Abstract
Weld overlaying is a promising and economical solution for repairing stainless steel structures. The present work deals with the deposition of ER-308L weld overlays on AISI 321 plate with Robotic gas metal arc welding setup. Microstructural examination depicts the presence of dendritic structures (equiaxed and columnar) and ferritic stringers. Energy-dispersive X-ray analysis confirmed the absence of precipitates and phases susceptible to corrosion in the as-deposited weld overlays. Specimens from ER-308L (weld overlay) and ER-308L&321-SS (interface) regions were subjected to electrochemical analysis using the Potentiodynamic polarization and double-loop electrochemical potentiokinetic reactivation measurements. Both overlay and interface sections exhibited comparable corrosion rate – 3.30 and 3.62 mils penetration per year, respectively with different passivation behaviour. The improved passivation behaviour of the ER-308L weld overlay authenticates the presence of δ-ferrite stringers. Double-loop electrochemical potentiokinetic reactivation tests highlighted the absence of sensitization on both weld overlay and interface specimens. This study presents the initial assessment results for repairing structural steel components subjected to corrosion damage.
Keywords
Introduction
Austenitic stainless steels (ASS) find extensive applications in the field of nuclear power plants, aircraft, thermal power equipment, etc., because of their superior corrosion rate (CR) along with excellent creep, and tensile strength. For these reasons, often they are considered as weld overlaying material. However, these alloys exhibit certain problems when subjected to service temperature of 400–800 °C by forming Cr23C6 phases, leading to sensitization in the components.1–3 Degree of sensitization (DOS) is a measure of assessing the sensitization or intergranular corrosion (IGC) in ASS and has a direct correlation with the volume of the ferrite phase. 4 AISI 321 is a titanium stabilized austenitic grade that prevents IGC attack by forming titanium carbide (TiC) and the addition of Ti does not influence the pitting resistance significantly.5,6 Stainless steels such as AISI 304 and AISI 321 are prone to pitting corrosion when exposed to saline environments during their service life.7,8 Javidi et al. 6 have mentioned that ASS 321 is not a suitable choice in chloride environments such as cooling systems, seawater treatment plants, power piping systems, etc. From the literature, it is confirmed that ASS 321 components subjected to a chloride-containing environment undergo corrosion damage due to pitting. Further, to restore the failed component, it must be replaced or repaired. Weld overlaying is a standard method to repair structural components in die and mold industries, marine industries, pressure vessels, and automobiles, thus reducing the replacement cost of the components.9–11 ER-308L is one of the recommended filler materials for welding or repairing ASS 321 structures exposed to corrosion damage. 12 This filler wire is preferred for overlaying ASS 321 structures due to its higher weight percentage of Chromium and Molybdenum. Cui et al. 13 evaluated the oxidation resistance and stress corrosion cracking behaviour of 308L claddings for pressurized water reactor applications in the range of 325–350 °C and reported the excellent SCC resistance due to its higher Cr content and closely networked ferrites. Investigations on the 308L clad deposits revealed a dendritic structure with δ-ferrite in the austenitic matrix without any precipitates and the δ-ferrite was 9% on the clad layers. 14 Li et al. 15 reported that the laser-cladded 308L on a 316L substrate revealed an average ferrite percentage of 10.48 in the austenitic matrix. This increase in the ferrite volume is attributed to the deposition of successive clad layers. Multi-layer weld overlaying leads to complex cyclic thermal history influencing the microstructure and mechanical features.14,15 According to the existing literature, various studies have been made on the CR of Ni-Cr-Mo coatings and the various steel welds in salt environments.16–18
Based on the mentioned literature, the research attempts emphasized the multilayer deposition of stainless steels, Ni-based superalloys as a coating material, and the influence of alloying elements on the microstructure and mechanical properties. However, few research attempts on the microstructural and corrosion behaviour of multilayer coatings deposited by the gas metal arc welding (GMAW) process have been published so far.19–24 GMAW is a process of direct metal addition to the base metal with the help of consumable filler metal in a shielded environment (inert gas). GMAW process is known for its higher deposition rate, stronger weld joints, welding wide range of thickness and is used in the repairing applications. 25 To fill the research gaps, an attempt has been made here to analyse the feasibility of employing ER-308L filler as an overlay material for protection against corrosion of AISI 321 substrate. The scope of this work is to deposit the ER-308L weld overlay on ASS 321 using GMAW and analyse their microstructural characteristics and corrosion behaviour.
Materials and methods
Multi-layer weld overlays were deposited using a Six-axis Robotic-GMAW system (OTC-Daihen FD-B6 robotic manipulator, Welbee-P500L power source) as shown in Figure 1. The filler metal employed for the study is ER-308L (Ø1.2 mm) and the substrate used is ASS 321 having dimensions of 80 × 80 × 12 mm3. The chemical composition of the filler metal and substrate measured with spark atomic emission spectroscopy following ASTM E1086-14 are shown in Table 1.
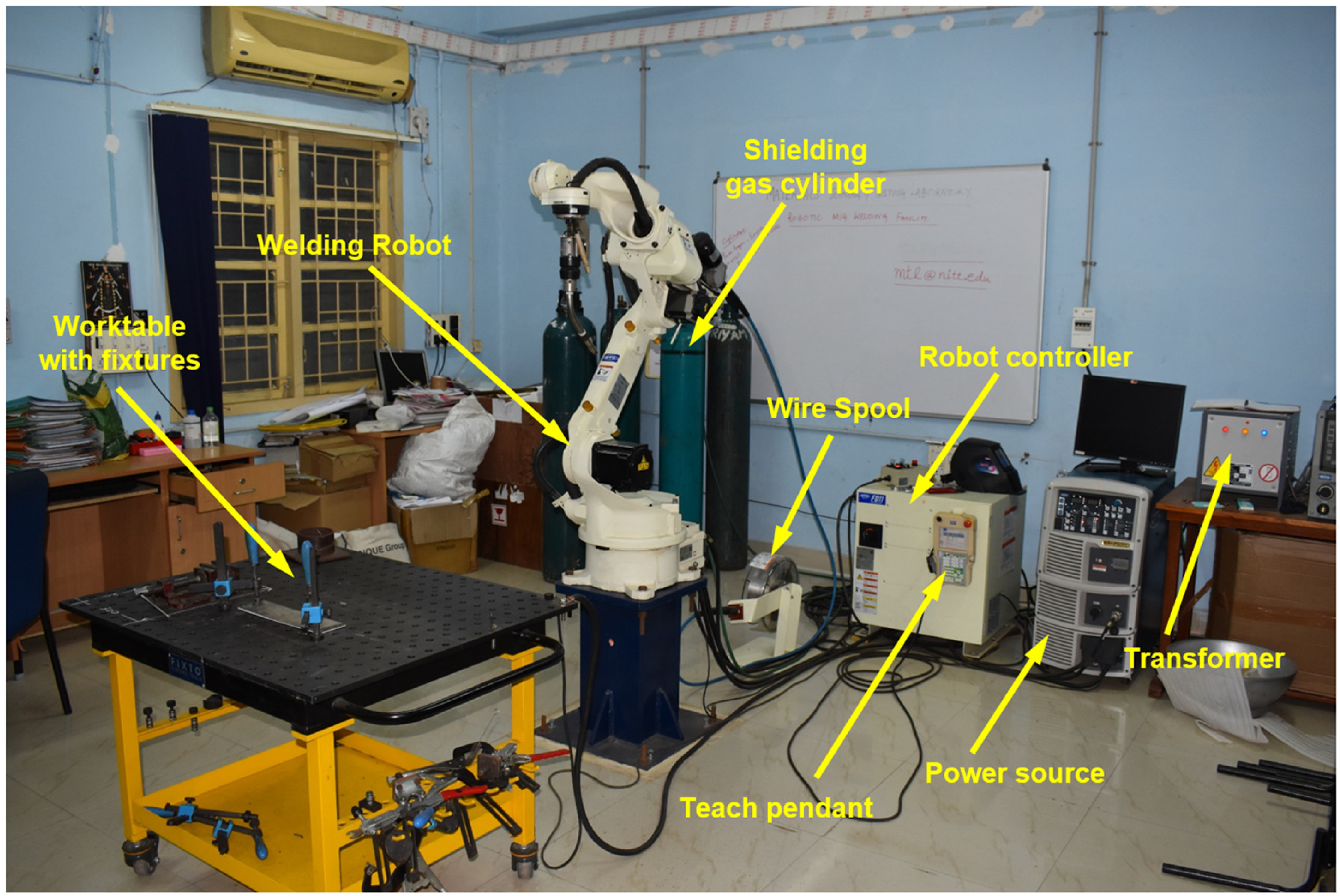
Experimental robotic gas metal arc welding (GMAW) system setup.
Elemental composition of ER308L filler wire and 321 SS.

In order to remove the impurities of the substrate plate, the plate was cleaned with a stainless steel wire brush and abrasive paper. The parameters like welding current and welding speed were identified as the potential parameters governing the weld bead shape of overlay deposition.26,27 Trial runs were conducted to identify the favourable weld deposition parameters such as welding current and welding speed. Welding current was in the range 100–140 Amps and the welding speed was in the range of 150–250 mm/min. Arc weaving technique was employed with the selected parameter which resulted in a defect-free deposit. Weaving the overlay deposit reduces the dilution ratio by widening the deposit with nominal reinforcement. 28 Sinewave type of weaving was employed while depositing the ER-308L overlay. Multi-layered overlay was deposited with an overlap ratio of 50% with the adjacent deposited layer.17,29 The parameter set was derived based on the study reported by the author in their previous research conducted on Ni-based superalloy deposition. 30 The deposition parameters are presented in Table 2. Microstructural analyses were conducted using an optical microscope and scanning electron microscope (SEM). Feritoscope (Fischer-FMP30) was employed to analyse the ferrite number (FN) in the weld overlays and substrate. Though, both the overlay and the substrate materials are ASS, the corrosion behaviour of the interface region is critical and requires systematic examination. The samples were designated as ER-308L for overlay region (top of overlay) and ER-308L and 321 SS for the interface section (transverse of the overlay deposit). Corrosion test was carried out using a three-electrode system in a naturally aerated NaCl (3.5 wt%) solution. In the three-electrode system, the saturated calomel electrode was the reference electrode, the counter electrode was graphite, and the samples were the working electrodes. Potentiodynamic polarization (PDP) tests were carried out at a scan rate of 1.33 mV/s and also electrochemical impedance spectroscopy (EIS) was performed on the same solution. To examine the intergranular susceptibility (IGC), double-loop electrochemical potentiokinetic reactivation (DL-EPR) test was conducted on a more aggressive solution of 2 mol/L H2SO4 + 1.5 mol/L HCl with a scan rate of 0.67 mV/s rather than using the standard solution of 2 mol/L H2SO4 + 0.01 mol/L KSCN + 0.5 mol/L NaCl.31,32 The potential range of −600 to 1200 mVSCE was considered for the forward scan while the same potential was used for the reverse scan at a scan rate of 1.67 mV/s. For both PDP and DL-EPR tests, each sample was immersed in the solution for a time of 30 min to stabilize and measure the open circuit potential (OCP) values. In DL-EPR test, forward scan and reverse scan were performed. In the forward scan, the test sample surface will get passivated and Ia is the maximum current density taken for passivation of the sample. 33 While in the reverse scan, depassivation begins and the passive film gets depleted with a decrease in the potential values. The maximum current density experienced in the reverse scan is the reactivation current density (Ir). DOS can be quantitatively measured with the ratio [(Ir/Ia)*100]. 34 Three samples were tested under similar conditions to determine the repeatability of the electrochemical measurements.
Process parameters for weld overlaying.

Results and discussion
From the visual examination the optimal parameter for overlaying is identified, the parameters which resulted in continuous bead without any surface cracks and lack of adhesion were chosen. Generally, a higher welding current provides higher weld bead width and height along with an increased depth of penetration. This is due to the fact that with increasing welding current wire feed rate increases proportionally as modern-day power sources work in synergic mode. In addition, the dilution percentage is directly dependent on the welding speed, i.e., higher welding speed reduces the over-melting of substrate with higher weld bead height. 35 Similar trends were observed during the overlaying of stainless steel. 36 Welding current and speed control the amount of heat input and the weld bead characteristics. 37 Deposits with good shape factor and height are preferred for this study. The photograph of the deposited ER-308L overlay is shown in Figure 2.

Photograph of the deposited ER-308L overlay on AISI 321 substrate.
The microstructural features of the as-deposited ER-308L at the overlay and interface sections at different magnifications were exhibited in the Figure 3. Microstructure of the overlay section is witnessed with various dendritic structures because of the repeated thermal cycles experienced while overlaying as shown in Figure 3(a)–(c). The higher wt% of ferrite-forming elements such as Chromium and Molybdenum in the filler wire aided the formation of δ-ferrite on the overlay (Figure 3(a)–(c)). In ASS's, solidification phases are influenced by their solidification modes. The chromium equivalent of the overlay is calculated as Creq = Cr + Mo + 1.5Si + 0.5Nb and for Nickel equivalent, Nieq = Ni + 30C + 0.5Mn. The measured Creq is 21.07 and Nieq is 12.78 using Schaeffler formula.38,39 The Creq/Nieq of the 308L overlay is 1.65, which follows the FA (ferrite to austenite) mode of solidification, i.e., FA type of solidification occurs when Creq/Nieq is in the range of 1.90 and it avoids hot cracking in ASS and their welds. Figure 3(c) confirms the distribution of delta ferrite stringers within the austenitic matrix. During solidification, δ-ferrite is the starting phase and follows austenitic transformation by a peritectic-eutectic reaction. 14 Most of the ferrite is transformed into austenite and a small fraction of ferrite is retained due to constitutional supercooling. The transformation of ferrite to austenite will happen till the temperature decreases and until preventing the elemental diffusion. The ferrite after solidification exists in the form of lathy and skeletal morphologies in weld overlays, 40 as shown in Figure 3(c).
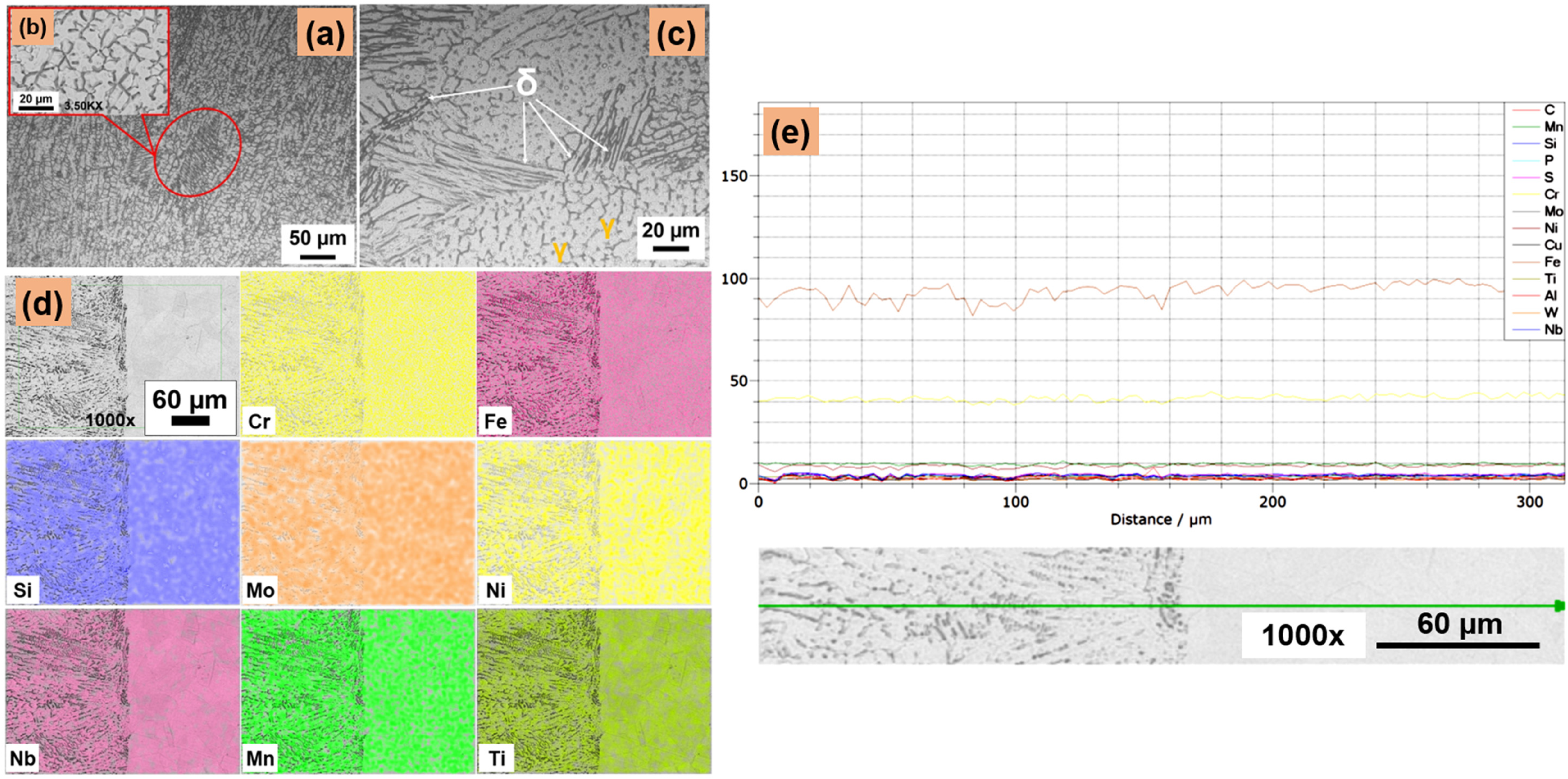
Microstructural results (a), (c) OM images of weld overlay, (b) SEM image (3.5Kx), (d) EDS elemental map (1000×) and (e) line map comprising the substrate and overlay (1000×). EDS: energy-dispersive X-ray analysis; SEM: scanning electron microscope; OM: optical microscope.
Epitaxial dendritic growth was observed at the interface as the substrate acted as a heat sink. 41 From the energy-dispersive X-ray analysis (EDS) elemental maps and line scan results shown in Figure 3(d) and (e), it is noticeable that no precipitates or inclusions exist in the overlays. EDS elemental maps highlight the absence of elemental variation at the interface, as both the substrate and the overlay material are ASS. The average amount of ferrite measured along the transverse direction of the weld overlay revealed 4.21 ± 0.5 FN on the ER-308L overlay, 1.46 ± 0.2 FN on the interface and 1.65 ± 0.32 FN on the substrate, respectively. The measured FN on the overlay section is consistent with the microstructure observed. The ferrite content got increased in overlapped layers of ER-308L due to multiple heating and cooling cycles. 15
PDP plot of the samples tested at 3.5 wt% NaCl solution are represented in Figure 4. The corrosion current density (icorr) and the corrosion potential (Ecorr) are extrapolated using Tafel method and are summarized in Table 3. The corrosion current density (icorr) indicates the CR and Ecorr implies the thermodynamic tendency of the material. 42 From the available literature, CR is lesser when the icorr is less and the Ecorr is higher.43,44 The pitting potential of ER-308L overlay was 269 mVSCE while 184 mVSCE for interface, above this potential, pitting begins. ER-308L samples exhibited signs of passivity at high anodic potential than ER-308L&321-SS samples. The corrosion current density of the 308L overlay was 8.03 µA/cm2 and on the interface it was 8.95 µA/cm2. Corrosion results are in agreement with previous findings on ER-308L filler multi-pass weld deposition on AISI 304L base metal. 45 The CR of the overlay is comparable because of the higher fraction of alloying elements that stabilize the critical corrosion-prone phases.
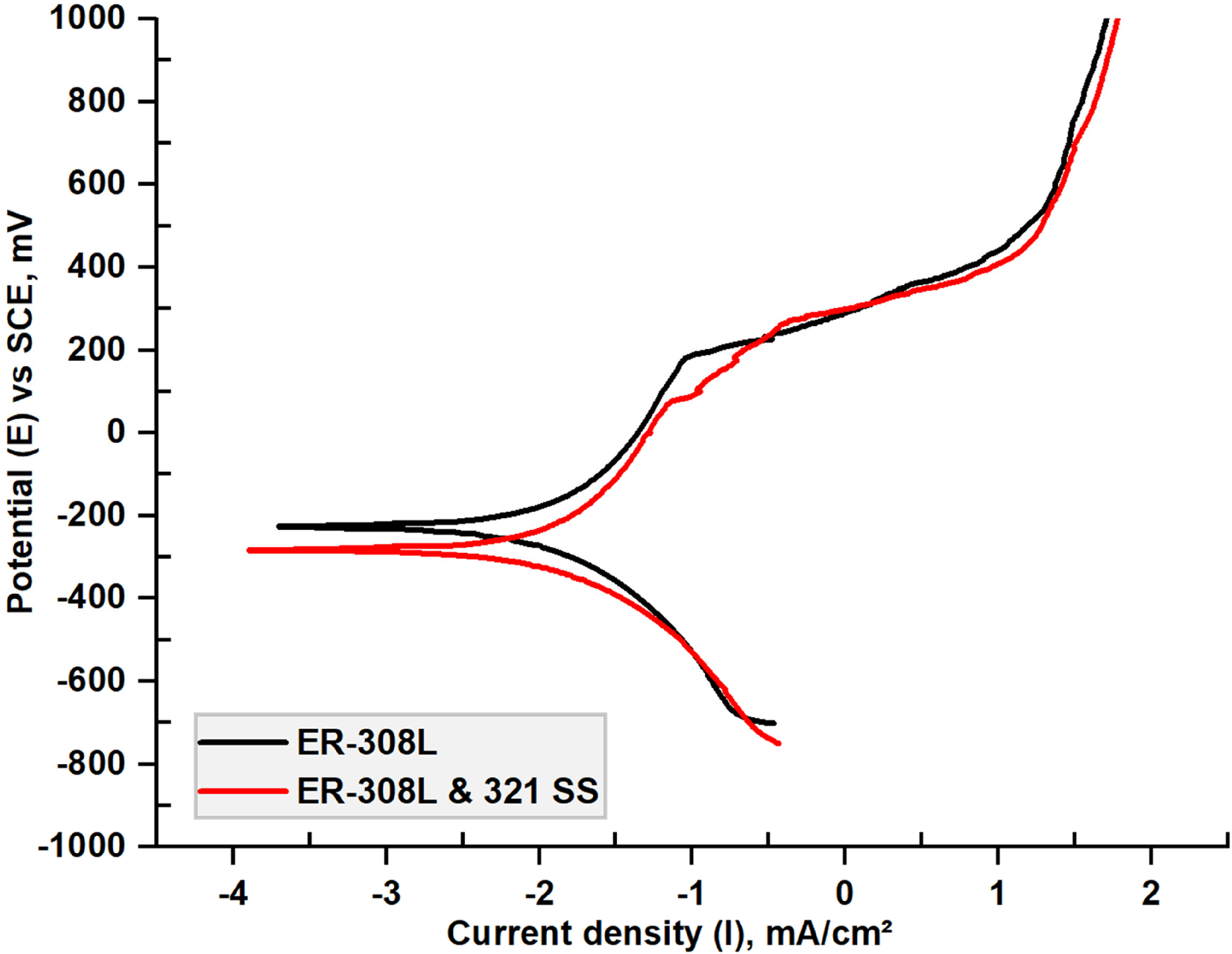
Potentiodynamic polarization curves.
Electrochemical corrosion parameters obtained from potentiodynamic polarization test.

EIS map, phase angle plot, bode plot and equivalent circuit of the weld overlay and interface samples are exhibited in Figure 5(a)–(d), respectively. The Nyquist plot exhibiting a higher radius of the semicircle highlights the increased polarization resistance on the ER-308L overlay. The maximum phase angle obtained for ER-308L & ER-308L&321-SS are 75° and 65°, respectively epitomizing their capacitive behaviour, usually found in passive materials. Higher the phase angle confirms the higher polarization resistance, and the trend is identical to the PDP results. The slopes of the Nyquist plots exposes the difference in CR, i.e., the increase in the radii of the semicircle denotes the increase in polarization resistance of the surface. 18 From Figure 5(a), it is evident that the samples exhibited different corrosion behaviours which are in line with the phase angle and bode plots. Impedance of the ER-308L was higher than ER-308L&321-SS interface revealing the formation of the enhanced passive film. Higher impedance modulus at lower frequencies is an indication of increased polarization resistance. At lower frequency, the impedance value of the ER-308L&321-SS interface is lesser than the ER-308L indicating the weak polarization resistance of ER-308L&321-SS samples. The equivalent circuit shown in Figure 5(d) was utilized to fit the impedance data of the sample at activation polarization mode. In the circuit, Rs denotes the resistance offered by the electrolyte, Rct denotes the charge transfer resistance and CPEdl denotes the constant phase element of the passive film. The CRs calculated were 3.30 and 3.62 mpy for overlay and interface samples, respectively. As reported by Fontana 46 when the CR is between 1 and 5 mpy, it denotes excellent CR being offered.
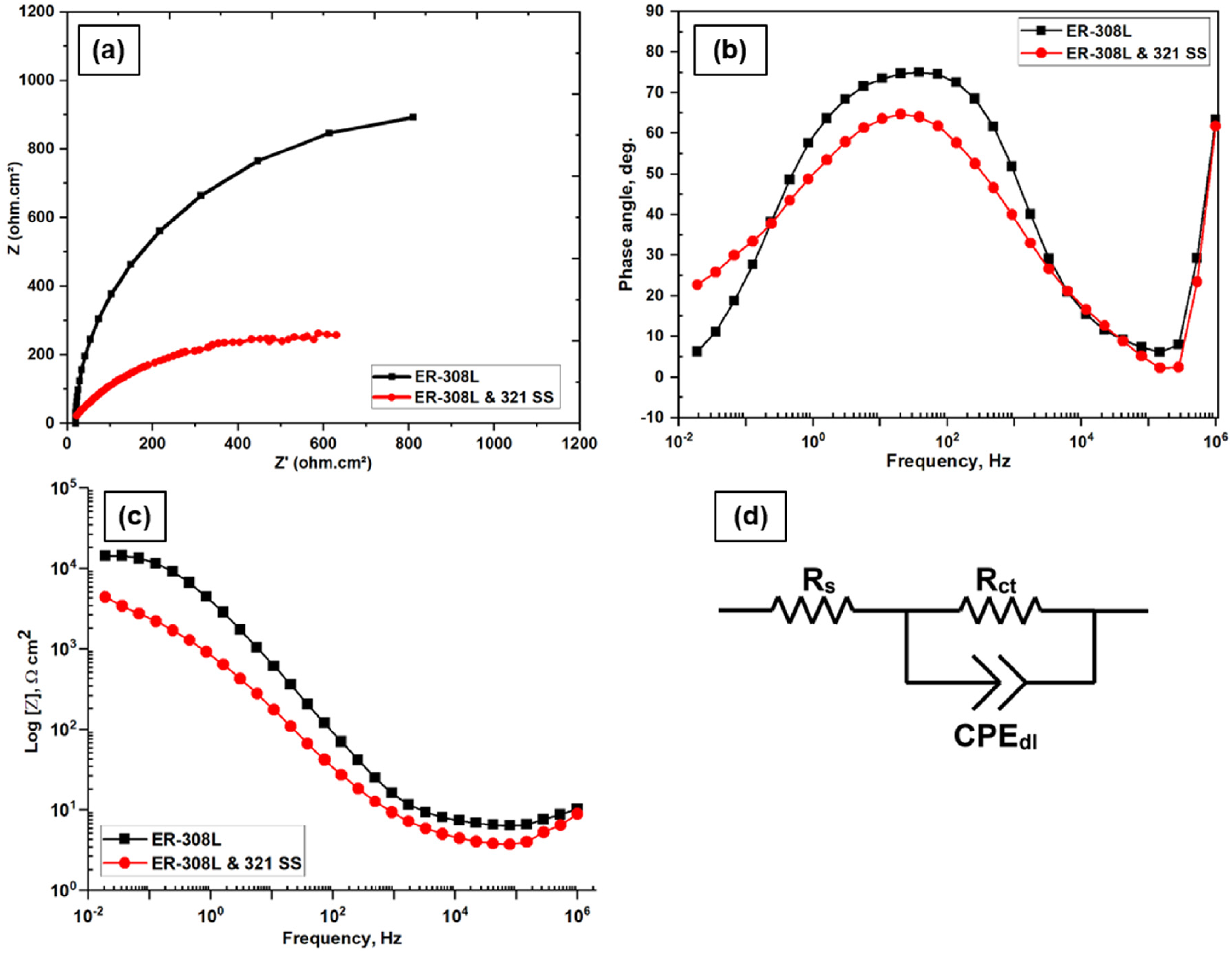
(a) electrochemical impedance spectroscopy map, (b) phase angle plot, (c) bode impedance plot and (d) equivalent circuit.
The pitting corrosion mechanism involves the breakdown of the passive film formed on the surface of austenitic stainless steels i.e., localized corrosion of the stainless steels. The passive film formed acts as a protective barrier and characteristics of the passive film depend upon the composition and the microstructure. 8 Chromium element provides better pitting resistance as it reacts with dissolved oxygen and forms a passive film over the surface of the sample. 47 The ferrite content rich in overlay region inferred from the ferrite measurement and the delta ferrite observation from the microstructure offers a good resistance to corrosion of the overlay sample. Pitting resistance was higher on the ER-308L overlay due to the presence of rich δ-ferrite content (4.21 FN).48–50 Therefore, a constant and efficient Cr-rich passive film formation is revealed in the anodic polarization.
The uniformly distributed alloying elements and the δ-ferrite stringers within the austenitic matrix in the overlay region resulted in improved pitting resistance.8,51 In addition, the interface has comparable CR since both the substrate and weld overlay material were austenitic grades. The residual ferrite, i.e., δ-ferrite influences the corrosion behaviour of the dissimilar welded Ni-based alloy with super duplex stainless steels. The pitting resistance on the root pass (FN – 10.8 and 8.9) was lesser compared to the cap pass (FN – 7.1 and 6.4) and this is attributed to their ferrite fraction. 52 Mirshekari et al. 45 have similarly reported that the δ- ferrite content increases with an increase in the number of passes and it considerably influences the pitting CR in the weld zone. Further, chloride ions available on the corrosive medium preferentially damage the ferrite phases leading to corrosion damage of the overlay sample. 53 It is also evident that the increase in the number of deposited layers favours the increase in the δ-ferrite content, which in turn improves the CR of the overlay sample. Overlay rich in δ-ferrite content traps the Sulphur rich inclusions in ASS welds on the boundaries of γ/δ and γ-matrix thus increasing the pitting resistance of the overlay. 48
Figure 6 reveals the plot of DL-EPR test conducted on two samples and their results are summarized in Table 4. The activation current density (Ia) in the forward scan on the ER-308L and ER-308L&321-SS sample was 0.016 and 0.018 µA/cm2, respectively. Both the ER-308L and ER-308L&321-SS samples have not exhibited any peaks on the reverse scan. 54 The absence of secondary phases in the weld overlay and the interface section is the reason for the low reactivation current density and therefore the ER-308L weld overlays and ER-308L&321-SS interface was confirmed as unsensitized material. The higher weight percent of Cr and extra low carbon content increases the IGC resistance. Hence ER-308L filler material is employed for welding stabilized grades such as SS 347 and SS 321 for ASME Boiler and Pressure Vessel Code Section III applications. The passive film formed during the forward scan on the surface restricts the damage by corrosion. While the results of Verma et al. 34 exposed 11% DOS of the weldments with high heat input due to the high ferrite content (41%) and Cr depleted grain boundaries. Further the author reported that the detrimental phases prone to IGC attack includes ferrite which gets transformed to sigma and secondary austenite (γ2) are responsible for higher DOS %. In the case of ER-308L multilayer overlays, no deleterious phases of this type were identified, and the ferrite content (4.21 FN) was lower, so there was no depletion of the passive film, so sensitization was not revealed.
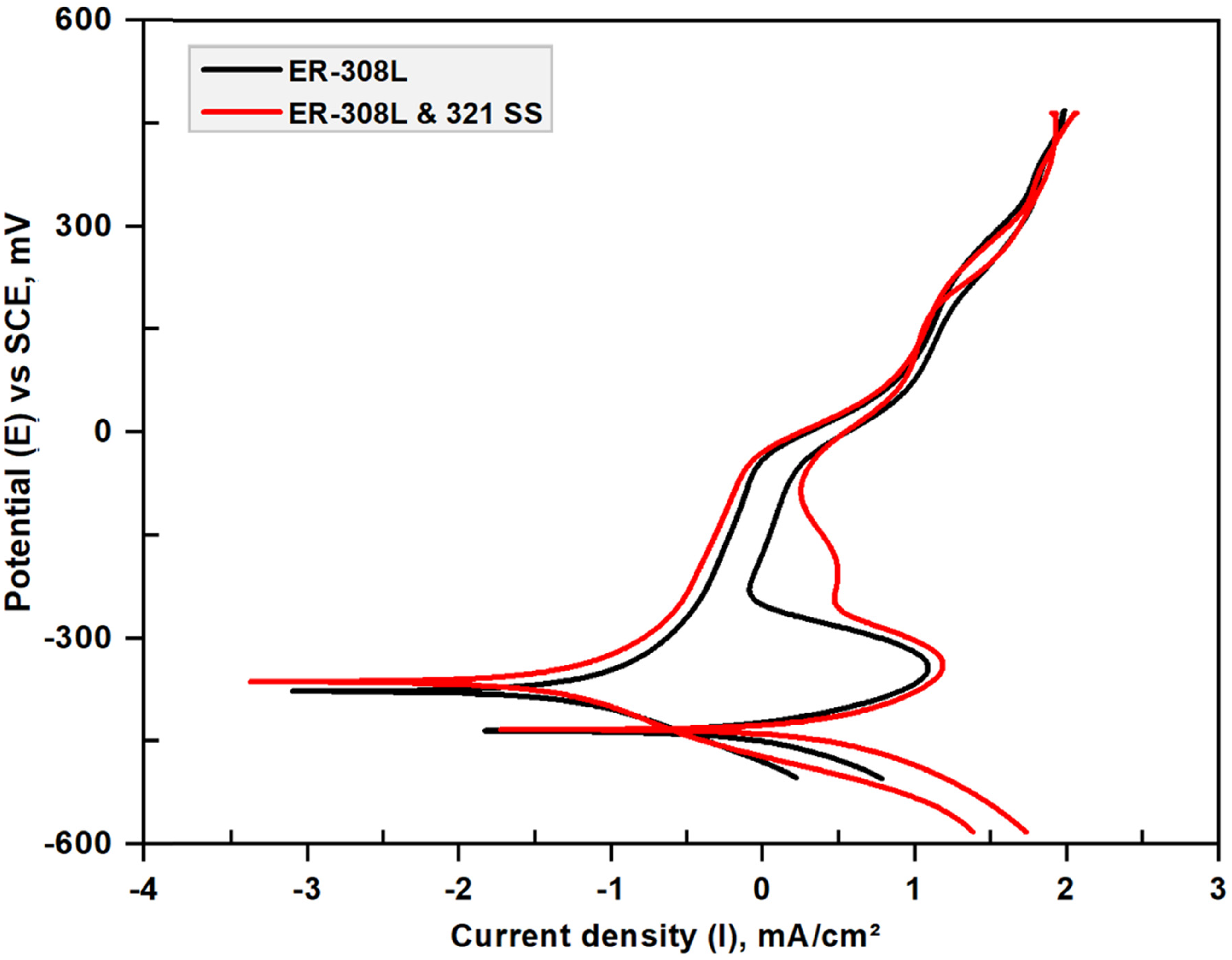
Potential versus current curves obtained in double-loop electrochemical potentiokinetic reactivation (DL-EPR) test.
DL-EPR results.

DL-EPR: double-loop electrochemical potentiokinetic reactivation; DOS: degree of sensitization.
Resistance to sensitization was strengthened due to the use of a low-carbon filler metal that reduces the reaction time of carbide precipitation and also the Ti-stabilized grade substrate minimizes the formation of Cr-rich carbide, which prevents intergranular attack. 39 Thus both the weld overlay and the interface showed insensitivity to IGC attack and confirmed from the DOS measurements.32,54
In Figure 7(a)–(c) the morphology of the pits in the deposited layers in the same place at different magnifications is shown. Figure 7(d) shows an image of pitting at 500× magnification in one place, while in Figure 7(e) and (f) the pitting surface at the interface at various magnifications elsewhere is shown. The pits visible in SEM images resemble the pitting behaviour of the samples. The smaller the pit size, the higher the pitting resistance offered, which is in line with the CRs. 18 The size of pit formation was higher on the ER-308L&321-SS sample than the ER-308L sample as shown in Figure 7(d)–(f). Usually, pit formation is closely in relation to weakening of the passive films on the Cr-depleted region. Cr-rich interdendritic regions prevailing on the ER-308L samples are resistant to pitting attack thus less pit size. 55 The size of the pit was in the range of 100–150 µm at weld overlay and interface regions as illustrated in Figure 7(a) and (d). The pit images confirm the attack on the passive film at the interface region while the passive film formed on the ER-308L overlay was more protective. From the available literature, it is known that the CR is significantly influenced by the FN in ASS weld deposits.33,34,56,57 The pitting resistance is high in the weld overlay section and is confirmed from the FN measurements. From Figure 7(b), (c) and (e), (f), it is evident a web of lace pit covers is formed and is partially soluble. 47 These lace-like pit covers are formed in the nests of delta ferrite and are known for long-term sustainability of the pits. 57 The pitting resistance is not significantly affected when the FN is less than 8% in ASS welds or overlays.18,38,49,58,59

Pitting images (a)–(c) ER-308L section and (d)–(f) ER-308L and 321-SS section.
Conclusion
ER-308L weld overlays were successfully deposited by the Robotic-GMAW process to examine the microstructural characteristics and corrosive behaviour. The conclusion of the study is as follows:
The grain morphology of the ER-308L overlay are mainly columnar dendrites with a few equiaxed dendrites on the top of the overlay. Ferrite content was higher on the overlay (4.21 FN) and existed in the form of skeletal and lath morphologies. The ferrite stringers formed during overlaying improved the CR as a result of higher Cr in the filler material. A strong protective passive film was formed on the multi-layer weld overlays and is in line with the EIS plots. The CR was between 3.30 and 3.62 mpy with better pitting resistance on the 308L overlay side than on the interface side. DL-EPR tests revealed no sensitization on both the overlay as well as the interface side due to the absence of detrimental phases. The results of this research will pave the way for the use of a cost-effective and compatible filler wire when repairing austenitic stainless steels by overlay welding, in terms of corrosion damage.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
