Abstract
Polyimide aerogel, presenting an outstanding new material for lightweight protective thermal insulation, has garnered significant attention in the aerospace field. Despite its potential, due to the high production cost of the supercritical drying method used for its preparation, its widespread application is limited. In this research, a series of lightweight polyimide aerogels (F-PIAs, 3,3',4,4'-biphenyl tetracarboxylic dianhydride (s-BPDA) and 4,4'-diaminodiphenyl ether (ODA)) with excellent thermal protection properties were prepared by lyophilization. The findings indicated that F-PIAs (Freeze-dried polyimide aerogels) exhibited comparable physical characteristics to S-PIAs (Supercritical-dried polyimide aerogels), while their preparation cost was even more economical. F-PIAs exhibits excellent flexible and compression behavior (compression strength up to 1.16 MPa, modulus up to 12.8 MPa), which provide a great structural basis for the production of molded thermal insulation protective materials. The average pore diameter of F-PIAs is in the range of 7∼11 nm. The nanoporous structure leads to the gas within the pores of the aerogel producing the Knudsen effect, thereby significantly enhancing the thermal insulation performance of the aerogel. The thermal conductivity of F-PIA-4 (Freeze-dried polyimide aerogels with a solid content of 4%) at room temperature is as low as 0.023 W·m−1·K−1, which is superior to the PIAs (0.043 W·m−1·K−1) prepared by lyophilization in literature. Its thermal diffusion coefficient changes from 0.108 mm2·s−1 to 0.18 mm2·s−1 in the temperature range of 27°C to 200°C, demonstrating good thermal insulation performance at high temperatures. Meanwhile, after heating on a 150°C flat plate for 11 min and 41 s, the surface temperature of F-PIA-4 was only 36.2°C, further verifying its excellent thermal insulation performance. The results of the TGA experiment on F-PIA-4 demonstrated the high-temperature stability of aerogel (Td5% is higher than 523°C, Td10% is higher than 566°C). The exceptional properties of F-PIAs hold significant practical reference value in helping to reduce the manufacturing cost of high-performance aerogel thermal insulation materials, thereby enabling their widespread application in aerospace, military, and civil field.
Introduction
Near-Space refers to the airspace 20∼100 km above the ground. Because of its significant potential for development and application, the international community has recognized it as a new space for technological and military purposes, drawing widespread attention. 1 Near-Space hypersonic aircraft, generally exceeding 5 Mach, is currently the focus of attention worldwide. To ensure that the aircraft can overcome the harsh hypersonic flight environment, it is crucial to develop high-performance, cost-effective thermal protection lightweight materials.2–9
Aerogel is a type of porous, lightweight material composed of macromolecular polymers, characterized by low density and a large specific surface area. Aerogel is recognized as “legendary material of the 21st century” because of its low density, high porosity, high specific surface area, low thermal conductivity etc. As early as 1931, Kistler took the lead in preparing silicon aerogel in the laboratory. The silica-based aerogels’ appearance made them one of the lightest thermal protection materials and caught the attention of aerospace applications. However, its development in the field of thermal insulation has been greatly limited by its poor mechanical properties, low flexibility, and high brittleness. The emergence of organic polymer-based aerogels, which have natural advantages in flexibility and mechanical properties, has made it a reality that aerogels are widely used in the aerospace field.10–13
In recent years, polyimide aerogels (PIAs), a new type of organic lightweight aerogels, have gradually emerged into people's vision. Polyimide is a type of polymer containing imide groups (-CO-N-CO-) in its main chain. The polyamide ring's own aromatic heterocyclic structure allows for improved bond energy and intermolecular interactions in the main chain, resulting in favorable thermodynamic properties. As a result, PIAs effectively combine the outstanding attributes of PI and aerogel. With their exceptional thermodynamic properties, high porosity, and mesoporous structure, PIAs serve as an exemplary thermal protective insulation material.14–17
Aerogel materials are derived from gel materials in which the liquid component has been replaced by gas, resulting in a fully interconnected solid structure and high porosity. Therefore, using the appropriate drying method is crucial in preparing aerogel, as it allows gas to replace the liquid without damaging the internal pore structure. Currently, the two primary methods for preparing aerogels are CO2 supercritical drying and lyophilization. The CO2 supercritical drying method involves transforming the precursor solution from a sol to a molded gel, substituting the pore-forming solvent with either acetone or absolute ethanol, and subsequently replacing the acetone or ethanol with liquid carbon dioxide. Finally, the liquid carbon dioxide is brought to a supercritical state by heating and pressurizing, and then released as a gas. The process avoids the surface tension caused by the direct gasification of liquid to damage the pore structure. The fundamental principle of lyophilization involves pre-freezing the precursor solution into ice blocks, followed by the release of the pore-forming solvent through vacuum sublimatio. This process creates a porous structure while also preventing the damage caused by liquid surface tension on the pore structure.18–26
Carbon dioxide supercritical drying can prepare nanoscale porous materials with uniform pore size, but its complex process and high operating cost have become the main factors limiting its widespread application. Lyophilization is relatively simple in process, with low operating costs, and often uses water and tert-butanol as gap regulators. Ma et al. 1 successfully prepared anisotropic polyimide aerogel materials with excellent performance by strengthening unidirectional pre-freezing. Zhang et al. 27 adopted the double crosslinking strategy and used lyophilization to regulate the skeleton structure of polyimide aerogel into a staggered honeycomb structure. However, water, as the pore-forming agent in the precursor solution, is susceptible to causing stress during pre-freezing, which can result in cracks in the material. The formation of porous are controllable pores, but the pore size is mostly micron-sized macropores, making it challenging to obtain nanometer-sized mesopores.28–31
The F-PIAs and S-PIAs with different solid contents described in this report were prepared using 4,4'-diaminodiphenyl ether and 3,3', 4,4'-biphenylenedicarboxylic anhydride as raw materials, respectively, through lyophilization and carbon dioxide supercritical drying. The results indicate that the physical properties of F-PIAs are close to S-PIAs. F-PIAs also have nanoscale pore structures, with average pore size and specific surface area close to S-PIAs. F-PIA-4 exhibits a high specific surface area of up to 269 m2·g−1, with average pore size ranging from 7 to 11 nm, a density of 0.082g·cm−3, and excellent flexibility. Its thermal conductivity at room temperature is as low as 0.023 W·m−1·K−1, which has more excellent thermal insulation performance than PIAs (0.043 W·m−1·K−1) prepared by lyophilization in literature. The thermal diffusion coefficient from 27°C to 200°C changes from 0.108 mm2·s−1 to 0.18 mm2·s−1, demonstrating good thermal insulation performance at high temperatures. Compared to aerogels prepared through the traditional process of hydrogel lyophilization, the F-PIAs prepared in this study have several advantages, including smaller pore size, higher specific surface area, and superior thermal insulation performance. These advantages offer practical reference value for reducing the manufacturing cost of high-performance aerogel thermal insulation materials and promoting their wider application in aerospace, military, and civil fields.
Experiment
Materials
The reagents used in this experiment include the following: 3,3', 4,4'- biphenyltetracarboxylic dianhydride (S-BPDA, AR), 1,3,5-tris (4-aminophenoxy) benzene (TAB, AR) are purchased from Shanghai McLean Biochemical Technology Co., Ltd, 4,4'- diaminodiphenyl ether (ODA, AR), acetic anhydride (AR), pyridine (AR), acetone (AR), 1-methyl-2-pyrrolidone (NMP, AR), and tert-butanol (AR) are purchased from Sinopharm Chemical Reagent Co., Ltd.
Synthesis of polyimide aerogel
Firstly, ODA (10.2 mmol, 2.041g), s-BPDA (10.5 mmol, 3.089g), and 37.620g NMP were added to a beaker and magnetically stirred in an ice water bath under nitrogen sealing conditions for 5 h to obtain a solution of PAA with acid anhydride sealing. TAB (0.2267 mmol, 0.0905g) was added and stirred for 15 min to obtain PAA solution with certain viscosity. Then add acetic anhydride and pyridine in turn, continue to stir for 5 min to obtain a transparent and viscous PI solution, quickly pour it into the mold within 10 min to complete the gel curing, and obtain a yellow PI gel block with a solid content of 12%. Similarly, polyimide gel blocks with solid content of 4%, 6%, 8% and 10% were prepared in turn. After the gel block obtained was aged in the mold for 24 h, the gel was put into pure acetone for solvent replacement for 24 h. The replacement was carried out for three consecutive times. Some samples with solid contents of 4%, 6%, 8%, and 10% were directly dried with carbon dioxide supercritical apparatus to obtain PIAs with different solid contents, which are denoted as S-PIA-X. Other samples with solid contents of 4%, 6%, 8%, 10%, and 12% were further solvent replaced with tert-butanol for 24 h. After three consecutive replacements, PIAs with different solid contents were prepared using lyophilization (pre-frozen at -60°C for 5 h), denoted as F-PIA-X. Figure 1 highlights the preparation process, the actual PIA sample, and provides a detailed description of the synthetic chemical reaction equation of PIA. (a) Preparation process of PIAs. (b) Molecuar structure of PIAs. (c) Physical picture of PIAs.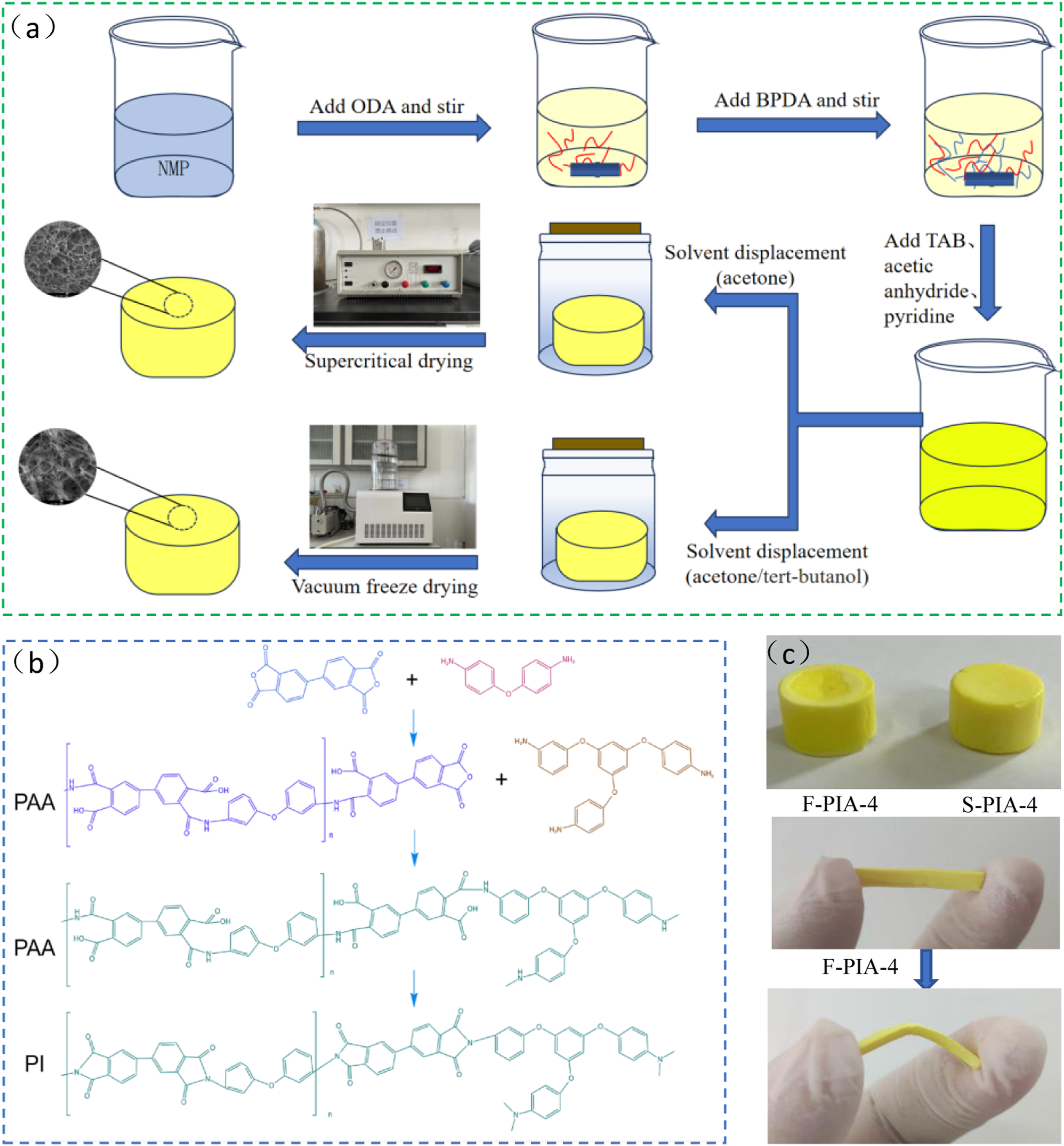
Characterization
The Fourier transform infrared spectroscopy (FT-IR) of F-PIA-4 was examined utilizing a Thermo Fisher IS 50 spectrometer (PerkinElmer, USA), with a resolution of 4 cm−1 and a scanning range of 4000 cm−1 to 400 cm−1. Solid state 13C nuclear magnetic resonance (NMR) spectroscopy was performed using the Brooke 400 M spectrometer. Thermogravimetric analysis (TGA) was performed by TA Q600 Instruments (Delaware, USA) at a heating rate of 10°·min−1. The sample density was measured using an electronic densitometer (MH-300A, Shanghai Qigong Instrument and Equipment Co., Ltd, China). The shrinkage of aerogel is determined by the ratio of sample diameter after drying to sample diameter before drying. The specific surface area and pore structure of the sample were analyzed using a fully automated multi-station specific surface and pore analyzer (JW-BK112T, Beijing Jingwei Gaobo Science and Technology Co., Ltd, China). Under 15 kV accelerating voltage, the morphology of aerogel was observed by scanning electron microscope (JSM-7610FPlus, JEOL Co., Ltd, Japan). The compression mode of the electronic universal testing machine (hr-500, Guangdong Hairui Testing Instrument Co., Ltd, China) is used for the mechanical performance test. The sensor is 1kN, the compression speed is 2 mm·min−1, and the temperature is room temperature. The compression performance of the aerogel is tested according to the National Standard of the People's Republic of China (GB/T 8813-2008). The thermal diffusivity was measured by laser thermal conductivity meter (LFA 457 hyperflash, Netzsch, Germany) in the temperature range of 27.7∼200°C. The thermal imaging image of the heating process was taken with an infrared thermal imaging camera (NUTRIC 326L, Shanghai Thermal Imaging Technology Co., Ltd, China). F-PIA-4 and S-PIA-4 were placed on a hot plate at 150°C for 11 min and 41 s to complete the heating process monitored by the infrared thermal imager. The sample was a block with a side length of 10 mm.
Results and discussion
Structural characterization of PIAs
The preparation process of PIA is shown in Figure 1. PI precursor solutions with varying solid contents were prepared, followed by the chemical imidization process to create wet gels. After replacing the solvent with acetone/tert-butanol and acetone, PIAs were obtained after drying with vacuum freeze dryer and carbon dioxide supercritical dryer. There is no discernible difference between F-PIAs and S-PIAs in appearance, as illustrated in Figure 1. Specifically, F-PIA-4 exhibits excellent flexibility.
As depicted in Figure 2(a), PIAs manifest distinct absorption peaks between 1200 cm−1 and 1350 cm−1, corresponding to the vibration of C-N bond in polyimide molecules, obvious characteristic absorption peaks between 1400 cm−1 and 1450 cm−1, corresponding to the deformation vibration of N-H bond in polyimide chain, and obvious characteristic absorption peaks between 1500 cm−1 and 1600 cm−1, corresponding to the vibration of C=C bond of aromatic ring in polyimide molecules. There are distinct absorption peaks between 1700 cm−1 and 1800 cm−1, corresponding to the carboxylic acid and imide bonds in the polyimide skeleton. In addition, the sample has no obvious characteristic peak at 1860 cm−1, which proves that the prepared PIA has no isoimide structure, indicating the successful synthesis of polyimide. Solid 13C NMR was also used to identify polyimide (Figure 2(b)). Peaks at ∼ 165 ppm and 120 ∼ 160 ppm represent the presence of imino carbonyl and aromatic carbon, which further proves the successful synthesis of polyimide. (a) FTIR curves of F-PIA-4. (b) 13C NMR curves of F-PIA-4.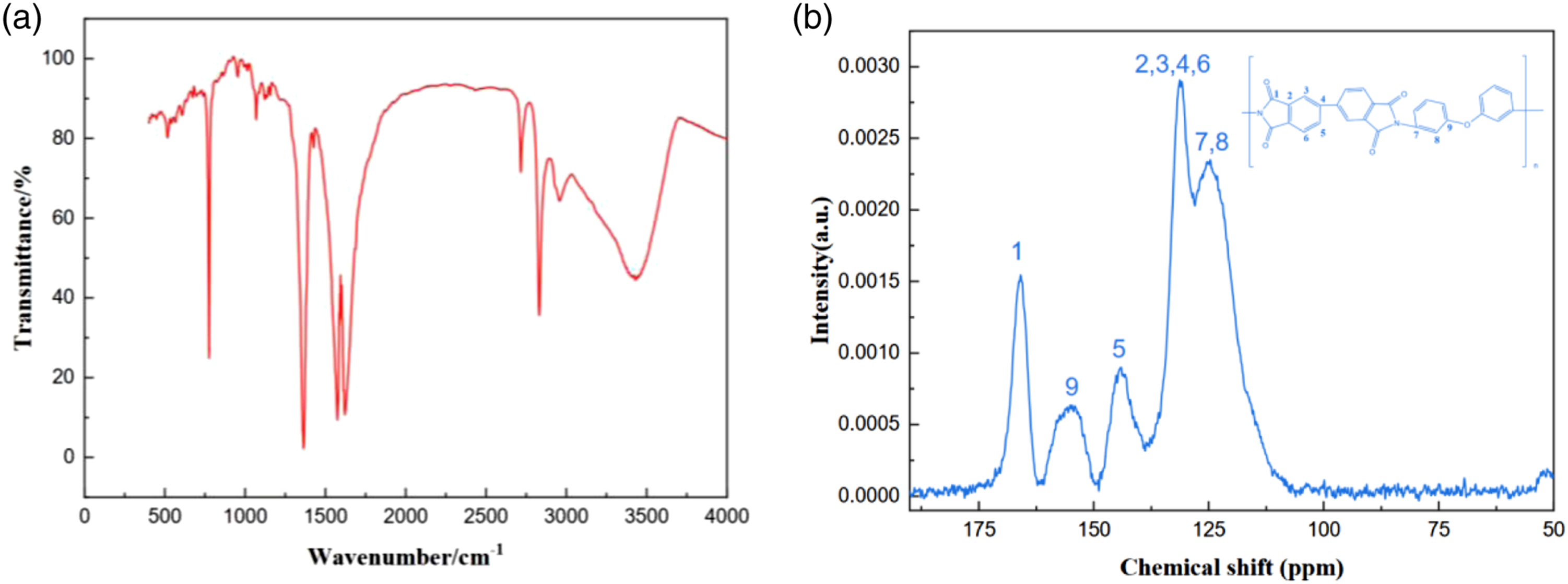
Figure 3(a) and (b) demonstrate the impact of solid content on the density and shrinkage of PIAs. With the increase of solid content, the physical properties of F-PIAs and S-PIAs showed the same trend and similar values. The density of F-PIAs increased from 0.082 g·cm-3 (F-PIA-4) to 0.208 g·cm-3 (F-PIA-12). With the increase of the solid content, the shrinkage rate increases gradually, which is mainly because during the drying process, with the increase of the solid content of the reactive sol, the probability of intermolecular collision and condensation in unit volume increases, the length and degree of crosslinking of the formed molecular chain increase, and the molecular cohesion increases, resulting in the increase of the degree of shrinkage. In this experiment, lightweight aerogel materials with low density and low shrinkage were successfully prepared by lyophilization. Physical properties and microstructure of PIAs. (a) The relationship between density and solid content of PIAs. (b) The relationship between shrinkage rate and solid content of PIAs. (c)-(g) Scanning electron microscopy images of F-PIA-(4-12). (h)-(k) Scanning electron microscopy images of S-PIA-(4-10).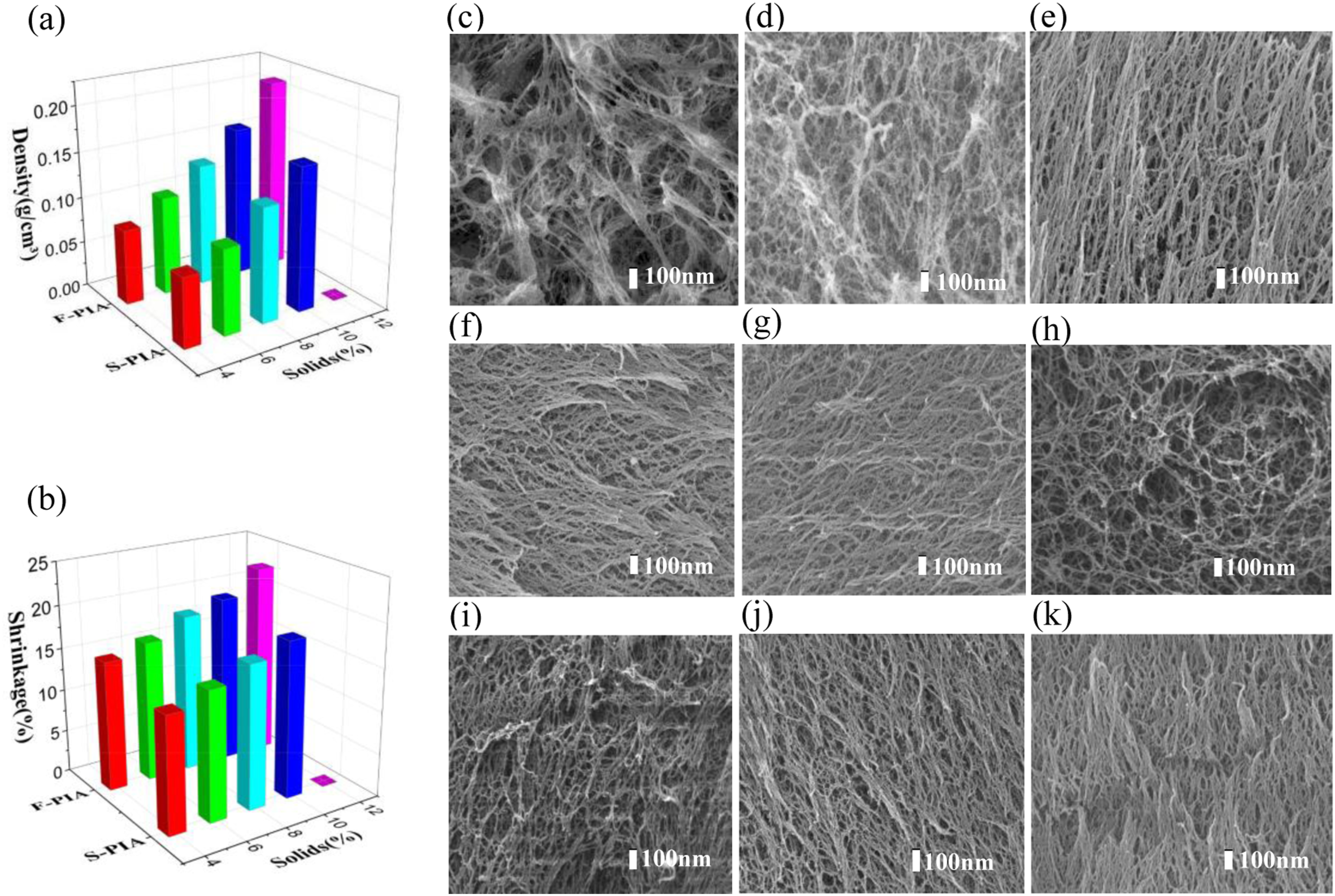
The microstructure of PIAs was demonstrated using the scanning electron microscope (Figure 3(c)–(k)). PIAs with varying solid contents displayed a crosslinking structure of the fiber network. The network pores were mesoporous with a diameter of ∼ 30 nm. These pores were formed by the effect of solvent when PI solution formed gel after polymerization reaction, and were occupied by solvent before drying. Due to the use of lyophilization and supercritical drying, the surface tension generated by the process of liquid phase transition to gas phase was avoided, and the porous structure is maintained to the greatest extent. With the increase of solid content, the network structure of PIAs becomes more compact from sparse, and the network skeleton becomes more and more slender. The microstructure reveals that the solid content significantly affects the pore size and network structure of PIAs. There is no discernible variation in the microstructure of the identical solid content between F-PIAs and S-PIAs. Based on Figure 4(a) and (b), it can be observed that the adsorption-desorption isotherms of PIAs, prepared using the two methods, exhibit a gradual increase during the first half. When the relative pressure (p/p
0
) increases to close to 0.8, the curve rises sharply and reaches adsorption saturation, indicating that the range of pore size distribution of the samples prepared by the two methods is relatively concentrated. According to the classification method of the International Union of Pure and Applied Chemistry (IUPAC), the adsorption type of the above curve belongs to the typical type IV adsorption-desorption isotherm, indicating that the tested mesh pores are mainly mesoporous, and the prepared polyimide aerogel is a typical mesoporous material. With the increase of solid content, the adsorption capacity of F-PIAs per unit mass sample at the highest point of nitrogen adsorption isotherm curve has a downward trend. Nitrogen adsorption-desorption curve and pore size distribution curve of PIAs. (a), ((b) The nitrogen adsorption-desorption curves of F-PIAs and S-PIAs, respectively. (c), ((d) The pore size distribution maps for F-PIAs and S-PIAs, respectively.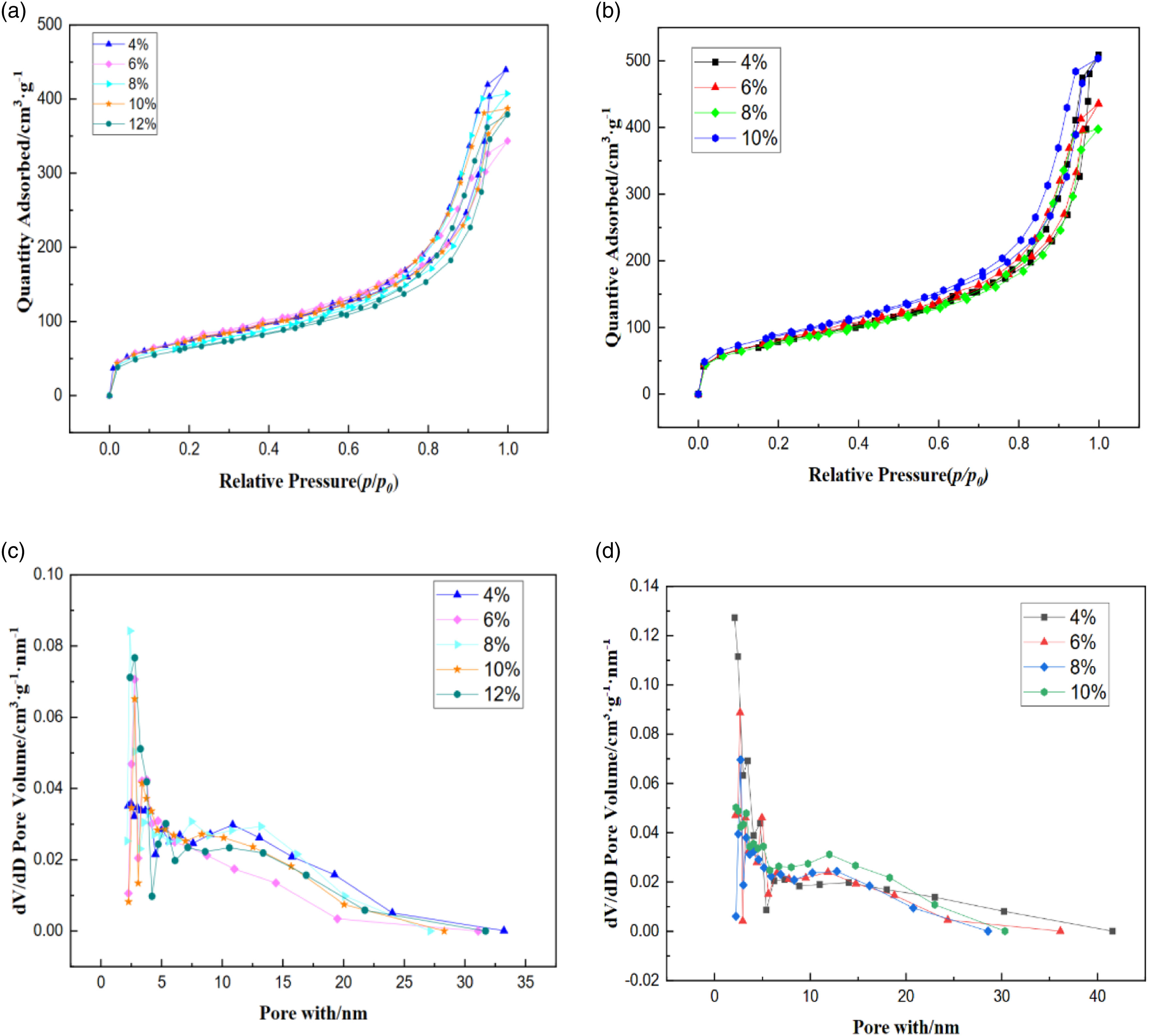
The pore size distribution curve of PIAs was obtained using the adsorption-desorption isothermal curve, following the BJH method, and shown in Figure 4(c) and (d). The number of pore size distribution of S-PIAs in the area with pore size of 2-5 nm was significantly more than that of F-PIAs. This could be attributed to the volume expansion of t-butanol pre-frozen into solid state as a lyophilizer, resulting in the expansion or conflation of some tiny pores, which also verifies the reason why the specific surface area of F-PIAs is slightly smaller than that of S-PIAs. Figure 4(c) illustrates that the overall trend of pore size distribution of aerogel with the increase of solid content of F-PIAs is to move to the left. The main reason is that with the increase of solid content, the size of particles formed by polycondensation remains unchanged, but the number of gel particles formed increases, which makes the average pore size of aerogel tend to decrease. This is consistent with the microstructural analysis of SEM.
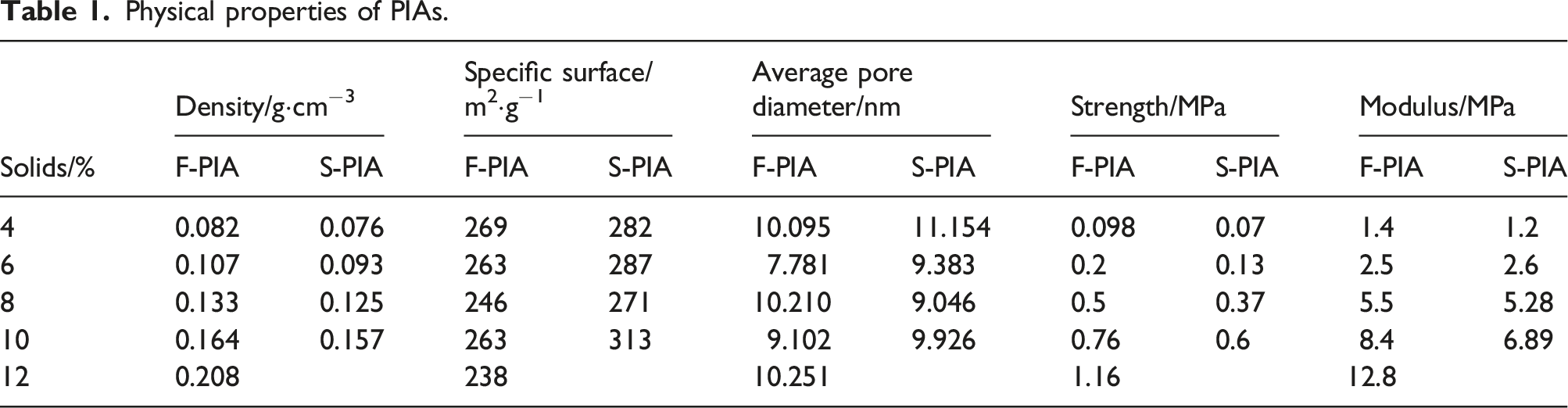
Physical properties of PIAs.
Mechanical properties of PIAS
The thermal insulation material must possess a specific mechanical strength once it is molded. Therefore, the compressive mechanical properties of F-PIAs and S-PIAs are studied in detail. The test results are presented in Figure 5. With the increase of solid content, the strength and modulus of F-PIAs and S-PIAs show an increasing trend, mainly because with the increase of polymer concentration, the denser the skeleton structure of aerogel, the better the mechanical properties. When the solid content is greater than 8%, PIAs show obvious yield state, which may be caused by the destruction of dense skeleton structure. It can be seen from Table 1 that F-PIAs and S-PIAs have proximity compression properties, while F-PIAs perform slightly better. The primary reason could be that the internal pore structure of F-PIAs undergoes a strengthening process due to the stress induced by solvent expansion during the pre-freezing process. F-PIA-4 has a strength of 0.098 MPa and a modulus of 1.4 MPa, and F-PIA-12 has a strength of 1.16 MPa and a modulus of 12.8 MPa, indicating excellent compression properties. Compressive properties of PIAs. (a) Compressive behavior of F-PIAs. (b) Compressive behavior of S-PIAs.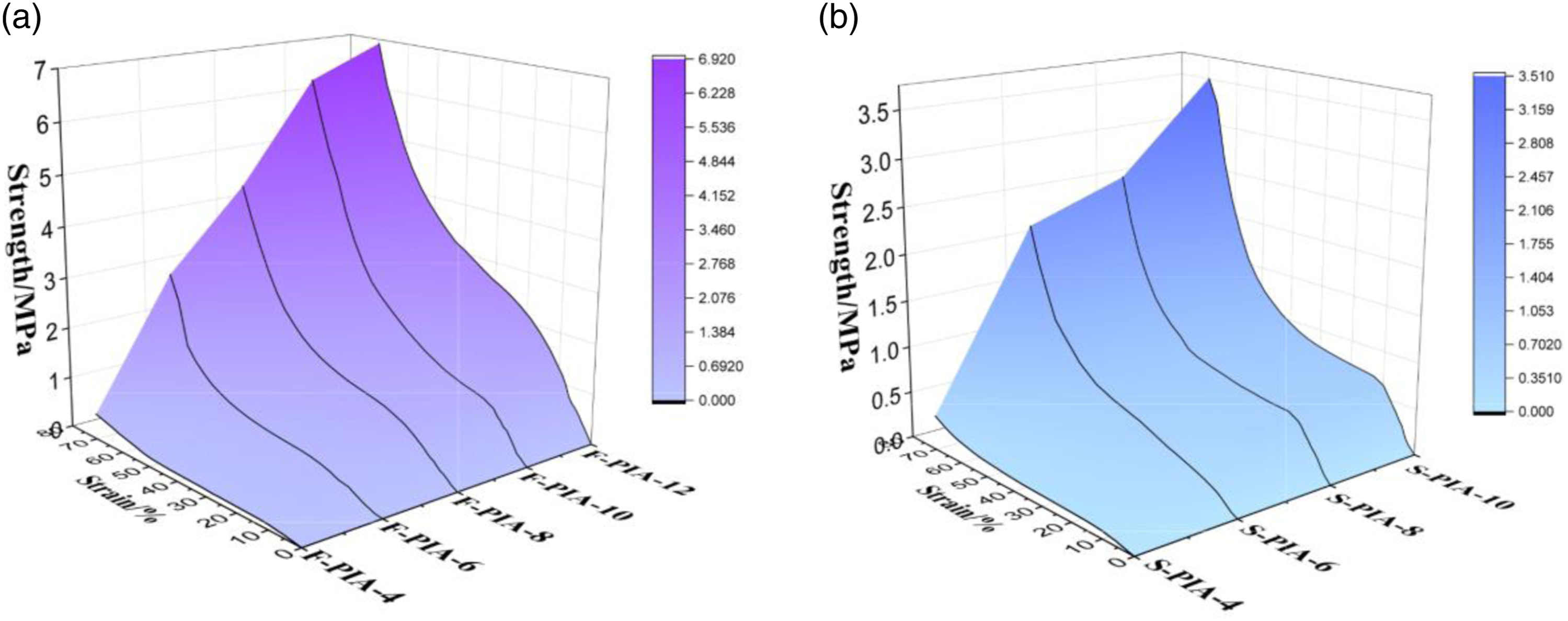
Thermal insulation and thermal stability of PIAs
The thermal diffusivity (α) of PIAs at room temperature and F-PIA-4 in the range of 27°C to 200°C was measured by laser flash method, as illustrated in Figure 6(a). A comparison of the results reveals that F-PIAs and S-PIAs exhibit a similar trend. With the increase of solid content, the thermal diffusivity also gradually increases. This is mainly due to the increase of polymer matrix improves the skeleton density of PIAs, forming a thermal conductivity network with polymer matrix as the main body, and thus improving the thermal conductivity of PIA system. With the same solid content, the thermal diffusivity of F-PIAs and S-PIAs is proximity, indicating that the thermal insulation properties of F-PIAs and S-PIAs are proximity. This is mainly because F-PIAs also have a porous structure with an average pore size of less than 20 nm. The gas in the pore produces Knudsen effect, namely, the average pore size is less than the average free path of the gas molecule. The gas locked by the pore is in a relatively static state, and the thermal conductivity of the internal gas will be reduced. Knudsen effect greatly reduces the thermal conductivity of the aerogel. It can be found from Figure 6(b) that the thermal diffusivity of F-PIA-4 increases slightly from 0.106 mm2·s−1 (27°C) to 0.18 mm2·s−1 (200°C), showing good thermal insulation performance at high temperature. According to the formula λ=ρ×α×c and the specific heat c of the known literature,
32
it is concluded that the thermal conductivity of F-PIA-4 at room temperature is as low as 0.023 W·m−1·K−1, which has better thermal insulation performance than the PIAs (0.043 W·m−1·K−1) prepared by lyophilization in the known literature.
1
Thermal management behavior of PIAs. (a) Thermal diffusivity of PIAs. (b) Thermal diffusivity of F-PIA-4 under temperatures of 27, 50, 100, 150 and 200°C. (c) TAG of PIAs. (d) Photograph of PIAs placed on a heated plate. (e), (f) Infrared images of F-PIA-4 and S-PIA-4 set on a 150°C hot plate for 5 min and 11 min. (g) Relationship between temperature and time of F-PIA-4 and S-PIA-4 on flat plate. (h) Relationship between height and temperature of F-PIA-4 and S-PIA-4 on flat plate.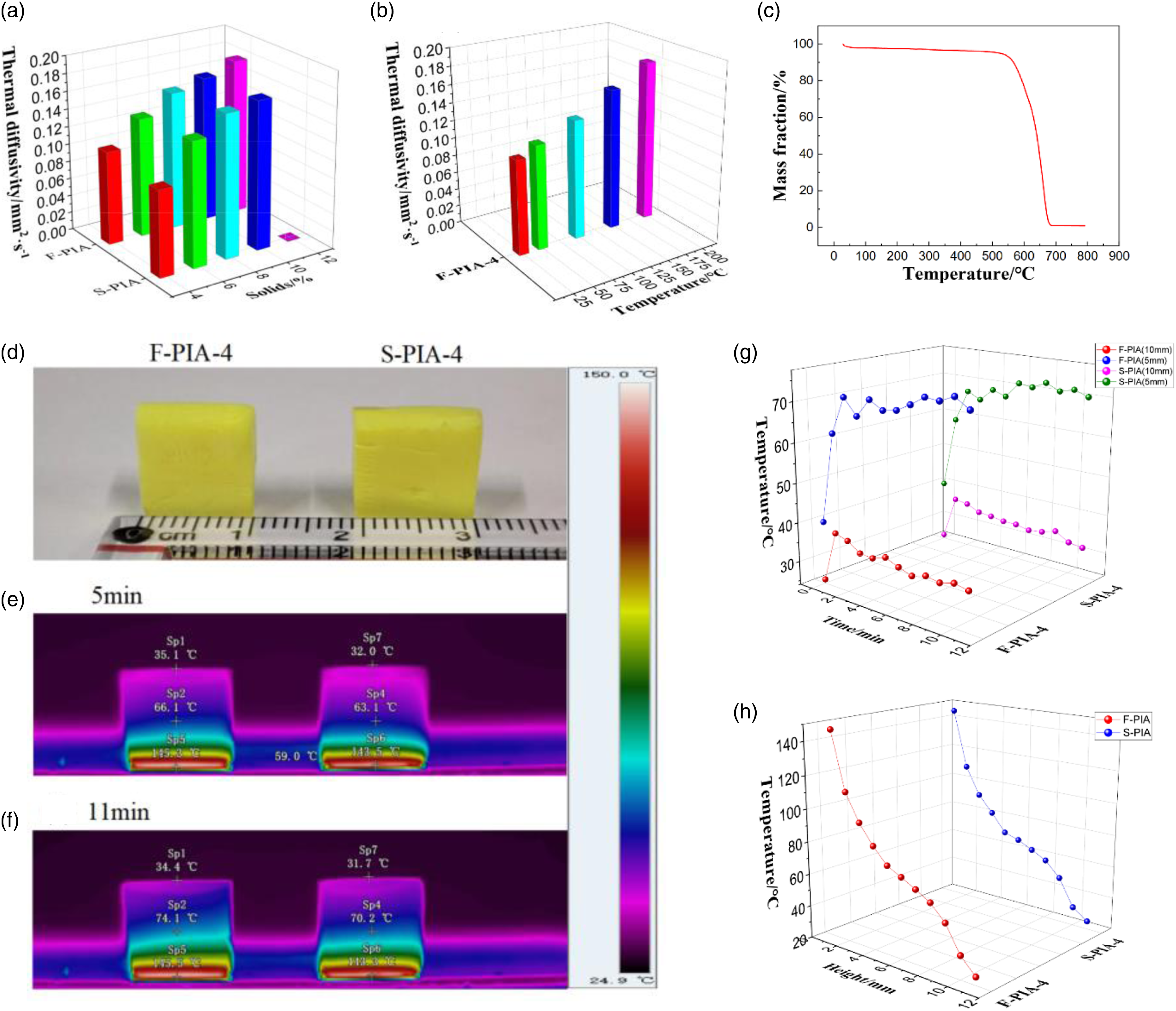
The thermal insulation behavior of PIAs is demonstrated by infrared thermal imaging images (Figure 6(d)–(g)). Place F-PIA-4 and S-PIA-4 on the heating plate to perform the heating process (150°C, lasting for 11 min and 41 s), and use infrared camera for monitoring. The temperature of aerogel increased with time. After about 4 min, the temperature of F-PIA-4 reached equilibrium at 63.8∼66.3°C (5 mm) and 34.6∼36.2°C (10 mm), and the temperature of S-PIA-4 reached equilibrium at 61.9∼64.4°C (5 mm) and 32.3∼34.2°C (10 mm). The maximum temperature of F-PIA-4 surface is 36.2°C, and the maximum temperature of S-PIA-4 surface is 34.2°C, which again shows that F-PIAs is close to S-PIAs in thermal insulation performance. After the temperature was maintained at 150°C for 11 min and 41 s, the relationship between the temperature and the hot surface height of PIAs was also studied (Figure 6(g)). The temperature of PIAs was the highest on the heating surface, close to 150°C, and the lowest on the upper surface (10 mm). The axial temperature range of F-PIA-4 was 36.2∼150°C, and the axial temperature range of S-PIA-4 was 34.2∼150°C, showing good thermal insulation performance.
To delve deeper into the high temperature thermal stability of PIAs, TGA experiments were conducted on F-PIA-4 in an air atmosphere, as illustrated in Figure 6(c). The results indicated that the 5% weight loss temperature (Td5%) was higher than 523°C, and the 10% weight loss temperature was higher than 566°C, which proved that PIAs exhibited excellent thermal stability at high temperature.
Conclusion
In summary, the wet gel was prepared by chemical imidization, followed by replacement of the solvent with acetone/tert-butanol and acetone. Subsequently, it was dried by lyophilizer and carbon dioxide supercritical dryer respectively to obtain PIAs. After undergoing tests and characterizations, it was found that F-PIAs and S-PIAs have proximity physical properties, with no significant difference observed in photomacrographs and SEM microstructure. F-PIAs exhibit excellent flexibility, and its compressive strength and modulus are up to 1.16 MPa and 12.8 MPa respectively, which provides a good structural basis for the production of molded thermal insulation protective layer materials. The average pore size of F-PIAs is in the range of 7-11 nm. The nanoporous structure in aerogel causes the gas within its pores to produce the Knudsen effect, leading to a significant enhancement in the thermal insulation capabilities of the material. The thermal conductivity of F-PIA-4 at room temperature is as low as 0.023 W·m−1·K−1, which is better than the PIAs (0.043 W·m−1·K−1) prepared by lyophilization in literature. The thermal diffusivity exhibits good high-temperature thermal insulation performance, varying from 0.108 mm2·s−1 to 0.18 mm2·s−1 in the temperature range of 27°C to 200°C. Furthermore, the excellent thermal insulation performance was confirmed once again when the surface temperature of F-PIA-4 reached a maximum of 36.2°C, after being heated on a 150°C plate for 11 min and 41 s. The TGA results of F-PIA-4 demonstrated the high temperature stability of aerogels (Td5% higher than 523°C, Td10% higher than 566°C). The outstanding performance of F-PIAs holds significant practical reference value in reducing the manufacturing cost of high-performance aerogel thermal insulation materials and fostering their application in aerospace, military, and civilian areas.
Footnotes
Acknowledgments
The authors greatly appreciate the financial supports from the Key Project of the Scientific Research Program of the Hubei Provincial Department of Education (D20201602) and the Hubei Natural Science Foundation (2021CFB560) and (2023AFB385).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Hubei Provincial Department of Education (D20201602) and Science and Technology Program of Hubei Province (2021CFB560 and 2023AFB385).
