Abstract
The aim of this study was to perform an initial assessment, in vitro, of the feasibility of using a glutaraldehyde cross-linked porcine mitral valve to retain acute functionality, focusing on assessing mitral regurgitation. Six porcine hearts were tested using an in vitro simulator. Testing was repeated following cross-linking of mitral valves; where cross-linking was achieved by placing them in a glutaraldehyde solution. The simulator enabled systolic pressure on the ventricular side of the valve to be mimicked. Following testing, mitral valve leaflets underwent Scanning Electron Microscopy of the ventricular surface of both the anterior and posterior leaflets (1 cm2 samples). The peak pressure withstood by cross-linked valves was significantly lower than for untreated valves (108 mmHg cf. 128 mmHg for untreated valves; p < 0.05). The peak pressure was typically reached 0.5 s later than for the untreated valve. While both cross-linked and untreated valves exhibited endothelium denudation, the unfixed valve had less endothelial loss. Glutaraldehyde cross-linking of porcine mitral valves may be of potential value in assessing improved bioprosthetic mitral valve replacements. However, a more immobile valve exhibiting endothelial denudation (i.e. sclerosis) was a possible concerns identified following in vitro acute assessment.
Introduction
The mitral valve consists of anterior and posterior leaflets connected to papillary muscles by chordae tendineae. 1 Chordae restrict the motion of the leaflets to prevent the prolapse of leaflets into the left atrium during systole. 2 Thus, the mitral valve’s function is to prevent blood backflow during systole (into the left atrium), while allowing unrestricted unidirectional flow during diastole; this relies on mobility, elasticity and structural integrity of the valve’s leaflets. 3 Mitral stenosis is the narrowing of the mitral valve orifice which obstructs blood flow through the valve, and can increase the risk of heart failure, stroke and atrial fibrillation. 4 Mitral regurgitation occurs, instead, when blood leaks backwards through the valve during systole.
Surgical correction can be required to reduce the risk of morbidity and mortality during severe mitral valve regurgitation. 5 However, repair cannot be performed clinically if the valve has become too damaged. Replacement is currently performed either with a mechanical valve or a bioprosthetic valve. Replacement with a mechanical device requires anticoagulants and carries a small constant annual risk of endocarditis6,7; whereas bioprosthetic valves have reduced durability. 6 Replacement valves tend to use an aortic valve configuration, despite the potential benefits of using a shape which conforms with the innate, but healthy, mitral valve. Increasing the functionality and longevity of bioprosthetic replacements is of interest given the prevalence of heart failure in an ageing global population. 8
Glutaraldehyde cross-linking (or fixation) is most commonly used for the preservation of bioprosthetic heart valves.9–11 Glutaraldehyde causes collagen molecules to internally cross-link, which is the formation of chemical bonds between the collagen fibrils which provides reinforcement to the tissue. 12 This provides the tissue with a range of benefits; it causes resistance to biodegradation, suppresses the antigenicity of collagen and ultimately improves durability.9–11,13 Paradoxically, cross-linking also increases the calcification rate of the tissue, which is the leading cause of heart valve failure and can result in tearing and rupture.12,14 As porcine mitral valves are structurally and geometrically similar to human hearts, 15 this structure might provide an alternative for mitral valve bioprosthetic replacements over the use of aortic valve based structures. Although the distinct anatomy of the mitral valve aids blood haemodynamics, 16 a potential issue would be the anticipated increase in tissue stiffness.10,13 Thus, it is unclear whether the potential benefits of using a mitral valve as a bioprosthetic device might outweigh the potential limitations.
The aim of this study was to perform an initial assessment, in vitro, of the feasibility of using a glutaraldehyde cross-linked porcine mitral valve to retain acute functionality, focusing on assessing mitral regurgitation. This in vitro technique has been used previously to assess mitral valve competence and regurgitation.17,18 In addition, valve leaflets have been analysed using Scanning Electron Microscopy (SEM) following in vitro testing. Elastic and viscoelastic properties of chordae from the heart valves have also been characterised but are presented separately; as Part-2 of this study. 19 The material’s testing data is reported separately because data obtained were not in broad agreement with expected results 20 ; therefore, they have been assessed in detail, aiming to account for the wider implications as regards tissue mechanics.
Methods
Specimen preparation
Six porcine hearts were obtained from a supplier (Fresh Tissue Supplies Ltd., West Sussex). On arrival, the hearts were individually wrapped in tissue soaked in Ringer’s solution (Oxoid Ltd, Basingstoke, UK) and stored in heat-sealed bags at −40°C. Before dissection and testing, the hearts were defrosted for 24 h at 4°C and after dissection, the hearts were then frozen until needed for testing. The protocol for storage, handling and dissection of the porcine hearts was in accordance with previous studies.17,18,20,21 The dissection method produced a specimen with a mitral annulus and papillary muscles connected to a ring of ventricular muscle, so that the sub-valvular apparatus was kept intact (Figure 1).
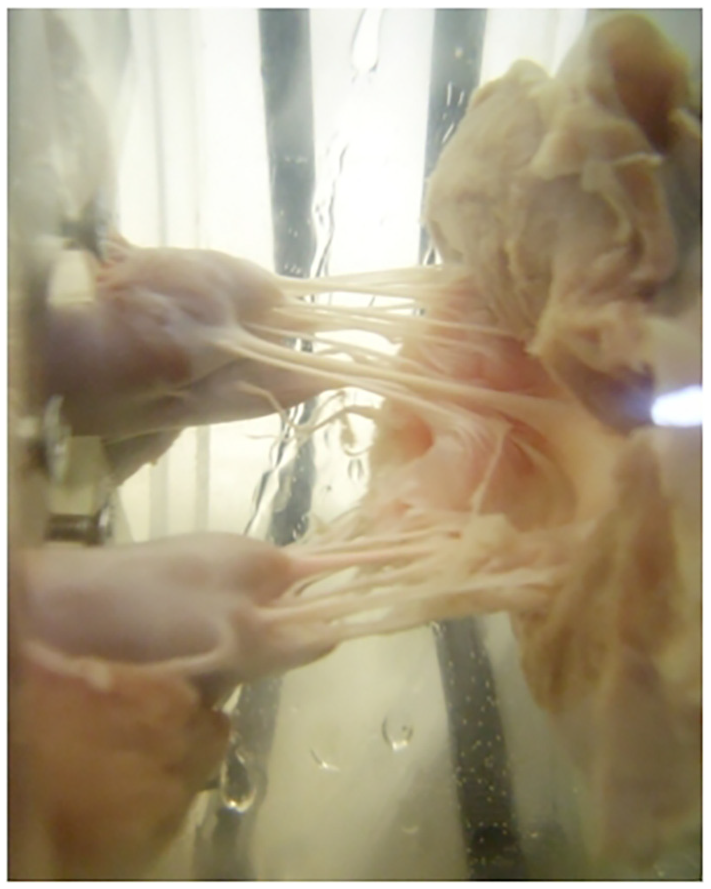
Mitral valve specimen within the in vitro simulator. The papillary muscles are connected to a papillary muscle annular ring of muscle, clamped in place within the simulator. A ring of muscle is maintained around the mitral annulus to facilitate suturing of the annulus on to rubber sheeting.
Mitral valve specimens were initially tested using an in vitro procedure (further described in section 2.3). Valves subsequently underwent cross-linking (section 2.2) before undergoing a repeat in vitro testing procedure. Following both cycles of in vitro testing, chordae were dissected from the mitral valve for mechanical testing; reported elsewhere. 19 Mitral valve leaflets were dissected and prepared for examination using Scanning Electron Microscopy (SEM; section 2.4). For clarity, the experimental protocol is outlined in Figure 2.
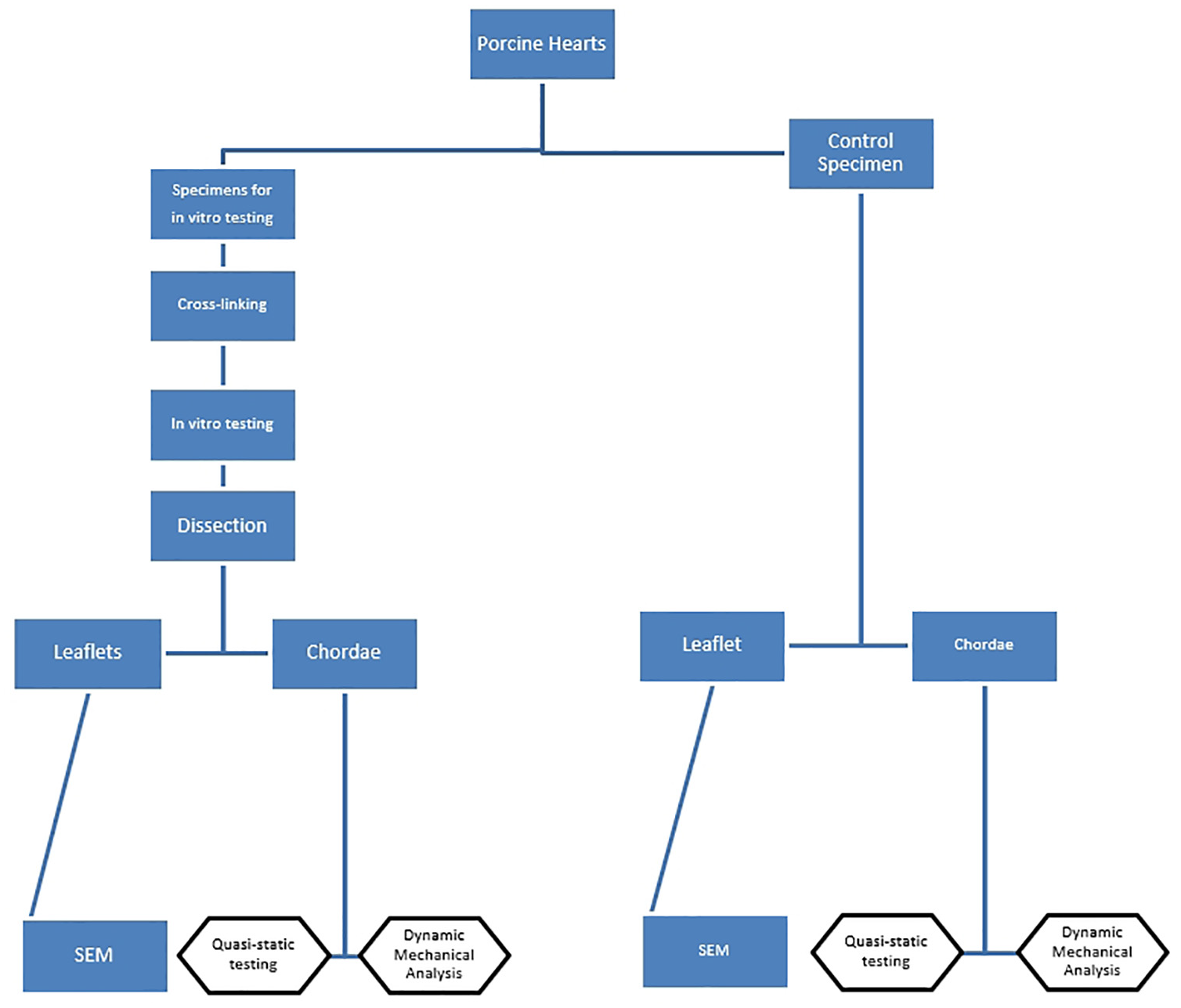
Outline of the experimental protocol used in this current study. Blue rectangles denote work reported in this current study; boxes with a black outline are reported in part-2 of this study. 19
Glutaraldehyde cross-linking
Cross-linking of mitral valves was achieved by placing them in a glutaraldehyde solution. Valves were placed in 1 L containers (approximately 10 times their volume) of 0.6% glutaraldehyde (12 mL of 50% glutaraldehyde; Fluka Analytical, Sigma Aldrich, St Louis, MO, USA) made up to 1 L with 0.2 mol/L and pH 7.4 phosphate-buffered saline (PBS) and left for 24 h.22–24 Subsequently, mitral valves were removed from the solution, and rinsed/soaked in PBS (three repeats) for 10 min. 21
In vitro simulator
The in vitro simulation system was set up so that water was pressurised, hydrostatically, from a pump (Model 12107-15, Cole-Parmer, London), via flexible tubing, and through to a test chamber corresponding to the ventricular aspect of the valve (Figure 3(a)). A digital pressure differential transducer (Omega Engineering Limited, Northbank Ind Park, Manchester, UK) was used to measure the pressure on the ventricular side of the valve. The pressure is reported in mmHg because of the clinical relevance (1 kPa corresponds to 7.5 mmHg).
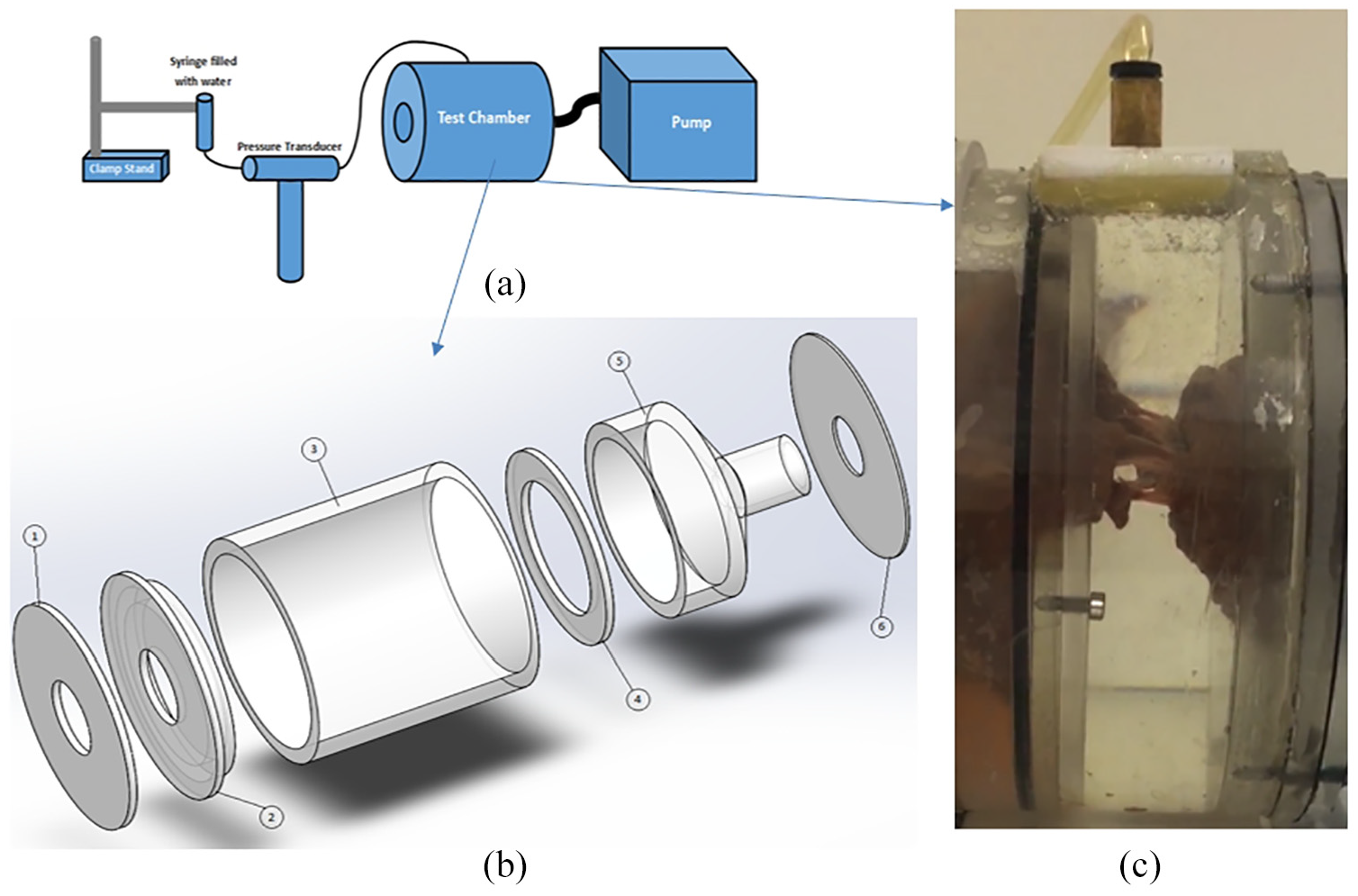
Mitral valve test apparatus. (a) Schematic outlining how the test chamber, which contains the mitral valve, is linked to the main components of the apparatus. (b) Exploded view of the test chamber components: 1 and 2 are perspex plates which fix rubber sheeting onto the main test chamber; 3 is the external cylinder which encases the test chamber; 4 and 5 hold the papillary muscles in place (they are placed within their holder into the cavity of component 5, and component 4 clamps their holder in place within this cavity); component 6 seals the test chamber. (c) A mitral valve is shown within the test chamber, closed under hydrostatic pressure.
The test chamber is described in further detail elsewhere.17,18 Briefly, the test chamber secured the mitral valve in place, with pressure applied from the ventricular aspect of the mitral valve (Figure 3). Water was used as a pressuring fluid within this quasi-static test involving hydrostatic pressure. The components of the test chamber are identified in Figure 3(b). This chamber consists of an outer Perspex cylinder (component 3) containing an inner cylinder (component 5). Papillary muscles are held in place through the method of dissection which leaves an annulus-shaped ring of ventricular muscle (which has been termed papillary muscle ‘annulus’; Figure 4(a)). This papillary-muscle ‘annulus’ is held in place via a mountable Perspex holder (Figure 4(a)) which fits within component 5, and fixed in place within the inner cylinder via component 4 (Figure 3(b)). Component 6 seals the test chamber against the outer cylinder. The annulus of the mitral valve was sutured onto 1 mm thick rubber (Figure 4), using braided polyester surgical sutures (2-0 Ethibond excel, Ethicon, Cincinnati, US). The rubber sheeting is then attached to the outer cylinder by two Perspex back-plates (Components 1 and 2; Figure 3(b)). The mitral annulus was sutured to a D-shaped hole in the rubber sheet (Figure 4(b)), with dimensions based on a 26 mm Duran annuloplasty ring. 17 The final set-up of the mitral valve specimen within the mounted test chamber, is shown with the valve under hydrostatic pressure in Figure 3(c); a close-up image of a mitral valve within the test chamber is provided in Figure 1.
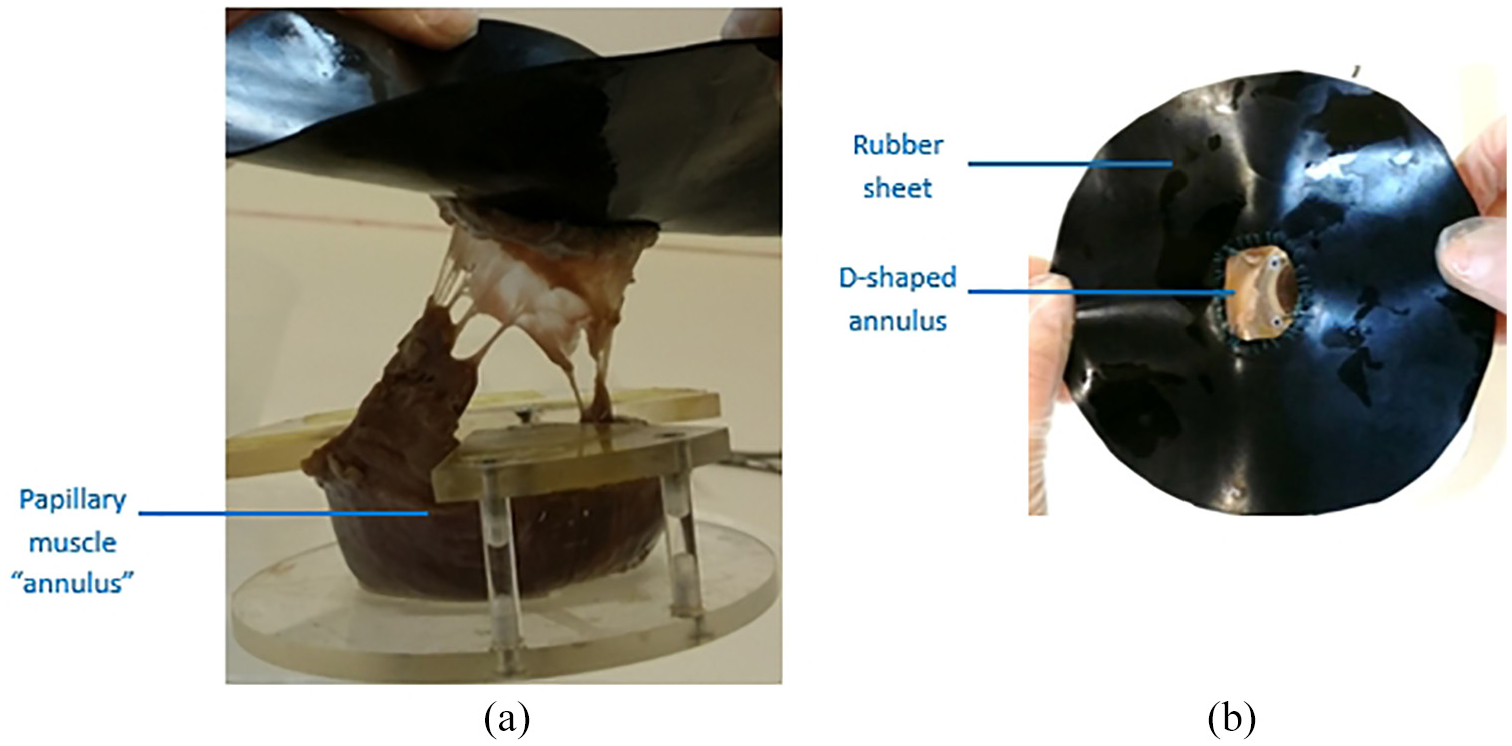
Attachment of a mitral valve specimen to: (a) a perspex holder which maintains papillary muscles in place via a ring of muscle tissue, termed papillary muscle ‘annulus’ and (b) rubber sheeting with D-shaped orifice onto which the annulus is sutured.
Mitral valves were tested up to pressures not exceeding 120 mmHg for eight loading cycles; the first two cycles were not used for analysis (the mechanical behaviour of soft connective tissues typically requires a couple of loading cycles before repeatable values are measured 25 ). Each loading cycle lasted 10 s, with the pressure increasing from 0 mmHg up to a peak pressure, followed by a pressure drop towards a mean stabilised pressure. Both a peak pressure and a mean stabilised pressure were recorded and used for analysis. The peak pressure was defined as the maximum pressure achieved during the initial stage of increased pressure (around 0–2 s; see Figure 5); while the mean stabilised pressure was defined to be the mean pressure measured after the peak pressure was reached, that is, the mean pressure which stabilises after around 3 s and held up to 10 s (see Figure 5).
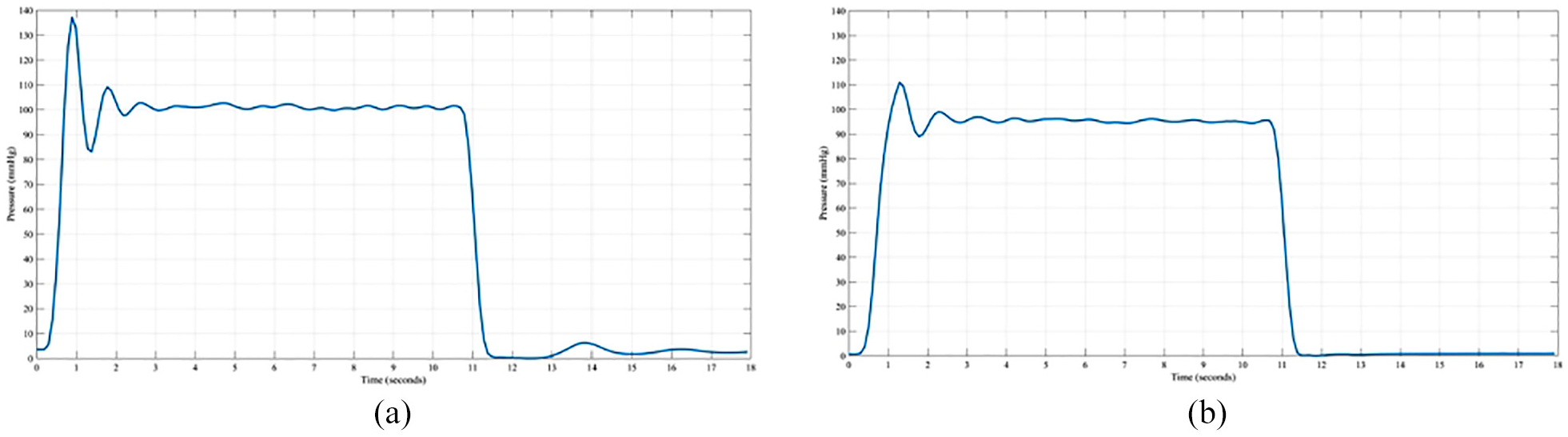
Sample pressure profiles for a valve tested (a) untreated and (b) following cross-linking.
Scanning Electron Microscopy
SEM was performed on the ventricular surface of both the anterior and posterior leaflets on 1 cm2 samples, following cross-linking and in vitro testing, using a Philips SEM microscope (Philips XL30 ESEM FEG, Amsterdam, The Netherlands). For comparison, an additional sample was scanned which did not undergo any further testing (i.e. the control specimen used in this part of the study; Figure 2).
SEM Samples were rinsed in Ringer’s solution following dissection prior to placement in a 2.5% glutaraldehyde solution (2 h). Once cross-linked, the samples were dehydrated using series of a graded ethanol concentrations 26 (70%, 90%, 100% and twice in 100% anhydrous ethanol) and placed in a critical point dryer (Polaron E3100, East Sussex, UK); where ethanol was replaced with liquid CO2 and heated under pressure to its critical point. Samples were mounted on stubs with silver paint to ensure a conductive path and coated with platinum using a sputter coater (Polaron SC7640, East Sussex, UK). 27
Data analysis
Statistical analysis was performed using the software SigmaPlot 13.0 (Systat Software Inc., London, UK). Paired comparisons were performed on unfixed versus fixed data for all the valves combined (p < 0.05). Data were tested for normality using the Shapiro-Wilk normality test. In addition paired t-tests (p > 0.05) were used to assess peak pressure (all data sets were normally distributed), and Wilcoxon Signed Rank tests (p < 0.05) were used for comparisons of the stabilised pressure (some data sets were not normally distributed).
Results
Cross-linking significantly reduced the peak pressure, but not the mean stabilised pressure withstood by mitral valves (Table 1; p < 0.05). The peak pressure withstood by cross-linked valves was 108 mmHg whereas the before treatment valves pressurised to 128 mmHg (Table 1). However, there was no significant difference in mean stabilised pressure (99 mmHg for untreated valves, cf. 87 mmHg for cross-linked valves; p > 0.05, Table 1). The cross-linked valve leaflets closed more slowly than that for untreated valves; the peak pressure was typically reached 0.5 s later than for the untreated valve (data for a sample valve is shown in Figure 5).
Mean peak and stabilised pressures (including standard deviation, SD) for porcine mitral valves before (untreated) and after glutaraldehyde cross-linking (Sample size, N = 6).
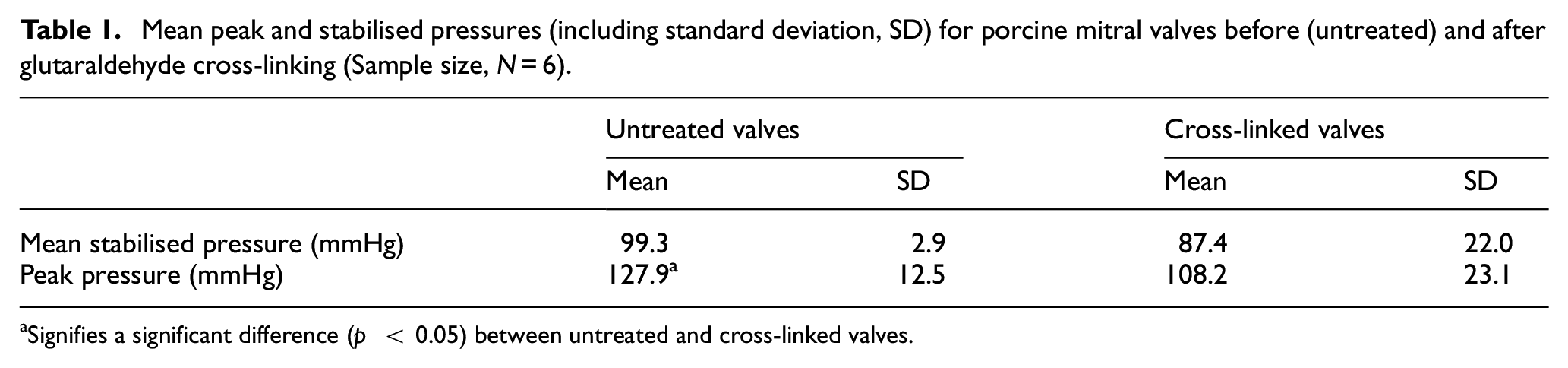
aSignifies a significant difference (p < 0.05) between untreated and cross-linked valves.
No leakage was observed from the valves before cross-linking. However, following fixation, some mitral regurgitation was observed consistent with the reduction in peak pressure. The anterior leaflet was observed to bulge when pressurised, but was less prominent in the fixed valve. The leaflets were also observed to be less mobile in the open position for cross-linked valves (consistent with the additional 0.5 s required to reach peak pressure, noted above). Indeed, for one valve, the leaflets were fully immobile and failed to close during the first three loading cycles (Figure 6). Such ‘bimodal’ function was not observed for untreated valves.
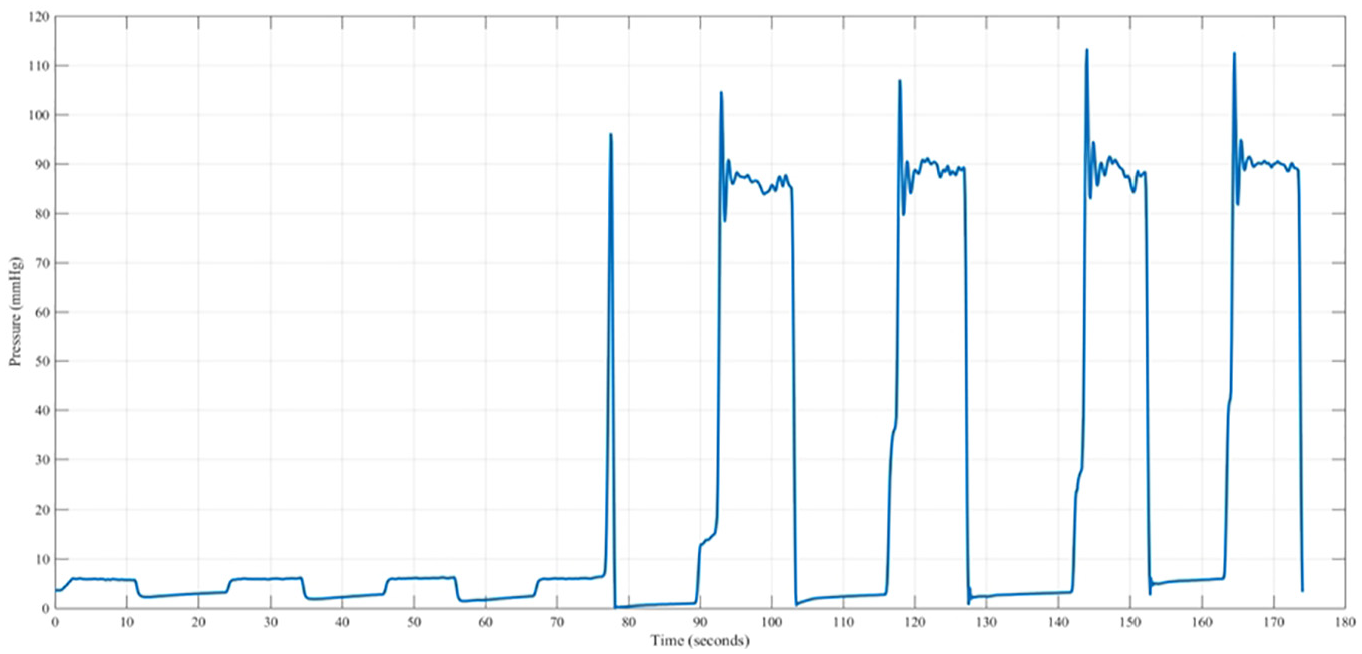
Time-dependent pressure for one cross-linked valve tested eight times. The valve failed to pressurise during the first three repeats of the test.
Both cross-linked and untreated valves exhibited endothelium denudation but to differing levels. The unfixed valve had less endothelial loss displayed by a smoother appearance which is representative of the uncovered basement membrane (Figure 7). The fixed valve had a more ‘fibrous’ texture which corresponds to the subendothelial components of collagen and elastin (Figure 8). On the surface of the fixed anterior leaflet the fibrils were intertwined (Figure 8(c)) and broken collagen fibrils can be observed on the posterior leaflet. At lower magnifications, transversely oriented parallel ridges were observed on the anterior leaflet of the fixed valve (Figure 8(a)); these ridges are partially visible on the unfixed anterior leaflet but are less pronounced (Figure 7(a)). The surfaces of the anterior and posterior leaflets of the unfixed valve are structurally similar and visually exhibit comparable degrees of endothelial denudation (Figure 8). This is also the case for the fixed valve apart from the lack of parallel ridges noticeable on the posterior leaflet (Figure 8(e)).
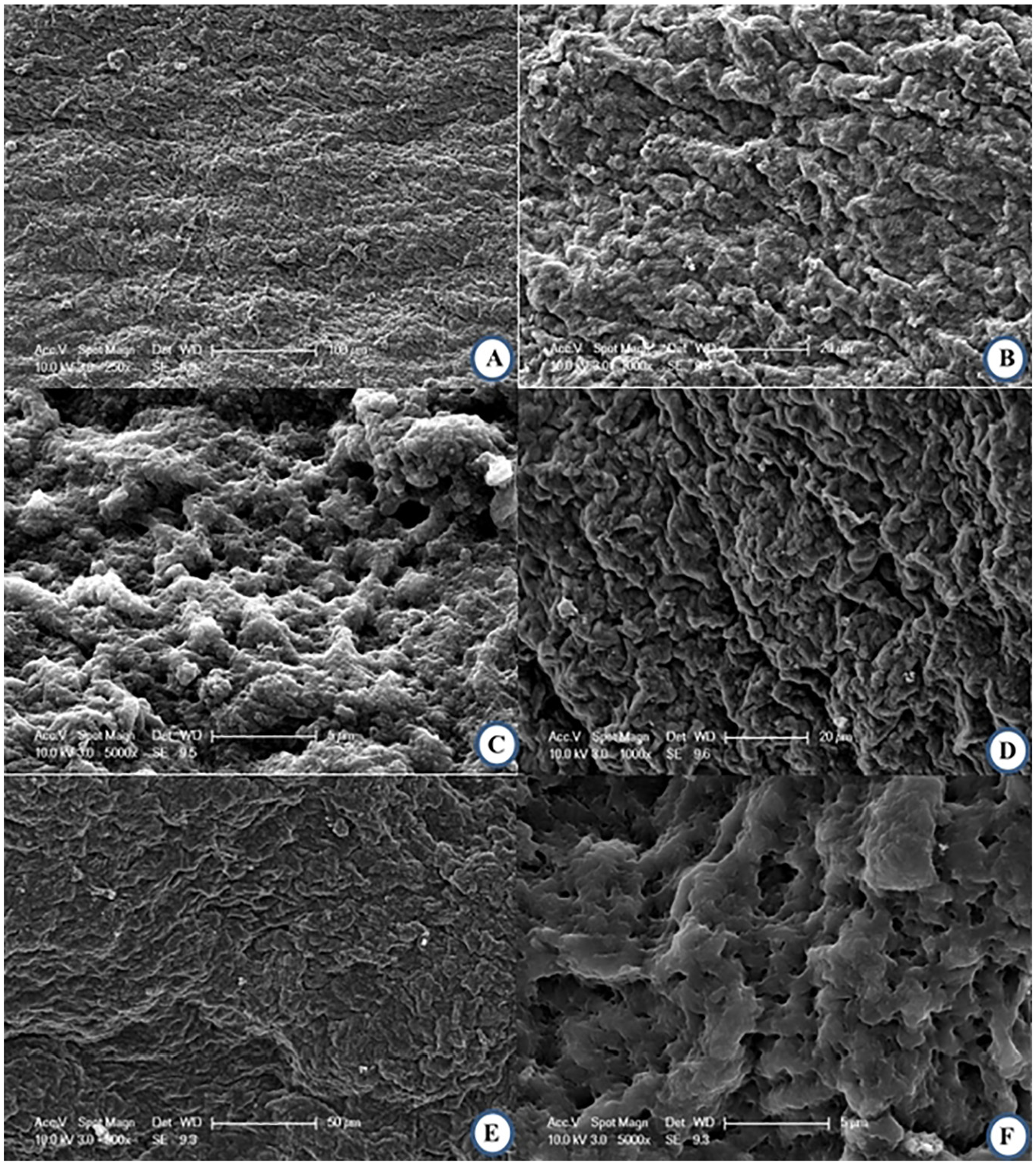
SEM images following testing, all images are of the ventricular or outflow surface. (a) 250× and (b) and 1000× magnification views of the same area on the anterior leaflet, (c) 5000× magnification of the anterior leaflet, (d) 1000× magnification of a wall of the posterior leaflet, (e) 500× and (f) and 5000× magnification of the same area on the posterior leaflet.

SEM images following fixation and testing, all images are of the ventricular or outflow surface. (a) 250× and (b) 1200× magnification views of the same area on the anterior leaflet, (c) 5000× magnification of the anterior leaflet, (d) 1000× magnification of a wall of the posterior leaflet, (e) 500× and (f) 5000× magnification of the same area on the posterior leaflet.
Discussion
This study has performed an acute, initial, assessment of using glutaraldehyde cross-linked porcine hearts under systolic loading. The cross-linking procedure has altered the nature of collagen fibrils for reinforcing the mitral valve tissue, subsequently affecting its mechanical behaviour. Also, the acute rise in peak-pressure is reached more slowly and to a lower value for the cross-linking procedure. However, peak-pressures remained within a physiological range. The increased time-scales, in response to pressure-based loading, appeared to relate to a more immobile valve. This study has also demonstrated that sclerosis is exhibited following cross-linking.
Collagen fibrils provide tensile reinforcement to extracellular matrix in the tissue, by withstanding the force that tends to extend and break the tissue, when the tissue is loaded under tension. 28 The lower peak pressure exhibited by glutaraldehyde cross-linked tissue may arise from an increased randomness in the fibril orientation. Kayed et al. have found that glutaraldehyde cross-linking alters the collagen fibril network organisation resulting in a more randomly orientated fibril network as compared to untreated tissue. 29 Clearly, cross-links act to constrain the fibrils and link them together mechanically. 29 In general, for a given applied stress, the fibrils will increasingly align in the direction of the applied force, therefore, and recruit into tension. 29 Collagen in the central body of the anterior leaflet is aligned circumferentially, 30 presumably to enable the anterior leaflet to expand radially during the early stages of systole. In doing so, the mitral valve covers the annular orifice which enables blood pressure to build up within the left ventricle without mitral regurgitation. 5 We, therefore, expect that a greater applied force, acting in a radial direction, would be necessary to lead to the same anterior leaflet strain. The following cross-linking deformation would be resisted by the fibrils which are aligned in that direction (reducing the rate of pressure build up due to mitral regurgitation). By an analogy to fibre reinforced composites, 31 the off-axis stress, σf, in the tissue may be modelled by σappcos 2 θ, where θ is the orientation of the fibrils and σapp is the stress applied to the tissue. By this definition, the σ1/σapp takes on a value between 1 and 0 where the upper limit implies that all the fibrils are oriented in the direction of the applied stress and the lower limit implies that all the fibrils are perpendicular to the applied stress. Owing to the varying orientation of the fibrils in a tissue, a proportion of the fibrils may not be recruited into tension; it has been observed that the spread of the orientation angles of the non-recruited fibrils (i.e. in the direction with respect in the applied stress) is larger in glutaraldehyde cross-linked tissue as compared to the untreated tissue, for a given strain. 29 We, therefore, expect σf/σapp to be larger in the untreated tissue as compared to the glutaraldehyde cross-linked tissue, with the exception of radial alignment (which would have tended towards 0 in the untreated valve). The natural randomness in the orientation of the fibrils in the tissue is exacerbated considerably by glutaraldehyde cross-linking, hindering the valve's capacity to enable pressure to build up behind the valve.
Decreased leaflet mobility was observed through the slower closing of the valve. In essence, the leaflets appeared to become stiffer. This is consistent with some previous studies demonstrating that valvular tissue stiffness increases after treatment with glutaraldehyde.10,32 The decrease in tissue compliance, 32 increase in internal shear properties under low strain rates 10 and decrease in extensibility 33 were all cited as effects of glutaraldehyde fixation. However, there is also contradictory evidence in literature which suggests that cross-linking actually reduced stiffness20,33 and reduced the ultimate tensile strength of chordae. 9 Mechanical testing of chordae tendineae has been used to assess how glutaraldehyde cross-linking alters their material properties and is presented in full in Part-2 of this study. 19
SEM imaging revealed the loss of endothelium on the surface of the fixed valve leaflets exposing subendothelial fibrils. This is similar to the damage that occurs during rheumatic heart disease 34 and is in agreement with imaging from previous studies of glutaraldehyde-fixed valves. 14 From SEM images, the entanglement of fibrils on the surface of the fixed anterior leaflet and broken collagen fibrils can be observed on the posterior leaflet. This is likely due to the mechanical forces exerted on the fixed valve during motion, thus causing the fibrils to break as the cross linking of the collagen by glutaraldehyde has caused an increase in flexural stiffness making the fibres more susceptible to injury. 12
Fibrils do not normally rupture when subjected to pressures within physiological conditions. So why did the fibrils rupture in the endothelium of the glutaraldehyde cross-linked tissue? One plausible explanation is as follows. Because a large proportion of the fibrils did not completely achieve alignment, they would not be as effective for load-bearing as compared to the proportion of fibrils which are aligned in the direction of the applied stress. Thus, the aligned fibrils have to bear higher load in the glutaraldehyde cross-linked tissue compared to the untreated tissue, for a given strain. Kayed et al. have observed that the latter fibrils also sustain larger strain compared to the former, as pointed out in a previous paragraph. 29 Consequently, these fibrils in the glutaraldehyde cross-linked tissue break when the magnitude of the stress in the fibril reaches the fibril rupture strength. Kayed et al. have found that the fracture stress of glutaraldehyde cross-linked tissue was lower than untreated tissue. 29
The results obtained demonstrate that glutaraldehyde cross-linking of porcine hearts may be of potential value in assessing improved bioprosthetic mitral valve replacements; therefore, there is potential to use the methodology presented as a tool for clinical assessment. However, a key area of concern is the reduced leaflet mobility. It is presumed that during diastole, reduced mobility could limit blood inflow and potentially increase pressure gradients. Another area of concern is the increased loss of endothelium. In our initial study, valves were assessed using an acute model (i.e. it does not assess long-term effects). Further assessment would require cyclic loading, which would only be expected to increase damage to the endothelium of valve leaflets. Indeed, extending existing computational models of mitral valves35–37 may enable better use of in silico assessment in aiding the development of a true bioprosthetic mitral valve replacement.
Limitations
There are several limitations worth identifying within the presented study. Our initial focus has been on the acute behaviour and understanding of valve mechanics following cross-linking with glutaraldehyde; which has been feasible despite a low sample size. All assessment was acute, though, with only 10 loading cycles performed per mitral valve. During previous studies, with similar sample size and loading regimes, statistical differences in peak pressure have been identified due to a given experimental variable (e.g. annular size, chordal rupture).18,38 However, longer-term performance has not been assessed and our results are limited to acute performance. For example, our observation of endothelial denudation highlights damage to the valve following acute testing, and this finding has uses in guiding hypotheses which could then be tested through longer-term, or in vivo, experimentation. However, it is not possible to infer directly from such acute observations how longer-term testing would alter the endothelium in vivo.
To enable direct observation of valve mechanics, and distinguish more easily between a peak pressure and a stabilised pressure, loading cycles of 10 s were used. A more physiological pressure would increase the clinical relevance, though it would also make clear observations more challenging. Some of these observations, such as on leaflet motion, would benefit from objective and quantitative measures not used during this study. In this study, the application of pressure was achieved using water rather than blood. This approach again limits certain physiological implications, however, the loading regime focused on increasing a hydrostatic pressure which is independent of the viscosity of the fluid. Mitral valves were held in place within the test apparatus by suturing the annuli on to rubber sheeting. The valve was sized against a fixed Duran annuloplasty ring. The advantage of using this approach was that it eliminated the annular diameter as an experimental variable, within our experimental set-up; the disadvantage of this approach is that it is a non-physiological condition.
The longer-term development of a mitral valve as an alternative to existing replacement devices is challenging. A key challenge, if the issues of cross-linking are overcome, is the shape and positioning of papillary muscles within the ventricle. Currently, the authors are unaware of a device which aims to mimic their placement, and for this to be possible technology would be necessary which enabled their placement without increasing the surgical complexity involved.
Conclusion
Glutaraldehyde cross-linking of porcine mitral valves may be of potential value in assessing improved bioprosthetic mitral valve replacements. Using an acute in vitro model physiological pressures were achieved. However, areas of concern include leaflet immobility and increased loss of endothelium.
Footnotes
Acknowledgements
The authors would like to thank Mr Paul Stanley and Mr Peter Thornton for technical assistance with sample preparation for SEM imaging and technical modifications to the experimental rig, respectively.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: H.E.B. was funded through an Engineering and Physical Sciences Research Council scholarship [M114612B]. This study was partly funded by an Innovation and Research Award from the Institute of Physics and Engineering in Medicine. The materials testing equipment used in this study was funded by an Arthritis Research UK Grant [H0671; now a part of Versus Arthritis].
Statement involving human and animal rights
This study did not involve any human participants or animal studies, and no animals were sacrificed specifically for this study. It is noted that animals from which porcine hearts obtained were otherwise destined for the food chain. Ethical approval was granted for this study by the University of Birmingham Research Support Group [ERN_15-0032].
