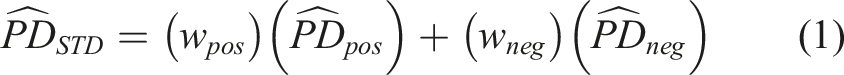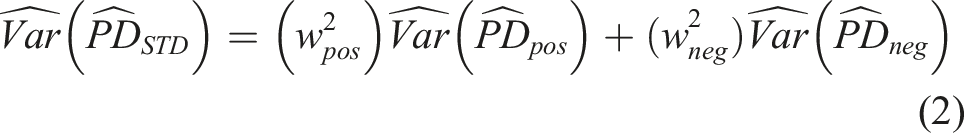Abstract
Background
To meet global targets for the elimination of mother-to-child HIV transmission, tailored approaches to HIV testing strategies need prioritizing. Herein, we sought to identify individual-level factors associated with male partner HIV testing.
Methods
We conducted a secondary analysis of data from two parallel randomized trials of pregnant women living with HIV and those HIV-negative in Lusaka, Zambia. Across both trials, control groups received partner notification services only, while intervention groups received partner notification services plus HIV self-test kits for their partners. Associations between baseline factors and male partner testing were estimated using a probability difference. The outcome of interest was uptake of male partner HIV testing of any kind within 30 days of randomization.
Results
The parent study enrolled 326 participants. Among the 151 women in the control groups, no clear associations were noted between maternal or male partner characteristics and reported uptake of male partner HIV testing. There were positive trends favouring partner testing among women who completed primary school education, had larger households (>2 members), and whose partners were circumcised. Likewise, no clear predictors of male partner testing were identified among the 149 women in the intervention groups. However, negative trends favouring no testing were noted among older, multiparous women from larger households.
Conclusion
No consistent predictors for male partner HIV testing across two compared strategies were observed. Our findings suggest that differentiated strategies for male partner HIV testing may not be necessary. Instead, consideration should be given to universal approaches when bringing such services to scale.
Introduction
Across the globe, significant progress has been made in prevention of mother-to-child HIV transmission (PMTCT). 1 Almost 2 million potential HIV infections in children were averted from 2010 to 2020, largely due to the increased availability of antiretroviral therapy (ART) to both pregnant and breastfeeding women. 2 To meet the ambitious global targets for the elimination of MTCT, tailored approaches for PMTCT need to be prioritized. 3 Engaging male partners during antenatal and postnatal care (ANC & PNC) is critical for optimizing PMTCT services and improving infant health outcomes.4–6 However, uptake of male partner HIV testing at ANC clinics remains low in many sub-Sahara African settings. 7 Several factors contribute to this trend, including: the societal perception that ANC is solely a woman’s obligation; that men should not participate in ANC/PMTCT services and ridiculing those that participate; the reluctance of men to learn their HIV status and their misconception that their partner’s HIV status was a proxy of their own; being unaware of ANC/PMTCT services; time conflicts with ANC/PMTCT; health system limitations, such as long waiting times at the clinics; and the male unfriendliness of ANC/PMTCT services. 8
Several evidence‐based strategies, including partner notification services, 9 home-based HIV testing, 10 and secondary distribution of HIV self-test kits, 11 have increased uptake of male partner HIV testing in antenatal settings across sub-Saharan Africa. However, uptake under these single-modality strategies has largely fallen short of the ambitious HIV testing targets set by the global HIV and AIDS community as they may not be enough to broaden coverage, enhance linkages and meet the HIV testing targets by themselves. 12 Further, several known single-modality strategies such as partner notification, HIV self-testing, and home-based HIV testing have been associated with moderate increases in male partner HIV testing. 12 Combination approaches that integrate two or more evidence-based strategies, may enhance uptake of male partner testing in antenatal settings,12,13 but are considerably more complex and resource intensive than single-modality strategies.
We conducted randomized trials that compared male partner HIV testing utilizing two strategies: partner notification services (control) versus partner notification services and HIV self-testing (intervention). Pregnant women living with HIV-positive and those HIV-negative were enrolled in parallel trials. In both populations, we found that male partners of women in the intervention group were more likely to undergo HIV testing of any kind, when compared to male partners in the control arm. 12 In this secondary report, we sought to identify factors associated with male partner HIV testing with each of these strategies for partner testing. Characterizing which groups are more likely to undergo male partner HIV testing can help to inform implementation of ANC/PMTCT services, potentially paving the way for differentiated approaches in service delivery. This was not the aim for the parent trials.
Methods
Trial and setting
We enrolled pregnant women aged 18 years or older whose male partners had not tested for HIV during the index pregnancy. The parent trials investigated a combination approach to increase HIV testing in male partners of pregnant women living with HIV and those HIV-negative. The trials were designed to address gaps in male partner HIV testing. In this secondary report, we sought to identify factors associated with male partner HIV testing with each of these strategies for partner testing. Characterizing which groups are more likely to undergo male partner HIV testing can help to inform implementation of ANC/PMTCT services, potentially paving the way for differentiated approaches in service delivery. The participants were recruited from the antenatal clinic at the Chipata First-Level Hospital in Lusaka, Zambia. The study team worked with community partners and hospital staff at the study site to provide information concerning the study and facilitate recruitment. At the time of being enrolled, the participants responded to questions on their sociodemographic characteristics, obstetrical history, and sexual health besides providing information about individual and primary male partner HIV testing history, current HIV treatment and prevention measures. Trial 1 enrolled pregnant women living with HIV while Trial 2 enrolled those documented as HIV-negative within the past 3 months during the current pregnancy. 12 Within each trial, participants were randomly assigned 1:1 to either a control or intervention arm. All participants received partner notification services as part of standard care for pregnant women living with HIV. In addition to partner notification services, those randomized to the intervention arm received counseling on the use of oral HIV self-test kits and were given up to five oral HIV self-test kits to give to male partners. Participants were asked to return for a follow-up visit approximately 30 days after enrollment. At this visit, they responded to questions about their primary partner’s HIV testing history, including the date, venue, and modality of their partner’s most recent HIV test. Methods for the parent trials have been described in further detail elsewhere. 12 Informed written consent was obtained from all the study participants upon informing them of the study procedures. The primary outcome was reported male partner testing at a health facility within 30 days after the randomization. Ethics committees at the University of Zambia and the University of North Carolina (Chapel Hill, NC, USA) approved the study protocol. The parent trial was registered at clinicaltrials.gov (NCT04124536).
Measures
The outcome of interest was uptake of male partner HIV testing of any kind, either at the health facility or via HIV self-test kit, within 30 days of randomization ascertained by female participant’s reports of HIV testing by the male partners. 12 Baseline predictor variables were selected prior to conducting association analyses.
Statistical methods
Analyses were conducted using an intention-to-treat approach, with women analyzed according to the arm they were randomly assigned to regardless of their subsequent distribution of HIV self-test (HIVST) kits to their male primary partner. Probability differences (PDs) were estimated with corresponding 95% Wald confidence intervals (CIs) to evaluate associations between baseline characteristics and uptake of male partner HIV testing within 30 days of entry. We conducted analyses within each study arm (intervention, control) because a strong effect of the intervention was observed in the parent study and the partner HIV testing modalities differed between the intervention and control groups. Our main analyses were pooled across HIV serostatus following a status-neutral approach. To help ensure external validity, direct standardization was used to construct pooled PDs because the study sample was not a random sample with respect to HIV serostatus (HIV prevalence in the antenatal clinic was 16% around the time of study recruitment, whereas 33% of study participants were living with HIV). A weighted average of serostatus-specific estimates was constructed using the following formulas:
Sensitivity analyses were also conducted separately by trial (i.e., HIV serostatus) to ensure that male partner HIV testing tendencies by female HIV serostatus were not obscured by pooling of data. For analyses performed separately by trial, bivariate linear-binomial regression models were used to estimate associations between baseline factors and HIV testing in the primary male partner. Point estimates were expressed as percentages and displayed graphically. Inferential tests were two-sided with a type 1 error level of 0.05 with no adjustment for multiple testing. All analyses were complete case, i.e., missing data were excluded, and conducted using SAS version 9.4 (SAS Institute, Cary, NC).
Results
Participant characteristics
Baseline characteristics of the study participants.
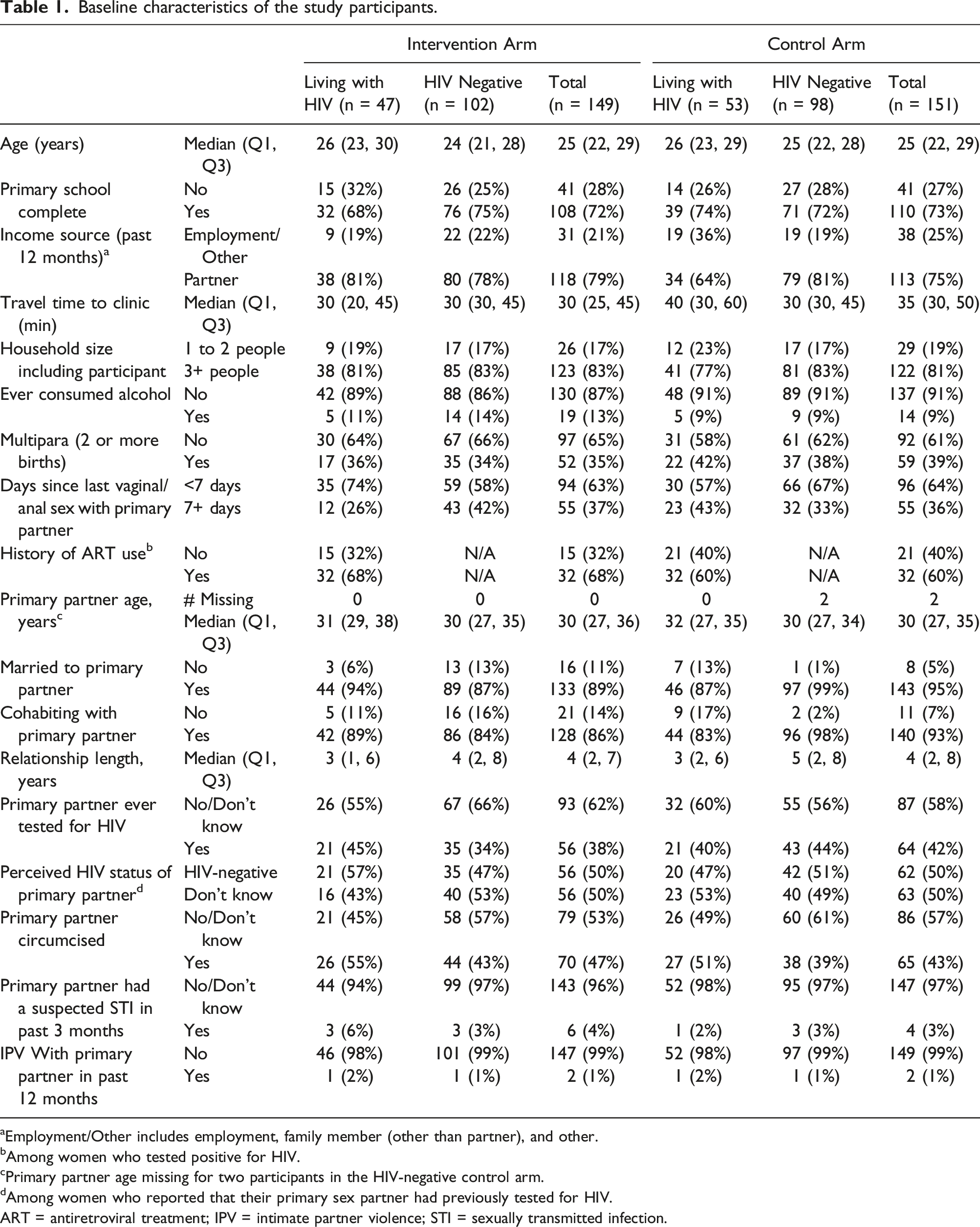
aEmployment/Other includes employment, family member (other than partner), and other.
bAmong women who tested positive for HIV.
cPrimary partner age missing for two participants in the HIV-negative control arm.
dAmong women who reported that their primary sex partner had previously tested for HIV.
ART = antiretroviral treatment; IPV = intimate partner violence; STI = sexually transmitted infection.
Partner notification services only (control groups)
Across the two trials, 151 women were randomized to the control groups, and there was no clear evidence of associations between the factors examined and male partner HIV testing as the 95% CIs covered the null (Figure 1). Nevertheless, the analysis pooled over serostatus suggested that male partner testing was an estimated 11 percentage points higher for women who lived in a household of 3 or more members compared to a 1 or 2 member household (PD: 11.2%, 95% CI: −11.0, 33.4%). Additionally, male partner testing was 10 percentage points higher among men who had been circumcised compared to uncircumcised men (PD: 10.2%, 95% CI: −7.1, 27.6%) and 11 percentage points higher if the female partner had completed primary school education compared to less than primary school (PD: 11.2%, 95% CI: −7.8, 30.2%). Baseline characteristics and association with male partner HIV testing: control arm, pooled across HIV serostatus.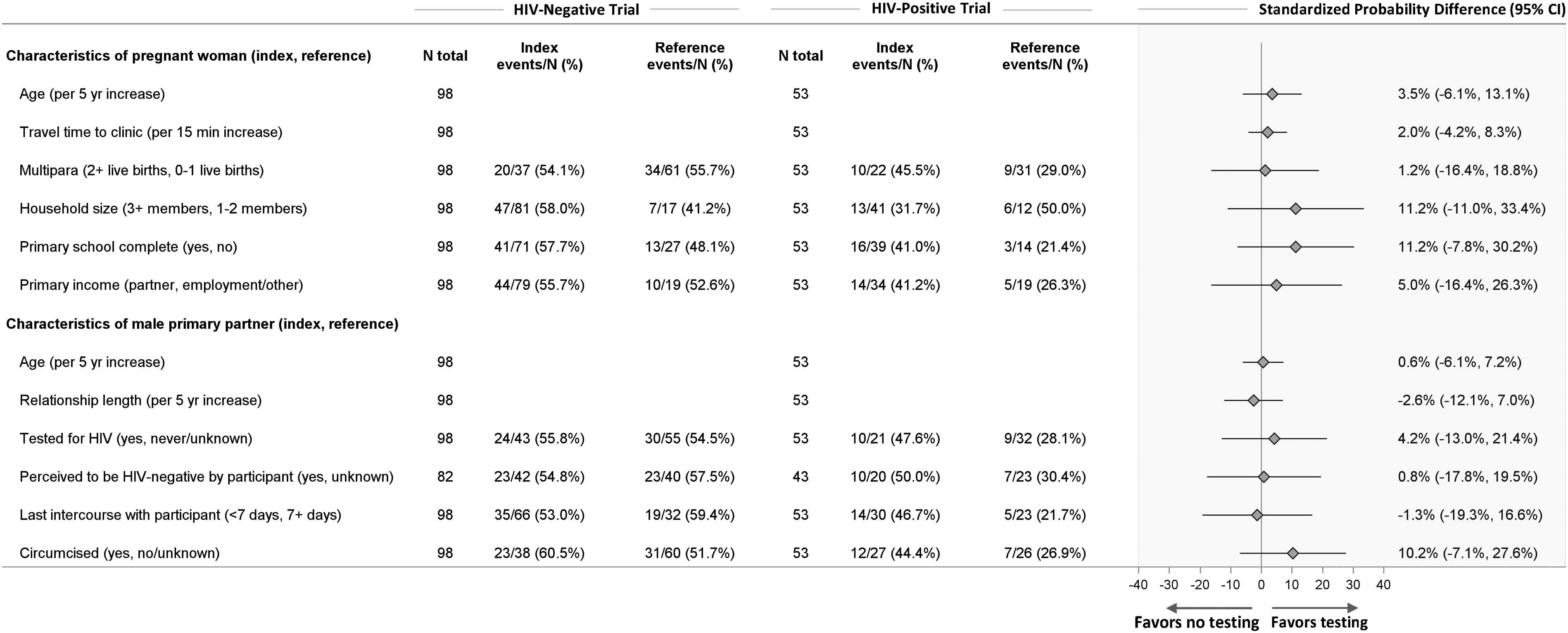
Partner notification services plus HIV self-testing (intervention groups)
Across the two trials, 149 women were randomized to the intervention groups. Estimates of association were imprecise and 95% CIs covered the null (Figure 2). Trends were largely consistent with those observed among women who received partner notification services only, with three exceptions. Among women in the intervention groups, male partner testing was an estimated 12 percentage points lower among women living in a household with 3 or more members, compared to a 1- or 2-member household (PD: −12.3%, 95% CI: −27.8%, 3.1%). Partner testing was also numerically lower among multiparous women compared to nulliparous or primiparous women (PD: −8.3%, 95% CI: 23.4%, 6.8%) and lower with each 5-years increase in the woman’s age (PD: −3.8%, 95% CI: −10.6%, 3.1%). For these variables, opposite trends were observed among women randomized to the control groups. Baseline characteristics and association with male partner HIV testing: intervention arm, pooled across HIV serostatus.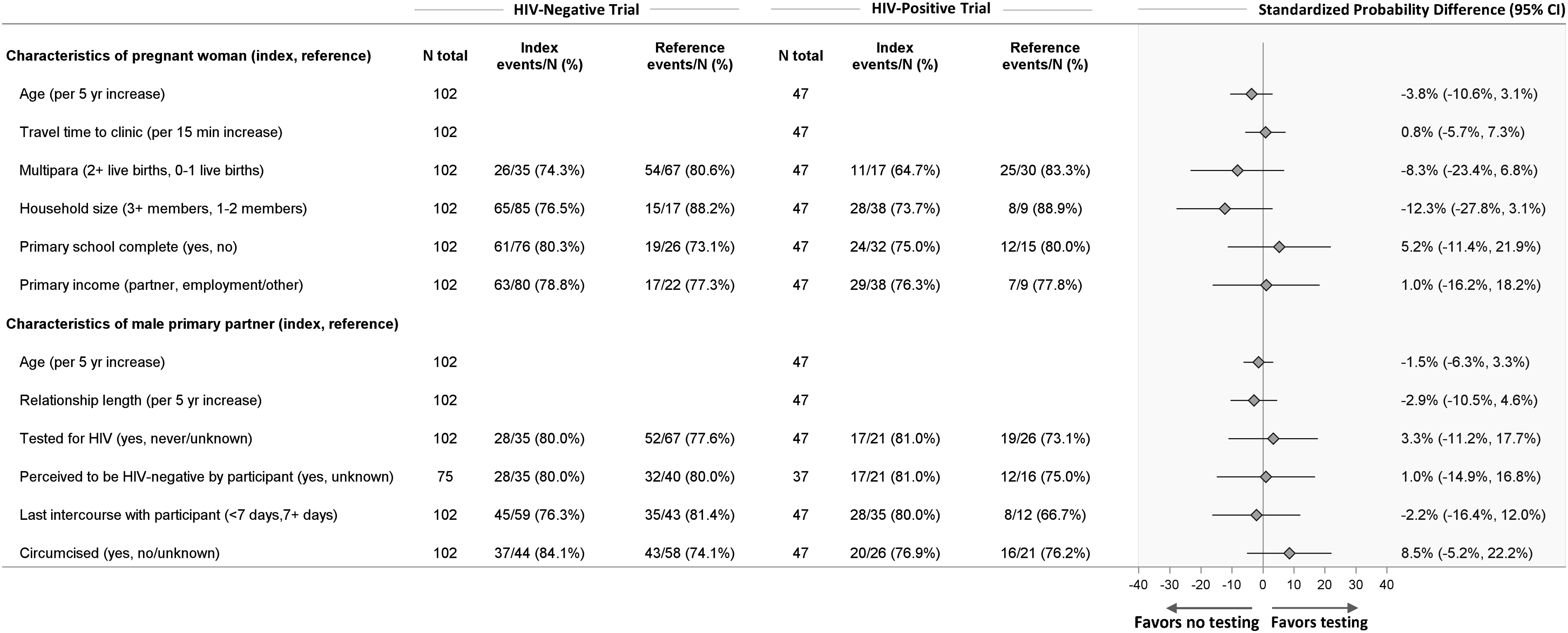
Sensitivity analyses by HIV serostatus
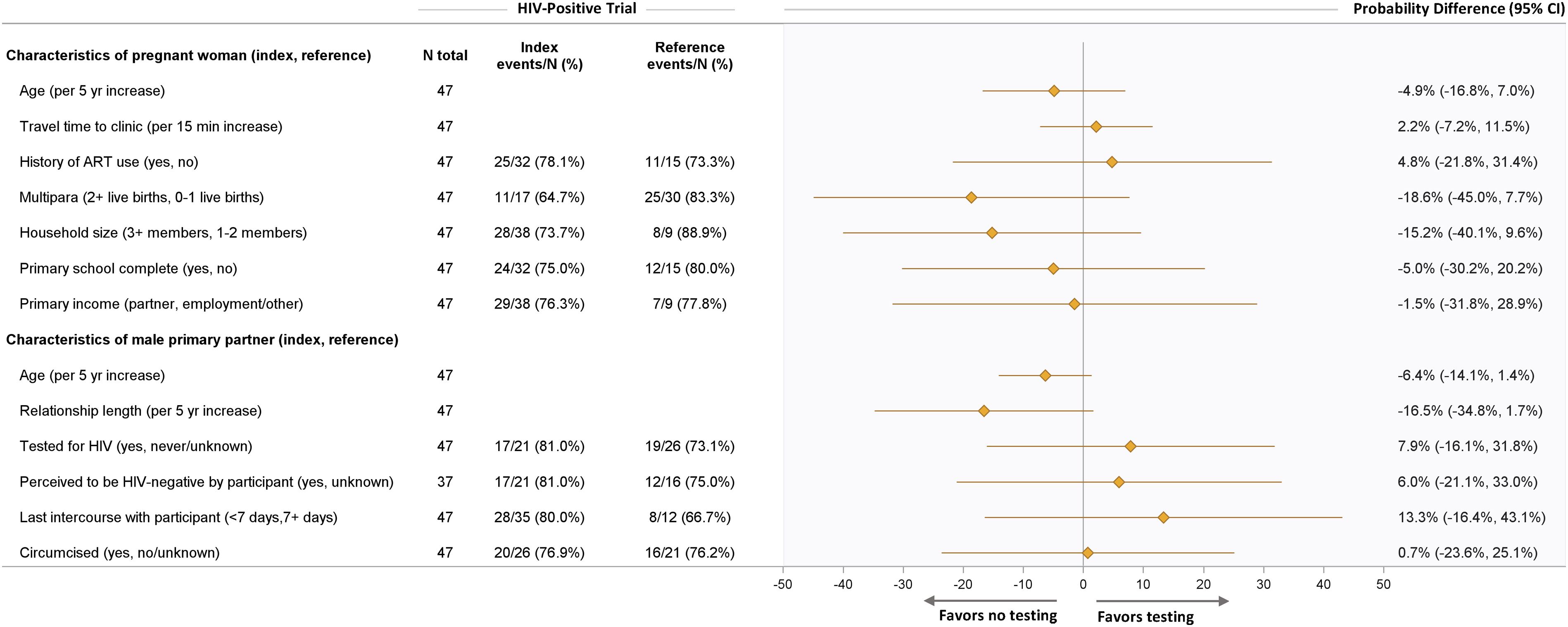
Within pregnant women living with HIV and those HIV-negative separately, we observed associations that were not present in the pooled analyses, but 95% CIs were wide (i.e., lacking precision). For example, among women living with HIV and in the control groups (Figure 3), male partners who engaged in intercourse with the female partner within the last 7 days had a 25-percentage point increase in male partner HIV testing compared to those who did not (PD: 24.9%, 95% CI: 0.4, 49.5%). Regarding years of age, for women living with HIV in the control groups (Figure 3), male partner HIV testing was an estimated 11 percentage points higher among men who were older (PD: 10.9% per 5-years increase, 95% CI: 0.9, 21.0%) and nearly 13 percentage points higher for female participants who were older (PD: 12.6% per 5 years increase, 95% CI: 0, 25.1%). However, among women in the intervention groups, these male partner testing tendencies reversed (i.e., decreased testing trends) when advancing age was considered (Figure 4). There was no evidence of associations between male partner HIV testing and the other covariates examined without pooling (Figures 3 and 4 and supplemental material S1 and S2). Baseline characteristics and association with male partner HIV testing: HIV-positive control arm. Baseline characteristics and association with male partner HIV testing: HIV-positive intervention arm.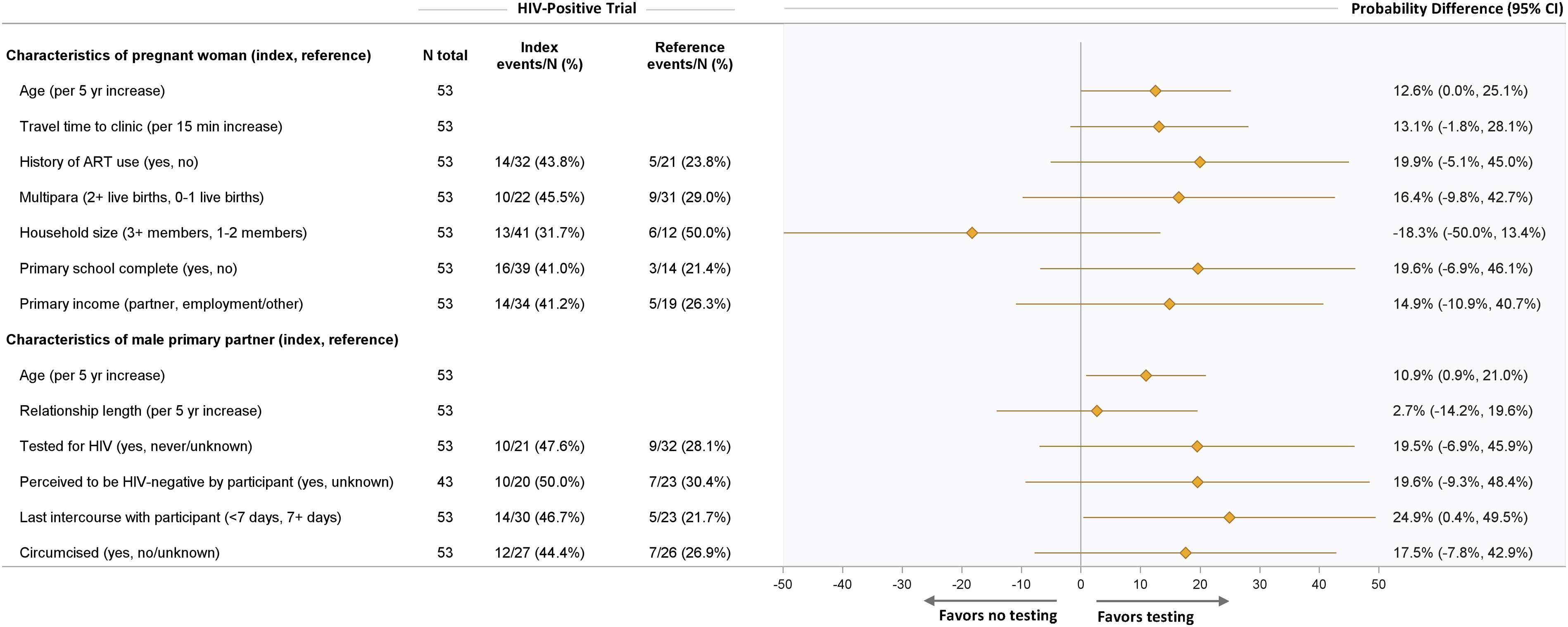
Discussion
From a pair of randomized trials among pregnant women in Zambia, we sought to investigate associations between individual-level factors and male partner HIV testing in two models of engagement: partner notification only (control) and partner notification plus HIV self-testing (intervention). We reasoned that, with this information, programs could better target pregnant women to increase the likelihood of male partner HIV testing in public health settings. However, no such associations were discovered in the main analyses that pooled across serostatus or the sensitivity analyses that stratified by serostatus.
In our parent trial, we compared two different strategies to increase male partner HIV testing. When combined with partner notification services, we found that the addition of HIV self-testing via secondary distribution resulted in higher rates of male partner HIV testing of any kind compared to partner notification alone (77% vs. 36%, in Trial 1 and 78% vs 55% in Trial 2). In the analysis that pooled across serostatus, no individual or partner characteristics were associated with uptake of male partner HIV testing among women. This was separately observed among women who received PNS alone and among women who received the partner notification services with HIV self-testing. While we noted a few trends (e.g., household size, male partner circumcision status), these were not consistent across the randomization arms.
Other studies have shown associations between male partner HIV testing and various factors, including the woman’s educational level,14–21 index pregnant woman’s or male partner’s age,14–17,19,22 and parity.23,24 Male partner circumcision has also been associated with male partner HIV testing. 25 In this regard, our findings were not consistent with previous studies and may have been a function of the interventions themselves or aspects of limited statistical power.
The overarching goal of this analysis was to identify subpopulations of pregnant women whose male partners would be likely to test for HIV. When resources are limited, such approaches can aid in program implementation and expansion and lead to greater efficiency in intervention uptake. Our findings suggest that such targeted approaches may not be feasible for either of these interventions. Instead, these data suggest that universal approaches may be needed when bringing either strategy to scale. In this regard, the parent study provides important data for such “status neutral” strategies, 26 by providing data about male partner HIV testing regardless of the index pregnant woman’s HIV status.
We acknowledge several limitations in the study. First, the trial enrolled pregnant women and relied on their report about male partner characteristics and HIV testing behaviours. This approach simplified the data collection process, but may have induced measurement error (e.g., reporting and social desirability biases).
It is possible that social desirability bias could lead to overestimation of HIV testing and underestimation of positive HIV test results with HIV self-testing. 27 In addition, some male partners could have an urge to hide their HIV status possibly for fear of being blamed for and exposing their sexual infidelity.28,29 Second, the sample size was small and this was reflected in the imprecision of the probability difference estimates. Precision was improved by pooling data from the two trials. Finally, these data were collected from a rigorously conducted clinical trial, which may limit the external validity of our results. While we believe our findings to be informative, additional observational studies that focus on “real world” populations would certainly enrich our understanding of how to target resources for improving the uptake of male partner HIV testing.
In summary, when we examined two different strategies for male partner HIV testing and we identified few—if any—strong predictors for our outcome of interest. While the combination strategy of partner notification plus HIV self-testing appears promising, it remains difficult to predict which male partners will (or will not) successfully undergo HIV testing and linkage to care. This is against reported high acceptability of HIV self-testing including kits provided through women in antenatal care from some qualitative studies.30,31 However, another area that might need further exploration is a focus on relationship factors. For instance, one study showed that the presence of joy, trust, open communication and pride in a relationship could have an impact on partner HIV testing. 32 Overall, our findings suggest that differentiated strategies for male partner HIV testing may not be necessary. Instead, consideration should be given to universal approaches when bringing such services to scale.
Supplemental Material
Supplemental Material - Two strategies for partner notification and partner HIV self-testing reveal no evident predictors of male partner HIV testing in antenatal settings: A secondary analysis
Supplemental Material for Two strategies for partner notification and partner HIV self-testing reveal no evident predictors of male partner HIV testing in antenatal settings: A secondary analysis by Andrew Kumwenda, Ann Marie Kathryn Weideman, Lauren Aiko Graybill, Matthew Kevin Dinwiddie, Kellie Freeborn, Mildred M Lusaka, Rose Lungu, Wilbroad Mutale, Nora E Rosenberg, Margaret Kasaro, Katie R. Mollan and Benjamin H Chi in International Journal of STD & AIDS.
Footnotes
Author contributions
AK, LAG, KRM and BHC led the conceptualization of the manuscript. AK, BHC, and AMKW wrote the original manuscript draft. AMKW, KRM, and MKD conducted the formal analyses. All authors critically reviewed the draft manuscript, participated in study interpretation, and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The primary study was funded by the National Institute of Allergy and Infectious Disease (NIAID) through award R01 AI131060. Additional investigator, trainee, and administrative support was provided by NIAID (K24 AI120796, P30 AI050410), National Institute of Mental Health (R00 MH104154), National Center for Advancing Translational Sciences (UL1TR002489), and the Fogarty International Center (D43 TW009340, D43 TW010558).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.