Abstract
Background
Single-tablet regimens (STRs) with integrase inhibitors, bictegravir (BIC) or dolutegravir (DTG), are favored in HIV treatment for their efficacy and convenience. This study compares persistence—time from initiation to discontinuation—between BIC/emtricitabine (FTC)/tenofovir alafenamide (TAF) and DTG-containing STRs at a Toronto HIV clinic
Methods
A retrospective cohort analysis was conducted on 1732 adults with HIV at Maple Leaf Medical Clinic who initiated or switched to BIC/FTC/TAF or DTG-containing STRs from 2016 to 2022. Persistence was measured in days until discontinuation. Kaplan-Meier curves and Cox models evaluated time-to-discontinuation and associated risks. Reasons for discontinuation were categorized into adverse events, patient preference, cost, compliance, physician preference, virologic failure, and others.
Results
Among 1732 participants (median age 48 years, 88.7% cisgender men), 387 (22.3%) discontinued their STRs after a median of 402 days. BIC/FTC/TAF had a lower discontinuation rate (18.9%) compared to DTG-containing STRs (29.9%) (HR = 0.74, 95% CI: 0.60–0.92). Adverse events were the primary reason for discontinuation, with BIC having lower rates (9.6% vs. 12.5% for DTG).
Discussion
BIC/FTC/TAF demonstrated higher persistence and fewer adverse events than DTG-containing STRs, aiding personalized HIV treatment decisions for better long-term outcomes.
Introduction
The advent of single-tablet regimens (STRs) revolutionized the antiretroviral treatment (ART) landscape for people with HIV.1,2,3,4 STRs-containing integrase strand transfer inhibitors (INSTIs), such as bictegravir (BIC) and dolutegravir (DTG), are now widely recommended in international guidelines as first-line ART due to their robust efficacy, tolerability, and simplicity. 5 By reducing pill burden, STRs promote adherence, thereby improving viral suppression and long-term clinical outcomes.2,6,7 However, real-world persistence, defined as the duration patients remain on a prescribed regimen, 8 is critical for assessing long-term success and tolerability outside controlled clinical trial settings. Understanding factors influencing regimen discontinuation informs clinical decision-making and supports the optimization of person-centered HIV care.
BIC/emtricitabine (FTC)/tenofovir alafenamide (TAF) and dolutegravir (DTG)-containing STRs have both demonstrated strong persistence in real-world studies.9,10 BIC/FTC/TAF is often noted for its favorable efficacy and long-term safety profile, once-daily dosing, and high barrier to drug resistance, contributing to high persistence rates across diverse patient populations. 11 Similarly, DTG-containing STRs remain widely prescribed due to their high efficacy and tolerability, particularly among people with HIV transitioning from established regimens or in specific clinical situations.12,13 Real-world evidence indicates robust persistence for DTG-based regimens despite challenges such as weight gain, 14 neuropsychiatric side effects,15,16 and gastrointestinal symptoms.17,18 While some studies suggest differences in discontinuation rates between BIC- and DTG-based regimens,10,19,20 further investigation into persistence trends and factors influencing regimen discontinuation, will help optimize the person-centered use of these widely prescribed therapies.
Evaluating real-world persistence requires a nuanced understanding of the reasons for ART discontinuation. Discontinuation may occur due to adverse events, patient preference, adherence challenges, cost or insurance coverage, and treatment failure. Comparing the persistence of BIC/FTC/TAF versus DTG-containing STRs across a large, real-world cohort can reveal important insights into regimen tolerability, durability, and person-centered outcomes, helping to meet the evolving needs of people with HIV beyond the goal of virologic suppression.
This study aimed to compare the real-world persistence of BIC/FTC/TAF and DTG-containing STRs in a large urban Canadian HIV center. Specifically, we aimed to: (1) assess regimen persistence, defined as time-to-discontinuation, between BIC/FTC/TAF versus DTG-containing STRs overall and by specific regimen, abacavir (ABC)/lamivudine (3TC)/DTG, 3TC/DTG, and rilpivirine (RPV)/DTG, (2) identify the primary reasons for discontinuation, including adverse events and patient preference, and 3) explore potential differences by regimen type.
Methods
Study design, setting and population
This was a retrospective cohort study conducted at the Maple Leaf Medical Clinic, a large urban HIV center in Toronto, Canada, specializing in HIV care and management with 14 physicians caring for over 3,000 people living with HIV. The study included adults aged 18 years or older, living with HIV who initiated or switched to BIC/FTC/TAF or a DTG-containing STR, between January 1, 2016, and December 31, 2022. 2016 was selected as the index year for cohort inclusion because it offered a methodologically balanced midpoint between the Canadian licensure dates of ABC/3TC/DTG (October 14, 2014) and BIC/F/TAF (July 10, 2018). This aimed to minimize temporal bias from artificially inflated discontinuation rates for and sufficient sample size of ABC/3TC/DTG during the study timeframe. DTG-containing STRs were categorized into three subgroups: ABC/3TC/DTG, 3TC/DTG, and RPV/DTG. Inclusion criteria further consisted of adults living with HIV who had a documented start date for the selected STRs within the study period. People were excluded if they had incomplete prescription data or were receiving other antiretroviral drug(s) in addition to BIC/FTC/TAF or DTG-containing STRs (i.e., they required to be solely taking BIC/FTC/TAF or a DTG-containing STR). Eligible participants were identified through the clinic’s electronic medical record (EMR) system. Participants were followed until December 31, 2023, or until the occurrence of discontinuation, loss to follow-up, transfer of care or death.
Data collection and study outcomes
Data were extracted retrospectively from the clinic’s EMR system to assess persistence and reasons for discontinuation of the BIC- and DTG-containing-STRs. Collected variables included demographic and clinical characteristics, such as age, sex, ethnicity, duration of HIV, baseline viral load and CD4 + cell count, hepatitis B and C status, and if they were ART-naïve or -experienced at the start of their STR. Regimen information, including the type of STR (BIC/FTC/TAF, ABC/3TC/DTG, 3TC/DTG, RPV/DTG), as well as the initiation or switch date were documented.
The primary outcome was regimen persistence, defined as the number of days from STR initiation to discontinuation. Discontinuation was identified as either a documented prescription switch to a different ART regimen or a delay in prescription refill exceeding 90 days from the last recorded STR prescription.
Reasons for discontinuations, which were identified through manual chart review, were categorized into predefined groups. These predefined groups included: (1) adverse events including central nervous system (CNS) effects, weight gain, gastrointestinal side effects, other side effects; (2) toxicity including renal toxicity, liver toxicity, other toxicity, and allergic reaction; (3) patient preference including switch to injectable, enrolled in a clinical study, preference for another medication, pill property (e.g., pill size), and other reason; (4) cost/coverage; (5) compliance issue; (6) physician preference including renal protection, cardiovascular risk reduction, drug efficacy for comorbidity, simplification, weight reduction, non-adherence, and other reasons; (7) virologic failure; and (8) other including drug interactions, pregnancy, other reasons, or discontinuation classified as unspecified. For each participant who discontinued their STR, the specific reason for discontinuation was carefully reviewed and documented by one of the co-authors (NM). Chart review data that was unclear was reviewed with three co-authors (ML, JM, GS) for verification and adjudication.
Statistical analysis
Statistical analyses were performed using R version 4.4.0 (R Core Team), and all tests were two-sided with a significance level of p < .05. Descriptive statistics were used to summarize baseline demographic and clinical characteristics of the cohort. Continuous variables, such as age, were reported as medians with interquartile ranges (IQRs). Categorical variables, including regimen type, were summarized as frequencies and proportions. Differences in baseline characteristics between the BIC/FTC/TAF group and the DTG-containing STR group were assessed using Wilcoxon rank sum tests for continuous variables and chi-squared tests or Fisher’s exact tests for categorical variables.
Persistence, the primary outcome, was first summarized as a proportion of those who discontinued their STR and then was defined as the median time (in days) from initiation or switch to an STR until discontinuation. Kaplan-Meier survival curves were used to estimate the time-to-discontinuation for BIC/FTC/TAF and DTG-containing STRs. The log-rank test was applied to compare survival distributions between the regimens. Cox proportional hazards regression models were used to calculate hazard ratios (HRs) and 95% confidence intervals (CIs) to assess the risk of discontinuation. Both unadjusted and adjusted analyses were performed to account for baseline demographic differences and subgroup variations. The final adjusted model includes baseline variables considered significant using backward selection. To ensure robust analyses, participants were only counted once in the dataset. For example, if a participant started and stopped BIC/FTC/TAF and then switched to a DTG-containing regimen (or vice versa), only their first regimen was included in the analysis to avoid duplication and bias.
To account for clinically insignificant switches, specific rules were applied. For participants switched from ABC/3TC/DTG to 3TC/DTG, this switch was treated as a “not clinically significant simplification”, given the similarities between the regimens and the known advantages of 3TC/DTG and not counted as a discontinuation. In contrast, any switch from ABC/3TC/DTG to DTG/RPV was considered a significant discontinuation in all analyses due to the clinical and pharmacological differences between these regimens.
Finally, subgroup analyses were conducted within the DTG-containing STR cohort to explore persistence differences across ABC/3TC/DTG, 3TC/DTG, and RPV/DTG subgroups. Reasons for discontinuation were compared across regimens using descriptive statistics with frequencies and proportions and compared using and chi-squared tests.
Research ethics approval
This study was approved by the Research Ethics Board (REB) at the University of Toronto and adhered to the principles outlined in the Declaration of Helsinki 21 and ethical principles of research with trans communities. 22 Given the retrospective nature of the study and the use of de-identified data, a waiver of informed consent was granted. All data were securely stored, accessed only by authorized personnel, and analyzed in compliance with confidentiality and privacy regulations.
Results
Cohort creation and study population
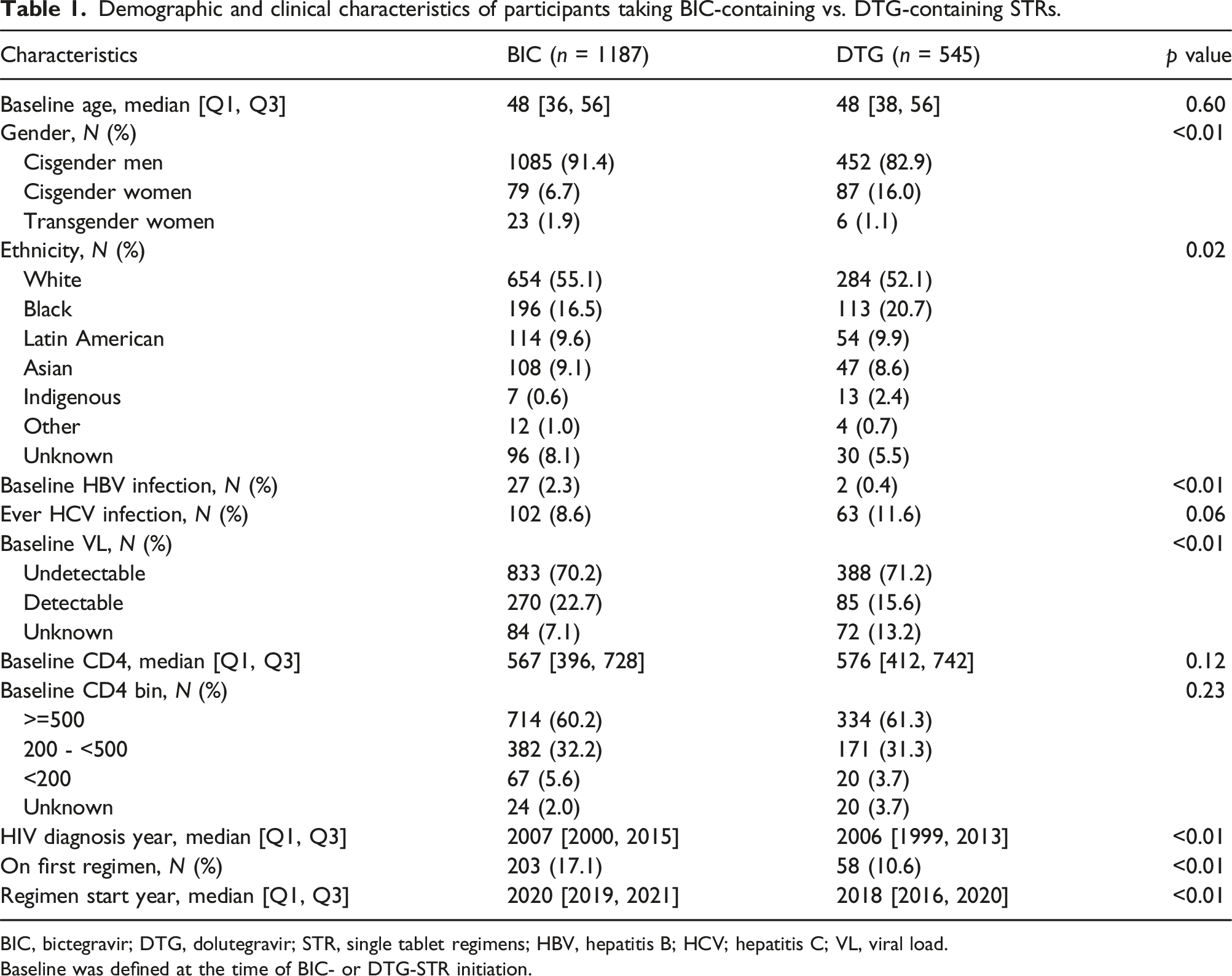
From 2016 to 2022, there were a cumulative 3,739 adults with HIV receiving care at the Maple Leaf Medical Clinic. Of these, 2,007 people were excluded from the analysis due to incomplete prescription data or receiving regimens outside the scope of this study. This left a final cohort of 1,732 participants who initiated or switched to BIC- or DTG-containing STRs (Figure 1). Of 1,732 participants, 1,187 initiated BIC/FTC/TAF, and 545 initiated DTG-containing STRs. Within the DTG-containing group, 359 (65.9%) were prescribed ABC/3TC/DTG, 113 (20.7%) were on 3TC/DTG, and 73 (13.4%) were on RPV/DTG. Among those on BIC/FTC/TAF (n = 1,187), 203 were new starts, while the remaining 984 switched from other regimens. For participants on ABC/3TC/DTG (n = 359), 53 were new starts and 306 were switches. In the 3TC/DTG group (n = 113), only five were new starts, and 108 had switched. Finally, all people prescribed RPV/DTG (n = 73) had switched from a prior regimen. The median age of study participants was 48 years (IQR = 36–56). Most of the sample identified as white (58.4%), cisgender men (88.7%), and had an undetectable viral load at baseline (70.4%). Participants had a median duration of HIV diagnosis of 17 years (IQR = 10–25). Further demographic and clinical characteristics of the study population by regimen (BIC vs. DTG containing STRs) are presented in Table 1. Demographic and clinical characteristics of the study population by specific STR, BIC/FTC/TAF, ABC/3TC/DTG, 3TC/DTG, and RPV/DTG are in Supplemental Table S1. Flow diagram for the creation of study participants for the current analysis. All active Maple Leaf Medical Clinic patients were screened for eligibility. 46% of the total clinic population met the inclusion criteria. Demographic and clinical characteristics of participants taking BIC-containing vs. DTG-containing STRs. BIC, bictegravir; DTG, dolutegravir; STR, single tablet regimens; HBV, hepatitis B; HCV; hepatitis C; VL, viral load. Baseline was defined at the time of BIC- or DTG-STR initiation.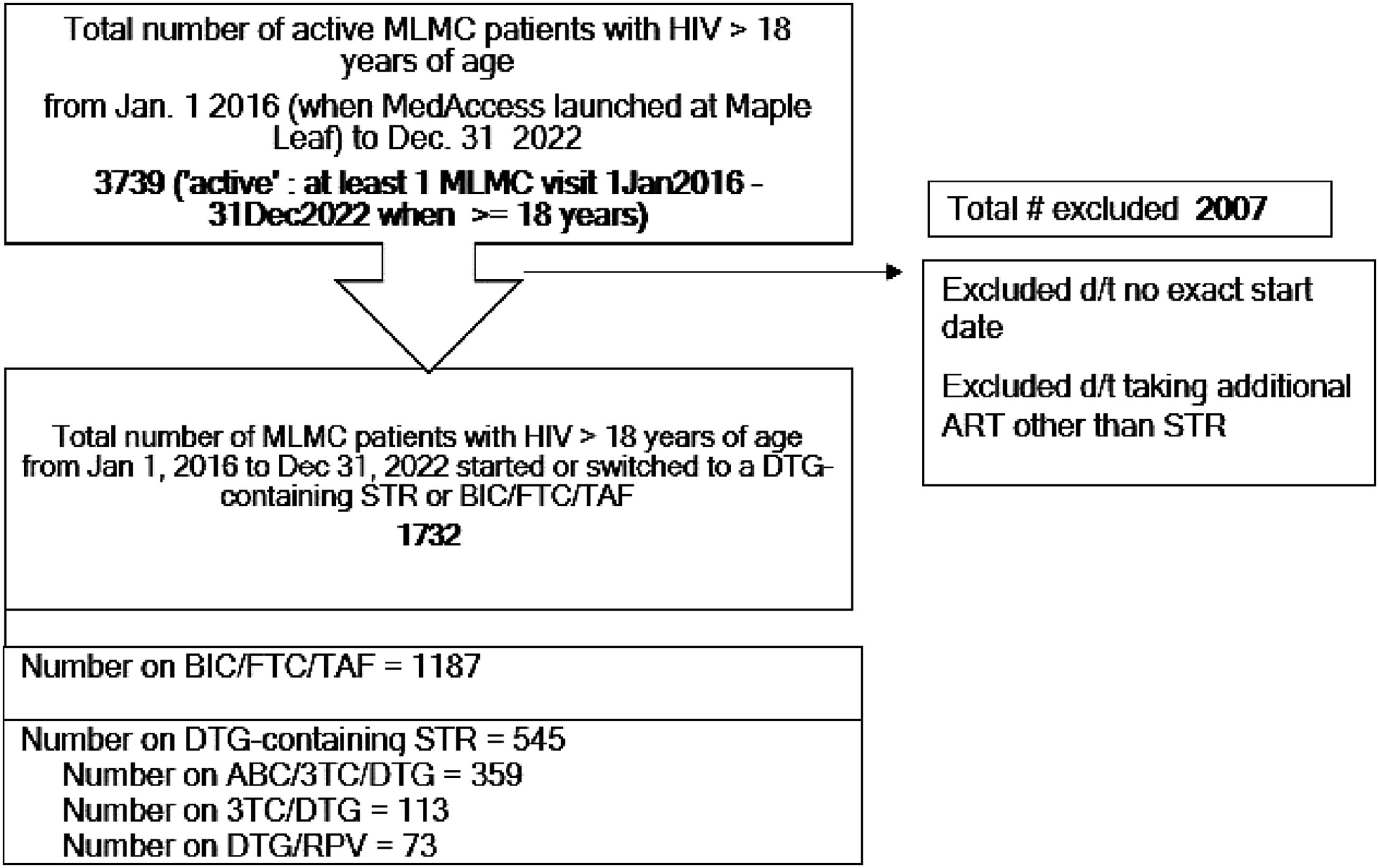
Persistence analysis
By the end of the analysis period (December 31, 2023), 387 (22.3%) had discontinued their STRs overall. Of the 1187 patients in the BIC/FTC/TAF group, 224 (18.9%) discontinued their regimen after a median time of 383 days (IQR: 140–774). In contrast, among the 545 patients in the DTG-containing group, 163 (29.9%) discontinued therapy, with a median time to discontinuation of 479 days (IQR: 156–1042). Kaplan-Meier survival analysis in Figure 2 demonstrated a significant difference in persistence between BIC/FTC/TAF and DTG-containing STRs (log-rank p < .01). Persistence Analysis. Kaplan-Meier curve comparison of persistence for bictegravir (BIC) vs. dolutegravir (DTG)-containing single tablet regimens.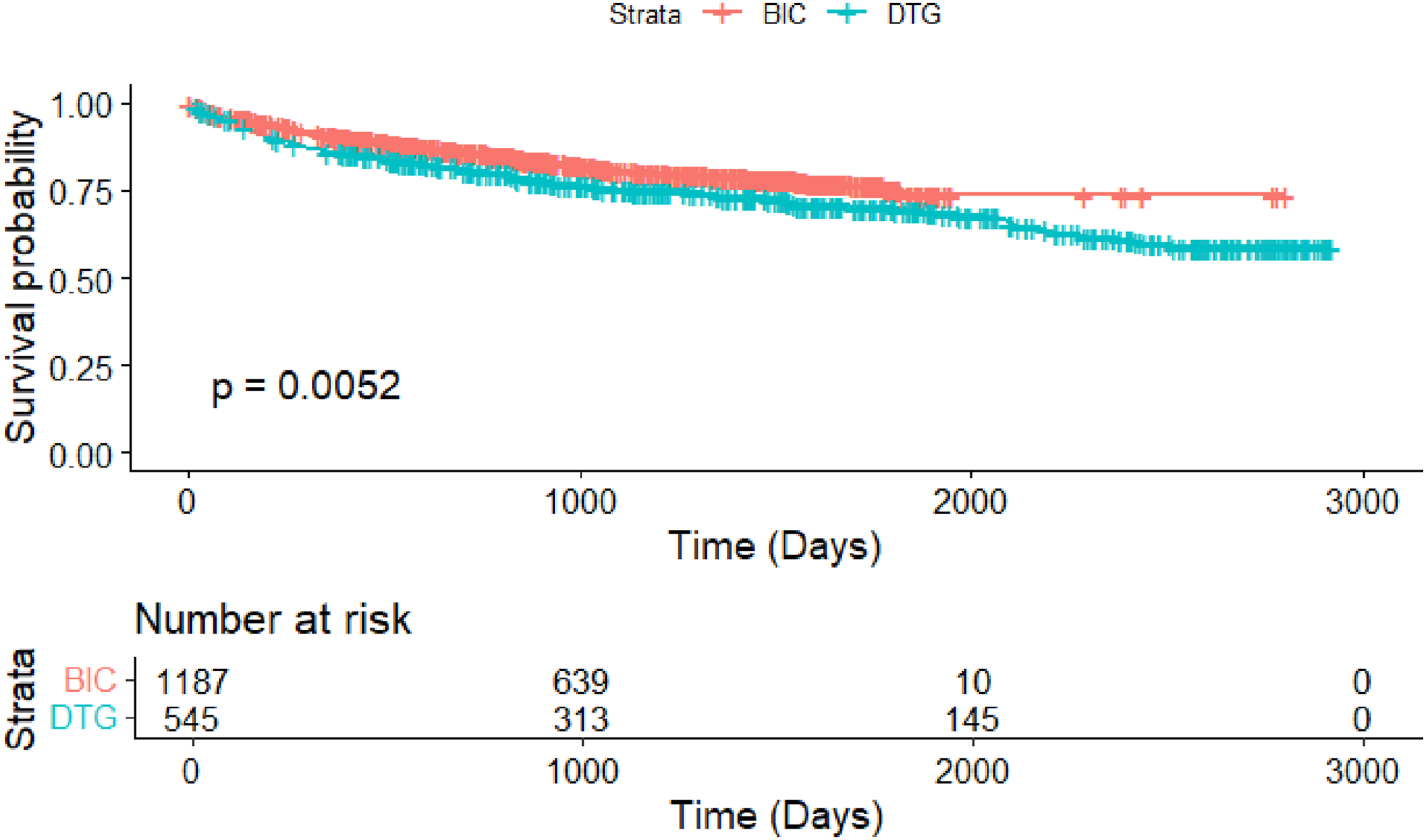
Univariate and multivariate Cox regression for discontinuation (by INSTI).
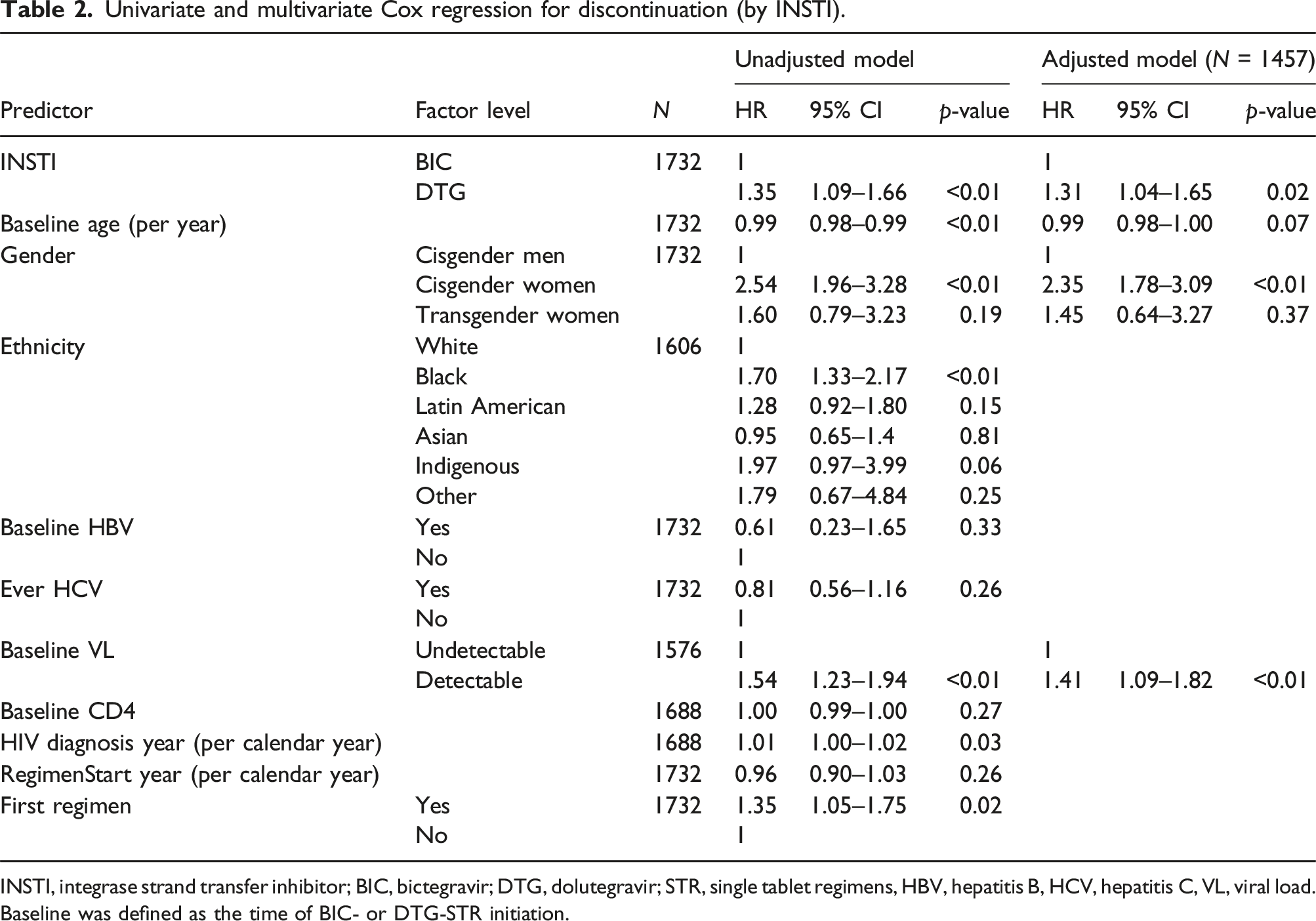
INSTI, integrase strand transfer inhibitor; BIC, bictegravir; DTG, dolutegravir; STR, single tablet regimens, HBV, hepatitis B, HCV, hepatitis C, VL, viral load. Baseline was defined as the time of BIC- or DTG-STR initiation.
Reasons for discontinuation
Overall, 22.3% (387/1,732) of patients discontinued their STR during the study period. Discontinuation rates varied by regimen: 18.9% (224/1,187) of patients on BIC/FTC/TAF discontinued treatment compared to 29.9% (163/545) on DTG-containing STRs. Among the DTG subgroups, discontinuation rates were 35.1% (126/359) for ABC/3TC/DTG, 15.9% (18/113) for 3TC/DTG, and 26.0% (19/73) for RPV/DTG.
Discontinuation reasons by INSTI.
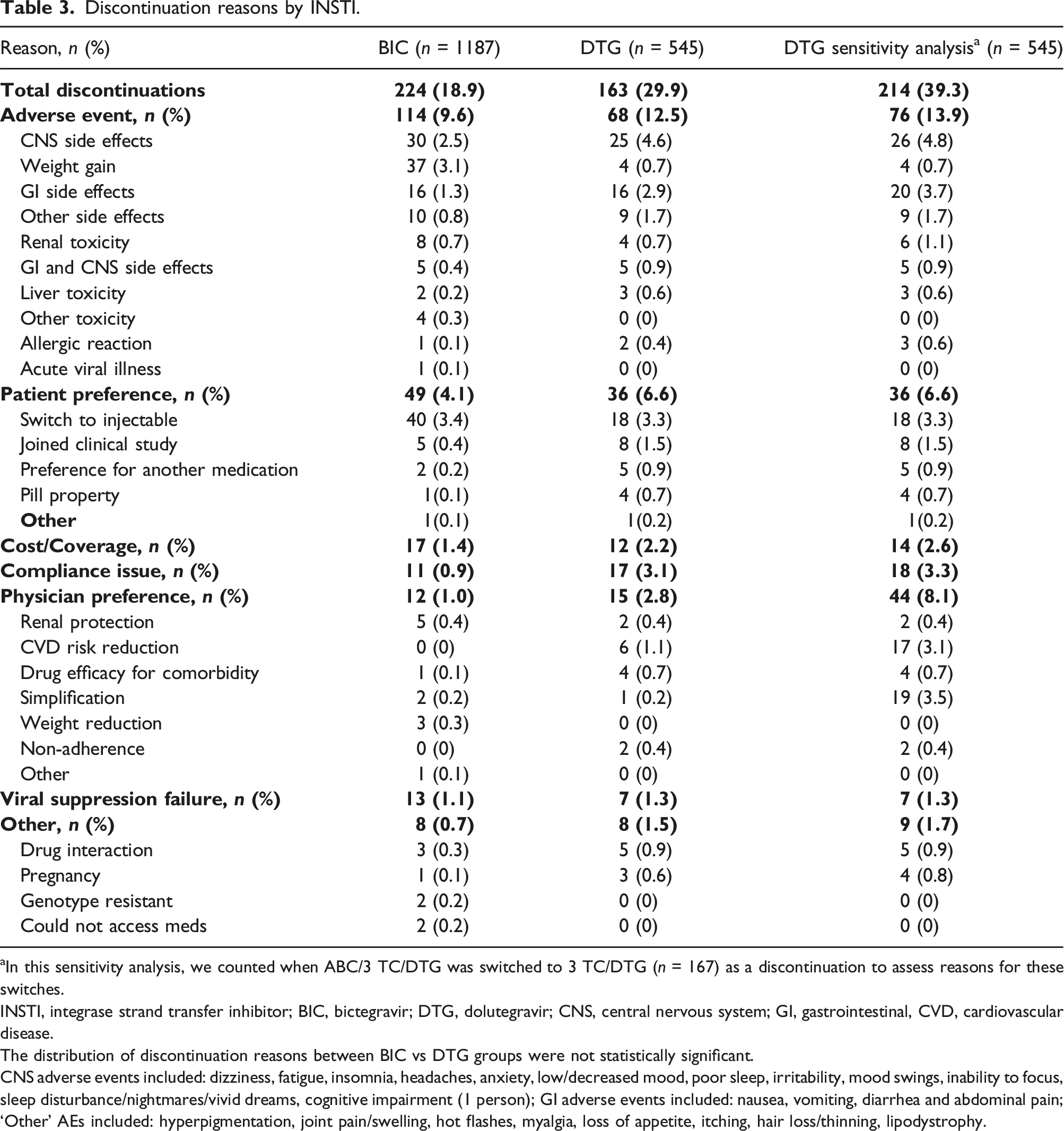
aIn this sensitivity analysis, we counted when ABC/3 TC/DTG was switched to 3 TC/DTG (n = 167) as a discontinuation to assess reasons for these switches.
INSTI, integrase strand transfer inhibitor; BIC, bictegravir; DTG, dolutegravir; CNS, central nervous system; GI, gastrointestinal, CVD, cardiovascular disease.
The distribution of discontinuation reasons between BIC vs DTG groups were not statistically significant.
CNS adverse events included: dizziness, fatigue, insomnia, headaches, anxiety, low/decreased mood, poor sleep, irritability, mood swings, inability to focus, sleep disturbance/nightmares/vivid dreams, cognitive impairment (1 person); GI adverse events included: nausea, vomiting, diarrhea and abdominal pain; ‘Other’ AEs included: hyperpigmentation, joint pain/swelling, hot flashes, myalgia, loss of appetite, itching, hair loss/thinning, lipodystrophy.
Patient preference was the second most common discontinuation reason, reported in 4.9% (85/1,732) of cases. This included dissatisfaction with regimen tolerability, concerns about pill burden, or perceived side effects. Patient preference-related discontinuation occurred at a higher rate in DTG-containing STRs (6.6%, 36/545) compared to BIC/FTC/TAF (4.1%, 49/1,187). Compliance issues, such as missed appointments, irregular ART use, or interruptions in care, accounted for 1.6% (28/1,732) of all discontinuations. These issues were more prominent in DTG-containing STRs (3.1%, 17/545) than in BIC/FTC/TAF (0.9%, 11/1,187). Cost or insurance-related challenges contributed to 1.7% (29/1,732) of discontinuations, with no substantial difference observed between the two regimen groups. Similarly, physician-directed switches, often driven by efforts to simplify treatment regimens or address specific patient concerns, represented 1.6% (27/1,732) of all discontinuations. Treatment failure, defined as virologic rebound or loss of viral suppression, was relatively uncommon, occurring in 1.1% (20/1,732) of participants. Rates of treatment failure were similar between BIC/FTC/TAF and DTG-containing STRs, indicating that both regimens maintained virologic efficacy in most patients. Finally, other, or unspecified reasons accounted for 0.9% (16/1,732) of discontinuations.
Discussion
With the simplification of ART that is afforded by STRs, understanding treatment patterns, such as persistence,8,23 is pertinent for the future of optimizing and personalizing HIV treatment. This study provides valuable insights into the persistence of BIC- and DTG-containing STRs in a real-world setting. Discontinuation rates were numerically lower for those on BIC/FTC/TAF (18.9%) compared to those on DTG-containing regimens (29.9%). While the median time to discontinuation was shorter at 383 days for the BIC/FTC/TAF than for the DTG group at 479 days, there was a higher risk of discontinuation observed in DTG-containing regimens (HR: 1.35; 95% CI: 1.09–1.66), especially in the ABC/3TC/DTG subgroup (HR: 1.43; 95% CI: 1.13–1.80). The longer time to discontinuation in the DTG group likely reflects the longer availability of ABC/3TC/DTG in the Canadian market, and that there were limited other regimens to change to at the time. The primary reason for discontinuation across all groups was adverse events, accounting for 47% (182/387) of cases. Higher rates were observed in the DTG- containing regimens group with CNS 24 and gastrointestinal symptoms being more common. Other discontinuation reasons included patient preference, which favored BIC/FTC/TAF, compliance issues, and cost. Several factors may contribute to patient preference for BIC/FTC/TAF, including the relative pill size compared to all other STRs. Virologic efficacy was maintained, as treatment failure rates were low and similar across both groups.
The study’s finding that 22.3% of participants discontinued their STRs aligns with previous literature8,19,20,22,23,24,25 reporting similar rates. The novel contribution of this study lies in its detailed comparison of specific regimens and in providing a comprehensive analysis of reasons for discontinuation. By including details of the adverse events and patient preference, and identifying variations in persistence among different DTG subgroups, the study offers valuable insights into regimen-specific challenges and supports more personalized treatment approaches.
The study highlights that the persistence of BIC/FTC/TAF was notably higher compared to DTG-containing regimens, with only 18.9% of patients discontinuing BIC/FTC/TAF therapy. The persistence pattern associated with BIC/FTC/TAF was also evident in the Kaplan-Meier analysis which demonstrated significantly higher persistence rates for BIC compared to DTG (p < .05). This persistence suggests that BIC/FTC/TAF is better tolerated, as reflected by the lower incidence of discontinuation due to adverse events (9.6%), compared to DTG regimens (12.5%). These findings are similar to those of other studies which showed higher persistence of BIC/FTC/TAF compared to other INSTI-based regimens.19,20,25 Our findings emphasize the potential of BIC/FTC/TAF as a more sustainable option for people with HIV, contributing to its favorable profile in maintaining long-term treatment persistence. This aligns with existing literature and supports its use as a preferred regimen in clinical practice.9,10
While our analyses demonstrated that people on DTG-containing regimens had a higher likelihood of discontinuation compared to those on BIC/FTC/TAF, DTG-containing STRs remain an important option for people with HIV. The higher discontinuation rates in DTG regimens, at 29.9%, highlight areas for improvement in managing adverse events and patient preferences. Also, the study’s findings do contribute to a nuanced understanding of regimen-specific persistence, reinforcing the importance of individualized treatment plans to optimize outcomes across diverse patient populations and contribute to long-term success for people with HIV.
In addition to the regimen type, several demographic and clinical factors emerged as predictors of discontinuation. Notably, younger individuals, cisgender women, and individuals identifying as Black exhibited higher discontinuation rates, corroborating findings from earlier studies that highlighted disparities in treatment persistence linked to gender26,27,28,29 and ethnicity.28,29,30 These insights signal an urgent need to explore challenges faced by cisgender women and Black individuals and tailor support and interventions for these populations to enhance persistence and ultimately improve health outcomes.
The reasons for discontinuation provided further depth to the findings of the current study. Adverse events emerged as the predominant reason, accounting for nearly half of all discontinuations. The specific nature of these adverse events—including CNS effects, weight gain, and gastrointestinal issues—suggests the need for careful monitoring and management of side effects in clinical practice. Moreover, patient preference and cost/coverage issues also played a significant role in discontinuation, highlighting the importance of addressing the broader social determinants of health that can impact treatment adherence and persistence. Overall, this study contributes to the growing body of evidence surrounding the effectiveness and tolerability of BIC/FTC/TAF in the HIV treatment landscape.9,10
The results of our study should be considered in context of the limitations which include its retrospective observational design, which may introduce selection bias and limit the ability to establish causality. The data primarily comes from a single medical center and the sample was predominately white cisgender men, which could affect generalizability to broader populations. Study findings should also be considered in light of the differences between the single BIC-containing regimen versus three DTG-containing regimens that were included. This limitation within our retrospective dataset was inherent due to the available STRs in Canada during the study period. The study also lacks long-term follow-up data beyond the analysis period, which could provide further insights into persistence over time, particularly for the newer two-drug STRs, 3TC/DTG and DTG/RPV. Lastly, variations in participant demographics and baseline characteristics might influence the outcomes and should be considered when interpreting the results.
Conclusion
This study supports growing evidence of the superior persistence of BIC/FTC/TAF compared to DTG-containing STRs in a real-world clinical setting. The lower discontinuation rates associated with BIC/FTC/TAF may reflect better tolerability and patient satisfaction, underscoring its utility as a first-line and switch ART option. Antiretroviral regimens that have a low potential for adverse events and toxicity may promote better quality of life and adherence which is integral to long-term success. Future studies should explore the long-term impact of treatment persistence on benefits beyond virologic suppression, such as its overall impact on the perception of symptom burden and health-related quality of life in people with HIV.
Supplemental Material
Supplemental Material - Real-world persistence of bictegravir versus dolutegravir single-tablet regimens: A retrospective cohort study in a large urban canadian HIV clinic
Supplemental Material for Real-world persistence of bictegravir versus dolutegravir single-tablet regimens: A retrospective cohort study in a large urban canadian HIV clinic by Mona Loutfy, Negin Mousadifar, Jennifer McCully, Angela Underhill, V Logan Kennedy, Dileesha Fernando, Dylana Mumm, Taban Saifi, Hugh Ngo, Soodi Navadeh, Graham S. Smith in International Journal of STD & AIDS.
Footnotes
Acknowledgements
The authors would like to thank the team at Maple Leaf Medical Clinic for their dedication and detailed management of the electronic medical record, making this analysis possible. They would also like to acknowledge the patients at Maple Leaf Medical Clinic whose data was used in the current analysis. Finally, thank you to Tali Cassidy from GSI for her contributions to the design and analysis of this study.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mona Loutfy and Graham Smith have received honoraria from Gilead Sciences and ViiV Healthcare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Gilead Sciences Canada, Inc.
Ethical approval
We confirm that this study has been reviewed and approved by the Institutional Review Board (IRB) of, under approval number 00046147. All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Individual consent is not obtained for this chart review study as the participating physicians gave consent for their data to be used for this research purpose.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
