Abstract
The integrated electrochemical grinding machining has received wide acceptance in the aircraft turbine industry for the machining of blades, vanes, and honeycomb seal rings. Also, medical devices, instruments and forceps, shells, precision nozzles, instrument coupling, and air rotor motors that produced from stainless steel and new materials have all successfully been accomplished with electrochemical grinding. To improve the metal removal rate and to reduce the surface roughness (Ra) of the electrochemical grinding at high voltages, an integration between the alumina abrasive jet and the electrochemical grinding machining has been performed. The effect of the Al2O3 abrasive content on the metal removal rate and the Ra of the K110 alloy steel using Everite electrochemical grinding 618 at different voltages, different feed rates, different electrolyte NaCl concentrations, and different depths of the cut were successfully investigated. The results revealed that the abrasive electrochemical grinding was better than the electrochemical grinding results. The maximum effect of the Al2O3 on the metal removal rate was achieved at 5 wt.%. The current density in the machining gap was affected by the addition of the Al2O3, where it was decreased at percentages over 5-wt.% Al2O3. The abrasive electrochemical grinding resulted in lower surface roughness than the electrochemical grinding process.
Keywords
Introduction
The electrochemical grinding (ECG) machining process is a process in which electrochemical and mechanical actions are combined to facilitate the process of metal removal from the surface of the work-piece. In this process, nearly 90% of the metal is removed by the electrochemical reaction and 10% by mechanical action. The process is utilized in many sensitive applications such as machining of sensitive materials to heat and machining high hardness materials that are difficult to machine by the traditional machining processes. It has long been adapted for use in regrinding of cutting and turning tools.1–7
Enhancing the efficiency of the material removal rate and improving the surface quality of the machining processes can be achieved by applying different cutting actions on the material being machined. In particular, the electrochemical and mechanical actions that applied in the ECG can be combined with respective mechanical interaction used in another unconventional machining processes like ultrasonic machining (USM) or abrasive water jet machining, jet electrochemical micro-drilling. 8
In the ECG process, the kinetic energy of the grinding wheel is considered untapped, so we tried to benefit from it to strike a particles of ceramic material with the electrolyte in the direction of the work-piece surface for remove any precipitated released ions that in the form of oxide to increase the metal removal rate (MRR) and to improve the surface roughness. The USM process as an advanced machining process is characterized by a low MRR and a good quality surface. 9 On the other hand, the electrochemical machining (ECM) process is characterized by a high MRR and a dull surface roughness compared with the USM. It has been shown that the integration between the two processes has the advantage of their virtues where the efficiency of both processes is, therefore, improved in terms of higher machining speeds and lower tool wear, compared to normal USM. 10 Also, the new integration participates in solving the problem of machining new composites that consist of metal and hard ceramics together, such as Al-Al2O3, Al-SiC, and Cu/WC-Co.11–14
Another example of a new hybrid machining process is the abrasive ECM. This new hybrid machining process integrates the mechanical abrasion action of the abrasive particles and the electrolytic dissolution of ECM to produce a defect-free and highly finished work-pieces. Fundamentally, in this process, the abrasives polish the work-piece surface mechanically while it is being machined electrochemically. 15
The effect of the SiC abrasive particles and the electrolytic concentrations on the MRR and surface roughness of the abrasive-assisted ECM of aluminum–boron carbide composites has been investigated.16,17 The experiments have been proceeded by mixing 5-wt.% silicon carbide (SiC) of 50-μm particles size with electrolyte and varying the electrolyte concentration (10%–30%) in steps of 5% at constant parameters voltage (14 V), current (150 A), tool feed rate (1 mm/min), electrolyte pressure (1.5 MPa), and electrolyte flow rate (15 L/min). The results revealed that the externally assisted SiC abrasives along with the electrolyte could remove the thin layer of the oxides which formed during the machining process that leads to improving the surface roughness. In addition, the MRR increased by increasing the concentration of NaCl in the electrolyte up to 20 wt.%.
Das S et al. 9 have investigated the effect of different boron carbide abrasives grains diameters (16, 24, 34, 44, and 63 µm) and abrasive slurry concentration of (50 and 60 g/L) on the MRR of the USM. The results show that the MRR increases as the abrasive grain diameters and its concentration increase.
A new hybrid machining process that combines the electrochemical jet machining and abrasive jet machining was established. The effect of abrasive electrolyte concentrations, machining gap, voltage, and jet pressure parameters on the machining efficiency of the electrochemical abrasive jet machining was investigated. The process has proceeded for the SUS304 stainless steel sheet. The processes combination participated in improving the surface quality and greatly improved processing efficiency.
The Al2O3, SiC, B4C, and the diamond are the most commonly used ceramic abrasive in the USM process.17,18 Because the Al2O3 is cheap and has many advantages such as high hardness, excellent corrosion resistance with high chemical stability, and low density, it is the preferred choice for this study. 9
This investigation has been performed for two reasons. The first is summarized in the shortage in studying the effect of the abrasive particles concentration on the machining efficiency of the new explained hybrid machining processes, and the second is summarized in the performance of the ECG process itself, where at the ECG process, at high voltages (9–12 V) a passive layer of oxides produces on the work-piece surface during the machining process, and this affects the MRR efficiency, by preventing the reaction of the electrochemical to continue. Also, it makes the surface of the work-piece being dull.
The main aim of this study is at enhancing the MRR and improving the surface quality of the ECG machining process at high voltages by combining the process with an additional mechanical cutting action. Also, optimizing the parameters of the abrasive electrochemical grinding (AECG) machining process by studying the effect of the Al2O3 at different voltages, feed rates, depths of cut, and different electrolyte concentrations.
Experimental work
Materials and methods
In this investigation, alumina powder of 0.5- to 3-µm particle size and density of 3.4 g/cm3, which purchased from “Zircar Co. LTD” the United States, is used as an abrasive for the electrolyte slurry. Figure 1 shows the morphology of the Al2O3 powder by the scanning electron microscope (SEM). As shown, the abrasives are irregular and have sharp edges.

Scanning electron microscope (SEM) of the alumina (Al2O3) powder.
The sodium chloride (NaCl) is used as the source of cations and of the electrolyte. The K110 alloy steel is used as the work-piece material, and its chemical composition is shown in Table 1.
Chemical composition of K110 alloy steel.

All tests are run on an Everite ECG 618 electrochemical surface grinder. The main parts of the ECG machine are the electrolyte tank, the electrolytic pump, the cathode wheel, the direct current power supply, and the mechanical operation system. The Al2O3 abrasive is added to the electrolyte to perform the AECG process. Figure 2 shows the experimental test rig of the AECG.
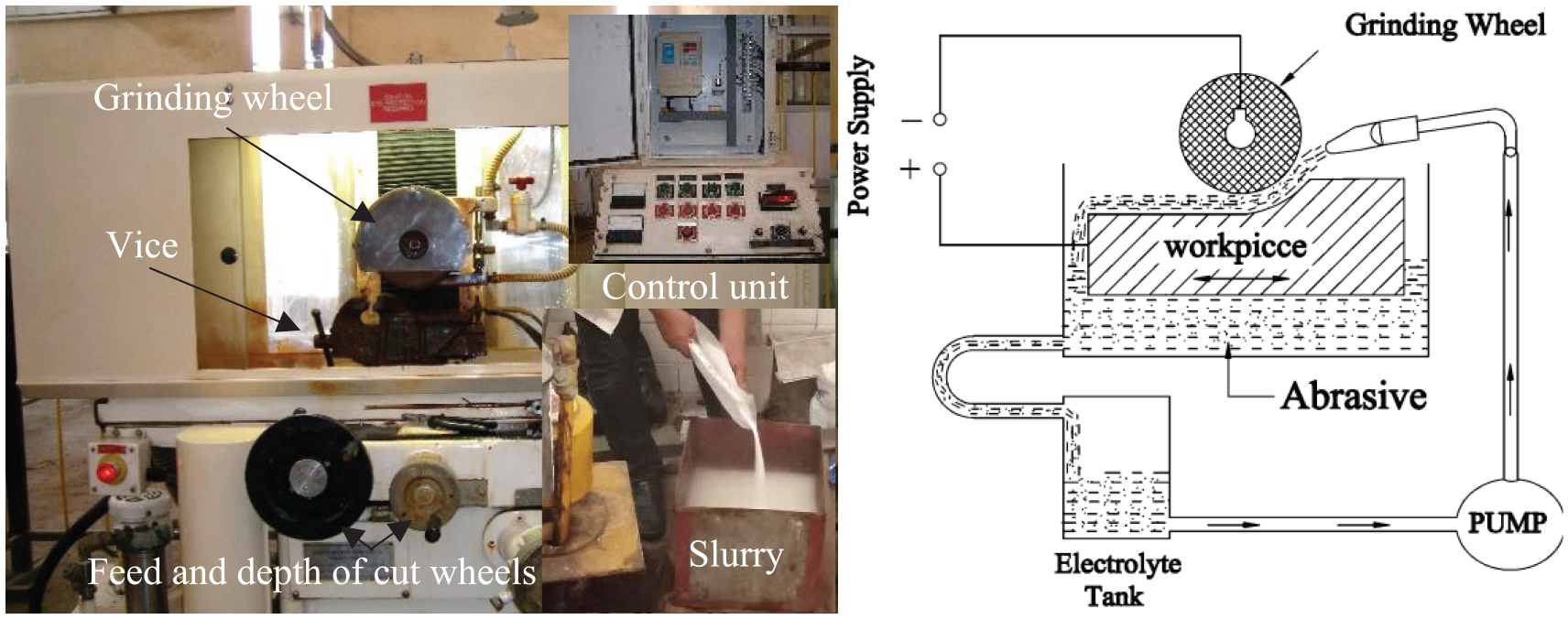
Hybrid abrasive electrochemical grinding (AECG) test rig.
The main parameters of the study are shown in Table 2. They are chosen according to values successfully applied in previous work1–6,14,15 and some preliminary experiments.
Electrochemical grinding (ECG) and abrasive electrochemical grinding (AECG) process parameters and their levels.

Based on the weight loss, the MRR is calculated using the following equations (1) and (2)
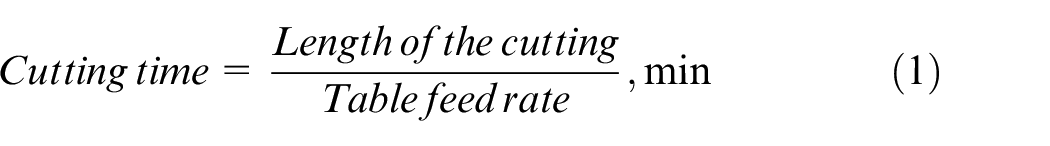
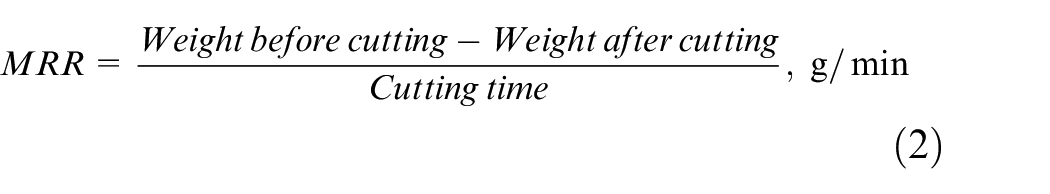
The Ra surface roughness of the machined material is evaluated using Surf Test 201, MITUTOYO, 178-950. Each surface roughness value is the average of five measurements at various positions of the work-piece. All the measurements are performed perpendicular to the cutting motion. The arithmetic mean deviation of the surface roughness is evaluated using equation (3)

where Ra is the arithmetic mean deviation of the surface roughness, N is the number of deviation, and rn is the sum of the deviations values.
Results and discussion
MRR of the ECG
Figure 3 shows the relationship between the feed rate and the MRR of the ECG machining process at 0- and 0.1-mm depth of cut (DOC) and 10-, 15-, and 20-wt.% NaCl electrolyte concentration. All the experiments have performed at the voltage v = 9 V. It is clear that the MRR increases by increasing both the feed rate and the electrolyte concentration.
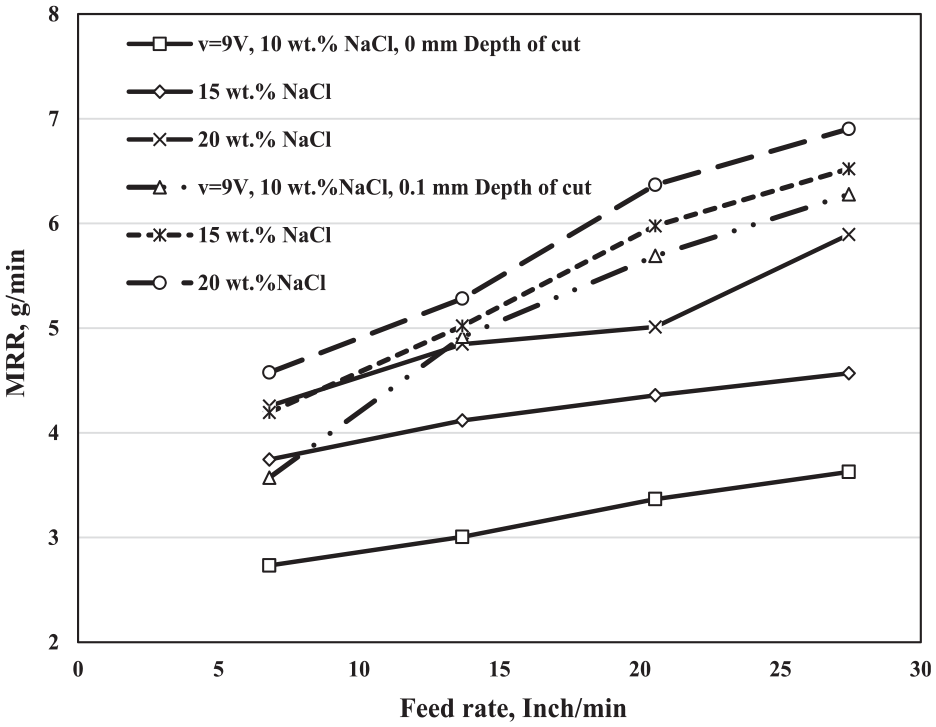
Variation of the MRR (g/min) with the feed rate at different depths of cut, different electrolyte concentrations, and at constant voltage v = 9 V.
As the feed rate increases, the time of the machining decreases, consequently the MRR increases. By increasing the feed, the effective time for cutting the required length decreases, because the relationship between them is inverse. The constant in the metal weight loss by decreasing the time makes the MRR increases. The figure reveals that the MRR increases by increasing the feed rate, and the effect of it at 0.1-mm DOC is higher than that of 0 mm. At 0.1-mm DOC, the gap between the work-piece and the cutting tool is less than of it at 0 mm. When the DOC was 0.1 mm, the mechanical action was greater than that of at 0-mm DOC, and the contact between the work-piece and the metal bonded grinding wheel increases; consequently, the effectiveness of the electrical current passing in the region of the cutting increases, that finally leads to an increase in the MRR. As a result of changing the feed rate from f = 6.81 in/min to f = 27.44 in/min at 10-wt.% NaCl and 0-mm DOC, the increment in the MRR was 0.894 g/min, whereas at 0.1 mm, it was 2.706 g/min at the same conditions. on the other hand, at 20-wt.% NaCl and 0-mm DOC, the increment in the MRR as the feed rate changed from f = 6.81 in/min to f = 27.44 in/min was 1.318 g/min, whereas at 0.1 mm and the same conditions, it was 2.327 g/min.
The electrolyte concentration, as the results show, has a great effect on the MRR of the ECG. Increasing the electrolyte concentration leads to an increase in the negative and positive charged, which is responsible for transferring the electrical current, consequently increases the MRR. Y Zhang et al. 19 showed that the conductivity of electrolytes increased by increasing its concentration leading to decrease machining time and consequently increases MRR.
The effect of the electrolyte concentration at 0-mm DOC on the MRR is better than the effect of it at 0.1 mm. At the 0-mm DOC, the gap between the work-piece and the grinding wheel is higher than that of 0.1 mm. The increasing gap allows the percentage of the positive and negative charge to increase, followed by increasing in the transferred current, and consequently increases the MRR. As the results show at the 0-mm DOC, the MRR increases from 2.733 to 4.256 mm at f = 6.81-in/min feed rate and from 3.627 to 5.894 g/min at 27.44-in/min feed rate, whereas at 0.1-mm DOC it improves from 3.572 to 4.576 g/min at f = 6.81-in/min feed rate, and from 6.278 to 6.903 g/min at 27.44-in/min feed rate.
Figure 4 illustrates the same relation between the feed rate and the MRR at different depths of cut and different electrolyte concentrations but at the voltage v = 11.5 V. It is clear from the results that the effect of the feed rate on the MRR at v = 11.5 V has the same trend of the results at v = 9 V where it increases by increasing the feed rate and the electrolyte concentration. The effect of the feed rate on the MRR at 0.1-mm DOC is higher than that of at 0 mm, whereas the effect of the electrolyte concentration at 0-mm DOC is better than the effect of it at 0.1-mm DOC, for reasons explained previously.
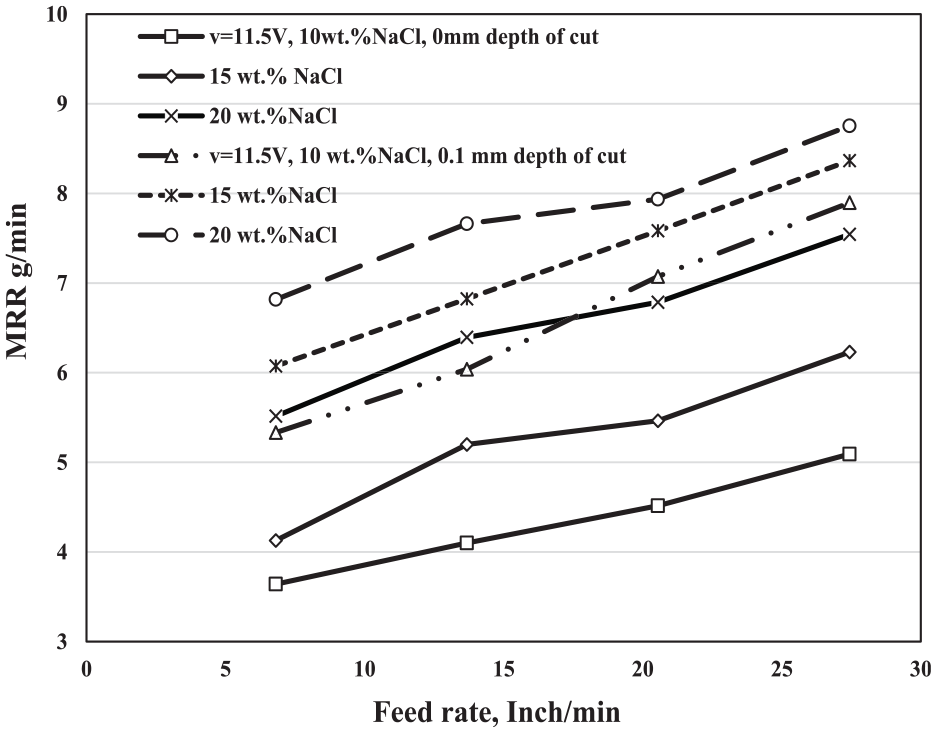
Variation of MRR (g/min) with the feed rate at different depths of cut, different electrolyte concentrations, and at constant voltage v = 11.5 V.
It is obvious from the results that the increment in the MRR at the different feed rates, 0.1-mm DOC, and at v = 11.5 V is less than that of at v = 9 V, that may be attributed to the increase in the passive layer that produced at v = 11.5 V. On the other hand, the effect of the electrolyte concentration at v = 11.5 V and 0-mm DOC is higher than that of at v = 9 V, that may be attributed to the increment in the electrical current that carries the negative and positive ions, consequently increasing the MRR.
As shown in Figure 5, at the voltage v = 9 V, the MRR increased from 3.572 to 4.576 g/min at f = 6.81 in/min and from 6.278 to 6.903 g/min at f = 27.44 in/min, whereas it increased from 5.332 to 6.816 g/min at f = 6.81 in/min and from 7.896 to 8.754 g/min at 27.44 in/min.
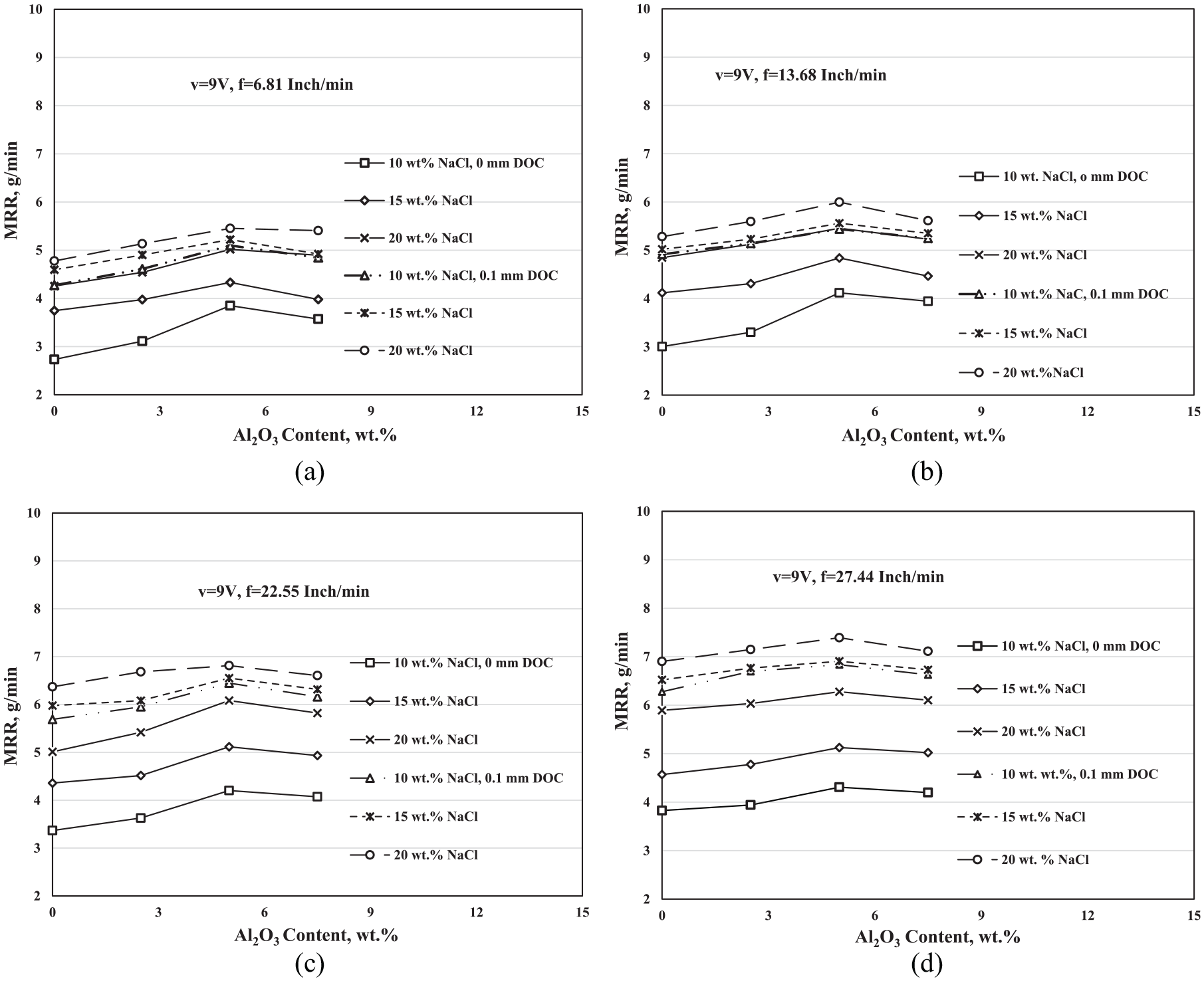
Variation of the MRR (g/min) of the ECG with the Al2O3 wt.% concentration at different electrolyte concentrations, different depths of cut, voltage v = 9 V, and feed rates (a) f = 6.81 in/min, (b) f = 13.68 in/min, (c) f = 22.55 in/min, and (d) f = 27.44 in/min.
The MRR of the AECG at v = 9 V
Figure 5(a–d) shows the effect of the Al2O3 abrasive addition on the MRR of the ECG process at different depths of cut and different electrolyte salt concentrations. Figure 5(a) is at f = 6.81 in/min, Figure 5(b) is at f = 13.68 in/min, Figure 5(c) is at f = 22.55 in/min, and Figure 5(d) is at 27.44 in/min. All the experiments were performed at the voltage v = 9 V.
As it is shown in all Figures 5(a)–(d), the MRR increased by increasing the Al2O3 weight percentage up to 5 wt.% then decreased. Also, they are shown the effect of the Al2O3 on the MRR of the ECG at 0- and 0.1-mm DOC. It is obvious from the results that the effect of the abrasive particles at 0-mm DOC is higher than the effect of it at 0.1-mm DOC, which may be attributed to the gap between the grinding wheel and the work-piece. At the 0-mm DOC, the gap in the cutting region is higher than that of at 0.1-mm DOC. At the higher gap, the chance of increasing the number of particles in the machining region increases, consequently increases the MRR. In addition, the high gap allows the grinding wheel to strike the alumina ceramic particles against the surface of the work-piece. As a result of hammering the abrasive particles by the grinding wheel, they acquire high velocity and subsequently high kinetic energy. Striking the surface of the work-piece with high force makes the particles erode the surface and aids in removing the precipitates passive layer, which is formed during the electrochemical reaction, leading to cracking and fracture of the work-piece surface, resulting into increasing the MRR.
Lowering the effect of the abrasive particles at 0.1-mm DOC may be related to the number of particles that can be embedded in the machining zone and share in the cutting during the machining process. At 0.1-mm DOC, the gap becomes minimum and may lead to restricting the motion of the particles in the machining zone, leading to a decrease in the MRR.
The figures illustrate the effect of the abrasive addition at different electrolyte concentration on the MRR of the ECG. It is manifest that the effect of the abrasive particles is decreased by increasing the electrolyte concentration and the effect of it at 0-mm DOC is more demonstrator than that at 0.1-mm DOC. As the electrolyte concentration increases, the positive and negative charge ions increase, and the electrochemical reaction becomes more active, and consequently the chance of the precipitated passive layer increases. The passive layer consumes a part of the kinetic energy of the abrasive particles during the cutting process and leads to a decrease in the performance of it, leading to a decrease in the effect of it on the MRR.
Moreover, the results show that the effect of the abrasive particles decreases as the feed rate increases. Increasing the feed rate leads to reducing the time of the machining process so that the effective time of the particles decreases, which finally leads to a decrease in the effect of abrasive particles by increasing the feed rate.
At the lowest feed rate, Figure 5(a) shows that the MRR at 10-wt.% NaCl and 0-mm DOC is increased from 2.733 g/min (when no abrasive particles are applied) to 3.849 g/min at 5-wt.% Al2O3 (the difference in the MRR is 1.116 g/min). At the same conditions and at 0.1 mm, the MRR is increased from 4.272 g/min (when no abrasive particles were added) to 5.098 g/min at 5-wt.% Al2O3 (the difference in the MRR is 0.826 g/min). The results at 15- and 20-wt.% NaCl take the same trend. On the other hand, at the highest feed rate, figure (d) shows that the MRR is increased at 10-wt.% NaCl and 0-mm DOC from 3.627 g/min (when no abrasive particles were added) to 4.708 g/min at 5-wt.% Al2O3 (the difference in the MRR is 1.081 g/min). At the same conditions and at 0.1 mm, the MRR is increased from 6.278 g/min (when no abrasive particles were added) to 6.836 g/min at 5-wt.% Al2O3 (the difference in the MRR is 0.558 g/min). Also, the results at 15- and 20-wt.% NaCl take the same trend.
From the explained results, it can be concluded that the best conditions for achieving the highest increment of MRR that at 0-mm DOC are 10-wt.% NaCl, 5-wt.% Al2O3, and at the lowest feed rate f = 6.81 in/min. At these conditions, the MRR is increased from 2.733 g/min (when no abrasive particles are added) to 3.849 g/min (when 5-wt.% Al2O3 added) with improvement percentage 40.8%.
Decreasing effects of the Al2O3 on the MRR of the ECG process at 7.5 wt.% may be related to the nature of the particles itself. Alumina is a non-conductive material, and the presence of it in the electrolyte reduces its electrical conductivity, and consequently the electrochemical reaction and MRR.
The MRR of the AECG at v = 11.5 V
The effect of the Al2O3 wt.% additions at voltage v = 11.5 V on the MRR of the ECG process shown in Figures 6(a–d). All the experiments were proceeded at different electrolyte concentrations, different depths of cut, and at f = 6.81, 13.68, 22.55, and 27.44 in/min, which are the same conditions at v = 9 V.
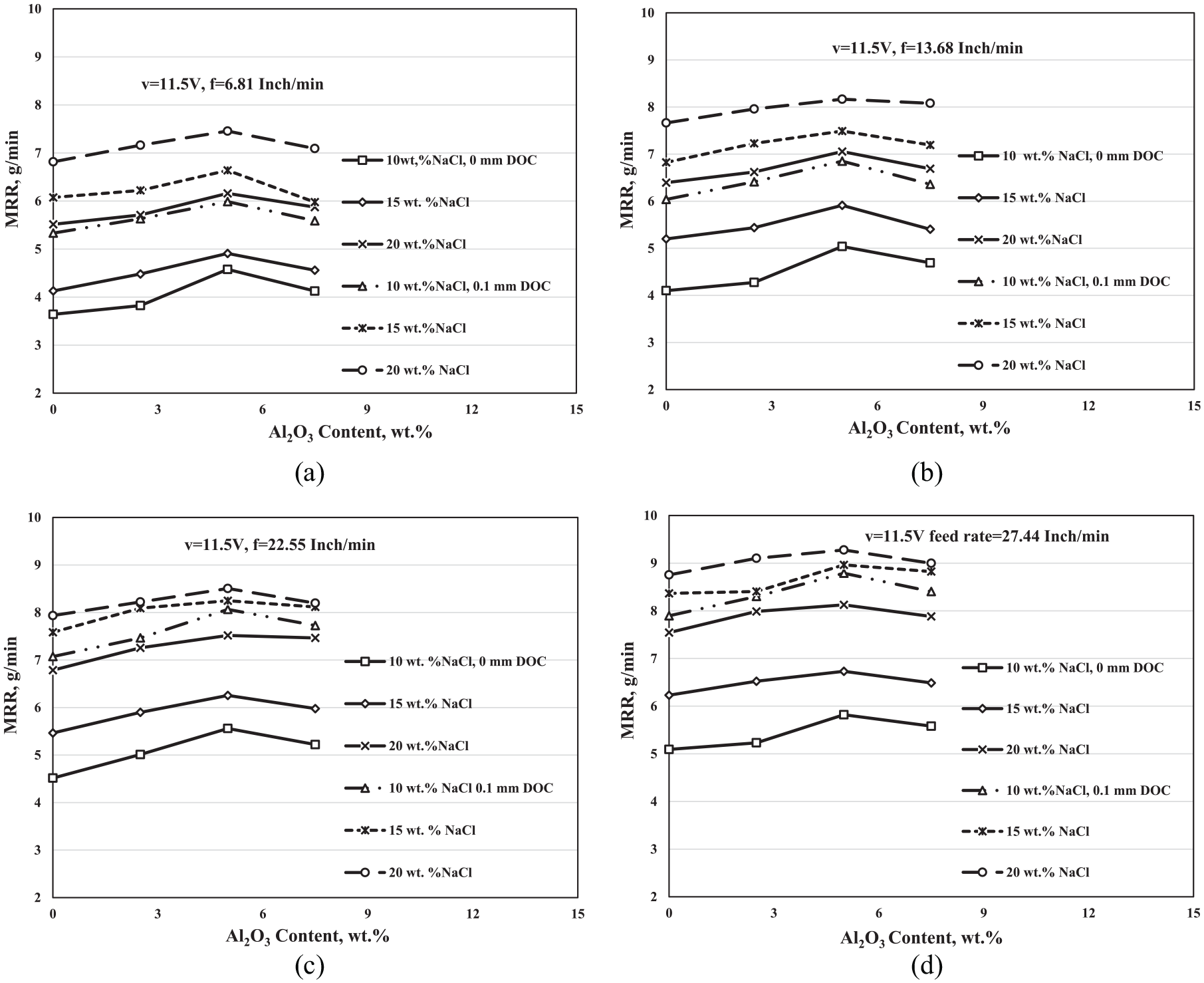
Variation of the MRR (g/min) of the ECG with the Al2O3 wt.% concentration at different electrolyte concentrations, different depths of cut, voltage v = 11.5 V, and at feed rates (a) f = 6.81 in/min, (b) f = 13.68 in/min, (c) f = 22.55 in/min, and (d) f = 27.44 in/min.
The results at v = 11.5 V showed the same effect of the Al2O3 wt.% additions on the MRR of the ECG at v = 9 V, where the MRR increased as the Al2O3 wt.% increased up to 5 Al2O3 wt.% then decreased. Also, the results revealed that the effect of the abrasive assist particles on the MRR of the ECG at 0-mm DOC is stronger than that at 0.1-mm DOC, which is the same effect at voltage v = 9 V. In addition, they show that the effect of the Al2O3 on the MRR is decreased by increasing the electrolyte concentration.
The difference only is that the effect of the Al2O3 wt.% at 0.1-mm DOC increases compared with v = 9 V. At the v = 11.5 V, the electrochemical reaction increases and the probability of propagating an overcut increases, followed by increasing in the gap between the cutting tool and the work-piece.17,20 The high gap allows high numbers of particles to a presence in the machining zone and consequently shares in the erosion and cutting process leading to an increase in the MRR of the ECG process.
At the v = 11.5 V, the strongest effect of the Al2O3 is at f = 22.55 in/min, 10-wt.% NaCl, and at 0-mm DOC, where the MRR increased from 4.515 to 5.559 g/min at 5-wt.% Al2O3 with an improvement percentage of 23.1%. It can be concluded from comparing the results of the MRR of the AECG at v = 9 V and v = 11.5 V that the effect of the Al2O3 at v = 9 V and 0-mm DOC is stronger than its effect at v = 11.5 V. At v = 9 V, the increment in the MRR at 10-wt.% NaCl and f = 6.81 in/min was 1.116 g/min, whereas at v = 11.5 V, the increment in the MRR at 10-wt.% NaCl and f = 22.55 in/min was 1.044 g/min.
Decreasing the effect of Al2O3 particle powder at v = 11.5 V may be due to increasing the passive layer on the surface of the work-piece as a result of increasing the voltage from 9 to 11.5 V. Passive layer consumes some value of kinetic energy of the particles that produce from striking it with the grinding wheel, which leads to decreasing the efficiency of it.
Effect of the Al2O3 wt.% additions on the current density
The experimental results of the current density of the AECG of K110 alloy steel have presented in Table 3. It is obvious from the table that the current density increases by increasing the electrolyte concentration and the feed rate. On the other hand, the current density influenced by the abrasive concentration, where it decreased at 7.5-wt.% Al2O3 addition. Al2O3 ceramic material is a non-conductive material and the presence of it in the electrolyte of the ECG process at a percentage of over 5 wt.% leads to hinder the motion of the ions, which leads to a decrease in the current density that passes during the electrochemical reaction, leading to a decrease in the MRR of the AECG.
Experimental results of the current density of the ECG and AECG.
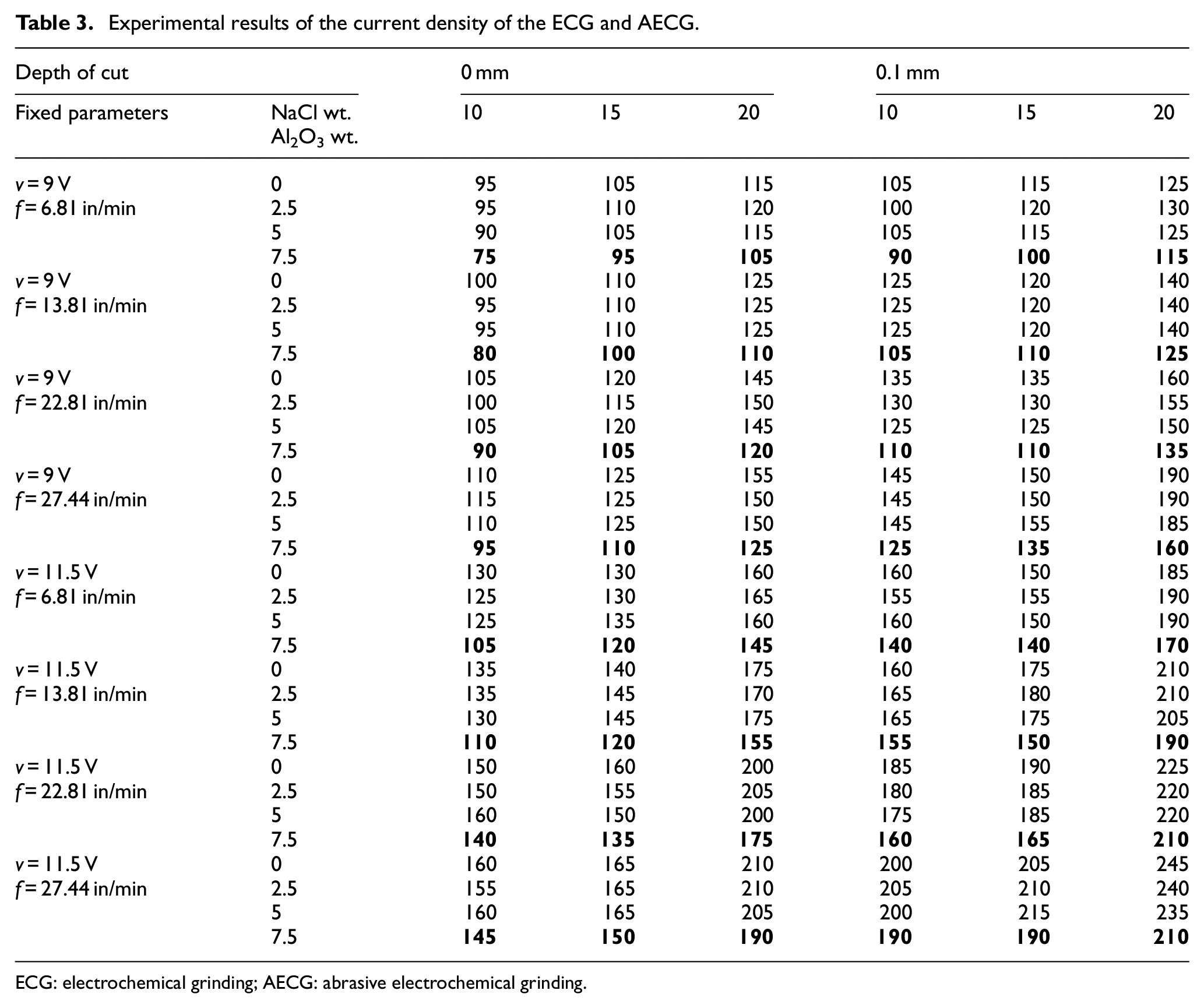
ECG: electrochemical grinding; AECG: abrasive electrochemical grinding.
Surface roughness measurements with Ra
The surface roughness (Ra) of the ECG at v = 9 V and v = 11.5 V
This portion is interested in studying the effect of the ECG process parameters on the surface roughness of the machined component surfaces. Figures 7(a) and (b) that are at v = 9 V and v = 11.5 V, consequently, illustrate the effect of the feed rate on the MRR at different electrolyte concentrations and different depths of cut. As the figures show, the surface quality of the machined surfaces improved gradually by increasing the applied feed rate. The results revealed that increasing the electrolyte concentration contributes to a decreasing in surface quality that may be attributed to the passive layer that precipitated during the machining process. As the electrolyte concentration increases, the current density that passes through the gap between the tool and the work-piece increases and the electrochemical reaction becomes more active, consequently the probability of increasing the passive layer is also increased,2–5 which may be the main reason in the decrease in the surface quality. M Sankar et al. 17 have investigated the variation of the surface roughness with the electrolyte concentration of the abrasive-assisted ECM and showed that the surface quality of the machined part decreases with increasing the electrolyte concentration. The effect of the feed rate on the surface roughness of the machined surfaces at 0.1-mm DOC is better than that at 0-mm DOC. It is observed from the results that the best surface quality (Ra = 1.06 μm) was achieved at v = 9 V, f = 27.44 in/min, 10 NaCl wt.%, and 0.1-mm DOC.
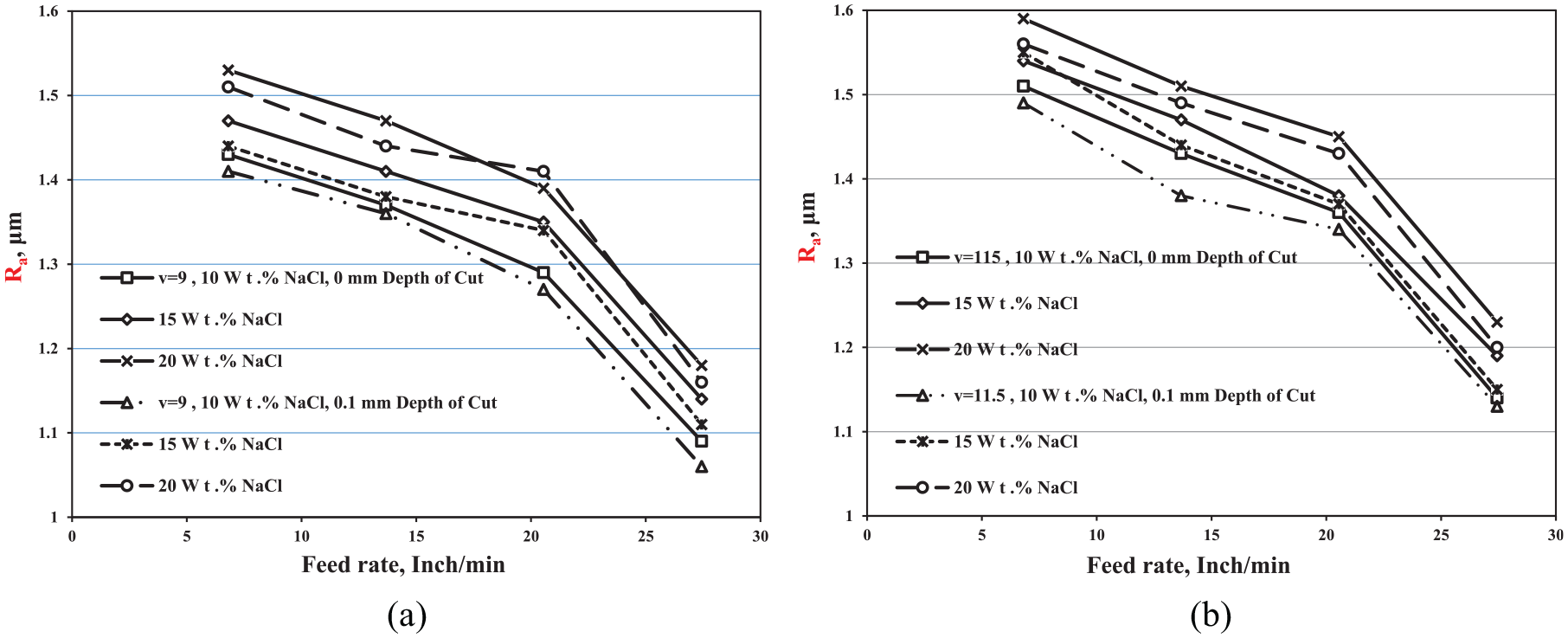
Variation of Ra with feed rate at different electrolyte concentrations and different depths of cut at (a) v = 9 V and (b) v = 11.5 V.
The surface roughness (Ra) of the AECG at v = 9 V
In this part, the only results of the Ra for the AECG at v = 9 V will illustrate, because the best result of the Ra for the ECG process was achieved at v = 9 V. The effect of the Al2O3 addition on the Ra of the ECG at different electrolyte concentrations and different depths of cut is shown in Figure 8 (a–d). Figure 8(a) shows the effect of Al2O3 wt.% at f = 6.81 in/min, (b) at f = 13.68 in/min, (c) at f = 20.55 in/min, and (d) at f = 27.44 in/min.
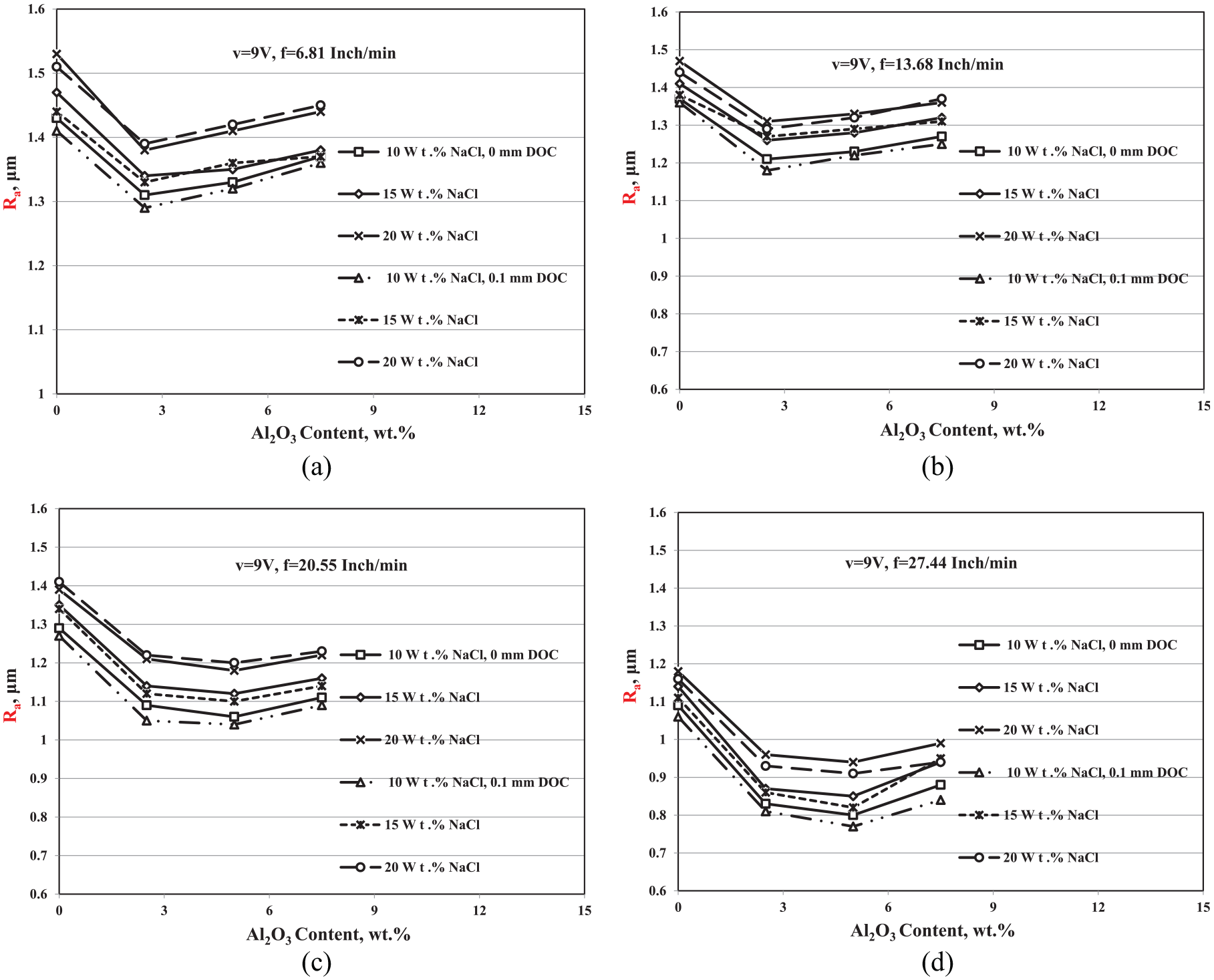
Variation of Ra with Al2O3 wt.% at different electrolyte concentrations and different depths of cut at (a) f = 6.81 in/min, (b) f = 13.68 in/min, (c) f = 20.55 in/min, and (d) f = 27.44 in/min.
It can be observed from the figures that the effect of the abrasive particles on the surface quality increases with increasing the feed rate. As Figure 8(a) and (b) shows, there is an enhancement in the Ra of the machined component surfaces as a result of increasing the Al2O3 up to 2.5 wt.%. Decreasing the surface quality at low feed rates and high abrasive percentages may be attributed to increasing the time of particles striking that may lead to an increase in the grooves on the surface of the machined part, consequently decreases the surface quality.
On the other hand, the Ra is improved up to 5-wt.% Al2O3, at feed rates 20.55 and 27.44 in/min, respectively, as it is shown in Figures 8(c) and (d). Because the cycle of the slurry is continuous during the machining process, the distribution of the abrasive in the electrolyte is uniform, and this makes the material removal in all area of the machined surface is same, resulting in a flat surface. As a result of impacting the machined surfaces, by the accelerated micro alumina abrasives, the peaks that produced on the surface are reduced; consequently, the quality of the machined surfaces is improved. Figure 9 shows the mechanism of the Al2O3 impact during the cutting process and its effect on the peaks produced from the ECG process.
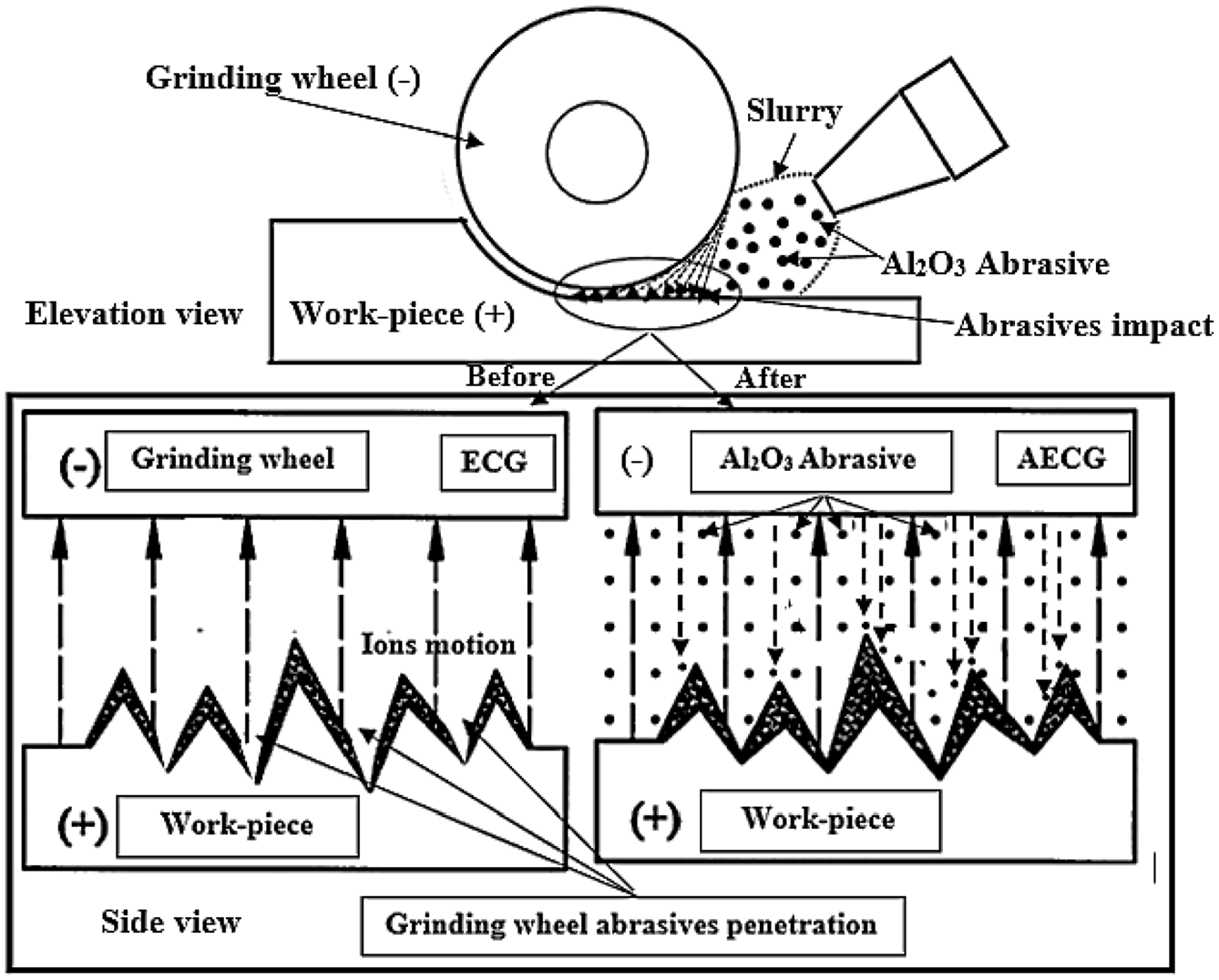
Mechanism of the ECG and AECG during the cutting process in two views that are elevation and side view.
It is also obvious that the surface quality deteriorates by increasing the electrolyte concentration in the presence of the abrasive particles. The 5-wt.% Al2O3, v = 9 V, f = 27.44 in/min, 10-wt.% NaCl, and the 0.1-mm DOC were the best conditions for establishing the lowest surface roughness, where the Ra changed from 0.106 to 0.77 μm.
Characterization of the machined component surfaces
Images for the machined component surfaces that established by the ECG and the abrasive-assisted ECG process are shown in Figure 10. As the images show, the machined component surface produced by the AECG is more bright and smoother than that machined by the ECG.

Images for machined component surfaces for ECG and AECG at 5-wt.% Al2O3, v = 9 V, f = 27.44 in/min, 10-wt.% NaCl, and 0.1-mm DOC.
Conclusion
In the present experimental investigation of the K110 alloy steel, the effects of the ECG parameters and the Al2O3 wt.% abrasive contents on the MRR and the Ra were studied. According to the results and its discussion, the conclusion of the research can be summarized in the following points:
The MRR of the ECG process was increased by increasing both the feed rate and the electrolyte concentration.
The effect of the feed rate on the MRR at 0.1-mm DOC was higher than that of at 0 mm.
The effect of the electrolyte concentration on the MRR was higher at 0 mm than that of at 0.1-mm DOC.
The increment in the MRR as a result of changing the feed rate at 0.1-mm DOC was decreased by increasing the voltage from v = 9 V to v = 11.5 V.
The increment in the MRR as a result of changing the electrolyte concentration at 0-mm DOC was increased by increasing the voltage from v = 9 V to v = 11.5 V.
The MRR of the ECG was increased by increasing the Al2O3 up to 5 wt.% then decreased.
The effect of the Al2O3 at 0-mm DOC was stronger than that at 0.1-mm DOC.
The effect of the Al2O3 on the MRR of the ECG was decreased by increasing the feed rate.
The best result of applying the Al2O3 was established at 0-mm DOC, 5 Al2O3 wt.%, v = 9 V, 10 NaCl wt.%, and at f = 6.81 in/min, with an improvement of 40.8%.
The surface quality of the machined surfaces by the ECG process was improved by increasing the feed rate, increasing the DOC, and decreasing the electrolyte concentration.
The best result of the Ra was achieved at v = 9 V, 0.1-mm DOC, 10 NaCl wt.%, and f = 27.44 in/min.
The greatest effect of the Al2O3 on the Ra was accomplished at 10-wt.% NaCl.
The effect of the Al2O3 on the Ra was more demonstrate at the high value of the feed rate that is 27.44 in/min.
The 5-wt.% Al2O3, v = 9 V, f = 27.44 in/min, 10-wt.% NaCl, and the 0.1-mm DOC were the best conditions for establishing the lowest surface roughness, where the Ra decreased from 1.06 to 0.77 μm with 37.66% reduction.
The presence of the Al2O3 abrasive with the electrolyte not only will remove the passive oxide layers that on the surface of the work-piece at high voltages, but also will prevent the positive ions to deposit on the surface of the grinding wheel and will dress it continuously.
Footnotes
Appendix
Acknowledgements
The authors wish to thank the technical staff of production technology at the Faculty of Industrial Education-Helwan University, Cairo, Egypt for their cooperation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
