Abstract
Risk preference impacts how people make key life decisions related to health, wealth, and well-being. Systematic variations in risk-taking behavior can be the result of differences in fitness expectations, as predicted by life-history theory. Yet the evolutionary roots of human risk-taking behavior remain poorly understood. Here, we studied risk preferences of chimpanzees (86 Pan troglodytes; 47 females; age = 2–40 years) using a multimethod approach that combined observer ratings with behavioral choice experiments. We found that chimpanzees’ willingness to take risks shared structural similarities with that of humans. First, chimpanzees’ risk preference manifested as a traitlike preference that was consistent across domains and measurements. Second, chimpanzees were ambiguity averse. Third, males were more risk prone than females. Fourth, the appetite for risk showed an inverted-U-shaped relation to age and peaked in young adulthood. Our findings suggest that key dimensions of risk preference appear to emerge independently of the influence of human cultural evolution.
Keywords
Risk preferences contribute to many of the decisions people make—whether to explore new areas in search of food, shelter, or mates; compete for social status; or engage in financial investments, to name but a few (Barseghyan et al., 2013; Brailovskaia et al., 2018; Clark & Lisowski, 2017; Dohmen et al., 2011; Mata et al., 2018; Schonberg et al., 2011; Slovic, 1987). As a key building block in decision processes that shape individuals’ health, wealth, and well-being, risk preference has the potential to influence the entire course of an individual’s life and therefore presents wide-ranging consequences for society. Yet its biological evolutionary roots remain poorly understood, and tests are scant for the proposition that “modern attitudes to risk are, at least in part, the product of biological evolution” (Robson, 1996, p. 397; see also Karlsson Linnér et al., 2019; Robson, 2001).
Here, we investigated whether risk preferences of chimpanzees (Pan troglodytes) are isomorphic to human risk preferences in important respects. Chimpanzees are an ideal reference (The Chimpanzee Sequencing and Analysis Consortium, 2005): They are one of humans’ two closest living relatives; they live in large, mixed-sex social groups; and they have similar developmental stages, with extended infant dependency, a period of adolescence, young adulthood, and long life expectancy of up to 50 years. Chimpanzees occupy a variety of habitats—from savannas to evergreen forests—and face myriad risks in their socioecological environments (de Waal, 1973; Goodall, 1968). There are theoretical reasons to expect evolutionary continuities in risk preferences. According to life-history theory, risk taking differs systematically across stages of life (Stearns, 1992). In the context of reproduction, sexual selection and differential parental investment costs (Trivers, 1972) predict lower levels of risk taking in females, who invest more in offspring and are therefore more discriminating in choosing mates. Among young males, reproductive competition for females is thus intense, and young human adult males are predicted and known to take more risks (young male syndrome; Wilson & Daly, 1985).
How can one study chimpanzee risk taking and what is known about it? Depending on the definition of risk, in psychology, risk preference (attitude) has often been interpreted to refer to the tendency to exhibit behaviors that, though rewarding, involve some potential harms or losses. In economic theorizing, risk preference refers to the willingness to engage in choices that involve higher variance in outcomes (Mata et al., 2018). Here, we will use both definitions. To behaviorally assess risk-taking behavior, comparative researchers have typically adapted human economic-choice paradigms for use in great apes (Rosati, 2017; Santos & Rosati, 2015). In the commonly used setup, subjects make foraging decisions between a safe option (reliable outcome; e.g., one piece of food) and a risky option (variable option with outcome variance; e.g., 50% chance of getting two pieces of food or nothing). Depending on the methodology and the rewards involved, some studies have found that chimpanzees prove to be—like humans—risk averse, consistently preferring the safe over the risky option (Haux et al., 2021; Keupp et al., 2021); other studies suggest, however, that chimpanzees are risk seeking (Haun et al., 2011; Heilbronner et al., 2008; Rosati & Hare, 2011, 2012, 2013). Furthermore, there is some evidence of an increased sensitivity to social risks (see Calcutt et al., 2019; Haux et al., 2021; Rosati & Hare, 2012). Notwithstanding these mixed findings, many questions about chimpanzees’ psychology of risk are unanswered. For instance, does chimpanzees’ willingness to take risks correlate across distinct domains (e.g., interactions with venomous animals, intergroup encounters, hierarchy fights) and distinct measures of risk—and, if so, to what extent? Can such a willingness, generalizing across several domains, be regarded as a traitlike preference? Furthermore, previous studies relied on sample sizes ranging from four to 24 subjects and examined relatively narrow age ranges (see De Petrillo & Rosati, 2021). Such sample sizes risk a relatively low statistical power to detect small to medium age and sex effects. Therefore, another open question is whether age- and sex-dependent behavioral effects, perhaps similar to those commonly observed in humans, will emerge.
In addition to the study of risk preferences, revealed in subjects’ economic choices, we asked long-time caregivers to rate subjects’ willingness to take risks in domains that represent major classes of risks in chimpanzees’ ecology. For instance, caregivers rated behaviors that involve the more psychological definition of risk, that is, behaviors entailing the potential for both rewards and some harms or losses. Consider, for example, the display of competitive behaviors to reach higher ranks in the dominance hierarchy. One question is whether, and to what extent, economic choice behaviors and observed behaviors (rated by caregivers) converge on similar inferences about individuals’ risk preferences.
Statement of Relevance
Risk preference is central to human activity and has the potential to influence the entire course of our lives. However, the evolutionary roots of human risk preference remain poorly understood. We used a multimethod approach to study chimpanzees (N = 86) from infancy to adulthood to determine whether the willingness to take risks in one of humans’ closest living relatives is similar to that in humans. Our results show that chimpanzee and human risk preferences share key structural similarities: Chimpanzees’ willingness to take risks manifests as a traitlike preference that is consistent across domains and measures, chimpanzees are ambiguity averse, male chimpanzees are more risk prone than females, and chimpanzees’ appetite for risk peaks during young adulthood. These evolutionary continuities are likely to reflect adaptations to similar dynamics in human and nonhuman primate life histories.
Other studies have investigated what has been or what could be interpreted as aspects of risk-taking behaviors beyond choice in economic paradigms. These studies involve, for instance, research on inhibitory control (e.g., MacLean et al., 2014; delay of gratification: Beran et al., 1999; expectancy: Stevens et al., 2011; patience: Rosati et al., 2007), temperament and personality (e.g., Herrmann et al., 2011, 2015; King & Figueredo, 1997; boldness: Massen et al., 2013; curiosity: Damerius et al., 2017; anxiety: Coleman & Pierre, 2014), cooperation and competition (e.g., Hare et al., 2000; trust: Engelmann et al., 2015; Engelmann & Herrmann, 2016; Haux et al., 2021), and new environments (e.g., unsafe areas: Hockings et al., 2006; responses to novelty: Forss et al., 2015; Kalan et al., 2019; xenophobia: de Waal, 2012; xenophilia: Tan & Hare, 2017). As it is not yet clear how these constructs and measures relate to each other as well as to risk preference as gauged here—for instance, Carter et al. (2012) showed that current boldness measures may not be interchangeable—we will not further interpret these findings.
Last but not least, our study rests on a cross-sectional design involving animals of different ages from infancy to adulthood. The multimethod approach combines observer ratings (Gosling & Vazire, 2002; McCrae, 1994) with behavioral choice experiments and builds on stated willingness ratings and experimental studies on risk and ambiguity preferences in humans (Dohmen et al., 2011; Ellsberg, 1961). These methodological choices allow us to compare the current results with well-established findings on human risk preferences. We focus on four structural aspects of risk preference. First, to what extent does chimpanzee—like human—risk preference (Frey et al., 2017) manifest as a traitlike preference that is consistent across domains in both behavior and observers’ assessments? Second, are chimpanzees—like humans (Ellsberg, 1961; Rosati & Hare, 2011; Trautmann & van de Kuilen, 2015)—ambiguity averse, that is, less willing to choose options with unknown risks? Third, are male chimpanzees more inclined to take risks than females, mirroring stable differences in human risk preference (Frey et al., 2021)? Fourth, do age differences in chimpanzees’ risk preference mirror those in humans, where risk taking peaks in adolescence and early adulthood and is lower in older age (Frey et al., 2021; Josef et al., 2016; Mata et al., 2016)?
Open Practices Statement
All data are included in this article or in the Supplemental Material available online. All code is available from the authors on reasonable request. The design and analysis plan for this study were not preregistered.
Method
Subjects
Eighty-six chimpanzees (47 females; age = 2–40 years) participated in the study (this sample represents all chimpanzees living in the sanctuaries; see Table S1 in the Supplemental Material for more detailed information about the subjects). Chimpanzees have access to large tracts of outdoor enclosures, including trees, bushes, and climbing structures, and live in large, mixed-sex social groups. All individuals stay in indoor enclosures overnight. Subjects are tested in familiar rooms and are never deprived of food or water for any reason. All testing was strictly voluntary. During testing, a subject could indicate its wish to stop participating at any time by, for instance, heading to the door or not making a choice. Chimpanzees are fed a combination of fruits, vegetables, and other species-appropriate foods three times daily. The research was noninvasive and carried out in accordance with the guidelines of the Pan African Sanctuary Alliance and the regulations of (a) Ngamba Island Chimpanzee Sanctuary, Wildlife Conservation Trust, in Uganda and (b) Sweetwaters Chimpanzee Sanctuary, Ol Pejeta Conservancy, in Kenya (see the Supplemental Material).
Observer report
Seventeen long-time caregivers rated the perceived willingness to take risks of 86 chimpanzees (47 females; age = 2–40 years; 49 chimpanzees from Ngamba Island and 37 from Sweetwaters). Every caregiver rated all chimpanzees in the respective sanctuary. The mean length of time caregivers knew the chimpanzee was 12 years at Ngamba Island Chimpanzee Sanctuary (SD = 2.38; seven caregivers) and 20 years at Sweetwaters Chimpanzee Sanctuary (SD = 6.39; 10 caregivers). Ratings were given for five relevant domains in chimpanzees’ daily lives that expose them to risks, making overt behaviors reveal individual’s willingness to take risks: foraging, interacting with snakes, escaping from their territory, competing for higher rank in the dominance hierarchy, and interacting with (human) strangers. Caregivers also gave a domain-general assessment of each chimpanzee’s willingness to take risks. Ratings were made on a 5-point scale ranging from 0 (not at all willing to take risks) to 4 (very willing to take risks). We calculated a composite risk score per chimpanzee as the mean of the six risk ratings of all caregivers and determined rank-order stability from caregivers’ rankings of the chimpanzees from the least to the most willing to take risks (see the Supplemental Material). Interrater reliabilities were calculated for each individual risk item, as well as for the composite risk score and the risk ranking, using the intraclass correlation coefficient (ICC) formula ICC(2,k). For both sanctuaries, interrater reliabilities were excellent for both the composite risk score (Ngamba Island: .85, 95% credible intervals [CI] = [.77, .90]; Sweetwaters: .9, 95% CI = [.84, .94]) and the risk ranking (Ngamba Island: .88, 95% CI = [.82, .93]; Sweetwaters: .94, 95% CI = [.88, .97]). Interrater reliabilities for the individual items are reported in Table S1.
Behavioral experiments
Fifty-five chimpanzees (a subsample of the observer ratings; 31 females; age 6–35 years; 33 chimpanzees from Ngamba Island and 22 from Sweetwaters) passed the familiarization steps (see the Supplemental Material) and subsequently participated in the behavioral experiments. Chimpanzees made choices under both risk and ambiguity (Ellsberg, 1961; Rosati & Hare, 2011): In the risky condition, the choice was between a safe and a risky urn (potential outcomes and probabilities were known), and in the ambiguous condition, the choice was between a safe and an ambiguous urn (potential outcomes and probabilities were uncertain). The safe urn always contained one reward; the risky/ambiguous urn contained either two rewards or nothing (50/50 chance; Fig. 1; see also the Supplemental Material).
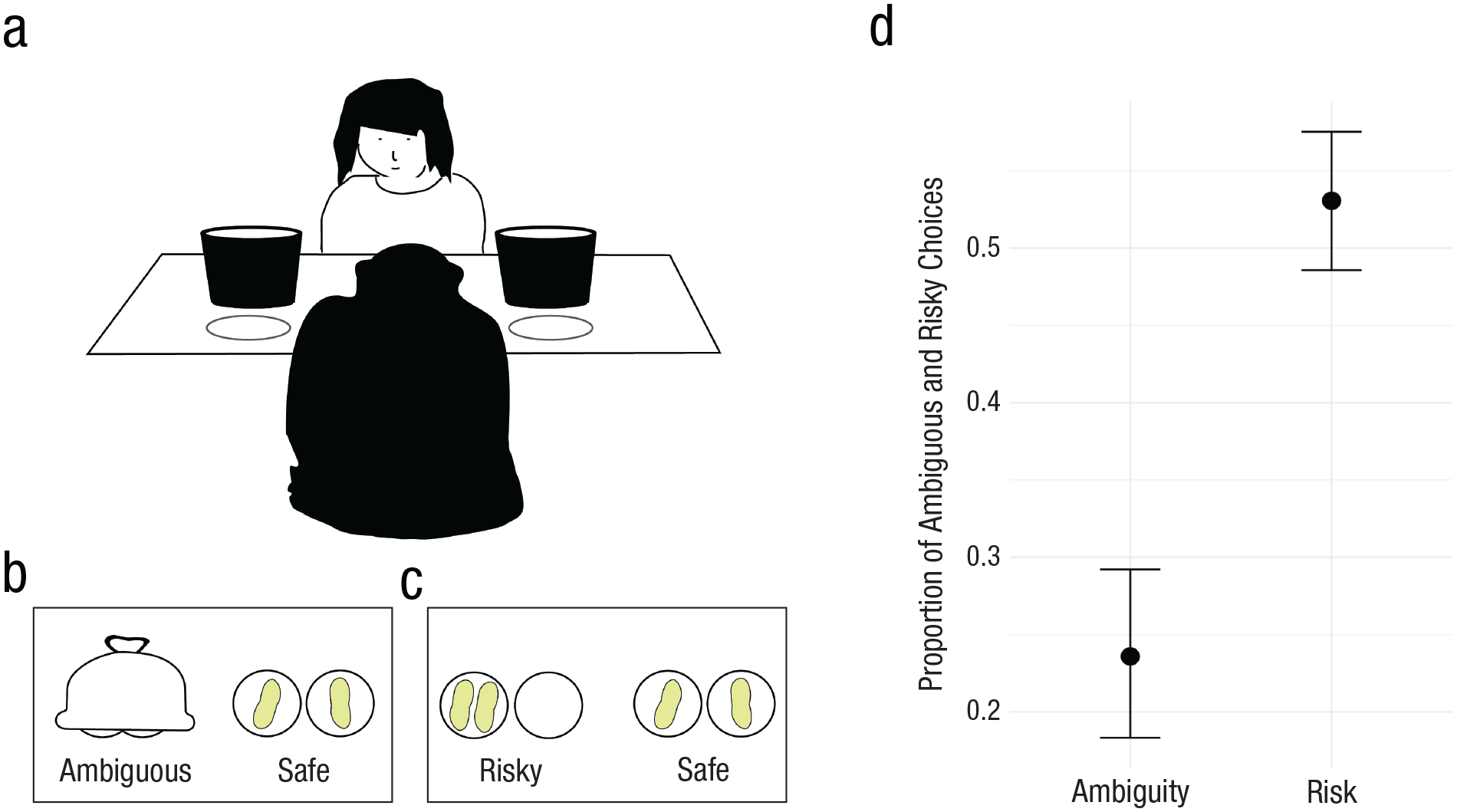
Ambiguous and risky choices in the behavioral experiments. (a) Experimental setup. (b) Ambiguous condition. (c) Risky condition. (d) Proportion of ambiguous and risky choices. Black dots represent means; error bars represent 95% credible intervals.
Results
Correlations across domains and measurements
To examine whether chimpanzees’ willingness to take risks manifests as a traitlike preference, we analyzed its consistency across domains and measurement approaches. For the observer ratings, we calculated the correlations among the general and five domain-specific risks (based on grouped means) and the risk rank order. We found high positive correlations between risk domains (Table S2 in the Supplemental Material): They were strongest between general and foraging risk (r = .85, 95% CI = [.77, .90]) and weakest (even though still relatively high) between escape and hierarchy risk (r = .48, 95% CI = [.30, .63]). The risk domains were positively correlated with risk rank order (composite risk score: r = .53, 95% CI = [.36, .67]). The sole exception was risk in interactions with strangers. It was only weakly correlated with the other risk domains (general risk: r = .10, 95% CI = [−.11, .31]) and negatively correlated with risk rank order (r = –.25, 95% CI = [−.44, –.04]). These divergent findings mirror past results indicating that social risk taking involves an element of trust and thus is rooted in or codetermined by social preferences (Engelmann et al., 2015; Engelmann & Herrmann, 2016; Fehr, 2009; Haux et al., 2021). Next, we investigated the relationship between the observer report and the behavioral risk measure (Table S3 in the Supplemental Material). Importantly, risky choices were positively related to general risk (r = .36, 95% CI = [.06, .60]) and risk rank order (r = .38, 95% CI = [.08, .62]). In humans, behavioral and stated risk measures correlate only weakly (M = .06, highest density interval = [.05, .06]; see Frey et al., 2017). In contrast, our behavioral and observational data both indicate that chimpanzees’ willingness to take risks manifests consistently across measures.
Ambiguity aversion
To find out whether chimpanzees are ambiguity averse, we investigated the effect of experimental condition (risky vs. ambiguous). The model estimate for the risky condition was positive (b = 1.30, 95% CI = [0.97, 1.65]; Bayesian generalized linear model [BGLM]; Table S4 in the Supplemental Material), suggesting that chimpanzees chose the safe urn more often in the ambiguous condition than in the risky condition. As Figure 1d shows, chimpanzees were clearly ambiguity averse (Trautmann & van de Kuilen, 2015), choosing the risky urn in 55% of trials but the ambiguous urn in only 25%.
Sex differences in risk preference
To investigate sex differences, we first analyzed the observer ratings. Male chimpanzees were rated as more risk seeking than females across all domains (Fig. 2a; composite risk score: b = 0.34, 95% CI = [0.04, 0.64], BGLM; Fig. S1a in the Supplemental Material). Consistent with life-history theory, results showed that the sex difference was most pronounced in the hierarchy domain (b = 0.43, 95% CI = [0.13, 0.75]; Table S5 in the Supplemental Material). To compare the size of the sex difference in chimpanzee and human risk preference, we calculated the percentage of the maximum possible (POMP) score for the general risk item of the observer ratings. Chimpanzee males had 8 percentage points of the maximum possible more than females. This corresponds closely to the effect size reported for sex differences in humans (e.g., 6 POMPs; see Dohmen et al., 2011). For the behavioral choices, the model estimate for the interaction of the risky condition and male sex was positive (b = 0.64, 95% CI = [0.01, 1.30]; Model 1.2 in Table S4), suggesting that males were more likely than females to choose the risky urn. Males chose the risky urn in 55% of trials and the ambiguous urn in 20%; females chose the risky urn in 50% of trials and the ambiguous urn in 25% (Fig. 2b). These results again echo those of human studies, showing that males are more risk seeking but also more ambiguity averse than females (Borghans et al., 2009).
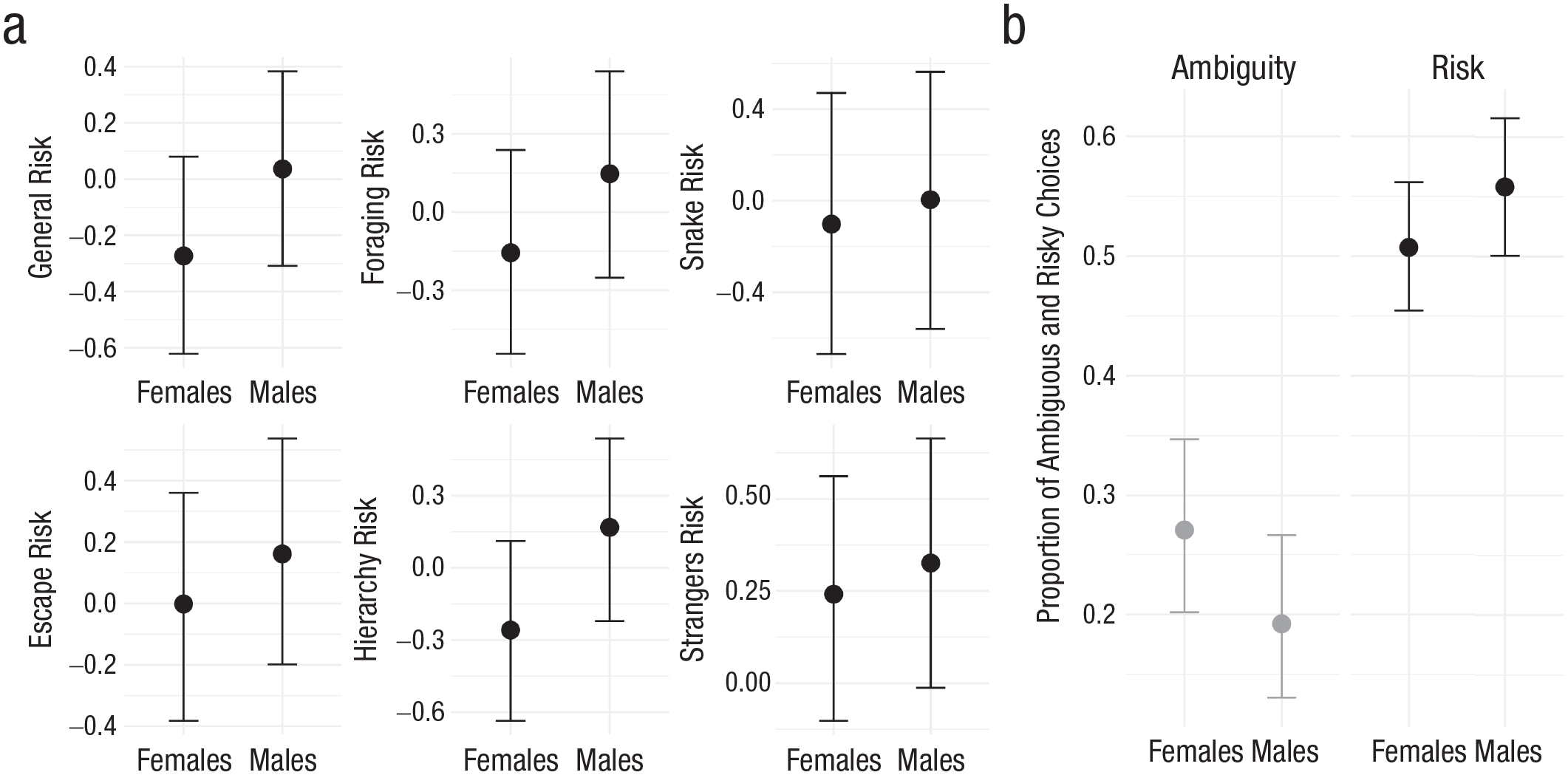
Sex differences in risk preference. Black dots represent means; error bars represent 95% credible intervals. (a) Sex differences in mean levels of general and domain-specific willingness to take risks. (b) Sex differences in proportion of ambiguous and risky choices.
Age differences in risk preference
To examine age differences (Fig. 3; see also Table S6 in the Supplemental Material), we used spline-based smooths (Bürkner, 2017). For the composite risk score, the standard deviation (b = 1.29, 95% CI = [0.36, 3.32], BGLM) and visual inspection indicated nonlinearity (Fig. S1b in the Supplemental Material). An inverted-U-shaped relationship emerged consistently across domains and was least pronounced for interaction with strangers (b = 0.76, 95% CI = [0.04, 2.43], BGLM; Fig. 3a and Table S6). This pattern echoes findings in humans showing that the willingness to take risks in the social domain peaks during early adulthood, is lower in older age, and remains relatively stable across the adult life span (Josef et al., 2016). Investigating the interaction of age and sex on the composite risk score revealed an inverted-U-shaped relationship for males (b = 1.62, 95% CI = [0.57, 3.91], BGLM) and a flatter curve for females (b = 0.66, 95% CI = [0.02, 2.61], BGLM; Fig. 4a; see also Fig. S1c and Table S7 in the Supplemental Material). We also investigated age differences in the behavioral measures (Model 1.3 in Table S4). For choices under risk, a small effect of nonlinearity emerged (b = 0.70, 95% CI = [0.03, 2.31], BGLM). Chimpanzees’ willingness to select the risky option was slightly lower in adulthood (Fig. 3b). The peak during young adulthood was less pronounced for the behavioral risk measure, corresponding to findings in humans showing that stated risk measures pick up age variations that behavioral risk measures do not detect (Frey et al., 2021). As in humans (Josef et al., 2016), male chimpanzees tended to be more risk seeking than females were across all age groups. The willingness to select the ambiguous option was highest during childhood and varied little across adulthood (b = 1.78, 95% CI = [0.24, 4.69], BGLM). Females tended to choose the ambiguous option more often than males did (Fig. 4b). These results mirror findings in humans showing that ambiguity preferences are invariant over the adult life span and that females are more ambiguity prone than males (Borghans et al., 2009).
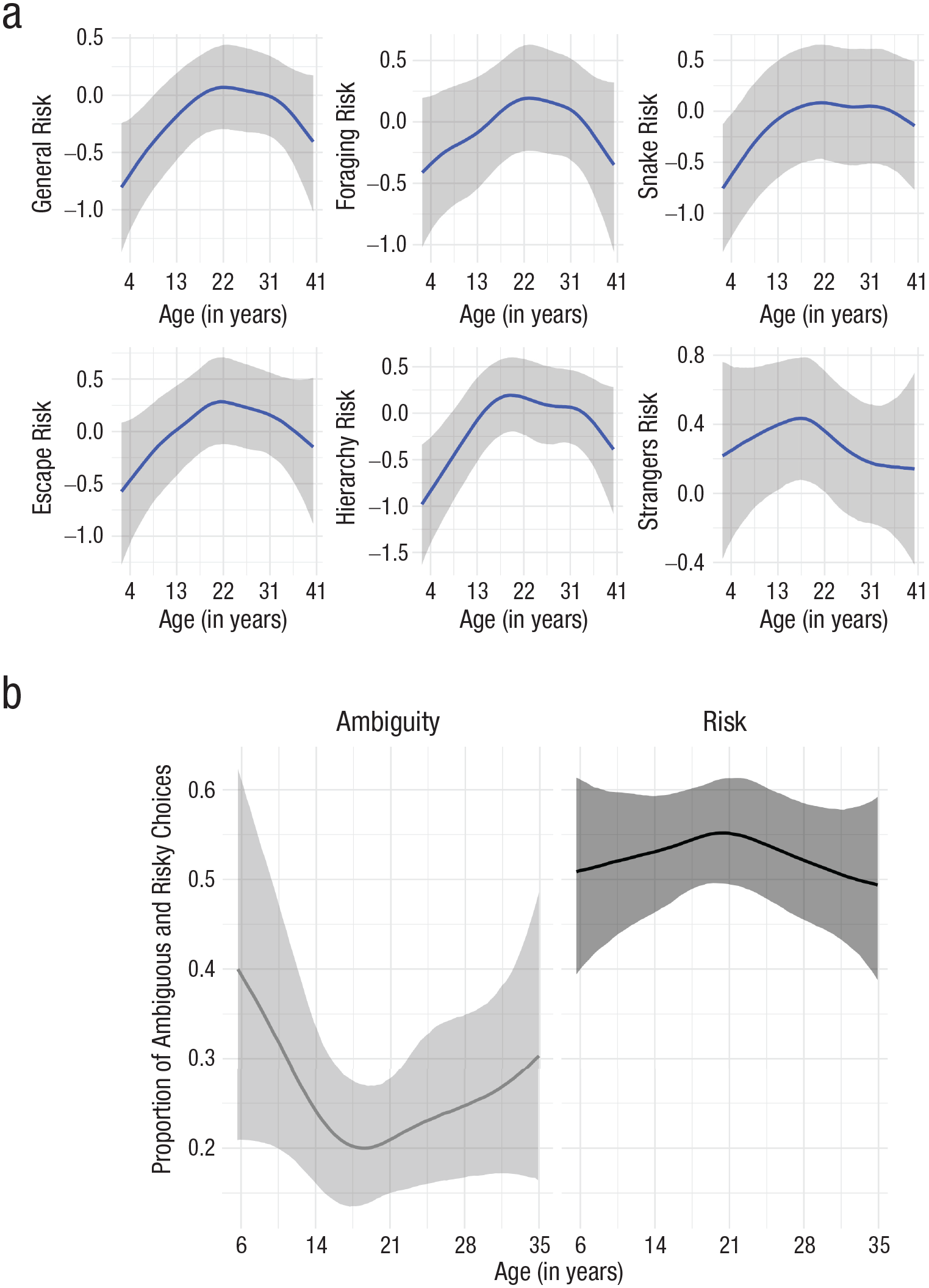
Age differences in risk preference: regression splines with shaded 95% credible intervals. (a) Age differences (aggregated across male and female subjects) in mean levels of general and domain-specific willingness to take risks. (b) Age differences in proportion of ambiguous and risky choices.
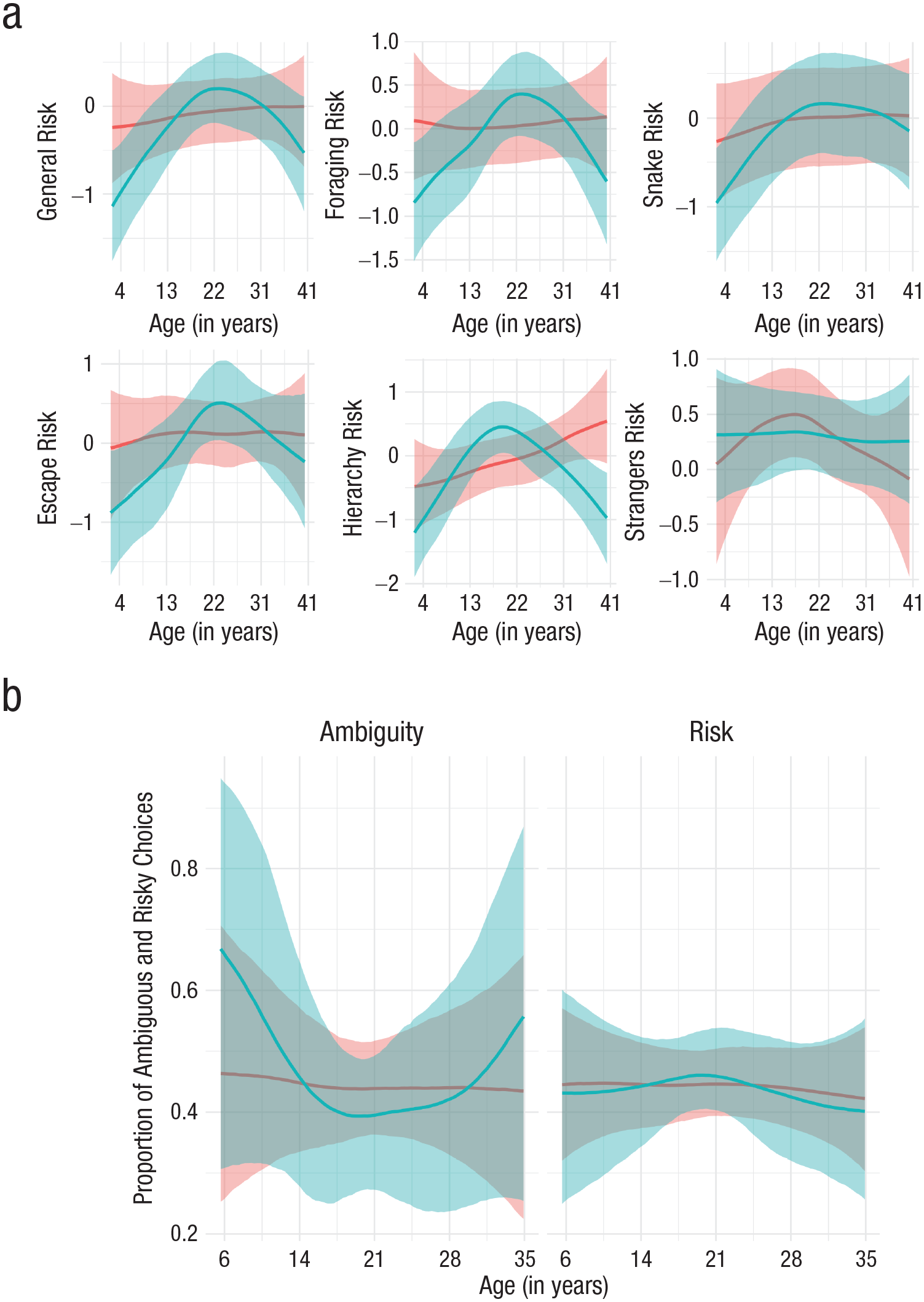
Age and sex differences in risk preference: regression splines with shaded 95% credible intervals. Red indicates female risk preference, and blue indicates male risk preference. (a) Age and sex differences in mean levels of general and domain-specific willingness to take risks. (b) Age and sex differences in proportion of ambiguous and risky choices.
Discussion
Risk preference is central to human and nonhuman behavior. The current results demonstrate that chimpanzee and human risk preference share key structural similarities and converge in crucial ways. Consistent with recent findings in humans (Frey et al., 2017), our results showed that chimpanzees’ willingness to take risks appears to manifest as a traitlike preference, with high rank-order stability across a set of important domains (based on observers’ assessments) and economic choice behavior. The social-risk domain proves to be the exception (Josef et al., 2016). Furthermore, chimpanzees are, like humans, ambiguity averse and less willing to choose options with unknown risks (Ellsberg, 1961; Trautmann & van de Kuilen, 2015). Our results indicate that males are more risk seeking than females, mirroring the same difference in human risk preference (Frey et al., 2021). Finally, chimpanzee risk taking shows an inverted-U-shaped relation to age, peaks in young adulthood, and is lower in older age—again mirroring similar trends in humans (Frey et al., 2021; Josef et al., 2016; Mata et al., 2016).
According to life-history theory, risk preference should be elevated in periods in which the goal of reproduction and associated proximal goals (e.g., gaining social status) is paramount (Stearns, 1992). Young adulthood is an indispensable transitional stage for male chimpanzees to learn the techniques of socially mature males in order to establish their own social position (Kawanaka, 1993; Watts, 2018). Consistent with the young male syndrome in humans (Wilson & Daly, 1985), our results show that male chimpanzees are especially risk prone around their 20s and particularly willing to take risks in order to get a better position in the hierarchy. Higher rank is associated with both relatively high mating (Kaburu & Newton-Fisher, 2015; Muller et al., 2011) and paternity (Boesch et al., 2006; Langergraber et al., 2013; Newton-Fisher et al., 2010; Wroblewski et al., 2009) success. Furthermore, our finding of heightened general risk tolerance during young adulthood converges with a recent overview of risk behavior in humans concluding that risk taking is heightened during emerging adulthood (Willoughby et al., 2021).
The current findings, in combination with the multimethod design, enrich the comparative approach in important ways. First, the general and traitlike preference for risk is congruent with evidence suggesting that animal personalities exist across a range of species and that risk-related traits are common characteristics (Wolf et al., 2007). Second, we found that the strangers risk domain was only weakly correlated with the other risk domains. This finding is in line with those of earlier studies indicating that trusting other individuals is not just a special case of risk taking but is based on important forms of social preferences, such as betrayal aversion (Fehr, 2009; Haux et al., 2021). Moreover, in humans, willingness to trust does not follow an inverted-U-shaped pattern but instead remains relatively stable across the adult life span (Josef et al., 2016). These results fit ideas that in humans and chimpanzees, the social domain remains prioritized across adulthood (Carstensen et al., 1999; Rosati et al., 2020). Third, chimpanzees’ observed ambiguity aversion is in line with results by Rosati and Hare (2011). They found that chimpanzees and bonobos are sensitive to ambiguity in the first trials of their experiment, suggesting that subjects dislike choosing options with unknown risks. Yet it remains unclear to what extent chimpanzees knew that only probability but not the information about outcomes was missing. This raises the question of whether ambiguity attitudes in Ellsberg’s (1961) implementation of the construct can be measured in nonverbal populations.
Fourth, our results suggest that chimpanzees are risk neutral to (minimally) risk prone. Past research offers inconsistent findings in this regard. The variation in risk sensitivity in studies may be explainable in terms of the presentation and experience of probabilities (see Hau et al., 2010; Heilbronner & Hayden, 2016; Hertwig, 2015; Wulff et al., 2018), that is, whether decisions were based on experienced frequencies (see Calcutt et al., 2019; Haux et al., 2021; Heilbronner et al., 2008; Keupp et al., 2021), or whether subjects had to infer probabilities from the task design (see Haun et al., 2011; Rosati & Hare, 2011, 2012, 2013, 2016). Furthermore, the possibility of coming away empty-handed—whether the risky option included the possibility of receiving nothing or always provided at least some amount of food—could also alter the decision (see Haux et al., 2021; Keupp et al., 2021).
Finally, the relatively small sample sizes and the concomitant sex and age distributions of past studies limit the generalizability of previous findings. Future research should expand the multimethod approach, for instance, by including several behavioral measures. This would allow researchers to examine the causes behind the variation in previous work and delineate which task characteristics influence chimpanzee risk sensitivity. Studies involving chimpanzees from different groups and environments will further enrich the discussion about the generalizability of behavioral variations. Because of their early-life experiences, sanctuary chimpanzees might display different risk preferences and behavior from those living in zoos and those in the wild (for a discussion on the generalizability across groups, see King et al., 2005; Laméris et al., 2021; Lutz et al., 2022; Weiss et al., 2007; Wobber & Hare, 2011). Last but not least, because it has been proposed that bonobos (Pan paniscus) and chimpanzees (humans’ two closest living relatives) show divergent risk preferences (see Rosati, 2017), for a complete reconstruction of our last common ape ancestors preferences, it is essential to also study bonobos’ willingness to take risks in more depth in the future.
In the behavioral sciences, risk preference is a mainstay of and a key building block in theories of choice (Barseghyan et al., 2013; Brailovskaia et al., 2018; Clark & Lisowski, 2017; Dohmen et al., 2011; Mata et al., 2018; Schonberg et al., 2011; Slovic, 1987). In recent years, the measurement and stability has been central to the debate about the nature of risk preferences (Frey et al., 2017, 2021; Josef et al., 2016; Schildberg-Hörisch, 2018; Weber et al., 2002). Our multimethod approach can enrich this discussion, indicating that across various aspects of risk preference, both observer ratings (for a discussion on the validity of human ratings, see the Supplemental Material) and behavioral choices are directionally consistent. These findings offer an important first step toward a general mapping of the construct of risk preference in chimpanzees. Understanding the degree of temporal stability and systematic individual change in chimpanzee risk preference will be an important endeavor for future research (Schildberg-Hörisch, 2018). In addition, “actuarial” data such as the frequency of injuries from hierarchy fights will offer another important addition to a multimethod approach to study risk preference in chimpanzees.
In humans, scholars have proposed that the willingness to take risks and to trust others is transmitted across generations through socialization experiences (Dohmen et al., 2012; Roberts et al., 2005; Slovic, 1966) but is also subject to genetic influences (Karlsson Linnér et al., 2019). Our findings suggest that human risk preference may in addition also have deeper phylogenetic roots than previously suspected. Structural similarities in risk preferences of humans and one of our closest living relatives are likely to reflect adaptations to similar dynamics in primate life histories.
Supplemental Material
sj-docx-1-pss-10.1177_09567976221140326 – Supplemental material for Chimpanzee and Human Risk Preferences Show Key Similarities
Supplemental material, sj-docx-1-pss-10.1177_09567976221140326 for Chimpanzee and Human Risk Preferences Show Key Similarities by Lou M. Haux, Jan M. Engelmann, Ruben C. Arslan, Ralph Hertwig and Esther Herrmann in Psychological Science
Supplemental Material
sj-docx-2-pss-10.1177_09567976221140326 – Supplemental material for Chimpanzee and Human Risk Preferences Show Key Similarities
Supplemental material, sj-docx-2-pss-10.1177_09567976221140326 for Chimpanzee and Human Risk Preferences Show Key Similarities by Lou M. Haux, Jan M. Engelmann, Ruben C. Arslan, Ralph Hertwig and Esther Herrmann in Psychological Science
Supplemental Material
sj-xlsx-3-pss-10.1177_09567976221140326 – Supplemental material for Chimpanzee and Human Risk Preferences Show Key Similarities
Supplemental material, sj-xlsx-3-pss-10.1177_09567976221140326 for Chimpanzee and Human Risk Preferences Show Key Similarities by Lou M. Haux, Jan M. Engelmann, Ruben C. Arslan, Ralph Hertwig and Esther Herrmann in Psychological Science
Supplemental Material
sj-xlsx-4-pss-10.1177_09567976221140326 – Supplemental material for Chimpanzee and Human Risk Preferences Show Key Similarities
Supplemental material, sj-xlsx-4-pss-10.1177_09567976221140326 for Chimpanzee and Human Risk Preferences Show Key Similarities by Lou M. Haux, Jan M. Engelmann, Ruben C. Arslan, Ralph Hertwig and Esther Herrmann in Psychological Science
Footnotes
Acknowledgements
We thank Lilly Ajarova and Joshua Rukundo, the board members and staff at Ngamba Island Chimpanzee Sanctuary andWildlife Conservation Trust in Uganda for their crucial support during all stages of this research. We appreciate permission from the Ugandan National Council for Science and Technology and the Uganda Wildlife Authority. We thank Richard Vigne, Samuel Mutisya, Stephen Ngulu, the board members and staff of Sweetwaters Chimpanzee Sanctuary in Kenya for their crucial support during all stages of this research. We appreciate permission from Ol Pejeta Conservancy, Kenya Wildlife Service, and the National Council for Science and Technology. Thanks go to Larissa Samaan and Lukas Eggeling for reliability coding and Susannah Goss for editing. We thank Michael Tomasello, Andrew Whiten and Amanda Seed for feedback. LMH was supported by the German National Academic Foundation and EH by the Minerva Programme of the Max Planck Society. R. Hertwig and E. Herrmann share senior authorship of this article.
Transparency
Action Editor: Lasana Harris
Editor: Patricia J. Bauer
Author Contributions
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
