Abstract
Additive manufacturing technologies are becoming more and more important for the implementation of efficient process chains. Due to the possibility of a near net shape, manufacturing time for finish-machining can significantly be reduced. Especially for conventionally hard to machine materials like gamma titanium aluminides (γ-TiAl), this manufacturing process is very attractive. Nevertheless, for most applications, a rework of these generative components is necessary. Independently of the mechanical material properties, electrochemical machining is one promising technology of machining these materials. Major advantages of electrochemical machining are its process-specific characteristics of high material removal rates in combination with almost no tool wear. But electrochemical machining results are highly dependent on the microstructure of the material regarding the surface roughness. Therefore, this article deals with research on electrochemical machining of electron beam melted γ-TiAl TNB-V5 compared to a casted form of this alloy. The difference between the specific removal rates as a function of current density is investigated using electrolytes based on sodium nitrate and sodium chloride. Moreover, the dissolving behavior of the electron beam melted and casted structure is analyzed by potentiostatic polarization curves. The surface roughness is heavily dependent on a homogeneous dissolution behavior of the microstructure. Thus, the mean roughness as a function of current density is investigated as well as rim zone analyses of the different structures.
Introduction
Assembling components layer-by-layer, additive manufacturing (AM) technologies are able to produce very complex shapes with internal channels or cavities. Integral constructions can be implemented more easily and finish-machining can significantly be reduced by a possible near net-shape manufacturing. Furthermore, unused powder can be reused efficiently. In particular, electron beam melting (EBM) is capable of working with many material classes. Using vacuum, the process is highly suitable for manufacturing materials with high affinity to oxygen such as titanium alloys. Nevertheless, due to their layer effect and powder size, AM technologies have relatively poor finish qualities, and rework strategies are usually required.1,2
Gamma titanium aluminides (γ-TiAl) are becoming more and more important for the turbomachinery industry. Due to their low density in combination with a high strength-to-weight ratio, oxidation, and corrosion resistance at high temperatures, these materials are suitable for replacing components made out of nickel-based alloys for some applications.
γ-TiAl are having disadvantages regarding the conventional cutting processes due to their low thermal conductivity and brittle deformation.3,4 Independently of mechanical properties, electrochemical machining (ECM) is a promising manufacturing method for machining these alloys.
First studies of ECM of γ-TiAl done by Clifton et al. focused on reproducible stable machining conditions of Ti-45Al-2Mn-2Nb+0.8 vol. % TiB2 using chloride and perchlorate electrolyte systems. 5 Further researches on the surface integrity and fatigue life of this material showed a relatively rough surface of the ECM specimen due to selective etching of individual lamellae. This behavior was identified for points of stress concentration and affected the fatigue life. 6 In Klocke et al.,7,8 further γ-TiAl alloys were investigated using sodium nitrate and sodium chloride–based electrolytes. Surface roughnesses machined with sodium nitrate electrolyte were improved, and the effects of the selective etching were reduced. Nevertheless, these investigations showed that the machining result of γ-TiAl is mainly affected by the microstructure due to the atomic dissolution.9,10 The microstructure of EBM-manufactured γ-TiAl TNB-V5 is very different from conventionally casted material. No investigations concerning different microstructures of γ-TiAl were published before. Hence, this article presents investigations on ECM and machining results for different machining routes of γ-TiAl.
Materials and experimental methods
The investigated workpieces, out of γ-titanium aluminide TNB-V5, were casted as well as electron beam melted. Nominal chemical composition of the different produced materials is equal and summarized in Table 1.
Chemical composition of TNB-V5.

The main active working principle of EBM is roughly comparable to that of a scanning electron microscope. The metal powder is melted by an electron beam emitted by a tungsten filament. The grain size of the used metal powder was approximately about 100 µm. The casted workpieces were produced in a vertical centrifugal casting. Compared to the conventional casting process, the microstructure consists of much less pores and blowholes. A lamellar microstructure is developed in the casted material with the typical colony structure. The lamellar structure has colonies in the magnitude of 100–300 µm.
The investigations on the electrochemical machinability of these different produced workpieces were carried out on two different machine tools due to two different electrolyte systems, sodium nitrate and sodium chloride. Nevertheless, the machining device, workpiece geometry, and machining parameters were identical. In Table 2, the machining parameters for the research studies are listed.
Machining parameters for EBM and ECM.
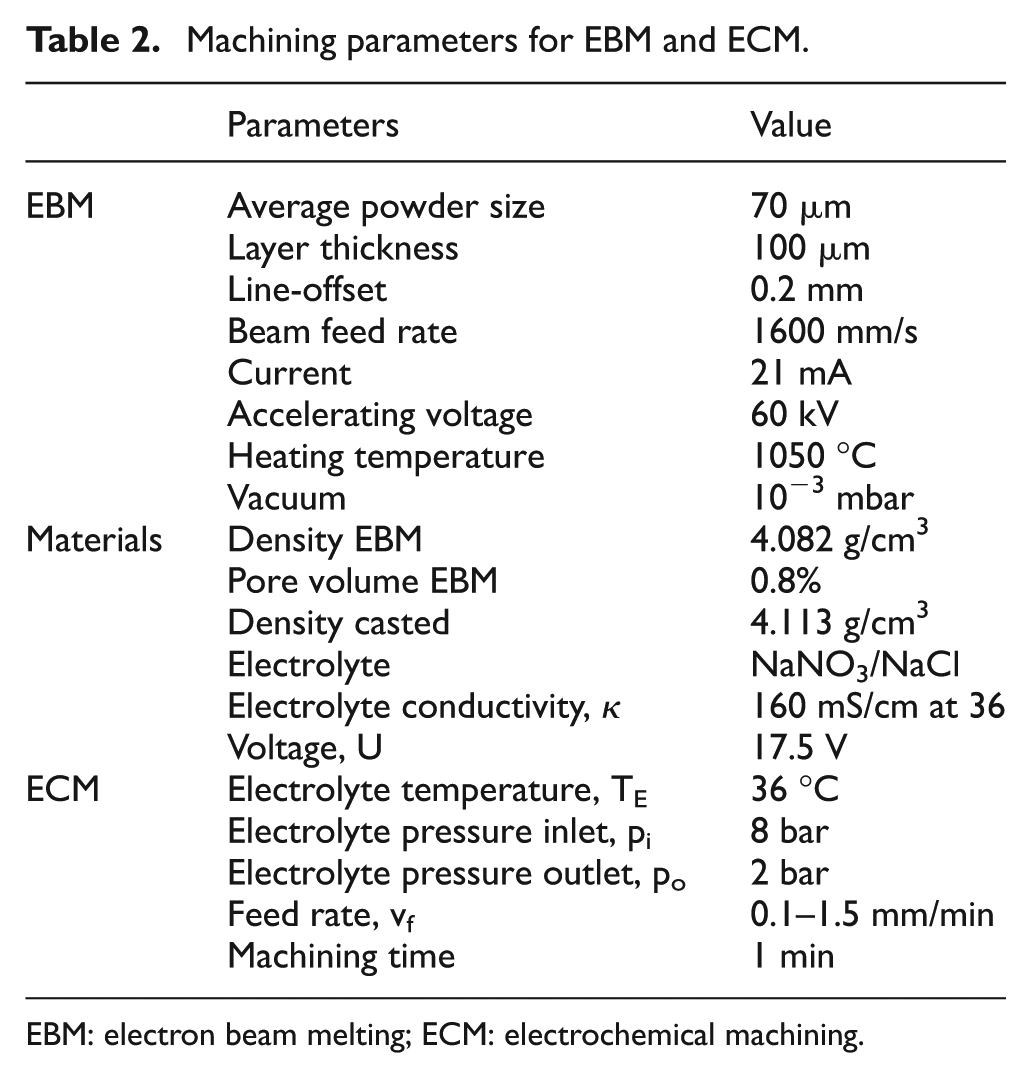
EBM: electron beam melting; ECM: electrochemical machining.
The materials were investigated on a standardized test setup for electrochemical classification. This method was first proposed by Degenhardt in 1972 and is performed at the WZL since that time.11,12 The standardized test setup ensures a good comparability of all investigated materials for the ECM sinking process because of similar conditions of electrolysis like electrolyte flow velocity, machining cross-sectional area, or changes in electrolyte conductivity (cp. Figure 1). The specimen surfaces were all ground before the ECM sinking process to avoid any influences by differences in the initial surface state. The specimen exhibits a surface roughness of Ra 0.1 µm after the grinding process.
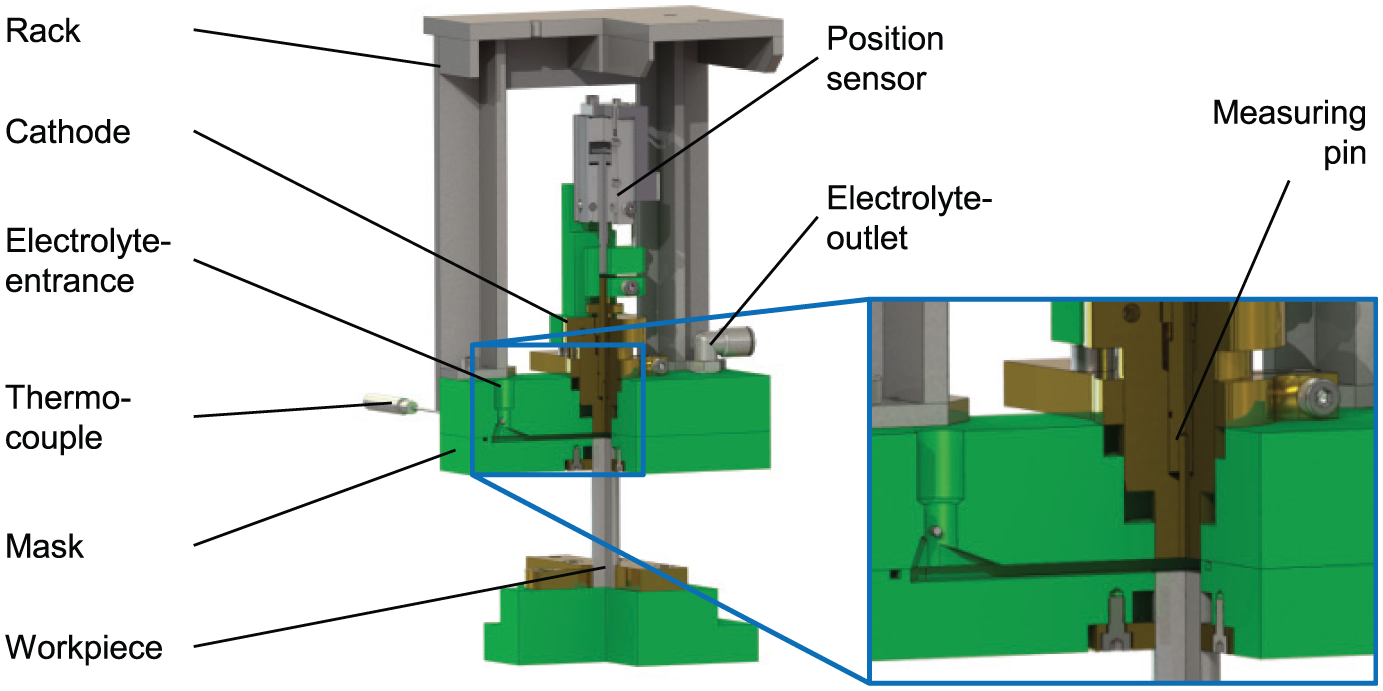
Standardized ECM device for electrochemical characterization.
For the investigations, the feed rate vf was set and the adjusting process current was measured. The frontal gap width s90 and mean surface roughness Ra of the different specimen were measured after the process. In conjunction with machining cross-sectional area and monitored current, the current density J can be calculated for the different feed rates. Due to the formation of an electric isolating oxide layer using sodium nitrate, standard titanium alloys, like Ti-6Al-4V, are machined with an aqueous sodium chloride solution. Concerning the higher amount of aluminum, it is feasible to ECM γ-TiAl alloys with a sodium nitrate electrolyte. Therefore, the experiments are performed with both types of electrolytes. Each experiment was performed three times.
Results and discussion
s90-J and vf -J curves
In Figure 2, the different s90-J and vf -J curves of TNB-V5 are shown. s90-J curves provide a measure of the manufacturing accuracy, whereas vf-J curves are used to determine the effective material removal rate Veff.. The s90-J curves show the typical hyperbolic behavior for ECM processes. Low current densities result in larger frontal gap widths; at a higher current density of more than 0.3 A/mm2, the frontal working gap is less than 300 µm. High accordance of both materials and electrolytes is noticeable in the s90-J curves. In contrast, the vf-J curves differ more from each other. The biggest difference can be noticed between the two electrolyte types. Due to the prevention of a passivation layer using sodium chloride electrolyte, the current efficiency is higher than using a sodium nitrate electrolyte. This behavior is well known and was observed in previous investigations.10,13,14 According to the vf-J curves (Figure 2), a higher current efficiency results in a larger slope of the line which is also declared as the effective material removal rate Veff.. The distance between the two electrolyte types increases with higher current densities.
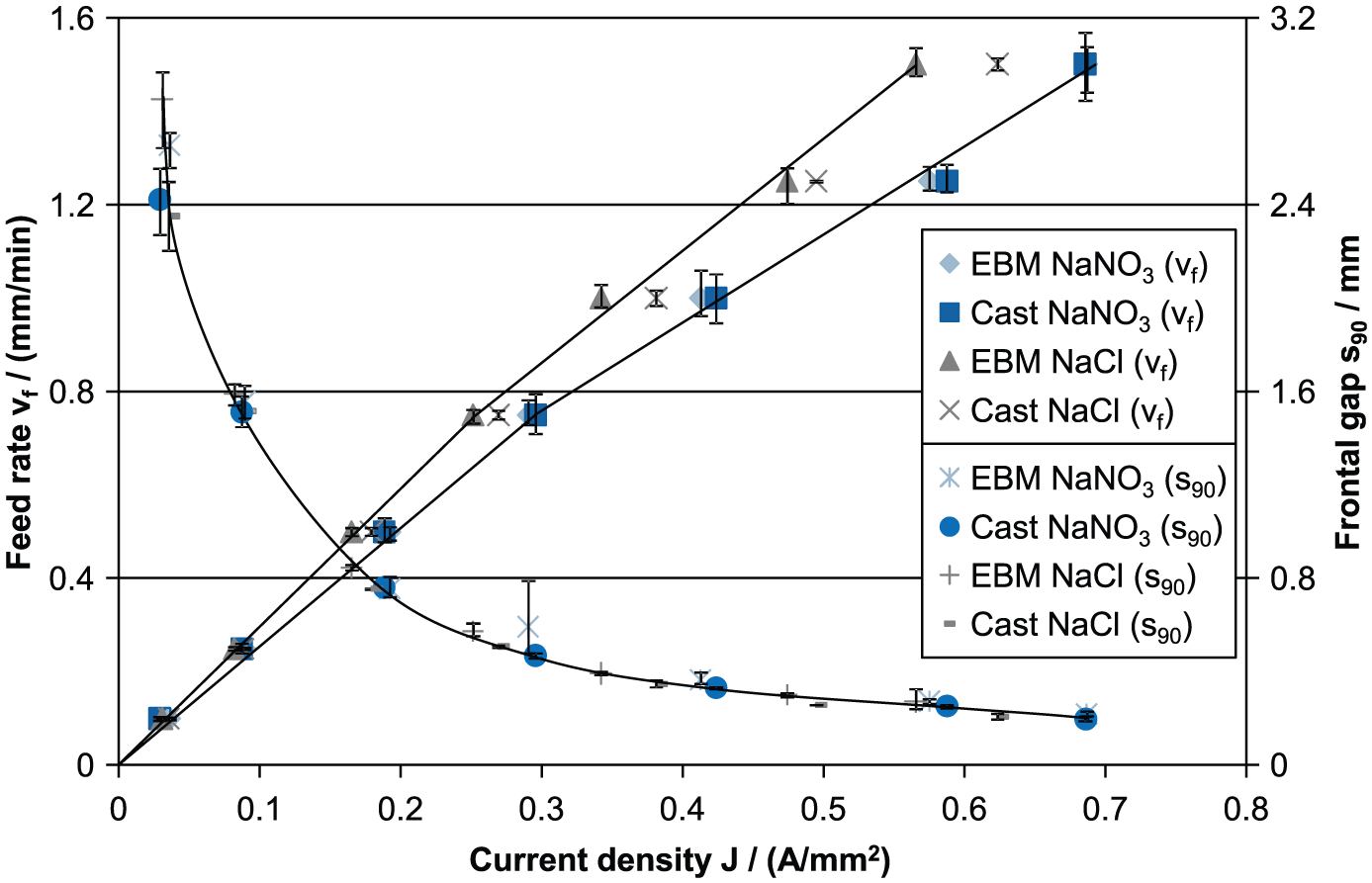
Feed rate–current density and frontal gap–current density curves of different microstructured TNB-V5 and different electrolyte systems.
In the following, the influence of the casted and electron beam melted specimen on material removal rates will be considered. The vf-J curves of the two materials machined in sodium nitrate and sodium chloride are quite similar. Nevertheless, it is recognizable that the electron beam melted material has a higher current efficiency than the conventionally casted material. Using sodium chloride the difference between both materials is higher than using sodium nitrate. These characteristics will be discussed with the help of the cross sections later on.
Moreover, a change in the electrolysis mechanism can be detected at current density of approximately 0.3 A/mm2 (cp. Figure 2). The break of the material removal rate can be identified significantly for both materials and both electrolytes. A change in the electrochemical valency of one or more alloying elements is responsible for this turning point in the curves. 9 The different material removal rates for the investigated materials are summarized in Table 3 and split into low (<0.3 A/mm2) and high (>0.3 A/mm2) current densities.
Material removal rates of EBM and casted materials with NaNO3 and NaCl split into low current density (lc < 0.3 A/mm2) and high current density (hc < 0.3 A/mm2).
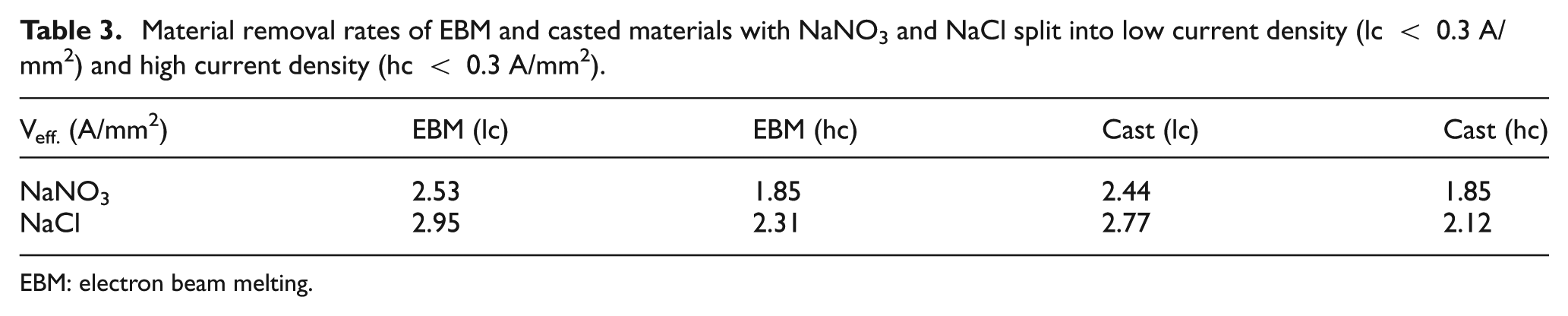
EBM: electron beam melting.
Ra-J curves
Figure 3 shows the resulting surface roughness according to the current density. First, an improvement in surface roughness is recognizable with an increase in current density. This behavior is typical for ECM. Furthermore, the achievable machining results regarding the surface roughness between sodium nitrate and sodium chloride are significantly different. For current densities above 0.2 A/mm2, the best surface roughness was achieved using sodium nitrate. An explanation for the poorer surface roughness using sodium chloride could be a different electrochemical behavior of the α- and γ-phase. This claim is later examined more closely by examining the cross sections.
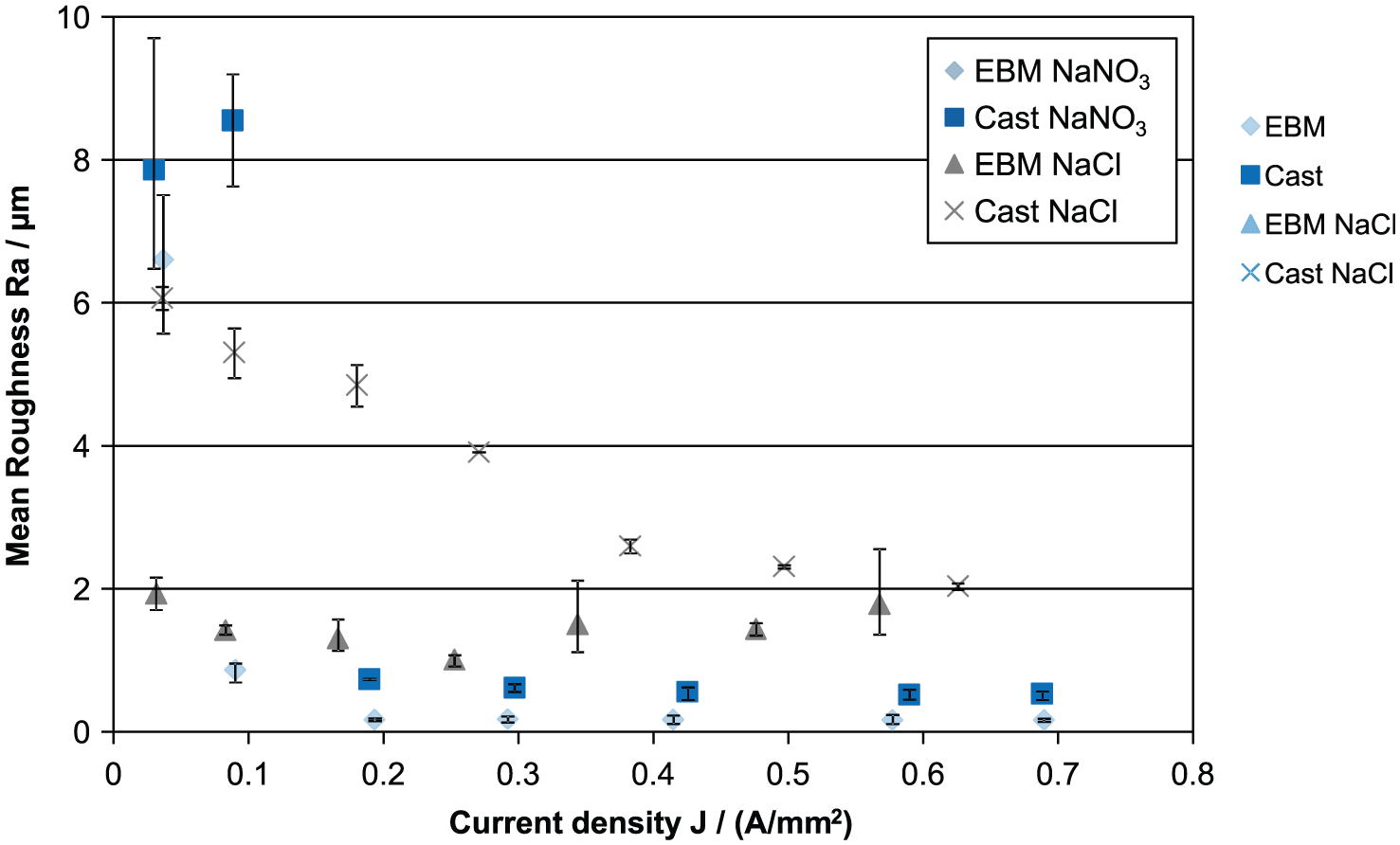
Surface roughness–current density curves of different microstructured TNB-V5 and different electrolyte systems.
Despite the differences in the surface roughness using the two electrolyte types, a contrast between casted and electron beam melted material can be recognized as well. The electron beam melted material generated usually a lower surface roughness using both sodium chloride and sodium nitrate. The only exception is seen at low current densities for sodium nitrate. For an explanation of these correlations, a closer look to the microstructure is necessary.
Cross sections of the different materials and electrolytes are shown in Figure 4. The workpieces were machined with a feed rate of 1 mm/min, which is equivalent to a current density of 0.4 A/mm2. For both materials, the microstructure shows no dependence on directional property. The ECM process does not affect the microstructure in the bulk material due to the chemical dissolution. Therefore, the microstructure of the bulk material is seen in Figure 4. The microstructure of the EBM and casted material is totally different. Due to the small melt pool and the rapid cooling, the microstructure of the EBM material exhibits an extremely fine grain size as already shown for other EBM materials in the literature.1,15 The casted material shows a near lamellar structure of the γ-TiAl alloy TNB-V5. The structure consists of colonies with α- and γ-lamellae and some small grains. This microstructure is also typical for casted γ-TiAl alloys. 16

Cross sections of electrochemical machined TNB-V5 with different microstructures and different electrolyte systems.
The surfaces of the electrochemical machined workpieces illustrate the different electrochemical behavior of the different phases to the respective electrolyte system. Due to its fine grain size, the EBM material exhibits a homogeneous dissolution behavior. Using sodium chloride the surface developed a slightly higher roughness with very fine and short sticks. The difference between the electrochemical behaviors of the casted microstructure in sodium nitrate and sodium chloride is more significant. In sodium nitrate, the dissolution behavior of the α- and γ-phase is nearly similar, but grain boundaries dissolve preferential, which affects the mean surface roughness of the casted material (compare Figure 3). Sodium chloride effectuates a faster dissolution behavior of the γ-phase compared to the α-phase. Therefore, an extremely rough surface develops. On closer inspection of the EBM and sodium chloride cross section, a faster dissolution of the γ-phase can be observed as well, but due to the fine grain size, this behavior is less significant. Different dissolution behavior of the phases influences the current efficiency as well. α-phases can be flushed mechanically out of the material due to the dissolution of ambient γ-phase.
The investigations of the cross sections enable two statements. Sodium chloride caused different dissolution characteristics of the α- and γ-phase in γ-TiAl TNB-V5. Furthermore, the dissolution is more homogeneous using sodium nitrate electrolyte and the fine grain size of the EBM material enables fine roughnesses.
Current density–electrode potential
To characterize the dissolving behavior of the two microstructures, investigations on the current density–electrode potential behavior were performed. Due to the prevention of a passivation layer using sodium chloride, no Flade potential and thus no passivation, activation, and breakdown potential exist. 17 The rest potential is located at the origin and new knowledge cannot be generated. Therefore, investigations were only performed using sodium nitrate.
Figure 5 shows the two curves of the EBM and casted structure with sodium nitrate electrolyte. The curve characteristics are similar regarding the rest, passivation, and activation potential. The biggest difference is recognizable in the breakdown potential. For the casted material, the breakdown potential is lower (1.42 V) than the EBM breakdown potential (1.65 V). Due to the nominal equal chemical composition, the declaration for this behavior can be traced back to the different microstructure. Lindenlauf 9 has shown that a more stable cover layer results in finer structures. Such a more stable cover layer results in higher breakdown potentials. The breakdown potential loss is not available for the material dissolution and has to be adapted by the machining parameter in advance.
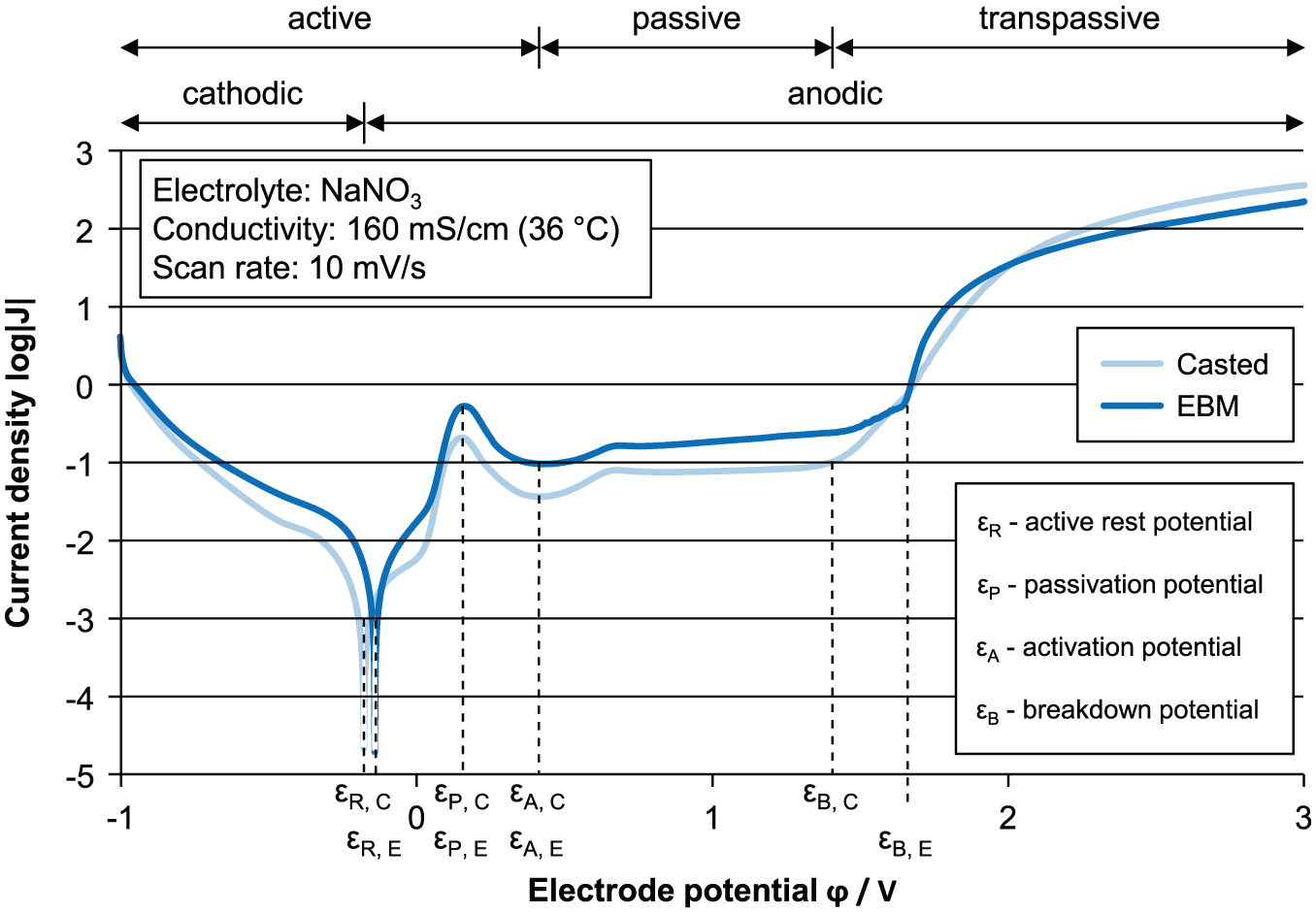
Current density–electrode potential of casted (c) and electron beam melted (e) TNB-V5.
Conclusion
ECM is capable of machining materials regardless of their mechanical properties. Furthermore, the workpiece rim zone is not affected thermally or mechanically. In combination with high material removal rates, ECM could be a capable manufacturing technology for γ-TiAl. Due to the chemical dissolution, the materials microstructure is highly important for the machining result. Therefore, this article presented investigations on the differences between casted and electron beam melted γ-TiAl TNB-V5 regarding their ECM behavior. To analyze the electrochemical machinability of the different microstructures, feed rate-, frontal gap width-, and surface roughness–current density curves were examined using sodium nitrate and sodium chloride electrolytes. Furthermore, cross sections of the machined workpieces and current density–electrode potential curves were investigated.
Due to the similar nominal chemical compositions, the machining differences were caused by the specific microstructures. While the frontal gap width–current density curves showed nearly no deviation between the casted and the EBM structure and different electrolytes, the feed rate–current density curves showed a higher current efficiency for the EBM material in both electrolytes. As expected, the current efficiency was improved by sodium chloride electrolyte for both materials. The largest differences were detected in surface roughness–current density curves. EBM structure generates a better surface roughness than the casted material. Cross sections of the different microstructures and electrolytes clarify the influence of the structure on the machining results. Sodium chloride caused a faster dissolution behavior of the γ-phase compared to the α-phase. Due to the fine grain size of the EBM material, a more homogeneous dissolution behavior effectuates, which leads to better surface roughness. In sodium nitrate, an increased dissolution of the grain boundaries in the casted material was observed.
Further investigations on the electrochemical behavior of the different microstructures were achieved by potentiostatic polarization curves. The general curve characteristic is very similar. Only significant difference constitutes the breakdown potential. Due to a more stable cover layer on the EBM material caused by the fine grain size, the breakdown potential is higher.
The investigations of this article showed a better machining result for the EBM material regarding the surface roughness compared to casted material. Higher material removal rates were also achieved with the EBM material. Future studies must demonstrate the mechanical fitness of the EBM microstructure. Depending on the application, an EBM production of γ-TiAl parts in combination with an ECM finishing could be a high-efficient manufacturing chain.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research and development project “ETurbo” is funded by the German Federal Ministry of Education and Research (BMBF) within the Framework Concept “Research for Tomorrow’s Production” (funding number 02PN2071) and managed by the Project Management Agency Karlsruhe (PTKA). The authors are responsible for the contents of this publication.
