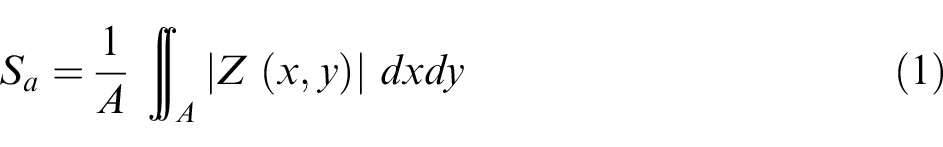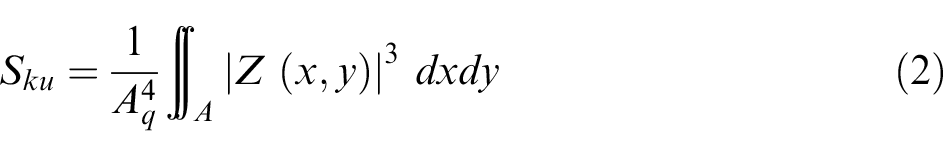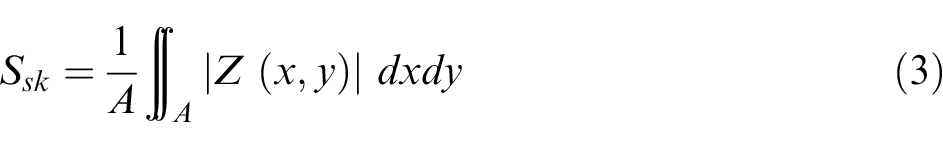Abstract
Metals such as titanium or Cr-Co alloys have been the most widely used materials in biomedical applications that require high mechanical properties, like implants. However, these materials present the disadvantage of releasing ion metals into the body. As an alternative, prostheses made of ceramic materials have been developed, as they produce less debris and have better durability. The aim of the present work is to test the biocompatibility of 3D-printed yttria-stabilized zirconia parts by culturing human bone-marrow-derived mesenchymal stem cells. Two different scaffols were 3D printed having a liner infill pattern, with 80 % and 95 % infill rate respectively. Results on surface roughness and biocompatibility tests confirmed that 3 mol % yttria-stabilized zirconia is a highly promising material as it presented high biocompatibility. In adition, similar results were obtained with or without the use of a type I collagen coating., which suggest that coating could be avoided when on zirconia substraes.
Keywords
Introduction
Metals have been the most widely used materials in biomedical applications that require high mechanical properties. Among the different utilizations, personalized medical implants 1 or dental ones 2 are the most common.
However, a major disadvantage of these materials is related to the release of ion metals. These may cause inflammatory and hypersensitivity reactions. 3 Additionally, a failure in medical implants such as hip or knee would require the removal of the device to implant a new one. On the other hand, research has also been carried out on 3D-printed metal-PLA (poly lactic acid) composite scaffolds for implants. 4 As an alternative, research in the use of ceramics is growing in popularity. Prostheses made of ceramic materials produce less debris and have better durability than those made of metals or plastics. 5
Amongst the different ceramics used in prosthetics (such as hydroxyapatite, 6 or alumina7,8), zirconia 7 offers several advantages: (1) reduction in the wear rate of implants 9 and the risk of toxicity 10 ; (2) excellent biocompatibility 11 and corrosion resistance 12 ; (3) outstanding mechanical properties. 11 Zirconia has a monoclinic structure at room temperature. As temperature increases, it changes to tetragonal and later to cubic structures. The transition between monoclinic and tetragonal leads to an important volume change. For this reason, yttria is often added to zirconia to stabilize it with a mix of tetragonal and cubic structures at room temperature. 13 Among the different parameters that can affect the performance of the ceramic grafts, surface roughness is of high importance. For example, in 3D printing processes lower layer height corresponds to lower lateral roughness. 14
Ceramics can be manufactured using different Additive Manufacturing technologies depending on the feedstock form according to Chen et al. 15 : (1) slurry-based (stereolithography -SLA-, digital light processing -DLP-, two-photon polymerization -2PP-, inkjet printing and direct ink writing -DIW-), (2) powder-based (selective laser sintering -SLS- and selective laser melting -SLM-), and (3) bulk-solid based (laminated object manufacturing -LOM- and fused deposition modeling -FDM-).
One of the most employed AM techniques for ceramics is Direct Ink Writing (DIW), an extrusion technique in which the ceramic paste is extruded and subsequently deposited layer-by-layer.16–18 It offers the advantage of being a cost-effective technique. Different ceramic materials can be printed with DIW, for example, kaolinite clay ceramics, 19 zirconia, 20 zirconia-toughened alumina, 21 or hydroxyapatite. 22
Yttria-stabilized zirconia (YSZ) is a biocompatible ceramic with high mechanical properties. A major advantage of YSZ is that it can be 3D printed to manufacture complex shapes such as prostheses or dental implants.23,24 Nevertheless, to the authors’ knowledge, few in vitro studies are known for proving the biocompatibility of 3D-printed YSZ.25,26 On the other hand, the surface finish of ceramics is known to affect their mechanical properties 27 and also influences their biocompatibility. 28 In addition, surface roughness is related to bacterial growth in dental implants. 29 The aim of the present research study is to culture human bone-marrow-derived mesenchymal stem cells (hBM-MSCs) on 3D-printed yttria-stabilized zirconia parts with different surface roughness values to assess their suitability to be used as biomedical material in implants.
Materials and methods
Zirconia parts preparation for cell culture
Zirconia pastes were prepared in a mixer. Mixing time was 2 min and rotational speed was 2000 min−1. Vacuum pressure was used to enable both dispersion of materials and the removal of bubbles (ARV-310P Thinky Corporation, Tokyo, Japan). Yttria-stabilized zirconia (YSZ) with a 3 mol% concentration (HSY-3B, Daiichi Kigenso Kagaku Kogyo Co., Ltd., Japan) and a 0.5 % wt dispersant (Dolapix PC75, Zschimmer & Schwarz, Germany) were mixed to a Pluronic F-127 (Sigma-Aldrich, UK) stock solution of 25 wt% concentration. The final solid percentage was 40 v%. The d50 value of the ceramic powder ranged between 0.7 and 1.5 μm (50% of the particles are smaller than that size).
Disk-shaped samples of diameter 10 mm and height 4 mm were printed (see Figure 1(a)) and subsequently sintered at 1550 °C, with a heating rate of 5 °C/min (see Figure 1(b)). The final dimensions of the disks were around 8.5 mm in diameter and 3 mm in height. As can be seen there is a shrinkage in the samples of 15% and 25% in the horizontal plane and vertical plane, respectively. A linear infill pattern of raster angle 0° was used, with different infill values of 80% and 95% respectively. Three replicates were considered for each experiment.

3D printed zirconia samples (a) before and (b) after sintering treatment.
A customized 3D printer was used, Dual Paste Extruder, from CIM-UPC (Figure 2). The nozzle diameter was 0.58 mm, the layer height was 0.3 mm, the printing speed was 5 mm/s and the extrusion multiplier was 100%.
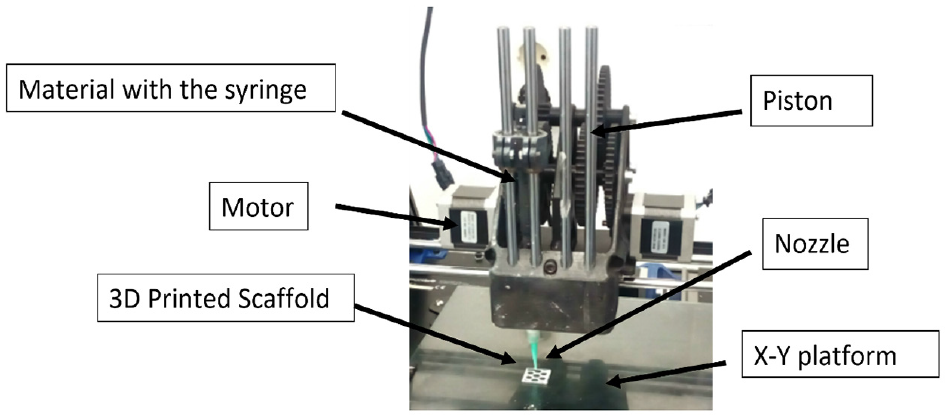
Printing head of the Dual Paste extruder from CIM-UPC.
Surface roughness characterization
According to the work by Deltombe et al., 30 the different roughness parameters can be divided into six groups: (1) amplitude, (2) spatial, (3) hybrid, (4) functional, (5) feature, and (6) other 3D parameters. In the present study, not only areal arithmetical mean has been studied, but also skewness (Ssk) and kurtosis (Sku) values were measured. Sa (equation (1)) is the average value of the heights, expressed as an absolute value, regarding the central plane. 20 Ssk (equation (2)) corresponds to the symmetry of the heights concerning the central plane. A positive Ssk value indicates the predominance of peaks, while a negative value corresponds to the predominance of valleys. 31 Kurtosis parameter Sku (equation (3)) is related to the peakedness of the surface. Sharp peaks and valleys correspond to Sku >3, while rounded peaks and valleys are related to Sku <3. 31 Both parameters are related to the friction control of surfaces. 32
A Smartproof 5 confocal microscope (Zeiss, Oberkochen, Germany) with a 20X magnification lens was used to measure areal roughness. Optical equipment was used to prevent damage to the samples and/or to the tip of a contact roughness meter. 33 The uncertainty of the microscope in the vertical direction is ± (0.1 µm + 0.008 × L), whereas in the horizontal direction it is ± (0.1 µm + 0.012 × L). Areal parameters were determined according to ISO 25178 standard. 34
where A is the measured area and Z (x, y) is a function that corresponds to the surface topography.
Roughness was measured on the upper surface of the disks, within an area of 0.5 ×0.5 mm2.
BM-MSCs culture
hBM-MSCs are commonly used in regenerative medicine and they are a widely accepted model to test the biocompatibility of new scaffold developments, as they have been used previously to test the biocompability of novel scaffolds based, for example, in silk composites. 35 hBM-MSCs were expanded and cultured following the manufacturer’s instructions (ATCC, VA). The medium was replaced every 2 days and cells were split from T-75 flasks after reaching 80% of confluence. Cells in passage 5 were used for this study. 3D printed zirconia scaffolds were sterilized by high pressure and vapor using an autoclave (Tomy SX-700E) before seeding the cells, and half of them were coated with type I collagen at a concentration of 0.1 mg/mL (Merck, US). Cells were seeded on the sterilized parts with a density of 4 × 104 cells/cm2 and cultured for 24 and 72 h. At the endpoint, samples were stained for DNA (NucBlue) to identify cell nuclei and imaged with a confocal microscope with a monitored X-Y stage (Nikon TI-HUBC, Japan) with a 10x objective.
Results and discussion
3D Printed samples shrinkage
The 3D printed samples showed, as mentioned before, a shrinkage of 15% and 25% in the horizontal plane and vertical plane, respectively. This is accordance with a previous study by Buj-Corral et al., 14 in which the shrinkage percentage values ranged between 19% and 28%. On the other hand, He et al. 36 3D printed using a DLP printer samples which displayed significant shrinkage after sintering, with the maximum shrinkage being 35.26%.
Surface roughness
Table 1 shows the average values of areal arithmetic mean, skewness, and kurtosis of both 80% and 95% infill experiments. Overall, both samples tested showed a smooth surface, without the presence of high peaks nor deep valleys. Figure 2.
Surface roughness of the upper surface of the yttria-stabilized zirconia samples.

The surface topographies showed that the different printed lines were fused, showing no holes among them. In Figure 3(a), several parallel crests can be observed, corresponding to the deposition of material along parallel lines in the linear structure. In Figure 3(b) only some crests can be observed, showing that, due to an excess of material, the paste became mixed. In Table 1, higher Sa values were observed for 95% infill (Figure 3(b)) than for 80% infill (Figure 3(a)). This suggests that, for a high infill value of 95% there was an excess of material that leaded to higher crests. A slightly negative value for Ssk was reported for 95% infill (Figure 3(b)), showing higher valleys than peaks. As for Sku, a value of 10.35 was obtained for 80% infill, corresponding to sharp peaks and valleys, with a lower value of 5.03 for 95% infill. In both cases, Sku was higher than 3, which would correspond to a normal distribution of heights.
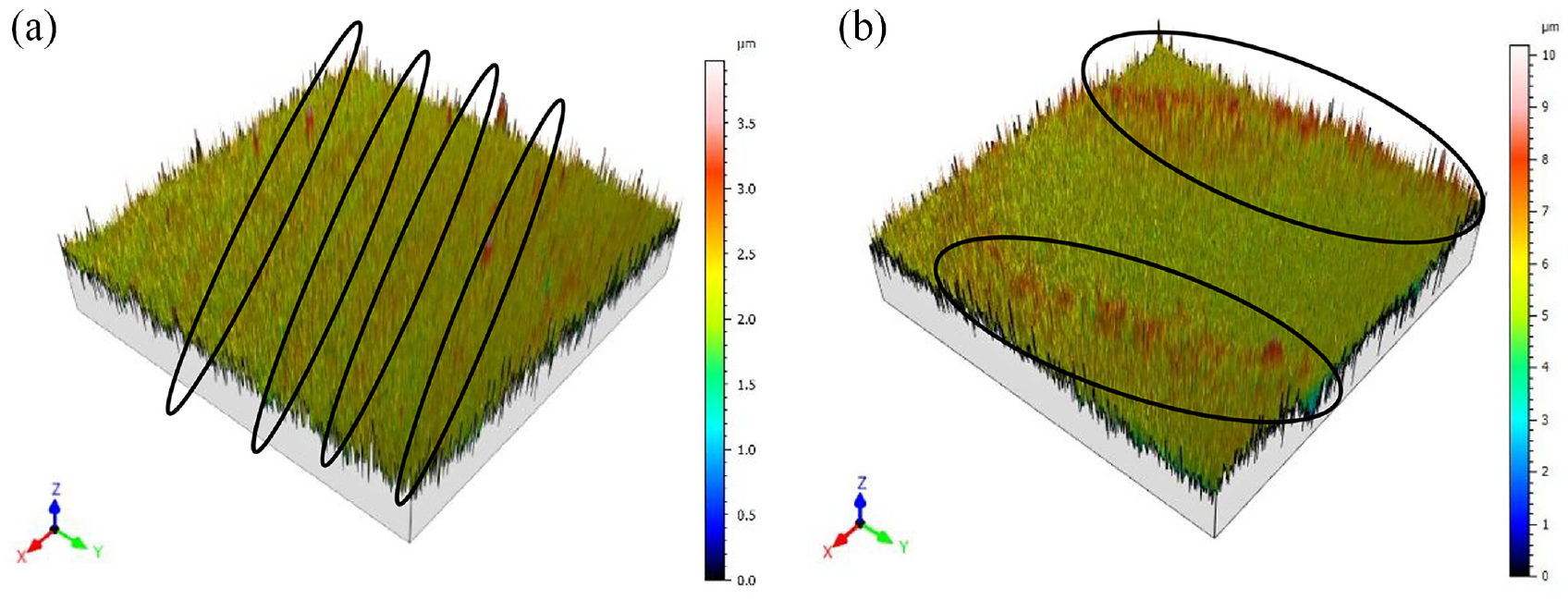
Surface topography of YSZ parts printed with: (a) 80% infill, (b) 95% infill.
Shao et al. 37 reported surface roughness close to 8 μm for zirconia-based gel materials. For implants, Sa values below or equal to 1 µm are considered to be smooth, while values above 1 µm are rough, from the point of view of osseointegration. 38 Thus, the samples in the present study can be considered smooth and, therefore, it is not necessary to undertake a subsequent polishing operation. Additionally, the surface roughness obtained in lateral walls of yttria-stabilized zirconia pastes depends on layer height and on print speed. 20 The lower layer height and speed, the smoother the surface is as demonstrated by our previous findings. 14 In the present work, however, the roughness was measured on the upper surface of the printed parts to assess its influence on cell compatibility.
In vitro biocompatibility assessment
As can be observed in Figure 4, hBM-MSCs were able to attach to the 3D-printed zirconia scaffolds and showed good viability after 3 days of culture (no differences were observed between 24 and 72 h of culture). It is noticeable that no differences were observed between uncoated and coated samples (type I collagen), showing that the bare material is biocompatible enough to allow direct cell culture on top without the need for functionalization, so unspecific adhesions formed by MSCs on bare scaffolds are enough for them to proliferate. Collagen has been proven to enhance corrosion resistance and biocompatibility mainly on metallic substrates such as titanium 39 or magnesium alloys 40 but it might not be so effective on ceramic substrates. Cells attached to the bare surface, suggesting that unspecific adhesions are being formed, although more tests should be done to confirm which cell proteins are mediating those adhesions to the surface. On the other hand, although a higher proliferation might be observed in the higher infill scaffolds, the difference is not significant enough to extract conclusions about the most appropriate infill parameter. Further tests on proliferation should be done to confirm these hypotheses.
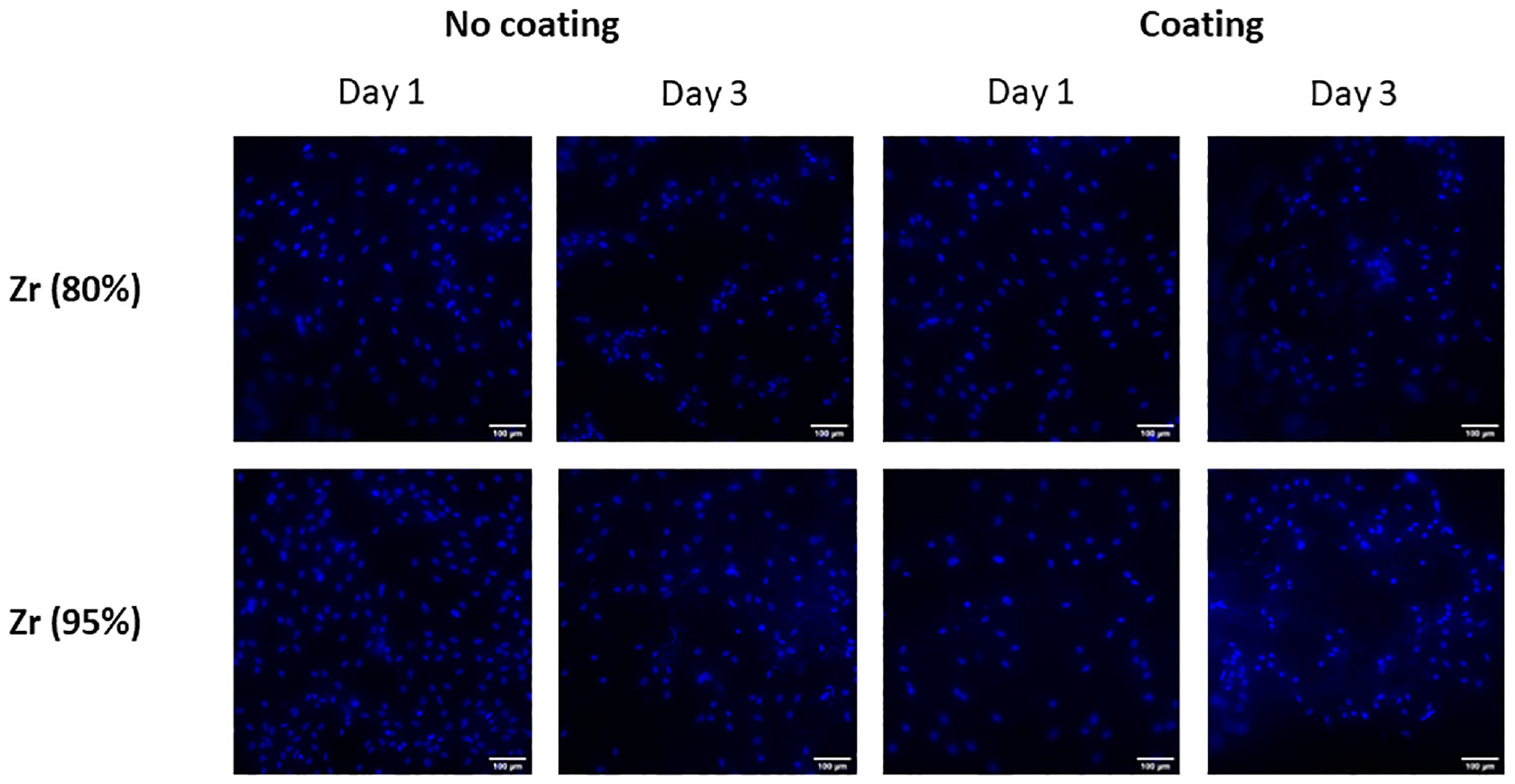
Fluorescent images (DAPI, cell nuclei) of hBM-MSCs cultured for 24 or 72 h on zirconia scaffolds (80% or 95% of density) with and without type I collagen coating.
The surface properties of the scaffolds directly affect cellular behavior. A too-smooth or too-rough scaffold could cause the cells not to adhere or to modify their behavior. For example, in soft hydrogels osteodifferentiation increased with surface roughness, being important from Rq values around 0.38 μm, while in stiff hydrogels higher osteodifferentiation was observed for intermediate roughness values. 41 In this work, the areal average roughness Sa measured in the above scaffolds, between 0.30 ± 0.026 μm and 0.69 ± 0.0 μm, led to a good cell proliferation behavior as shown in Figure 4. Thus, it seems that scaffolds’ roughness makes them suitable for MSCs culture, even when the material remains uncoated. This indicates that cells are generating unspecific adhesions with the printed parts that allow them to engraft.
Conclusions
In the present work, 3D-printed yttria-stabilized zirconia parts have been presented to be used as a novel material in implants. We found that the physical properties of the material, such as the surface topography, made the material a suitable candidate for prosthetics. Human BM-MSCs cultured on top of the printed scaffolds showed to be highly viable. Interestingly, we observed no differences between bare printed parts and those coated with an extracellular matrix protein to increase cell adhesion, thus indicating that the 3D-printed scaffolds could be used without further processing. This opens a door in the field of personalized medicine, as scaffolds could be directly printed with the needed shape before the intervention in the patient. Further research in vivo, by implanting the developed scaffolds in animals, would be needed to evaluate the scaffolds presented herein, but in vitro results suggest that biocompatibility would not be a limiting factor in those experiments.
Footnotes
Acknowledgements
Authors would like to thank Alejandro Domínguez for his help with the experimental tests.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was co-financed by the Spanish Research Agency, project number PID2020-115647RB-C21. The authors would like to thank the Catalan Government for the quality accreditation to their research group TECNOFAB (2021 SGR 01034) as well as for the quality accreditation and funding given to their research group DIOPMA (2021 SGR 00708). DIOPMA and TECNOFAB are certified agents TECNIO in the category of technology developers from the Government of Catalonia.