Abstract
Introduction
Late diagnosis of HIV is associated with increased morbidity and mortality, and an increased risk of non-infectious comorbidities. On a societal level, late diagnosis leads to higher treatment and healthcare costs and is a major driver of HIV transmission. Despite improvements in other areas of the HIV care pathway, late diagnosis remains an individual and public health concern globally.
Objective
To examine the barriers to HIV testing and highlight successful strategies to improve prompt diagnosis. This review describes the prevalence of late diagnosis in the UK and discusses key factors that contribute to late diagnosis, including the effect of the COVID-19 pandemic. Late HIV diagnosis is lower in the UK than in most other European countries. In this review, pilot projects and ongoing initiatives that have reduced late diagnosis in the UK are highlighted; moreover, further strategies for improving prompt diagnosis are suggested.
Conclusions
Insufficient testing is the fundamental reason for late HIV diagnosis, with societal, systemic, and individual factors all contributing to inadequate testing. Improving access to testing, removing barriers to health-seeking behaviour, and ensuring all people with HIV indicator conditions are promptly tested are key to reducing the rates of late diagnosis globally.
Introduction
Modern combination antiretroviral therapy has transformed outcomes for people living with HIV, but prompt HIV diagnosis and linkage to care are critical for successful treatment. Late diagnosis (Box 1) is associated with higher mortality, morbidity, and healthcare costs compared with prompt diagnosis.1–3 Across Europe from 2010 to 2016, the incidence of AIDS and mortality in the first year after diagnosis was estimated to be nine times higher in people who were diagnosed late than in those who were diagnosed promptly. 4 Late HIV diagnosis is also associated with a higher prevalence of non-infectious comorbidities. 2 Quality of life, which has been suggested as the fourth ‘90’ to supplement the UNAIDS targets, 5 may also be negatively affected by late diagnosis, owing to pain/discomfort or depression/anxiety.6,7 On a societal level, late HIV diagnosis is associated with higher healthcare costs: total medical costs in the first year after diagnosis are approximately twice as high in patients who are diagnosed late compared with those who are diagnosed early.2,3 Additional avoidable healthcare costs are also likely incurred before an HIV diagnosis is made, but these are not well quantified. 3 Furthermore, even if a person who is diagnosed late recovers their health following treatment, they may have lost their employment and savings through ill health and therefore may require additional state support. 3 It is imperative to improve prompt diagnosis to optimise clinical outcomes, improve patient health-related quality of life, and reduce costs for the health service and wider society.
Every year, two-fifths of new HIV diagnoses in the UK are late diagnoses.
8
Nevertheless, late HIV diagnosis has been consistently lower in the UK than in most other European countries in the past decade, with numbers falling dramatically in certain regions. Here, we highlight regional trends and demographic groups who are more likely to receive a late diagnosis in the UK and we identify barriers to prompt diagnosis, including the effect of the COVID-19 pandemic. Crucially, we evaluate pilot projects and ongoing initiatives that have successfully reduced late diagnoses in the UK and could be adopted in other regions and countries facing similar challenges. The key objective of this review is to provide readers with practical advice for reducing late HIV diagnoses. With strong leadership and commitment from stakeholders, including national bodies, to test in community settings, we can work to achieve zero late HIV diagnoses. Alongside further improvements in engagement and retention in care, this will contribute to ending HIV transmission.
9
Definition of late HIV diagnosis. Late HIV diagnosis is defined as a CD4 count below 350 cells/mm³ of blood within 91 days of diagnosis, excluding individuals with evidence of recent HIV acquisition (either a negative test in the 24 months before their first positive HIV test, or the result of a Recent Infection Testing Algorithm).10,11
Methods
A UK-based expert author group – comprising HIV clinicians, pharmacists, industry professionals, and a social scientist – was assembled to reflect the collaborative networks of people working together to end the HIV epidemic in the UK. Data on HIV late diagnoses were sourced from UK public health bodies and the World Health Organization (WHO). References were identified through a targeted literature search of PubMed articles published from 01/01/2018 to 08/12/2022 using the search terms ‘HIV’, ‘late’, and ‘diagnosis’; the resulting 1073 titles were reviewed to identify all UK studies and highly relevant articles from other countries. Additional articles and grey literature were identified via internet searches and from the authors’ own libraries.
Prevalence of late HIV diagnosis in the UK and patient characteristics
Prevalence of late diagnosis in the UK
In 2021, 43% of HIV diagnoses in the UK were late.
8
The proportion of people receiving a late HIV diagnosis remained relatively steady between 2013 and 2019, ranging between 32% and 38% in England.
12
Since 2019, late HIV diagnoses have exceeded 40% in many regions of the UK (Figure 1). England has seen an increase in late diagnoses, from 41% (961/2343) in 2019 to 46% (786/1715) in 2021, although absolute numbers have fallen.
10
Scotland had an HIV outbreak among people who inject drugs (PWID) that peaked in 2015, but has since achieved a remarkable reduction in new HIV infections and late HIV diagnoses.
13
In 2019, just 27% of HIV diagnoses in Scotland were late and this fell further to 16% in 2021 (Figure 1).
13
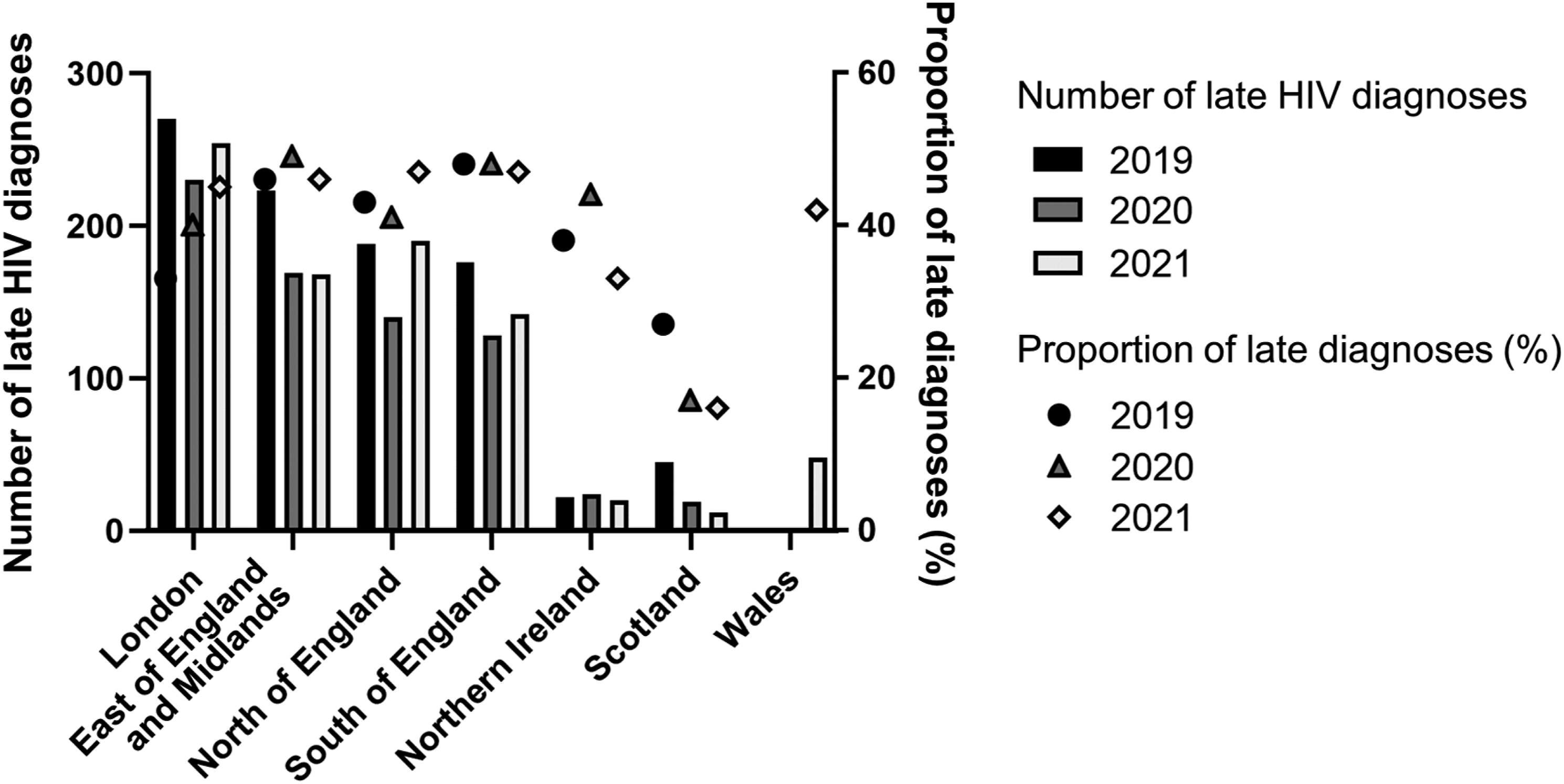
Despite this overall upward trend in the UK during the COVID-19 pandemic, the proportion of late HIV diagnoses in the UK is still lower than that reported for many other European countries, including Germany (61.1%), France (57.2%), Italy (63.2%), Spain (50.0%), Ireland (58.5%), and the overall EU/EEA average (55.5%). 8 These countries also saw an increase in late diagnosis during the COVID-19 pandemic.8,16
Risk factors for late diagnosis in the UK
Although HIV in the UK is most commonly diagnosed in men, in individuals between 25 and 49 years of age, and in persons of white ethnicity, late diagnosis was most prevalent in women (47–53%), older individuals (56–61% in those aged 50 to 64 years, and 58–73% in those aged 65 years and over), and individuals of Black African ethnicity (55–61%) in England from 2017 to 2021. 12 Demographic traits do not exist independently of one another and may compound the risk of late diagnosis.
Heterosexual contact now accounts for more new HIV diagnoses than sex between men in the UK. 12 The majority of men (54–63%) and half of women (48–51%) who were likely to have been exposed to HIV via heterosexual contact were diagnosed late. 12 For those born outside the UK, this trend may be more pronounced. Among 1256 migrant men and women who acquired HIV via heterosexual transmission, most (70–80%) acquired HIV premigration with a mean time from HIV infection to diagnosis in the UK of more than 5 years. 17 This is consistent with patterns seen among migrant populations elsewhere in Europe. 18
The impact of the COVID-19 pandemic on late HIV diagnosis
In response to the COVID-19 pandemic, the UK Government imposed a strict national lockdown from March 2020. Routine HIV screening was disrupted during this time. For example, in Northern Ireland, first-episode HIV screens decreased by approximately 70%. 19 Health-seeking behaviour changed dramatically: in England, primary care consultations dropped in 2020, only fully recovering in May 2021. 20 This may have been due to the fear of COVID-19, concerns about NHS capacity, and reduced or adapted services. Diagnosis across a range of conditions fell significantly. 20 This widespread reduction in routine testing and health-seeking behaviour (including reduced access to sexual health services) may have contributed to the increase in the proportion of late HIV diagnoses seen in 2020 and 2021. 10 Mortality also increased markedly during the COVID-19 pandemic, underlining the consequences of late diagnosis. In 2020, people who were diagnosed late in the UK were 17 times more likely to die within a year, up from six times more likely in 2019.10,12
Strategies to improve prompt diagnosis of HIV
Insufficient testing is the fundamental reason for late HIV diagnosis. Societal, systemic, and individual factors all contribute to inadequate testing. Individuals may face structural, interpersonal, and internalised discrimination based on their age, gender, ethnicity, or sexual orientation; this complex network of factors results in barriers to routine testing, health-seeking behaviour, and testing in response to clinical disease (Figure 2).
21
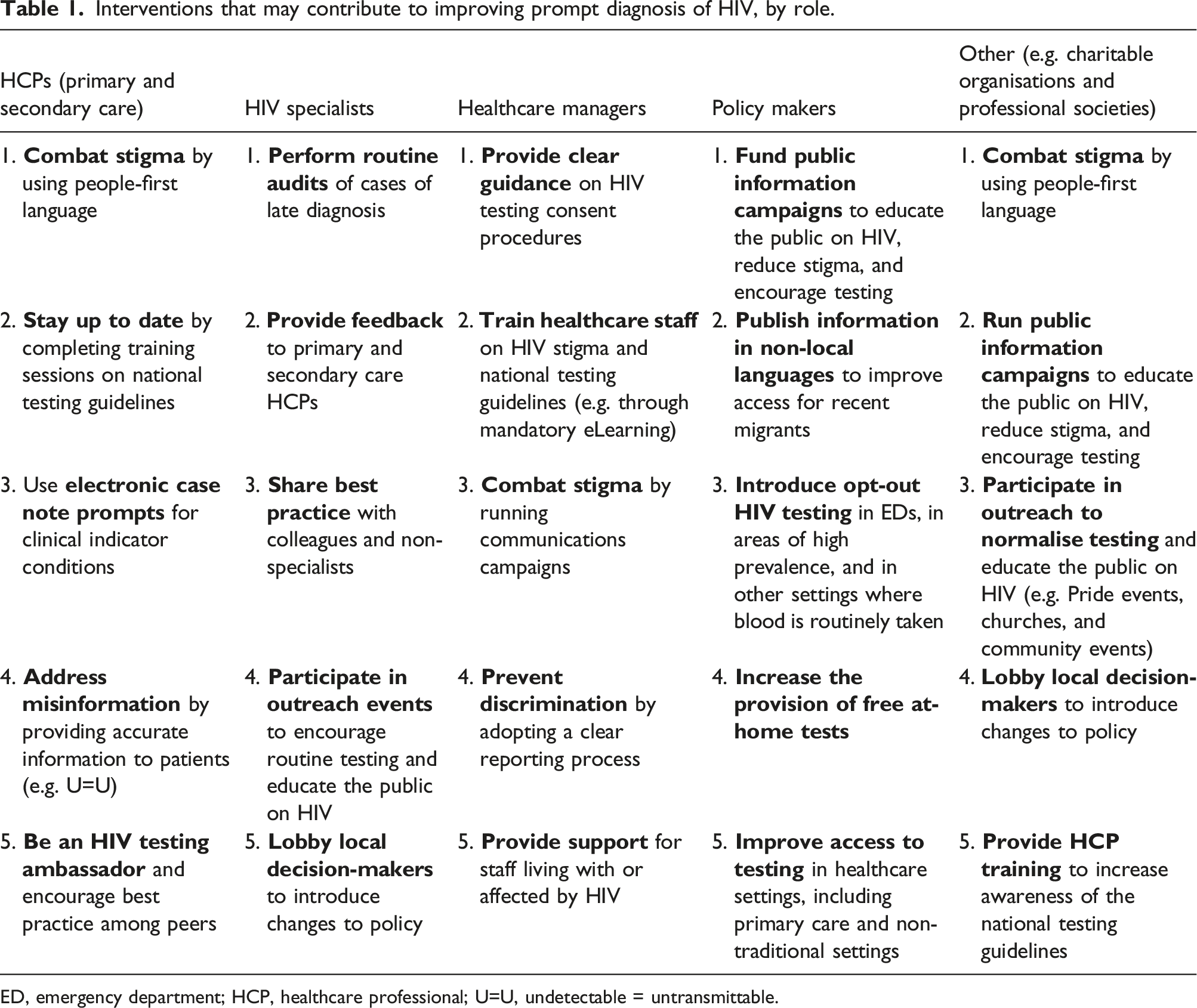
Interventions that could facilitate prompt diagnosis if more widely adopted are detailed below (Table 1). Common barriers to HIV testing and key routes to prompt diagnosis. Interventions that may contribute to improving prompt diagnosis of HIV, by role. ED, emergency department; HCP, healthcare professional; U=U, undetectable = untransmittable.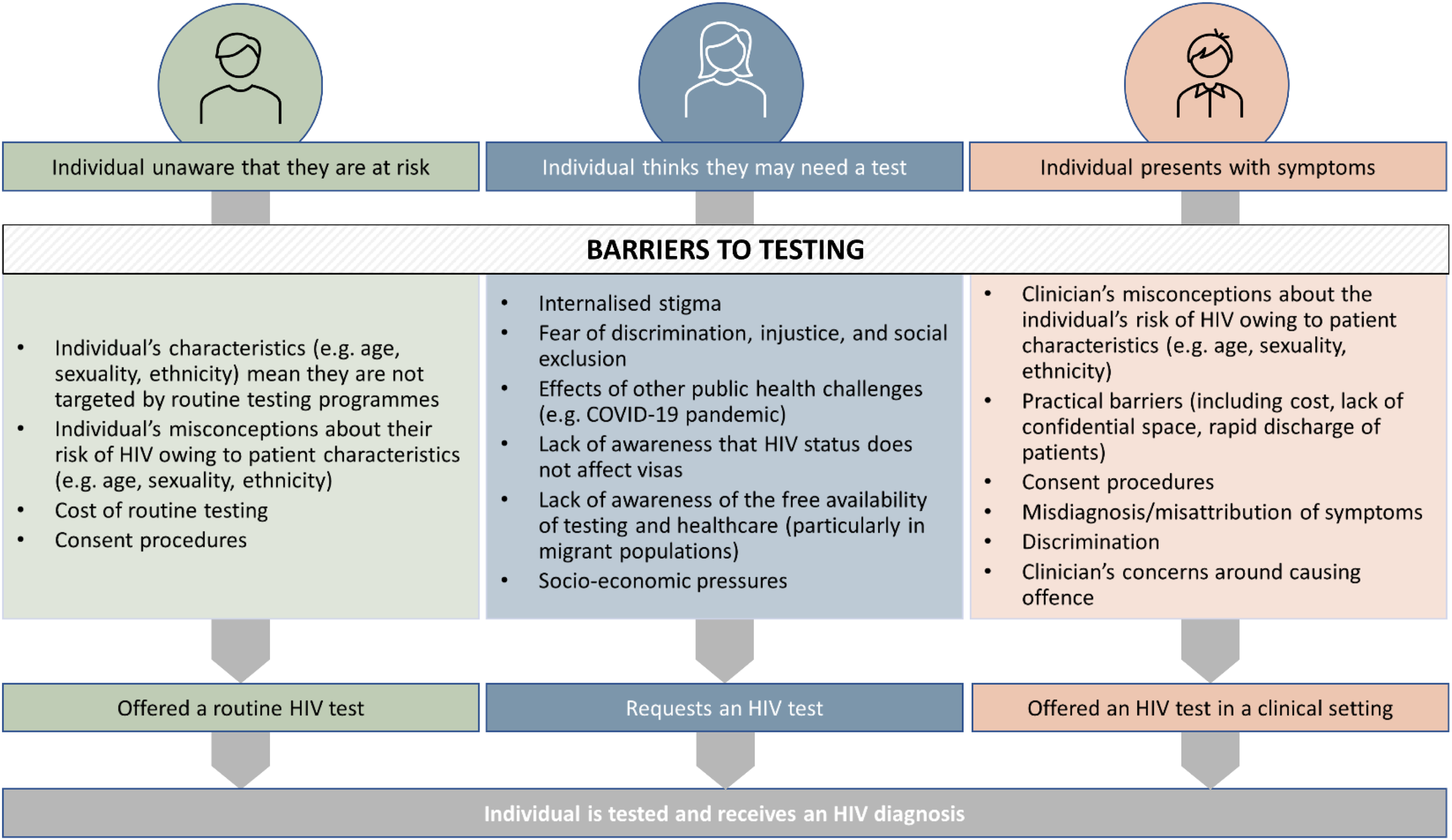
Health-seeking behaviour
Many factors may prevent someone from getting an HIV test. 22 Some people are unaware that they are at risk of HIV or have misconceptions about who is likely to be exposed to HIV. For example, older people may have the impression that HIV is only relevant to young people and may misattribute their symptoms to an already diagnosed comorbidity, and healthcare workers may have the same misconceptions. 23 Socioeconomic factors are also strong influencers of health-seeking behaviour. 24 Furthermore, anticipated stigma can deter people from getting tested. 25 Many of these barriers to health-seeking behaviour can be overcome with targeted actions.
Educate the public about HIV
Knowledge of HIV among the general population has not kept pace with clinical developments and HIV remains highly stigmatised. 26 Individuals may fear the consequences of diagnosis, 22 and so educating the public that people living with HIV – when given appropriate treatment promptly – can have a normal life expectancy, could motivate people to get tested. Targeted public information campaigns and community-specific outreach programmes can reduce the fear and misconceptions around HIV. 27 ‘Undetectable = Untransmittable (U=U)’ and ‘Can’t Pass It On’ are public health campaigns highlighting that when a person with HIV has an undetectable viral load they cannot transmit HIV through sexual contact. In a study of attitudes of the UK general public, a greater understanding of U=U was associated with lower HIV stigma. 28 This represents an opportunity to improve awareness and reduce stigma. Big, bold, community-led movements have the potential to change attitudes among the wider public and challenge outdated ideas about who is at risk of HIV. 29
Combat ignorance and stigma surrounding HIV in the healthcare setting
Proposed strategies to reduce stigma in healthcare settings include training staff about HIV stigma, running campaigns within organisations, adopting a clear reporting process for people who experience discrimination, and providing support for staff living with or affected by HIV. 29 A UK pilot of mandatory HIV stigma eLearning for healthcare staff, which included interviews with people who had experienced HIV-related stigma in healthcare, was successful and could be rolled out more widely with the support of senior leadership. 30 People-first language, language that puts the person before a diagnosis as outlined in the People First Charter, should always be used in healthcare settings to help tackle stigma.31,32 Moreover, the language employed should reflect that a person with HIV is not responsible for their late diagnosis. ‘Late presentation’ and ‘late presenter’ terminology may imply that the patient is responsible for not seeking health services, but in reality, late diagnosis is often a result of missed opportunities for testing in the clinical setting.
Increase access to testing among those born abroad
Information about healthcare entitlements should be made available in languages spoken by local migrant communities because those who move to a new country may be unfamiliar with the healthcare system. 33 For example, in the UK, information should confirm that HIV testing and treatment is free, confidential and will not impact an individual’s immigration status. Opt-out HIV testing is recommended when patients born abroad register with a primary care provider, particularly for those from countries with a high rate of HIV infection.33,34
Routine testing provision and access
Sexual health clinics (also called genitourinary medicine clinics) are traditionally the most common setting for new HIV diagnoses in the UK (accounting for 69% of all new diagnoses), followed by emergency departments (8.6%), general practice (6.4%), and antenatal services (5.5%). 35 Notably, diagnoses outside of sexual health clinics are more common among Black African and Black Caribbean individuals than among individuals of other ethnicities. 35 Those diagnosed outside of the sexual health clinic setting are more than twice as likely to be diagnosed late; 35 therefore, improving access to routine HIV testing in non-genitourinary medicine settings may reduce late diagnosis.
Universal testing in emergency departments
Universal HIV testing for pregnant women, established in the 1990s in the UK, has been highly successful and cost-effective. 35 Trials of a similar universal testing approach for blood-borne viruses in Emergency Departments (EDs) found that testing was feasible and effective.36–38 Opt-out testing has been introduced in EDs across London and other areas with high prevalence of HIV. 39 In the first 9 months of the programme (April–Dec 2022), 665,746 HIV tests were carried out and 282 new diagnoses were made.40,41 This programme is estimated to have increased HIV testing in UK EDs eight-fold between April 2022 and April 2023, making EDs the second most common healthcare setting for testing. 40 In Portugal, opt-out testing in EDs was successful in reducing late HIV diagnoses; 42 therefore, a wider rollout of this approach across the UK has the potential to significantly reduce late diagnoses. A British HIV charity, the Terrence Higgins Trust, is campaigning for the expansion of opt-out testing in EDs across England. 43 Opt-out testing when someone registers with a primary care provider, or in settings where blood is routinely taken, is also recommended in areas of high prevalence. 34
Improved access to home HIV testing
Discomfort in sexual health clinic waiting rooms is a barrier to HIV testing, which may be particularly significant for people from ethnic minority backgrounds. 44 Self-testing kits offer a discreet and convenient way to test at home. During the COVID-19 pandemic, these services became vital as many people were unable to attend in-person appointments. Across much of the UK, free home tests are available to order from services such as SH:24 and Free Testing HIV.45,46 Such services were vital during the pandemic; for example, testing via SH:24 accounted for 24% of all HIV testing in Northern Ireland in 2020.19,45 However, in some regions free online tests are only available for those most at risk of HIV infection, including MSM, women and men from Black African communities, and trans women. 46 HIV self-tests are typically £20–30, which may be a barrier for individuals who do not have access to free tests or are unaware of these services. Improving access to and awareness of free HIV home testing services could improve prompt diagnosis, and particularly support those on low incomes.
Testing in non-traditional settings
Routine HIV testing in non-traditional settings can prevent individuals from feeling targeted or judged, help to diagnose those with HIV who are not in an at-risk group, and more broadly help to tackle stigma. 47 For example, The Test@Work programme provided general health checks with optional HIV testing at UK construction worksites, resulting in a high uptake of opt-in HIV testing. 47 Other non-traditional settings may include prisons, pharmacies, termination clinics, and addiction and substance misuse services. 48 Dedicated community work can improve testing among minority groups who are not engaged with formal healthcare services; for example, the Naz Project London employed one Latin American community worker to discuss HIV and offer free HIV tests within his network, resulting in 26 new diagnoses in 18 months. 48 This strategy is particularly effective in areas with a high incidence of HIV in specific populations. Public health campaigns featuring celebrities and influencers, such as the National HIV Testing Week ‘Give HIV the finger: a finger-prick test is all it takes’ campaign organised by HIV Prevention England, are also important for raising awareness and normalising testing. 49 Expanding successful programmes and encouraging innovation are crucial to reaching underserved populations.
Targeted testing in response to a local HIV outbreak
Targeted action to improve access to HIV testing among more traditional at-risk groups still has a role in reducing late diagnoses. An expansion of testing across multiple settings in response to an ongoing HIV outbreak in Scotland increased testing from 33% to 67% among PWID. 50 This increase in targeted testing is likely to have contributed to low levels of late diagnosis reported in Scotland in recent years (Figure 1).
Testing in response to symptoms in the clinic
National testing guidelines for HIV, which list clinical indicator conditions and symptoms of primary HIV infection, were first introduced in 2008 in the UK and most recently updated in 2020.51,52 Unfortunately, knowledge of and adherence to these guidelines is not universal, contributing to inadequate testing and late diagnoses. In a local UK audit, 37% of individuals who received a late diagnosis in 2019 had a clinical indicator disease in their past medical history and, of these, 83% had a probable missed opportunity for diagnosis. 53 Poor testing rates may be influenced by providers’ misconceptions about HIV risk, operational barriers, inadequate training, or a lack of time. 54 Stigma and stereotyping by healthcare professionals about who is likely to be living with HIV may also be hindering testing. 55 Discrimination (in the form of sexism, heterosexism, transphobia, racism, xenophobia, ageism, ableism, and other prejudices) may also play a role in missed diagnoses. Health inequalities and inequity based on gender, age, and ethnicity are evident from the data on late diagnosis in the UK, 10 suggesting that these factors have a real impact on patient care.
In line with national testing guidelines, clinicians should offer a test to all patients presenting with an HIV indicator condition, regardless of demographic factors. 52 The widespread education of healthcare professionals on consent procedures for testing, or the simplification of these procedures, may also be useful. 55 Educational campaigns may be run by national bodies or in partnership with industry. For example, the ‘HIV in the UK today’ digital educational campaign targeted non-HIV healthcare professionals in primary and secondary care and directly engaged over 7700 healthcare professionals. 56
Testing in primary and secondary care
Analyses of very late diagnoses in England and Wales found that approximately 60% of missed opportunities for testing occurred in primary care and approximately 30% in secondary care.57,58 In the hospital setting, high staff turnover, competing clinical priorities, and confusion around consenting procedures may contribute to missed opportunities for diagnosis. 55 Peers encouraging best practice and the provision of up-to-date information to physicians via trusted sources (e.g. in the UK, the British HIV Association [BHIVA], British Medical Association conferences, or the Royal College of General Practitioners) are essential to improving testing. Practical interventions have also been shown to be effective: the introduction of electronic testing prompts in primary care resulted in a substantial initial increase in testing.59,60 Similarly, in a secondary care setting, healthcare professional education combined with case note prompts highlighting the presence of an indicator condition (a highly visible sticker manually inserted into clinical notes) increased HIV testing from 3% to 34%. 61 Treating missed opportunities for HIV diagnosis as a patient safety issue in cases of patient death may also facilitate individual and institutional learning. 57
Learning from local audits
Identifying the local barriers to testing, learning from cases of late diagnosis, and sharing best practice within countries and internationally are all fundamental to making structural improvements that will reduce late diagnoses.
It is vital to identify gaps relating to testing and to adopt formal, routine audit processes to learn from any cases of late diagnosis. BHIVA’s good practice position statement on recording and investigating late HIV diagnoses offers practical advice, protocols, and case studies.
3
These free resources support organisations to identify areas for improvement and communicate lessons to the relevant teams. This was successfully implemented in Greater Manchester, where improvements in late diagnosis were reported following audits in 2016 and 2019.
53
However, in a national audit in 2016, 55% of HIV services had not reviewed cases of late diagnosis, owing to there being no clearly defined process, poor access to health record data, concerns about stigma, patient embarrassment or confidentiality, and concerns about apportioning blame.
57
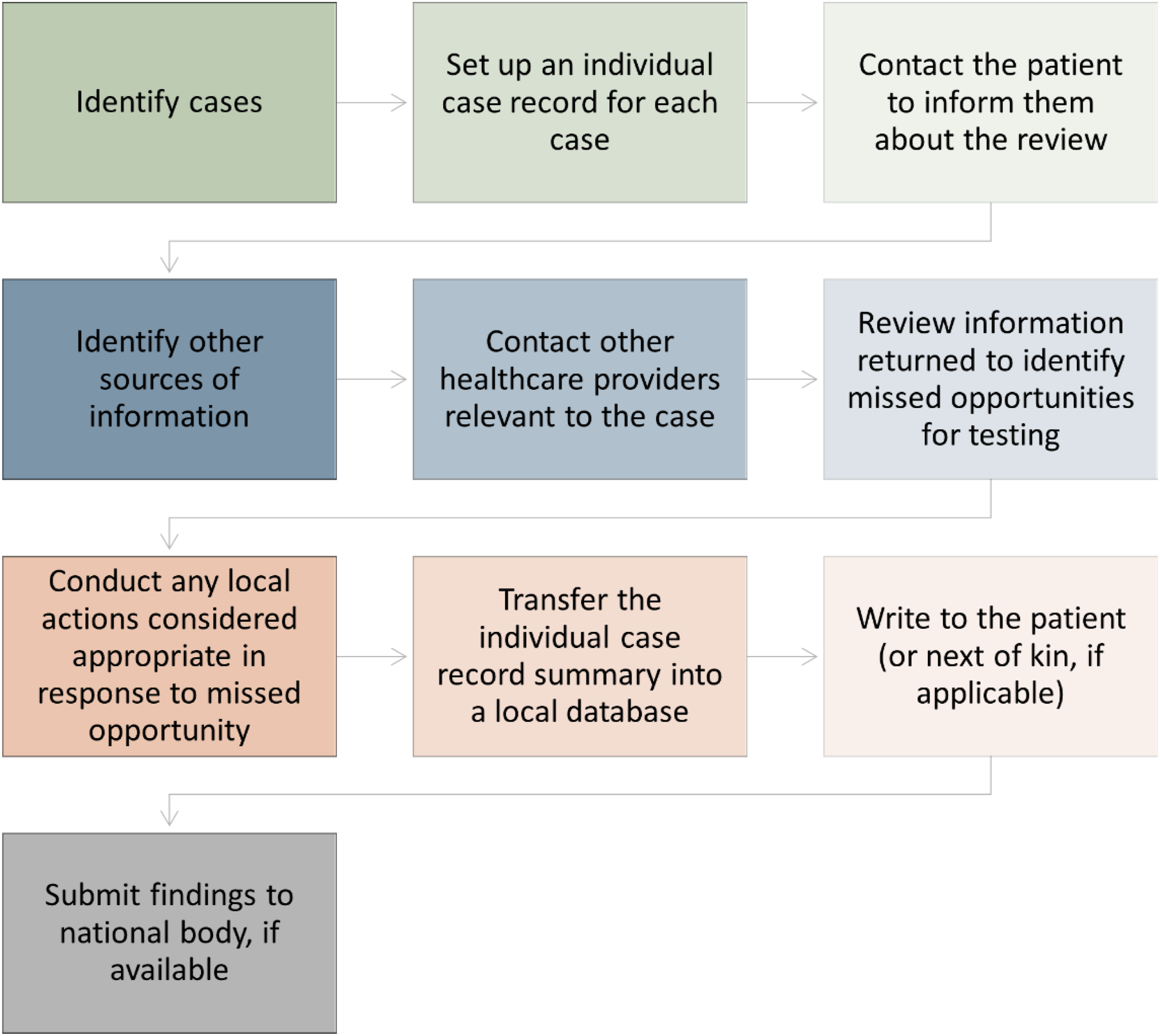
More recently, the BHIVA Late Diagnosis Review Protocol was published (Figure 3).
62
This supports units in assessing whether there were missed opportunities for testing and if the patients were harmed as a result, and whether serious incident review should be performed. The overarching aims are (1) to provide direct feedback to clinical areas that are failing to test for HIV according to national guidelines, and (2) to provide data nationally on late diagnoses and missed opportunities, which can be shared to encourage learning from these missed opportunities and improve testing. Overview of the procedure for investigating late diagnoses and potential missed opportunities for HIV testing. Adapted from the British HIV Association Late Diagnosis Review Protocol.
62
Best practice sharing and global partnerships
The limitations of this review include the focus on the UK experience as a case study, the potential for selection bias in our literature review, and the small number of UK pilot studies. There may be differences in policies (e.g. immigration rules) and healthcare provision (e.g. availability of free testing) in other countries, but even within the UK there are regional differences, and different solutions are relevant locally. Best-practice sharing, nationally and internationally, offers the opportunity to learn from others facing similar challenges. Globally, partnerships between charities, industry, and the community are contributing to innovative solutions. Fast-Track Cities is a global partnership that aims to share knowledge and experience to maximise collective efforts to end the HIV, tuberculosis, hepatitis B virus, and hepatitis C virus epidemics, and has published useful resources on the issues surrounding late HIV diagnosis. 63 RADIAN is an example of successful collaboration between a charity (the Elton John AIDS Foundation), industry, and local stakeholders, aiming to improve HIV prevention and care in Eastern Europe and Central Asia. 64 Furthermore, the industry-sponsored ‘Zeroing In: Ending the HIV Epidemic’ grant programme and ‘HIV Prevention Fund’ support organisations and community-led initiatives to end the HIV epidemic globally.65,66 Proactive sharing of successful initiatives to reduce late diagnosis will allow all countries to tackle this problem in an evidence-based way.
Conclusion
Absolute numbers of new HIV diagnoses in the UK are low and declining owing to advancements in treatment and prevention, but since the COVID-19 pandemic, 43% of new diagnoses are late and this warrants action. 8 Societal, systemic, and individual factors contribute to the insufficient testing that results in late diagnosis. By identifying the barriers in our local communities, appropriate interventions to improve testing can be implemented. HIV stigma and discrimination must continue to be addressed and lessons must be learnt from cases of late diagnosis. Recent advances include improved access to home testing and a successful pilot of opt-out testing in London EDs. Although these initiatives have been successful locally and may be helpful blueprints for others, the UK still has a long way to go on the journey to zero late HIV diagnoses. Continued sharing of best practices between countries is essential for these goals to be met globally. Ultimately, this will optimise clinical outcomes, improve patient health-related quality of life, and reduce costs for healthcare services and wider society.
Footnotes
Acknowledgements
The authors would like to thank Public Health Wales for kindly providing data on late HIV diagnosis, and Lucy Eberhard, PhD, of PharmaGenesis London, London, UK, for providing medical writing support, which has been funded by Gilead Sciences, Inc., Foster City, CA, USA, in accordance with the Good Publications Practice 2022 (GPP 2022) guidelines (![]() ).
).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Marta Boffito received travel grants, speaker and advisor fees, and research grants to the organisation from Janssen, Roche, ViiV, Bristol Myers Squibb, Merck Sharp & Dohme, Gilead Sciences, Mylan, Cipla, Novavax, Valneva, GSK, Atea, Moderna, Teva, AZ and Pfizer. David R Chadwick is a trustee/executive member of the British HIV Association and has recently chaired a sub-group on guidelines for Emergency Department opt-out testing using implied consent. He has also received research grants from Gilead Sciences. Samuel Kabagambe is an employee of Gilead Sciences and may own stock options in Gilead Sciences. Bakita Kasadha has previously received speaker honoraria and consultancy fees from Gilead Sciences. Cindy Elliott is an employee of Gilead Sciences and may own stock options in Gilead Sciences. Emily Boardman, and Emily Cheserem declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Gilead Sciences, Inc., Foster City, CA, USA, provided funding for medical writing support.
