Abstract
Electrochemical machining is a modern machining technique that plays an important role in the applications of aerospace, die and electronic industries. Electrochemical machining is used to machine difficult-to-machine materials and complex shapes. Machining of alloy steels especially 20MnCr5 is very important for its wide variety of applications such as piston bolts, spindles, camshafts, gears, shafts and other mechanical controlling parts. Machining of the above components with conventional machine tools is a burdensome task. Hence, in this work, an investigation was made to study the electrochemical machining characteristics of 20MnCr5 alloy steel. Two electrolytes, namely, aqueous sodium chloride (NaCl) and potassium dichromate (K2Cr2O7) mixed aqueous NaCl, were used to investigate the machining performance. K2Cr2O7 was selected for its oxidizing characteristics and was included in small proportions in aqueous NaCl bath. The influence of predominant electrochemical machining process parameters such as applied voltage (V), inter electrode gap and electrolyte concentration was studied on the material removal rate and surface roughness (Ra). Scanning electron microscope photography of surface of the 20MnCr5 specimen machined with electrochemical machining was studied to understand the effect of electrolytes during the machining. The contour plots were generated to study the effect of process parameters as well as their interactions. It was noted in the study that the presence of K2Cr2O7 in aqueous NaCl electrolyte increases the material removal rate significantly. The process parameters are optimized through genetic algorithm-desirability function. Optimized operating conditions were found to be quite close with experimental results.
Keywords
Introduction
Electrochemical machining (ECM) has been in the industrial interest for the last few decades due to its specific advantages such as no tool wear, mechanical forceless machining, no thermally influenced machining zones, high surface quality, low roughness and stress-free surface products. ECM is the reverse of electroplating process. It is one of the advanced machining processes (AMPs) capable of machining hard materials and complicated shapes. ECM is a controlled atomic-level anodic dissolution process of the workpiece that is electrically conductive by a shaped tool through an electrolyte. In ECM, workpiece is the anode and the tool is the cathode and the electrolyte is pumped into the inter electrode gap (IEG) between the tool and the workpiece. High current is passed through this electrochemical circuit to dissolve metal from the workpiece. The movement of ions is accompanied by electrons’ flow, in the direction opposite to the positive current in the electrolyte. The amount of substance dissolved is directly proportional to the amount of current flow into the circuit according to Faraday’s law. The metal hydroxides and other by-products generated during the machining are flushed away from the gap by the high-velocity electrolyte jet. ECM and its hybrid processes such as electrochemical honing (ECH) and electrochemical discharge machining (ECDM) are gaining momentum in the industrial applications.1,2 ECM of metals, particularly steels, plays an important role in many industrial applications. The use of alloy steel 20MnCr5, a good wearing resistance material in piston bolts, spindles, camshafts, gears, shafts and so on, fascinated the authors to study its ECM characteristics. Yielding higher material removal rate (MRR) of workpiece is most important in this machining process in order to utilize its full potential. The dissolution rate is highly reliant on the selection of electrolytes and its current-carrying capacity.
3
Electrolyte composition (ratio of NaNO3:NaCl), temperature and concentration play significant role in the improvement of surface quality of workpiece.
4
Higher concentrated electrolyte solution increases the dissolution rate. But excess concentration allows the crystal formation, which damages the accessories of ECM and reduces the volume of electrolyte in the flow pipes. Weak solution makes intermittent passivity on the workpiece resulting in poor machining. The conductivity of electrolyte depends not only on the concentration but also on the ionic interaction. Thus, the current-carrying process done by the base electrolyte is small, but H+ and OH− ions produced in electrolysis of water play vital role.
3
Water–isopropanol–glycerin chloride blend was considered as electrolyte for high anodic dissolution of hafnium.
5
The results point out that the anodic potential or the temperature of the electrolyte bath has significant effect on the hafnium anodic dissolution rate. Change in pH value of electrolyte, during the machining, contributes more to the dissolution rate than the hydroxyl ion’s catalytic effect.
6
Three prominent ECM characteristics such as metal dissolution, gas evolution and the solid surface film formation can be observed during machining. At low current density, exclusive oxygen evolution is observed. With increasing current density, formation of cation species is enhanced, while the oxygen evolution diminishes. At high current density, surface film formation such as metal oxides is very less. Haisch et al.
7
experimented with NaNO3 and NaCl salt solutions in order to study the ECM characteristics of 100Cr6 steel. The formation of more Fe3+ ions at the current density of 21 A/cm2 suggests the increase in current efficiency (
Experimentation
Electrical conductance
Electrical conductance of NaCl was tested at different concentrations in distilled water, and the results are shown in Figure 1. The concentration of 150 g of NaCl/L of water was considered as the optimum concentration to test the concentration requirement of K2Cr2O7. Conductance was measured for different concentrations of K2Cr2O7 in the selected concentration of NaCl in water. Here, the concentration of NaCl in water was taken as 150 g/L with different concentrations of K2Cr2O7 in it. As shown in Figure 2, at 5 g of K2Cr2O7 per l00 mL of aqueous NaCl, pH value is slightly above 4, which is adequate to accelerate the anodic dissolution rate.
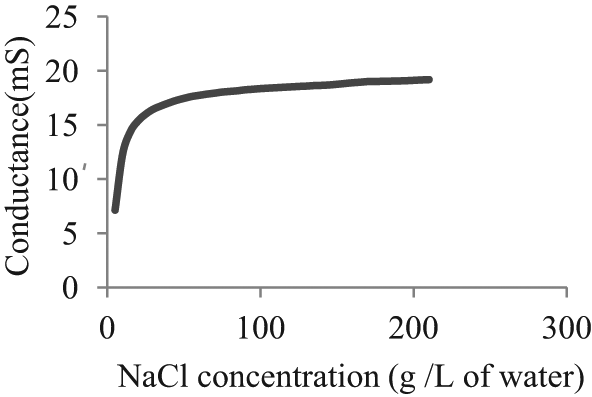
Concentration of NaCl with conductance.
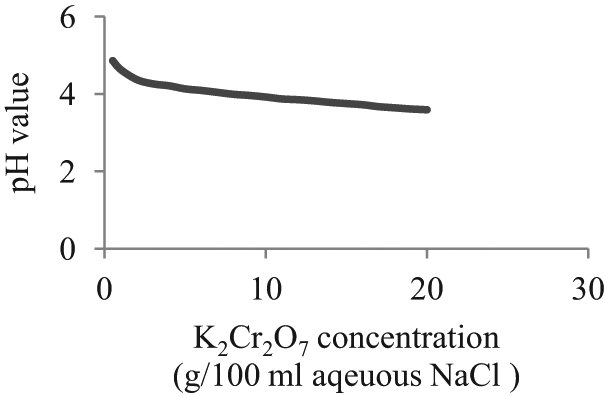
pH value change with concentration of K2Cr2O7.
Experimental work in ECM
Schematic diagram of ECM setup is shown in Figure 3. It comprises a power supply system, electrolyte supply and filtering system, tool and tool feed mechanism, work holding and position system, control panel and frame and housing. A 5–30 V direct current (DC) potential with current adjustable up to 300 A is applied across the IEG between a brass tool and an anode workpiece. The tool is fed against the workpiece, which is firmly fixed on the vice. IEG, sufficient voltage (V) and current are set in the control panel.
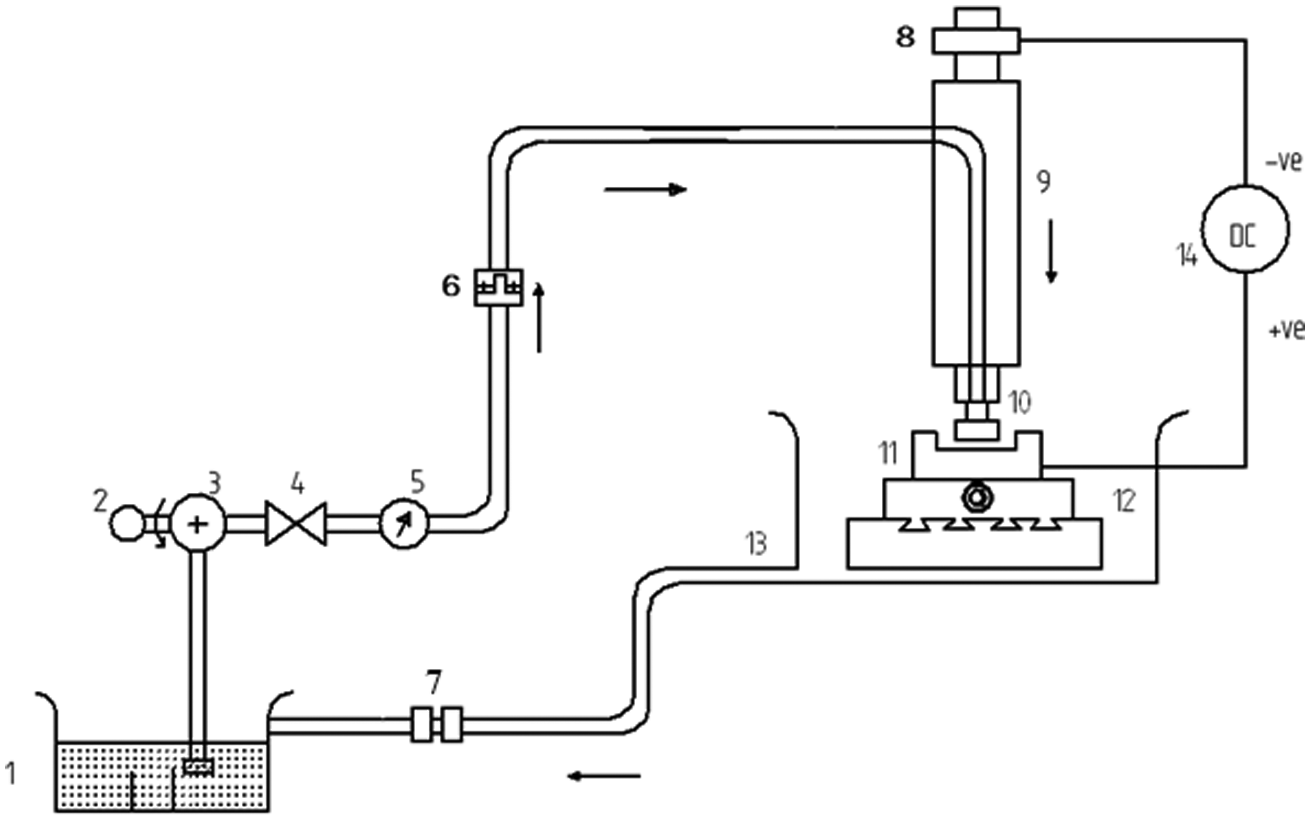
Schematic diagram of ECM apparatus.
Commercially available 20MnCr5 alloy steel specimens with Φ 40 mm diameter and 10 mm height were used in the experiment. The composition of the steel is as follows—iron: 97%, C: 0.20%, Mn: 1.25%, Si: 0.25%, P: ≤0.035%, S: ≤0.035%, Cr: 1.15% and Al: ≥0.020. Aqueous NaCl and K2Cr2O7 mixed aqueous NaCl electrolytes are axially fed into machining zone. Figure 4 shows photographic view of experimental setup of the ECM system. Figure 5 presents the three-dimensional (3D) model and cross-sectional view of the tool. The experiments were based on central composite design (CCD) of response surface methodology (RSM) and were designed with the help of the software Design-Expert 7.1. 13 RSM is a collection of mathematical and statistical techniques useful for modeling and optimizing the response variable models involving quantitative independent variables. 14 RSM was effectively implemented in ECH process and its effectiveness was proven. 4 In this work, 20 tests were carried out with different parameter combinations. At low current densities, the MRR is low. In order to have a high current efficiency, higher current density of 110 A/cm2 is used.

Electrochemical machining apparatus.
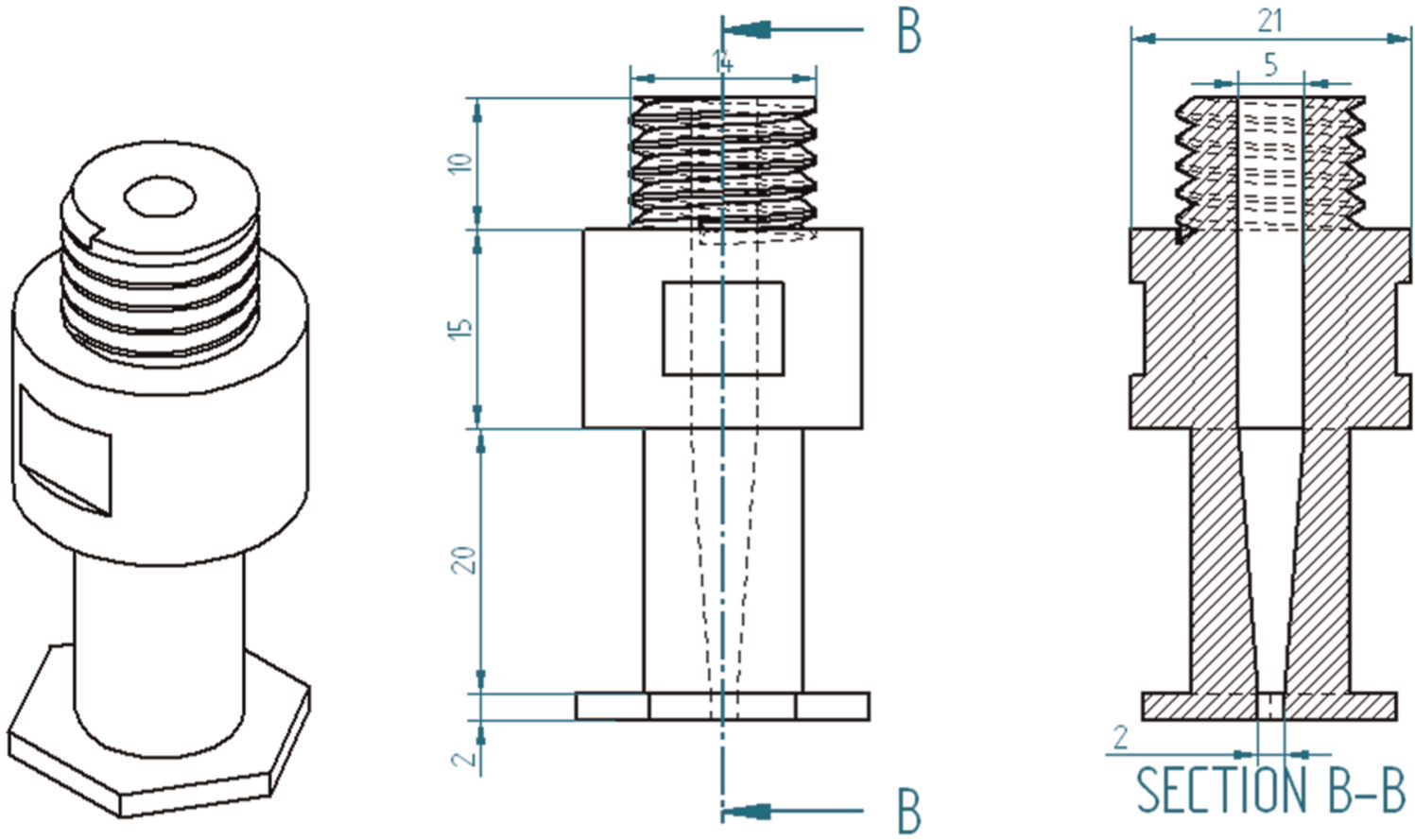
Tool 3D model and cross-sectional view.
The main process parameters governing the ECM process are voltage (V), IEG and EC according to the literature. Therefore, these parameters are considered in this work and its levels are shown in Table 1.
Process parameters and their values at different levels.

In this work, MRR was measured based on weight loss during machining time

Weights were measured using weighing machine with least count of 1 mg and maximum weight of 1 kg. The surface roughness (Ra) was measured with the Mitutoyo surface roughness tester with sampling length of 10 mm. The results are the average of the three measurements. Table 2 shows the experimental results of aqueous NaCl and K2Cr2O7 mixed aqueous NaCl electrolyte.
Central composite design and experimental results.
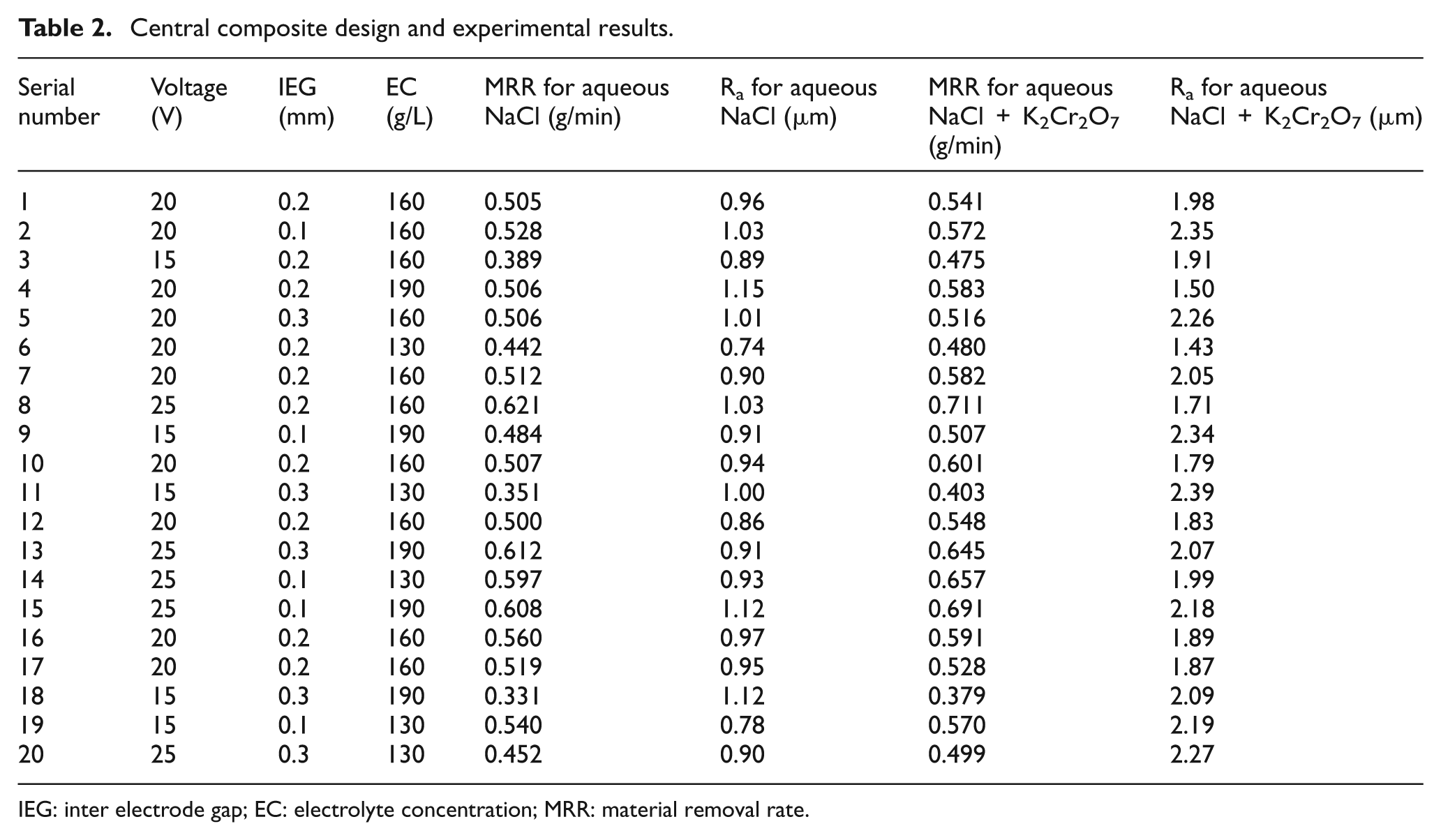
IEG: inter electrode gap; EC: electrolyte concentration; MRR: material removal rate.
Electrolyte and reactions
The following parameters were set as constant during the experimentation. These values have been set after conducting several tests and were found to be optimal:
Current (
Machining time (
Flow rate (
Feed rate (F): 0.1 mm/min;
Concentration of K2Cr2O7: 5 g/L of water.
A higher feed rate produces higher MRR. However, higher feed rate of tool reduces the IEG and it makes more difficult to allow the full flow of the electrolyte. This circumstance leads to short-circuiting, intermittent sparks that damage the tool and workpiece. Moreover, feed rate is directly proportional to MRR. Hence, constant feed rate (F) of 0.1 mm/min is considered for good surface finish. The experiments were conducted in turbulent electrolyte flow conditions with the flow rate (
Aqueous NaCl solution dissociates as
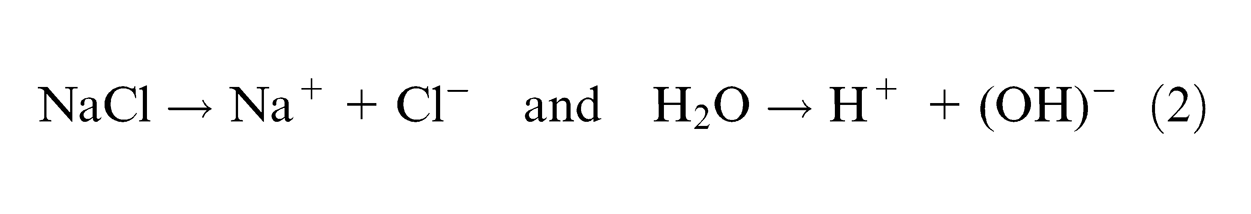
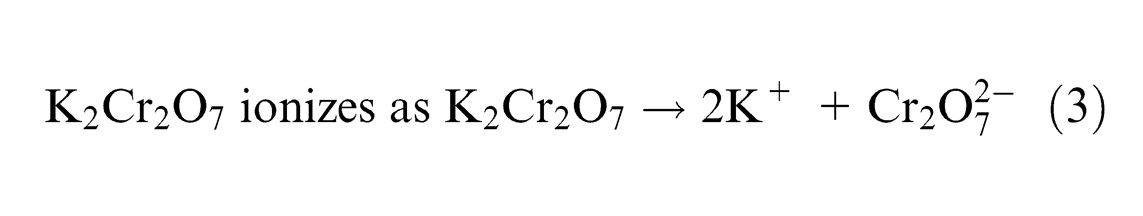
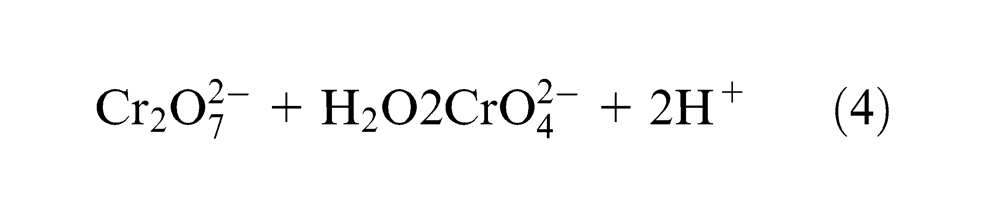
Dichromate ions react with excess hydrogen ions and form water as
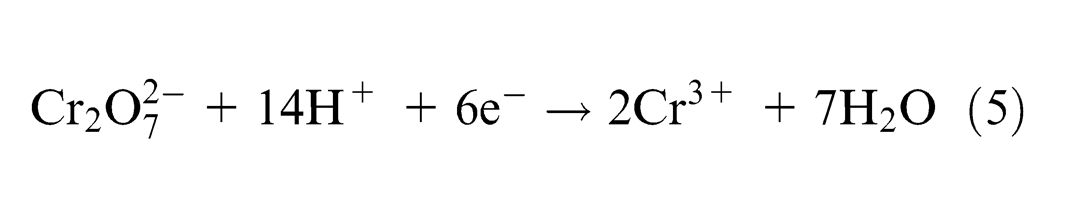
The alloy steel 20MnCr5 workpiece (anode) undergoes an oxidation reaction by which the atoms are ionized.
The divalent ferrous dissolution occurs at the current efficiency (
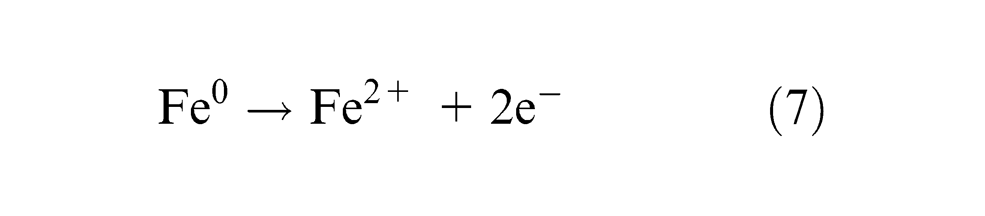
The trivalent ferrous dissolution occurs at the current efficiency (
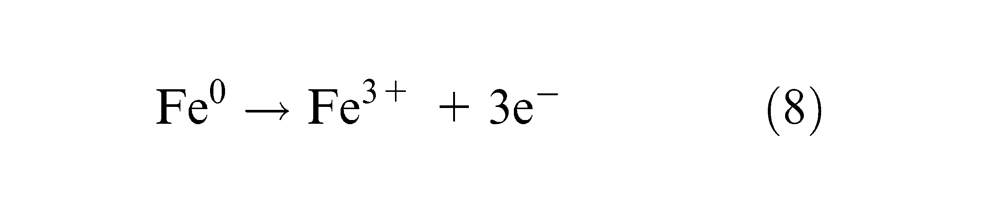
Hydroxyl ion (OH)− evolves oxygen at the anode as
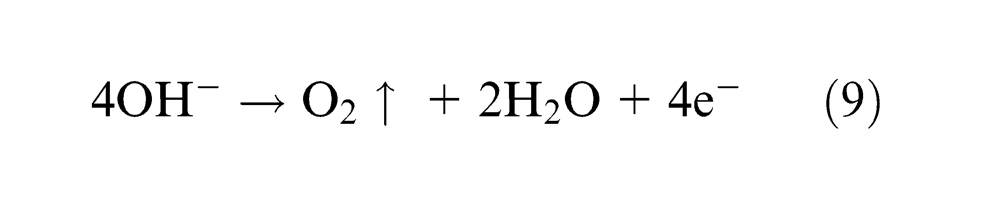
This oxygen formation dominates if no metal dissolution occurs. For NaCl solution, oxygen formation is less.
The hydrogen ions will take away electrons from the cathode (tool) and form hydrogen gas at the cathode
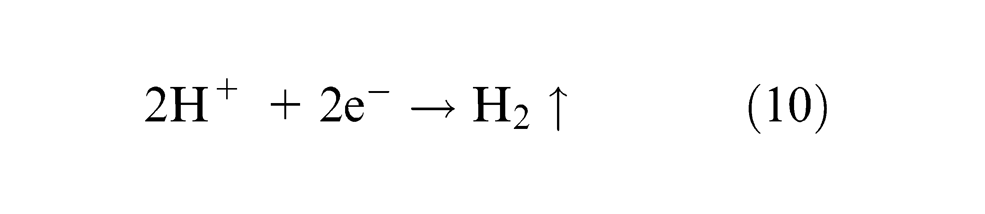
Choking of the flow of electrolyte by hydrogen bubbles is avoided by the high velocity of electrolyte jet. Within the electrolyte, iron ions would combine with chloride ions to form iron chloride and similarly, sodium ions would combine with hydroxyl ions to form sodium hydroxide
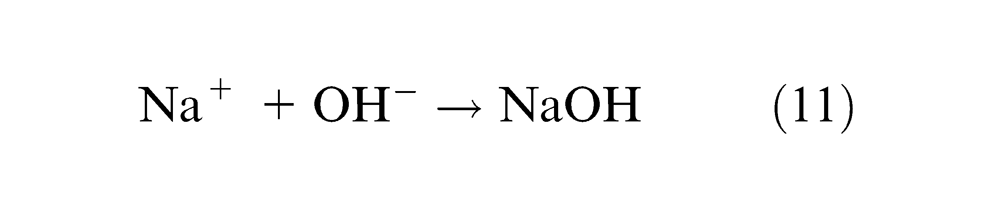
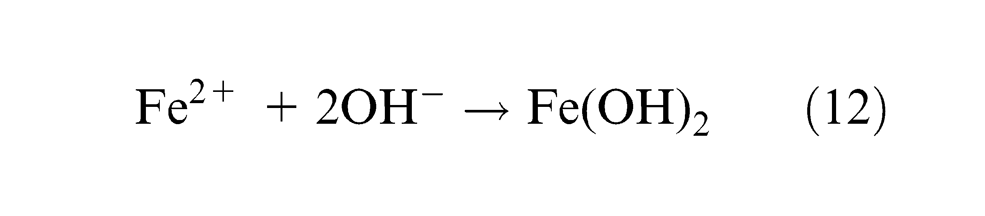
Fe(OH)2 are formed and precipitated as sludge. Due to further reaction, some amount of Fe(OH)3 is also possible. Dichromate oxidizes Fe2+ to Fe3+ ions, which confirm the more electrochemical dissolution on the surface of the workpiece. The above reactions confirm the gradual machining of workpiece and the sludge gets precipitated. As the material removal takes place due to atomic-level dissociation, the machined surface is of good surface finish and stress free. The experimental results of working of this electrolyte are tabulated in Table 2. Since K2Cr2O7 is a good oxidizing agent, it electrochemically reacts with aqueous NaCl solution. During the process of ECM, the hydroxyl ions’ formation is important for machining. Because these ions react with workpiece material to form metal hydroxide which is considered as material removal in this machining.
Analysis of variance of the models
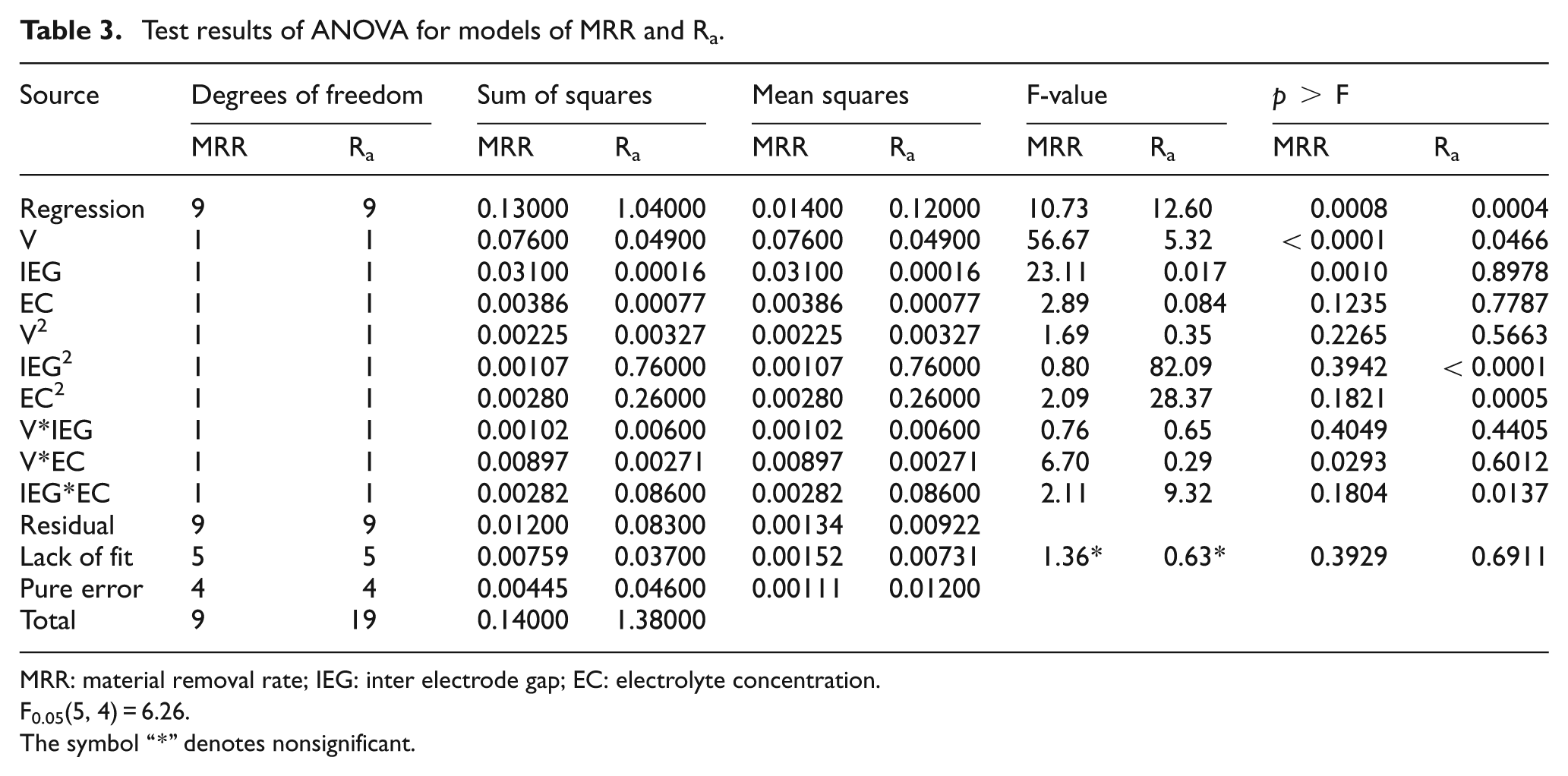
The analysis of variance (ANOVA) and Fisher’s test (F-ratio) have been performed to justify the goodness of fit of the mathematical models. 14 In order to study the effect of the ECM process parameters, a second-order polynomial model with input variables is fitted in the following equation. Table 3 refers to the ANOVA results for models MRR and Ra
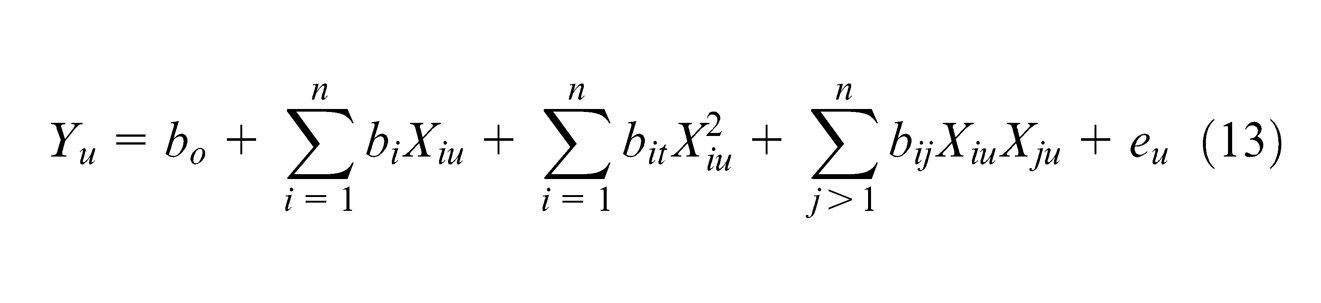
Test results of ANOVA for models of MRR and Ra.
MRR: material removal rate; IEG: inter electrode gap; EC: electrolyte concentration.
F0.05(5, 4) = 6.26.
The symbol “*” denotes nonsignificant.
where
The MRR model F-ratio value of 10.73 implies that model is statistically significant. In this case, V, IEG and V*EC are significant model terms. The value of R2 = 91.47% means that the regression model provides an excellent explanation of the relationship between the variables and the response MRR. Standard F-ratio (F 0.05,5,4) for “lack of fit” of this model is 6.26. The calculated F-ratio value of 1.36 is smaller than the standard, which means that there is no strong evidence of lack of fit. Thus, the model equation (14) is adequate

The Ra model F-value of 12.60 implies that the model is significant. In this case, V, IEG2, EC2 and IEG*EC are significant model terms. The value of R2 = 92.65% means that the regression model provides an excellent explanation of the relationship between the variables and the response Ra. The calculated F-ratio value for “lack of fit” of 0.63 is smaller than the standard (F0.05,5,4) = 6.26. Thus, the developed model equation (15) can be used for the further analysis

Results and discussion
The amount of different substances dissolved by the same quantity of electricity is proportional to their chemical equivalent weights. Since the electrolyte serves as the conductor of electric current, Ohm’s law can be applied to this conductor. The simple expression of Faraday’s law can be described as
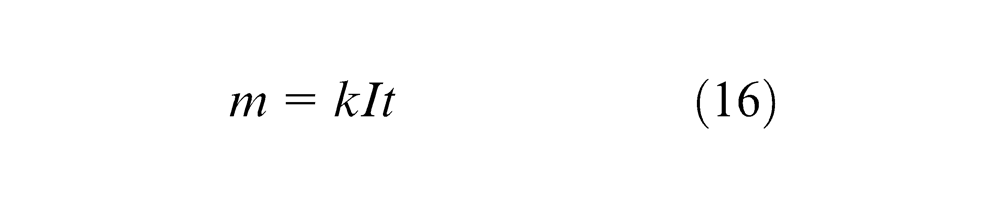
where
In this work, 20MnCr5 alloy steel comprises different elements. The amount of mass (
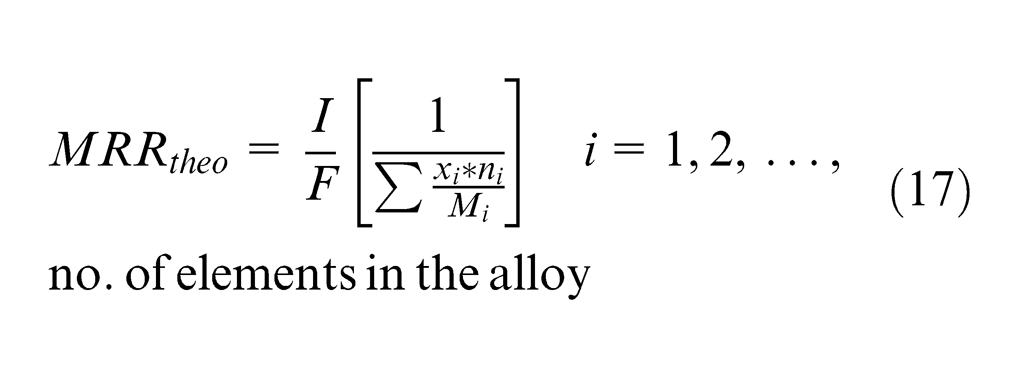
where
Faraday’s law of electrolysis predicts the rate at which metal is removed from a workpiece. The current efficiency (
The type of electrolytes used in the process affects the quality of surface finish of jobs machined with ECM. Sodium chloride (NaCl) electrolyte tends to produce matte finish with alloy steels. Higher voltages are applied to break the oxide film and its disruption is non-uniform, which results in deep grain boundary attack of the metal surface. Pitting on metal surfaces occurs sometimes due to the evolution of the gas bubbles, which rupture the oxide film. Identifying the best electrolyte selection plays an important role in ECM. In Figure 6, it is observed that current efficiency is slightly higher with K2Cr2O7 mixed aqueous NaCl electrolyte for all tests. This is because of the oxidation due to K2Cr2O7 in addition to regular metal hydroxides (sludge) and erosion on the surface.
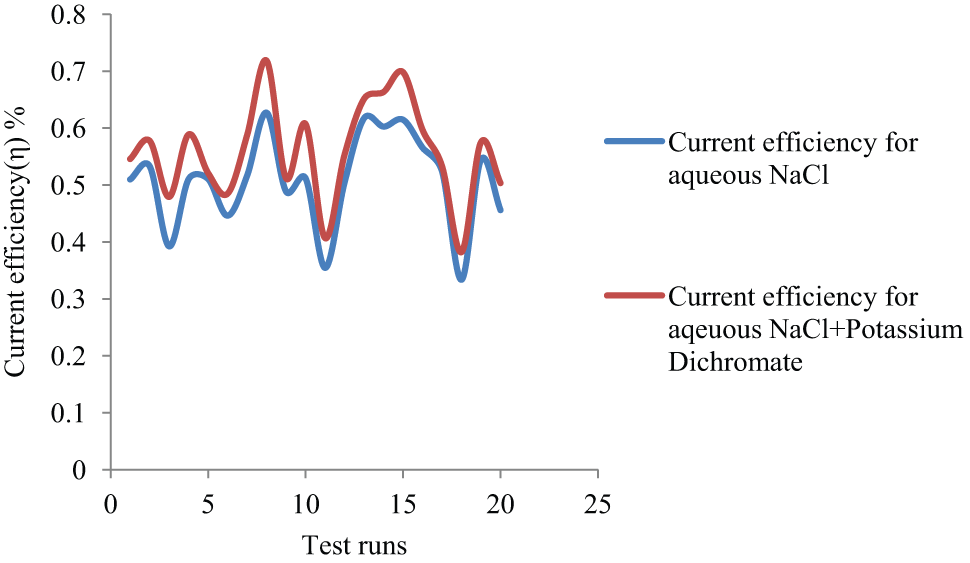
Current efficiency versus different electrolytes.
Parametric influence on MRR and Ra for K2Cr2O7 mixed aqueous NaCl electrolyte
In order to analyze the effect of the various process variables on MRR and Ra, contour plots were drawn for various combinations. Figure 7 shows the influence of the variables on the MRR for K2Cr2O7 mixed aqueous NaCl electrolyte. Increase in voltage increases the machining current in the IEG, thereby increasing the MRR.
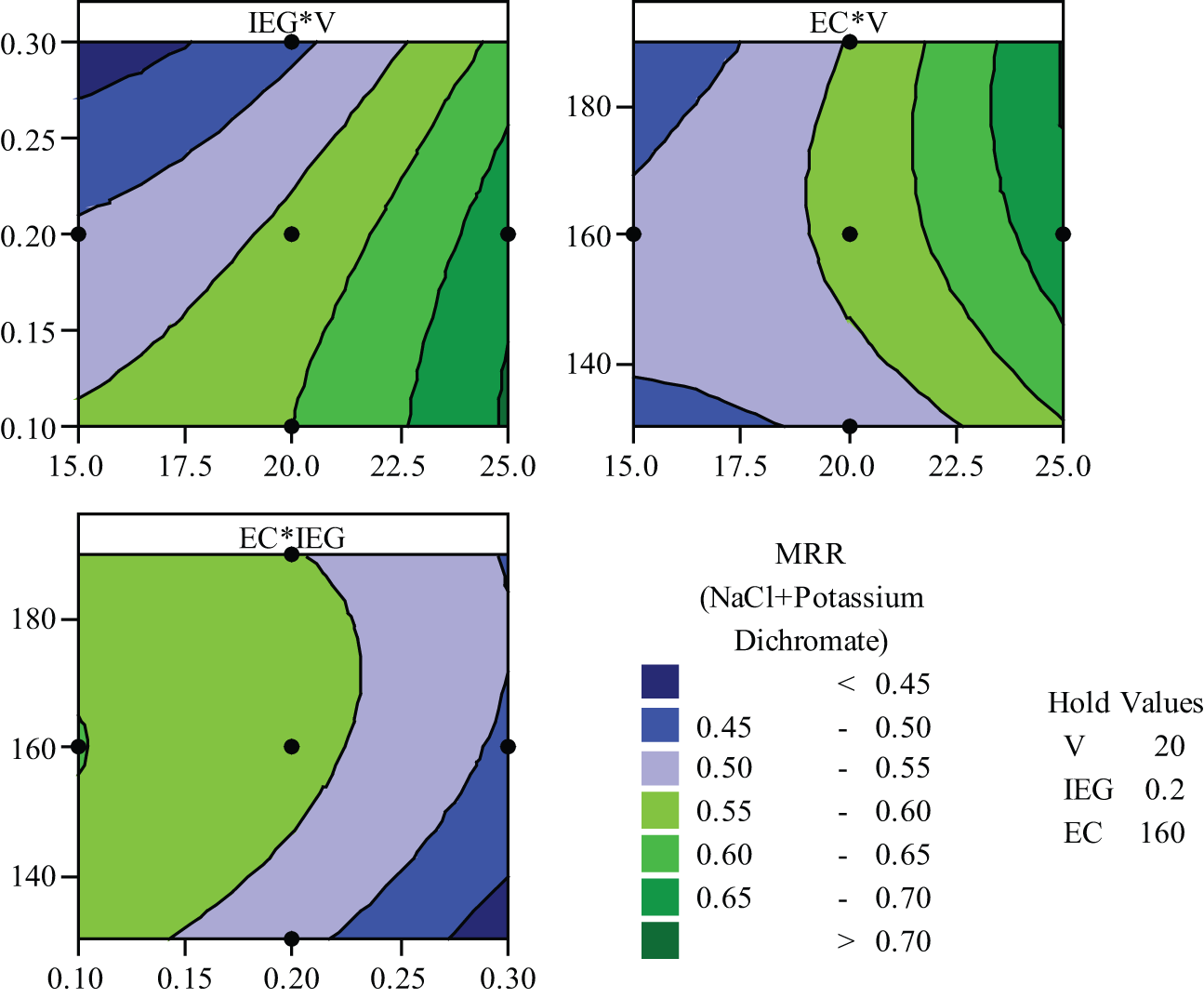
Parametric influence on MRR.
This can be seen in scanning electron micrograph of machined components at low voltage (15 V) in Figure 9(a) and (b). Surfaces look rough at low voltage (15 V). Figure 9(b) confirms more erosion on the surface of workpiece due to the presence of oxidation power of K2Cr2O7. Smaller IEG increases the rapid anodic dissolution as a result of higher current density. This confirms to the fundamental machining mechanism of ECM. It can be observed that the increase in EC increases MRR. This is attributed to the increase in the conductance of the electrolyte with the increase in concentration. Increase in both voltage (V) and EC enhances the MRR enormously. This can be seen in the second graph of Figure 7. The third graph in Figure 7 represents the effect of IEG and EC on the MRR. Since smaller IEG increases the current density and higher concentration allows more ions for ionization, its combined effect increases the MRR. Figure 8 shows the influence of process variables on the Ra for aqueous NaCl with K2Cr2O7 electrolyte. K2Cr2O7 presence influences more on the surface of the specimen. Change in pH value of electrolyte mixture during the machining affects the surface structure. IEG of 0.2 mm with voltage of 25 V produces good surface roughness (Ra). Higher concentrations of NaCl in water produce small Ra. A mild oxide deposits on the metal surface, due to the presence of K2Cr2O7, is further oxidized and is eliminated due to high velocity of electrolyte jet. On the whole, sensible non-uniform metal dissolution is created due to K2Cr2O7, which results in slightly higher Ra. Figures 9(b), 10(b) and 11(b) confirm the more anodic dissolution as well as corrosion due to K2Cr2O7 presence in the aqueous NaCl electrolyte.
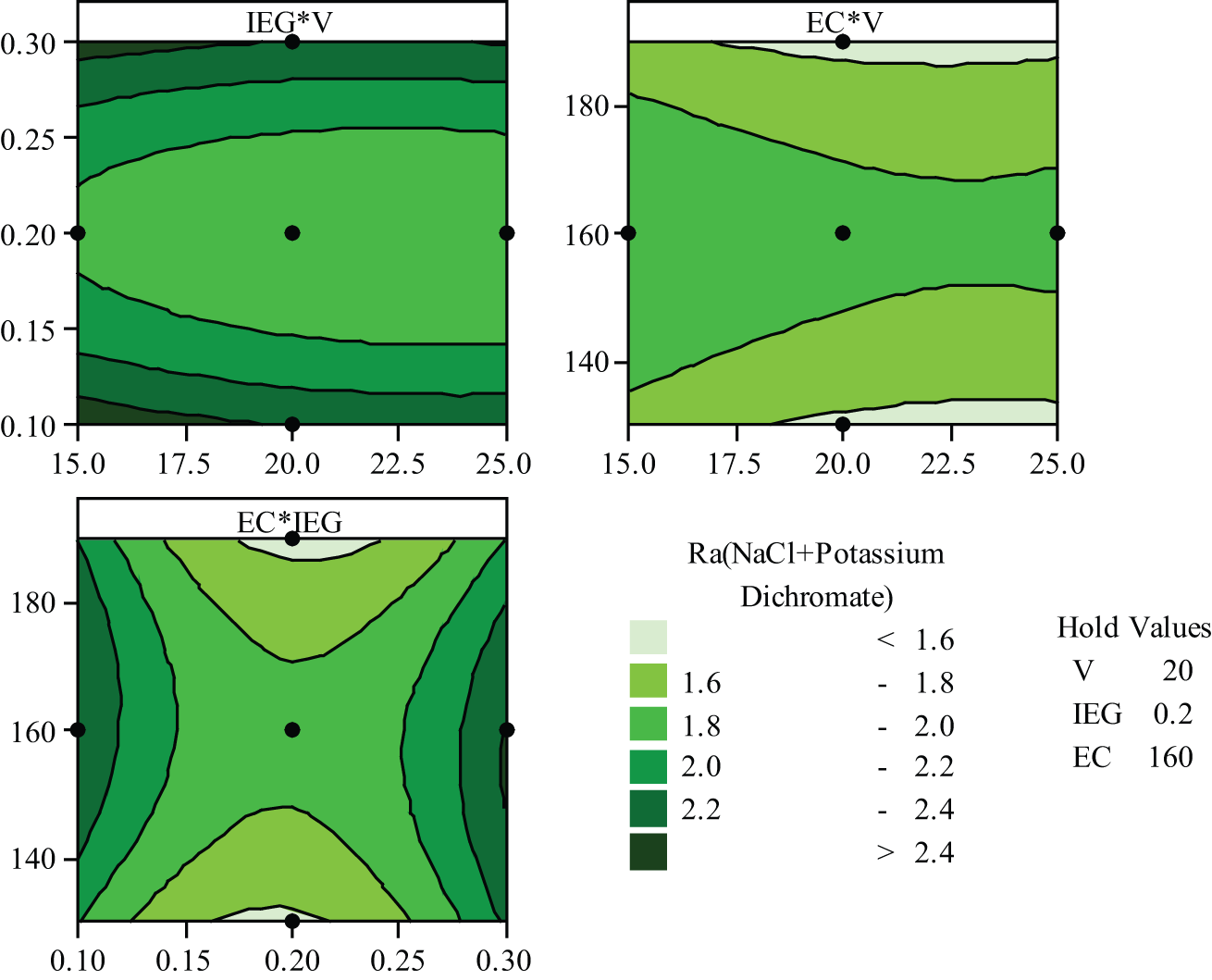
Parametric influence on Ra.

(a) Machined surface at 15 V with aqueous NaCl, (b) Machined surface at 15 V with K2Cr2O7 mixed aqueous NaCl.

(a) Machined surface at 20 V with aqueous NaCl. (b) Machined surface at 20 V with K2Cr2O7 mixed aqueous NaCl.

(a) Machined surface at 25 V with aqueous NaCl. (b) Machined surface at 25 V with K2Cr2O7 mixed aqueous NaCl.
Multi-response optimization by GA-DF optimizer
DF
DF is one of the most extensively used methods for multi-response optimization.
17
It transforms each response
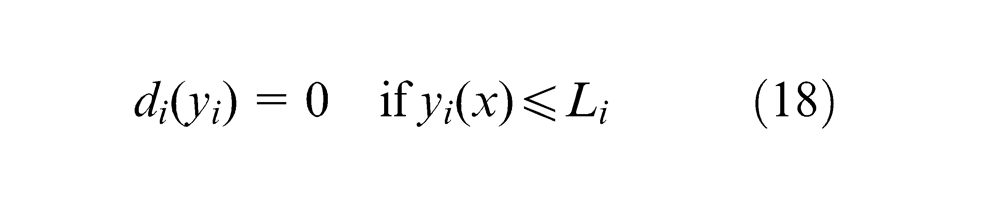
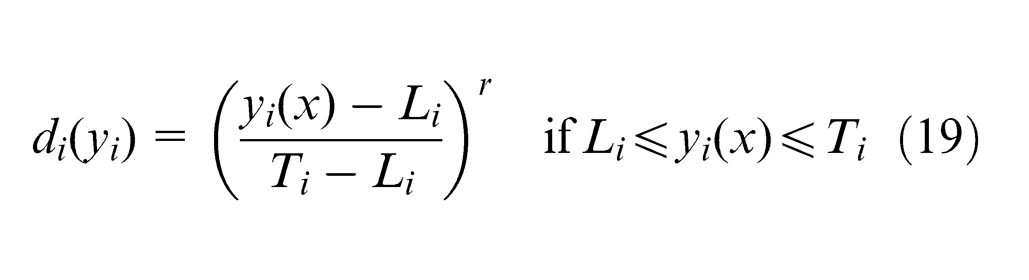
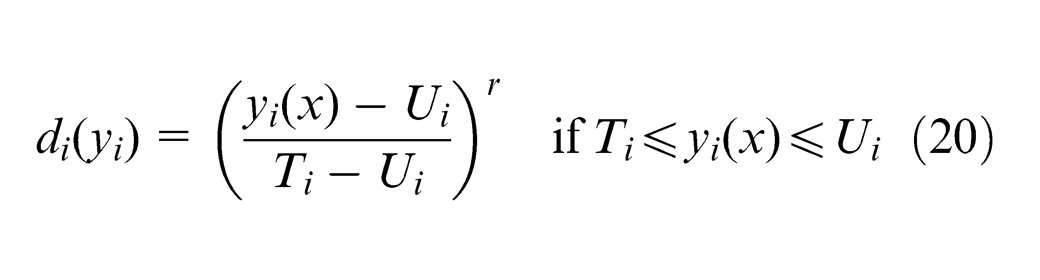
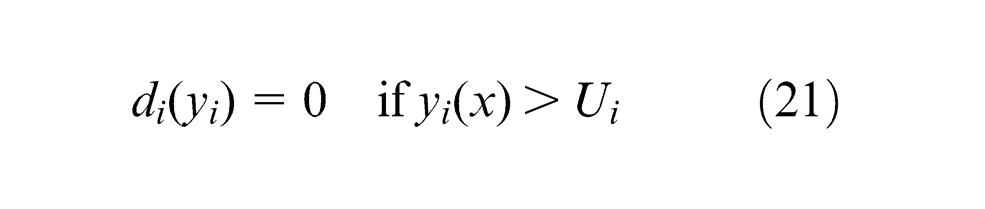
where
The individual DFs are then combined using the geometric mean, which gives the
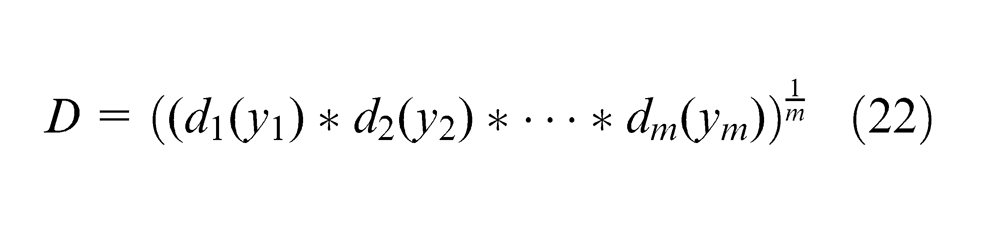
where
The composite desirability (
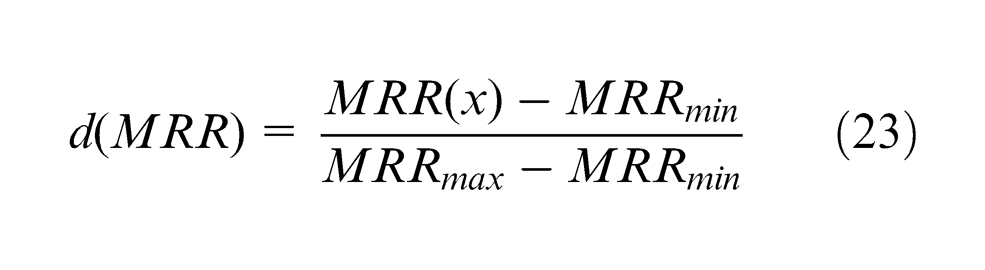
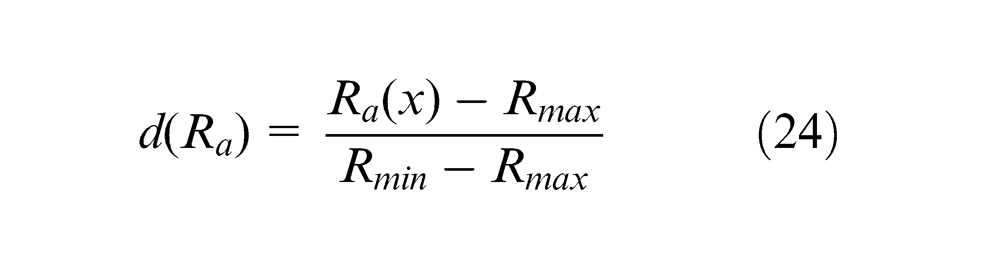
The r-value is 1 as the shape of individual desirability is considered as linear function. The composite desirability (
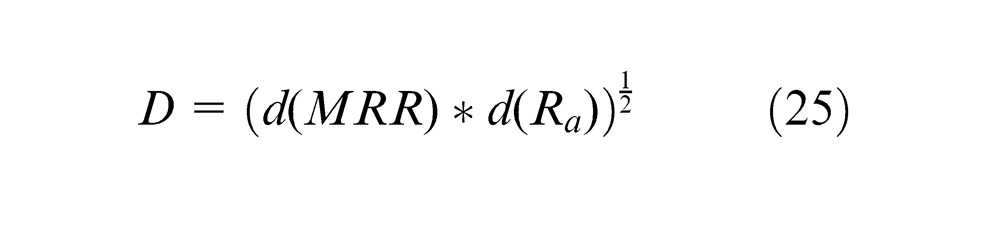
GA-DF optimizer steps
GA was developed by John Holland inspired from the evolution of biological systems.
18
GA is integrated with DF in such a way that GA-DF optimizer is developed. Optimality search is conducted to get the maximum composite desirability (
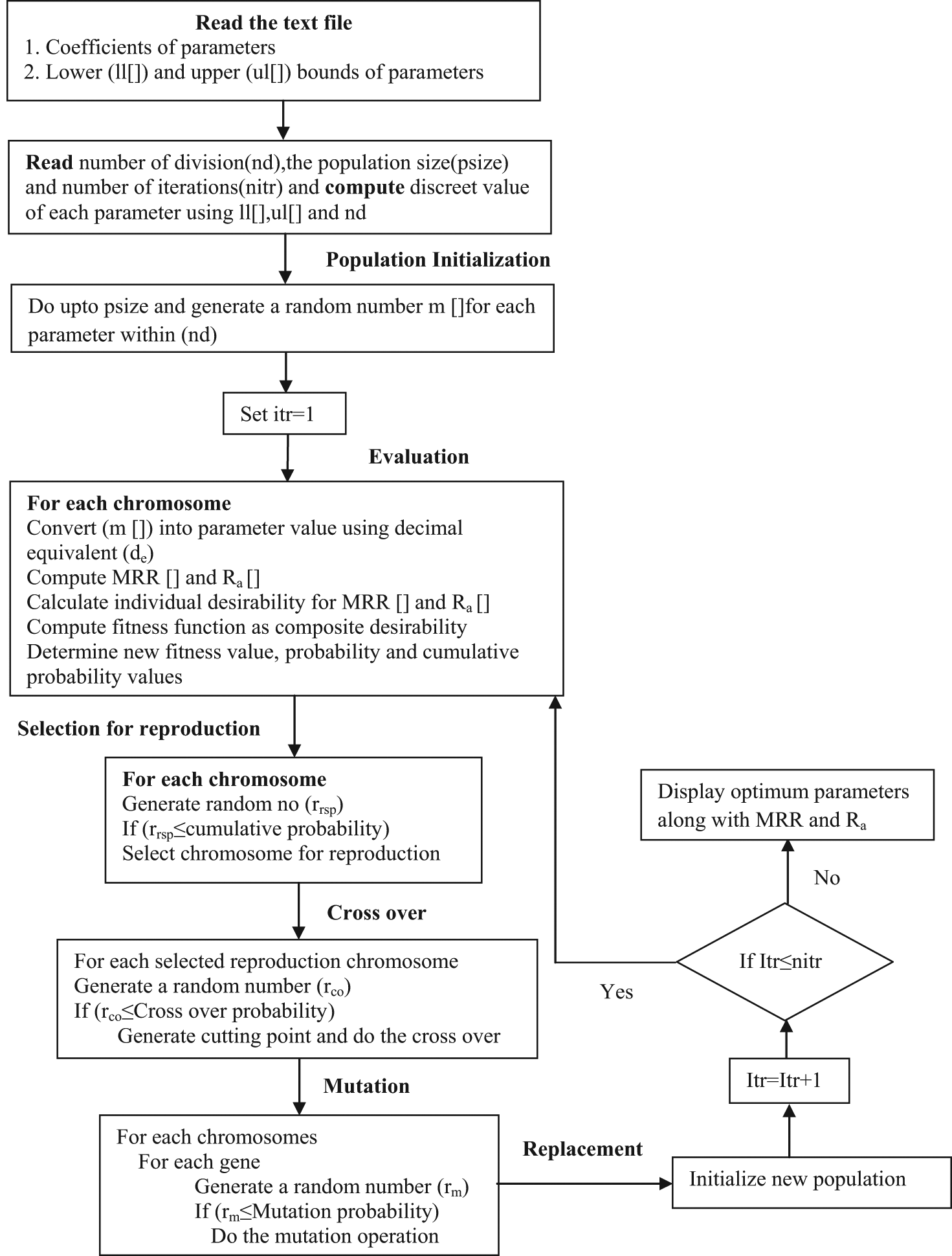
Scheme of GA-DF algorithm.
Initialization
In this work, population size and the number of generations considered are 20 and 500, respectively.

Variables (genes) are assumed in the range of 0–1000.
Parameters are initialized based on the expression
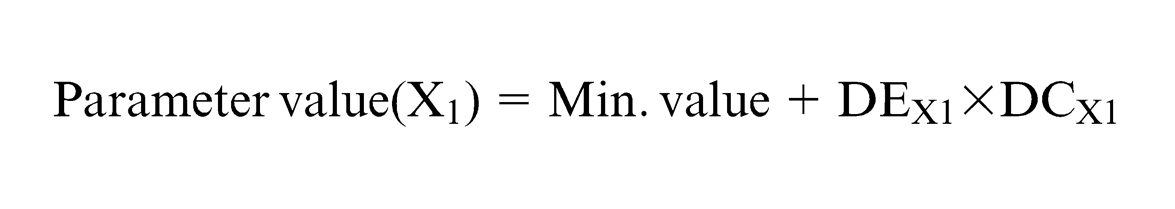
where
Five chromosomes are considered for the explanation. The evaluation for MRR and Ra and its desirability values are given in Table 4.
Initialization and evaluation.
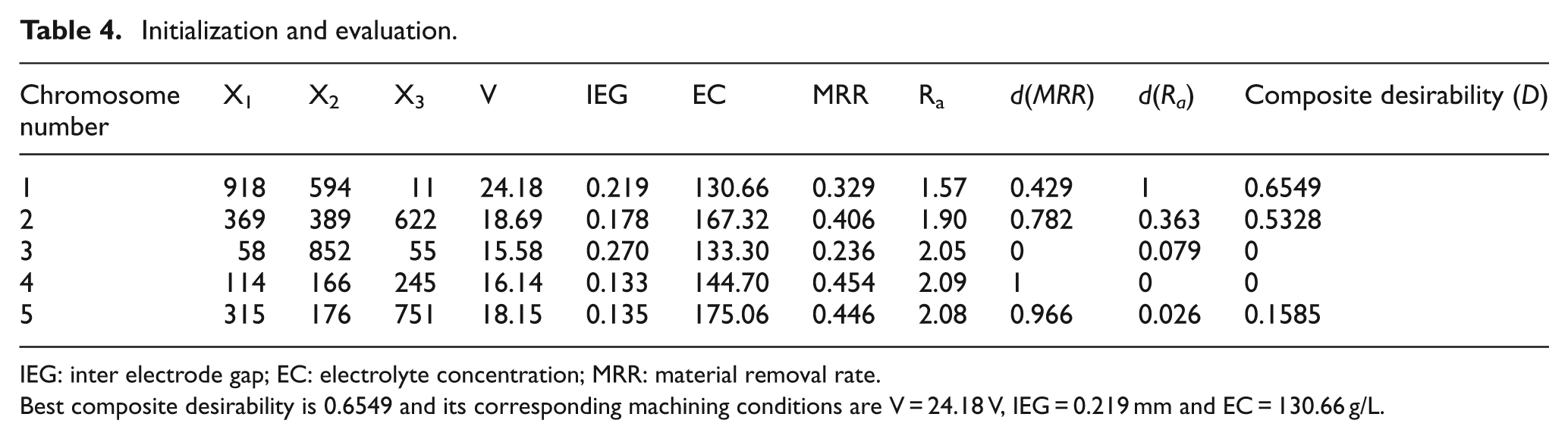
IEG: inter electrode gap; EC: electrolyte concentration; MRR: material removal rate.
Best composite desirability is 0.6549 and its corresponding machining conditions are V = 24.18 V, IEG = 0.219 mm and EC = 130.66 g/L.
Selection for reproduction
The chromosomes are selected according to roulette wheel selection method. The modified fitness value for each chromosome is calculated by the expression
The probability of chromosomes is determined using the following expression
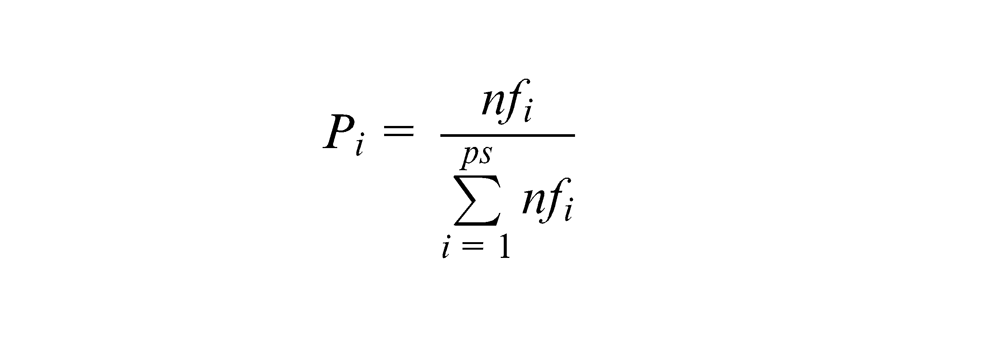
where
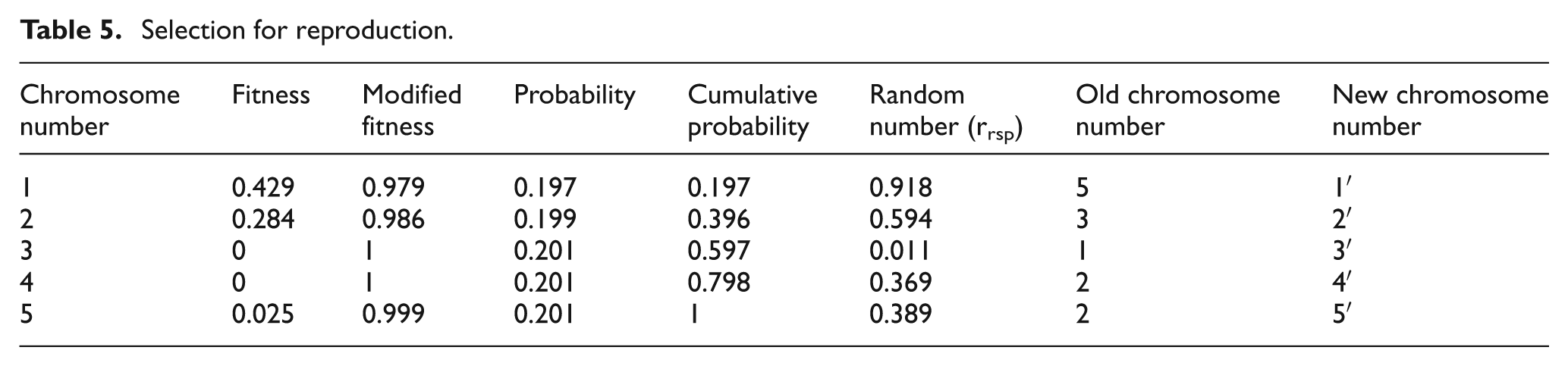
Probability and cumulative probability are listed in Table 5. Random number (rrsp) is generated for each chromosome. Chromosomes are selected corresponding to its cumulative probability value that is the next higher value than rrsp and renamed as chromosome number 1′, 2′ and so on. Table 5 represents the selected chromosomes for reproduction.
Selection for reproduction.
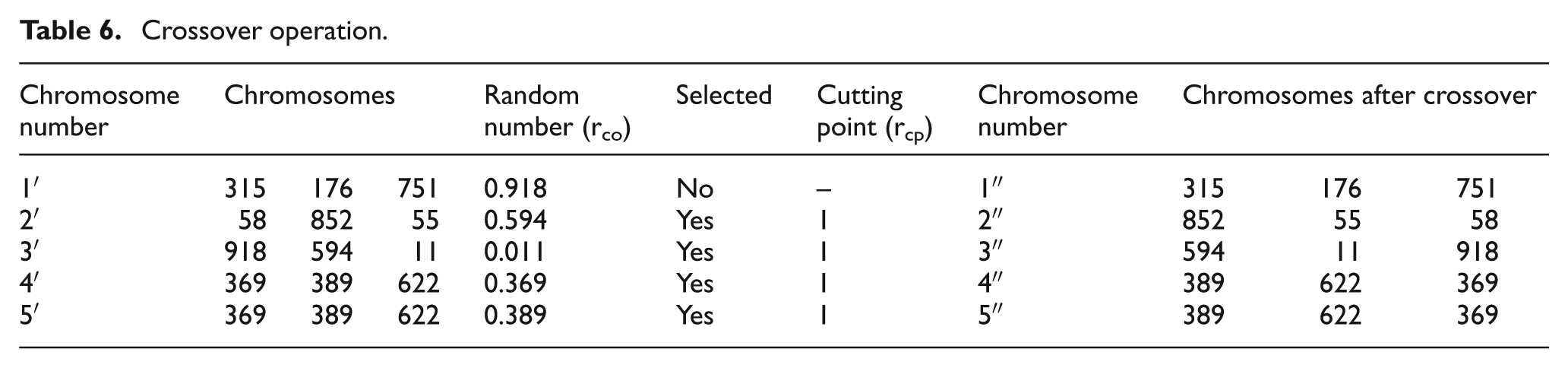
Crossover
A crossover probability is assumed as 0.62 and a random number (rco) is generated for each chromosome. The chromosome is selected for crossover operation, only if rco is less than or equal to crossover probability. Single-point crossover is considered in this work.
A random number (rcp), as cutting point, is generated within in the number N < number of parameters considered. So either 1 or 2 is generated for each chromosome for crossover. The genes after and before cutting point (rcp) are interchanged and presented in Table 6. Those chromosomes not selected for the crossover will be retained as such after crossover.
Crossover operation.
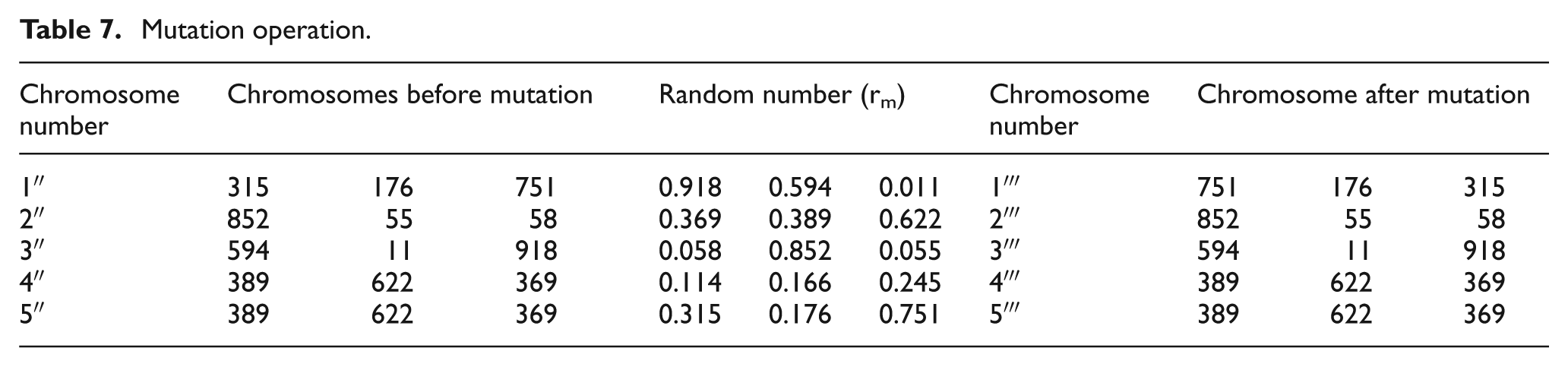
Mutation
A value of 0.05 is assumed as mutation probability and a random number (rm) is generated for each gene of all chromosomes. If rm is less than or equal to mutation probability, then the corresponding gene is mutated with neighbor gene and it is shown in detail in Table 7.
Mutation operation.
The chromosomes after mutation are assumed as new population. This new population is evaluated to find the best fitness value and is repeated till 500 iterations.
Best composite desirability for the simultaneous optimization of MRR and Ra in the aqueous NaCl and K2Cr2O7 environment is 0.98 (Figure 13). Optimum machining conditions are V = 25 V, IEG = 0.192 mm, EC = 184.3 g/L, MRR = 0.489 g/min and Ra = 1.65 µm.
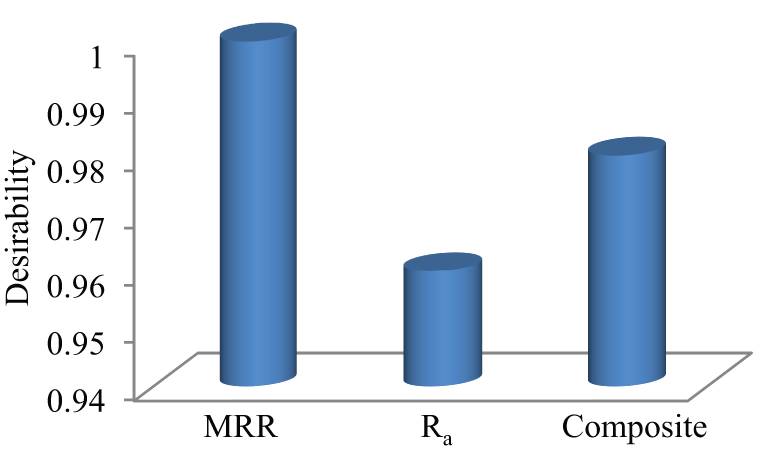
Desirability results of GA-DF optimizer.
Confirmatory experiment
In order to test the validity of the above machining conditions, confirmatory experiment was conducted and its results are given in Table 8.
Comparison of predicted and experimental performances.

The percentage of error is very acceptable.
Conclusion
The experimental analysis highlights the ECM characteristics of 20MnCr5 alloy steel using aqueous NaCl solution and K2Cr2O7 mixed aqueous NaCl. RSM was used in this research work to prove its adequacy to analyze the ECM process. From the investigations, the following conclusions can be drawn. The process parameters, voltage (V), IEG and EC, significantly affect the MRR and Ra. Potassium dichromate presence in the aqueous NaCl proves to be very effective in improving the MRR of workpiece. Increase in MRR is noted from 1% to 14% due to the addition of 5 g of K2Cr2O7/L of aqueous NaCl solution. MRR is improved due to the oxidation effect of K2Cr2O7. Due to the affinity of steel toward oxygen, more oxygen elements in K2Cr2O7 are attracted into the electrochemical reaction. Due to this high oxygen availability in the electrolyte, more FeO is formed, which is further oxidized and washed away by the electrolyte jet. Mathematical models developed based on RSM approach for correlating process parameters with MRR and Ra are tested through the ANOVA and proved adequate. The GA-DF optimizer was used for multi-response optimization. Optimum machining conditions, in the K2Cr2O7 mixed aqueous NaCl solution environment, are found out. Hence, it is concluded that the MRR is increased when K2Cr2O7 mixed aqueous NaCl is used as an electrolyte and slight increase in Ra is also observed.
Footnotes
Appendix 1
Acknowledgements
The authors wish to gratefully acknowledge the TEQIP scheme of World Bank for funding the ECM apparatus at the Government College of engineering, Salem-636011, Tamilnadu, India.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
