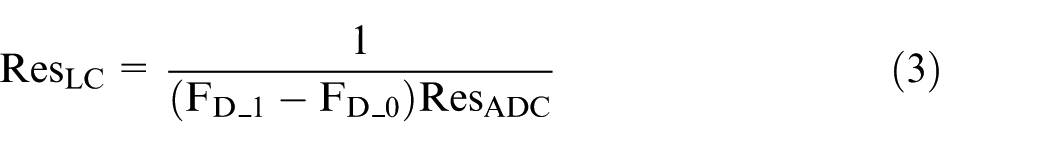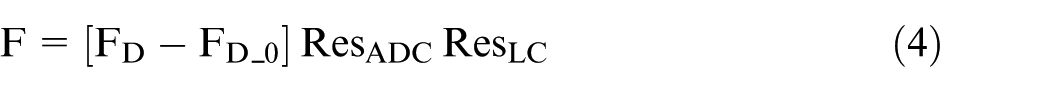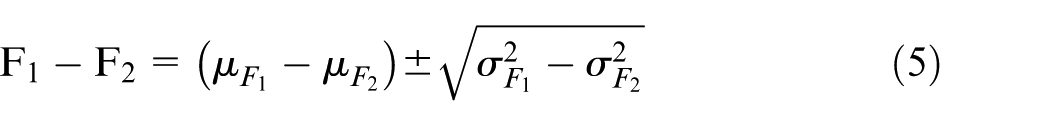Abstract
Procedural-related musculoskeletal pain is common among orthopaedic surgeons, often caused by the repetitive use of high-force bone-cutting tools. Ultrasonic cutting devices, which can operate with lower force, may help reduce this physical burden. In this study, three practising orthopaedic surgeons each performed two cuts on three fresh cortical bone samples, harvested from excised femoral necks from three patients undergoing hip replacement surgery. The study was conducted using an ultrasonic cutting device in a controlled yet clinically reflective environment. A novel setup captured real-time data on surgeon-related parameters, including vertical cutting force and vertical and horizontal cutting speed. Consistent with previous research, we confirmed that ultrasonic devices enable low force cutting (average 1.91 N). However, our findings revealed significant variability in how each surgeon interacted with the device – including how much force each surgeon applied, and how the device was manoeuvred which can influence device performance, thermal effects, and overall clinical outcomes. Given the critical importance of surgeon-related factors, our results highlight the need to understand how each surgeon interacts with these devices differently. This insight can inform training and device optimisation strategies; help translate bench testing results into effective clinical use and ultimately improve surgical performance and patient outcomes. Additionally, our findings support the potential benefits of integrating ultrasonic devices with robotic platforms to maintain consistent cutting parameters. Future research should investigate optimal cutting parameters, evaluate different blade profiles, assess result generalisability and compare outcomes before and after training or system enhancements.
Keywords
Introduction
Orthopaedic surgeons face many occupational hazards during their practice, including procedural-related musculoskeletal pain and hand arm vibrating syndrome, 1 which is a known risk factor for developing carpal tunnel syndrome. 2 A recent survey by McQuivey et al. demonstrated high prevalence of procedural-related musculoskeletal pain among arthroplasty surgeons, 3 which may occur due to repetitive use of bone saws during surgery. Ultrasonic bone surgery devices were first commercialised for oral and maxillofacial surgical procedures to perform osteotomies,4–21 then were later extended to neurosurgical22–26 and orthopaedic27–30 surgeries. These devices cut bone tissue with low force, which helps to reduce the physical burden on surgeons during surgery and also reduces their vibration exposure as ultrasonic vibrational amplitudes are insignificant compared to those produced by a mechanical saw. 31 Compared to other surgical instruments, ultrasonic devices boast many advantages that have the potential to accelerate post-operative bone healing, including reduced cutting force, 32 increased precision,26,33 controlled blood loss23,24 and reduced collateral damage to surrounding tissues.23,26 Furthermore, while surgical robotic technology has not yet reached a level where completely replacing surgeons is feasible,34,35 many common orthopaedic procedures such as total hip arthroplasty (THA) and total knee arthroplasty (TKA) are increasingly being performed with robotic assistance.36,37
However, as ultrasonic surgical devices largely rely on making multiple small, shallow cuts, 38 which enables precise cuts in small bones, this limits their applications to orthopaedic large bone surgeries where large cuts are required. There are many measurable and controllable parameters that can affect device performance and clinically relevant outcomes, which can be largely categorised as surgical device related, patient related or surgeon related. Device related parameters include ultrasonic transducer design and blade design (including blade material, vibration frequency and amplitude, and cutting-edge geometry). Patient related parameters include bone density, bone type, bone orientation and state of the bone, including hydration or moisture content and temperature. Finally, surgeon related parameters include cutting force, cutting speed, length of cut, angle of approach, the time that the device is in contact with the bone sample being cut and number of passes of the surgical device on the bone sample.
While numerous in vitro studies have investigated the effect of ultrasonic cutting on bone stress (to minimise bone mechanical damage),39–45 reduce cutting temperature46–52 and improve device performance, including blade design,53,54 device design, 55 transducer design,56–59 and cut parameters,60,61 these studies have been conducted in highly controlled laboratory environments using mechanical test rigs, and with most studies using non-human bone (bovine,46,48,50,62,63 porcine,43,45,49,61 synthetic 64 or specific bone not mentioned 60 ). Alternatively, in silico modelling has been used where input cutting parameters (normally surgeon related parameters) and the cutting sample are idealised.38–42,47,51 While highly controlled lab-based experiments are critical to understand how to optimise surgical device operation, they do not reveal how different surgeons interact with these devices under the complex and often unpredictable conditions encountered in clinical practice. Understanding how surgeons use and interact with ultrasonic surgical devices can provide insights that help translate bench testing results into clinical use.
In this study, we present a novel experimental setup, and demonstrate feasibility of the setup, that allows practicing orthopaedic surgeons to perform cuts on fresh human cortical bone using an ultrasonic surgical device within a controlled yet clinically realistic environment. The setup can capture surgeon related parameters, including applied cutting force, time-stamped tool trajectory from which the horizontal and vertical components of cutting speed can be calculated, and bone penetration rate. Our results represent an approach that facilitates detailed analysis of surgeon–device interaction to inform future strategies for training and optimising device usage, using information gained from controlled lab tests. The ultimate aim is to improve surgical performance and patient clinical outcomes.
Methodology
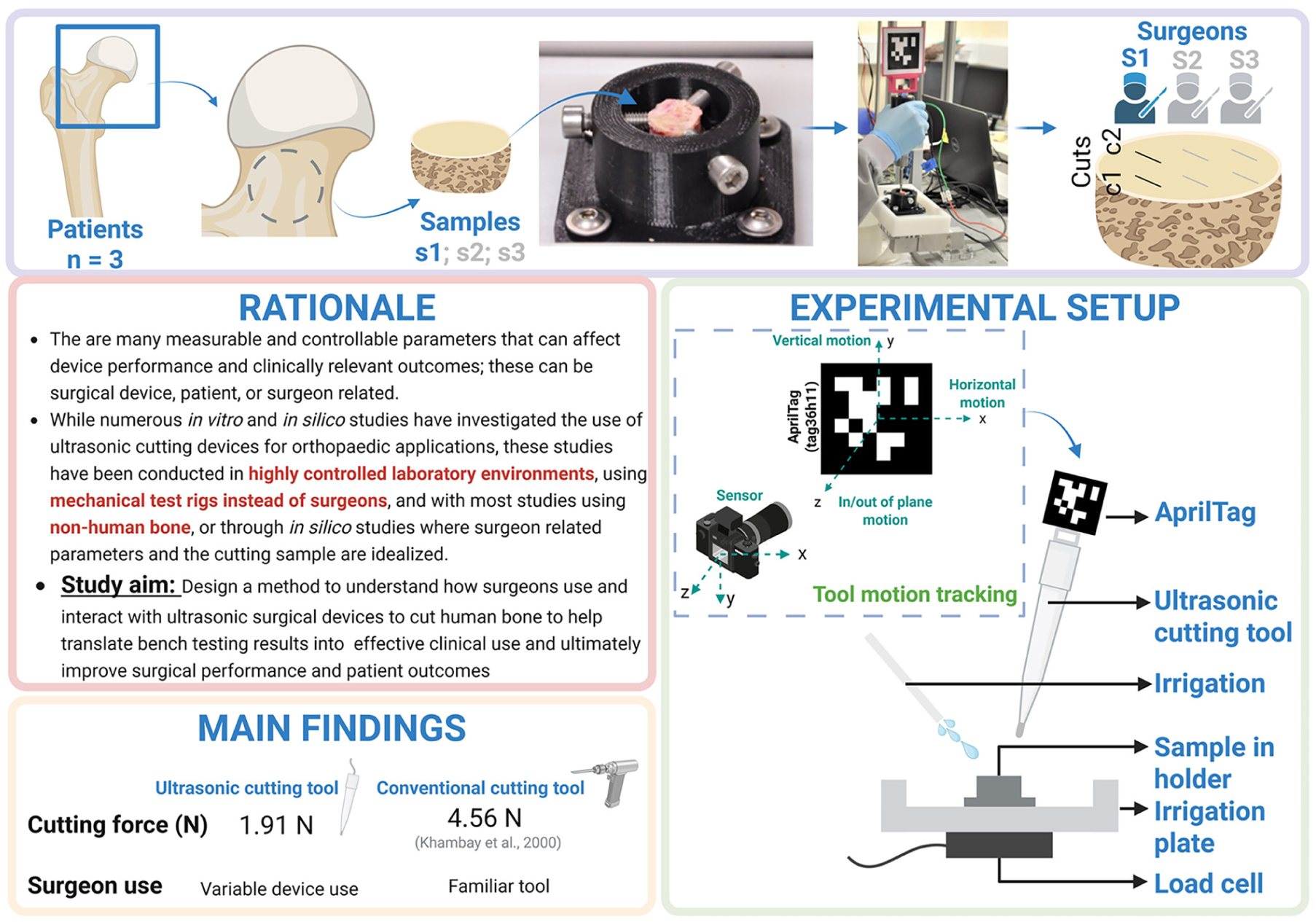
We set up a robot operating system (ROS) coding suite 65 to record and synchronise time stamped surgical device position and vertical force from the Arduino microcontroller.
To record the cutting force, a custom designed test clamp was connected to an irrigation plate, which was mounted on a 6-wire single point load cell (1000 N capacity) and the assembly was fixed to a workbench. To measure force, the load cell was connected to an Arduino Due microcontroller for processing. To track the surgical device position, an AprilTag (tag36h11) was mounted onto the ultrasonic surgical device using a custom 3D printed tag holder66–68 (Figure 1).

Experimental setup showing orthopaedic surgeon cutting a human cortical bone sample using a custom ultrasonic tool, tracked using an AprilTag.
To visualise the tag, a SpotLight Pro Webcam with LED lights (Trust International B.V.) was used in conjunction with the usb_cam (a ROS driver for V4L USB Cameras), 69 which had been calibrated using the Kalibr Calibration toolbox70,71 using a Aprilgrid with 36 AprilTags (6 × 6) printed and glued onto a rigid piece of cardboard. For calibration, a pinhole-radtan model was chosen and the camera frame rate was set to 4 Hz. The maximum reprojection pixel error was 1.2 with most of the assessed data points lying below 1 pixel (Figure 2).
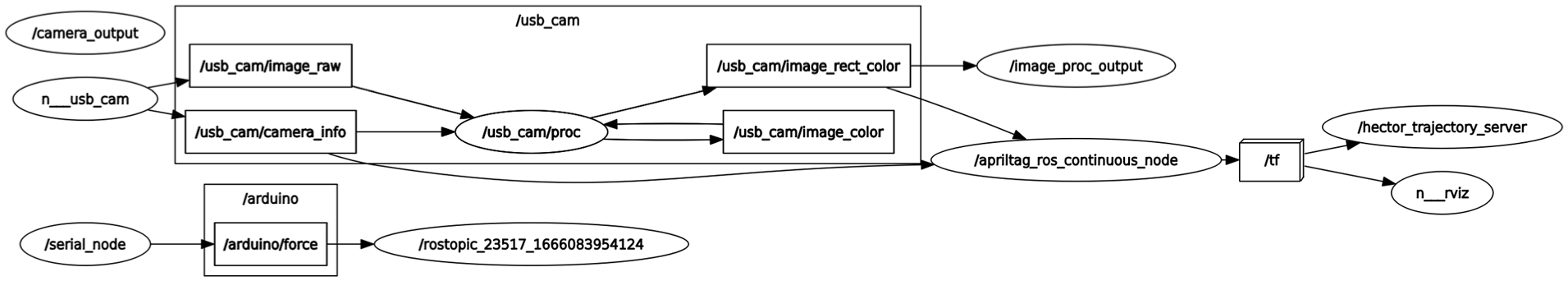
A plot demonstrating the ROS system (nodes and topics) used in this study.
Ultrasonic bone cutting devices operate at a low ultrasonic frequency (typically between 20 and 35 kHz). 57 In a preliminary study, we compared a 20 kHz and a 35 kHz device and found that the 35 kHz device exhibited less damage to the bone tissue at the cut site. 72 Therefore, in this study we used a 35 kHz device with a previously described rounded blade. 73 Detailed description of the surgical device design and characterisation, including measurement of the ultrasonic vibration response, have been reported previously. 30 Briefly, three orthogonal components of vibration velocity were measured at a grid of points on the device surface using a 3-D laser Doppler vibrometer. Frequency Response Functions (FRFs) were computed from excitation and response signals using dedicated software, followed by curve fitting and modal analysis to extract FRF magnitude and phase and hence the modal parameters. 30 During cutting, the device’s electro-mechanical characteristics of impedance, resonance frequency and consumed acoustic power were measured using a commercial ultrasonic generator and resonance tracking unit (PDUS210 Piezodrive).
The ultrasonic device was driven by the generator with 800 Vp-p output voltage and power load of 210 W, equivalent to a surgical device tip displacement amplitude of 36 µm. While this is lower than some of those reported for commercial surgical devices, ranging from 40 to 60 µm, 74 it has been determined in experiments that cortical bone can be cut effectively with a surgical tip displacement amplitude of 20 µm.
Force calculations
The signal from the force transducer was connected to an Arduino microcontroller via a strain gauge amplifier, which provided both signal amplification and analogue-to-digital (A/D) conversion. The force measurement range, denoted as
The strain gauge amplifier output was configured to a range of 0–10 V, with the Arduino Due only capable of reading voltages from 0 to 3.3 V. The amplifier gain was adjusted accordingly so that a force of 55 N corresponded to 3.3 V at the Arduino input. To convert voltage readings into force, the resolution of the load cell at 0 and 1 N was first calculated (equation (3)), followed by the actual force calculation using the ADC readings (equation (4)):
Eliminating the force of the irrigation fluid
As irrigation was used during the experiment, it was required to eliminate the force of the irrigation fluid from the force readings. The force during 1 min (equivalent to each bone cutting experiment duration) of irrigation with PBS was measured and the average force and standard deviation was calculated (equation (5)).
The measured average force due to irrigation, for a 1 min duration, was 0.38 ± 0.14 N.
The measured average force for each experiment is also calculated using equation (5), however,
Measuring cutting parameters during device operation by a clinician.
Cutting tests
During cutting tests, samples were irrigated with Phosphate-Buffered Saline (PBS) at 25°C with the irrigation controlled using a MINIPULS® 3 Peristaltic pump set to 38 revolutions per minute. Three practising orthopaedic surgeons from the Edinburgh Royal Infirmary hospital volunteered to take part in the study. Each clinician was instructed to find a comfortable position at the workbench, which allowed an adequate arm rest without contacting the assembly. All surgeons chose to perform the cuts in a standing position, reflecting as closely as possible their clinical handling of a cutting instrument in surgery. Before performing the cuts, each surgeon was given a bone sample and asked to practice a few cuts to familiarise themselves with the surgical device. Once they were comfortable with using the surgical device, cortical bone samples were placed in the test clamp. Each surgeon was instructed to perform two separate cuts on three separate cortical bone samples (6 cuts per bone sample) and to ensure that each cut was made without pausing (3 surgeons × 2 cuts × 3 bone samples, total of 18 cuts). For these tests, the following nomenclature is adopted: surgeon (S) number (1–3), followed by sample (s) number (1–3) and cut (c) number (1–2), such that for example, S1.s2.c1 pertains to surgeon 1, sample 2, cut 1.
Cutting speed
To calculate the instantaneous horizontal (
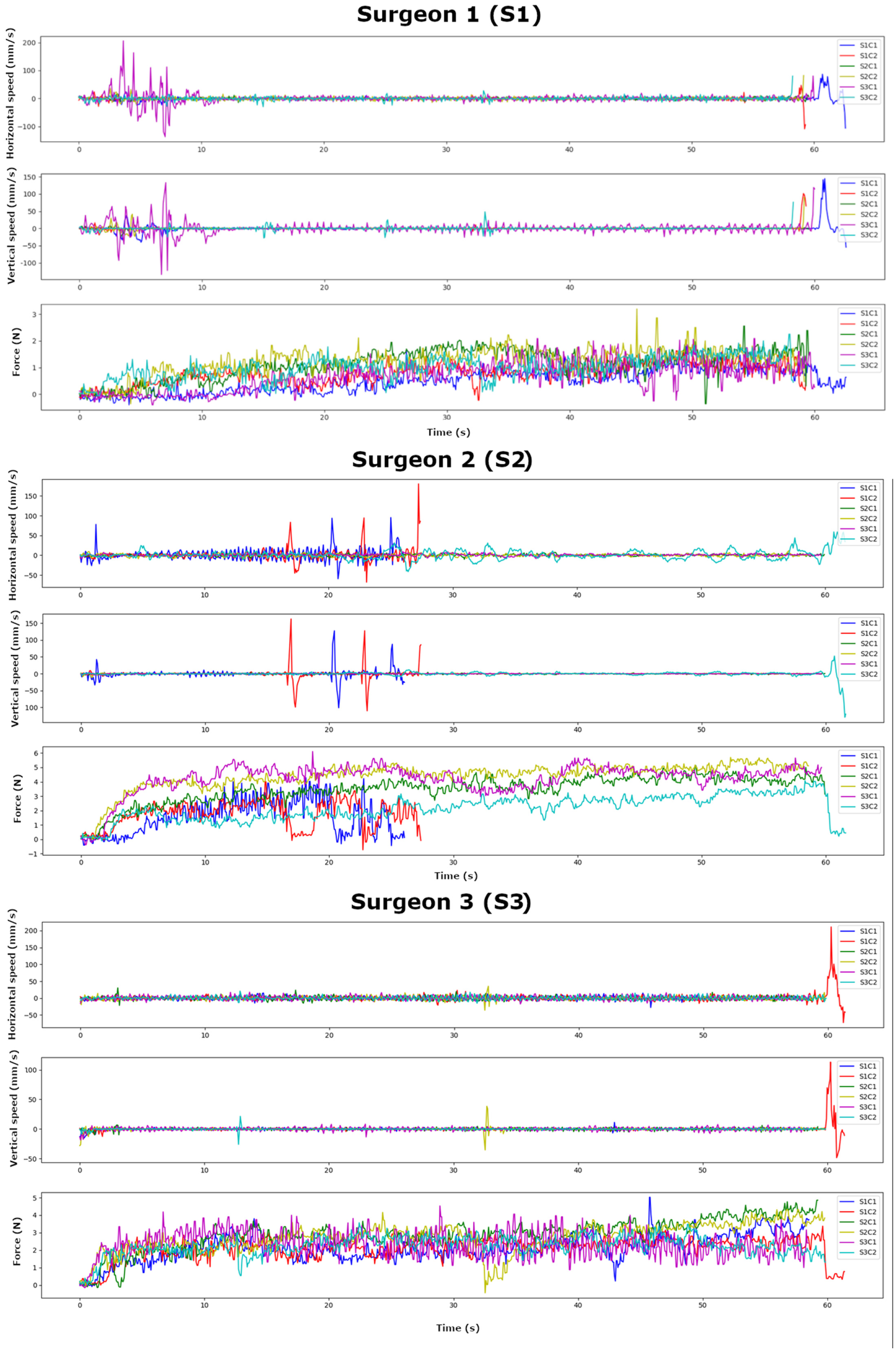
Horizontal and vertical cutting speeds (mm/s), and cutting force (N): Surgeon 1, Surgeon 2 and Surgeon 3.
Bone penetration rate (BPR)
Unlike the device vertical speed which was measured from the AprilTag, the bone penetration rate (BPR) was calculated from equation (4):
Where:
For any sample with a total cutting time of less than 1 min, the BPR was not computed.
Bone mineral density (BMD) and cut depth
To remove the considerable effects on cutting tests of low bone density, we confirmed that none of the patients had any reported history of osteoporosis by measuring the BMD of the bone cores. To convert the measured Hounsfield Units (HU) into BMD, we calibrated the micro-CT scanner using two 4 mm core diameter rods of calcium hydroxyapatite phantoms with known mineral densities (
Bone mineral density (BMD) for the three core cortical bone samples used for cutting.

To measure the cut depths, the bone samples were embedded in agar and imaged using the micro-CT scanner and settings as described for scanning the bone phantoms. Images of the bone samples were reconstructed using SkyScan software (V1.7.4.2). The depths of the cut were measured using stacked micro-CT scan images analysed with ImageJ software (version 1.54f). To ensure measurement consistency, the software was calibrated with the pixel-to-micron ratio of the micro-CT specifications, and the images were stacked sequentially for proper alignment. Consistent regions of interest (cut site) were defined across all images using ImageJ’s region of interest (ROI) manager. The depth was then calculated by measuring the distance between the surface and the bottom of the deepest point in the sagittal plane of the cut.
Testing using human cortical bone samples
Discard tissue (excised femoral heads) from three hip osteoarthritic patients undergoing hip replacement surgery was collected from the Edinburgh Royal Infirmary hospital, following approval from an institutional review board (ethics approval number: SR1342) and prior informed consent from all patients. Excised femoral heads were submerged in Dulbecco’s Modified Eagle Medium (DMEM; 41965-039; Invitrogen, Paisley, UK) and stored in a sterilised container in a refrigerator (−4°C), ready for same day collection. Following same-day sample pick-up, samples were transported to a controlled laboratory setting for experimental testing. We used a hand powered 10 mm core drill to obtain a bone sample core from the femoral neck region from each of three different hip replacement patients, where cortical and trabecular bone were deemed available (Figure 4). To ensure bone tissue integrity could be retained as much as possible, all bone samples were tested within 1 week of harvesting. Each bone sample was wrapped individually in sterile gauze soaked in Phosphate-Buffered Saline (PBS) and fresh-frozen at −20°C until required. On the day of testing, the samples were thawed for 2 h before use. For testing, cortical bone samples were mounted one at a time onto a custom designed test clamp, 3D printed in Polylactic Acid (PLA; Figure 5).
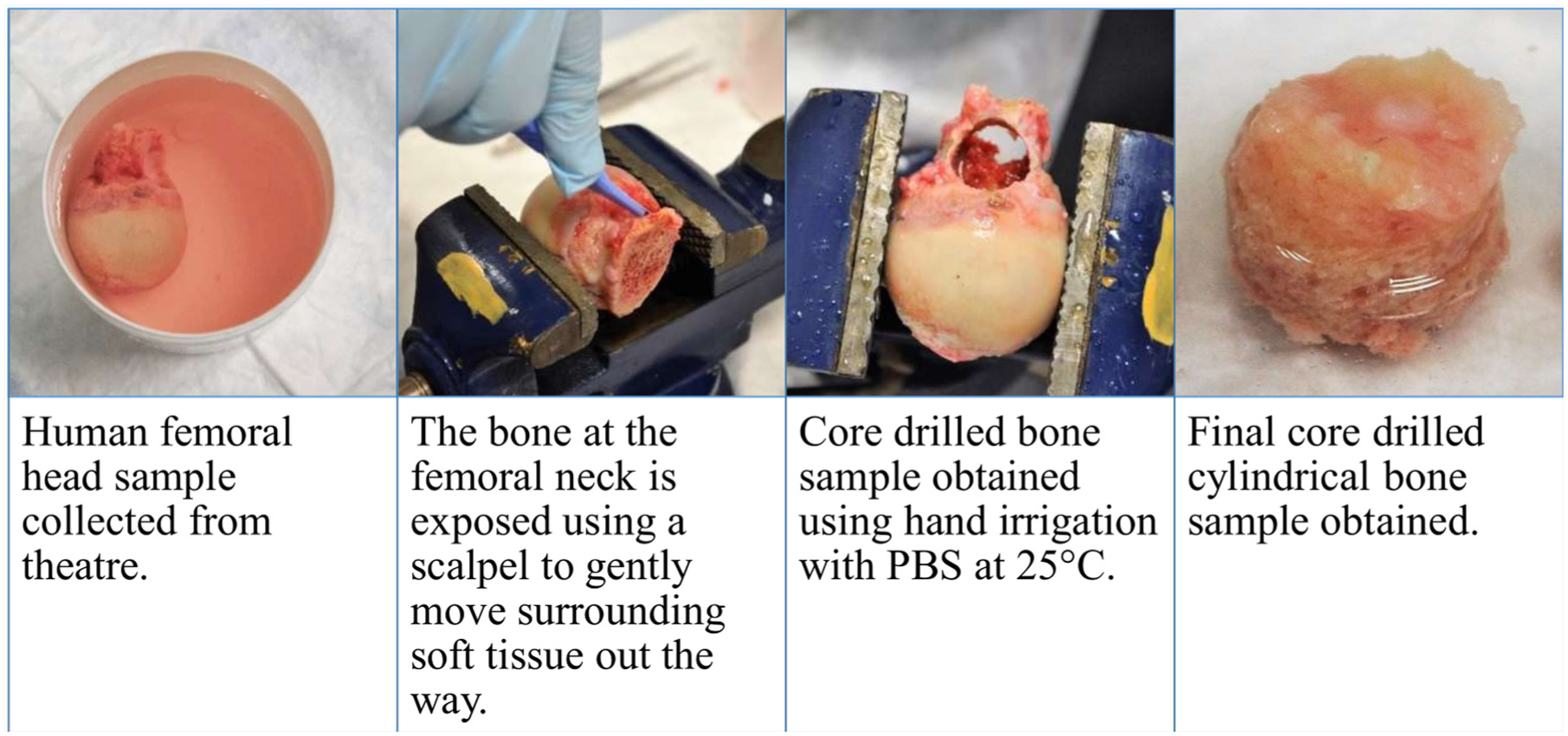
Process to obtain cylindrical human bone core samples.

Human bone sample used for cutting testing held in custom designed 3D printed clamp.
Results
The overall designed system is depicted in Figure 6.
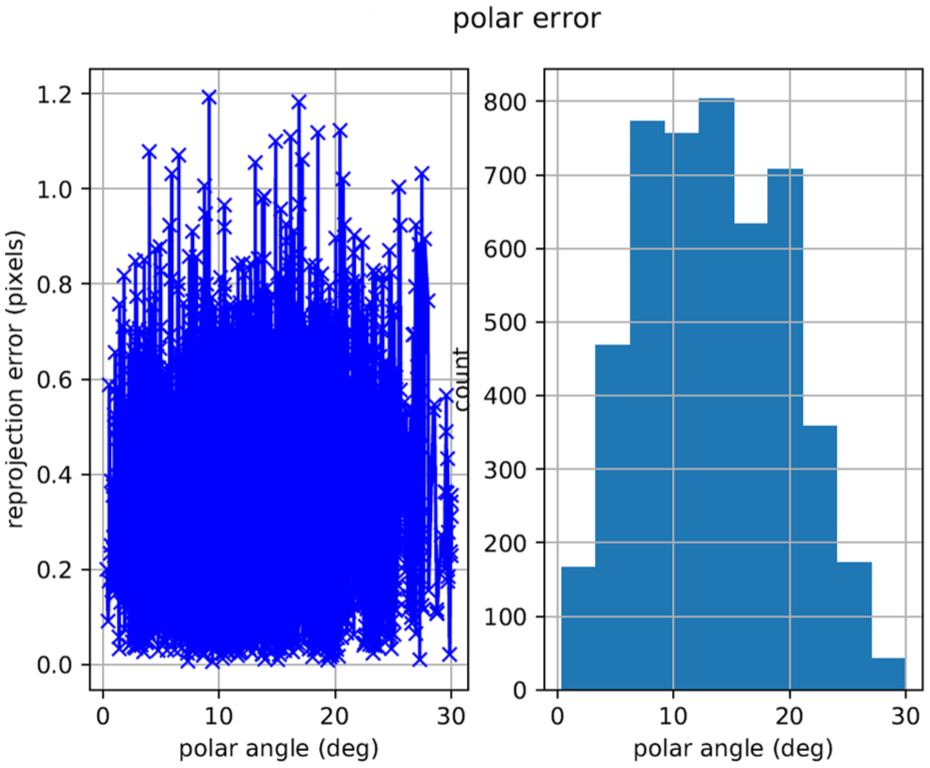
Re-projection error results from Kalibr calibration.
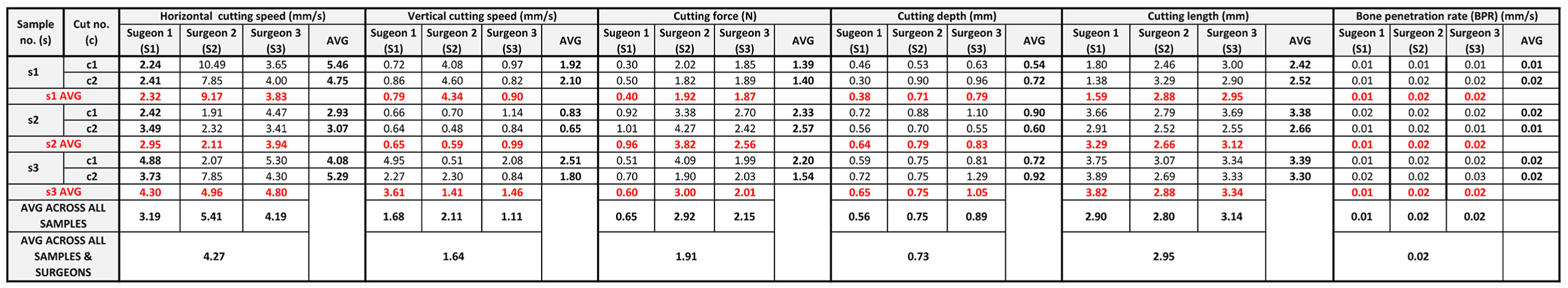
The average horizontal cutting speed, vertical cutting speed, vertical cutting force, cutting depth, cut length and the calculated BPR across all surgeons and all samples were 4.27 mm/s, 1.64 mm/s, 1.91 N, 0.73 mm, 2.95 mm and 0.02 mm/s (Table 2).
Surgeon (S1, S2 and S3) vertical cutting speed (mm/s), cutting force (N) and cutting depth (mm) for sample 1 (s1), sample 2 (s2) and sample 3 (s3).
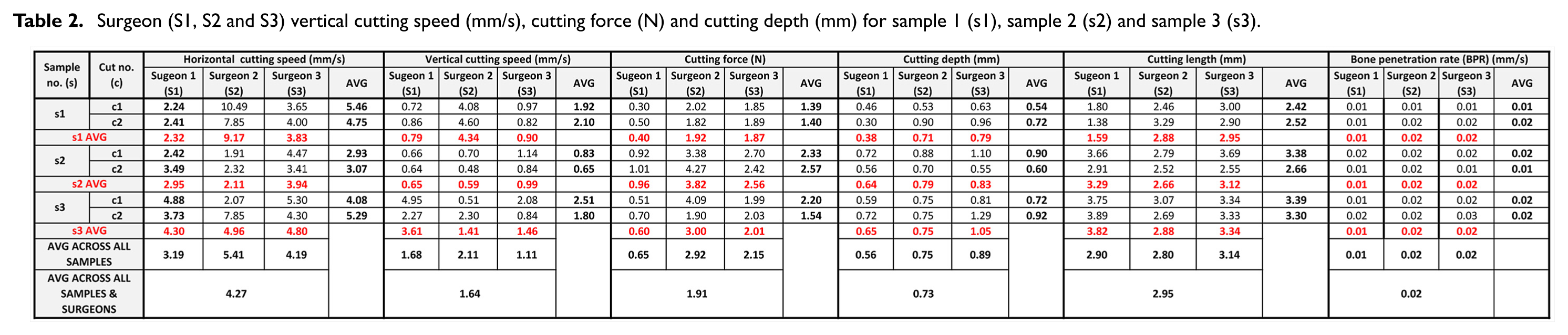
S1 and S3 had a consistent approach to cutting bone for all samples and cuts (Figure 3). However, for S2.s3.c2 the surgeon struggled to advance the cutting blade into the bone. The surgeon pivoted the surgical device at the cut site to try to perform the cuts, which can be seen in the larger variation in the surgical device horizontal speed for this sample (Figure 3).
For S2.s1.c1 and S2.s1.c2, the resonance tracking system (Piezodrive) overloaded, which results from the surgical device being advanced too quickly into the bone, so this experiment was prematurely aborted (the cutting time was <1 min, the force of the irrigation fluid was adjusted accordingly). For the remaining samples and cuts for S2, cuts were carried out for the full cutting time of 1 min.
Discussion
There are multiple potential advantages of using ultrasonic surgical devices for orthopaedic surgeries. In addition to the very low cutting forces and minimal vibration displacements compared to conventional bone cutting tools, 31 ultrasonic surgical devices offer enhanced safety due to their selective cutting of mineralised bone while sparing adjacent soft tissues and the elimination of rotary cutting motion which reduces the risk of soft tissue entanglement.25,76 These properties also reduce the risk of bone microcracks, improve surgical visibility by limiting bleeding and debris and enable greater control in confined or anatomically complex regions. Furthermore, the applications of ultrasonic cutting tools for orthopaedic surgeries have expanded to explore the use of ultrasonic bone cutting devices as a potential strategy for chondroprotection during cartilage injury repair. 30
However, despite the numerous in vitro and in silico studies investigating the use of ultrasonic cutting devices for orthopaedic bone cutting applications,39–61 these studies have been conducted in highly controlled laboratory environments, using mechanical test rigs instead of surgeons, and with most studies using non-human bone,43,45,46,48–50,61–64 or through in silico studies where input cutting parameters (normally surgeon related parameters) and the cutting sample are idealised.38–42,47,51 However, different surgeons will each interact with these devices differently, and the conditions encountered in clinical practice are complex, variable, and often unpredictable. While numerous clinical studies have investigated the use of ultrasonic cutting surgical devices for cutting various bones in the axial skeleton, including dental,4,5 craniomaxillofacial,6–13 mandibular11,14–21 and spinal22–26 bones, and in the appendicular skeleton, including bones in the hand, 29 these studies have typically measured clinical outcomes for example, amount of bleeding,4,9,14,16,19,21,23,24 precision for example, cutting length/depth,5,18,26,29 amount of soft tissue damage,6,7,15,23,26 total operative time,6,9,14,16,19,21,23,24,29 bone cutting time, 7 duration of inpatient stay,9,16,24 post-operative healing/functional capacity8,16,19–21,23,29 (e.g. pain 77 ), blood pressure, 9 blood values, 9 required medication, 9 among other clinical parameters specific to the surgical procedure,9–12,17 without reporting how the devices were operated. Only one clinical study tracked the tool position during surgeon cutting of facial bones in patients with a variety of pathologies. The study confirmed that tracking ensured continuous position control, however, the authors did not report the surgeon related parameters which drive overall device performance, and clinical outcomes. 13 Understanding these interactions is crucial, as surgeon-related factors directly influence device performance and clinical outcomes. Analysing surgeon–device interaction can inform training and device optimisation strategies, helping translate bench testing results into effective clinical use and ultimately improving surgical performance and patient outcomes.
In this study, we designed a novel experimental setup that allows measuring surgeon related cutting parameters of practicing orthopaedic surgeons performing cuts on fresh human cortical bone using an ultrasonic surgical device within a controlled yet clinically realistic environment. To demonstrate feasibility of our developed setup, three practising orthopaedic surgeons each performed two separate cuts on three separate human cortical bone samples (six cuts per bone sample) (3 surgeons × 2 cuts × 3 bone samples, total of 18 cuts). As the AprilTag was located at the top end of the cutting device near to where surgeons were holding the device, our setup enabled surgeon related parameters to be captured. This analysis captured how surgeons manipulated the cutting device in real time, using the AprilTag’s position and motion to quantify the device’s trajectory relative to the bone sample. Therefore, the results reflect how a practicing orthopaedic surgeon uses an ultrasonic cutting device to cut human bone samples within a controlled yet clinically realistic environment. Across all surgeons and samples, we observed an average horizontal cutting speed of 4.27 mm/s, a vertical cutting speed of 1.64 mm/s, and a vertical cutting force of 1.91 N (Table 2). These kinematic and force measurements were directly linked to micro-CT-derived outcomes, which showed an average cut depth of 0.73 mm and a cut length of 2.95 mm. From this, we calculated a Bone Penetration Rate (BPR) of 0.02 mm/s. The discrepancy is not a shortcoming of our method, but rather a revealing insight into the complex biomechanical interactions during manual bone cutting. Several contributing factors may explains the difference: (1) bone tissue exhibits both elastic and viscoelastic properties, meaning some portion of vertical tool displacement is absorbed through deformation rather than resulting in direct material removal; (2) hand and skin compliance in the surgeon’s grip may introduce damping effects not accounted for in rigid-body assumptions; and (3) the cantilever-style load cell used for force measurement may introduce minor mechanical compliance. However, perhaps most important, our tracking method – based on the AprilTag affixed near the surgeon’s grip – captures the actual hand-driven movement of the tool, accurately characterises the functional cutting behaviour during a controlled, linear cutting task and offering high-fidelity data on how motion intent translates into cutting performance. By decomposing the surgical trajectory into horizontal and vertical components, we provide a time-resolved view of the cutting dynamics that reflects real-world variability in device use technique. The BPR thus serves as a performance-linked metric that captures how each surgeon’s motion strategy influenced effective vertical cutting progress. Overall, our method provides a robust, interpretable, and surgeon-specific assessment of bone cutting performance, grounded in both motion capture and force data. Future work may explore recording device rotations using the AprilTag, from which it would be possible to derive precise tip motion using the known tag-to-tip transformation matrix and the overall device geometry. Unlike purely theoretical or tool-tip-only tracking approaches, our system reflects the actual dynamics of surgeon-device interaction, offering superior insight into both technique and provide insight into device performance outcomes.
The results herein offer several useful insights. We confirmed that when surgeons use ultrasonic surgical devices to cut human cortical bone, it can significantly reduce intra-operative cutting force. The average measured cutting force across all surgeons and human cortical bone samples in this study was 1.91 N. These results are comparable with a previous study, where practising oral surgeons cut a fresh heifer (bovine) femur cubic sample with an approximate volume of
The currently reported BPR of 0.02 mm/s is very likely too slow for practical implementation in large-scale orthopaedic bone cutting procedures. Given a mean articular tibial plateau width for males of 82.48 mm, 85 our current surgeons would require at least 4124 s (68.73 min) to cut across the width using the device in this study. To begin to translate bench testing results into effective clinical use and ultimately improve surgical performance and patient outcomes, we must begin by understanding from our results how a given surgeon may be interacting with a device. The average force of S2.s1.c1 and S2.s1.c2 of 1.92 N is comparable to the 1.87 N for S3.s1.c1 and S3.s1.c2, however, the vertical speed for S2 was 4.34 mm/s, which was over four times that for S3 (0.90 mm/s). The stalling observed in the ultrasonic device during S2 trials is likely due to this higher vertical feed rate, which may have exceeded the device’s ability to dynamically adjust excitation frequency and power in real-time. This highlights the importance of balancing applied force and vertical feed rate during ultrasonic cutting. While previous work using porcine bone suggested that a contact load of 150 g (1.5 N) resulted in the greatest cut depth, 86 that study used a different cutting configuration – one focused on vertical penetration rather than lateral cutting. As such, its relevance here is limited, but it does underscore the broader point that applied load has a strong influence on cutting performance. An optimal cutting strategy would aim to maintain the lowest effective applied vertical force necessary to achieve effective cutting, while operating within a feed rate that does not overwhelm the device’s control system or generate excessive heat or mechanical damage. These exact limits and ranges are still under investigation, specifically for large bone studies.
To mitigate the risk of device overload and improve consistency, integration with robotic surgical platforms offers a clear advantage. These systems can be programmed to maintain an optimal and consistent applied force during cutting, reducing the risk of excessive tissue damage, transducer stalling or high cutting temperatures.
Given the clear value in measuring and understanding how the surgeons’ hands move the cutting device relative to the bone sample, for example, the vertical and horizontal cutting speeds, future work should explore the value of the ‘surgeon use path’ under realistic surgical conditions for making a cut and look at how that influence different cutting parameters such as for example, optimal parameters, the effect of using different blade profiles on cutting parameters, identify how generalisable the results reported in this study are and investigate how results compare before and after implementing surgeon training on optimal parameters.
Conclusions
Ultrasonic cutting devices, which operate with low force, may reduce MSP and the physical burden during surgery. This study presents the successful development of a novel and clinically realistic experimental setup for measuring surgeon-controlled parameters during ultrasonic cutting of fresh human cortical bone within a controlled yet clinically realistic environment. By capturing real-time data on cutting force, speed and tool motion from practicing orthopaedic surgeons, we offer insights into how different surgeons each interact with these devices differently. Consistent with previous studies, our findings confirm that ultrasonic devices enable low force cutting (1.91 N). However, our results highlight significant variability in surgeon-device interaction, including how much force each surgeon applied, and how the device was manoeuvred which can influence cutting efficiency, thermal effects, and overall surgical outcomes. Given the critical importance of surgeon-related factors that directly influence device performance and clinical outcomes, our results highlight the need to understand how a given surgeon may be interacting with a device. This can inform training and device optimisation strategies, help translate bench testing results into effective clinical use and ultimately improve surgical performance and patient outcomes. Our results also support the potential of integrating ultrasonic devices with robotic platforms to maintain controlled cutting parameters and enhance consistency. Future work should explore optimal parameters, the effect of using different blade profiles on cutting parameters, identify how generalisable the results reported in this study are and investigate how results compare before and after implementing surgeon training on optimal parameters.
Footnotes
Acknowledgements
The authors thank Louise Robiati, Joe Esland, Mohsem Khadem, Stephanie Collishaw, Christine Beadle, Pauline Irvine for their assistance with this study.
Author contributions
Irene Yang: Writing – review & editing, Writing – original draft, Visualisation, Validation, Software, Methodology, Investigation, Formal analysis, Data curation, Conceptualisation. Russell Buchanan: Writing – original draft, Visualisation, Validation, Software, Methodology, Investigation, Formal analysis, Data curation. Nisreen Al-Namnam: Methodology, Investigation, Formal analysis, Data curation. Xuan Li: Methodology, Writing – review & editing. Margaret Lucas: Writing – review & editing, Supervision, Resources, Project administration, Funding acquisition. A. Hamish R. W. Simpson: Writing – review & editing, Supervision, Resources, Project administration, Funding acquisition, Conceptualisation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by an Engineering and Physical Sciences Research Council (EPSRC) Programme grant: Ultrasurge – Surgery enabled by Ultrasonics (EP/R045291/1).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.