Abstract
Specimens of AA2099-T8 were immersed for different times ranging from 2 to 180 min in 0.1M NaCl. The development of corrosion around intermetallic particles was studied by scanning electron microscopy (SEM) with energy-dispersive X-ray spectrometry (EDXS). The degradation evolution of each major intermetallic particle after exposure of the alloy to electrolyte allowed to determine which particle corroded during the first stages of immersion. The corrosion of lithium-containing particles was also investigated by transmission electron microscopy (TEM). The earliest stages of attack started with localised corrosion of Al2CuMg (S-phase) particles resulting in dealloying which was followed by trenching around these particles. After 10 min, trenching was observed around Al7Cu2Fe(Mn) particles and then progressed to AlCuFeMnSi particles after 90 min.
Introduction
Aluminium alloys are a relatively low-cost, light-weight metals which can undergo heat treatment and can be loaded to relatively high level of stresses [1]. Lithium-containing aluminium alloys are promising substitutes for conventional aluminium alloys (2024-T3 or 7075 T6) in the aerospace industry regarding the improvement of payload and fuel efficiency of aircraft [2]. Al–Cu–Li alloys have received increasing attention during the last two decades due to their low density, high strength and excellent fatigue crack resistance, according to Lei et al. [3]. Adding 1 wt-% of lithium will contribute to a 6% increase in the elastic modulus and to a 3% decrease in weight [4]. However, the presence of lithium has a significant effect on the corrosion performance of aluminium alloys [3,5–8].
According to Prasad et al. [5] and Szklarska-Smialowska [9], intermetallic particles play a major role in the pitting corrosion of aluminium alloys. The microstructure, in addition to influencing the corrosion of the alloys, determined the mechanical properties, particularly the type, size, volume fraction and distribution of precipitates within the grains and at the grain boundaries [5,10,11]. Prasad et al. [5] indicated that more varied precipitation is enabled by the addition of copper and magnesium to binary Al–Li alloys. The addition of copper favoured θ (Al2Cu) and T1 (Al2CuLi) precipitation, while the addition of magnesium will increase the S-phase (Al2CuMg) precipitations. The balance between the S- and T1-phases in aluminium lithium alloys depends on the relative concentration of all three elemental additions: high Cu and Li contents relative to Mg result in the predominance of T1, while high Cu and Mg contents relative to Li result in the predominance of S-phase. From the works of Ma et al. [2], Prasad et al. [5] and Lei et al. [3], it is known that AA2099-T8 is constituted of ß′(Al3Zr) dispersoids, precipitates of T1 (Al2CuLi), δ′ (Al3Li), θ (Al2Cu), S-phase (Al2CuMg), Al20Cu2Mn3, Al7Cu2Fe(Mn) and T2 (Al6CuLi3). The S-phase was found to be the most predominant intermetallic particles, representing 60% of the total of the intermetallic, by Snihirova et al. [12] in their review. As this phase is very active in the presence of chloride or moisture, it will play an important role in the corrosion process of lithium–aluminium alloys.
The T1 phases were also identified as a critical actor in the corrosion process of lithium–aluminium alloys by Li et al. [13]. They observed that this phase was initially anodic to the alloy matrix during the early stages of corrosion but became cathodic due to the preferential dissolution of aluminium and lithium from the T1-phase.
In recent works, Ma et al. [2,14] observed that particles in AA2099 can be divided into two groups: high- and low-copper-containing particles. These particles can be single or multiphases. The high-copper-containing particles are usually surrounded by the low-copper-containing phase. Ma et al. [2] showed by electron energy-loss spectroscopy (EELS) that the content of lithium was reduced for the low-copper-containing phase particles in comparison to the high-copper-containing phase. They concluded that trenching around the particles is the main mode of degradation induced by localised corrosion associated with low-copper-containing Al–Fe–Mn–Cu (Li) particles. In the case of high-copper-containing phase, the trenching around the copper-rich particles was shown to happen only after the dealloying of aluminium and lithium from the particles.
Tao [7] indicated that particles containing Cu, Fe and Mn act as a cathode and promote matrix dissolution at their periphery while particles containing Mg (e.g. Al2CuMg) show anodic behaviour and dissolve with preferential dealloying of Mg and Al. The trenching of the matrix surrounding intermetallic particles may be interpreted as a galvanic corrosion between the matrix and the intermetallic particles [7,15]. This observation was confirmed by Ma et al. [2] who showed that the importance of the galvanic coupling effect was increasing with the increase of the concentration of copper in the particle.
According to Luo et al. [10], the oxygen reduction reaction (ORR) takes place preferentially on the copper-rich intermetallic particles such as S-phase (Al2CuMg), θ-phase (Al2Cu), α-phase (Al–Cu–Fe–Mn–Si) and T1-phase (Al2CuLi). By immerging different aluminium–lithium alloys in 4% NaCl solution, they showed that the T1-phase actively undergoes severe lithium dealloying leading to the switching from an anode to a cathode behaviour after the enrichment of the particle with copper. The precipitate remnant and corroded intermetallic particles induced dissolution in the periphery of the particles as it was demonstrated by Tao [7].
Zhu et al. [16] reported that the trenching around clusters of particles and particle dissolution serves as the basis of metastable and stable pit formation. From Lei et al. [3], the development of the pitting process can be divided into an initial slow nucleation stage (stage I), a fast development stage (stage II) and a stabilised growth stage (stage III). The accumulative number of pitting sites rises more quickly with an increased content of lithium.
The microstructure of conventional aluminium alloys has been studied in several papers [16–19], but only few studies have been conducted on the localised corrosion of isolated intermetallic particles (IMPs) on AA2099-T8 [5,20].
The aim of the present work is to examine how corrosion develops on AA2099-T8 during the early stages of exposure to chloride solution. Consequently, the microstructure of AA2099-T8 and more precisely the corrosion behaviour of intermetallic particles were studied using scanning electron microscopy (SEM) equipped with energy-dispersive X-ray spectrometry (EDXS), scanning transmission electron microscopy (STEM) with EDXS and transmission electron microscopy in the bright field (TEM-BF).
Materials and method
Samples preparation
Chemical composition of aluminium alloys determined by ICP-OES (wt-%).

This alloy has been heat treated, cold worked for strain hardening and then artificially aged to achieve precipitation hardening. It is, therefore, characterised by a T8 temper.
The samples of 2099-T8 with a size of about 1.1 × 1.2 cm were embedded in an IP epoxy resin, grinded (SIC 220) and polished (down to 0.04 µm diamond paste). The polished samples were then cleaned ultrasonically in ethanol.
Corrosion initiation: Immersion test
Polished specimens of AA2099-T8 were immersed in aerated conditions, horizontally at ambient temperature (i.e. 20°C ±1) for different times ranging from 2 to 180 min in 0.1M NaCl solution prepared from analytical grade chemicals and deionised water. After immersion, the samples were removed, rinsed in deionised water and dried in laboratory air.
Scanning electron microscopy
SEM investigations were performed using a Hitachi SU3500, with EDXS ThermoScientific NSS312. All specimens were carbon coated at about 50 nm.
Secondary electron and backscattered electron imaging were performed using beam energies of 20 kV and probe currents of approximately 140-154 pA.
From X-ray spectra obtained by X-ray spectrometry, the composition of each particle was identified.
Transmission electron microscopy
The two types of TEM were used to investigate the corrosion of lithium-containing particles. A STEM-EDX CM20feg and TEM-BF HF 3300 with a Hitachi microscope. Secondary electron and backscattered electron were performed using beam energies of, respectively, 200 and 300 kV.
Electrochemical investigations
The open-circuit potential (OCP) in immersion was followed during the duration of the test (e.g. 3 h) on AA2099-T8 in 0.1M NaCl using a Gamry R600 potentiostat and a saturated reference electrode. Two runs were performed on the alloy.
Results and discussion
A first evaluation was performed after 2 min of immersion. At this time, no corrosion on the surface of AA2099-T8 was noticed. It was necessary to wait 5 min to observe localised corrosion and only the S-phase (Al2CuMg) was corroded. The corrosion process results in a dealloying followed by trenching. This process was completed after 7 min as presented in Figure 1.
(Left) Secondary and (right) backscattered electron images of Al2CuMg particles after 7 min exposure to 0.1M NaCl.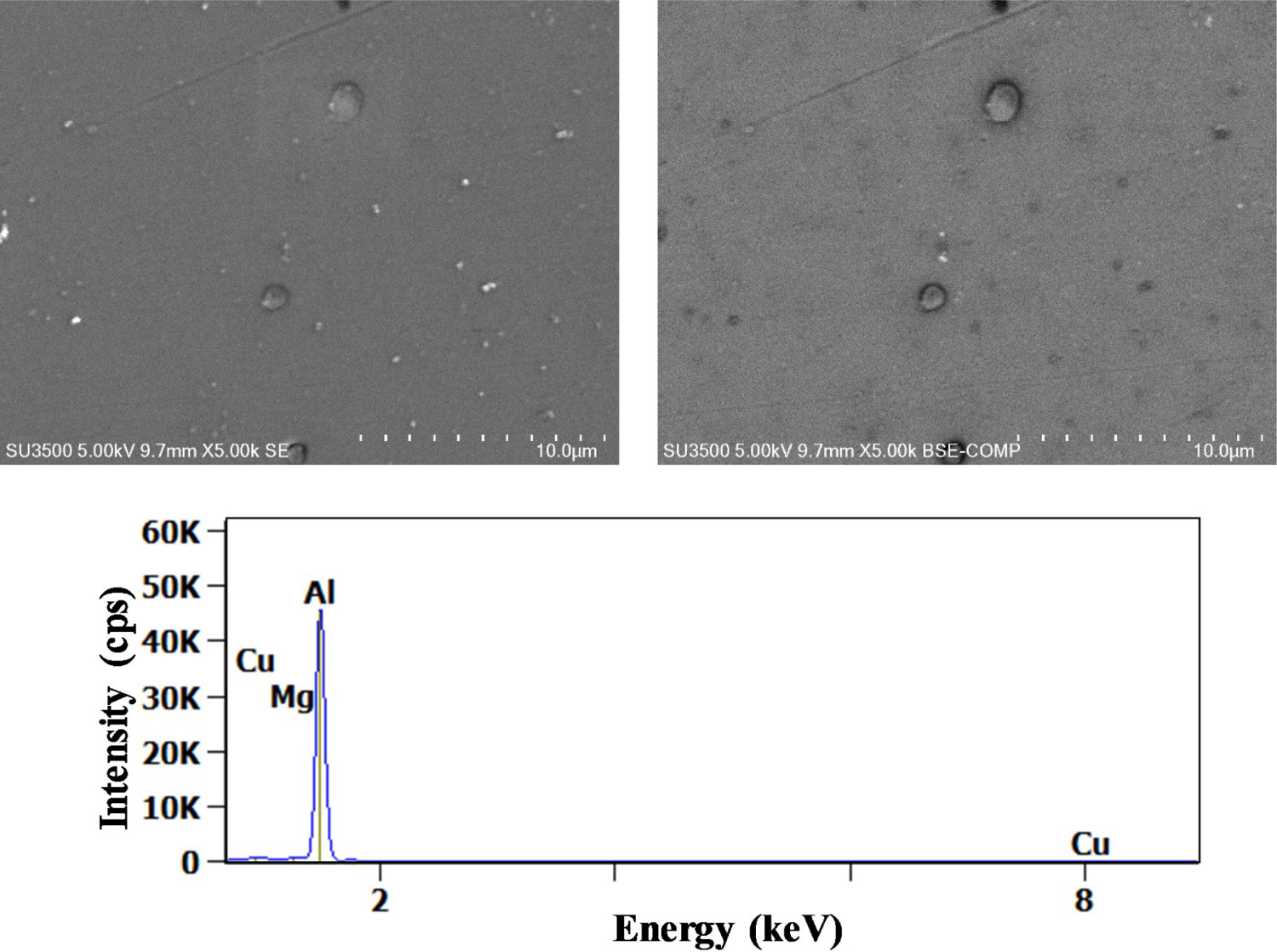
According to Snihinova et al. [12], the S-phase can act both as an anode (dealloying) and a cathode (trenching). Once, the dealloying was completed, the attack was located in the matrix around the particle with a preferential dissolution of aluminium (trenching). Hence, the development of trenching indicates that the particles act as active cathodes. As explained by Boag et al. [19], the activity of these sites can be due to the difference in the electrochemical behaviour between the S-phase and the matrix of AA2099-T8. Indeed, they showed with OCP measurements in 0.1M NaCl that this phase was more positive than the surrounding matrix [19].
After 7 min, the S-phase particles are replaced with a Cu-rich remnant as illustrated in the literature [12,16,19]. When trenching around the S-phase was completed, corrosion activity started at other intermetallic particles, such as those rich in Fe or Mn. The OCP measurements were carried on AA2099-T8 for 3 h. As shown in Figure 2, after 9 min, the aluminium alloy AA2099-T8 reached a stable OCP at about −703 ± 3 mV in accordance with the work of Carrick et al. [21].
Measurement of the corrosion potential of AA2099-T8 immersed in 0.1M NaCl during 3h.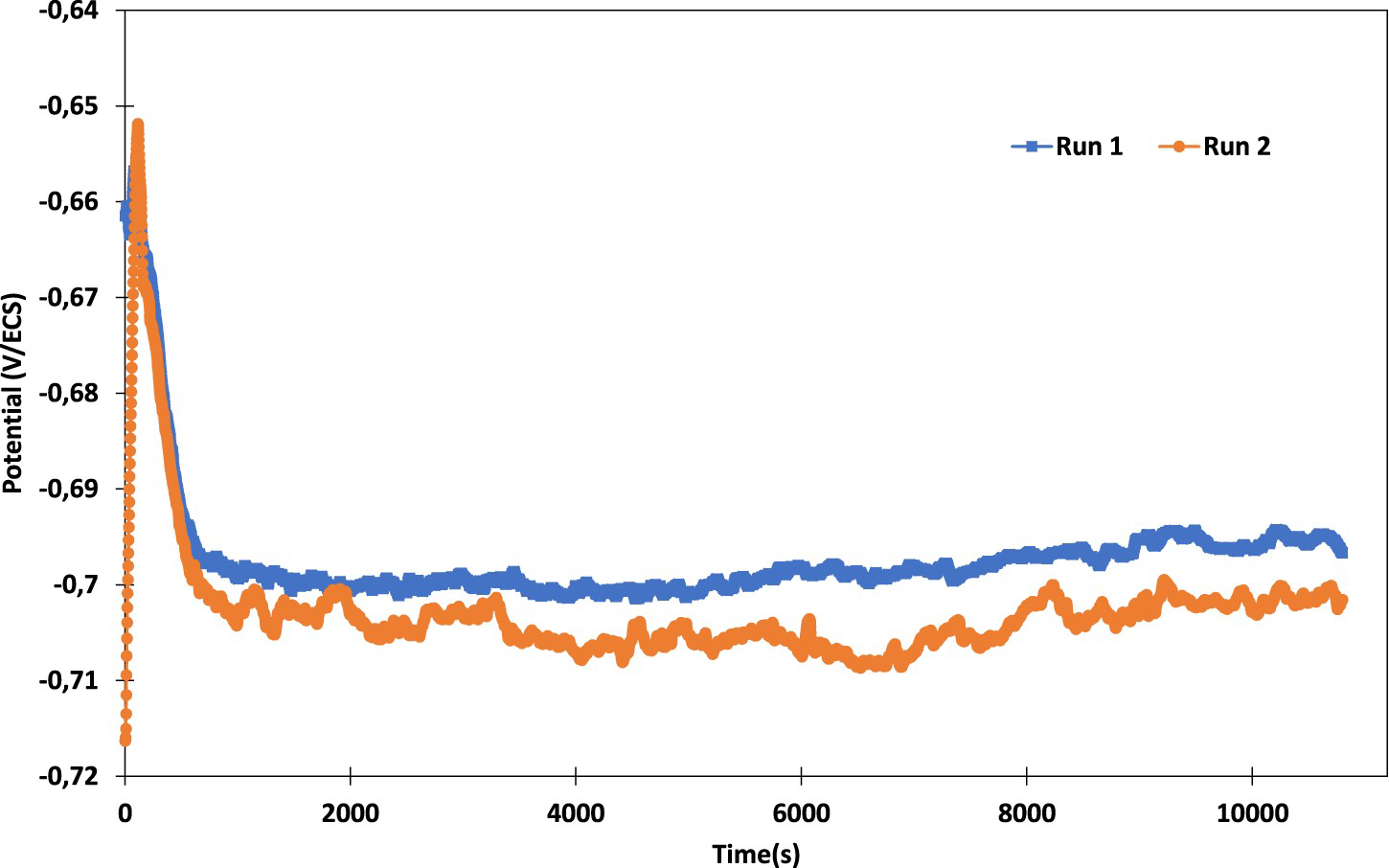
After 10 min, trenching was visible on Al7Cu2Fe(Mn) particles (see Figure 3), while it took about 30 min to observe this phenomenon for Al7Cu2Fe particles (see Figure 4). The influence of the particle's nature on the time for trenching occurrence is in agreement with the literature. Indeed, it was shown that the efficiency of the cathodic reaction was dependent on the Cu/Fe ratio.
(Left) Secondary and (right) backscattered electron images of Al7Cu2Fe(Mn) particles after 10 min exposure to 0.1M NaCl, X-ray spectrum for the intermetallic particle. (Left) Secondary and (right) backscattered electron images of Al7Cu2Fe particles after 30 min exposure to 0.1M NaCl, X-ray spectrum for the intermetallic particle.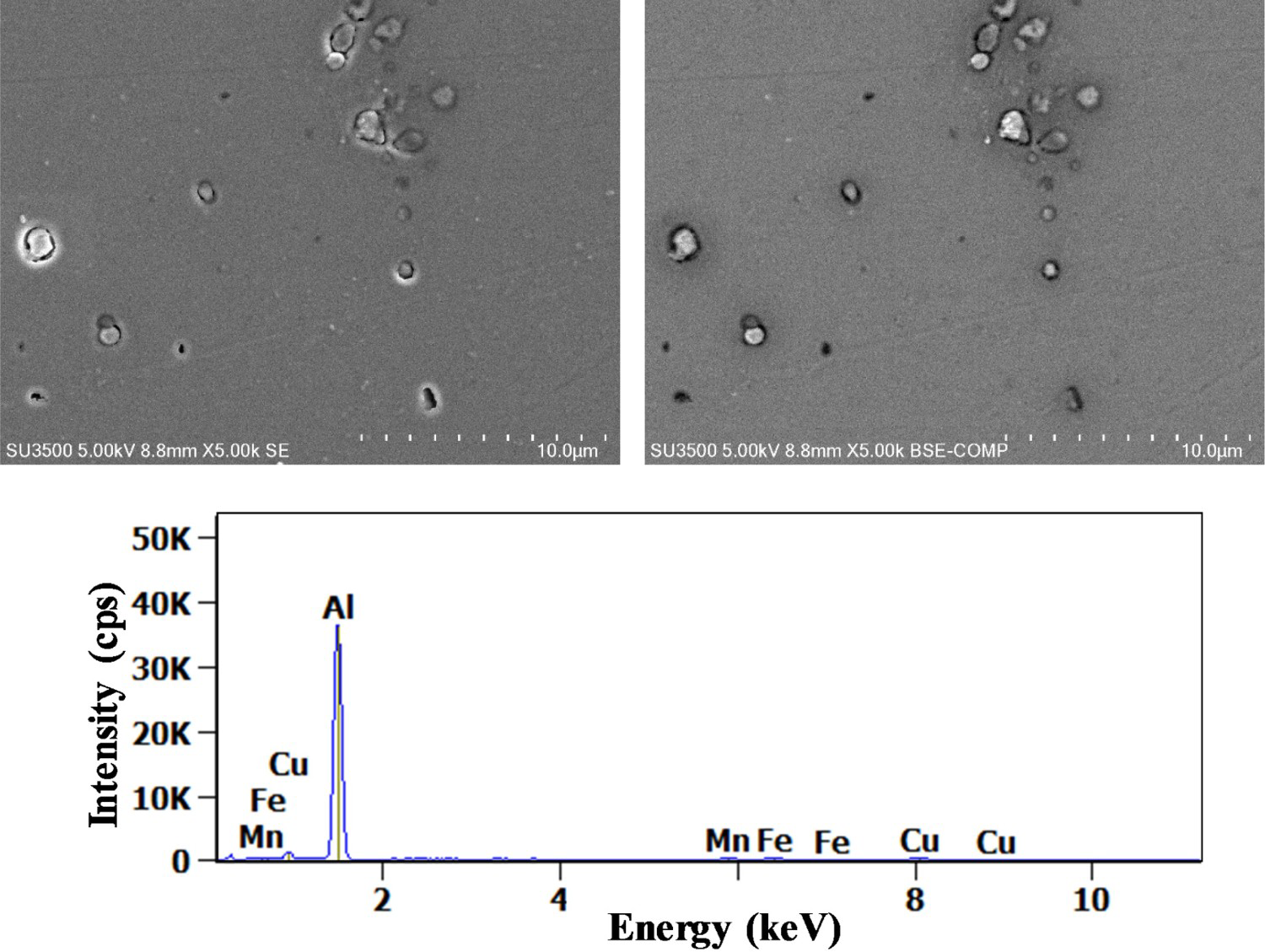

According to Boag et al. [19], two groups of intermetallics can be defined. One group of intermetallic particles are classified as (Al,Cu)
x
(Fe,Mn)
y
Si particles. This group of intermetallic particles tend to have a low Cu/Fe ratio and variable Si contents. The second group contained Al,Cu,Fe and Mn elements and is defined by a higher Cu/Fe ratio. From Zhu et al. [16], it appears that faster trenching is observed for particles from the second group due to a more efficient cathodic reaction in comparison to particles of the first group. Figure 5 compares the difference of the Cu/Fe ratio of Al7Cu2Fe, Al7Cu2Fe(Mn) and (Al,Cu)
x
(Fe,Mn)
y
Si IM particles after 90 min of immersion. Al7Cu2Fe and even more Al7Cu2Fe(Mn) exhibited a higher Cu/Fe ratio which is in agreement with Boag et al. [19] and Zhu et al. [16] observations.
Cu/Fe atomic ratios for Al7Cu2Fe, Al7Cu2Fe(Mn) and (Al,Cu)
x
(Fe,Mn)
y
Si IM particles after 90 min of immersion.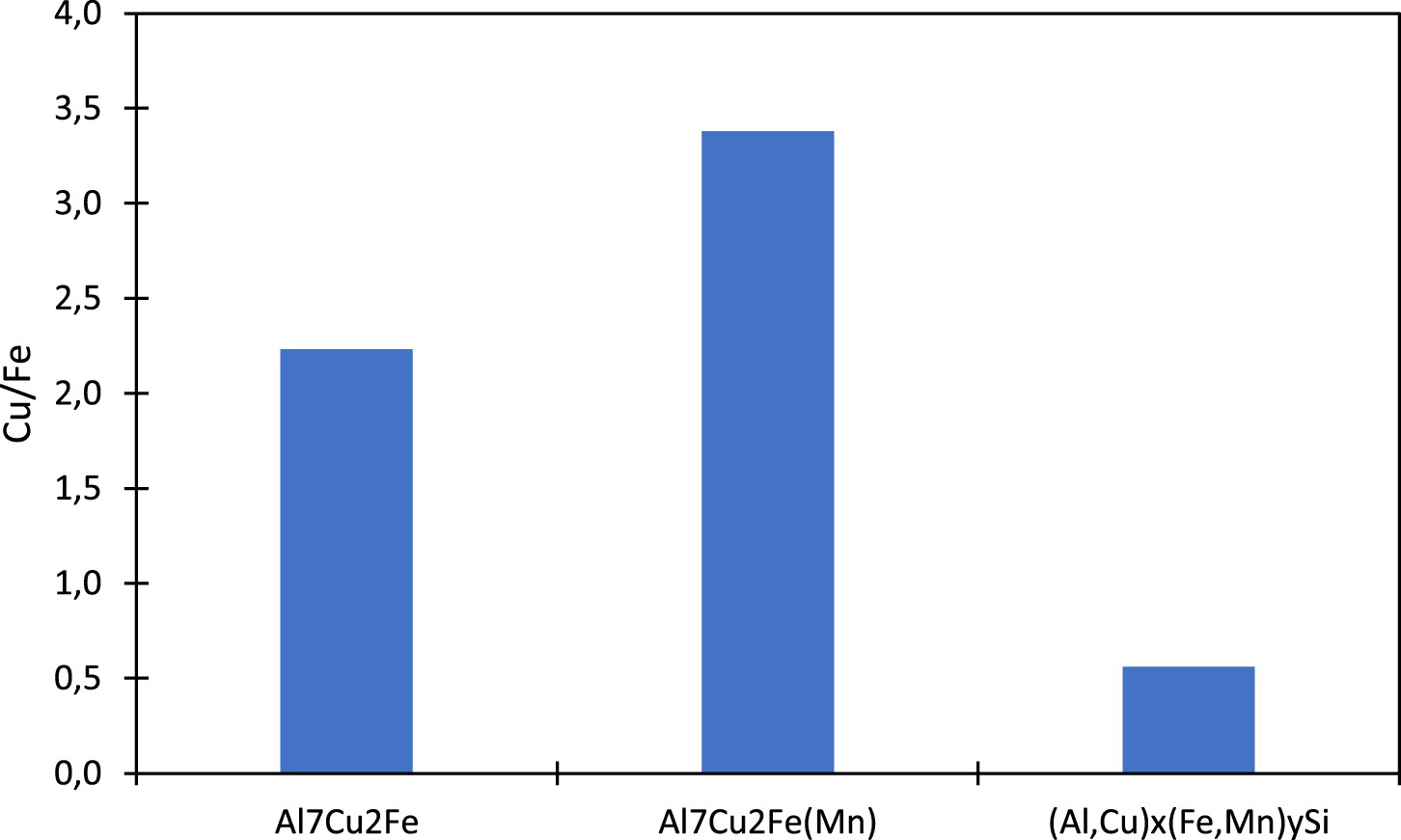
Another phenomenon observed during the corrosion of the AA2099-T8 was the copper enrichment on Al7Cu2Fe(Mn) particles. This enrichment happened between 7 and 90 min with an increase of Cu content from 3.2% to 11% as shown in Figure 6. These results are in good agreement with the literature [12,19]. According to the different authors, this enrichment was due to the initial aluminium dissolution. Consequently, after or during the selective aluminium dissolution, some areas enriched in copper exit and will be the preferential site of reduction reaction of oxidising species.
Highlighting of the copper enrichment as a function of the immersion time on Al7Cu2Fe(Mn) particles.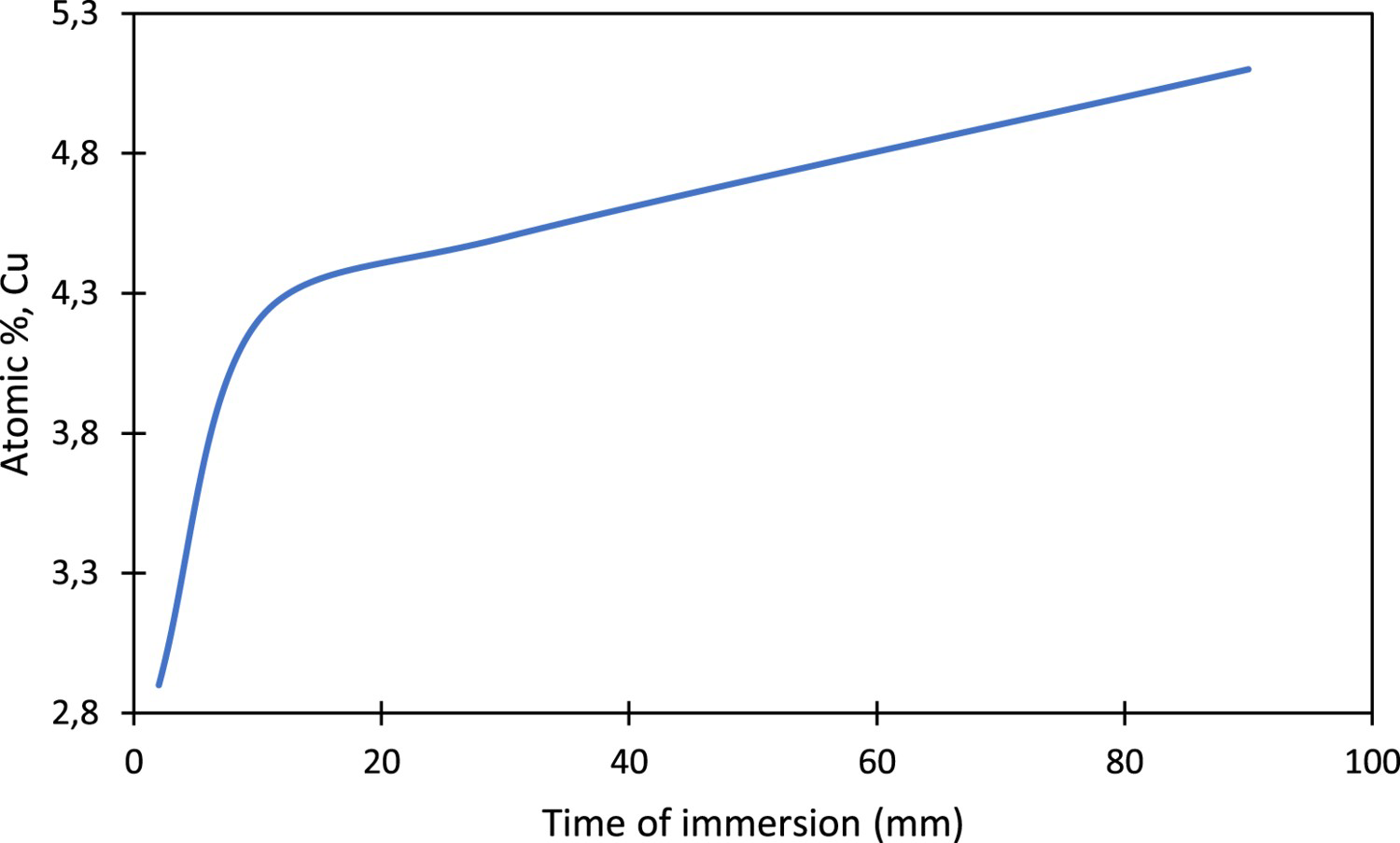
It should be noted that TEM investigations were also performed on the alloy AA2099-T8. However, it was not possible to observe neither the distribution of lithium in the alloy, nor the time of the first attack as presented in Figure 7. The area of observation may be too limited or the distribution of lithium unequal to allow the observation of lithium. It is also possible that TEM resolution was not sufficient to observe lithium particles due to their extremely small size.
Transmission electron micrograph of 2099-T8 aluminium alloy.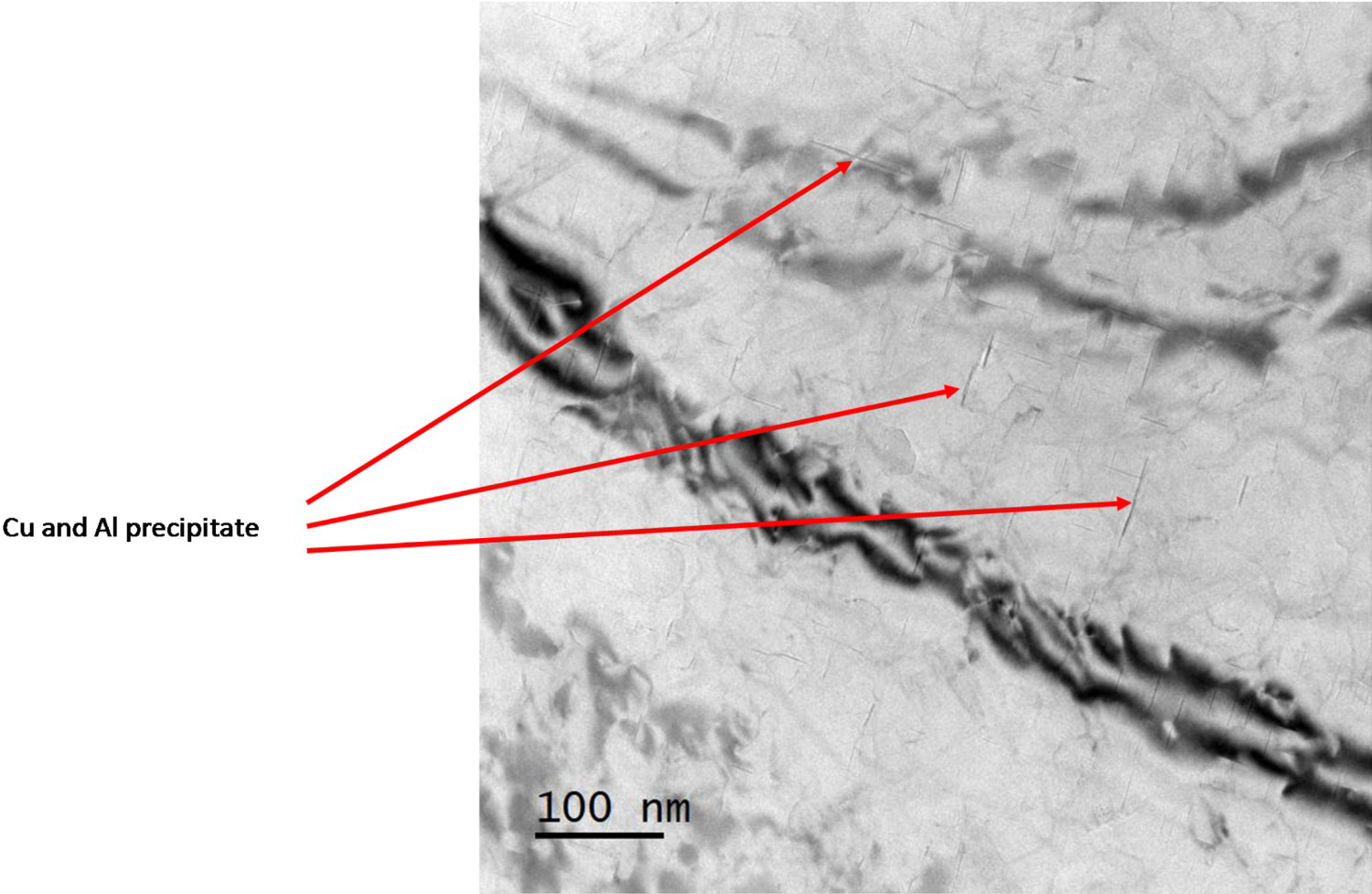
Nevertheless, according to Ma et al. [2], we can assume that lithium is present in the high-copper-containing particles. Consequently, the presence of a relatively high level of lithium and copper in this type of phase blocked the formation of a protective oxide film on the particles and allowed the selective dissolution.
Concerning AlCuFeMnSi particles, trenching occurred only after 90 min as presented in Figure 8.
(Left) Secondary and (right) backscattered electron images of (Al,Cu)
x
(Fe,Mn)
y
Si particles after 90 min exposure to 0.1M NaCl, X-ray spectrum for the intermetallic particle.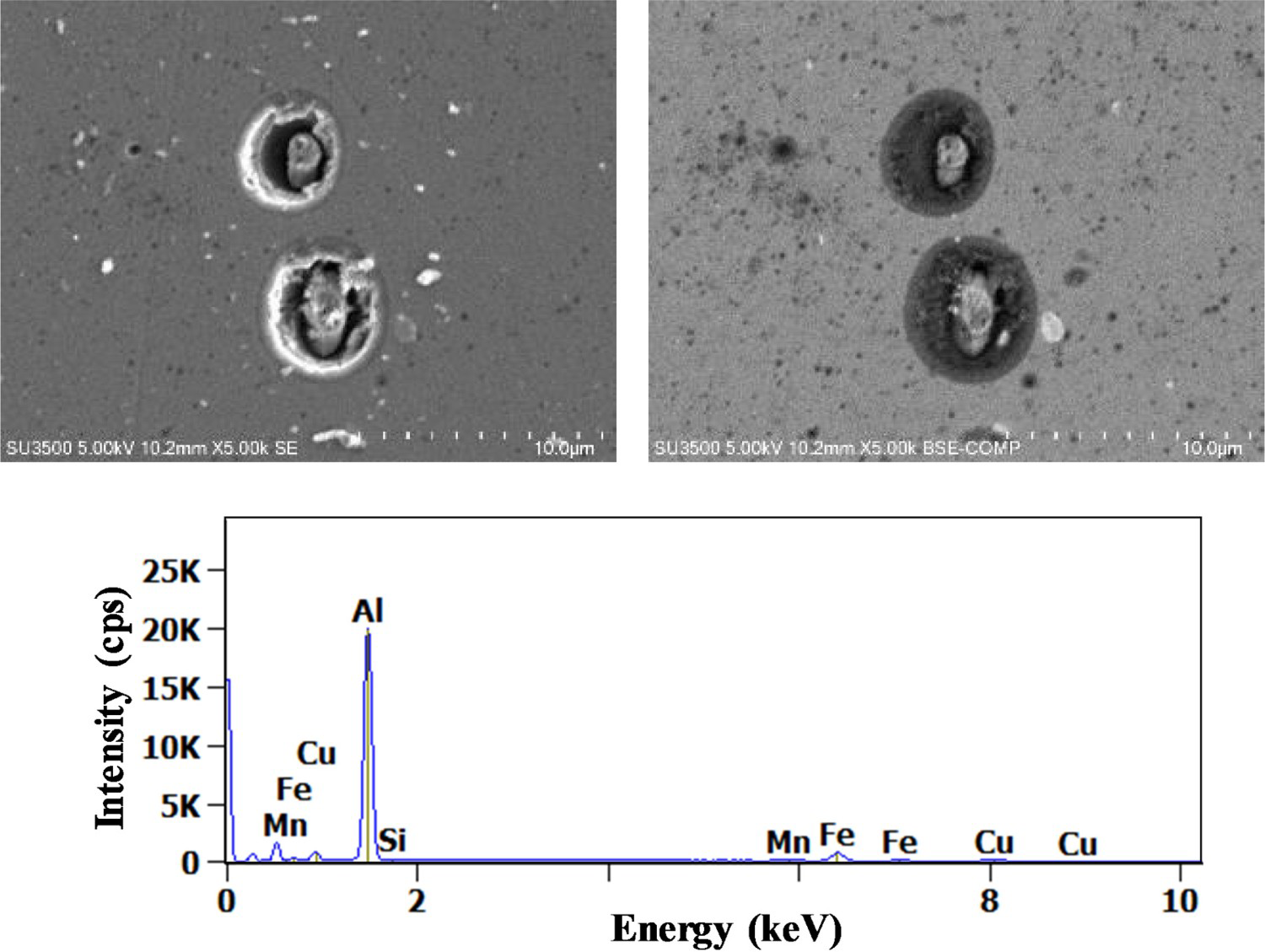
These results are in agreement with the work of Boag et al. [19] and Schneider et al. [22]. In addition, to have a low Cu/Fe ratio, as demonstrated previously, it was determined that the Si levels for these particles varied over time. Indeed, the Si levels in the (Al,Cu) x (Fe, Mn) y Si IM particles were around 2% after 7 min of immersion which is close to results observed by Boag et al. [19]. After 90 min, the Si content decreased to around 0.6%. According to the authors, Si dissolution mechanism can be explained by the destabilisation of the IM particles surface by the alkalinisation generated at cathodic sites when these particles became active sites of corrosion as both aluminium and silicon dioxides are soluble in alkaline media.
After 180 min, all particles were corroded except Al20Cu2(MnFe)3 particles as shown in Figure 9. This observation was in good agreement with Ilevbare and Scully [23] work which reported the resistance of this type of particle to self-dissolution. They highlighted a higher impedance in NaCl for Al20Cu2(MnFe)3, while Al2CuMg presented a relatively lower impedance indicating an active dissolution.
(Left) Secondary and (right) backscattered electron images of Al20Cu2(FeMn)3 particles after 180 min exposure to 0.1M NaCl, X-ray spectrum for the intermetallic particle.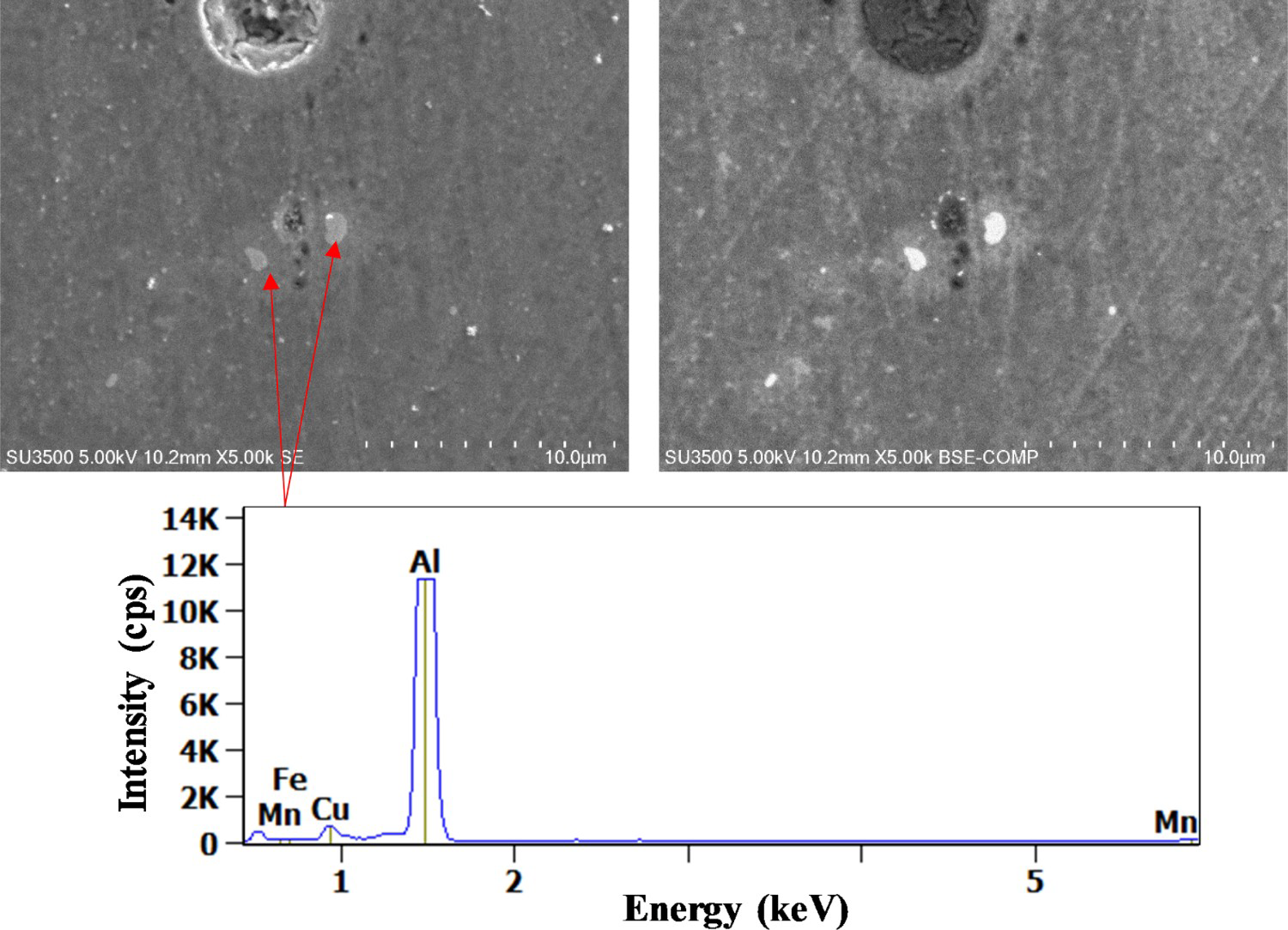
According to Karayan [24], these particles are formed at high temperatures and controlled recrystallisation behaviour and grain size. Li et al. [13] noticed that this type of particle acts as a cathode because of its relatively more positive corrosion potential compared with the surrounding matrix. Furthermore, it shows enhanced ORR kinetics when compared with pure aluminium. This is due to the presence of Cu clusters in the Al oxide film on the particle surface, which increases the conduction of electrons through the oxide film.
From these results, it was possible to determine a hierarchy of attack which appeared to be related to the activity of the IM particles as illustrated in Figure 10.
Hierarchy of localised corrosion attack observed on AA2099-T8 immersed for various times up to 180 min in 0.1M NaCl.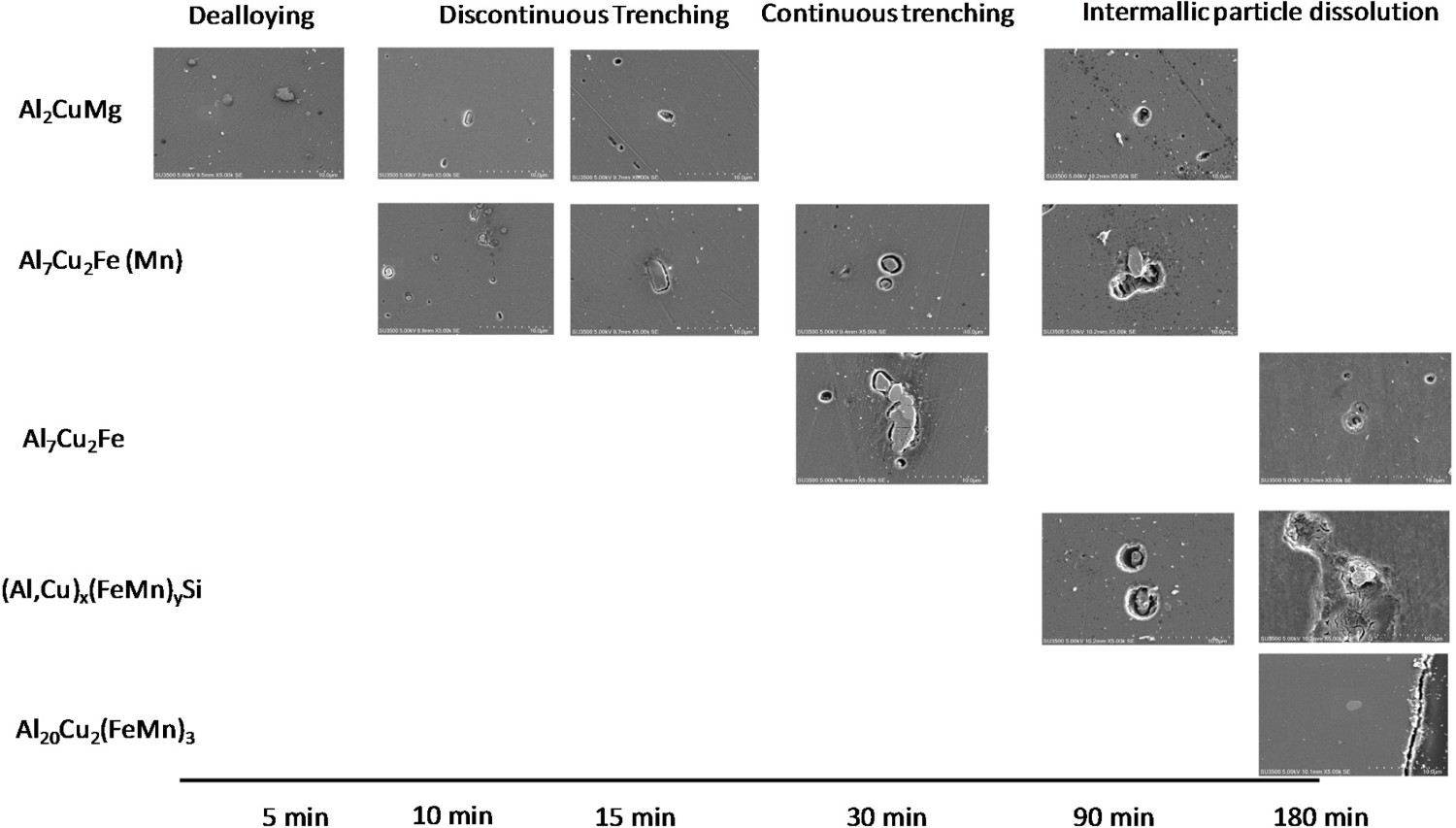
Conclusions
SEM studies of isolated intermetallic particles in AA2099-T8 revealed a range of localised corrosion process occurring at the surface in the first 180 min of immersion in 0.1M NaCl solution.
A progressive attack upon Al2CuMg and Al7Cu2Fe(Mn) then Al7Cu2Fe particles and finally (Al,Cu) x (Fe,Mn) y Si particles was evidenced. After 180 min, Al20Cu2(MnFe)3 particles were still intact. Despite the different techniques used in this work, it was not possible to precisely investigate the corrosion of lithium-containing particles during the first stages of immersion. This would be, therefore, a line of research for future work.
Footnotes
Disclosure statement
No potential conflict of interest was reported by the author(s).
