Abstract
Identifying predictors of cognitive decline in old age helps us understand its mechanisms and identify those at greater risk. Here, we examined how cognitive change from ages 11 to 70 is associated with cognitive change at older ages (70 to 82 years) in the Lothian Birth Cohort 1936 longitudinal study (
Keywords
This work addressed individual differences in cognitive aging from a novel perspective. Rather than studying how differences in age-related cognitive decline are associated with other factors, we examine cognitive-change consistency across the life course. We and others have shown that level of cognitive ability ascertained in childhood relates strongly to level of cognitive ability in older age (Deary, 2014; Rönnlund et al., 2015; Schalke et al., 2013). Here, instead, we asked whether individual differences in earlier life-course cognitive trajectories (age 11–70 years) predict subsequent cognitive change from age 70 to 82—a life period which generally sees more rapid and clinically important shifts. Individual differences in cognitive aging probably reflect an accumulation of small influences from numerous factors (Corley et al., 2018), many of which are likely to be already present in early life and midlife (e.g., genetic factors, early-life cognitive ability, physical fitness, smoking). Therefore, it is essential to characterize the relationship between earlier-period and later-period cognitive trajectories across the life course.
Cognitive decline is among the most feared aspects of aging. It will affect more people as the world population ages: In many countries, the proportion of older adults is increasing (Rousson & Paccaud, 2010; United Nations DESA, 2015), and longer life expectancy is not always matched by prolonged health (Abbafati et al., 2020; Prince et al., 2015). Even nonpathological cognitive decline can affect daily life and activities: Reduced cognitive functioning is associated with lower quality of life, leading to loss of autonomy, illness, and death (Batty et al., 2016; Deary et al., 2009; Schaie et al., 2009; Yam et al., 2014). Thus, the clear personal, societal, and financial consequences of cognitive aging, even among the nonclinical majority, motivate urgent scientific investigation. Beginning approximately at age 70, the risk of cognitive decline increases substantially (Deary et al., 2009; Marmot et al., 2003; Salthouse, 2019), as does the risk of dementia (Berr et al., 2005; Jorm & Jolley, 1998; Santoni et al., 2015).
There is considerable interindividual variability within the general trend of cognitive aging (e.g., Schaie & Willis, 2010; Zaninotto et al., 2018). Understanding the nature, predictors, and mechanisms underlying individual differences is essential for tackling the disruptive effects of cognitive decline and designing paths to successful aging. In this context, when some cognitive changes begin in early adulthood (Salthouse, 2019; Schaie & Willis, 2010; Tucker-Drob, 2019), the timing of interventions becomes an especially complicated matter (Plassman et al., 2010). The accurate prediction of trajectories is critical because it improves understanding of potential mechanisms and identification of those at relatively higher risk (Brayne, 2007; Deary et al., 2009; Salthouse, 2019).
Longitudinal studies emphasize the need to distinguish cognitive change from cognitive level; they show that an individual’s cognitive level at any given age is, at best, weakly associated with their cognitive trajectory (Karlamangla et al., 2009; Tucker-Drob, 2019). Accordingly, factors related to peak cognitive level in adulthood do not necessarily have a comparable association with cognitive-decline rates (Corley et al., 2018; Lövdén et al., 2020; Marden et al., 2017; Ritchie et al., 2016; Rönnlund et al., 2017; Tucker-Drob, 2019). Research on cognitive aging correlates has tested genetic, sociodemographic, health, and lifestyle factors. Among the stronger predictors of steeper cognitive decline are sex (being male), lower physical fitness, and possession of the
We are unaware of research examining whether differences in cognitive change from childhood to later adulthood are predictive of the subsequent cognitive-decline gradient in older age. This is an important omission. If we knew that individual differences in cognitive change between, say, ages 11 and 70 were associated with cognitive changes from ages 70 to 82, we would have more confidence that addressing factors operating before older age could ameliorate cognitive decline in older age.
Statement of Relevance
Age-related cognitive decline is a significant threat to quality of life in older age. Its economic and social impact on society will increase together with the steadily rising life expectancy. How can we preserve cognitive health in older age? Researchers have made significant advances in identifying protective and risk factors. However, most studies focus on a limited age range, and cognitive-change mechanisms are not yet completely understood. This work took advantage of almost-life-spanning longitudinal data to test whether cognitive trajectories across childhood and adulthood can predict cognitive trajectories in older age. Our findings show that earlier change is associated with later change. Some factors related to individual differences in cognitive change might thus operate over much of the adult life course, and certainly before older age. This knowledge can help us identify individuals at higher risk of decline and understand the mechanisms and factors responsible.
Here, we tested the hypothesis that cognitive change in general and domain-specific abilities after 70 (i.e., visuospatial, memory, and processing speed) might be predicted by cognitive change up to age 70. We used longitudinal data spanning 71 years from the Lothian Birth Cohort 1936 (LBC1936).
Method
Participants
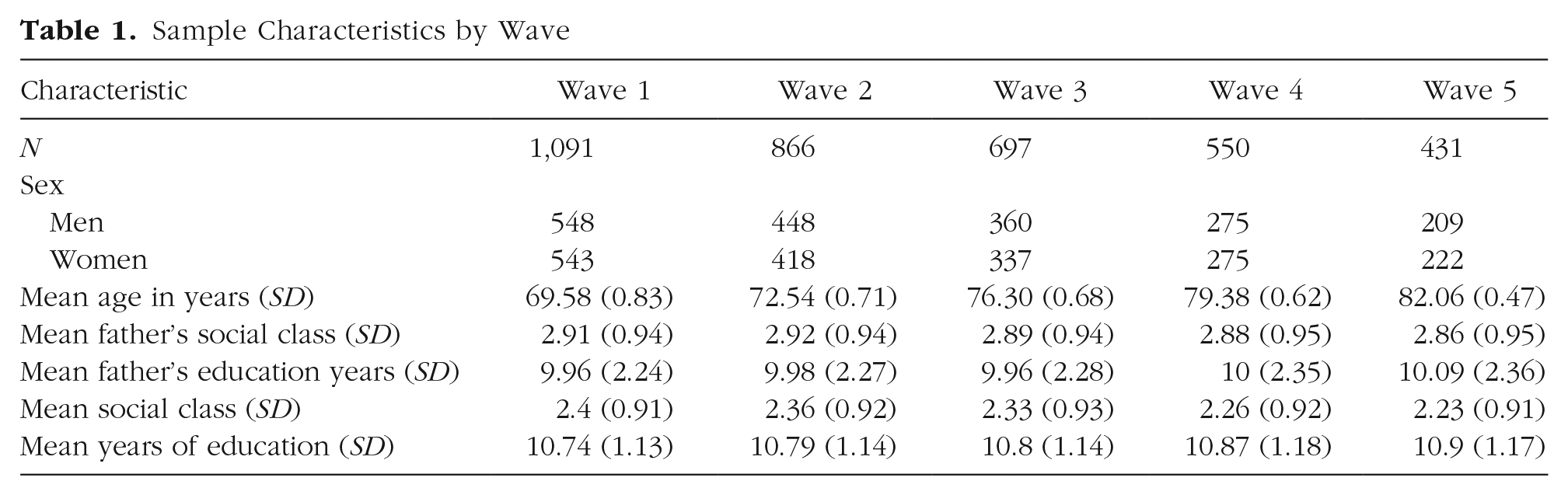
The LBC1936 is a longitudinal study of cognitive, brain, and general aging. Participants were all born in 1936, and most took a test of general mental ability, the Moray House Test No. 12 (MHT), at age 11 years, as part of the Scottish Mental Survey of 1947 (SMS; Scottish Council for Research in Education, 1949). Between 2004 and 2007 (i.e., at about age 70), 1,091 probable SMS participants living in the Lothian area joined in the first wave of follow-up testing to form the LBC1936. As of 2022, the LBC1936 participants have taken part in five assessment waves at approximately 3-year intervals from age 70 to age 82. A description of the types of data collected at each wave is given in Taylor et al. (2018).
At baseline (Wave 1), the LBC1936 sample consisted of 1,091 individuals (543 females, age:
Sample Characteristics by Wave
Measures
The MHT was completed by participants at age 11 years and age 70 years (Wave 1) in the present study. It was called a verbal-reasoning test, but its items assess a range of abilities, including word classification, reasoning, analogies, arithmetic, spatial reasoning, and following directions. The test provides a single general cognitive-ability score, with a maximum value of 76. The MHT score correlated at about .80 with the Stanford-Binet scale in a validation test conducted during the SMS (Deary, 2014; Scottish Council for Research in Education, 1949).
Cognitive ability from age 70 to 82 was assessed using a battery of 10 tests related to three cognitive domains, administered at each wave from 1 to 5. Three tasks evaluated visuospatial ability: Matrix Reasoning and Block Design from the Wechsler Adult Intelligence Scale III UK (WAIS-III UK; Wechsler, 1998a), and Spatial Span forward and backward (the sum score of the two was used in the analyses) from the Wechsler Memory Scale III UK (WMS-III UK; Wechsler, 1998b). Three tests from the WMS-III UK evaluated verbal memory: Verbal Paired Associates immediate and delayed, Logical Memory immediate and delayed (for these two tasks, total scores were the sum of scores in the two conditions), and Digit Span backward. Finally, speed of information processing was ascertained by the Symbol Search and Digit-Symbol Substitution tasks from the WAIS-III UK by a visual inspection time task (Deary et al., 2007), and by a four-choice reaction time task (Deary et al., 2001). In the analyses, reaction times were multiplied by −1, so that, for all tests, higher scores indicated better performance. For a detailed description of test characteristics and administration, see Deary et al. (2007).
Socioeconomic indicators
Participants reported their own and their father’s principal occupation (prior to retirement), which were grouped into social-class categories scored from 1 to 5: professional, managerial, skilled nonmanual, skilled manual, and semiskilled/unskilled. For women, spouse occupation was considered if higher than their own. Father’s and own years of education were also reported (Table 1).
Statistical analysis
We hypothesized that individual differences in cognitive change observed between the ages of 11 and 70 would be significantly associated with individual differences in cognitive change between the ages of 70 and 82. To test this hypothesis, we conducted the following steps, which are described in greater detail below: (a) estimate cognitive change from ages 11 to 70 using the MHT scores measured at both ages; (b) build measurement models for cognitive abilities from ages 70 to 82 using data from the larger set of 10 cognitive tests; (c) test the degree to which cognitive change between 11 and 70 predicts cognitive aging between 70 and 82; and (d) test whether cognitive change between 11 and 70 is independently predictive of change between 70 and 82, beyond just age-70 cognitive level (Fig. 1a).
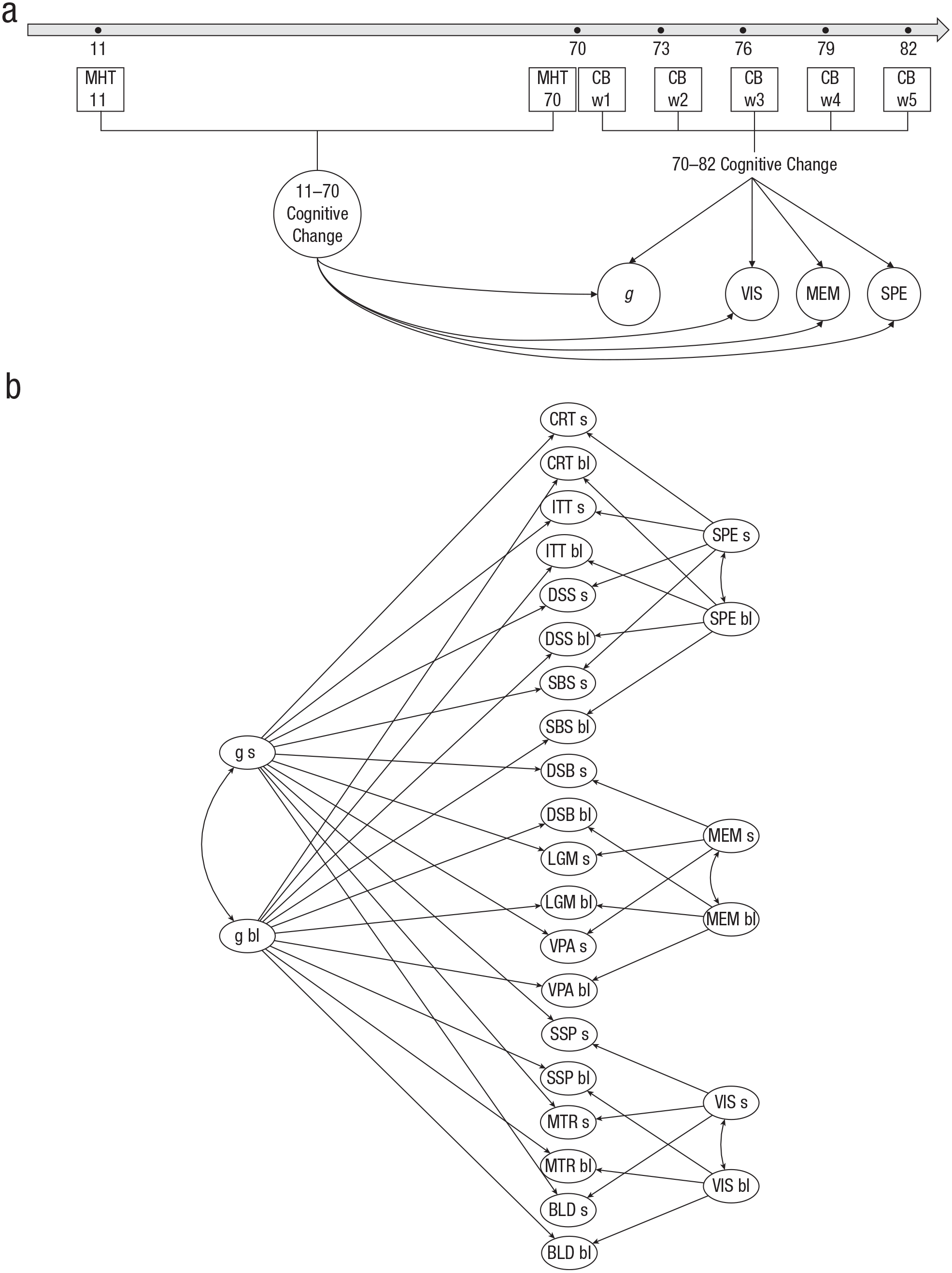
Analysis diagram and bifactor model representation. The diagram in (a) shows the main analysis: cognitive change from age 11 to 70 as a predictor of cognitive change from age 70 to 82. Cognitive change from age 11 to 70 was estimated from Moray House Test No. 12 (MHT) scores. Change in general cognitive ability (
Deriving measures of cognitive change
Cognitive change from 11 to 70 was modeled as the unstandardized residuals of the regression between MHT scores at Wave 1 (age 70) and age-adjusted MHT scores at age 11. This procedure has been used in previous Lothian Birth Cohort studies, such as Cherrie et al. (2018).
Cognitive change from age 70 to age 82 was estimated using a factor-of-curves (FOCUS) model (McArdle, 1988). At the lowest level of the FOCUS model, 10 linear latent growth curves estimated change for each of the 10 cognitive tests. Wave 1 (age 70) scores were considered the origins of the curves, and scores from subsequent waves (ages 73, 76, 79, 82) were weighted on the basis of the mean number of years that had passed since Wave 1. The latent growth curves provided, for each cognitive task, a baseline-level parameter, representing mean scores at Wave 1 (age 70), and a slope parameter, representing mean change per year for the subsequent 12 years.
At the higher level of the FOCUS model, baseline level and slope for each of the three cognitive domains (speed, memory, and visuospatial) and for
Estimating associations between cognitive change from ages 11 to 70 and ages 70 to 82
We asked whether our measure of cognitive change between ages 11 and 70 predicted subsequent cognitive declines in older age. To do so, we introduced change between ages 11 to 70 in the model of cognitive change from ages 70 to 82 (previously constructed; see above), as a predictor of the baseline (age 70) levels and slopes of general and domain-specific cognitive abilities within older age. To aid model convergence, we fixed factor loadings and intercepts obtained from the measurement model, whereas regression coefficients and residual-factor variances were freely estimated.
We introduced sex, the Sex × Cognitive-Change From 11 to 70 interaction, childhood and adult socioeconomic status indicators, and MHT score at age 11 as covariates alongside our main predictor. Following peer review, we ran follow-up analyses excluding either cognitive change between ages 11 and 70 or MHT score at age 11 from the regression model to test how the use of one versus both predictors influenced education associations with cognitive level and change.
Finally, we ascertained whether the measure of cognitive change between 11 and 70 accounted for unique variance in decline in
Supplementary analyses
We calculated our main cognitive predictor (i.e., MHT change from ages 11–70) as a regression-based score because these are arguably less affected by random measurement error compared with raw difference scores (Campbell & Kenny, 2002; Cronbach & Furby, 1970). However, we recognize that there is no clear consensus on the optimal measurement of change. Therefore, we conducted a supplementary analysis in which we used a raw difference score, also accounting for change reliability (see Supplementary Methods in the Supplemental Material available online).
Even though the data benefited from a narrow age range, there were small age differences for each assessment wave in older age. To ensure that these age differences did not substantially impact our results, we fitted a second version of the cognitive measurement model, covarying the observed task scores with mean-centered age in days at the time of assessment.
Finally, in supplementary results, we present the association between cognitive change between 11 and 70 and individual cognitive domains, without partialing out general cognitive variance.
Peak-based measures of cognitive change
The longitudinal data from the LBC1936 cohort provides insight on cognitive change over most of the human life course. The lack of assessments between ages 11 and 70 makes it difficult to identify specific phases of cognitive change, such as childhood development or the beginning of decline in adulthood. However, we can use some existing data to partially fill the 60-year gap. One of the other measures collected in the LBC1936 is the National Adult Reading Test (NART; Nelson & Willison, 1991). The verbal skills assessed by the NART improve throughout adulthood and are robust to some normal and pathological decline (Lezak et al., 2004). Various follow-up studies of the SMS, using the MHT, have validated the NART as an estimate of prior or premorbid cognitive ability (Crawford et al., 2001; Deary & Brett, 2015; McGurn et al., 2008). Deary et al. (2004) showed that NART-included cognitive-change estimates correlate strongly with measures of actual lifetime cognitive change. As a counterpoint to our primary analysis, we used NART score at age 70 as an estimate of peak cognitive ability in adulthood. We then computed two additional regression-based indicators of cognitive change: from childhood to estimated adulthood peak (i.e., MHT at age 11 to NART at age 70); and from estimated adulthood peak to age 70 (i.e., NART at age 70 to MHT at age 70). The intention was to distinguish a phase of cognitive development from a phase of decline prior to age 70. Consistent with the main analysis, MHT score at age 11 was adjusted for age before regressing NART on it. Each of these indicators was tested as a predictor of cognitive change from 70 to 82 by introducing it in the cognitive-measurement models in the same way as cognitive change from 11 to 70.
Software, fit, and multiple-comparison correction
All models were estimated in the R environment (R Core Team, 2020) using the
Results
Deriving measures of cognitive change
MHT scores showed a general improvement between age 11 (
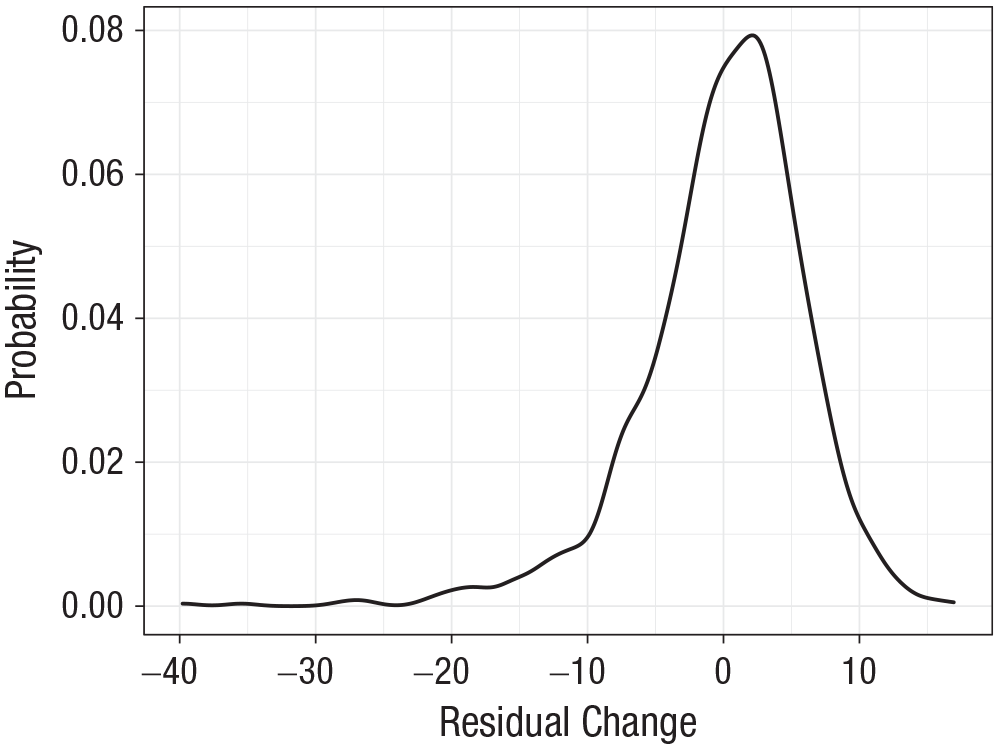
Density plot of residual-change scores of individual participants, showing residuals of the regression of Moray House Test scores at age 70 on age-corrected Moray House Test scores at age ~11.
Bivariate Correlations Between Measures of General Cognitive Ability and Cognitive Change From Ages 11–70
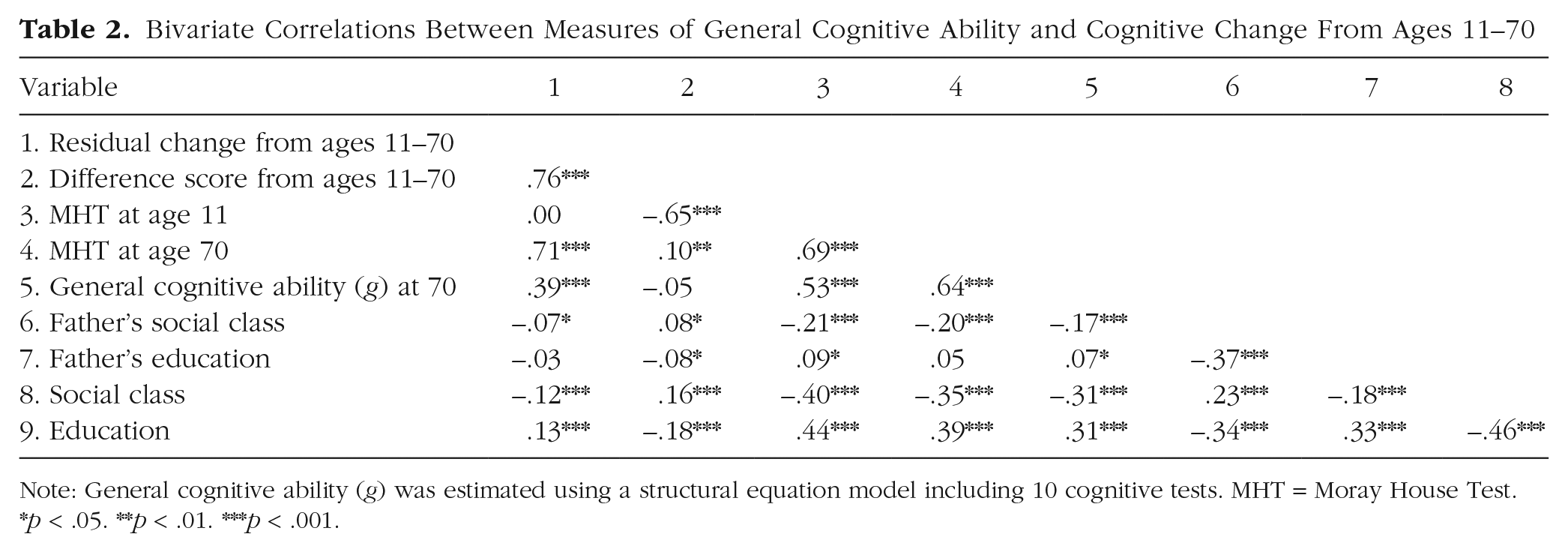
Note: General cognitive ability (
Fit indices for the bifactor model of the baseline levels and slopes of the 10 cognitive tests are presented in Table S1; factor loadings are presented in Table S2.
The cognitive-measurement model fit the data well. An average of 43% of task variance in baseline levels was shared within
Cognitive change from ages 11 to 70 as a predictor of individual differences in later-life cognitive trajectories
Results of the principal analyses are presented in Table 3 (top). Table S1 reports model-fit indices, which were good. A greater relative improvement in MHT score between ages 11 and 70 was associated with slower decline in
Associations Between Cognitive Change From Age 11 to 70 and Later-Life Trajectories of General and Domain-Specific Cognitive Abilities
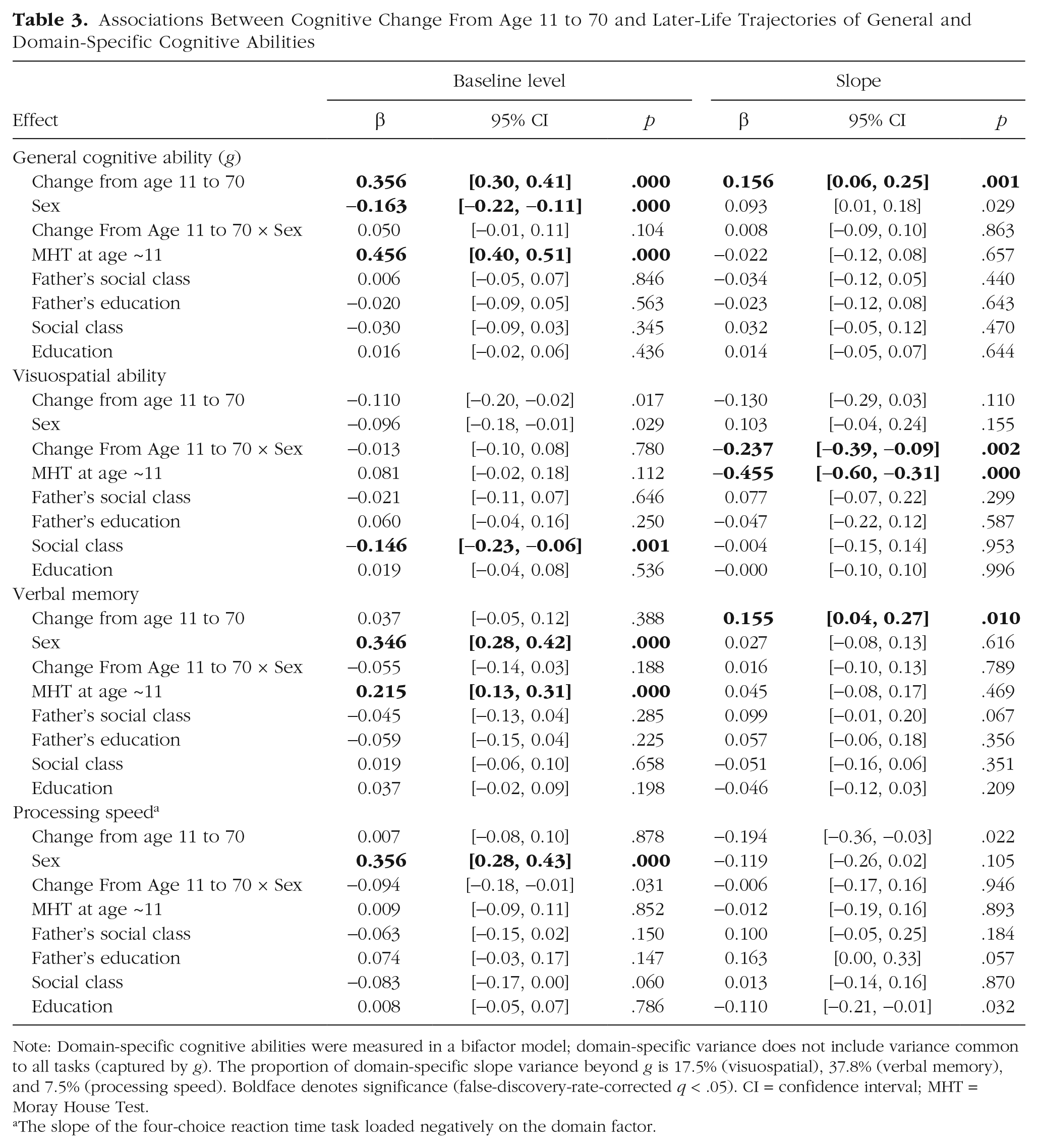
Note: Domain-specific cognitive abilities were measured in a bifactor model; domain-specific variance does not include variance common to all tasks (captured by
The slope of the four-choice reaction time task loaded negatively on the domain factor.
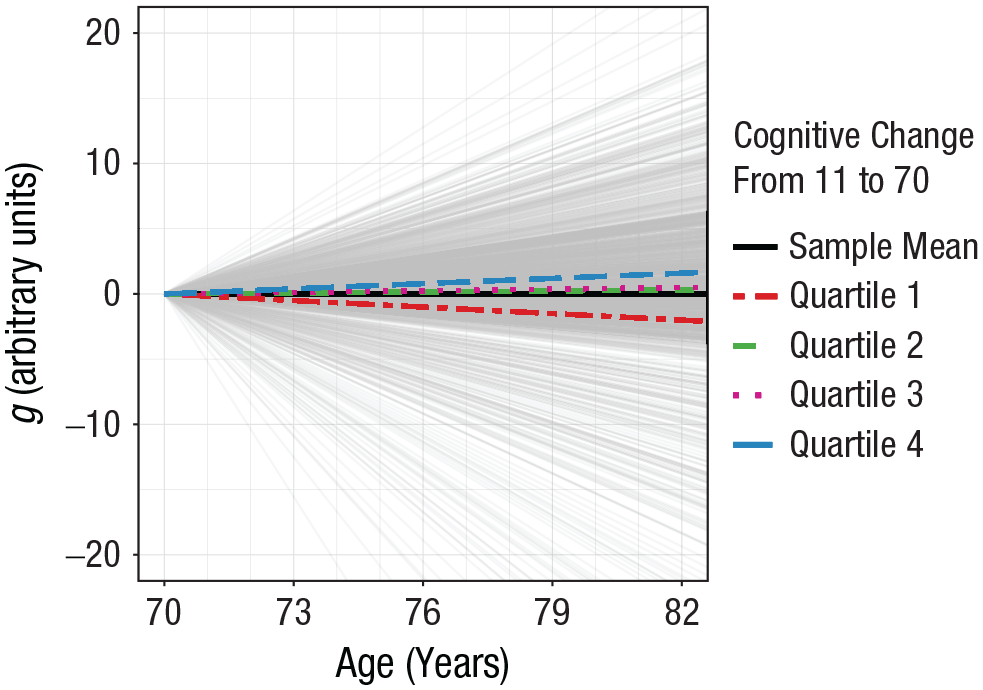
Estimates of standardized change in general cognitive ability (
MHT change between 11 and 70 remained a significant predictor of cognitive decline from 70 to 82 even after
The next analyses involved changes in the cognitive domains from which variance in
Supplemental analyses
The Supplemental Material presents results from our analyses (a) measuring MHT change at ages 11 to 70 with a difference score, first on the entire sample and then on the subsamples showing reliable change in scores; (b) correcting for within-wave age differences; and (c) fitting individual domain models without partialing out general variance.
When conducting analyses on raw MHT change at ages 11 to 70, the direction and magnitude of effects on
Controlling for age differences within each wave of testing had no significant impact, as illustrated in Table S4.
The direction and size of effects in individual domain models (see Table S5 in the Supplemental Material) were essentially similar to those observed on
NART-based measures of cognitive change as predictors of individual differences in later-life cognitive trajectories
We found that MHT change from ages 11 to 70—that is, across nearly six decades—predicted subsequent cognitive changes from ages 70 to 82. We then used NART scores at age 70 as an estimate of peak adult cognitive ability to investigate whether change from childhood to peak ability, or from peak to age-70 ability, might be differentially important. We kept the same bifactor cognitive-measurement model as in the main analysis (see Section 2.3.1 in the Supplemental Material). Cognitive change from age 11 to peak was
Having higher NART scores than expected on the basis of MHT at age 11 was associated with higher baseline level in
Associations Between Cognitive Change From Age-11 to Peak and Later-Life Trajectories of General and Domain-Specific Cognitive Abilities
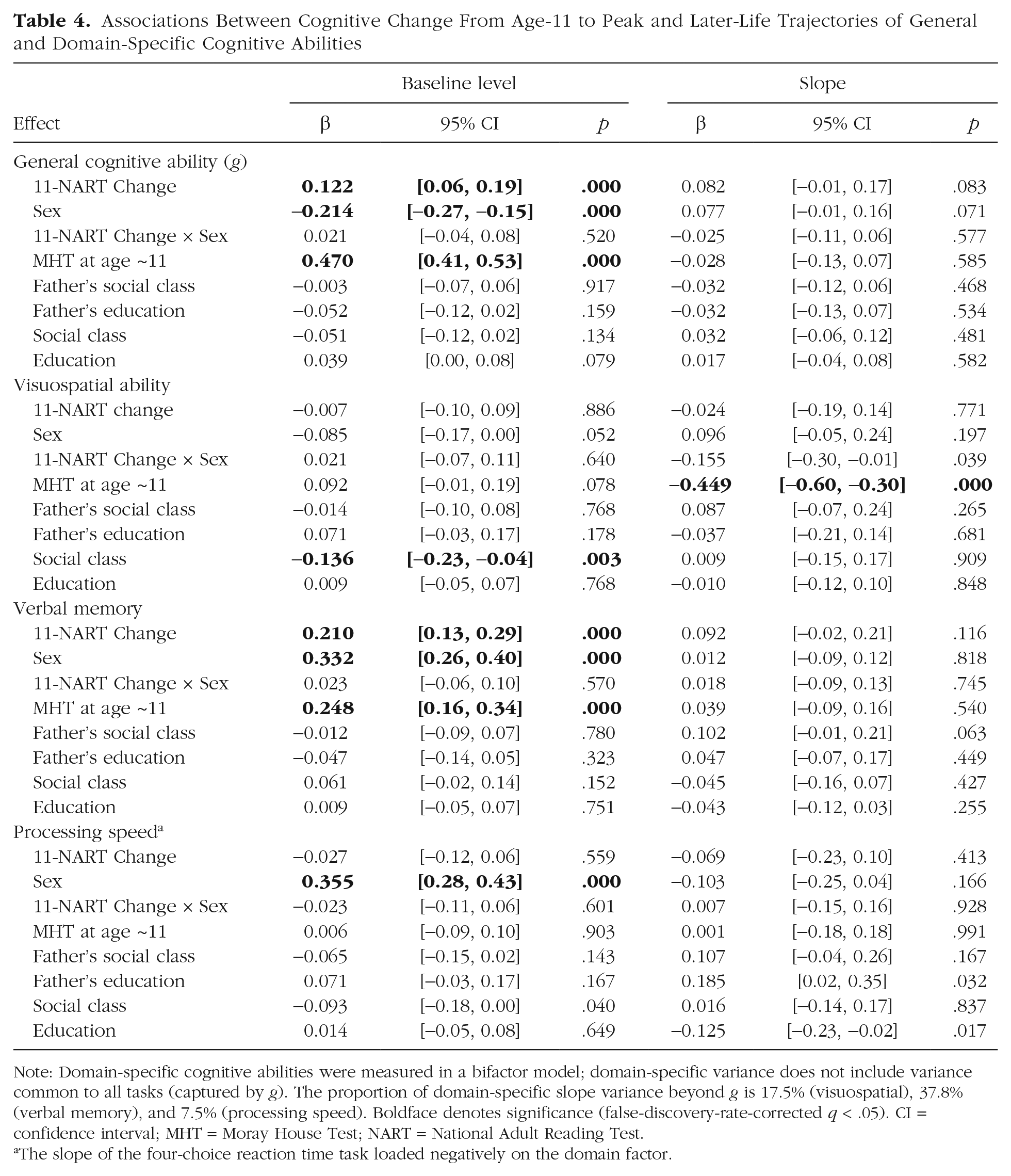
Note: Domain-specific cognitive abilities were measured in a bifactor model; domain-specific variance does not include variance common to all tasks (captured by
The slope of the four-choice reaction time task loaded negatively on the domain factor.
Associations Between Cognitive Change From Peak to Age 70 and Later-Life Trajectories of General and Domain-Specific Cognitive Abilities
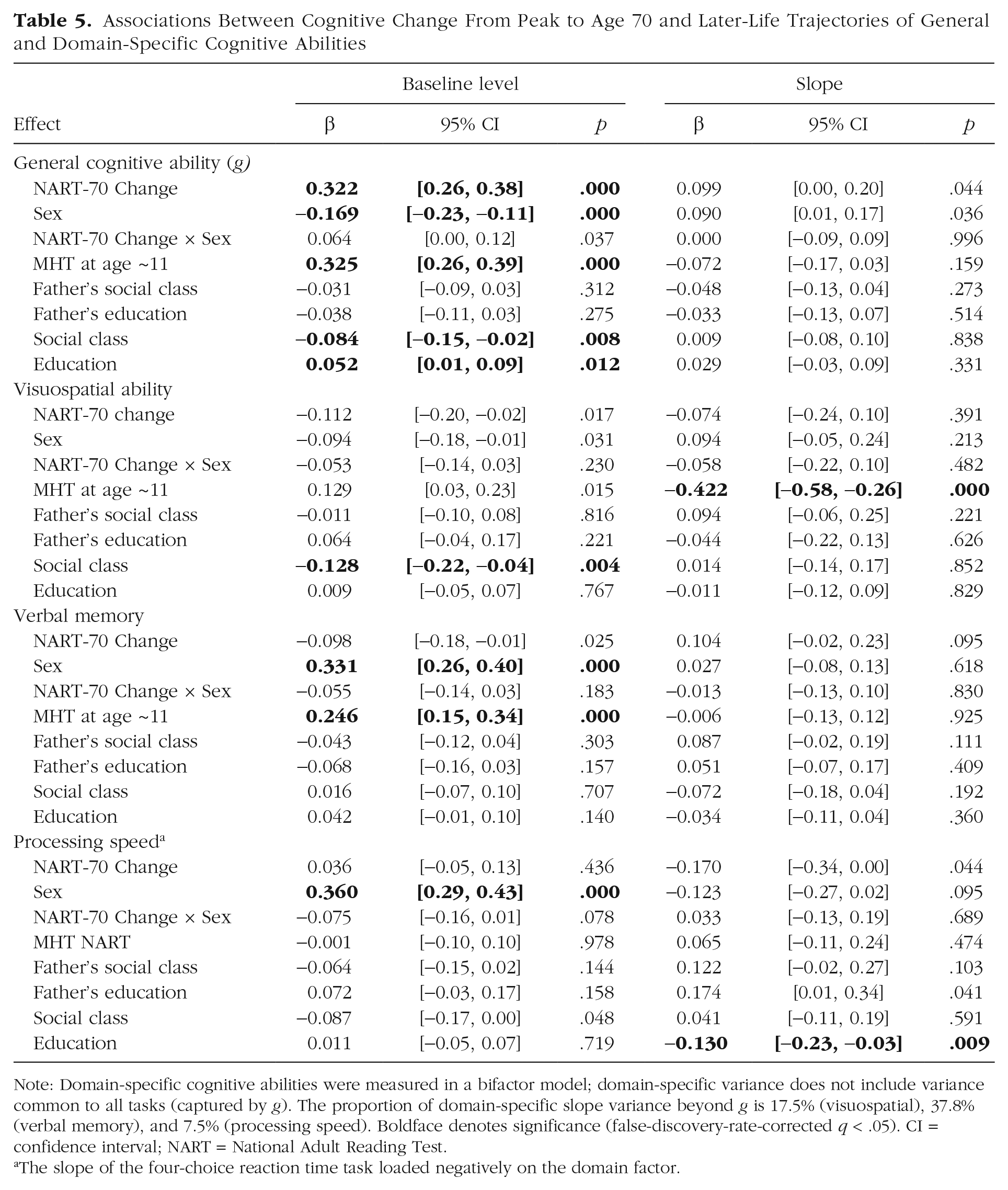
Note: Domain-specific cognitive abilities were measured in a bifactor model; domain-specific variance does not include variance common to all tasks (captured by
The slope of the four-choice reaction time task loaded negatively on the domain factor.
Discussion
Our main finding is that individual differences in cognitive change between ages 11 and 70—measured on the same general-ability test—significantly predicted differences in
We did not detect any significant association between years of education and cognitive level at age 70, in apparent contrast with previous LBC and meta-analytical investigations (Lövdén et al., 2020; Ritchie et al., 2016). However, according to follow-up analyses, the result is due to the simultaneous inclusion of childhood cognitive ability and cognitive change between 11 and 70 as model predictors, both of which correlate with education and consequently attenuate its associations with cognitive differences at age 70.
The bifactor model differentiated
The association of earlier cognitive change (ages 11–70) with later decline in
The predictive effect of cognitive change from 11 to 70 seemed pervasive, being significant also with regard to domain-specific decline. Greater relative improvement in MHT scores from age 11 to 70 was associated with better preservation of verbal memory and with steeper decline in visuospatial abilities at later ages (the latter only in women). These effects were less stable than those on
Why did change between 11 and 70 predict change in
New questions arise as to what lifetime period might be most informative about age-related cognitive decline: Would it be, say, between childhood and early adulthood, or from mid- to later life? We partially answered such questions using the NART at age 70 as an indicator of peak cognitive ability and assessing change in rank orders from age-11 MHT to NART and from NART to age-70 MHT. Previous Lothian Birth Cohort studies showed that NART-based cognitive-change estimates correlate strongly with actual cognitive change (Deary et al., 2004). Despite this, the absence of significant associations with rates of change in
Limitations
Limitations should be considered when interpreting our results and may help inform future research. The Lothian Birth Cohort studies provide direct measures of participants’ cognitive abilities in childhood and older age. However, only a single MHT total score is available at each time point, preventing the testing of factorial invariance or latent-change-score modeling. Despite this, existing literature (e.g., Terman & Merrill, 1937) and consistency of results between main and supplementary analyses support our estimate of MHT test-retest reliability.
No cognitive tests were administered between ages 11 and 70. Thus, our measure of cognitive change likely reflects the lifelong influence of multiple factors and encompasses nonlinear changes (e.g., cognitive development and early decline). However, the effects of cognitive change between 11 and 70 were robust to the introduction of cognitive and socioeconomic covariates, and we could use a strong index such as the NART to further specify cognitive trajectories. We hope that further research can identify potential critical periods during which earlier-life cognitive change anticipates later-life decline.
Compared with the population average, LBC1936 cohort members tend to be healthier and better educated and tend to perform better on cognitive-ability tests (Taylor et al., 2018), likely leading to some restriction of range and a slight reduction in effect sizes (e.g., Johnson et al., 2011). Finally, participants were all born in a single year and come from a particular geographical setting, thereby limiting our results’ generalizability, albeit removing the possibility of cohort effects in a mixed-age sample.
Conclusion
Research indicates that individual differences in cognitive decline arise from many diverse factors, each exercising a small influence (Corley et al., 2018; Deary et al., 2012). Tracing cognitive trajectories back through the life course requires data that are rarely available. The present study shows that cognitive change between ages 11 and 70 is independently informative of cognitive trajectories from ages 70 to 82, beyond cognitive level at age 11 or 70. Therefore, the results support identifying individuals at higher risk of cognitive decline before the critical years in which dementia risk accelerates. The positive side to the findings is that, to some extent, those who fare better cognitively from ages 11 to 70 tend to be at lower risk of cognitive decline from 70 to 82. To quote a sentence often attributed to Fred Astaire (1899–1987), “Old age is like everything else . . . to make a success of it, you’ve got to start young.”
Supplemental Material
sj-docx-1-pss-10.1177_09567976221100264 – Supplemental material for Cognitive Change Before Old Age (11 to 70) Predicts Cognitive Change During Old Age (70 to 82)
Supplemental material, sj-docx-1-pss-10.1177_09567976221100264 for Cognitive Change Before Old Age (11 to 70) Predicts Cognitive Change During Old Age (70 to 82) by Federica P. Conte, Judith A. Okely, Olivia K. Hamilton, Janie Corley, Danielle Page, Paul Redmond, Adele M. Taylor, Tom C. Russ, Ian J. Deary and Simon R. Cox in Psychological Science
Footnotes
Acknowledgements
We thank the participants in the Lothian Birth Cohort 1936 and the research team who collected, entered, and checked the data used in this article.
Transparency
F. P. Conte, I. J. Deary, J. Corley, J. A. Okely, O. K. Hamilton, and S. R. Cox contributed to the conceptualization and methodological design of the study. In addition, F. P. Conte was responsible for the formal analysis and writing of the original draft; A. M. Taylor was involved in the investigation and project administration; I. J. Deary and S. R. Cox were responsible for project administration, funding acquisition, and supervision. D. Page contributed to investigation and data curation. P. Redmond contributed to data curation. All authors reviewed and edited the original draft and approved the final version for submission.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
