Abstract
This study explores the elongation of submerged arc-welded joints by using developed agglomerated fluxes. The response surface methodology technique was used to conduct the experiments. MnO, CaF2, NiO, MgO, and Fe-Cr were added to the fluxes comprising CaO, SiO2, and Al2O3 as the main constituents. The effect of the addition was studied by means of mechanical performances of the welds in terms of elongation. The elongation of weld metal increases with the increase in MnO, MgO, and Fe-Cr constituents, but CaF2 and NiO constituents did not reveal favorable sign unlike the other three constituents mentioned here. The recommended optimal conditions were verified by conducting confirmation experiments.
Introduction
The submerged arc welding (SAW) is a versatile process and is used for a wide range of applications that include the critical applications, for example, joining of pressure vessels, thick plates, ship hulls, and so on. In SAW, the arc is covered by a flux. The constituents, present in the flux, control arc stability, bead shape, and the mechanical and chemical properties of weld metal. 1 The SiO2 content, for example, in the synthetic fluxes improves the welding performance.
As is determined through metallographic examination, the presence of a metallic mist in the matrix of the flux and its amount get increased with the addition of silica contents. 2 Colvin 3 in his study propelled that the basic fluxes are more or less associated with the improved metallurgical properties of weld than the acidic fluxes. The study of commercial acidic fluxes by Bennett 4 has correlated the slag detachability and weld bead surface appearance to their chemical composition. Several other studies have revealed the effect of various flux constituents onto the transfer of elements to the weld metal. As a result, the mechanical and metallurgical properties of the weld metal get affected.5–20 This work studies the elongation of low-carbon steel weld joint, which is the most important property to be imparted to the weld metal.
Selection of flux constituents and their ranges
In the study under consideration, the fluxes have been designed on the basis of binary and ternary phase diagrams for different oxide and fluoride systems. 21 In these fluxes, varying amount of oxides and fluorides was added to study the impact of these constituents onto the elongation of the weld metal. After ascertaining the compositions and constituents of fluxes, they were prepared by agglomeration method using the potassium silicate as its binder.
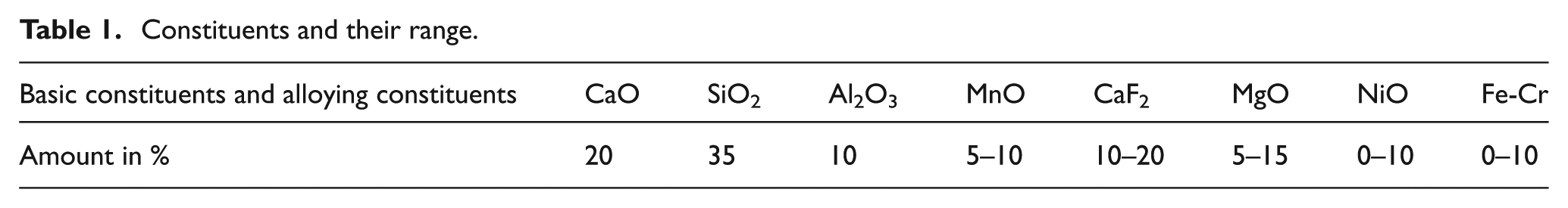
The selection of flux constituents is generally based on the composition of base plate and electrode employed to welding. CaO-Al2O3-SiO2–based flux systems have been selected for the study, as these are the most widely used fluxes at the commercial level. The details of basic constituents, alloying constituents, and their percentage are reported in Table 1.
Constituents and their range.
Response surface methodology technique
The response surface methodology (RSM) was used to design and point out the relationship between the response of interest and the variables (alloying constituents of flux) as well as to determine the conditions of these variables that were actually able to optimize this response. In this study, five constituents of flux taken as a process variable are used as levels that maximize the yield (Y) of a process as shown in equation (1)
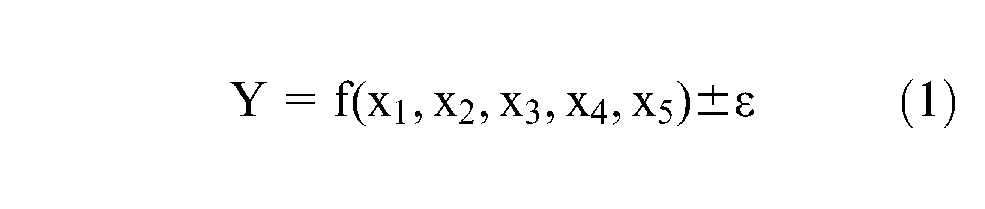
where Y is the response, f is the response function, ε represents the experimental error, and x1, x2, x3, x4, and x5 are the independent constituents in the developed agglomerated flux. The expected response (elongation) is defined by E(Y) = f(x1, x2, x3, x4, x5) = η, where η is called the response surface. To establish the relationship between response and the independent variable, the first-order model is employed and the above equation can be written as shown in equation (2)
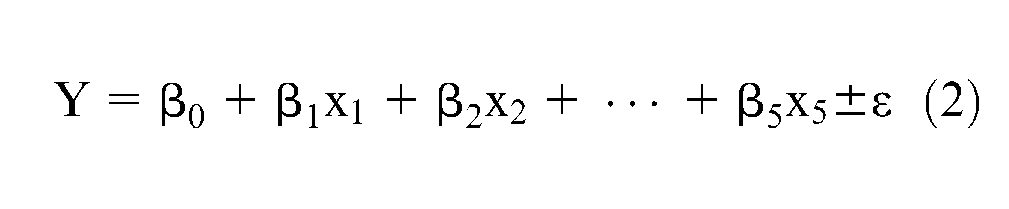
The behavior of the system can be described by the following second-order polynomial as shown in equation (3)
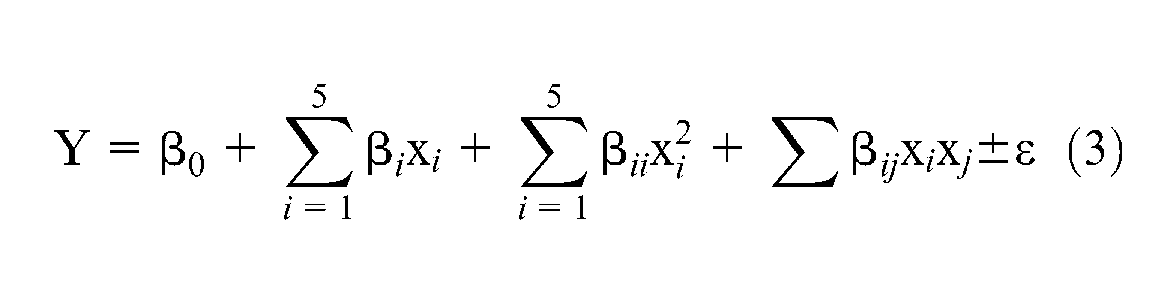
where Y is the response,
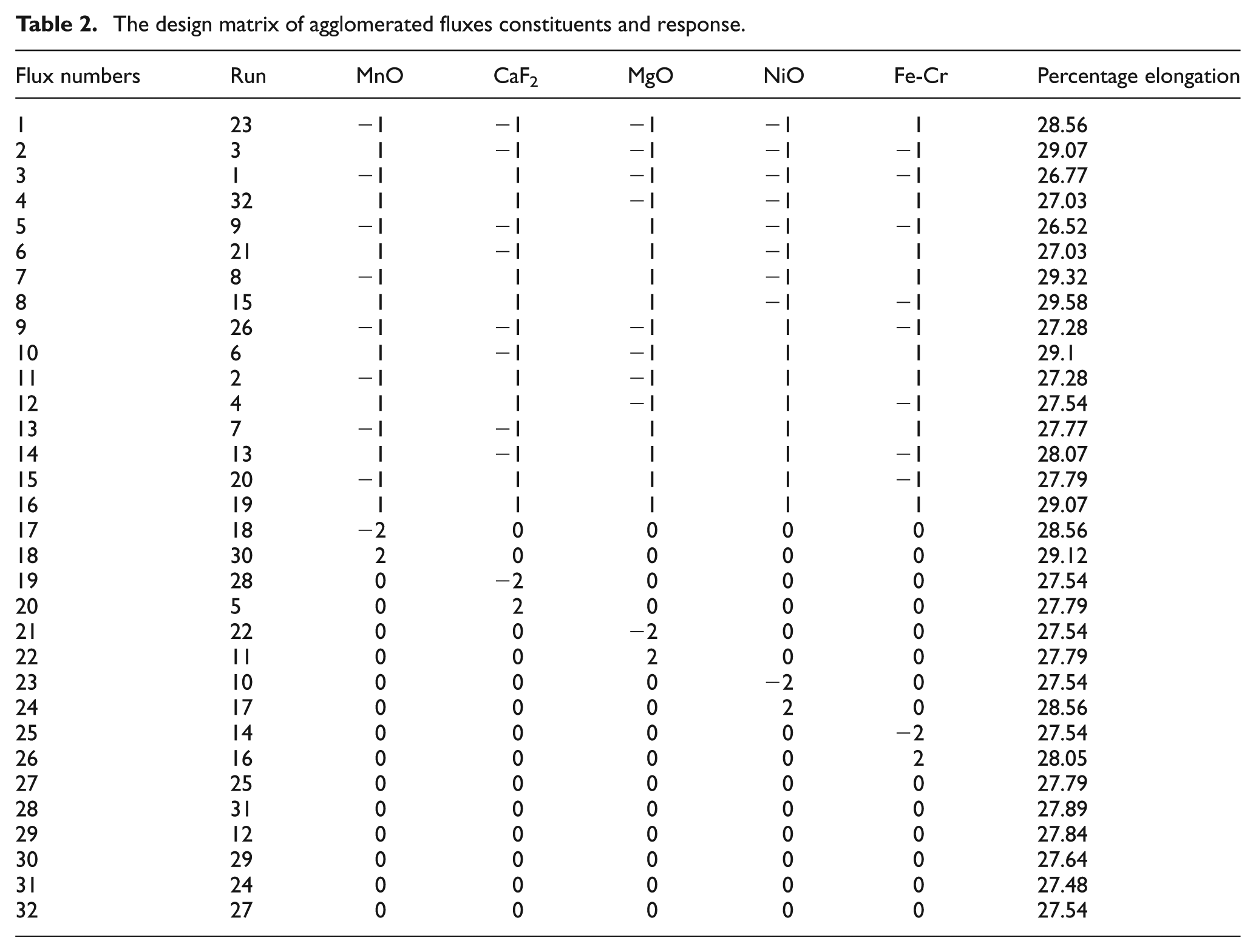
The design matrix of agglomerated fluxes constituents and response.
Flux preparations
The agglomeration method is used for flux preparation by taking small batches of (2 kg) weighed quantities of powdered chemicals, with binder, which is in turn thoroughly mixed in a ball miller. The prepared powdered flux is dried and baked in an electrically heated furnace for 3 h at 800 °C. The dried flux is allowed to cool down to room temperature before storing it in a moisture-free box.
Selection of process parameters of SAW
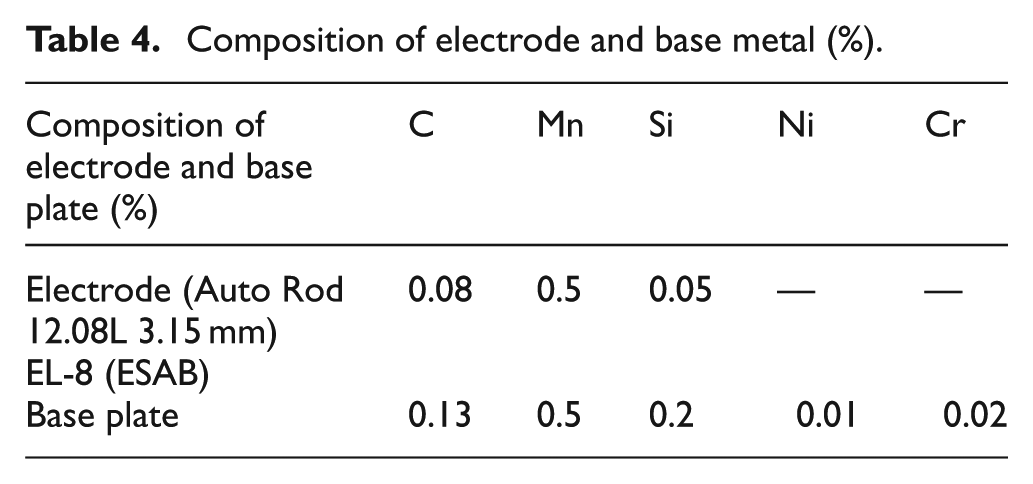
The process parameters of SAW were selected on the basis of good slag detachability, weld appearance, arc initiation, and arc stability. The higher and lower values of welding parameters may lead to poor welding properties. The welding parameters were chosen on the basis of above properties, as reported in Table 3. The composition of base metal and electrode is given in Table 4.
Process parameters.

DCEP: direct current electrode positive.
Composition of electrode and base metal (%).
Experimentation
The weld joints were made on low-carbon steel plates of 300 × 300 × 20 mm (V-groove at 45°) size by using 32 developed agglomerated fluxes by SAW machine, with fixed process parameters mentioned in Table 3. The specimens for measuring elongation were prepared from the welded joints as per the standard specification shown in Figure 1. The elongation of prepared samples was measured with the help of Tensometer (KIPL-PC 2000) testing machine (highest limit 18 kN), with a 0.8 mm/min uniform test speed. The specimens before and after testing are shown in Figure 2. The responses of elongations for each welded joint are given in Table 2.
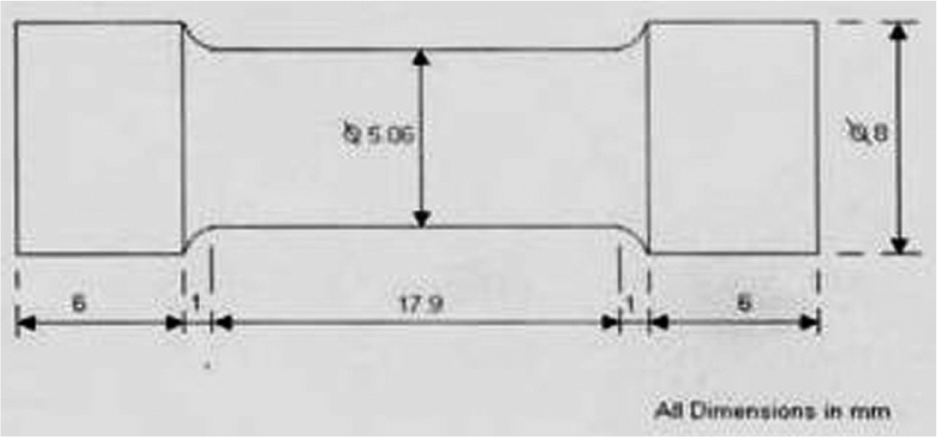
Specification of specimen.

Specimens (a) before and (b) after the elongation measurement.
Results and discussion (response surface modeling of percentage elongation)
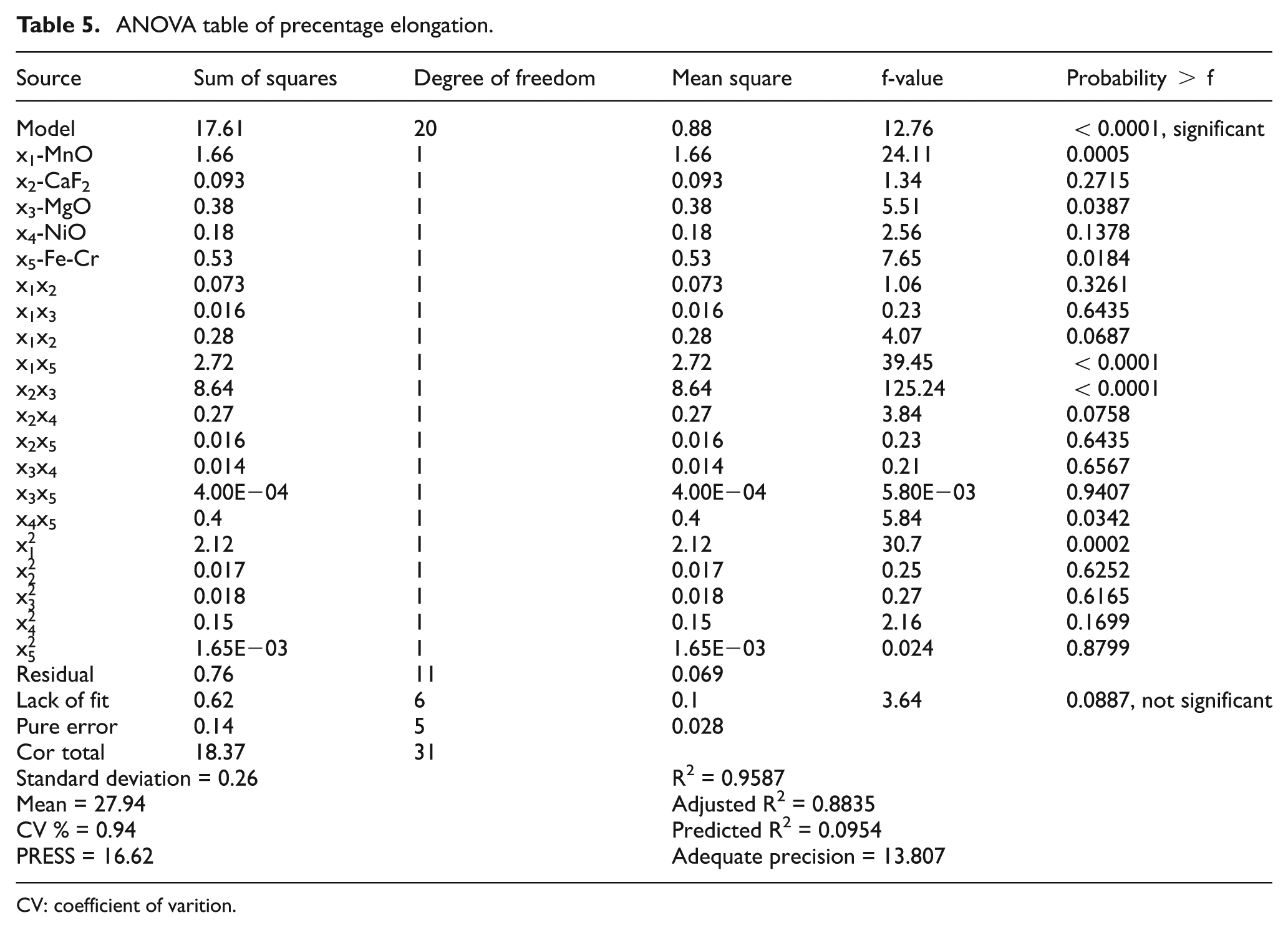
The analysis of variance (ANOVA) and pooled ANOVA have been reported in Tables 5 and 6. It revealed that the model was actually significant. The f-value of 25.18 in pooled ANOVA and the p-value<0.0001 demonstrated that this regression was statistically significant at 99% confidence level. The value of R2 was found to be 0.9115 and fulfilled the requirement of ANOVA. According to ANOVA, R2 must lie between zero and one, the larger values in this range being more desirable. 25 The values of probability > f for the model less than 0.05 (i.e. α = 0.05, or 95% confidence) indicate that the model terms were significant. The lack-of-fit term is not significant in the model as per the ANOVA condition. In this study, the factors MnO, MgO, and Fe-Cr, the combined effect of constituent MnO with constituent Fe-Cr, the combined effect of constituent CaF2 with constituent MgO, the combined effect of constituent NiO with constituent Fe-Cr, and second-order term of constituent MnO have significant effect altogether.
ANOVA table of precentage elongation.
CV: coefficient of varition.
Pooled ANOVA table of percentage elongation.
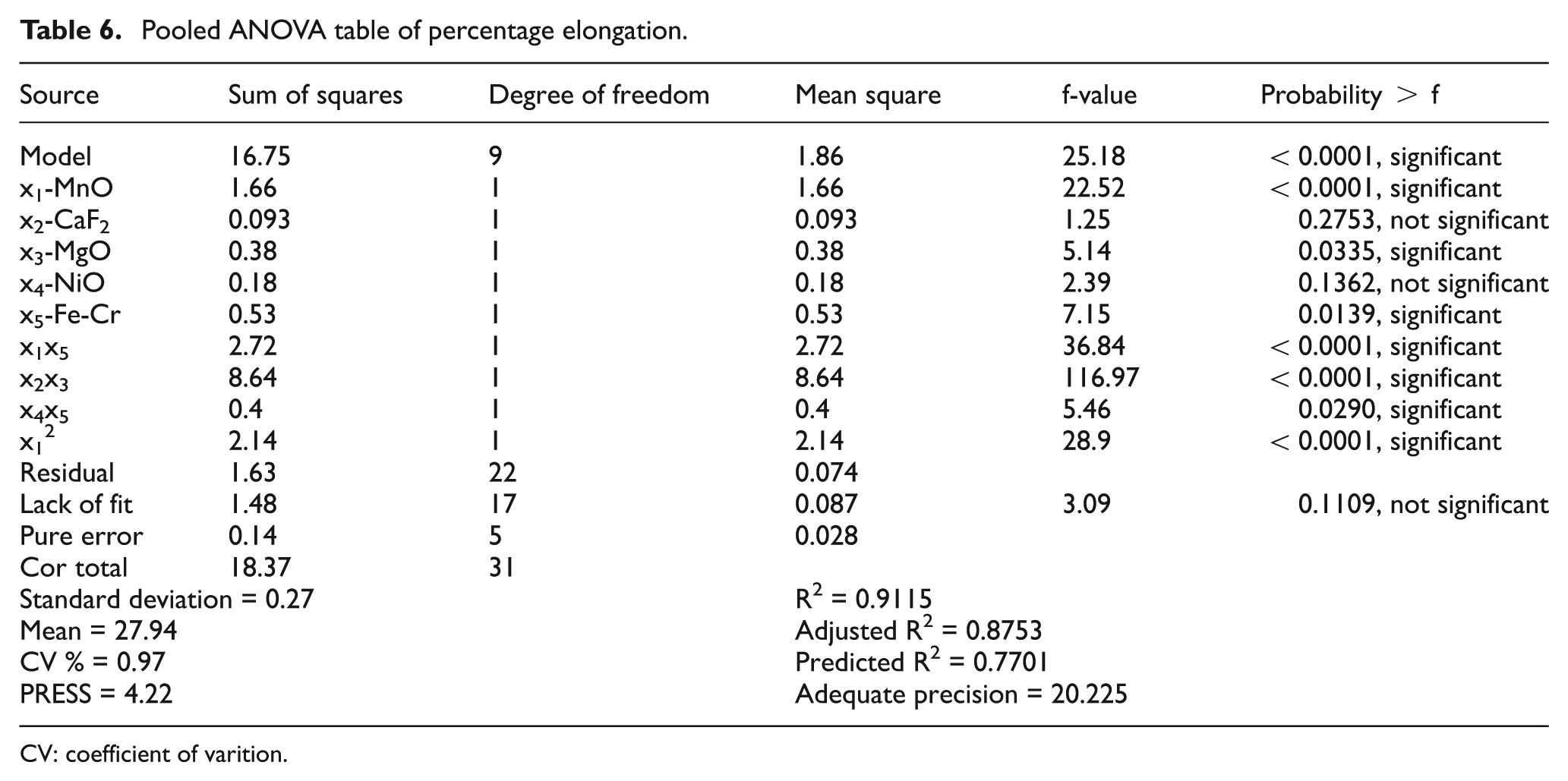
CV: coefficient of varition.
The mathematical model that describes the final response equation for elongation is given in equation (4)
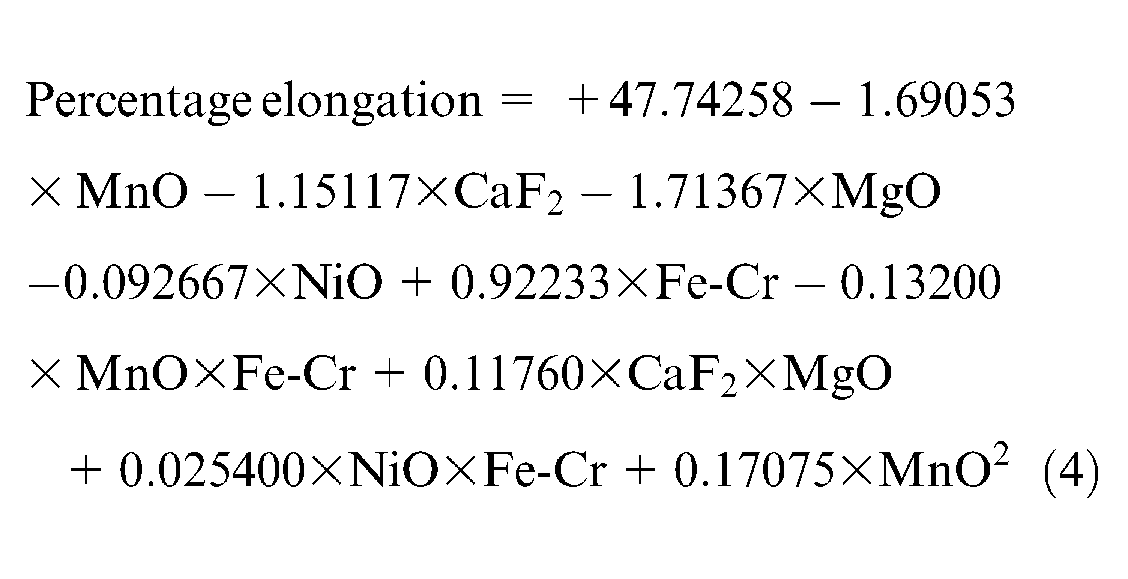
Figure 3 describes the normal probability plot of the residuals for percentage elongation. Figure 4 shows the values of percentage elongation predicted by the model and actual values obtained by the experiment. It is observed that the distribution of points around the line for this response is fit to linear model.
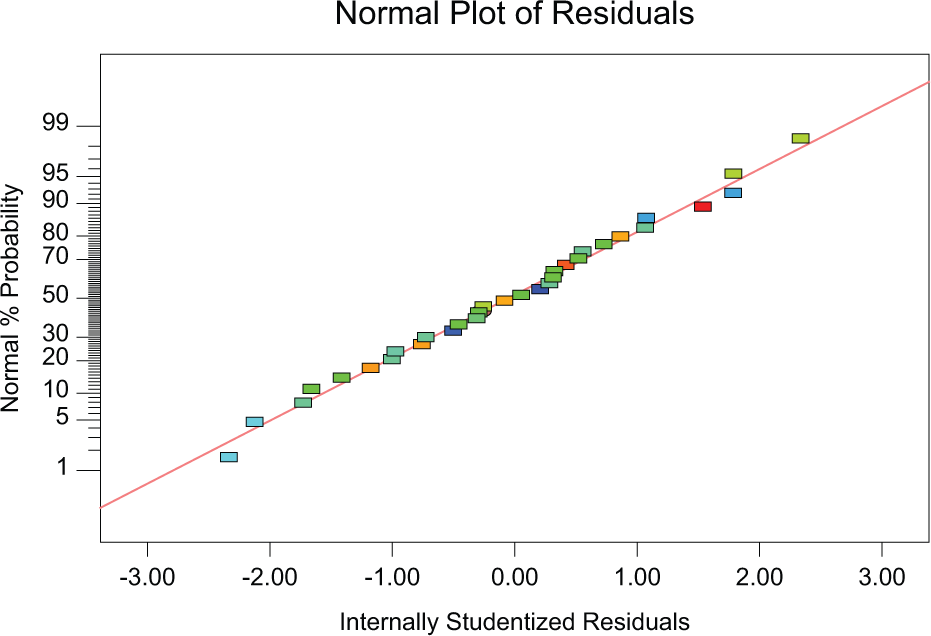
Normal probability plot of the residuals for percentage elongation.
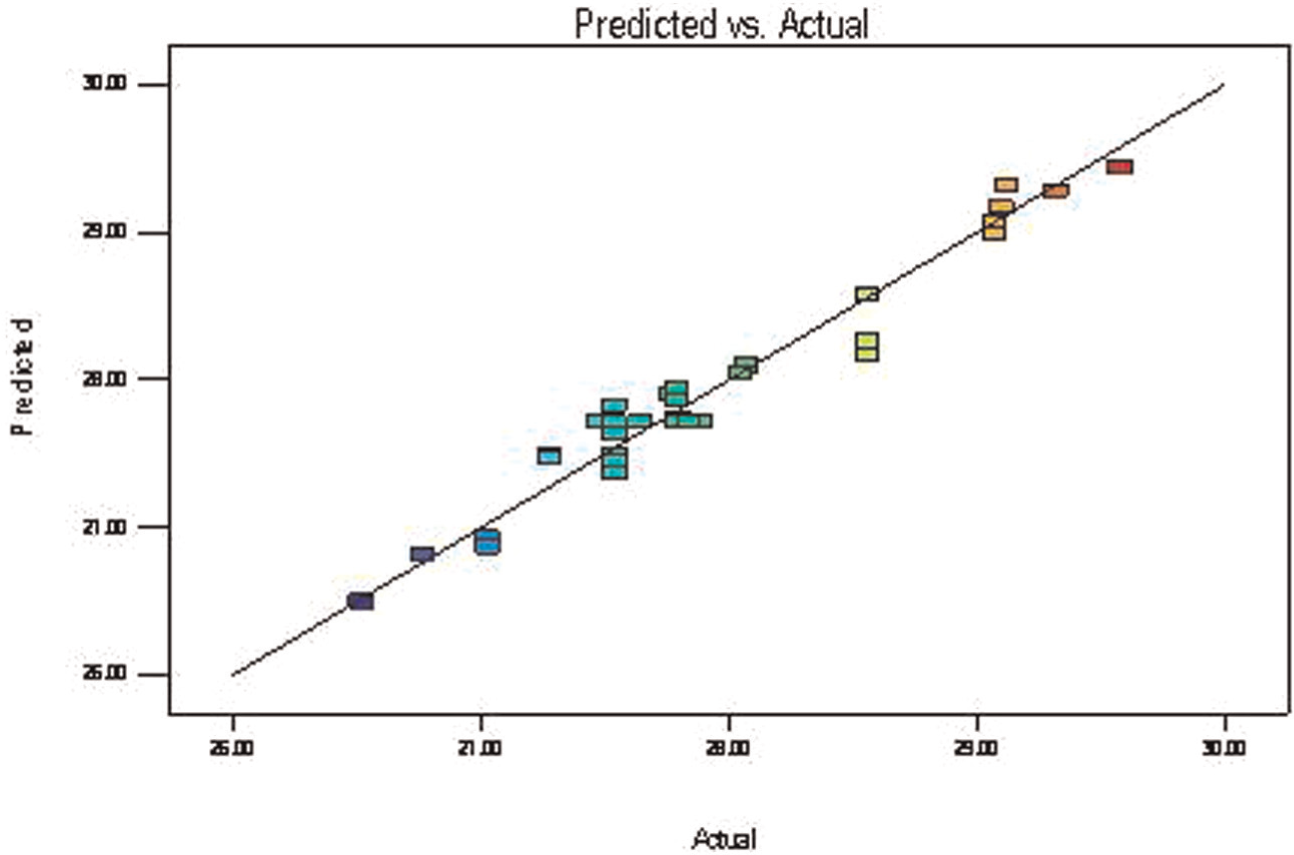
Predicted versus actual percentage elongation value.
Figure 5 depicts the response surface for percentage elongation in relation to the flux constituents CaF2 and MnO. In this figure, the percentage elongation marginally increases with increasing percentage of CaF2 constituents because it lowers the high oxygen in submerged arc welds. The value of percentage elongation also increases with the increasing percentage of MnO constituents because it is a transition metal oxide.
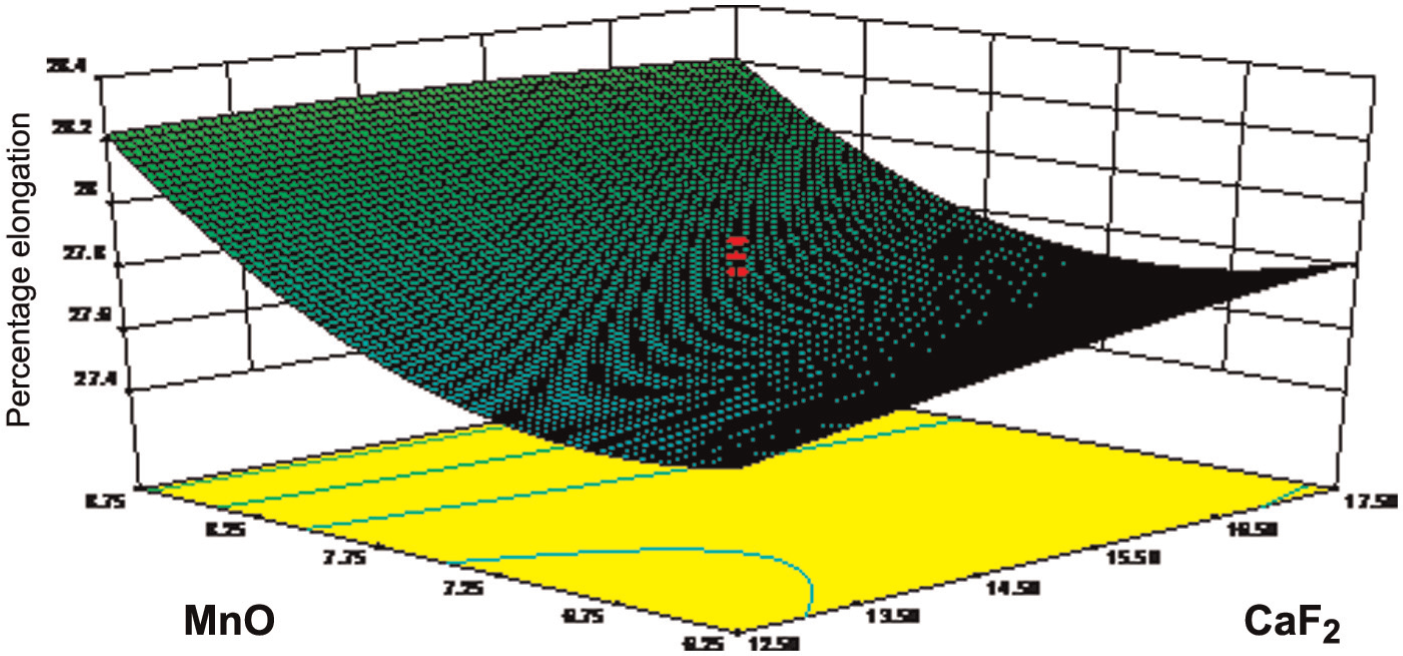
Effect of CaF2 and MnO on percentage elongation.
The response surface of CaF2 constituents and MgO constituents on percentage elongation is shown in Figure 6. This figure shows that the value of percentage elongation increases with CaF2 constituent, the reason for which has been explained earlier. The value of percentage elongation also increases with the increasing percentage of MgO because it is very stable (nonreactive) with the combination of CaF2, which reduces the oxide nature.
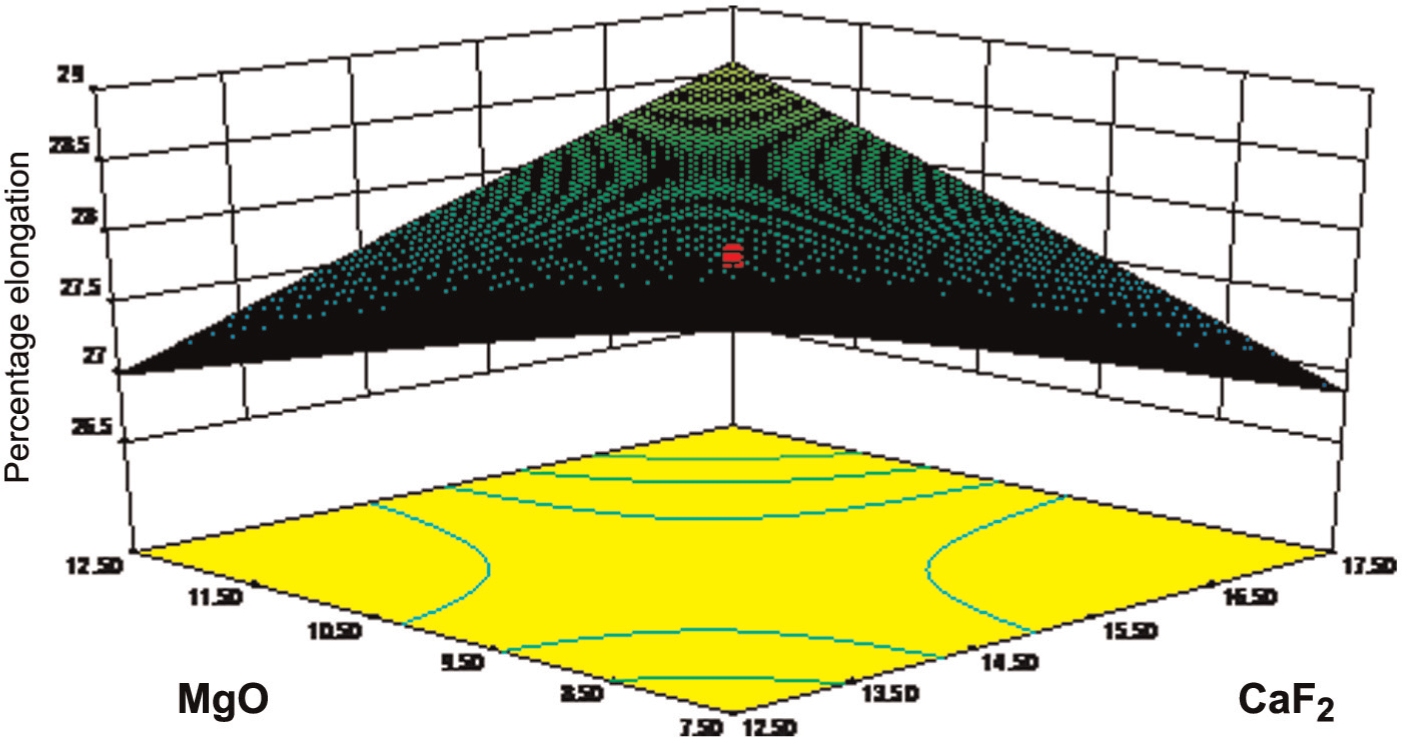
Effect of MgO on elongation.
Figure 7 shows the response surface of behavior of CaF2 and NiO constituents. The effect of CaF2 on elongation was explained earlier, but the effect of NiO on percentage elongation was slightly constant because Ni increases the toughness of the weld metal, which imparts slightly constant elongation at the weld joint. The presence of NiO in the combination of CaF2 is really helpful to increase the percentage elongation.
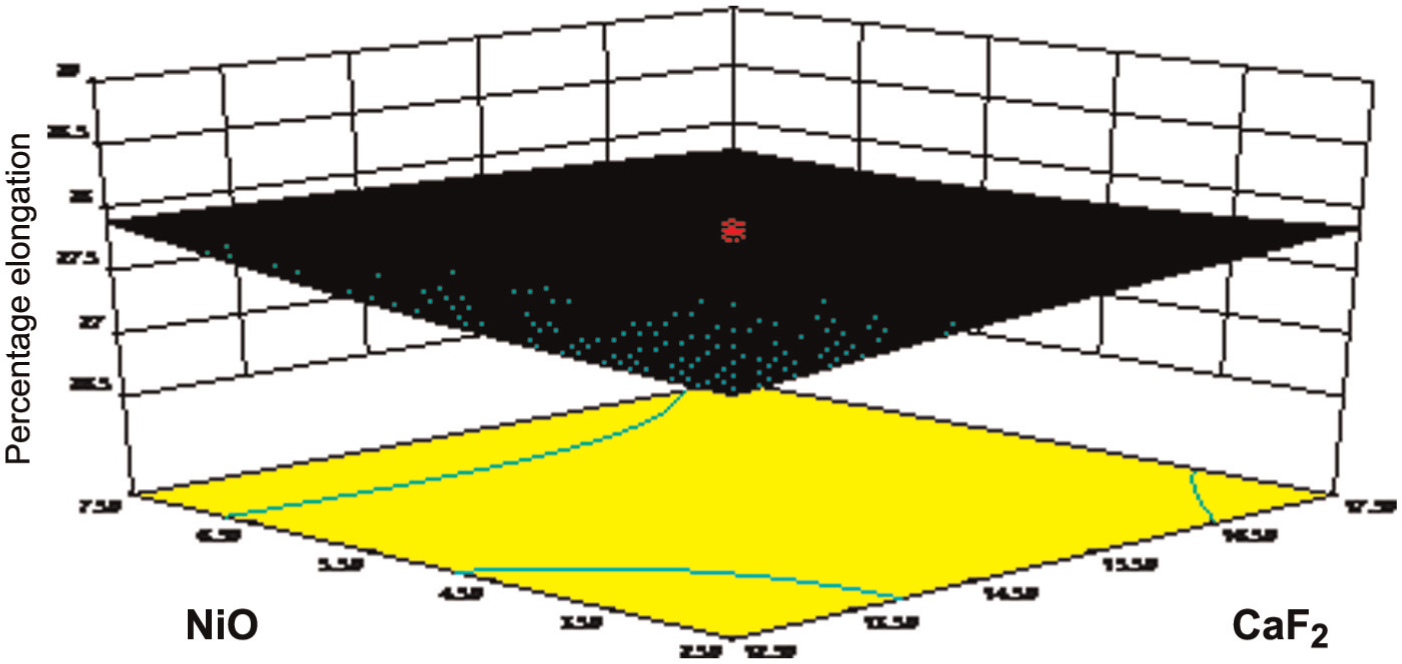
Effect of NiO on elongation.
Figure 8 shows the response surface behavior of CaF2 and Fe-Cr constituents. The effect of CaF2 on elongation was explained earlier, but the effect of Fe-Cr on percentage elongation gave positive impact on percentage elongation because Fe and Cr are good transition metals.
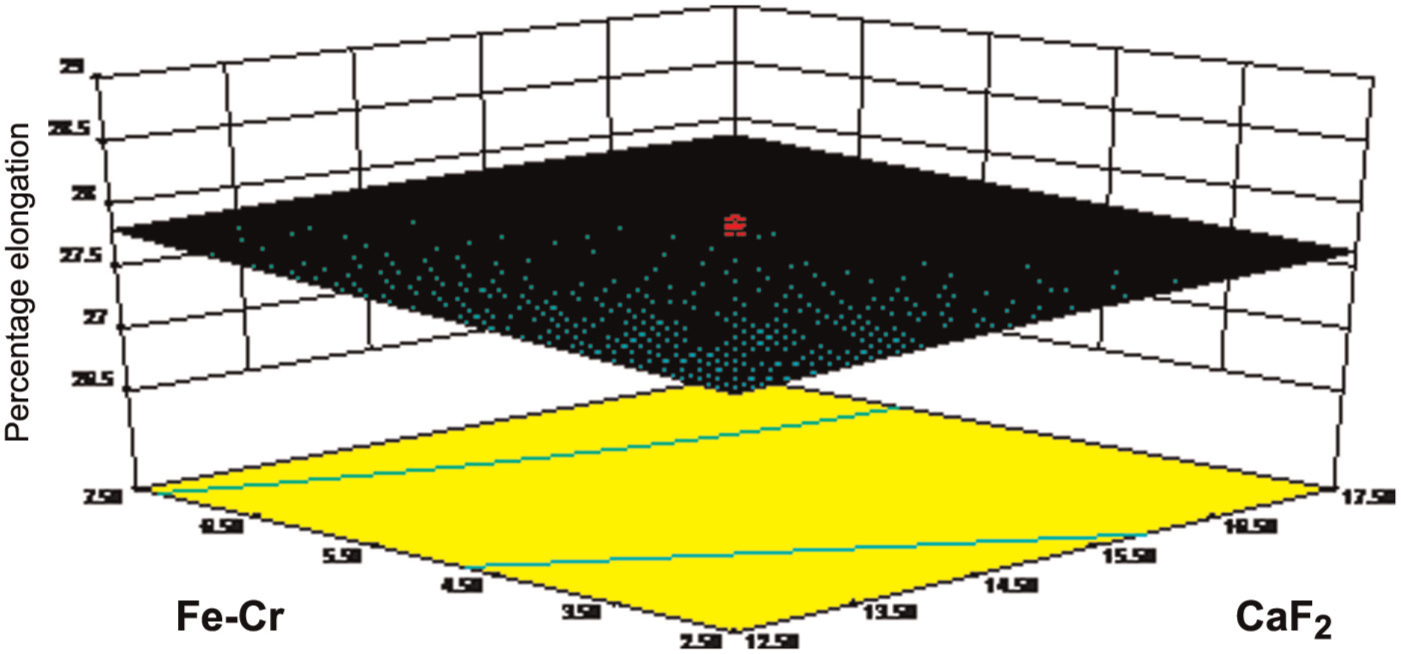
Effect of Fe-Cr on elongation.
Confirmation experiments
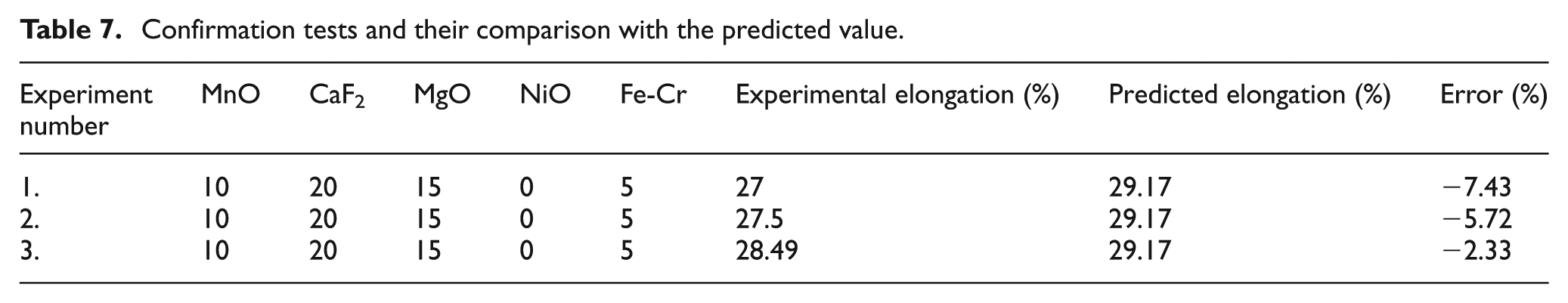
Since for validation, the confirmation tests must be performed, so the three confirmation experiments were performed for elongation. In the confirmation tests, experimental values were compared with the predicted value, obtained by the mathematical model equation of percentage elongation. The confirmation is given in Table 7. From the confirmation tests, it is observed that the calculated error is small and tolerable in the field of welding.
Confirmation tests and their comparison with the predicted value.
Conclusion
The ANOVA revealed that the factors MnO, MgO, and Fe-Cr have the positive impact on elongation but CaF2 and NiO give little impact on elongation.
The value of percentage elongation at the optimum parameters MnO (10), CaF2 (20), MgO (15), NiO (0), and Fe-Cr (5) is 29.17%.
The confirmation tests show that the errors between experimental and predicted values of elongation are −7.43%, −5.72%, and −2.33%.
Footnotes
Acknowledgements
The authors would like to acknowledge the support of Departments of Mechanical and Chemical Engineering, Noida Institute of Engineering and Technology, Greater Noida, Department of Manufacturing Processes and Automation Engineering, Netaji Subhas Institute of Technology, New Delhi, and Department of Mechanical Engineering, Jamia Millia Islamia, New Delhi.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
