Abstract
Hot stamping is increasingly used to improve automobiles by improving safety, reducing weight and containing cost, but with the new focus on improving resource efficiency, novel manufacturing techniques are required. This article presents an innovative hot stamping process step and describes how steel can be austenitized and surface treated simultaneously. The proposed method has the potential to reduce both energy and material usage by respectively combining two manufacturing steps and minimizing the coating of material that does not form part of the finished product. The proposed process is explained using an example, describing how 22MnB5 can be austenitized and zinc coated simultaneously.
Introduction
The use of ultra-high-strength materials is becoming more important in the automotive industry. These materials allow the automakers to build stronger and safer cars while adhering to ever increasing customer expectations of better performance and improved fuel efficiency. 1 Production resource efficiency is also starting to become a important consideration for car makers and a joint study between several Fraunhofer institutes have shown that there are large quantities of resources to be saved in automobile manufacturing. 2 The so-called ‘6R’ concept (recover, reuse, recycle, redesign, reduce and remanufacture) 3 is suggested to help with the approach to improve, not only the resource usage of cars during its useful lifespan, but also before and after. It has been shown that advanced high strength steels (AHSS) can play a significant role in achieving these goals. At the moment press-hardened steel is of particular interest, this process, also known as hot stamping, is currently used to manufacture components with tensile strengths of up to 1500 MPa, or higher in some cases. One of the most popular steels used in hot stamping is 22MnB5, it is commonly used for components such as B-pillars and other safety critical components, but is also becoming a popular selection to reduce the weight of other components. The superior strength of hot stamped components, combined with relatively little spring back, makes it ideal for a number of applications throughout the car body. Hot stamping 4 can be subdivided into indirect press hardening (IPH) 5 and direct press hardening (DPH). 6 In the IDH process (Figure 1) the sheet metal blank is first formed in its cold state (soft), and then heated to a temperature above its Ac3 temperature before it is calibrated and cooled in the same, or a similar, forming tool causing the tensile strength to increase significantly.
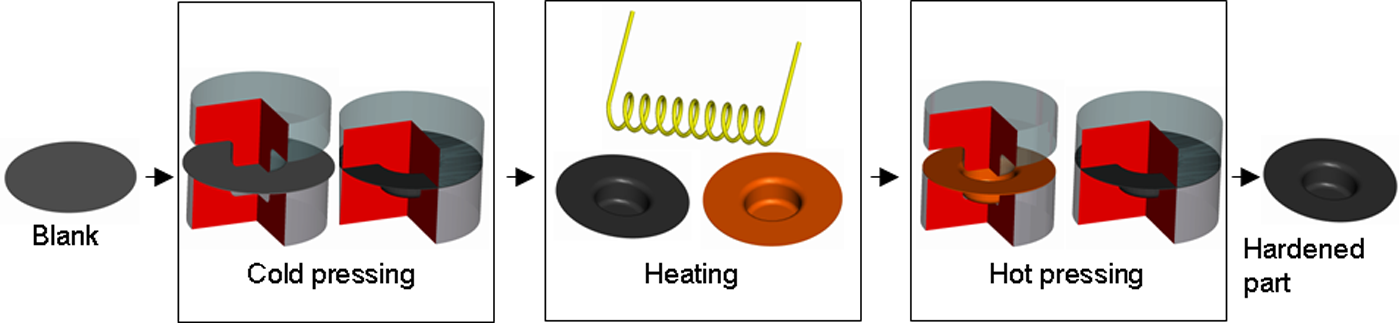
IDH process.
With the DPH process (Figure 2) the blank is first heated to a temperature above its Ac3 before it is formed and cooled in a single step, thus eliminating the cold forming step.
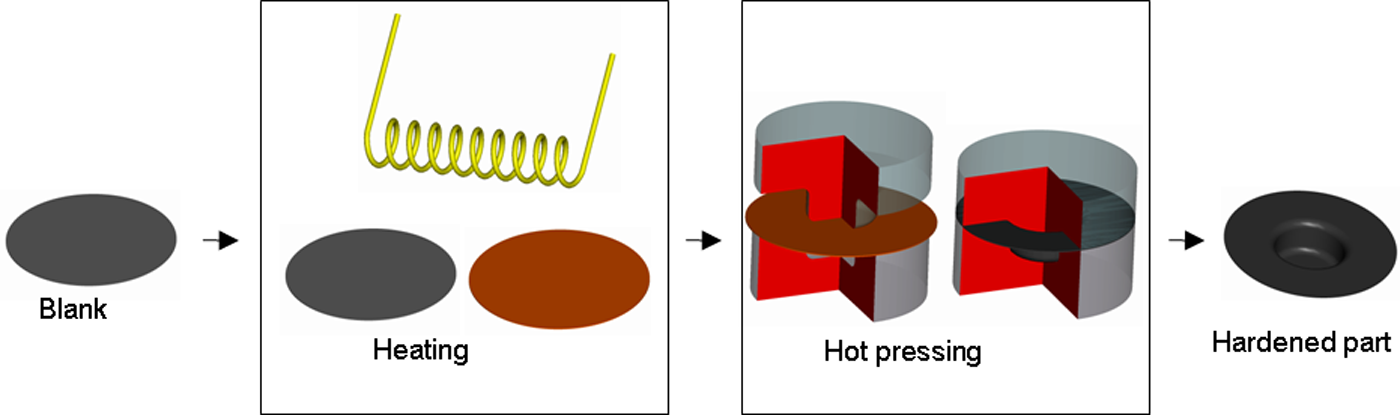
DPH process.
The actual Ac3 temperature (the temperature where the austenitization process is completed) of steel depends on its alloying elements as well as the heating rate. Uncoated steel is seldom used, because of the scale that forms on the surface during heating. The surface coatings used, in general, are zinc, 7 aluminium–silicon (AlSi)8,9 and X-Tec®, 10 but corrosion protection oils 11 are also being investigated. Uncoated and zinc-coated steels are employed in combination with the IPH process and AlSi with the DPH process, whereas the X-Tec® coating can be used in either process. Each of these combinations has unique advantages and disadvantages, but in general the hot stamping process has problems with long cycle times, corrosion of the sheet metal, final part surface quality and tool surface preservation. The main problem with the IPH process is that it contains more steps, resulting in a higher component unit cost than DPH, but the upside is that it can be used to form zinc-coated sheet metal. Zinc-coated sheet steel is ideal for automotive applications as a result of the anodic corrosion protection that the zinc provides, but is not used in DPH owing to the phenomenon of liquid-metal-assisted cracking 12 and because the process is protected by two patents.13,14
The most popular heating strategies used to heat the sheet metal, for and during the hot stamping processes, are convection, conductive and inductive heating. Convection heating is the most versatile, but slow, takes up a lot of floor space and is normally not energy efficient when compared with the other methods. Conductive heating is compact, but can only be used on simple shapes, like pipes, steel strips and rods. When using inductive heating the heating coil needs to be adapted for each new component design and even then uniform heating cannot be guaranteed. Fluidized bed heating 15 is also being investigated as an alternative heating strategy, but more research must still be done.
New process
In the realm of contradictory requirements for building cars bigger and lighter, faster and safer, customized with fewer components and more comfortable and cheaper, new manufacturing approaches are needed. To do this traditional manufacturing methods must be reassessed continuously, even eliminated if possible, in an effort to simplify the complete manufacturing chain. In this spirit we would like to propose a process that does just that. It eliminates one link in the manufacturing chain of surface-coated steel and it has the potential to provide parts, similar in quality as those from current processes, while using fewer resources (both energy and raw material).
The proposed process (Figure 3) is one where the blank is heated by dipping it in a bath with molten alloy to simultaneously heat and apply the surface coating. After heating the blank it gets transferred to the press where it is formed and cooled in a normal hot stamping tool.
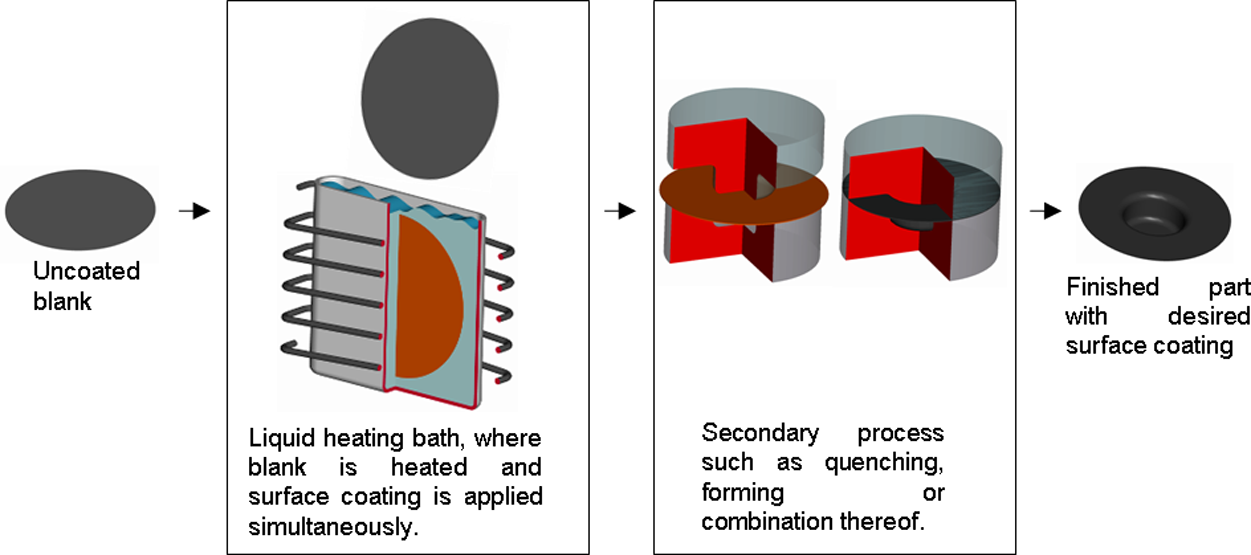
Simplified layout of proposed liquid bath heating and surface coating process, with hot stamping as a secondary process to illustrate the idea.
When a piece of steel is dipped into a molten metal such as a zinc or aluminium alloy, a reaction between the steel surface and the alloy takes place. This reaction rate is governed by the alloy composition and the temperature. If the alloy reacts too fast with the base metal, too-thick a surface coating will be formed. If it reacts too slow, the total heating–coating process will become unnecessarily long. It is important to understand the intermetallic phase behaviour, because some of these phases could be brittle, promoting cracks, and others could be malleable, thus preventing cracks from forming, and the thickness of the different intermetallic phase layers is dependant on the alloy composition, substrate alloy, substrate surface preparation, process temperature and time. Looking at the zinc–iron (Zn–Fe) phase diagram an (αFe)p or αFe + liquid phase can be expected, but when other alloying elements are introduced, ternary phase diagrams will become relevant. The alloy must have a melting point low enough to prevent overheating of the substrate and a boiling point higher than the Ac3 temperature of the steel in question; otherwise the heating effort would be rendered useless. The heating rate of the dipped component would be high as a result of the very low thermal resistance between the sheet metal and the heating alloy, the good conductivity of the steel itself and the molten alloy. The high specific heat capacity of the alloy, together with its total mass, acts like an energy store, with enough energy to heat a blank to the required temperature. There will naturally be a temperature drop in the alloy when a component is dipped and it must be managed to ensure that the temperature of the alloy does not dip below the Ac3 temperature of the steel. The temperature drop is managed by ensuring adequate alloy mass and a high-enough temperature at the start of the dipping cycle. The rate at which heat is replenished into the bath must be regulated as with any other heating system.
The heating alloy used should ensure an acceptable surface finish on the final product and the process has to be designed in such a way that the alloy does not oxidize excessively as is often the case at high temperatures (>800 °C). The ideal heating alloy, for press hardened components, would contain a large percentage of zinc, because of the anodic corrosion protection it provides to steel, thus ensuring lasting structural integrity. Although extensive research has gone into galvanizing, most of the research was conducted at temperatures relevant to hot-dip galvanizing and no research could be found for temperatures in the typical Ac3 temperature range of steel. Therefore, there is currently little information available on how zinc and its alloys interact with steel at these elevated temperatures.
There are a number of possible advantages to this approach.
It combines the surface-coating process with the austenitization process, thus the energy usage is more efficient and one manufacturing step (continuous galvanizing) eliminated when compared with current process chains.
The surface coating is only applied to the material that will be formed and the uncoated scrap can be recycled with minimal coating material wastage.
The steel is heated in an oxygen-free environment (because the sheet is dipped into the molten alloy), therefore eliminating corrosion of the sheet metal during heating. There are several non-vacuum methods available to protect the alloy itself from oxidization.
The high heating rate will improve the productivity over convection ovens resulting in a smaller construction size, thus simplifying heat insulation of the oven.
Neither the efficiency nor the effectiveness are influenced by the blank geometry, as in the case of inductive and conductive heating.
Development of the process is currently under way, taking into account the substrate, the heating alloy and the forming tool. The heating alloy not only has an influence on the substrate and its corrosion protection, but also plays a significant role in the forming process. One of the most important research areas would be the tribological pairing of the heating alloy with different tool surface coatings, and here friction properties, similar to or better than, that of AlSi-coated steel sheet should be aimed for.
A practical consideration that must be considered during the research of this method is the free zinc sticking to the surface of the sheet metal when the heated parts are removed from the heating bath. It is, however, believed that this could be managed with a concept similar to the air–knife systems used on continuous galvanizing lines.
Conclusion
The aim of this communication is to present a concept that combines the surface coating and austenitization steps into one. It explains how a piece of steel can be heated in a molten alloy bath (typically zinc alloy) to a temperature above its Ac3 temperature, while the alloy reacts with its surface to form a reliable surface coating, which can have corrosion or any other predetermined protection properties. The presented concept has the potential to save resources in the form of energy, time and coating material. The time saving is the result of the fast heating time achievable owing to the good heat transfer between the substrate and the heating alloy. The time saving has the potential positive knock-on effect of a smaller oven footprint, thus reducing the floor space requirement in the factory. These potential savings are attractive in the light of modern manufacturing tendencies and the process will be developed further towards industrial implementation.
Footnotes
Acknowledgements
This article arose in the context of the Cluster of Excellence ‘Energy-Efficient Product and Process Innovations in Production Engineering’ (eniPROD®).
Funding
The project is funded by the European Union (European Regional Development Fund) and the Free State of Saxony (Germany).
Conflict of interest
The authors declare that there is no conflict of interest.
