Abstract
The high-temperature corrosion is a crucial issue of stainless-steel components such as boilers and superheaters in power plant industries when it operates at elevated temperatures due to impurities in the environment, which deteriorate the properties of the materials. The current research work goal is to investigate the cyclic corrosion characteristics of the bare and Ni–20%Cr, a high-velocity oxy-fuel sprayed coating on AISI 321 steel in Na2SO4–60%V2O5 salt atmosphere at 900 °C. Thermogravimetric study was used to examine the corrosion kinetics by weight change technique. Surface morphology and compositions of the coating and oxide scale morphology were characterized. X-ray diffraction analysis divulges that Fe2O3 is the primary scale in the uncoated specimen. Corrosion attack across the depth and scale thickness is analyzed by a cross-sectional study. More oxide scales spalling could be observed in the uncoated specimen during the corrosion study due to nonprotective Fe2O3 formation. Compared with the bare specimen, the coated steel provides superior corrosion resistance at 900 °C. This is ascribed to the occurrence of NiO, NiCr2O4, and Cr2O3 scales in the coated specimen, which provides sufficient protection to the specimen.
Keywords
Introduction
Due to cost-effectiveness and high mechanical strength, carbon steel and stainless-steel materials are widely prepared for the power plant superheaters and boiler components. The austenitic stainless steel AISI 321 is broadly used for high-temperature industries because of its excellent corrosion and oxidation resistance. This material is mainly preferred for power plant applications such as boiler tubes and superheater materials. Presence of a higher amount of Cr and Ni provides corrosion resistance to the material at elevated temperatures.1–2 However, the components of the boilers and turbines are vulnerable to accelerated oxidation when exposed to corrosive elements at elevated temperatures. The corrosive elements such as sodium, sulfur, and vanadium in the low-grade fuel combine to form fused salt during combustion. The steel materials suffer severely from corrosion attacks in the boiler environment due to the deposition of salts such as Na2SO4 and V2O5 from coal. At elevated temperatures, the salts combine, forming low-melting eutectic compounds.3–4 In power plant environments, the fused salts are deposited on the boiler components, further accelerating the corrosion rate and reducing the component's life resulting in leakages of the boiler tubes and plant shutdown. Continuous working of the plant was interrupted due to the degradation of boilers and other components. In boiler environments, the hot sections of the components must be protected from corrosive elements to enhance their life. Various researchers suggested the employment of corrosion-resistance metallic coatings to protect the components at elevated temperature.5–7
Coatings are developed on the steels and superalloy materials to impart corrosion and oxidation resistance at elevated temperatures. 8 Several techniques were employed to prepare metallic coatings, such as chemical vapour deposition (CVD), sputtering, diffusion, electroplating, and thermal spraying systems. However, the protective coatings are generally made by the thermal spraying process on high-temperature materials without disturbing any other properties of the substrate materials. Thermal spray systems, though widely used, are also readily available, cost-effective, and most convenient. In the thermal spray process, the melted powder particles are sprayed over the substrate material.9–10 The microstructure of the coatings is mainly depending on the alloying elements and thermal spraying methods. High-velocity oxy-fuel (HVOF) is a widely used technique to deposit erosion, wear, and corrosion resistance coatings among a variety of thermal spray methods. HVOF coating is the most promising process to producing metallic, cermet, and carbide-based ceramic coatings on the steels and superalloy components in boilers, biomass, waste heat incinerators, and turbine industries for wear, erosion, and corrosion resistance applications. In HVOF method, powder particles and wires are used as promising candidate material. It is significant to optimize the process parameters to employ quality and defect free coating. HVOF technique has several advantages over the plasma spray process in terms of the coating quality, for instance, homogeneous, higher hardness, high density, and more flexibility with desired thickness of coating can be achieved. In addition, HVOF coating exhibits low-cost coating deposition and greater performance with low oxide contents and porosity. HVOF coating shows the high adhesive strength due to high impact powder particles velocity, and the coating produces the splat-like structure.11–13
Due to easy fabrication, cost-effectiveness, and better resistance to corrosion characteristics, Ni–20%Cr coating is extensively designed for corrosion resistance applications at elevated temperatures against hot corrosion in boiler and gas turbine atmospheres. Depositing the metallic coatings on the structural components could enhancing the life and performance. Cr-containing Ni-based feedstock materials are more interesting and significantly used as a coating material in various industrial sectors such as coal fired boilers, reheaters, and superheaters to improve the oxidation and corrosion resistance. At elevated temperature, Cr2O3 scale formed rapidly on the coating surface, which protects the high temperature structural material from hot corrosion. Many researchers have developed corrosion-resistant coatings for low alloy boiler steel and superalloy materials through the HVOF process. Muthu et al. 8 studied the hot corrosion performance of the Ni–20%Cr coated and bare alloy 825 in a V2O5–40% Na2SO4 atmosphere. They concluded that the coating improved the resistance to corrosion and spallation. The bare specimen was vulnerable to severe spalling of oxide scales and high weight gain. Sidhu et al. 12 developed stellite-6 and NiCrBSi coatings on alloy 718 using the HVOF technique to preserve the specimen from the actual boiler environment. They reported that stellite-6 coating preserved the specimen better than NiCrBSi coating and bare sample. Bala et al. 14 employed HVOF and cold spray Ni–20%Cr coating on steel to enhance the corrosion performance in the superheater zone of an actual boiler atmosphere. It was noticed that the HVOF sprayed steel offers better performance than cold sprayed steel. Muthu et al. 15 deposited Ni–Cr coating on Alloy A-286 by the HVOF method to reduce the corrosion rate. The authors suggest that the coatings protect the specimens from the aggressive salt environment owing to the protective oxide scales development on the coating, and the bare alloy suffered severely by corrosion attack. Mervi et al. 16 deposited NiCr and NiCrBSi on carbon steel S275 by APS, HVAF, and HVOF methods and studied the corrosion behavior in 0.5 and 3.5% NaCl solution by immersion and electrochemical method. The authors reported that HVOF sprayed NiCr exhibited the better resistance than other methods. Herman et al. 17 used HVOF spraying method to deposit Ni–20Cr, Ni–Cr–B–Si–Fe and WC–CrC–Ni coatings on AISI 4340 steel to enhance the corrosion and wear performance. Ni–20Cr coating employed by HVOF improved the both corrosion and wear resistance than other materials. Santhosh et al. 18 deposited Ni–20Cr and Ni–Al coating on the SA 516 and T22 boiler grade steels by wire arc spraying method and erosion–corrosion performance was evaluated in simulated boiler environment. Ni–20Cr coating reduced the E-C rate of the steels than Ni-Al coating due to high hardness and the development of protective NiO, NiCr2O4, and Cr2O3 scales.
It is elucidated from the literature that no stated works are available on the corrosion features and thermal spray protective coatings on AISI 321 steel at high temperature. Therefore, it is significant to examine the effect of coating on steel material at high temperatures. The main goal of this study is to evaluate the cyclic corrosion behavior of the bare and HVOF sprayed Ni–20%Cr coating on AISI 321 steel in the salt atmosphere at 900 °C, which is not stated in the literature so far. The oxide scale phases, morphology, and compositions were evaluated. Corrosion attack and scale formation have been studied by cross-sectional analysis. In addition, the elemental mapping technique was used to analyze the distributions of the corrosive elements in the cross-sections of the specimens.
Materials and experimental procedures
Substrate and preparation of coating
AISI 321 steel has been selected as the substrate material primarily used for fabricating boilers components and superheaters in a power plant application. The specimen is cut into rectangular shape with a dimension of 20 × 10 × 5 mm using the wire electrical discharge machining (WEDM) process. The specimens are polished with SiC emery sheet on all sides to remove impurities and oxide scales from the surface. Optical emission spectroscopy is used to analyze the actual composition of the AISI 321 steel, which is reported in Table 1.
Chemical composition of austenitic stainless steel AISI 321.
In this study, Ni–20%Cr powder is deposited on the AISI 321 steel by the HVOF method. The coating is carried out on all the sides (six) of the specimen. Before coating, the specimen is polished with SiC papers, followed by alumina disc polishing to remove the dirt, unwanted oxides, and surface irregularities. To ensure proper adhesion, it is necessary to enhance the roughness; hence, alumina particles were used to grit blast the specimen, which significantly improves the bonding strength of the coating to the substrate. Then, the steel sample was washed with acetone. The optimized process parameters employed for the HVOF coating deposition, which have been displayed in Table 2.
HVOF coating process parameters.

Morphology of the feedstock Ni–20%Cr powders was analyzed using scanning electron microscope to evaluate the particle shape and size. X-ray diffraction (XRD) analysis was done on the powder and coating using Brucker D8 diffractometer to identify the phases. The coating characterization such as surface roughness, micro-structure, and micro-hardness examination was performed. Mahr surface roughness tester was employed to measure the roughness of the coating in the as-sprayed condition at three different places. Then, average value was taken. The structure and thickness of the coating were examined using scanning electron microscope (SEM: Carl Zeiss EVO 18) and optical microscopy (OM: Carl Zeiss) analysis. In addition, a hardness investigation was done at the coating cross-section (vertical direction) using Vickers microhardness (Matsuzawa MMT-X) tester. A diamond shape indenter was used for make indentation. For hardness measurement, a load of 50 g was applied for a dwell time of 10 s with a regular interval of 50 µm. Three replications were performed on the coated sample. Then average value was taken.
Hot corrosion test
The corrosion study is carried out on the bare and HVOF Ni–20%Cr coated AISI 321 steel specimens in Na2SO4–60%V2O5 corrosive medium at 900 °C for 50 cycles. The weight measurement was done on the specimens before and after applying salt. The molten salt is deposited on the specimens in the range of 3–5 mg/cm2 by camel hairbrush. While applying salt, the specimen temperature is maintained at 250 °C to enhance the proper adhesion of salt on the specimen surface. To remove the moisture, the salt-coated specimens with crucibles are preheated at 120 °C for 2h. The specimens are kept in the furnace with an alumina crucible at 900 °C. After each cycle, the specimen's weight is noted with an alumina crucible using an electronic weighing balance (M/s Mettler Toledo) with 0.0001 g accuracy. The specimens were placed inside the furnace after weight measurement for next cycle. During the corrosion study, scales spall into the alumina boats, which are also included in weight measurement. The weight change method is used to establish the total corrosion rate. The scales formed on the specimens after the corrosion run were analyzed using SEM-energy dispersive x-ray spectroscopy (EDS) and XRD. The corroded specimens were cut across the cross-section using a diamond saw cutter. The specimens were subjected to a standard metallographic procedure for the cross-sectional study to obtain a mirror finish. The scale formation and damage due to corrosion attack on the scales were characterized using SEM cross-sectional analysis.
Results
Coating characterization
Mahr surface roughness equipment is used to measure the roughness of the HVOF Ni–20%Cr coated AISI 321 steel. The coating has an average roughness value of 5.13 µm. SEM image of the powder (Figure 1(a)) shows that the spherical and irregular shape powders were used for HVOF coating deposition and the powder particle sizes were varied in the range of 30–60 µm, which was confirmed by particle size analyzer (Figure 1(b)). XRD results (Figure 1(c) and (d)) depicts that the existence of Ni and Cr elements in both powders and coating. SEM surface micrograph of the as-sprayed coating (Figure 1(e)) has few micropores and cracks. Also, it indicates the existence of partially and unmelted particles. SEM cross-sectional morphology (Figure 1(f)) indicates that coating displays a dense structure and well bonding to the substrate, and the coating thickness value is found in the range of 230–240 µm. It reveals that some micropores and cracks were in the coating cross-section, and some inclusions are found in the interface. The hardness profile (Figure 2) reveals that the coating region shows high hardness value than the substrate due to metallurgical change. The interface shows a high hardness than the substrate due to the high impact velocity of coating particles like the micro shoot peening effect. 15 The hardness values tested in substrate, interface, and coating regions are 212, 448, and 595 HV, respectively.
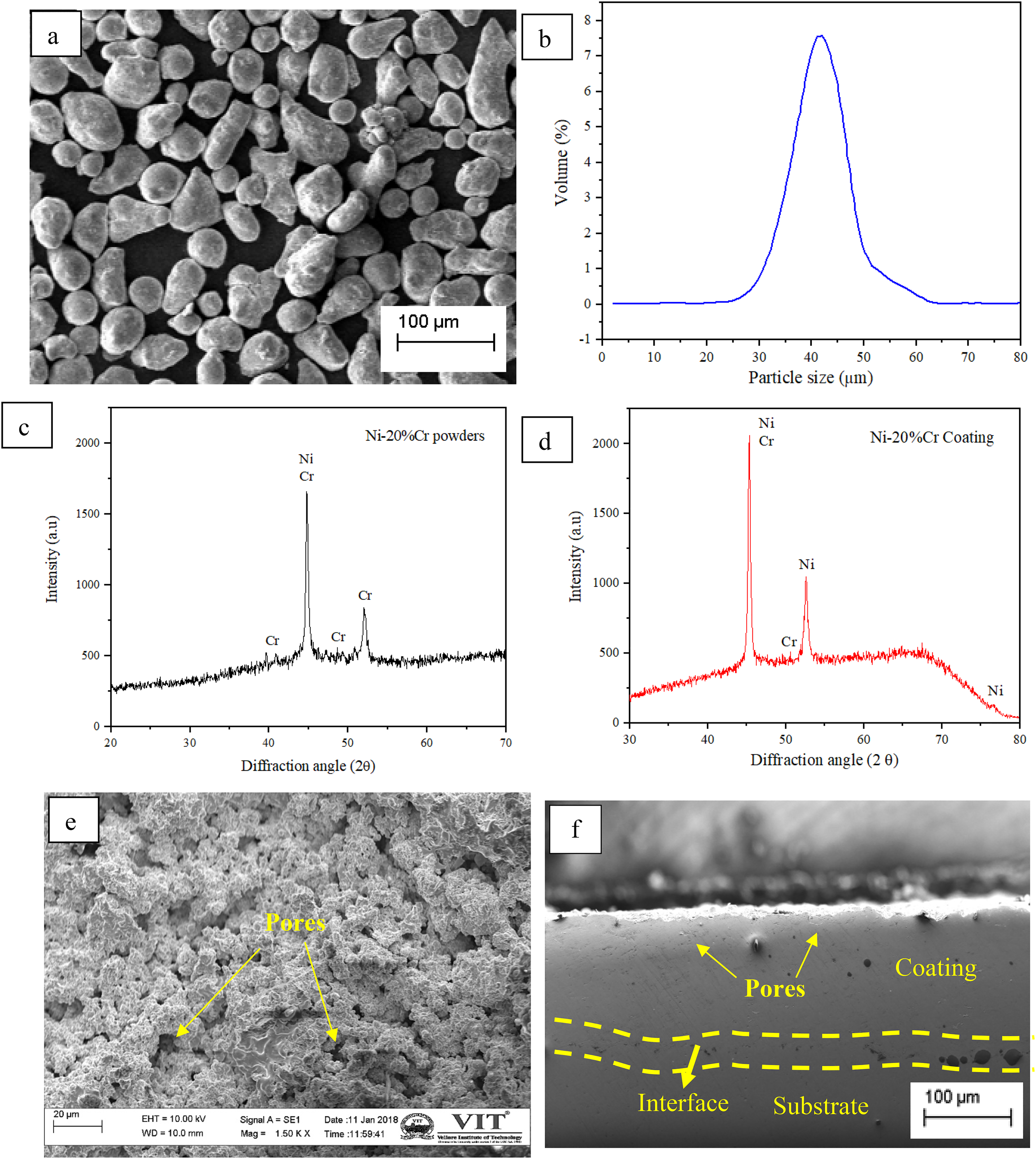
It shows (a) SEM powder morphology; (b) particle size analysis; (c) XRD analysis of the Ni–20%Cr powder; (d) XRD analysis; (e) SEM surface morphology, and (f) OM cross-sectional microstructure of the Ni–20%Cr coating. OM: optical microscopy; SEM: scanning electron microscope; XRD: X-ray diffraction.
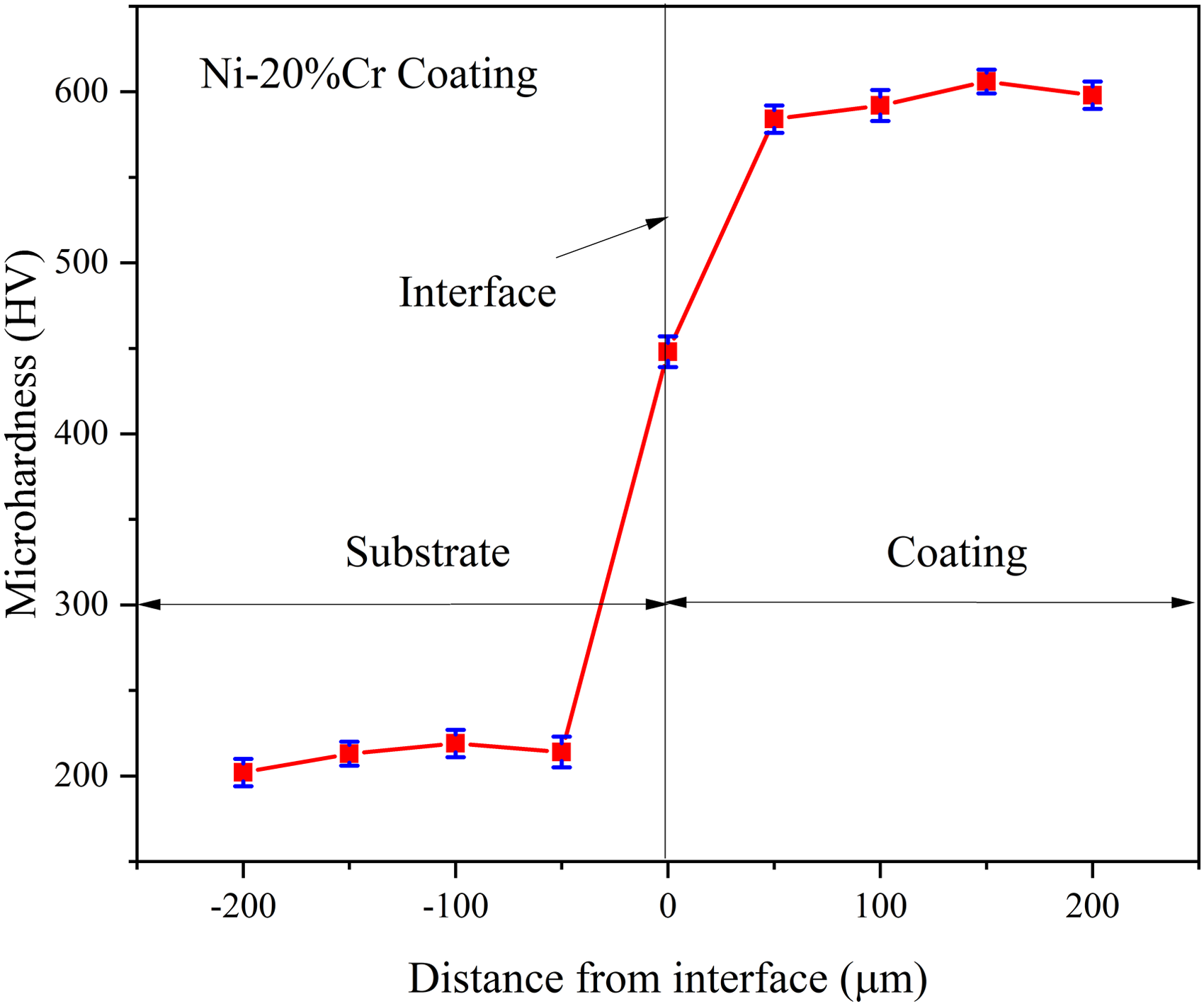
Microhardness profile of HVOF sprayed Ni–20%Cr coating. HVOF: high-velocity oxy-fuel.
Visual inspection
The corrosion test was performed in tubular furnace and the schematic diagram was illustrated in Figure 3. The macrograph of coated and bare steels are shown in Figure 4 during corrosion study in the corrosive medium at 900 °C. In the uncoated specimen, the scales start to spall from first cycle. The oxide scales are spalled out from the surface during the cooling process due to thermal stress. Oxide scales spalling and sputtering were noticed from the initial cycle, which may be attributed to cyclic heating and cooling. Spalling rate of scales were increases with an increase in exposure time. No weight gain was found till the end of the exposure. After the second cycle, the specimen color changed to brownish, which further turned into dark black after the fourth cycle. In the case of the coated specimen, the scales color turns dark black after a few cycles. No oxide scale spallation is noticed up to the 18th cycle. After the 18th cycle could be seen minor oxide scale spallation. No visible cracks were found on the coated specimen. Protective oxide scales are formed on the coated sample during the corrosion study, which protects the specimen from molten salts.
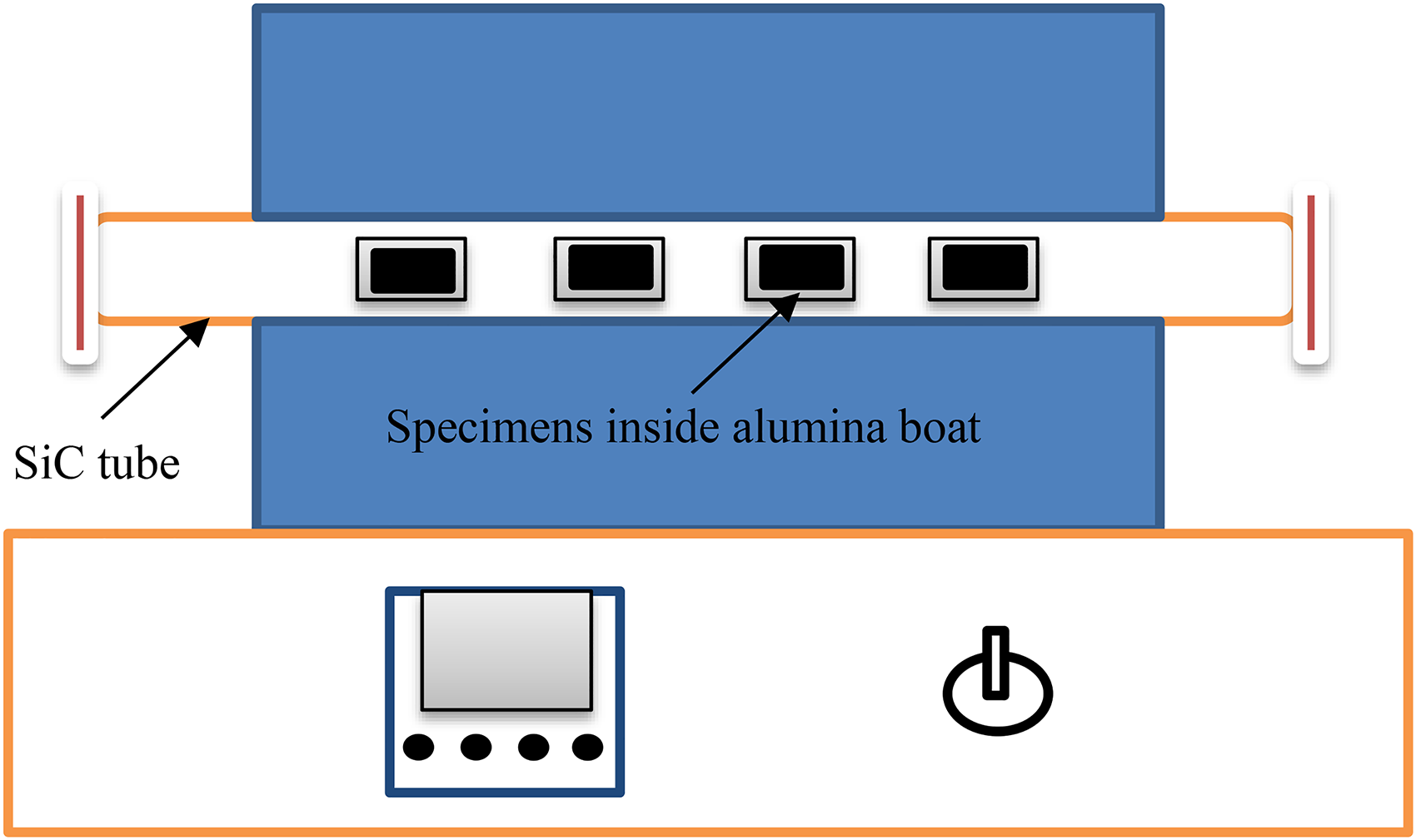
Schematic diagram of hot corrosion setup.

Shows the photograph of hot corroded bare and HVOF coated steel AISI 321 during hot corrosion study. HVOF: high-velocity oxy-fuel.
Thermogravimetric analysis
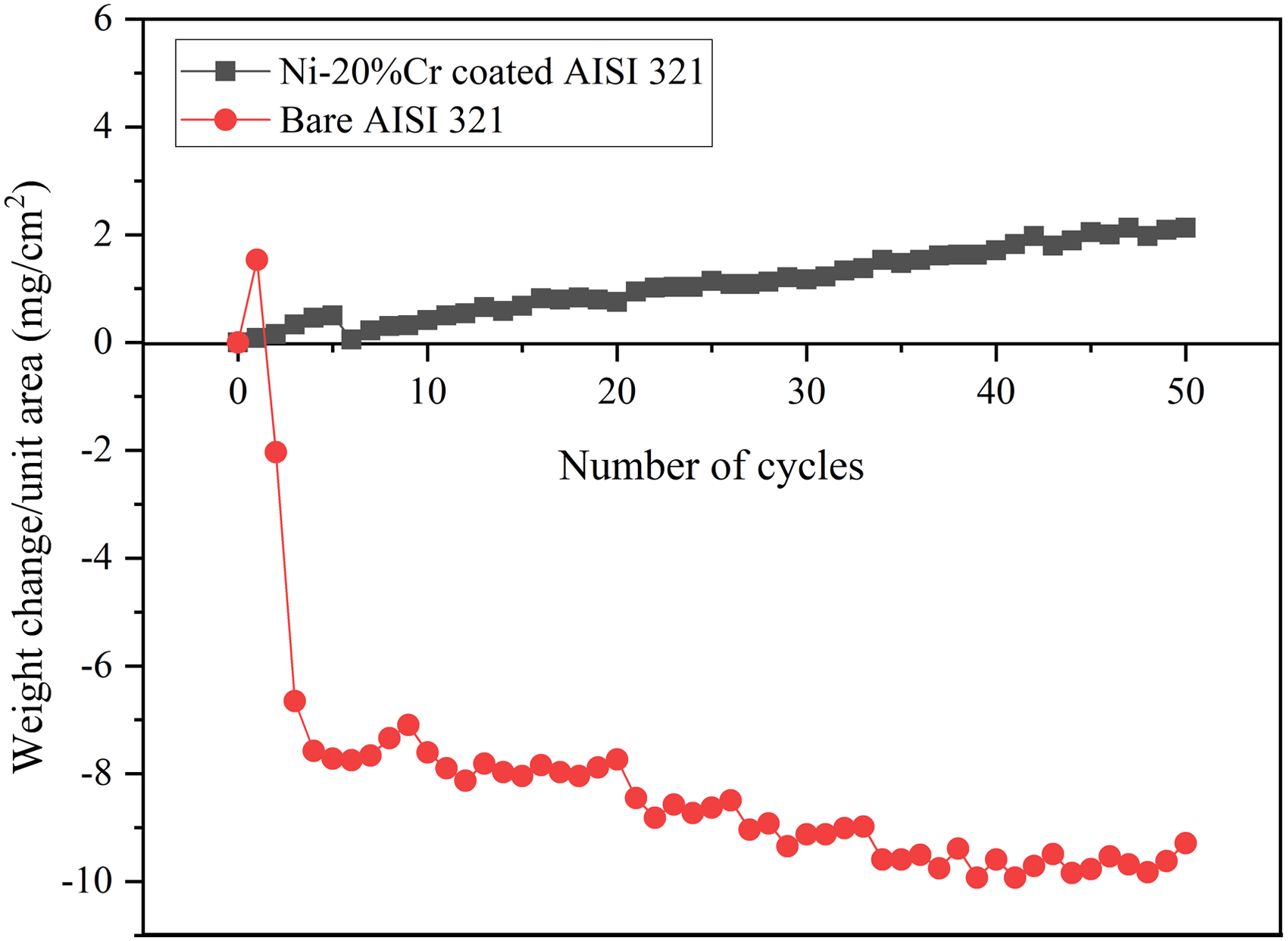
Corrosion kinetics of the specimens is established by the thermogravimetric technique. Figure 5 shows the thermogravimetric chart plotted for hot corroded stainless steel AISI 321. In the case of the uncoated specimen, more scales are spalled inside the alumina boat, which is also included during weight measurement. From the initial stage, weight loss was only noticed due to the oxide scale spalling outside of the crucible. Therefore, monitoring the weight gain and corrosion rate is challenging and difficult. From thermogravimetric analysis, weight gain is found on the coated specimen and less oxidation because of protective oxide scales formation. This suggested that the coated specimen exhibited excellent resistance to corrosion and showed a weight gain of 2.1285 mg/cm2 after 50 cycles. The following formula is used to calculate the change in weight and corrosion kinetics (Kp) of the coated steel.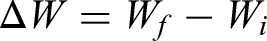
where ΔW is weight change, Wf is final weight, and Wi is initial weight. Thermogravimetric (weight gain/unit area vs number of cycles) plot for bare and coated steel after subjected to hot corrosion in Na2SO4–60%V2O5 environment at 900 °C for 50 cycles.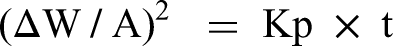
XRD analysis
The XRD analysis (Figure 6) shows that Fe2O3 (PDF # 330664) is the predominant phase on uncoated AISI 321, along with NiO (PDF # 893080), FeS (PDF # 653408), Cr2S3 (PDF # 654142), NiS (PDF # 653687), and Cr2O3 (PDF # 381479) are observed. NiO, Cr2O3, and NiCr2O4 (PDF # 850935) are the main phases, along with NiS, and Cr2S3 is detected in the coated specimen. The existence of Cr and Ni in the coating gets oxidized to form Cr2O3 and NiO layer, which protects the specimen from the corrosive medium. Due to the reaction of salt and the elements in the substrates, the corrosive phases were found.
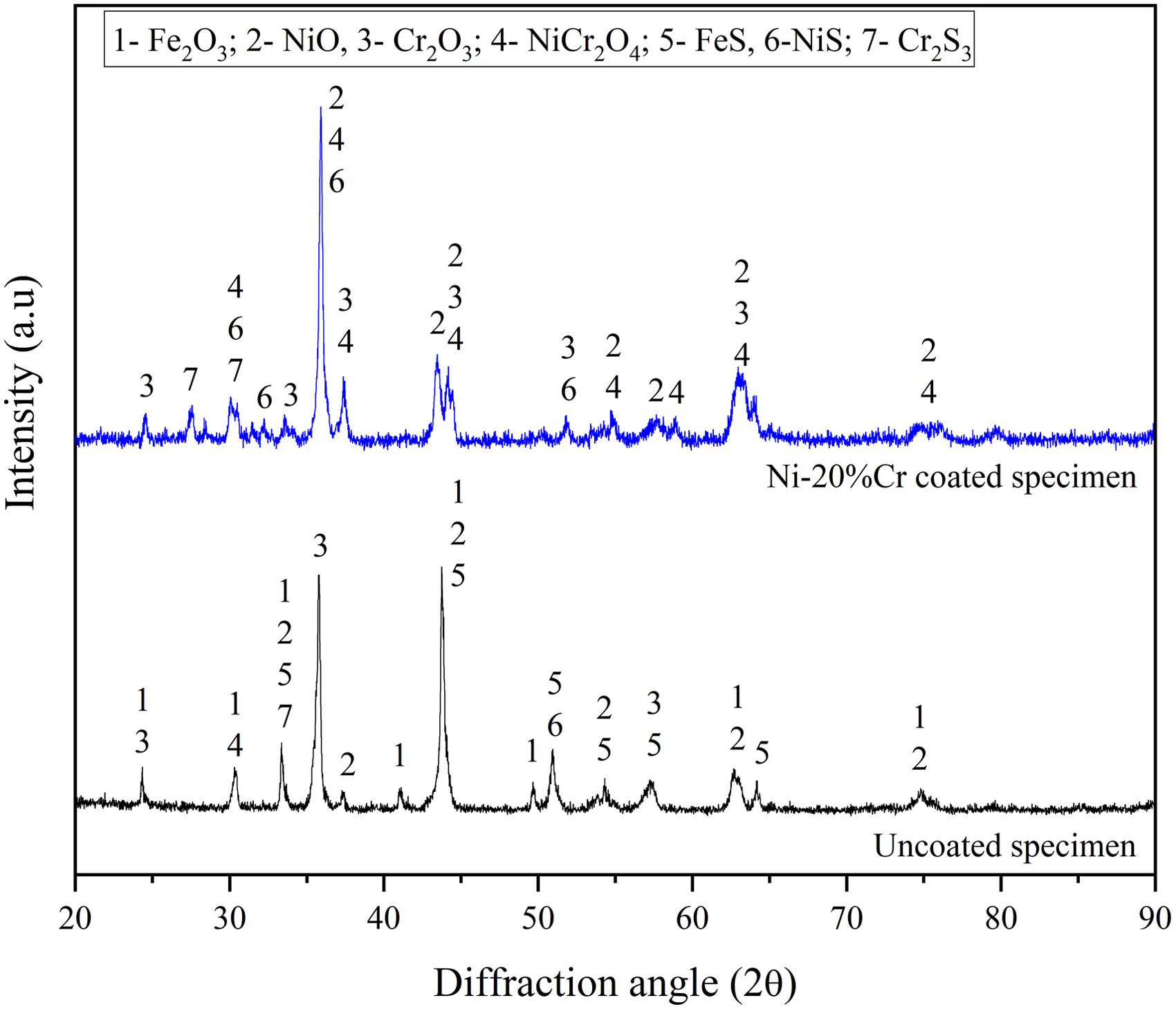
X-ray diffraction analysis of the hot corroded AISI 321 steel in Na2SO4–60%V2O5 environment at 900 °C.
SEM/EDS analysis
SEM surface microstructure of bare hot corroded steel (Figure 7) shows the rod shape structure. It is noted from the EDS analysis that Fe and O are the main elements, and also Ni, S, Na, Cr, and V are observed in minor amounts. This suggesting that Fe2O3 is the major scale along with minor NiO and Cr2O3. SEM microstructure of the coated steel (Figure 8) shows the irregular and rough morphology after corrosion. The EDAX analysis reveals that corroded coating mainly consists of O, Ni, and Cr, and S and Na are observed in minor levels. This divulges the possibility of NiO and Cr2O3 scales along with corrosive phases.

SEM/EDS analysis of the bare AISI 321 steel after hot corrosion in Na2SO4–60%V2O5 environment at 900 °C. SEM: scanning electron microscope.
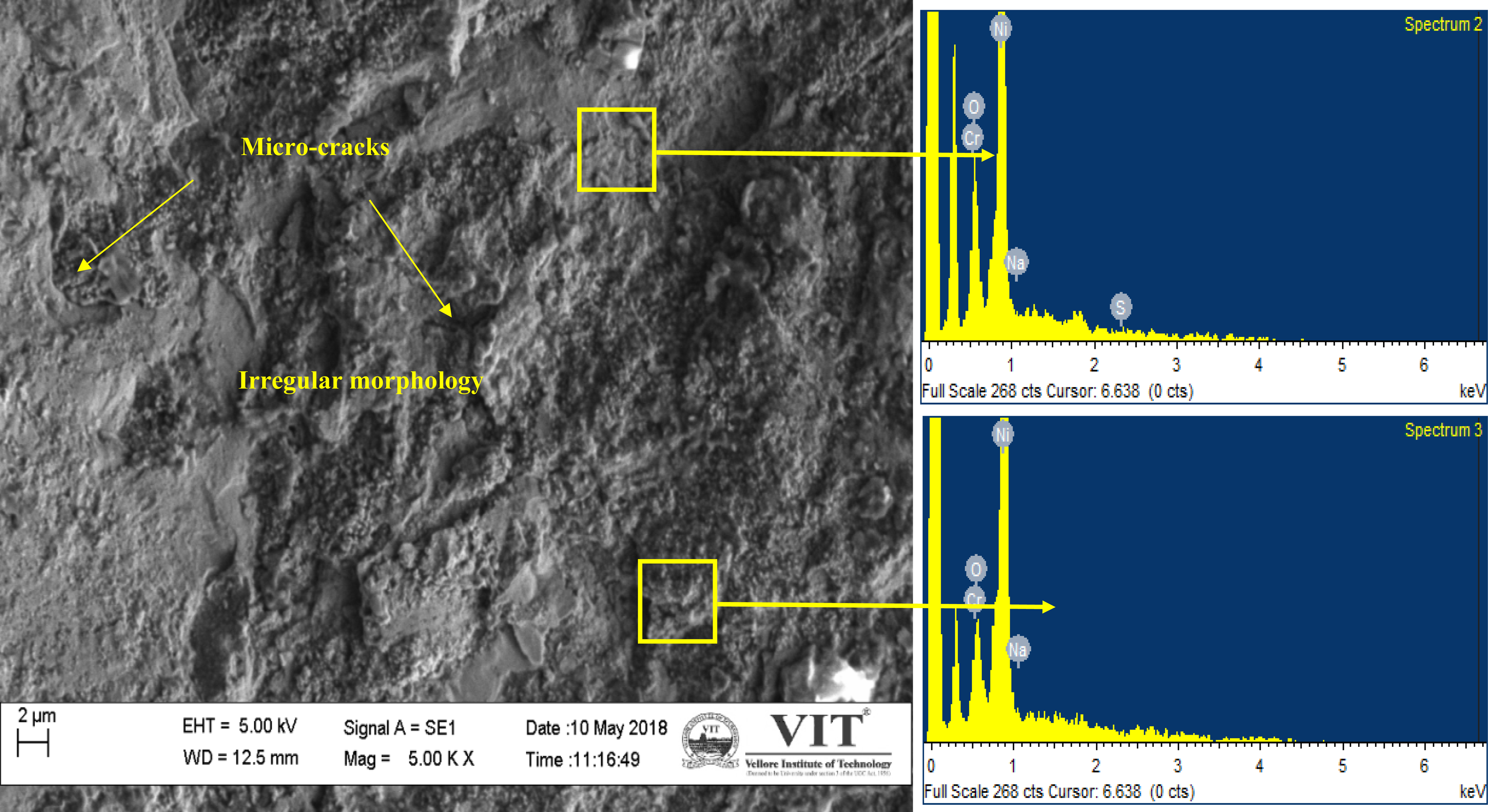
SEM/EDS analysis of the HVOF sprayed Ni–20%Cr coated AISI 321 steel after hot corrosion in Na2SO4–60%V2O5 environment at 900 °C. HVOF: high-velocity oxy-fuel; SEM: scanning electron microscope.
Cross-sectional analysis
SEM images of the corroded uncoated specimen at cross-section (Supplementary Figure 1) clearly show the scale, cracks, and spallation. The fragile scale was noticed on the surface with the thickness range of 20–30 µm. The bare steel's top surface contains major amounts of Fe, O, Ni, and Cr, and a higher amount of V (25.84%) is present, resulting in an increased oxidation rate. This inferred that the V plays a vital role in more oxidation than Na and S. The presence of O and V contents decreases with an increase in the depth of analysis. However, V and O were diffused in the specimens till 90 µm. Supplementary Figure 9(b) reports the SEM and EDS analysis of the coated steel at the cross-section. It is noticed from the EDS study that the scale contains Ni, O, and Cr, which may indicate the existence of NiO, Cr2O3, and NiCr2O4. In addition, corrosion products S and Na are observed. Oxygen levels is decreasing as the depth of analysis increased. The elements present in the scales are reported in Supplementary Tables S1 and S2.
X-ray mapping analysis
X-ray mapping was done to study the elements distribution in the corroded specimens, and the results are depicted in Supplementary Figures 10 and 11. The scales formed on the uncoated steel (Supplementary Figure 10) are fragile and additionally pores. Some minor cracks are observed, indicating that the steel's top scale is severely corroded by molten salt. X-ray mapping analysis shows that O, Fe, Ni, Cr, and V are highly distributed, and the existence of the minor amount of corrosion products of S and Na is in the cross-sections of the uncoated steel. The mapping result (Supplementary Figure 11) shows that the top region of coated steel contains O, Ni, and Cr, which supports the development of NiO and Cr2O3 scales along with Na and S are partially present. Fe is noticed in the coating interface region, which divulges Fe diffusion from the substrate.
Discussion
The powder form of Ni–20%Cr was deposited on the AISI 321 steel using the HVOF coating process. Besides, the corrosion characteristics of the steels were examined in a eutectic mixture of Na2SO4–60%V2O5 atmosphere at 900 °C. The macro images (Figure 4) clearly show that the uncoated specimen suffered severely from intense sputtering and spallation. More scales were developed on the specimen due to rapid oxidation. While cooling at room temperature, the scales were subjected to thermal stress as resulting the cracks were initiated.4,8 At elevated temperature, the eutectic mixture of salt is melted and then diffused into the substrate through the cracks in the layer, which further increases the oxidation rate of the specimen. After 50 cycles, the surface of the bare specimen has rough, irregular, and more pits.
Weight change of the specimens were used to determine the corrosion kinetics. In thermogravimetric plot (Figure 5), X-axis represents the number cycle (exposure time) and y-axis represents the weight gain per unit area. The weight loss of the uncoated specimen increases with increasing exposure time. There is no weight gain found from the initial period due to spallation. The weight loss of the uncoated specimen was 9.285 mg/cm2, which may be attributed to the scales that are spalled outside of the crucible due to cyclic heating and cooling.8,9 Due to more sputtering and spalling of scales in the bare steel, it is difficult to find the corrosion rate. This kind of similar result was noticed by Sidhu et al. 9 during the alloy 718 being exposed in salt condition at 900 °C. In the case of the coated specimen, gradual weight gain is noticed from the initial cycle, which indicates the oxide layers formed on the coating. No weight loss was observed till end of the corrosion test due to the development of protective scales. The coated steel exhibited the superior resistance to corrosion against Na2SO4–60%V2O5 salt condition. The maximum mass gained by the coated specimen after the 50th cycle is 2.1285 mg/cm2. The kinetics of corrosion (Kp) of the coated steel is 1.258 × 10−9 g2 cm−4 S−1.
The bare steel shows poor corrosion resistance due to higher amounts of sputtering and spallation of oxide scales. The coefficient of thermal expansion plays a major role in oxide scale spallation. The EDS result shows that the bare steel is rich in O and Fe along with less Ni and Cr. The reaction products Fe2O3, along with other NiO and Cr2O3 phases, were formed on the uncoated steel is indicated by XRD results. The following equations (1)–(3) clearly explains the oxide layer formation12,19,20: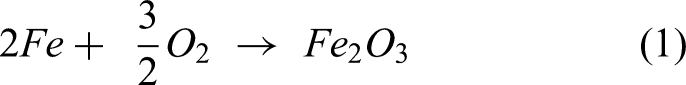
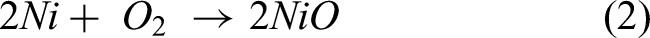
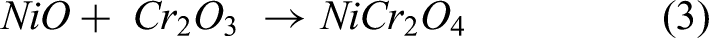
At 900 °C, salts of Na2SO4–60%V2O5 melts and combine to form sodium vanadate (NaVO3), which is explained by equation (4). The melting point of NaVO3 (610°C) is significantly less than our test temperature. During hot corrosion study, NaVO3 melts and initiates the fluxing mechanism and also leads to rapid oxidation, which is reported by Kotla et al.
6
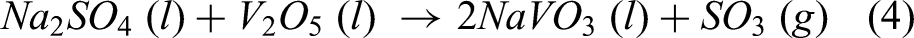
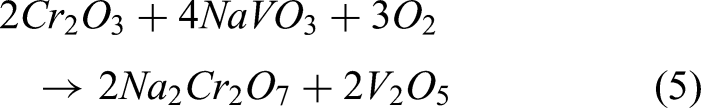
The HVOF sprayed steel shows the least weight gain and also provides better protection. There is no weight loss was found and also minor oxide scale spallation was observed than bare specimen during exposure. The EDS analysis reveals that the scales present on the coated steel indicate protective NiO, Cr2O3 and spinel NiCr2O4 scales, which are supported by XRD analysis. The formed scales are protective and act as a barrier to the diffusion of salts, which provides the necessary protection to the steel. The existence of NiO is partially porous in nature, and it might let the corrosive elements to react with coating. Protective Cr2O3 scale was formed on the coated specimen, which is stable till 900 °C and has high melting point, which does not permit the corrosive species. XRD analysis confirmed that the spinel NiCr2O4 oxide was observed on the coated, which also provides the necessary protection to the specimen.21,25
Although the Cr2O3 layer is stable till 900 °C, its high melting point does not allow the penetration of corrosion products. However, XRD analysis supported the occurrence of spinel oxide NiCr2O4 in coated steel, providing the necessary protection to the specimen
The reaction between the sulfur from Na2SO4 and elements in the substrate and coating leads to form the metal sulfides explained by the following equations (6)–(9). The XRD analysis confirmed NiS, FeS, and Cr2S3 in the specimens after exposure to the salt atmosphere. In the case of bare specimen, the sulfidation takes place, followed by oxidation through the pores and cracks in the specimens. Therefore, the bare specimen is severely prone to sulfur attack causing more spallation. The coating significantly prevents the oxygen and salt penetration by NiO and Cr2O3 scales, causing no sulfides in the coating-substrate interface. However, metal sulfides were found in the top region.15,24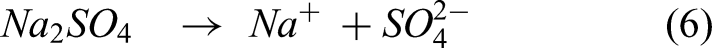
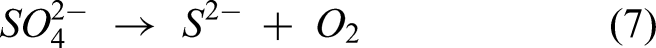
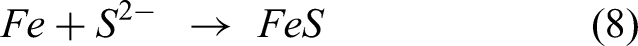
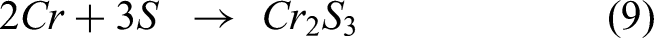
Conclusion
HVOF coating has successfully been employed to deposit Ni–20%Cr powders on AISI 321 steel in the 230–240 µm thickness range. The cyclic corrosion performance of the HVOF and bare steel AISI 321 is examined in Na2SO4–60%V2O5 salt condition at 900 °C. The following conclusions were drawn from the test results:
The bare steel is severely susceptible to corrosion attack, and weight loss could be witnessed during exposure due to nonprotective Fe2O3 scale formation, which might induce intense spalling and sputtering of scale. The cross-sectional analysis indicates that the fragile scales are formed on the uncoated specimen in the range of 20–30 µm. Furthermore, the EDS method clarified that the corrosion products were diffused into the specimen till 80–90 µm. Ni–20%Cr coating on steel provides better preservation against corrosion and spallation, owing to the formation of protective NiO, NiCr2O4, and Cr2O3. These scales act as a barrier to the diffusion of salts into the scales resulting in no corrosion attack in the substrate region. A very thin layer was found in coated steel, and no more corrosion attack was noticed in the coating. Fe is partially present in the coating interface region, suggesting that Fe diffusion from the substrate. Both acidic and basic fluxing mechanisms were found. In addition, high-temperature oxidation and sulfidation occur in vanadium and sulfur environment. Corrosion performance of the materials are closely related to the presence of alloying elements. High Cr in the Ni–20%Cr coating accelerated the formation of thin Cr2O3 passive layer, which resists the further oxidation by salt. High Fe in the bare steel promoted the nonprotective Fe2O3, resulting in more oxides growth and severe damage.
Supplemental Material
sj-docx-1-pie-10.1177_09544089221143341 - Supplemental material for Microstructure and hot corrosion characteristics evaluation of high-velocity oxy-fuel sprayed Ni–20%Cr coating on AISI 321 steel with Na2SO4–60%V2O5 salt deposits
Supplemental material, sj-docx-1-pie-10.1177_09544089221143341 for Microstructure and hot corrosion characteristics evaluation of high-velocity oxy-fuel sprayed Ni–20%Cr coating on AISI 321 steel with Na2SO4–60%V2O5 salt deposits by Shanmugam Mannan Muthu, Moganraj Arivarasu, Babu Rebin Sanford, Veeman Dinakaran and Shubham Gupta in Proceedings of the Institution of Mechanical Engineers, Part E: Journal of Process Mechanical Engineering
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
