Abstract
Feasibility of partial remelting process in producing the feedstock for semi-solid processing of M2 high-speed steel (HSS) has been investigated. The effect of reheating parameters, that is, heating rate, holding temperature and holding time on final microstructure of M2 HSS, has been studied. For this purpose, a series of experiments based on Taguchi method was employed to determine the required reheating cycles. The experiments were carried out in a resistance furnace and in an argon-controlled atmosphere to avoid any oxidation and decarburization. During any reheating cycle, the temperature of surface and core of the samples were recorded and compared with the furnace temperature. Microstructural evolution of the samples was then studied and the formation of a globular microstructure in holding time has been evaluated. The particle shape factor and globule size in each stage of experiments have been derived and its relation to other parameters has been studied. Finally, appropriate reheating cycle and parameters has been determined and two-dimensional plots of parameters versus shape factor are introduced as a process window for reheating cycle derived.
Introduction
Semi-solid processing of metals is a novel field, which has been under investigation for not more than four decades. The first experiments that led to foundation of semi-solid processes were hot shear tests on Sn-15Pb alloy by Spencer et al. 1 in early 1970s. They sheared Sn-15Pb alloy during cooling down and observed a remarkable reduction in shear stress. The mixture of spheroidal solid particles in a liquid matrix is able to move freely against each other by means of translation and rotation movements and behaves like a liquid slurry with an effective low viscosity and flow at much lower shear stresses than would be expected for a dendritic material of the same solid fraction. Consequently, this kind of microstructure is particularly well-adopted to produce components with complex geometry, fewer forming steps, minimum forming loads and less die wear.
Generally, the semi-solid process is composed of two steps: preparation of the feedstock with equiaxed non-dendritic microstructure and forming step. Until now, many processes have been investigated by researchers, 2 such as magneto-hydrodynamic (MHD) stirring, 3 stress-induced melt-activated (SIMA) process,4,5 spray forming 6 and liquidus/near liquidus casting. 7 Reheating and partial remelting of an as-cast material is another process low-temperature weight alloys 8 and especially on high-temperature alloys like steels. 9 Reheating the metallic alloy to solid-liquid temperature leads to remelting of grain boundaries and formation of spherical solid particles, which are more favorable energetically. For heating purpose, either resistance or inductive furnace can be used. Nevertheless, accurate temperature control and holding time of the samples in a constant temperature within a narrow tolerance are somehow very difficult or impossible with these systems. Moschini 10 reported the use of a classical furnace in the semi-solid process. Hirt et al. 11 mentioned that reheating using classical electric resistance furnace based on radiation/convection case is less expensive but requires a long time and a robust control structure. Induction heating requires less time and provides the possibility of flexible process control. Induction heating systems are mostly used in industrial environments because of its less required time. Most of materials were tested using both techniques. The main advantage of inductive systems is their fast heating rates, which reduce grain coarsening consequence. Bolouri et al. 12 and Shang et al. 13 used an electrical resistance furnace in reheating the Al7075 and Al6061 alloys. Induction heating systems were used to reheat aluminum–silicon casting alloys, 14 7075 aluminium alloy 15 and steels such as 100Cr6 and X210CrWl2. 16 In order to achieve a successful semi-solid process, a fine microstructure of spherical solid particles suspended in a liquid matrix is required. Therefore, the parameters of reheating process like heating rate, holding temperature and holding time have to be completely investigated and understood.
In this research, feasibility of partial remelting process in producing feedstock for semi-solid processing of M2 high-speed steel (HSS) in as-received condition has been studied. The final goal of this research is to find a process window for reheating cycle parameters to attain a fine globular microstructure. It is clear that such a process window is a valuable tool in laboratory and industrial scale as it is a starting point in any semi-solid process.
Experimental procedure
The BÖHLER Uddeholm GmbH M2 tool steel in as-received (rolled and annealed) condition without any modification or refinements was used in the present research. Table 1 shows the chemical composition of the used material for experimental evaluation. The semi-solid temperature range of this steel was between 1228.22 °C and 1453.1 °C, which is calculated by thermal analysis using STA 409 PC Netzsch DSC-DTA analyzer (Figure 1). From this figure, it is seen that during heating with the rate of 432.9 J/g, melting starts at 1228 °C, while liquidus temperature was estimated at 1353.1 °C. The red color curve shows liquid fraction phase versus temperature. For example, at 1349 °C, the liquid fraction is 0.5. The samples were cut from the rods of 25 mm diameter and were then machined into the desired size of ϕ24 × 12 mm. A small blind hole was drilled along the axis of each sample allowing the use of a thermocouple in it. This was intended for close control and measurement of samples’ inside temperature.
Chemical composition %X of experimented M2 steel.
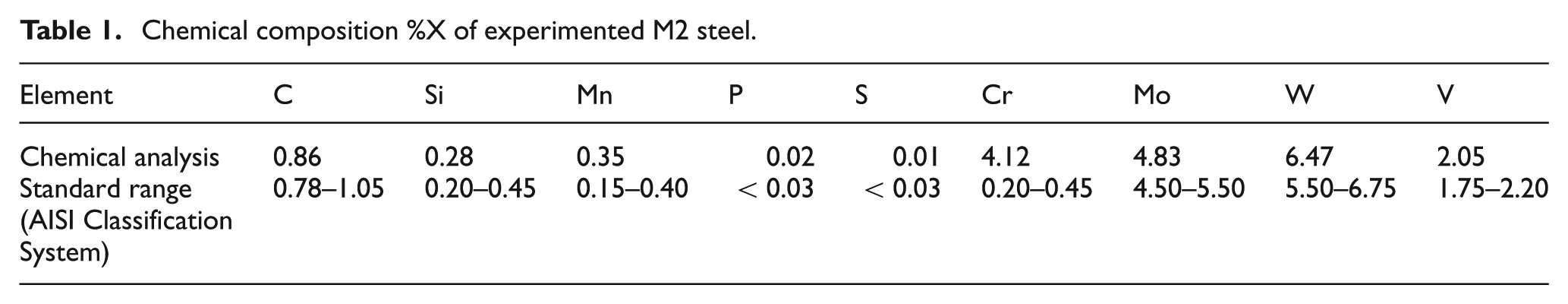
DSC-DTA analysis of M2 samples.
In order to investigate the influence of reheating parameters on the microstructure, four different values for each parameter were chosen according to Table 2. Since the number of the parameters influencing the final morphology of the samples was high (these parameters include cooling slope angle, ram velocity, temperature and holding time), the final number of the experiments required would be n 3 , in which n is the number of the states of the parameters (four in our case). In order to reduce this amount, design of experiments based on Taguchi method was employed (design L16). Using the Taguchi L16 design, only 16 tests were necessary to investigate the aforementioned parameters. Two extra tests were also done as verifiers of the final results of Taguchi analysis.
Heating cycle parameters and their values.

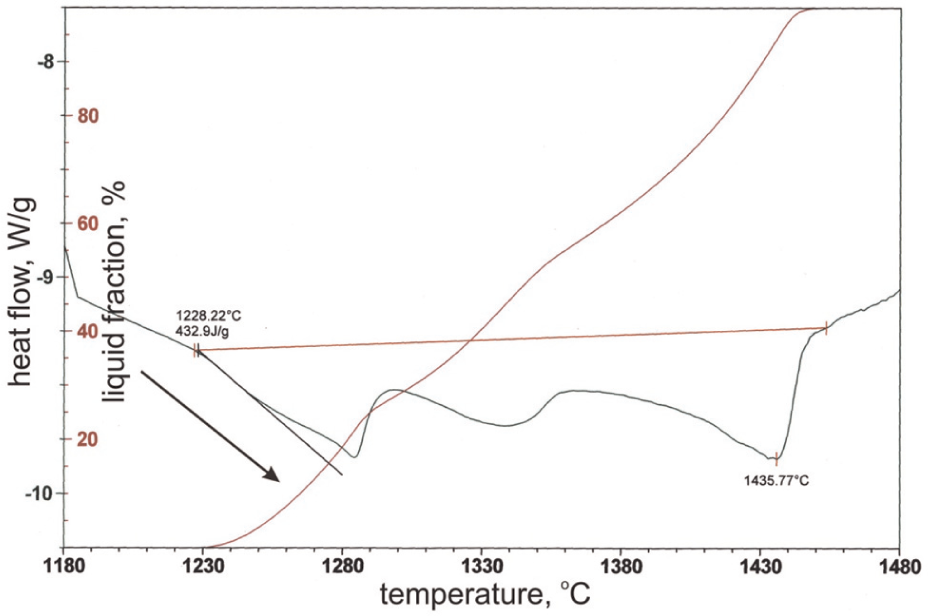
In this research, the tests were started by placing a prepared sample in a programmable logic controller (PLC)-controlled resistance furnace and studying one of the parameters. Heated argon gas atmosphere was flushed into the furnace in order to avoid oxidation. Temperatures are measured and controlled by means of three platinum–20% rhodium (type S) calibrated thermocouples at the surface and inside of samples and in order to compare them with the temperature of the furnace (Figure 2).
Temperature distribution of a M2 sample in a typical reheating cycle.
The samples were heated up to 700 °C with the rate of 28 °C/min and remain at that temperature for 10 min and the involved parameter study is investigated. After the completion of each cycle, the sample was quenched immediately in water and the samples were then cut and prepared for metallography. The samples were polished and etched in Nital (2%) solution. Microstructural images of the samples were taken from different points using an optical microscope and the scanning electron microscope (SEM; VEGA-TESCAN). The commercial image analysis software (Clemex Vision PE) was used to analyze the morphology and the shape factor, and globule size for each sample is calculated. An X-ray diffraction analysis was performed using Bruker D8-Advance X-ray Instrument according to BS-En 13925 standard.
The results of 16 different tests for each alloy were entered into Taguchi results section and were analyzed using Minitab software. Therefore, the results for remaining experiments were calculated. For the verification of the results, the obtained shape factors and mean globule size of the verifier specimens were compared with the calculated result. The effect of each parameter individually on the final microstructure of the samples and also the percentage of their effects were then calculated and plotted.
Results and discussion
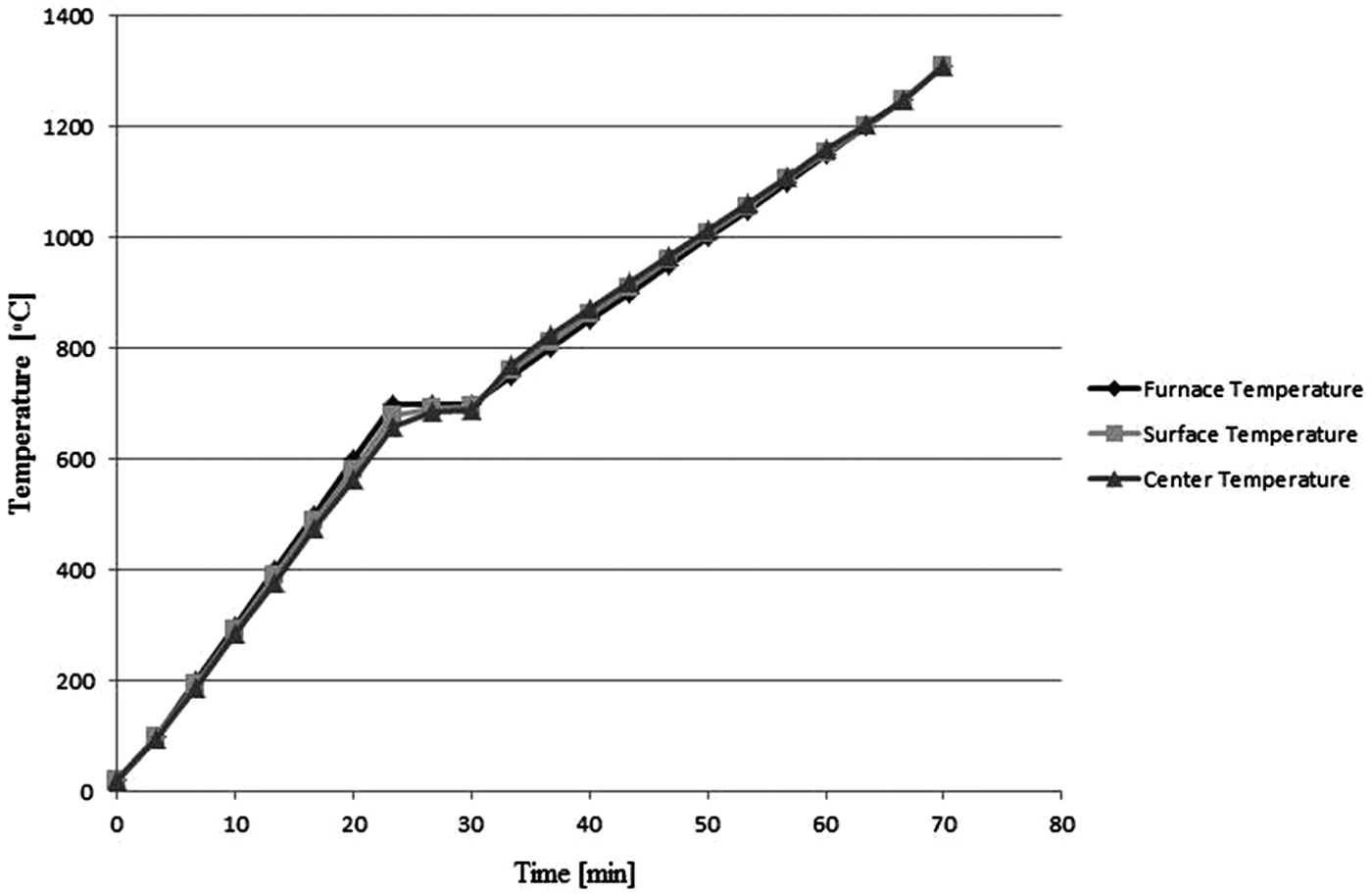
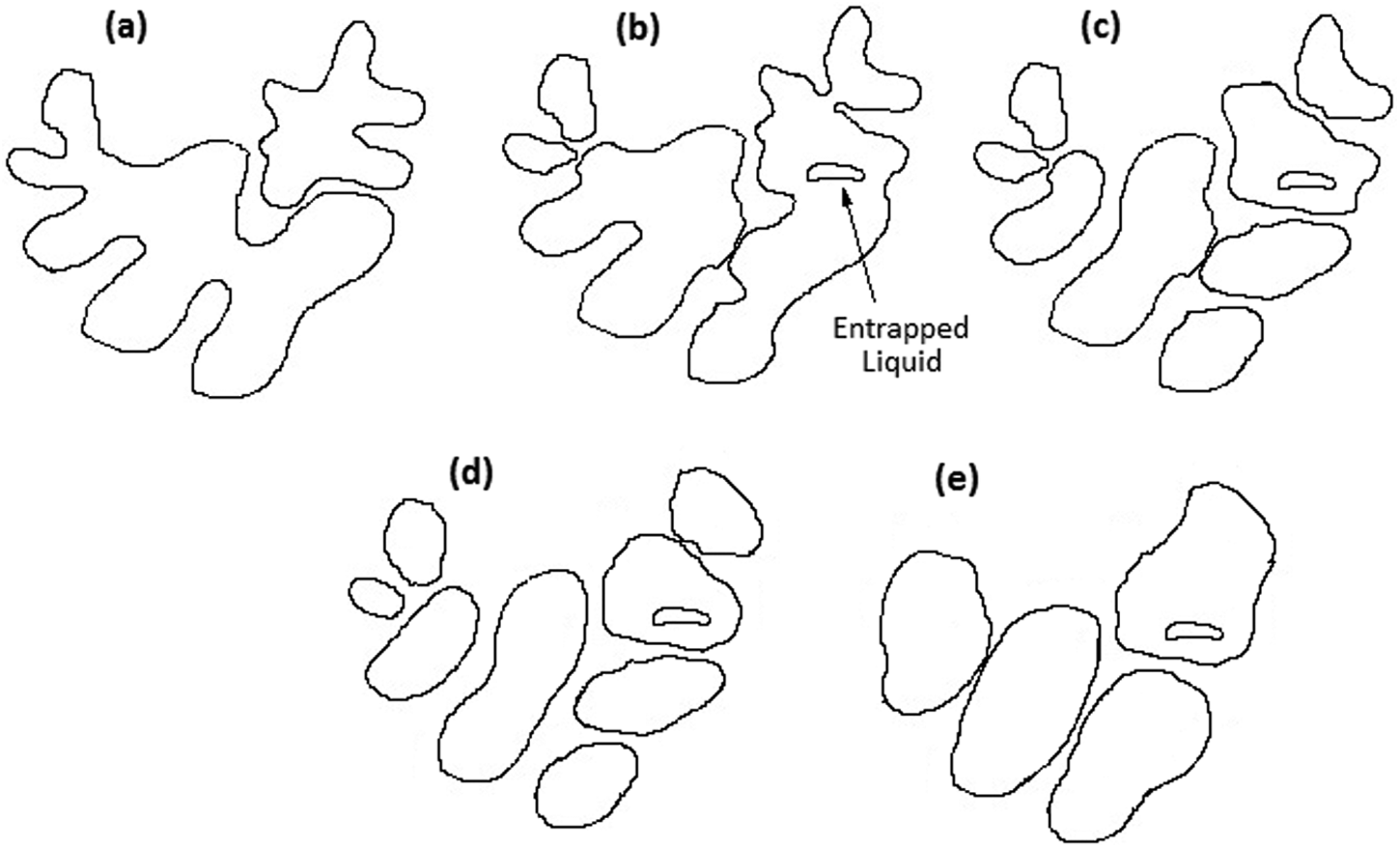
Figure 3 shows the microstructure of M2 HSS in rolled annealed condition and after reheating cycles. The fragmented microstructures of Figure 3(b) and (c) show that holding rolled annealed samples in semi-solid temperature range results in a globular microstructure, which is favorable for semi-solid processes. The formation of such a globular microstructure from originally dendritic microstructure is related to several mechanisms. Figure 4 illustrates the generation of a globular microstructure via isothermal holding of a conventionally dendritic structure. In the initial stages of holding time, the dominant mechanism is the dissolution of last solidified phase of low melting temperature, which formed as a result of segregation during solidification. In M2 HSS, these are metal carbides (MC) and M6C carbide-rich regions, which have been confirmed by SEM and X-ray analysis (Figure 5). Holding the samples above the solidus temperature results in penetration of the liquid phase into the original grain boundary, causing fragmentation of that grain, as shown in Figure 4(b). To be more specific, wetting of the grain boundaries originally starts at the root of dendrite arms. The fragmented dendritic arms are then evolved into ellipsoidal and subsequently spheroidal grains to form an energically favorable boundary, as shown in Figure 4(c) and (d). Coarsening follows after this period, as shown in Figure 4(e). Several coarsening mechanisms are in operation during isothermal holding period. One is the coalescence of grains, in which two adjacent grains join together and form a bigger grain; another mechanism is the Ostwald ripening in which the larger grains grow while the small grains remelt.

Optical micrograph of M2 high-speed steel (HSS): (a) rolled annealed, (b) holding at 1320 °C and (c) holding at 1365 °C.
Schematic of microstructure evolution during holding in semi-solid state.
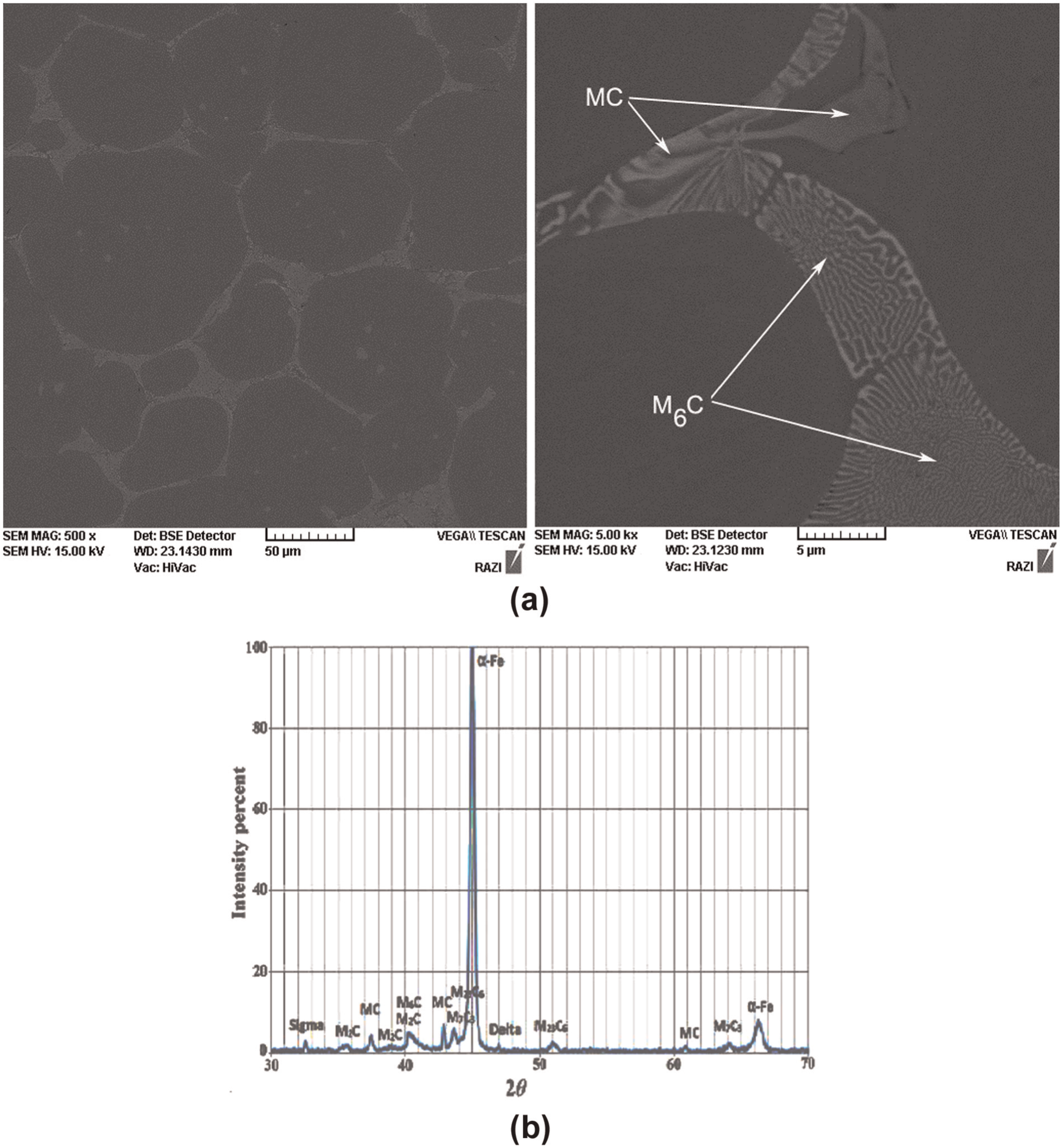
SEM micrograph of M2 high-speed steel (HSS) after holding at 1320 °C.
Entrapped liquid is another phenomenon observed after holding in semi-solid state, which is a result of coalescence mechanism, as shown in Figure 4(b). This causes the actual and active liquid fraction of M2 samples to be less than the calculated numbers and it must be taken into account in practice. Figure 6 shows a typical microstructure of the M2 samples after holding at 1350 °C with small entrapped liquids. It is concluded that rolling process produces fine broken carbides, which enhances the formation of small entrapped liquid in semi-solid microstructure of the samples. Liquid phase also forms at high angle grain boundaries if the samples are held at the semi-solid state. Coarsening of dendrite arms occurs by coalescence mechanism right after holding the samples at the semi-solid range. Coalescence mechanism occurs quickly if dendrite arms of the same grain have preferred direction.
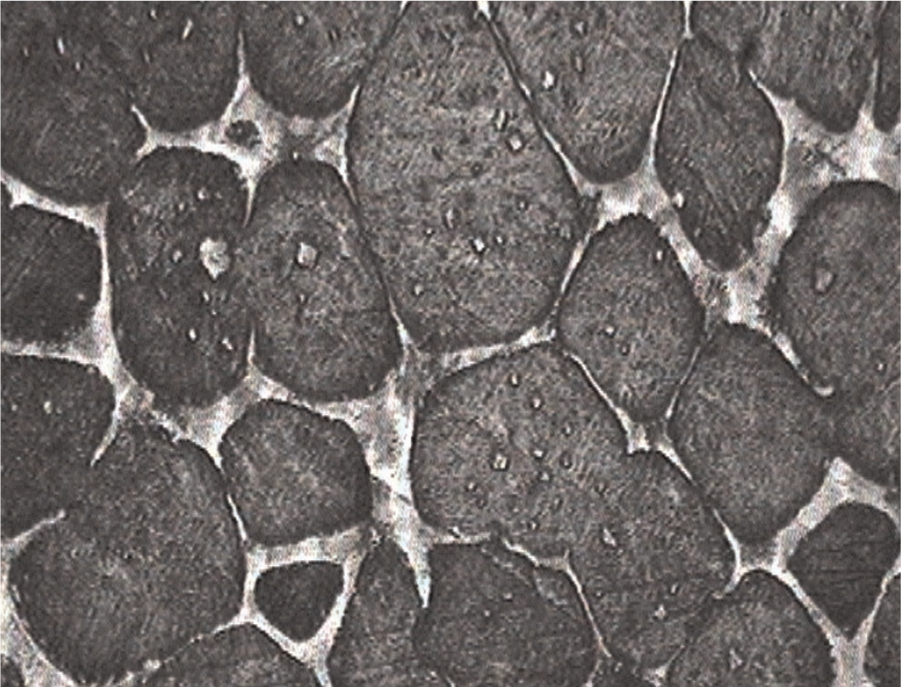
Optical micrograph of M2 high-speed steel (HSS) after holding at 1350 °C, indicative of the small entrapped liquids.
Globule size
In practice, the samples with the fine globules are preferred because of their better flow characteristics and higher mechanical properties. Hence, a measure of globule size can be a comparison gauge between a desirable and undesirable microstructure. Image analysis software was employed to measure the size of the globules in the prepared samples. Distribution of the globule size within the samples along with the mean value of the globule size for different holding temperatures is plotted in Figure 7. As shown in this plots, increasing of the temperature from 1320 °C to 1365 °C causes the growth of average globule size from 72.5 to 85 µm for M2 samples. Two important factors can be derived from these plots: general globule size of the microstructure of the samples, which is in the range of 72.5–85 µm, and size distribution of the globules, which is almost in a narrow range (smaller standard deviation). The reason may be related to the rate of carbides density distributed in the microstructure of samples. It is obvious that M2 samples possess more carbide solutions in their microstructure, hence obtaining finer and uniform globules, which is more favorable in semi-solid processes. After all, as was mentioned earlier, it is not only the globule size, which determines a desirable microstructure for semi-solid processing.
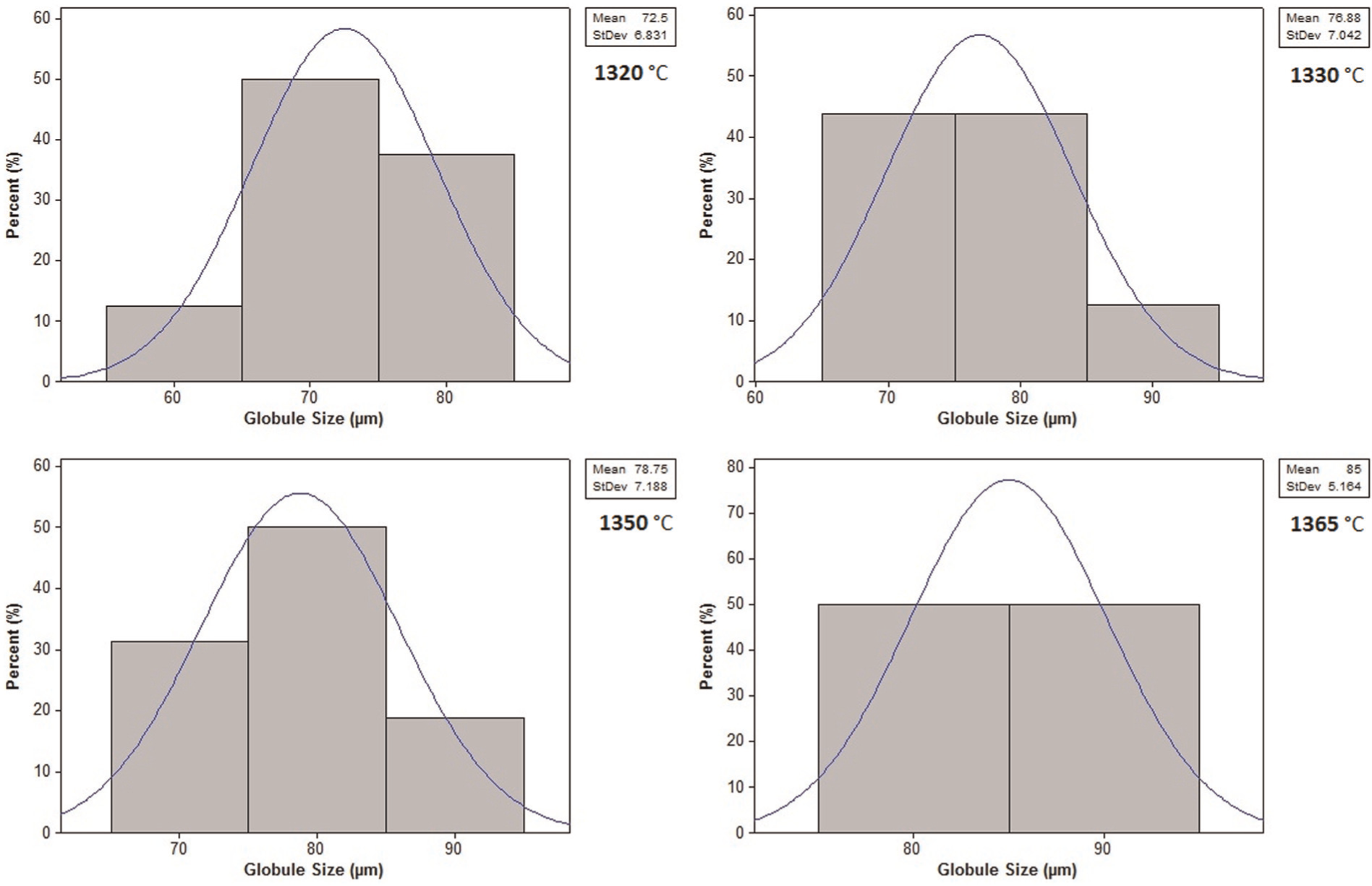
Distribution of globule size for different holding temperatures for M2 samples.
Shape factor
Another interrelated variable for investigation of morphology is the shape factor, which is discussed in this section. Ideal spherical solid particles suspended in a liquid matrix are the key for a successful semi-solid process. A measure of globularity of the structure of samples can be achieved by determining their shape factors. The following equation is used

where
As shown in Figure 8, increase of temperature up to 1350 °C resulted in a better globular microstructure, while after that the quality of globules decreased rapidly. The reason is that the increase of temperature in the first stages resulted in remelting of eutectic phase of initially low-temperature phase and thus refinement of globules. While after that point, ripening and grows of globules increased the shape factor. Holding time also had a different influence on the microstructure of M2 samples. Holding the samples in a constant semi-solid temperature contributed the formation of a finer globular microstructure. Heating rate also had a two-sided effect on the final microstructure of the samples. Increasing the heating rate up to 45 °C/min decreased the shape factor of the samples, while after that point, the shape factor increased. The reason again is related to the time in which the samples are kept under heating. In order to become the globularity of the samples, less time is not acceptable. Too much time also is not preferable because of ripening and growth phenomena. As a result, there is an optimum time of soaking the samples under heat.
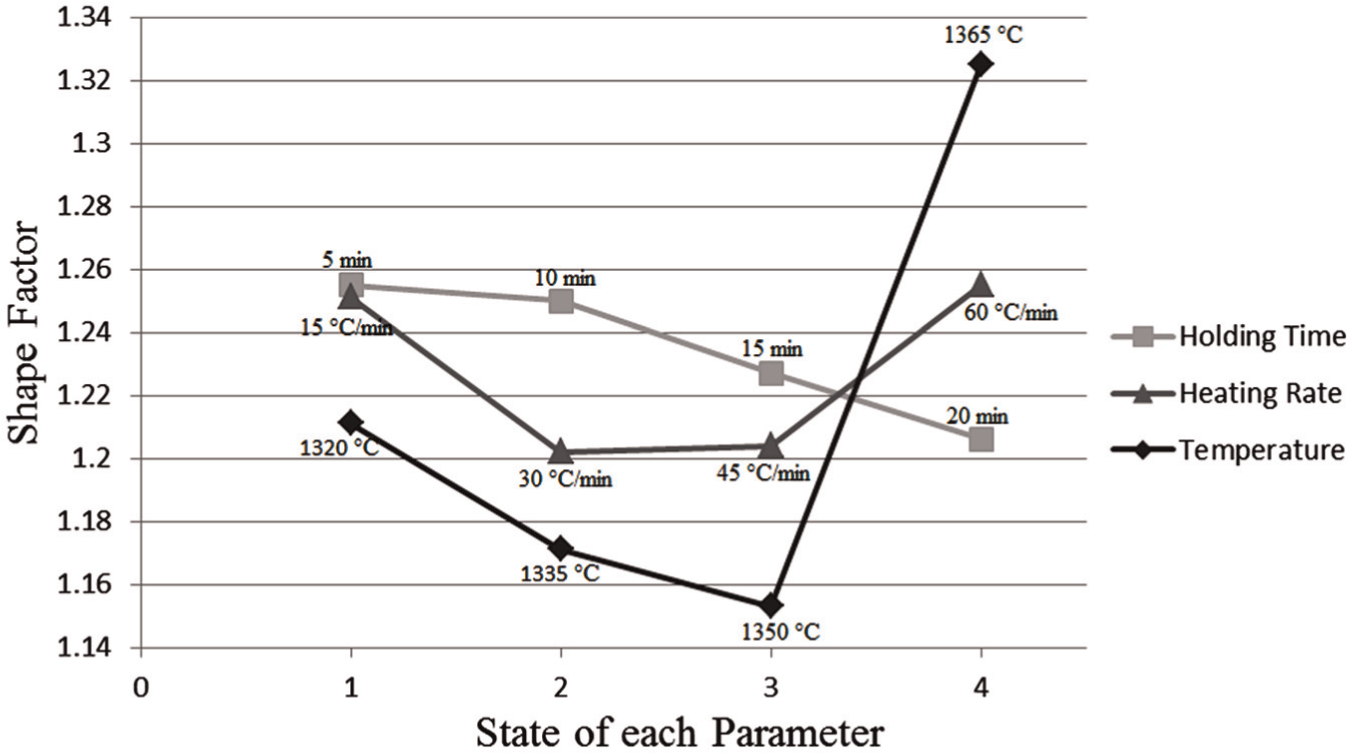
Mean influence of each parameter on the shape factor for M2 samples.
For better view of parameters effects on the shape factor of the samples, two-dimensional (2D) contour of the holding time and temperature has been plotted in Figure 9. The optimum range of parameters for a globular structure is the areas with lighter colors, which is located in the middle of the plot. The plots with consideration of Figure 7 reveal that for M2 HSS, an appropriate processing temperature in semi-solid state would be about 1350 °C, which will result in an average globule size of about 78 µm.
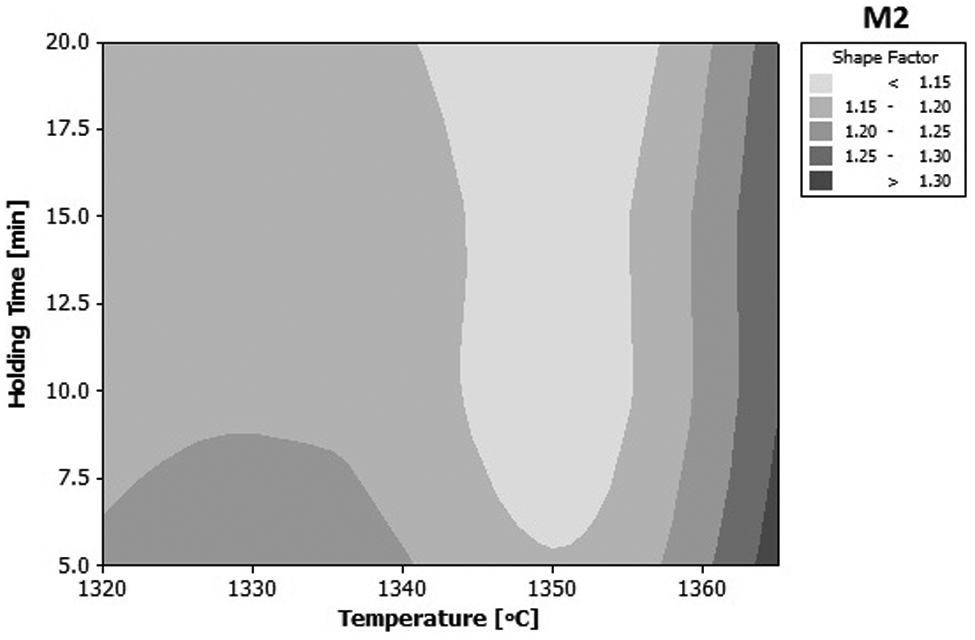
Contour plot of shape factor versus temperature and holding time for M2 tool steels (mean value of heating rate is considered).
Importance of parameters effect on the final microstructure of the samples is plotted in Figure 10. The percentage for each parameter has been calculated from the data in Figure 9, by calculating the maximum deviation of shape factor for each parameter and then plotting all three values on a percentage scale. It can be observed that temperature with 63% effectiveness has the greatest effect on the final microstructure of the samples and has to be carefully chosen and controlled.
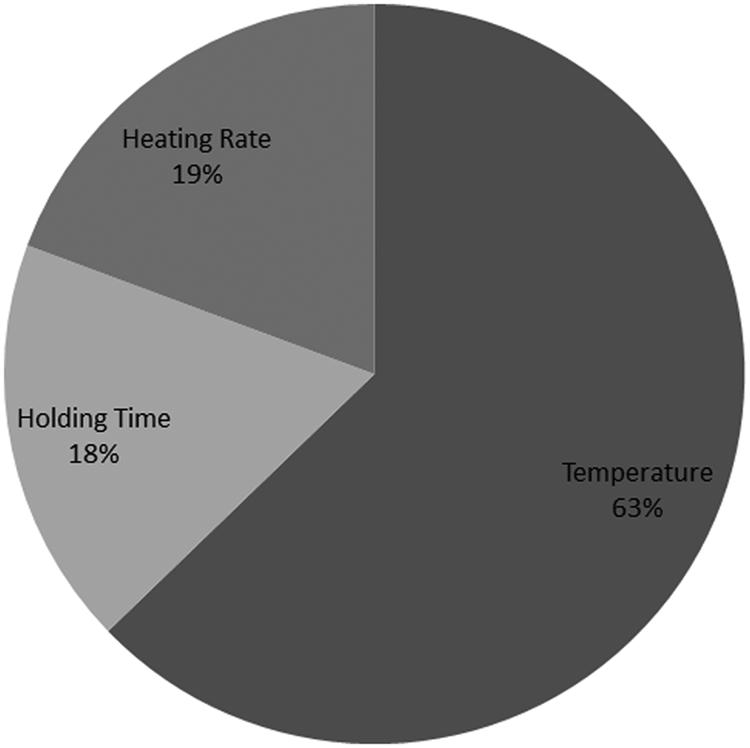
Importance of parameter analysis results for different reheating cycles of M2 samples.
Conclusion
The following conclusions are drawn as follows:
Metallography images showed that the M2 tool steel is suitable for semi-solid processing by partial remelting without any preprocessing operations for feedstock.
Globularization phenomena started in the first stages of holding cycles, when the low melting point MC and M6C carbide region started to melt.
Holding temperature in semi-solid state had greatest effect on the globularity and globule size of the microstructure of samples. For M2 tool steel, increasing the temperature up to 1350 °C corresponding to almost 50% liquid fraction resulted in a better globular microstructure, while after that the quality of globules decreased rapidly.
The contour plot of the shape factor shows that in the temperature above 1360 °C, the shape factor exceeds of 1.3 for the mean value of heating rate.
For a successful semi-solid process, a great care must be taken in choosing and controlling the temperature of the processed parts. Also, an optimum holding time is needed for the semi-solid billet become globular. Less times result in a weak globularity while higher times increase the shape factor of the microstructure by ripening phenomenon.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
