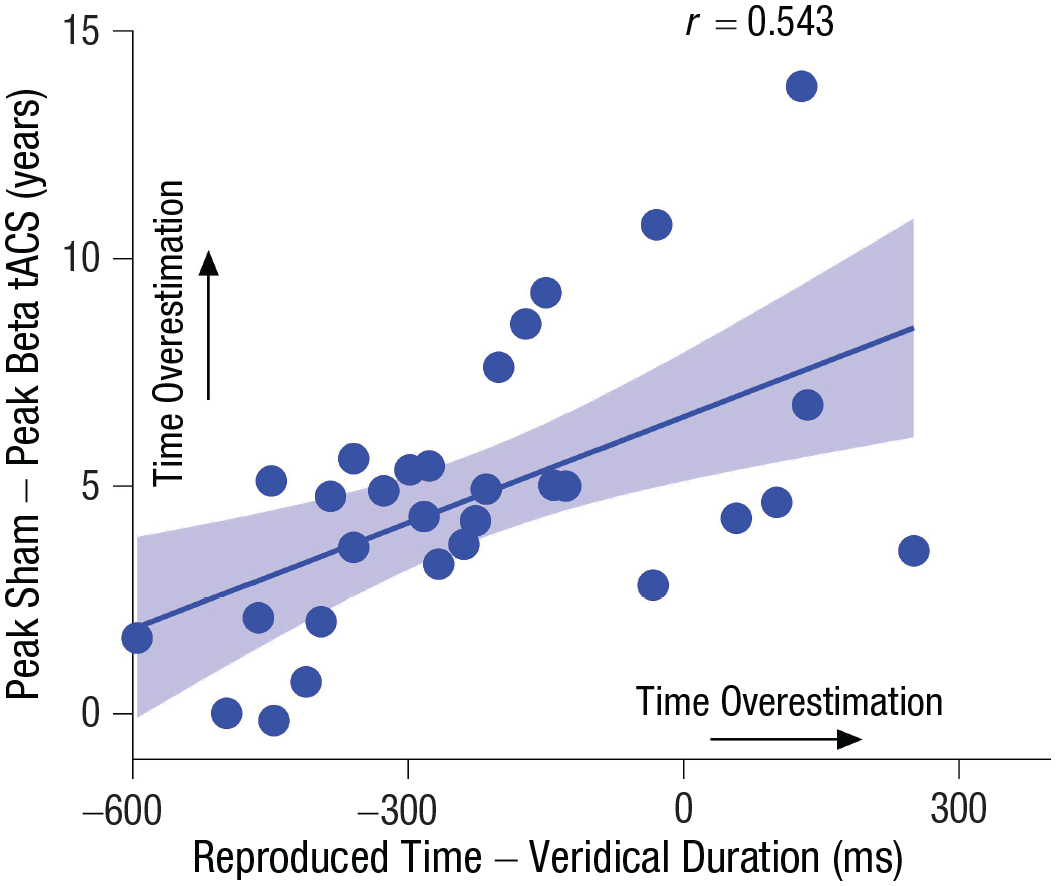
Correlation between overestimation of seconds-long-interval timing and the effect of beta transcranial alternating current stimulation (tACS) in Experiment 2. The solid line represents the best-fitting regression. The shaded region reflects the 95% confidence interval.
Experiment 3
Method
We conducted another experiment in order to (a) replicate the correlation between short-interval timing and past MTT and (b) further explore the relationship between future MTT and short intervals. To do so, we recruited another group of 30 participants (age:
Results
Following the same procedure as in Experiment 2, we computed the average estimated time interval and subtracted it from the average veridical time (2,000 ms) to obtain an index of participants’ time overestimation. For each participant, we then calculated the mean of the Gaussian distribution fitted on the proportion of “likely” responses in the past-projection condition (mean
Discussion
We showed that MTT into the past is modulated by parietal beta frequencies, such that participants underestimated temporal distances following beta tACS. This suggests that beta oscillations are crucial for computing temporal distances during past MTT, in addition to their role in processing short durations. Beta oscillations have emerged as a mechanism for coordinating timing functions in the human brain (Kononowicz & van Rijn, 2015; Wiener et al., 2018). Beta power indexes temporal durations, such that higher power is linked to longer durations (Bartolo et al., 2014; Bartolo & Merchant, 2015). More recently, Wiener and colleagues (2018) reanalyzed their EEG data from an auditory temporal bisection task, in which participants classified an auditory stimulus as long or short (Wiener & Thompson, 2015; Wiener et al., 2012), and found that beta power covaried linearly with the duration of the previous trial, with higher beta power for longer durations. This was taken to suggest that beta oscillations index the duration each presented value was compared with, serving as a memory standard to which different durations are categorized as longer or shorter. Our results indicate that beta frequencies are also crucial for computing longer temporal distances when the self is projected into the past. This may suggest a common computational principle between time processing of short durations and longer temporal distances involved in MTT, although we did not test the role of beta oscillations in short-interval timing in the current study.
Experiment 2 explored the link between temporal processing of short durations and MTT in more detail. We found a correlation between the reproduced time intervals of a few seconds in a temporal-reproduction task and participants’ responses in the past-projection MTT task. Participants who overestimated temporal intervals in the seconds range also overestimated temporal distances in the past-projection/sham tACS condition, indicating people pictured in older face stimuli as more likely to have experienced a given life event. Moreover, participants who overestimated temporal intervals of a few seconds and overestimated temporal distances in the MTT task also showed a stronger effect of beta tACS in the past-projection MTT task. Thus, participants who overestimated temporal periods may have had longer memory standards in the range of both seconds and years. Beta frequencies may play a key role in encoding and building time priors involved in processing time both in the range of a few seconds and for mentally travelling in time. Critically, beta tACS led to an underestimation of time, consistent with results of Wiener and colleagues (2018), who found an underestimation of short temporal intervals during beta tACS. Both results can be explained in terms of tACS reducing beta power and therefore leading participants to a shorter memory standard for time durations. However, as Wiener and colleagues (2018) noticed, the impact of tACS on oscillatory power is highly state dependent, and it is yet to be demonstrated whether tACS enhanced or lowered power in the current study (see also Silvanto et al., 2008).
It may be argued that our task did not necessarily require participants to mentally travel in time, so instead they may have used a strategy based on the age of the face stimuli to perform the task. However, such a strategy would not explain the tACS modulation itself. Indeed, our control experiment showed that tACS did not modulate participants’ ability to judge the stimuli’s age. Moreover, the past- and future-projection conditions had completely different distributions of responses, although these two conditions were presented randomly and not in a blocked fashion. Therefore, it is implausible that participants adopted the same age heuristic for both conditions. For these reasons, we think that time underestimation during beta tACS is still the best explanation of our data.
A key aspect of our study is that beta stimulation targeted the posterior parietal cortex. Even if tACS spatial resolution is notoriously poor (Chai et al., 2018), our data are consistent with those of previous studies showing that the posterior parietal cortex is crucial for both seconds-based time processing and MTT (Hayashi et al., 2015; Magnani et al., 2013). For instance, time intervals are underestimated after the right parietal cortex is inhibited with transcranial magnetic stimulation (Oliveri et al., 2009; Wiener et al., 2012), and a similar time underestimation has also been observed in patients with right parietal brain damage (Magnani et al., 2013; Oliveri et al., 2013). More recent studies based on functional MRI adaptation have shown that duration-tuned neural populations in the right parietal cortex reflect our subjective experience of time rather than the actual physical duration of stimuli (Hayashi & Ivry, 2020). In a similar vein, the posterior parietal cortex is also a key area for MTT (Anelli et al., 2018; Arzy et al., 2009; Casadio et al., 2022). Patients with right parietal damage underestimate time intervals and are impaired at processing past MTT, being unable to distinguish between events that already happened and those that are yet to happen (Anelli et al., 2018). Taken together, these studies indicate that the posterior parietal cortex is a common node for both time processing of short durations and past MTT.
We are not, however, claiming that MTT and time estimation are exclusively supported by parietal beta oscillations. MTT comprises many cognitive processes, involving a large network of brain areas and different cortical oscillations. In addition to beta oscillations, frontal theta oscillations, probably phase locked to hippocampal theta, are sensitive to temporal durations (Liang et al., 2021). Our study suggests that, of all the complex cognitive processes involved in MTT and time processing, parietal beta oscillations play a role in computing and comparing temporal quantities of different scales. Future studies will further investigate the exact role of beta oscillations in MTT, for instance by systematically varying the length of the temporal distances or by exploring other neural and computational components of MTT, such as episodic and prospective memory, mental imagery, and self-orientation with respect to the temporal order of different events.
Beta tACS unsuccessfully modulated future MTT. Moreover, consistently with the absence of beta tACS effect in the future MTT, our findings did not show any significant correlation between future MTT and short intervals. This result strengthens our idea that the specific beta tACS effect in past MTT is due to a common role of beta frequencies in processing short interval durations and longer temporal distances. However, given the current data, providing a univocal interpretation to the null effects on future MTT may be premature. The posterior parietal cortex may be mainly involved in the processing of past events, rather than in future MTT. This evidence is in line with an open debate currently focusing on whether brain regions implicated in MTT may contribute differently to past and future self-projection. Future self-projection recruits brain areas such as the dorsolateral prefrontal cortex, the lateral temporal lobe, and the frontopolar cortex (Addis et al., 2007; Schacter et al., 2012), which are usually associated with executive functions or with processing intentions and personal goals (Irish & Piguet, 2013; Weiler et al., 2011). Moreover, neuropsychological studies have shown that lesion or dysfunction of the lateral (Berryhill et al., 2010; de Vito et al., 2012) and ventromedial (Ciaramelli et al., 2021) prefrontal cortex affect future but not past MTT. Therefore, a correct computation of temporal distances, supported by the parietal cortex, may be more relevant for past than future MTT. However, these interpretations should be taken with caution because the neural dissociation between past and future MTT is quite controversial. Moreover, in our task, participants reasoned about events regarding other people and not about their own personal events. Therefore, possible differences between past and future MTT, as well as the relationship between future MTT and seconds-long-interval processing need to be explored further. Finally, future studies with concurrent tACS-EEG recording need to replicate and further test the role of beta oscillations in computing longer temporal distances.
In conclusion, we found that past MTT was modulated by parietal beta tACS, such that participants underestimated temporal distances. Thus, beta oscillations are crucial for MTT, in addition to their role in computing short-interval timing. Moreover, our data provide a link between temporal processing of short durations and MTT because participants who overestimated intervals in the seconds range also overestimated temporal distances in the past MTT task and displayed a stronger effect of beta tACS. Overall, these data build, for the first time, a bridge between the currently distinct literatures on short-interval timing and on MTT, providing a unique window into the way the brain projects the self in time.
Footnotes
Acknowledgements
We thank Mariagrazia Benassi for statistical support and Biagio Dell’Omo and Alfredo Carrelli for help in data collection.
Transparency
Francesca Frassinetti and Marinella Cappelletti equally contributed to the study
