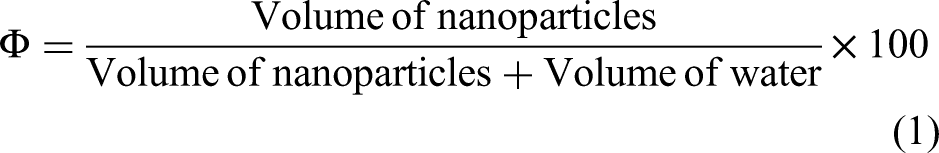Abstract
Nanofluids are well known for their enhanced thermal properties. In spite of their excellent properties, there are certain hindrances to their applications on large scale. The issue of nanoparticle agglomeration in the base fluid with the consequent stability related issues is one of the main obstacles to the usage of nanofluids. Stability is crucial because the longer the nanofluids remain stable, the better their capacity to retain their thermal properties. Hence there is a need to evolve long-term stable nanofluids. Since there are a lot of factors, which are affecting the stability of the nanofluids, there is a need to optimize the process parameters. In this regard, central composite rotatable design (CCRD) was applied in this study to optimize the independent parameters of stability of ferric oxide nanofluids. For this, the performance of nanofluids was assessed by measuring the nephelometric turbidity units (NTU), based on the independent variables such as percentage of intensity of the nanoparticle, pH of the base fluid, and percentage volume concentration of the surfactant. All the parameters that are affecting individually and mutually were validated statistically using analysis of variance (ANOVA). A regression equation to evaluate the NTU was developed. The obtained results showed that the values predicted by the model and that obtained from the experiments were in good agreement with each other. It is observed that more than 99.65% of the variation could be predicted by the model developed for NTU. The response surface methodology (RSM) has revealed that the ideal process parameters for greater stability of nanofluids are 0.01% particle volume intensity, pH 3.2, and 0.6% surfactant intensity.
Introduction
Nowadays, developing technologies for high-performance heating and cooling applications has been the foremost priority of industries, buildings, and other energy-consuming sectors primarily to curb the energy demand and environmental footprint which is relevant from the perspective of low or zero carbon economy future. 1 The research on nanosized particles, especially metal and metal oxide-based ones, has emerged as one of the core areas of research nowadays due to their widespread applications in several fields of science and technology. 2 Nanofluids find applications in heat exchange processes such as refrigerators, heating, ventilation, and air conditioning (HVAC), automobile radiators, high-power lasers, electronic cooling, solar and nuclear systems, biomedical systems, and gas recovery systems, etc. 3 Over the past few years, a range of augmentation techniques have been developed to improve the heat transfer in heat exchange processes. Some of them include the use of treated, rough, and extended surfaces, insertion of displaced augmentation, swirl flow, surface tension appliances, additives of liquids and gases, usage of mechanical supports like vibration on surfaces, vibration over fluids, and electrostatic fields. 4 Existing techniques can be categorized into active, passive, and compound methods. 5 Dispersion of nano-sized particles less than 100 nm size into the carrier fluids has a profound technological implication for heat transfer. Studies on various aspects of the nanofluids like mechanism of stability, thermal, rheological, and morphological properties, and convective transfer of heat showed that nanofluids exhibit enhanced thermo-physical properties in a wide range of applications. 6 Owing to these superior properties, nanofluids have garnered the interest of researchers globally in the field of heat transfer in recent times. However, lack of sufficient data for the same set of conditions on the transport and convective behavior of nanofluids hinders their applicability. Lack of conception of the mechanisms to predict the transport and convective behavior of nanofluids is another hindrance. 7 Rheological features rely on numerous variables such as size, shape, type, intensity of nanoparticles, surfactant type and the intensity of the carrier fluid, etc.8,9 Various parameters such as particle size, shape, properties, temperature of the carrier fluid and concentration affect the thermal conductivity of the fluid. 10 Augmentation in the concentration of the nanoparticles causes agglomerations. Iron oxide nanoparticles augment the transfer of heat of the nanofluids under the magnetic field. 11
Different parameters influence the stability of nanofluids that include the type of surfactants and their concentration, sonication time, adjusting the pH of the carrier fluid, concentration of nanoparticles, size and shape of nanoparticles, dispersant used, etc.
Numerous research works have been reported studying the effect of type of surfactant used on the stability of the nanofluids. Ali et al. 12 employed different surfactants like sodium dodecyl sulfate (SDS), cetyl trimethyl ammonium bromide (CTAB), tannic acid and ammonia solution, dodecyl benzene sulfonate, and sodium deoxycholate. From their findings, it is known that the surfactants CTAB and tannic acid + ammonia solution resulted in a longer stability period comparatively (more than two months) with the ultrasonication mixing time being maintained at six hours. However, the addition of higher amounts of surfactants (>0.5 mL) may lead to reduced thermal conductivity with a marginal increase in viscosity and density. Wang et al. 13 investigated the use of a variety of surfactants on the stability and thermal properties of different nanofluids consisting of Ferric oxide-water, carbon nanotubes (CNT)-water, and Ferric oxide-CNT-water within a temperature range of 278–313 K. The surfactants used were trisodium citrate dihydrate, polyvinyl pyrrolidone, cetyl tri methyl ammonium bromide, tetra methyl ammonium hydroxide (TMAH), acacia senegal, sodium dodecyl benzene sulfonate, sodium dodecyl sulfate (SDS) and sodium lauryl sulfate (SLS). They concluded that the addition of surfactants resulted in better stability and increased thermal conductivity. Ouikhalfan et al. 14 prepared surface-modified titanium dioxide (TiO2) nanoparticles by treating them with surfactants and dispersed them into distilled water. The treated nanoparticles were stable for 2 weeks, and in addition to that thermal conductivity also increased.
The stronger attraction between the particles makes the nanoparticles tend to form agglomeration, leading to different sizes and surface treatments of each particle as per the application field to which they are prepared. 15 The traditional techniques of dispersion such as ball milling and jet milling due to their higher milling speeds contaminate the dispersion, and their dispersion ability is also comparatively lower. To address this issue, ultra-sonic dispersion can be a potential solution that can control aggregation. 16 The supersonic waves can be effectively used to attain the suspension of the nanoparticles in the carrier fluids. The effect of ultrasonication is to break down the large agglomeration of nanoparticles into smaller ones thereby ensuring the stable suspension and more uniform distribution of nanoparticles to the base fluid.17–19
In recent times metal-oxide-based nanomaterials such as TiO2, ZnO, and SnO2 are being given considerable importance in the research for systhesis to prepare nanomaterials with controlled size and shape to ensure better stability. 20 The factors such as ultrasonication time, power, and ultrasonication device not only influence the stability of nanofluids but also influence their thermal properties as well.21,22 Thus in order to achieve better stability, the determination of optimum ultrasonication time is essentially important. Asadi and Alarif 23 showed that extending the sonication time up to 60 min is found to be effective in improving the stability of the nanofluids, which, however, deteriorated beyond 60 min. The ultrasonic horn/probe devices are found to be better than ultrasonic bath devices. 24 Huang et al. 25 studied the stability, thermal conductivity, and viscosity of hybrid nanofluids consisting of SiC-MWCNT/Oil. From their findings, it was known that a dispersant content of 3 wt% could result in better stability of nanofluids. Moreover, the thermal conductivity of hybrid nanofluid enhanced by 22.58% with a volume concentration of 1% at 80°C.
Xian-Ju and Xin-Fang 26 studied the effect of pH on the thermal conductivity and viscosity of Cu-water and Al2O3-water nanofluids. They confirmed that at an optimal value of pH, the increase in thermal conductivity of both nanofluids Cu-water and Al2O3-water was 15% and 13%, respectively at 0.4 wt.% when compared to the base fluid without nanoparticles. Goudarzi et al. 27 experimentally verified the effect of adjusting the pH of nanofluids CuO-water and Al2O3-water with 0.1 wt.%, nanoparticle each, in a solar application. They used sodium dodecyl sulphate as a surfactant for improving the stability of the nanofluids. It was observed that when compared with the pH of 10.5, pH of 3 (acidic condition), the CuO-based nanofluid enhanced the solar collector performance by 52%, on the other hand, with Al2O3-water nanofluid, the enhancement was 64.5% at a pH of 10.5 as opposed to that of a pH of 9.2 (basic condition). Umar et al. 28 synthesized two nanofluids Cu-water and Al2O3-water using the surface charge modifiers NaOH, NH4OH, and acetic acid. They reported that the stabilization pH value of Al2O3-water nanofluid is 4.59 (acidic condition) whereas CuO-based nanofluids exhibited poor stability in comparison with that of Al2O3-based ones under similar conditions. They conclusively reported that better stability could be observed either at a relatively low pH value of nanofluid or pH higher than the isoelectric point.
Chen et al. 29 used cationic chitosan as a dispersant to study its effect on 15 nm Al2O3-water nanofluid's thermal conductivity, dispersion, and suspension. The two-step method was employed in this study to synthesize the nanoparticles. It was shown that 0.05 wt.% dispersant could increase the thermal conductivity of 1.0 wt.% nanofluid by 4.1% and 12.6% at temperatures of 15°C and 55°C, respectively. The dispersant can potentially improve the dispersion and suspension and in addition, it can reduce the tendency of nanoparticles to stick to the pipe walls. Cationic chitosan was also employed by Hung et al. 30 as a dispersant in a heat pipe application that used Al2O3-water nanofluid. There was an enhancement of 22.7% to 56.3% in working efficiency as opposed to that of substrate solution. Zhu et al. 31 considered sodium dodecylbenzene sulfonate (SDBS) dispersant in Al2O3-water nanofluid to study the influence of suspension behavior at different pH values. The enhancement in the thermal conductivity of nanofluid was about 10% at the nanoparticle weight concentration of 0.0015%. Wusiman et al. 32 used two different surfactants namely SDBS and sodium dodecyl sulfate (SDS) in an aqueous solution of multi-walled carbon nanotubes (MWCNT). They found that nanofluids showed better dispersion and stability using two surfactants. Thermal conductivities of the nanofluids showed a reduction with an increase in the concentration of either of the surfactants. The thermal conductivity of MWCNT nanofluid with surfactant SDBS was higher than that with SDS.
Classical approaches can be utilized for optimizing the parameters that affect the stability of the nanofluids. However, they have been identified as time engrossing and cost-associated approaches. Moreover, these methods are not very efficient in determining quadratic terms and introducing curvature into the response.33–36 The predictive models developed should be accurate enough to predict accurately the properties such as density of nanofluids so that the design of heat exchange devises becomes easier and faster. 37 Basir et al. 38 carried out sensitivity analysis of cobalt-CeO2/kersone hybrid nanofluid using RSM method. They found that the rate of heat transfer and coefficient of skin friction reduce with increase in the melting parameter. The developed model can be useful in applications such as nano-based microscopic propulsion and electromechanical systems.
Ali et al., 39 in their review on stability of nanofluids, have discussed about the challenges that exist in the preparation of nanofluids, stability evaluation mechanisms and stability enhancement procedures. They stated that if nanofluids have longer settling time, it can also conserve its thermal conductivity for a longer period. Uniformity in particle suspension depends on the type of method of preparation of nanofluid and this will have an effect on the thermo-physical properties of the nanofluid. Two methods are generally used for the preparation of nanofluids: one-step method and two-step method. The most popular method is the two-step method. With this method, smaller quantities of the nanofluids of any type can be prepared. But the problem that is associated with the two-step method is agglomeration. In order to avoid this problem, researchers came up with another method, which is called one-step method. Though this method can effectively minimize agglomeration, the disposal of contaminants that are formed during the process is a drawback.
Xia et al. 40 carried out experimental investigation to study the effect of surfactant on stability and thermal conductivity of Al2O3 nanofluids. They reported that the surfactant can have negative influence on the base fluid. The longer the alkyl chain length, the lower the thermal conductivity. Nevertheless, the addition of surfactants significantly improves the stability of Al2O3/de-ionized water nanofluids.
Hung et al. 41 studied the dispersion behavior of water-based Al2O3 and copper nanofluids. They measured the zeta potential and absorbency of the nanofluids under different pH values. They reported that the stability of the nanofluids is highly dependent on pH. Also, an optimal pH value of the base fluid can result in the highest stability of the nanofluids.
From the literature, it was found that many factors such as sonication time, pH of the base fluid, % weight of the surfactant and nanoparticle volume concentration play an important role in the stability of the nanofluids in terms of zeta potential. Optimum quantities of these factors are required for getting stable nanofluids that prevail for a longer time. Out of these parameters, pH of the base fluid, % weight of the surfactant, and nanoparticle volume concentration are the predominant factors. Many research works aimed at evaluating the effect of these parameters individually on the stability of the nanofluids in terms of zeta potential. But in reality, the mutual effects of these parameters on the stability of the nanofluids have to be taken into consideration to ascertain how the stability is improved without compromising the thermo-physical properties of the nanofluids. Thus there is a necessity to study the individual effects of these parameters and their mutual effects on the stability of the nanofluid. In view of this, this work attempts to determine the individual and mutual effects of these parameters on the stability of the nanofluid by using response surface methodology (RSM) approach.
There are several statistical techniques for optimizing the output responses like Taguchi, Response surface methodology, Artificial Neural Networks (ANN) etc. Each has its own advantages and disadvantages. Taguchi method is preferred when the experiments are to be carried out are less. But when the goal is to attain the global maxima, and experimentation is not a constraint, then RSM is preferred over Taguchi. Also, RSM is preferred for such situations where large amount of information can be obtained from limited number of experiments. If large information is available, then ANN techniques are quite useful. Herein RSM is used to find the optimal parameters that lead to the maximum stability of the nanofluid in terms of NTU. For this, the parameters considered for modeling and optimization are particle volume concentration, pH, and concentration of the nanofluids. The combined effect of various factors like concentration of the nanofluid, weight percentage of the surfactant, and pH of the base fluid using RSM is studied. In addition, optimization of the process parameters which affect the stability of the nanofluids is also carried out.
Preparation of ferric oxide nanofluids
Ferric oxide, nanopowder of reagent grade with a size of 100–200 nm, was purchased from Hychem Laboratories, Hyderabad. The two-step method was employed for preparing the nanofluids. In this method, Nanoparticles were dispersed in to 100g of the base fluid (water), and mechanical stirring was carried out for 3 hours. Double distilled water was used to prepare nanofluids to ensure the purity of the base fluid. Nanoparticles were dispersed into 100 g of the base fluid (water), and mechanical stirring was carried out for 3 h. During this process, sodium lauryl sulphate (SLS) (1/10th of the weight of the nanoparticles) is added as a surfactant to the solution in proper proportions to avoid the formation of agglomeration (formation of sedimentation of the particles in the carrier liquid). Since there is a massive difference between the density of ferric oxide nanoparticles and the double-distilled water, particles are to be prevented from settling in the water.
Under atmospheric conditions, the particles form loose clusters of size ranging from micrometers to nanometers. However, they can be quite successfully dispersed in the liquid by breaking the groups by sonication. The solution was stirred in an ultrasonic bath sonicator for about 2 h. Clustering of the nanoparticles is avoided by inducing a charge on the surface of the particles. It can be obtained by adjusting the pH of the carrier liquid. The base fluid's pH is adjusted by adding a small amount of hydrochloric acid. A pH meter is used to monitor the pH of the base fluid. At various pH levels of the base fluid, nanofluids were prepared. The particle volume concentration of the ferric oxide nanoparticles was estimated using the equation below.
Weights of the ferric oxide nanopowder and surfactant at various particle volume concentrations.
Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) analyses are carried out to confirm the morphology of the ferric oxide nanoparticles. Photographic views of SEM and TEM of ferric oxide nanoparticles are shown in Figure 1(a–b). SEM and TEM results depict that the particles have spherical shapes. XRD (X-ray diffraction: Siemens, D-500, 45 kv, and 40 Ma) was utilized to study the phase characterization of synthesized ferric oxide nanoparticles. Figure 2 shows the XRD pattern of ferric oxide nanoparticles. As can be seen from Figure 2, the nanoparticles comprise magnetite and oxygen as the constituents. The diffraction peaks are extended since the size of the crystallites is ultra-low.

(a-b). SEM and TEM photographs of Fe3O4 nanoparticles.
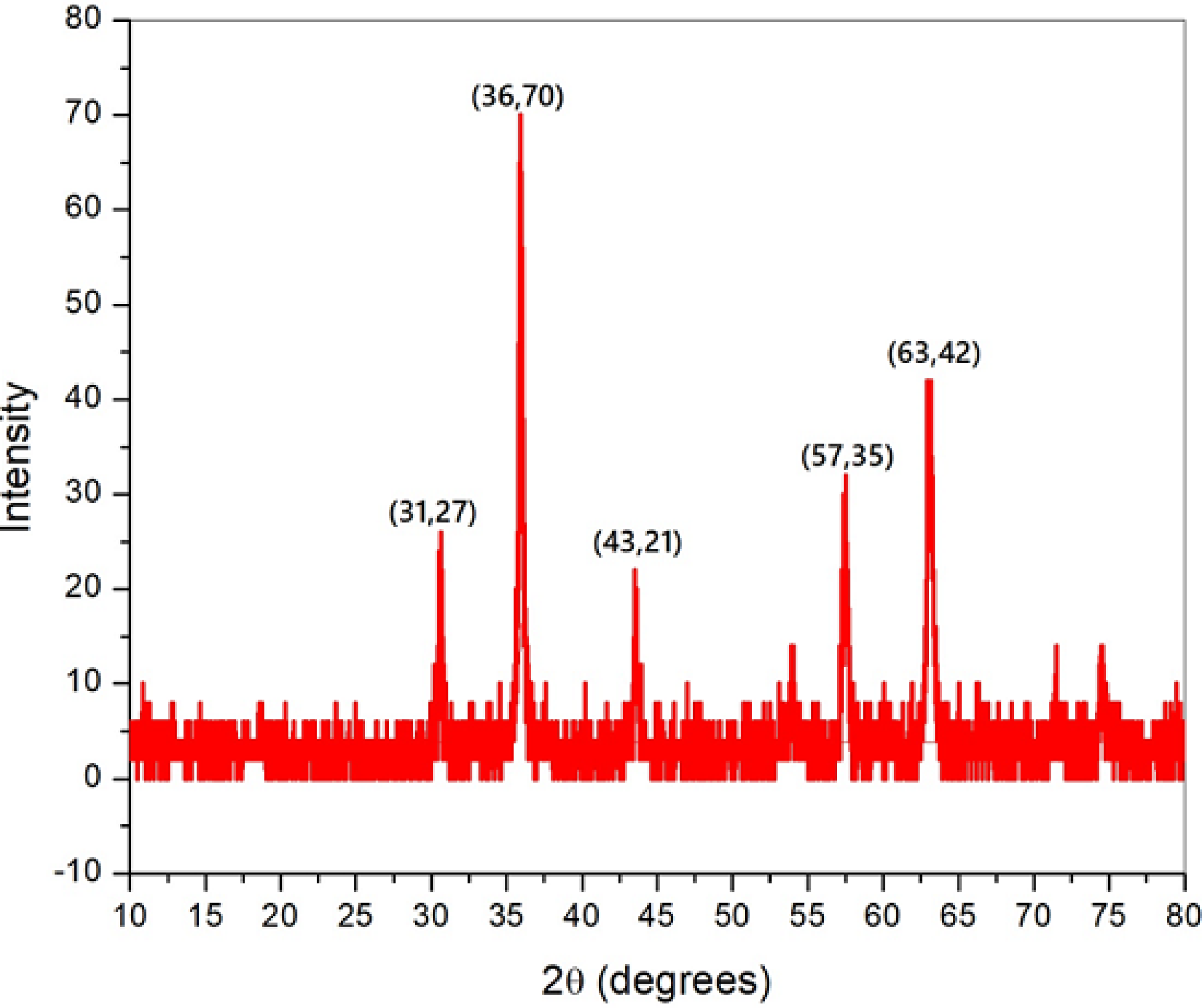
XRD pattern of synthesized Fe3O4 nanoparticles.
The diffraction peaks found were indexed using cubic structure of ferric oxide (JCPDS no.19-629) to enable high phase purity for the magnetite. EDX is utilized to examine the presence of Fe and oxygen in the nanopowder and is displayed in Figure 3. From EDX, it is found that oxygen constitutes 63.99% while Fe is 36.01%. The average size of the crystallite is determined using the Scherer equation
42
given below
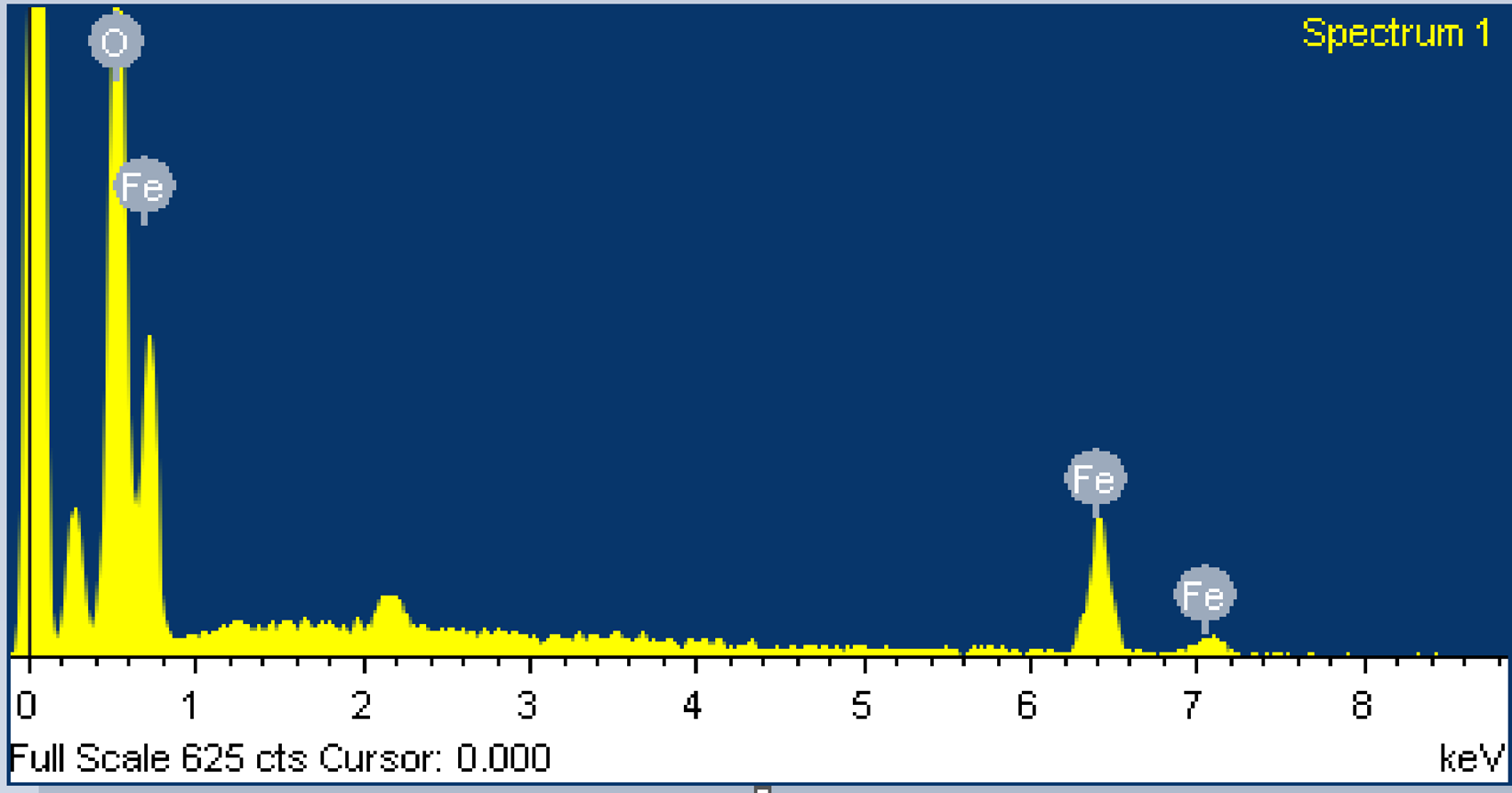
EDX pattern of Fe3O4 nanoparticles.
The average size of the nanoparticles was estimated from the data obtained from the XRD analysis and Scherer equation. The average size of the crystallite is 26.73 nm. FWHM is 0.3263 and theta is obtained as 35.940. Ferric oxide nanofluids of various combinations of concentrations and surfactants are shown in Figure 4. The dispersion stability of the particles in the nanofluid is represented in terms of turbidity. Turbidity is measured in terms of nephelometric turbidity units (NTU). NTU is measured by using a nephelometer. A Tungsten filament source is used for this purpose. It is measured by the reflection of light from the fluid sample. Initially, double deionized water was filled in the cuvette of the nephelometer and it is placed in the test chamber. NTU value is set to zero. Then the prepared nanofluid was filled in the cuvette of the nephelometer. It was placed in the test chamber and left undisturbed until a stable reading was obtained. At different volume concentrations of ferric oxide nanoparticles, NTU values were measured. A graph is plotted between particle volume concentration (%) and NTU, which is shown in Figure 5.

Fe3O4 nanofluids of various combinations of concentrations and surfactants.
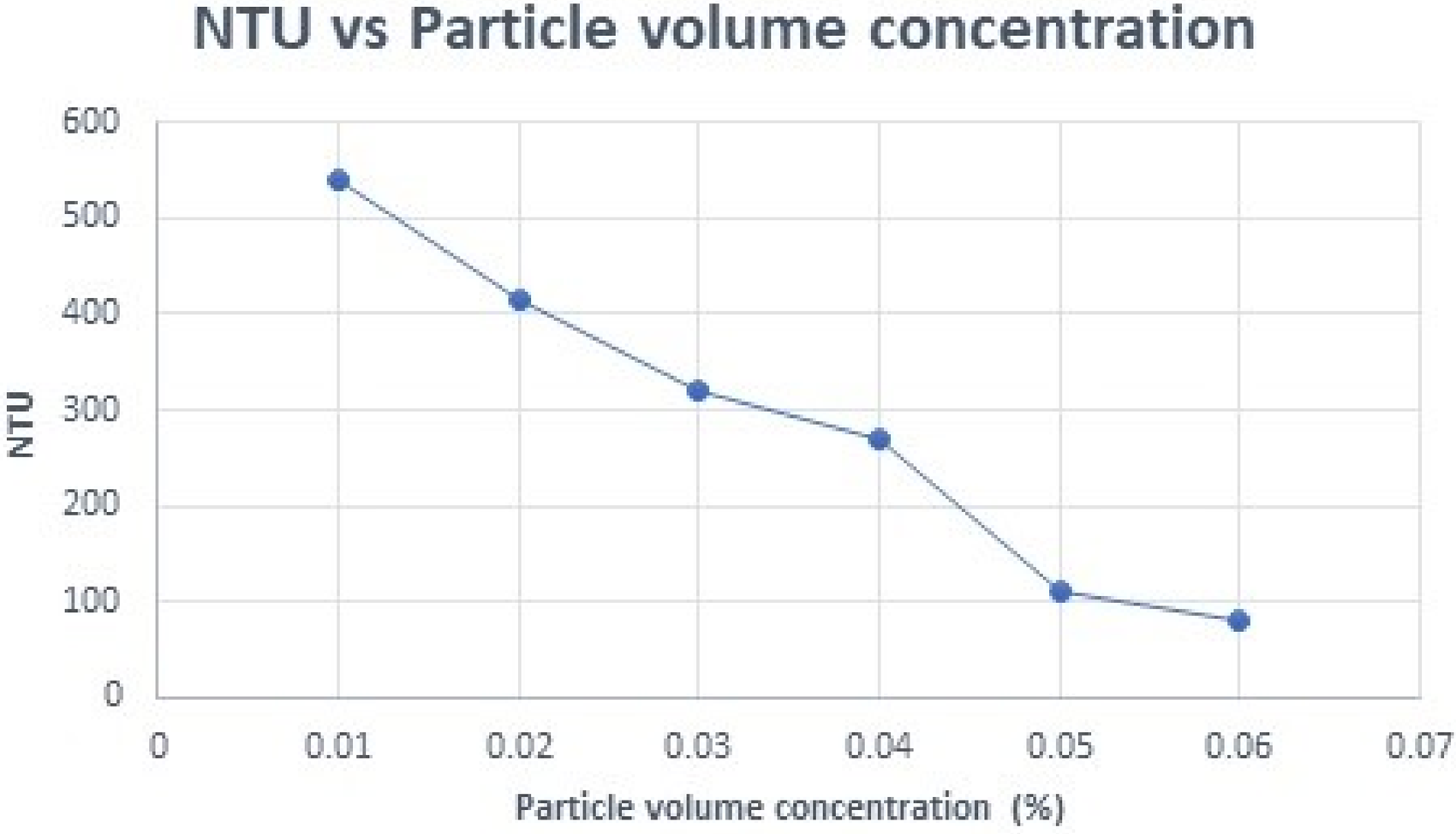
NTU vs particle volume concentration.
Measurements were made without surfactant and immediately after the sample preparation to study the effect of the particulate. The results projected a reduction in NTU with augmentation in the particle volume intensity of the nanofluid, and the relationship is found to be linear. Therefore, to produce stable nanofluids, the effect of various input parameters like particle volume concentration of the nanofluid, pH of the base liquid, and percentage concentration of the dispersant has to be studied.
Response surface methodology
RSM is a widely used mathematical and statistical approach that is based on the design of experiments for the optimization of the process variables since it is possible to evaluate the influence of several parameters and their interactions using one or more response variables.43–45 The main advantage of this method is it could minimize the number of trials and recognize the process variables’ influence on the removal process.46,47 Quadratic terms of two can be solved by considering at least three levels for every individualistic variable. Since the number of independent variables increases up to 3, this process becomes tedious, and experiments are time-consuming. If it involves cost, then this technique is not feasible. Therefore, the central composite design (CCD) of RSM, which uses mathematical and statistical methods, is employed to develop a model that reduces the factors to a 2-level factorial design, thereby simplifying the problem.
In the present analysis, RSM-based CCD was utilized in optimizing the independent process parameters of stability, which are useful in heat transfer applications. Design-Expert software version 12 (stat Ease Inc.) was employed to optimize the input parameters to improve NTU’ response parameters. The selected parameters for this investigation were particle volume intensity (X1), pH of the base fluid (X2), and % intensity of the surfactant (X3). Coded as well as actual data are presented in Table 2. For the factors selected, the quadratic model comprises 23 factors with six axial and eight cubic points. The central composite design representation for three self-reliant variables is shown in Figure 6. A polynomial equation of the second order was used to develop an empirical relation between the input parameters and response variables to optimize the process.
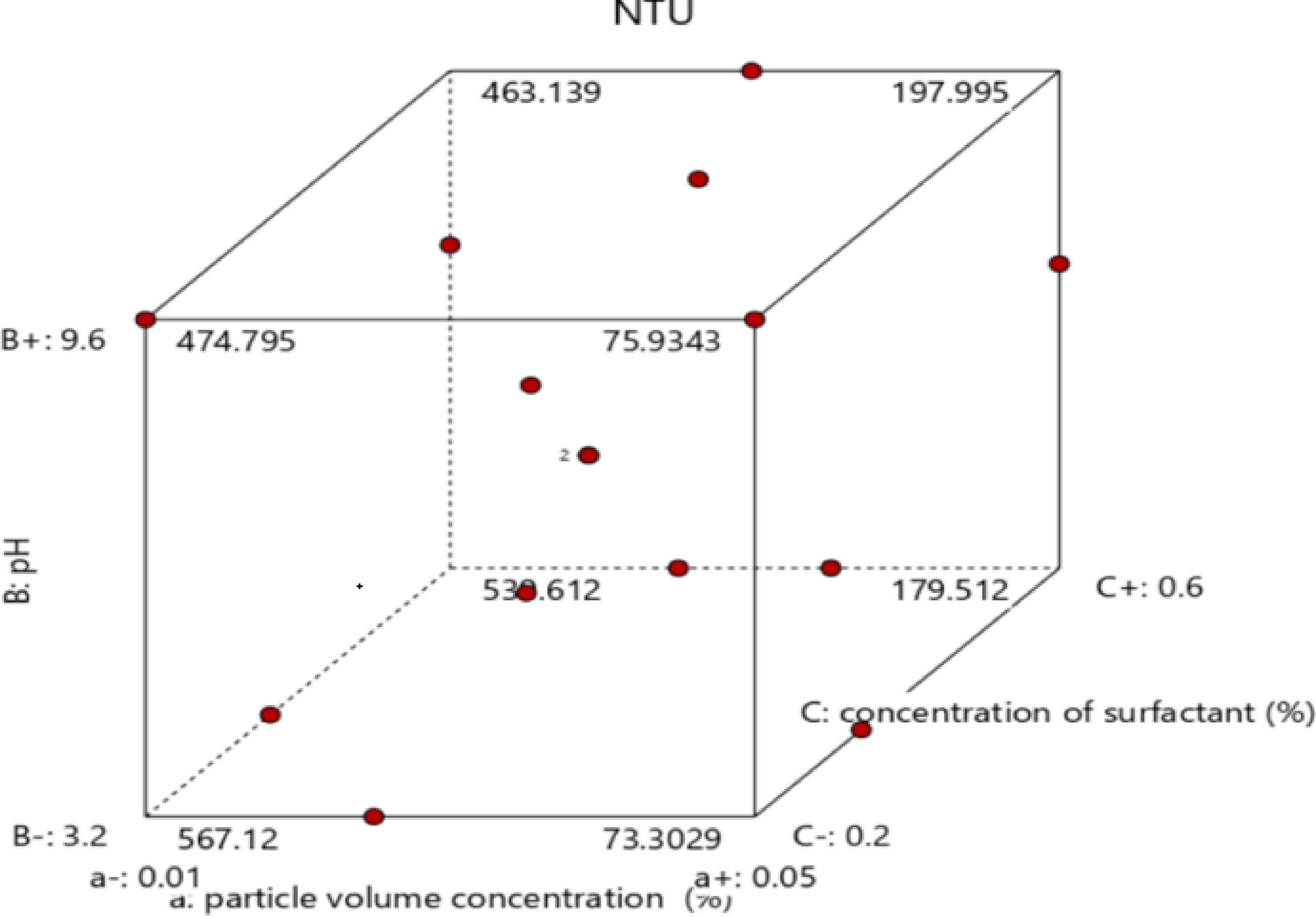
CCD for independent variables.
Selected individual parameters for central composite design.
The mathematical equation that was used in the present study is Identification of the predominant factors which influence the stability of the nanofluids and to find the upper and lower limits of the chosen factors. Development of the experimental design matrix. Performing the experiments as per the design matrix. Analyzing the effect of various parameters on the stability of the nanofluid using Analysis of Variance (ANOVA).
Identification of the predominant factors which influence the stability of the nanofluids and to find the upper and lower limits of the chosen factors
Process parameters that affect the stability of the nanofluid are nanoparticle concentration of the basal fluid, size of the nanoparticles, concentration of the surfactant, pH of the base fluid, stirring time, and ultra-sonication time. Among the above, the nanoparticle concentration of the basal fluid, concentration of the surfactant and pH of the base fluid are the predominant factors. During the process of preparation of the nanofluid, due to the difference in the densities of the base fluid and the nanoparticles, the particles form agglomerations in the base fluid. In order to avoid this problem, there are two ways: one is to induce electrostatic repulsive forces between the nanoparticles and the other is to add surfactants. Electrostatic forces are related to zeta potential which in turn is related to pH of the base fluid. pH limits are chosen from 3.2 to 9.6 for this study. Brownian motion and van der Waals forces result in the formation of clusters in the nanofluid. This can be avoided by the addition of surfactant in the base fluid. Sodium Lauryl sulphate is used as the surfactant. The lower and upper limits for the surfactant are fixed based on the electric double-layer adsorption model with respect to the volume fraction of the nanoparticles. The lower and upper limits for the surfactant are fixed at 0.2% and 0.6%, respectively. Since the nanofluids possess superior properties at low particle volume concentrations, the concentrations are set between 0.01% and 0.05%, respectively.
Development of the experimental design matrix
To optimize the experimental conditions, a three-level central composite rotatable matrix was used. In the three level, there is an intermediate level
One extra point is added at the center to give roughly equal precision for the response. The method of developing such a matrix is shown in Reference 48. Shankar et al. 49 carried out similar analysis using RSM on Al2O3 nanofluids.
Performing the experiments as per the design matrix
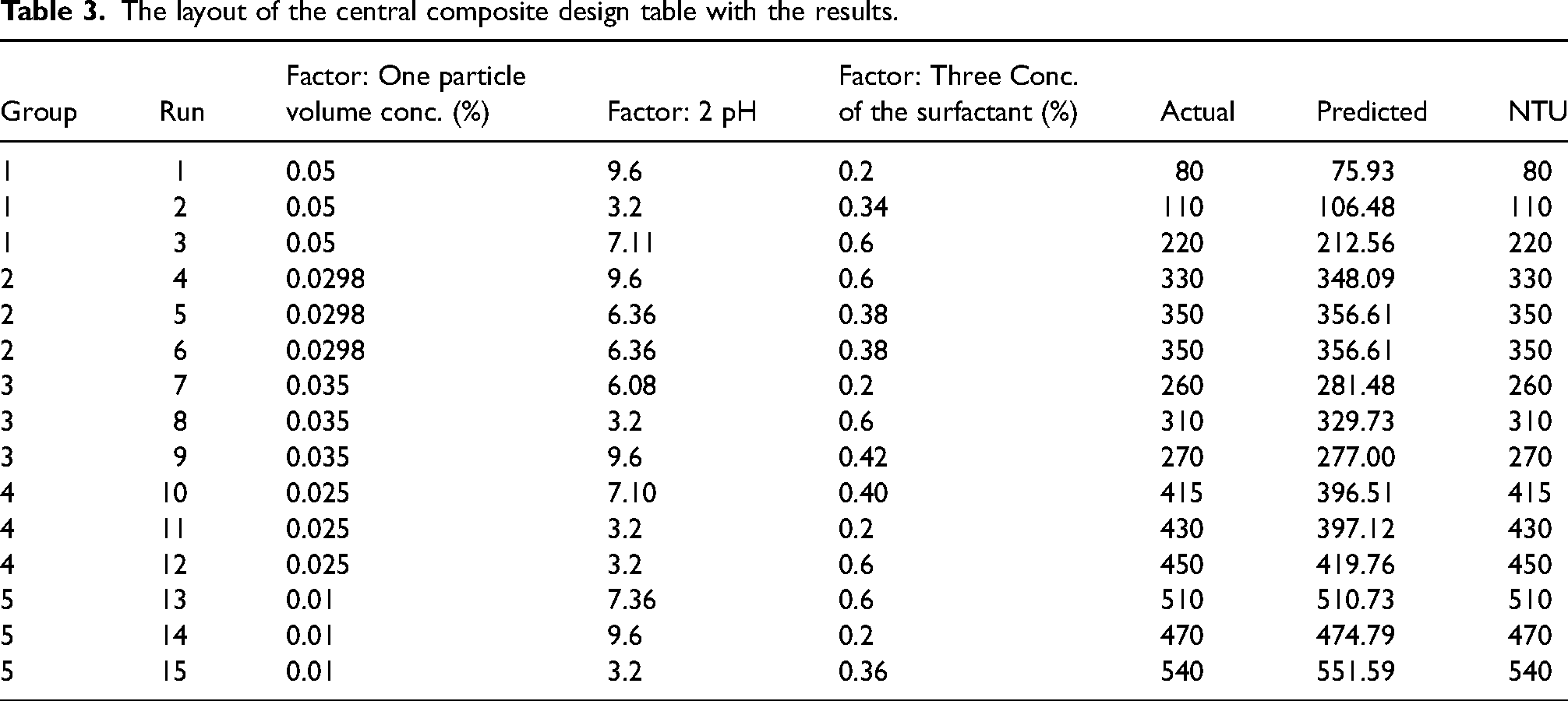
The objective of this study is to analyze the effect of three individual parameters such as particle volume concentration of the nanoparticles, concentration of the surfactant, and the pH of the base fluid as well as the mutual effects of each of the parameter on others. For this, ferric oxide nanofluids were prepared by adding sodium lauryl sulphate as the surfactant. After adding the surfactant and the nano powder, mechanical stirring was carried out for 30 min. This solution was kept in ultrasonic bath sonicator and sonication was carried out for 30 min. pH of the solution was varied by adding hydrochloric acid. All the samples were prepared as per the design matrix and were observed for 20 days. Stability of the nanofluids was measured in terms of NTU. RSM identifies different combinations of process parameters for preparing stable nanofluids. Fifteen different combinations of process parameters are developed by central composite design (CCD). Nanofluids are prepared based on these combinations, and corresponding NTU values are measured and tabulated in Table 3. NTU is considered as a response variable for optimizing the input process parameters by RSM. Schlegel50 studied the optical properties of ferric oxide and found that when incident light energy goes beyond 12ev reflectivity decreases. Thus, turbidity decreases as ferric oxide concentration increases in the fluid. Hence, lower NTU value indicates the larger concentration of ferric oxide nanoparticle in the nanofluid and consequently superior stability of these particles after the nanofluid remains motionless for a given period of time. The present investigation is aimed at optimal parameters for attaining maximum stability. Since the lower NTU, i.e, higher concentration of the nanoparticles results in maximum stability, 0.05% nanoparticle volume concentration and 0.2% surfactant concentration are the optimum values for better stability out of all the combinations considered which is evident from Table 3. It shows that the lower NTU is 80 for the combination of 0.05% nanoparticle volume concentration and 0.2% of surfactant.
The layout of the central composite design table with the results.
Analyzing the effect of various parameters on the stability of the nanofluid using analysis of variance (ANOVA)
ANOVA Table is generated from the CCD-based RSM optimization and is tabulated in Supplementary Table 4. In this optimization, the confidence level is selected as 95%, and the condition is chosen as “more extensive is the better.” The confidence level shows that the P-values are less than 0.05 (5%), which are the most influential parameters on NTU. The P-values of lack of fit of X1, X2, and X3 show that the process parameters individually and mutually influence the NTU. Equation 3 shows the regression equation, which is obtained from the CCD-based RSM optimization. The predicted NTU values are obtained by the regression equation and are displayed in Supplementary Table 5. It clearly shows that there is no significant difference in the measured and predicted NTU values. The relation between different individual coefficients and interaction coefficients is shown as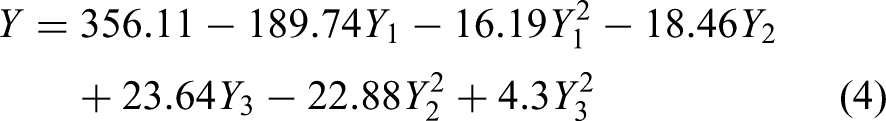
In the randomized designs, statistical plots like probability plots, internally studentized plots over the independent variable play a vital role in analyzing the errors in a normal distribution, model's acceptability, and scattering the observations in random order. The normal distribution of the probability is shown in Figure 7.
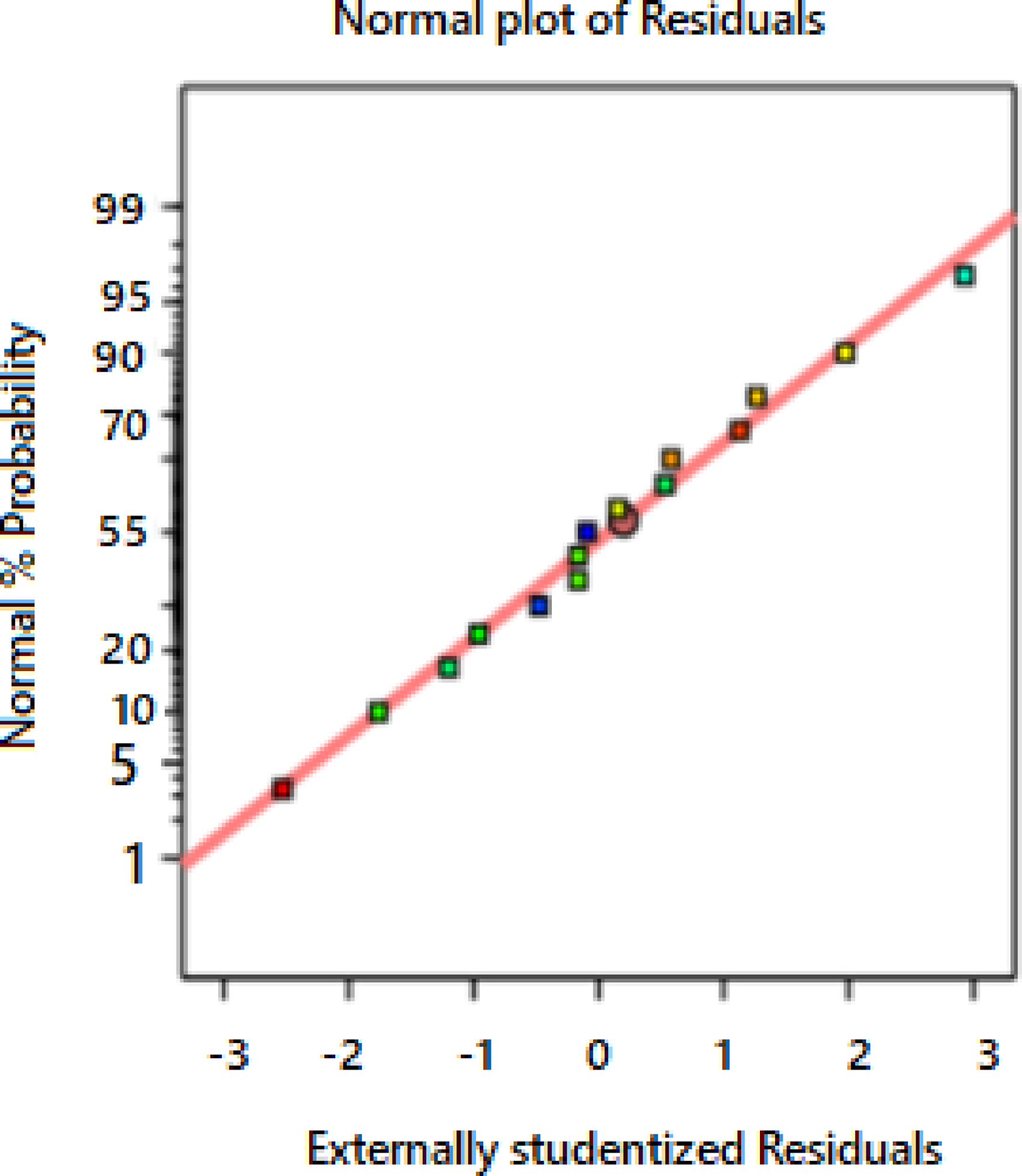
Normal probability plot.
The residuals are very close to the slope, and it clearly shows no significant variation in the residuals. The constant variance for every independent variable error is verified for the different individual variables by arranging the residuals over variables such as predicted values, time, and the model. Supplementary Figure 8 shows the studentized residuals over the expected responses. Since there is no concatenation of positive and negative forms or model of a regression equation, the model chosen is correct. The residuals over another independent factor are also verified (not displayed here). It is found that over a constant range, the variance is constant.
Results and discussion
Effect of concentrations of nanoparticle volume and surfactant
The effect of concentrations of nanoparticle volume and surfactant on NTU is shown in Supplementary Figure 9. The stability reduces with the augmentation in both particle volume and surfactant's intensities. Due to the augmentation in the particle volume intensity, the clustering of ions shoots up. This results in a decrease in the zeta potential, which in turn reduces the suspension capability of the nanofluid. The decrease in zeta potential is due to particle aggregation and flocculation of the nanoparticles, and the reason for flocculation is van der Waals forces. With the enhancement in particle volume intensity, particle aggregation enriches. However, with the increase in the concentration of the surfactant, both the surface tension of the liquid and surface energy reduce, resulting in the dispersion stability enhancement of the nanoparticles in the base fluid. Thus there is a decrease in the aggregation. This results in the enhancement of the nanofluid's dispersion stability. As the particle volume concentration and concentration of the surfactant are increased, the NTU rises from 100 to 400. This indicates an increase in the stability of the nanofluids.
Effect of ph and particle volume concentration
The mutual effects of pH and the particle volume concentration against the NTU are shown in Supplementary Figure 10. As there is an enhancement in the particle volume intensity of nanoparticles and pH of the base liquid, there is a reduction in the NTU of the nanofluid. As the particle volume concentration and pH are increased, the tendency of NTU is observed to be decreasing from 500 to 200. This demonstrates that the stability decreases with the increase in pH and particle volume concentration.
If the suspension's pH is adjusted in a particular manner that it is away from iso-electric point (IEP), then the suspension's repulsive forces become stronger. It makes the nanofluid to be more stable. The lower the pH values of the base fluid, the higher are the NTU values. If the pH of the nanofluid is remarkably less or considerably more significant than the IEP, superior stability will be obtained.
Effect of ph and concentration of the surfactant
Contour plots for the combined effect of pH and the surfactant's intensity are shown in Supplementary Figure 11. It is found that the value of NTU remains unchanged with the enhancement in the pH. With the enhancement in the surfactant, there is an augmentation in the value of NTU. But with the enhancement in the surfactant, there is a decrease in the formation of agglomeration and sedimentation. The agglomerations are reduced by the surfactant, which allows an increase in the value of NTU. As the pH and concentration of the surfactant are increased, it is observed that the NTU, which is initially low, decreased further from 200 to 100. This shows a decrease in the stability of the nanofluid.
Optimization of operating conditions
For optimizing statistical parameters, a desirability factor will be chosen. The desirability factor chooses a desirability value between 0 and 1. If the desirability factor shows 1, it represents optimal performance, while 0 represents poor performance. Response surface plots are considered to analyze the influence of various combinations of the variables for obtaining maximum NTU. Contour plots of the mutual effect of pH and particle volume concentration are shown in Supplementary Figure 12 (a-b). The desirability factor chosen is 1. The predicted value of NTU obtained is found to be 539.612. Optimum stability can be obtained with the combined effect of pH and particle volume concentration of the nanofluid. Contour plots for the combined effect of concentration of the surfactant and pH are shown in Supplementary Figure 13(a-b). The desirability factor chosen is 0.99764. That means, even though the desirability factor is not 1, it is approaching 1. It indicates that the optimum stability can be achieved with the combination of the concentration of the surfactant and the concentration of the nanofluid. The optimum value obtained for NTU is 539.612. Supplementary Figure 14(a-b) shows the mutual effect of desirability and predicted contours of the concentration of the surfactant and pH. It is found that out of all the combinations, the mutual effect of concentration of the surfactant and pH gives the most optimum stable conditions. Maximum NTU that can be achieved is 560. The ideal combination is 0.01% of the particle volume concentration, 3.2 pH, and 0.6 surfactant concentration.
Conclusion
RSM was utilized successfully to develop a model for predicting the optimum process parameters influencing the stability of ferric oxide nanofluids. SEM and XRD analyses were carried out to check whether there are any agglomerations, peaks, and valleys of the crystals in the nanoparticles. The developed model was validated using the ANOVA table. The desirability approach was carried out to optimize the process parameters like nanofluid volume concentration, pH of the base liquid, and surfactant concentration. The response variable is chosen as NTU. From the above modeling and analysis, the following conclusions were drawn.
In this work, RSM was used to determine the predominant factors and the interactions that influence the stability parameters of the ferric oxide nanofluids. Particle volume concentration of the nanofluid (%), weight (%) of the surfactant, and pH of the base fluid are taken into consideration as input parameters. RSM showed that the weight % of the surfactant has a positive effect while pH has a negative effect on the stability of the nanofluid. Optimal values obtained for attaining maximum NTU for the input parameters of nanoparticle volume concentration, the intensity of the surfactant, and pH of the base fluid are as follows: particle volume concentration of the nanofluid (Y1) = 0.01%; pH of the base fluid (Y2) = 3.2 and intensity of the surfactant (Y3) = 0.6%. This data helps the researchers aiming to improve the stability of the nanofluids.
Supplemental Material
sj-docx-1-pie-10.1177_09544089231151541 - Supplemental material for Prediction of stability parameters of ferric oxide nanofluids using response surface methodology based on desirability approach
Supplemental material, sj-docx-1-pie-10.1177_09544089231151541 for Prediction of stability parameters of ferric oxide nanofluids using response surface methodology based on desirability approach by KPV Krishna Varma, Kavati Venkateswarlu, PV Durga Prasad and Uday Kumar Nutakki in Proceedings of the Institution of Mechanical Engineers, Part E: Journal of Process Mechanical Engineering
Supplemental Material
sj-docx-2-pie-10.1177_09544089231151541 - Supplemental material for Prediction of stability parameters of ferric oxide nanofluids using response surface methodology based on desirability approach
Supplemental material, sj-docx-2-pie-10.1177_09544089231151541 for Prediction of stability parameters of ferric oxide nanofluids using response surface methodology based on desirability approach by KPV Krishna Varma, Kavati Venkateswarlu, PV Durga Prasad and Uday Kumar Nutakki in Proceedings of the Institution of Mechanical Engineers, Part E: Journal of Process Mechanical Engineering
Footnotes
Acknowledgements
The authors thank the RAK Research and Innovation Center, American University of Ras Al Khaimah, Ras Al Khaimah, United Arab Emirates and the Management of CMR College of Engineering & Technology, Hyderabad for providing the required facilities to carry out the research work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.