Abstract
UK drug discovery and development is increasingly being shaped through a complex interaction of research, policy and practice. However, our understanding of this innovation system is partially due to the dearth of systems-level empirical studies and to simplistic conceptual approaches. This study uses a Triple Helix systems approach to illustrate how a novel database of Research Excellence Framework 2014 impact case studies may be used both to advance empirical understanding of UK drug discovery and development and for theory development. The authors refine the Triple Helix system by identifying relationships between its three components (academia, government and industry) and various social actors. The paper also make two contributions to practice, concerning the relative unimportance for impact generation of geographical clusters relative to strategic alliances, network linkages and knowledge spillovers, and the strong bias towards national and Anglo-American academic–practitioner linkages with few or no links to emerging knowledge economies.
Keywords
UK drug discovery and development is increasingly being shaped through a complex interaction of research, policy and practice, in response to technological advances and changing demographic and socioeconomic trends (Corbett et al., 2017; Deloitte, 2017; NHS England, 2017). Big Pharma business models have largely shifted from vertical integration to network or systems integration, emphasising precompetitive initiatives and public–private partnerships (Cooke, 2004; Hunter and Stephens, 2010; PricewaterhouseCoopers, 2009; Ràfols et al., 2014; TBR and CBSL, 2016). The UK government has adopted increasingly supportive policies, including smart specialisation strategies, the Life Sciences Sector Deal and the Industry Strategy (BEIS, 2017; Bell, 2017; DBIS, 2015), and is encouraging academia to engage in impact, defined as ‘an effect on, change or benefit to the economy, society, culture, public policy or services, health, the environment or quality of life, beyond academia’ (HEFCE, 2011).
In order to design effective policy and business strategies in this changing landscape, the division of innovative labour (what actors are involved in innovation, their functions and interactions) must be clearly understood to avoid the hyping up of some and the neglect of others (Lazonick and Mazzucato, 2013). However, the dearth of systems-level empirical studies exploring UK drug discovery and development means that our understanding of the system is partial and likely to be anecdotal or ‘myth-laden’ (Corbett et al., 2017; Hopkins et al., 2007; Light and Lexchin, 2012; Munos, 2009; Scannell et al., 2012; Schmid and Smith, 2005). To illustrate, studies of impact-related innovation focus on knowledge commercialisation and industry engagement at research-intensive higher education institutions (HEIs), and typically neglect teaching-led HEIs, non-profit organisations, government bodies and patient groups, service and process innovation, and localised practice-based innovations (Abreu et al., 2016; Barlow, 2017; Caron-Flinterman et al., 2007; Dalziel, 2007; Hughes and Kitson, 2012; Smith et al., 2015; Savory, 2009).
The inclusion of an impact assessment in the UK’s 2014 Research Excellence Framework (REF2014) constitutes the first national-level systematic evaluation of academic research not only according to academic outputs but also to effects on society (Hinrichs-Krapels and Grant, 2016). REF2014 has left a rich database of case studies submitted by all UK HEIs describing the significance and reach of impact underpinned by research during the period January 1993 to December 2013 (KCL and Digital Science, 2015), how impact emerged and who was involved in its emergence. It offers a new longitudinal perspective on the division of innovative labour in a range of UK innovation systems.
In this paper, we illustrate how REF case studies may be used both for theory development and as a means to understand or advance insight into the UK drug discovery and development system. We develop a three-stage review approach to do this: Choosing a conceptual model with acceptable explanatory power. We critically appraise conceptual models of how research, policy and practice inform and interact with each other, and identify the Triple Helix systems model (Ranga and Etzkowitz, 2013) as being appropriate to explore impact case studies. Systematic review of REF case studies. We employ a systematic review methodology (Tranfield et al., 2003) to interrogate the REF2014 database and identify 268 case studies relevant to drug discovery and development. Integrative synthesis. We adopt a systematic combining approach (Dubois and Gadde, 2002) to develop a Triple Helix systems perspective of UK drug discovery and development.
Our work adds to existing research in three important ways. First, our novel methodology constitutes the first exploration of REF data within a particular context of application rather than a field of research (Greenhalgh and Fahy, 2015; Hole, 2017; Pidd and Broadbent, 2015), and thereby offers a template for the exploration of subsequent REF exercises. Second, we refine the Triple Helix system by identifying relationships between its three components (academia, government and industry) and social actors, including non-governmental organisations (NGOs), the media and supranational organisations. Third, we expand empirical knowledge concerning how academics and academic research contribute to the UK drug discovery and development system. We make two contributions to practice, concerning the relative unimportance of geographical clusters relative to strategic alliances, network linkages and knowledge spillovers, and the strong bias towards national and Anglo-American academic–practitioner linkages with few or no links to emerging knowledge economies.
Stage 1: Choosing a conceptual model with acceptable explanatory power
Three broad approaches have been used to conceptualise how impact is derived from research: linear, relational and system models (Best and Holmes, 2010). Linear models envisage a unidirectional flow of knowledge from basic research to applied research to commercialisation (Hara, 2003). This type of model is problematic, and has been described as a ‘folk model’ (Balconi et al., 2010) and a ‘classic straw man’ (Edgerton, 2004) never intended to be an analytically useful concept. It is technologically deterministic (neglecting market pull or societal need) and its use has led to a disproportionate emphasis on scientific research and a neglect of both the business processes required to bring technology to the market (Balconi et al., 2010; Edgerton, 2004; Tait and Williams, 1999) and the role of industry (Vallas and Kleinman, 2008).
Relational models are more sophisticated and emphasise knowledge sharing and the development of partnerships, networks and clusters consisting of stakeholders with common interests. There is a large body of literature which explores the factors that facilitate or impede relationships between academia and industry (Ankrah and Al-Tabbaa, 2015; D’Este and Patel, 2007; Perkmann et al., 2013), although other actors (local and national government, public funding bodies, the third sector, and intermediary organisations) have received less attention. However, relational models often neglect the fact that knowledge diffusion and relationship forming are mediated by structures containing actors with varying priorities, expectations, worldviews, capabilities and vocabularies. As a consequence, systemic factors that influence the development of impact from research, such as institutional biases (Abraham, 2009; Davis and Abraham, 2011) or societal concerns about the accountability of scientific research (Stokols et al., 2008) are generally ignored.
Systems models are the most sophisticated model currently available (Carlsson et al., 2002). These recognise that innovation occurs in dynamic and constantly changing complex adaptive systems, shaped by culture, structures, priorities and the capacities of, and feedback between, actors (Best et al., 2009). Various system types (national, regional, sectoral, technological) have been studied, but rarely in the context of UK drug discovery and development. Using a combination of hand-searching Nature journals and a selective search of Web of Science, we found just four papers relevant to the UK context, adopting national, regional and technological innovation systems perspectives (Cooke, 2004; Herrmann and Peine, 2011; Kooijman et al., 2017; Lawton Smith, 2004). The ‘ecosystem’ approach, a concept based on the national system of innovation, is beginning to be employed in the grey literature (Bell, 2017; Deloitte, 2017; HM Government, 2017; TBR and CBSL, 2016). However, these examples rarely explore impediments caused by the conflicting worldviews or cultures of partners, and are therefore more likely underpinned by relationship models.
National, regional, technological and sectoral systems approaches have been criticised on the grounds that many accounts are overly descriptive, and that geographically or sectorally delimited systems do not adequately account for the increasingly open and globalised nature of research and innovation (Weber and Truffer, 2017). Furthermore, they position the industrial firm as the primary locus of technological innovation. As Big Pharma increasingly adopts the role of network or systems integrator rather than innovation locus, the explanatory power of these types of systems approaches may become increasingly limited (Cooke, 2004; Ràfols et al., 2014). Based on these considerations, we chose the Triple Helix systems (THS) model (Ranga and Etzkowitz, 2013) as our conceptual foundation. The THS is based on the broader Triple Helix approach, one of the most developed conceptual and empirical accounts of the changing nature of university–industry–government relationships in knowledge economies (Etzkowitz, 2003, 2011). The Triple Helix approach positions academia as the primary locus of innovative knowledge creation, and is therefore consistent with the perspective offered by REF case studies.
Stage 2: Systematic review of drug discovery and development case studies
Our review of impact case studies was based on the systematic literature review methodology developed by Denyer and Tranfield (2009). This consisted of five steps: question formulation; locating studies; study selection/evaluation; analysis/synthesis; and reporting/using results.
Question formulation
The scope, research question, protocol and inclusion/exclusion criteria were established in dialogue with a guidance panel consisting of academics and pharmaceutical industry practitioners under the auspices of the British Pharmacological Society.
Locating studies
The REF2014 impact case study database was selected as it represents the first national level systematic evaluation of research impact globally (Hinrichs-Krapels and Grant, 2016). REF2014 was conducted by the Higher Education Funding Council for England (HEFCE) on behalf of all UK research funding councils. It assessed the quality of HEI research published between January 2008 to December 2013, and both the significance and reach of impact underpinned by research during the period January 1993 to December 2013 (KCL and Digital Science, 2015). A total of 154 (out of 164 publicly funded) UK HEIs submitted 6,975 impact case studies for assessment, of which 6,679 have been made publicly accessible (HEFCE, 2014). These are spread across four panels: life sciences, engineering and physical sciences, social sciences, arts and humanities.
Case studies were not assessed for academic rigour during the REF exercise and so may be considered an example of grey literature – that is, ‘knowledge artefacts that are not the product of peer-review processes characterising publication in scientific journals’ (Adams et al., 2017: 433). We characterise REF2014 case studies as first-tier grey literature according to Adams’s taxonomy. This differentiates along two dimensions: outlet control (the extent to which content is produced, moderated or edited in conformance with explicit and transparent knowledge creation criteria) and source expertise (the extent to which the authority of the producer of content can be determined). In terms of outlet control, case studies have significant retrievability in that they have been made available on a publicly accessible website maintained by UK Research and Innovation (UKRI). In terms of source expertise, case studies have significant credibility in that they were written by or with the involvement of academics who led research projects which underpinned impact. In addition, impact claims in case studies were required to be supported by evidence for the purposes of REF2014, and there is evidence that legitimate claims were sometimes not made due to difficulties in establishing proof (Manville et al., 2014). Thus we consider relevant REF2014 case studies to offer credible accounts from the perspective of focal academics of the UK drug discovery and development THS.
Study selection/evaluation
Our search strategy (Figure 1) involved identifying case studies in the REF2014 database relevant to a drug discovery and development context. In consultation with our mixed academic and practitioner guidance panel, we developed a list of keywords relevant to this context (Appendix 1). This method of drawing on the tacit knowledge of stakeholders was chosen in order to improve legitimacy and validity of the ensuing synthesis (Dixon-Woods, 2011). We combined these keywords into a number of search strings and used these to search the REF database and extract relevant case studies. In all, 3,645 case studies were extracted in this way.
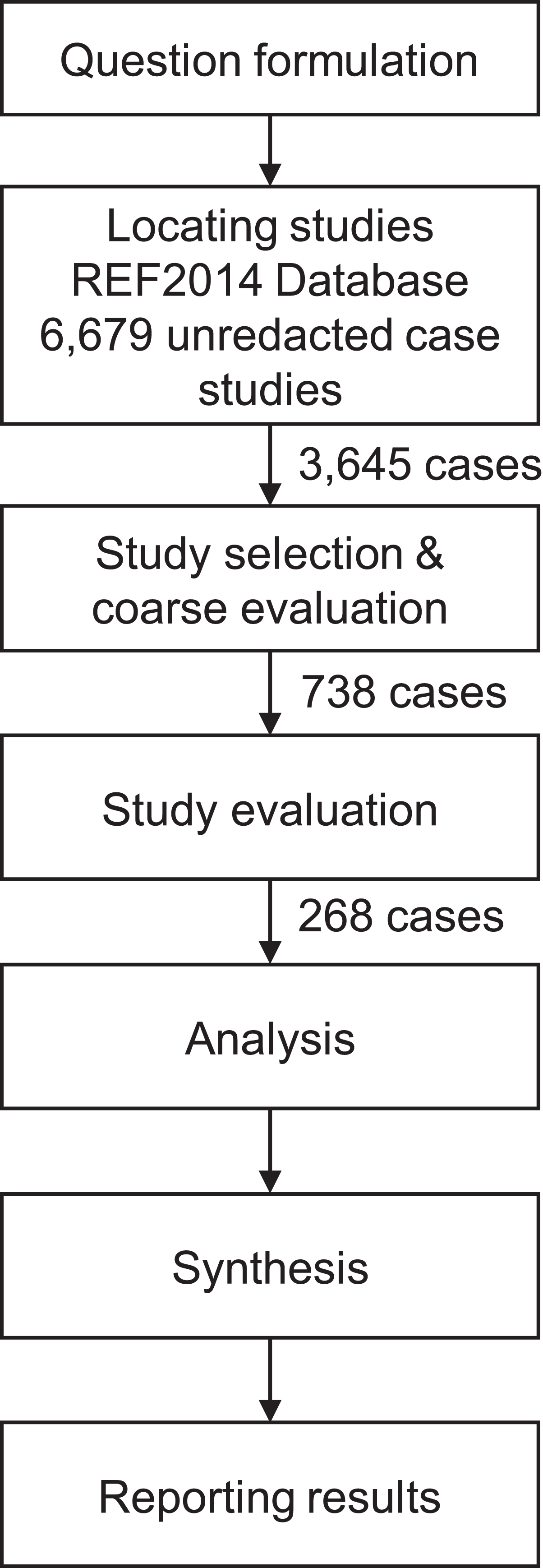
Search strategy.
In order to reduce our pool of extracted case studies, we adopted a fitness-for-purpose approach whereby quality appraisal was suborned to the relevance of the case studies in contributing to synthesis and understanding of the research problem (Briner et al., 2009; Pawson, 2006; Van Aken and Romme, 2009). This was a two-stage process, consisting of a coarse evaluation, based on a reading of the ‘Summary of impact’, followed by a fine evaluation, based on a reading of the ‘Details of impact’. Thus no study was excluded on the basis of quality, but rather studies were included on the basis of relevance if the relevant sections of the case study included at least one of a list of inclusion criteria developed by the research team (Table 1). Reliability was achieved by exceeding an 80% agreement threshold among a research team consisting of both academic and industry subject matter experts. In this way, we reduced our pool to a shortlist of 268 relevant case studies.
Inclusion criteria.

We defined the term ‘drug’ to mean medicine not restricted to use in humans, and therefore include small molecule drugs as well as other approaches (e.g. biologics, stem cells). For the purposes of this categorisation we included the reporting of new therapeutic avenues as a result of fundamental research as well as significant milestones in the drug development process. We also included evidence of a stratified/personalised approach; that is, the use of genetic/biomarker information or techniques to inform the discovery of new drugs. We excluded medical devices, advances in drug disposal and surgical interventions.
Analysis/synthesis
Case studies are not generalisable to a population and are typically unsuitable for statistical generalisation (Yin, 2009), although they may be employed in the exploration of likely variables and the explanation of relationships between them using deductive reasoning (Easton, 1995). Case studies are regarded as suitable for two types of generalisation (Mantere and Ketokivi, 2013; Tsang, 2014; Yin, 2009). The first, analytic or theoretical generalisation, involves generalisation to a theoretical proposition, enabling theory building. The second, falsification, involves the rejection of theoretical propositions based on case evidence and the consequential refinement of existing theory, for example changing its proposed relationships among concepts or its boundary conditions.
In this paper, we chose the former because of the variety of established theoretical approaches to research impact discussed previously. Our aim was to refine the THS model by studying it in the context of UK drug discovery and development. We adopted a systematic combining approach (Alvesson and Sköldberg, 1994; Dubois and Gadde, 2002), involving a systematic and iterative process of data analysis in which an emerging conceptual model is developed and refined, based on recurrent themes and patterns in the data. The final aim is to create a solid theoretical and empirical base, while at the same time strengthening the practical validation of the research by making the results relevant for relevant organisations.
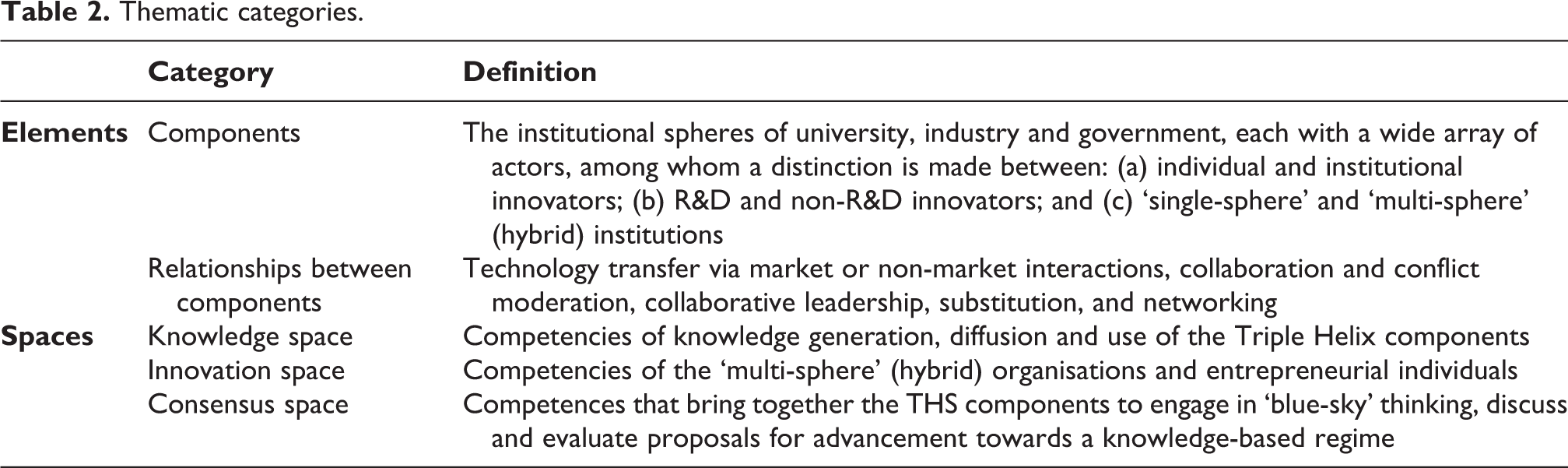
Our model was based on the THS (Ranga and Etzkowitz, 2013), defined as a set of three elements and embedded in three spaces, which we used as thematic categories (Table 2).
Thematic categories.
Using examples of impact listed in the REF2014 guidance instructions for sub-panel reviewers (HEFCE, 2012) as a set of a priori codes, we categorised impacts in the sequential order in which they are recorded in case studies. We then mapped these, as well as the actors cited in case studies, onto the framework in order to develop a perspective of the system. Data were extracted to specially designed spreadsheet pro-formas. We triangulated findings by drawing on secondary data from external sources referenced in case studies, specifically the institutional affiliations of co-authors of underpinning research papers and financial reports of academic spin-offs listed at Company House.
Descriptive summary
We find REF2014 drug discovery and development case studies to be skewed, variable and geographically distributed.
Geographically distributed
Our finalised shortlist of 268 drug discovery and development case studies was submitted by 63 of the 154 HEIs that took part in REF2014. All categories of UK universities were included in our shortlist, with the civic universities contributing the greatest number of case studies (88). Just less than half (46%) were submitted by institutions within the Oxford–Cambridge–London Golden Triangle, the world’s third largest technology cluster (Bell, 2017), while clusters in the Midlands, the North of England and the Edinburgh–Glasgow corridor were also well represented.
Variable
While case studies adhere to a common template, they show a significant amount of variability in data recorded. For example, research funding through Quality Related (QR) funding from funding councils, equity finance or the proceeds of technology sales are specified rarely or with a low level of specificity, while peer-reviewed funding received from UK research councils, the EU and charities, and to a lesser extent industry, are specified in greater detail. Analysis of institutional affiliation of underpinning research co-authors enables identification of actors not identified in the case study itself. This reinforces our view that deductive approaches to case study analysis should be treated with caution.
Skewed
Building on recent work on research evaluation (Hinrichs-Krapels and Grant, 2016), we assessed the impacts claimed in case studies according to three categories: effectiveness, efficiency and equity. We employed a definition of R&D effectiveness as the ‘ability of an R&D system to produce outputs with certain intended and desired qualities’ (for example, medical value to patients or commercial value) (Paul et al., 2010: 204). We also adopted Paul et al.’s (2010: 204) definition of R&D efficiency as the ‘ability of an R&D system to translate inputs (e.g. ideas, investments, effort) into defined outputs (e.g. internal milestones that represent resolved uncertainty for a given project or product launches)’. Finally, we defined R&D equity as the ability of an R&D system to translate inputs to produce outcomes among certain beneficiaries or addressing specific health needs.
We found that impacts cited in case studies were skewed towards R&D effectiveness and efficiency. Our shortlist contained 135 case studies (50%) that described the discovery or development of a drug or therapeutic approach underpinned by academic research, including 176 named small molecules and 69 biologics, the majority of which were novel. As an indication of the degree of ‘newness’ of these drugs, 14 were described as first-in-class, 1 was fast-tracked by the US Food and Drug Administration (FDA) and 1 was designated as a breakthrough therapy by the FDA. Our shortlist also included 142 (53%) studies that described the development of a technology which improved the efficiency of a stage of the drug discovery and development process. These included imaging or formulation technologies, catalysts, databases, prediction software and cognitive tests.
By contrast, consideration of R&D equity is relatively rare. For example, employment creation is typically cited by case studies in terms of number of jobs rather than where these jobs are created, suggesting a gap in the assessment of impact concerning inclusive growth (‘improvements in the social and economic wellbeing of communities that have structurally been denied access to resources, capabilities, and opportunities’) (George et al., 2012: 661). By comparing the number of jobs created by academic spin-offs (gathered from case studies and financial returns) to a geographical distribution of average worker earnings (Inclusive Growth Commission, 2016), we found a comparatively minor contribution to employment in poorer regions (Figure 2).
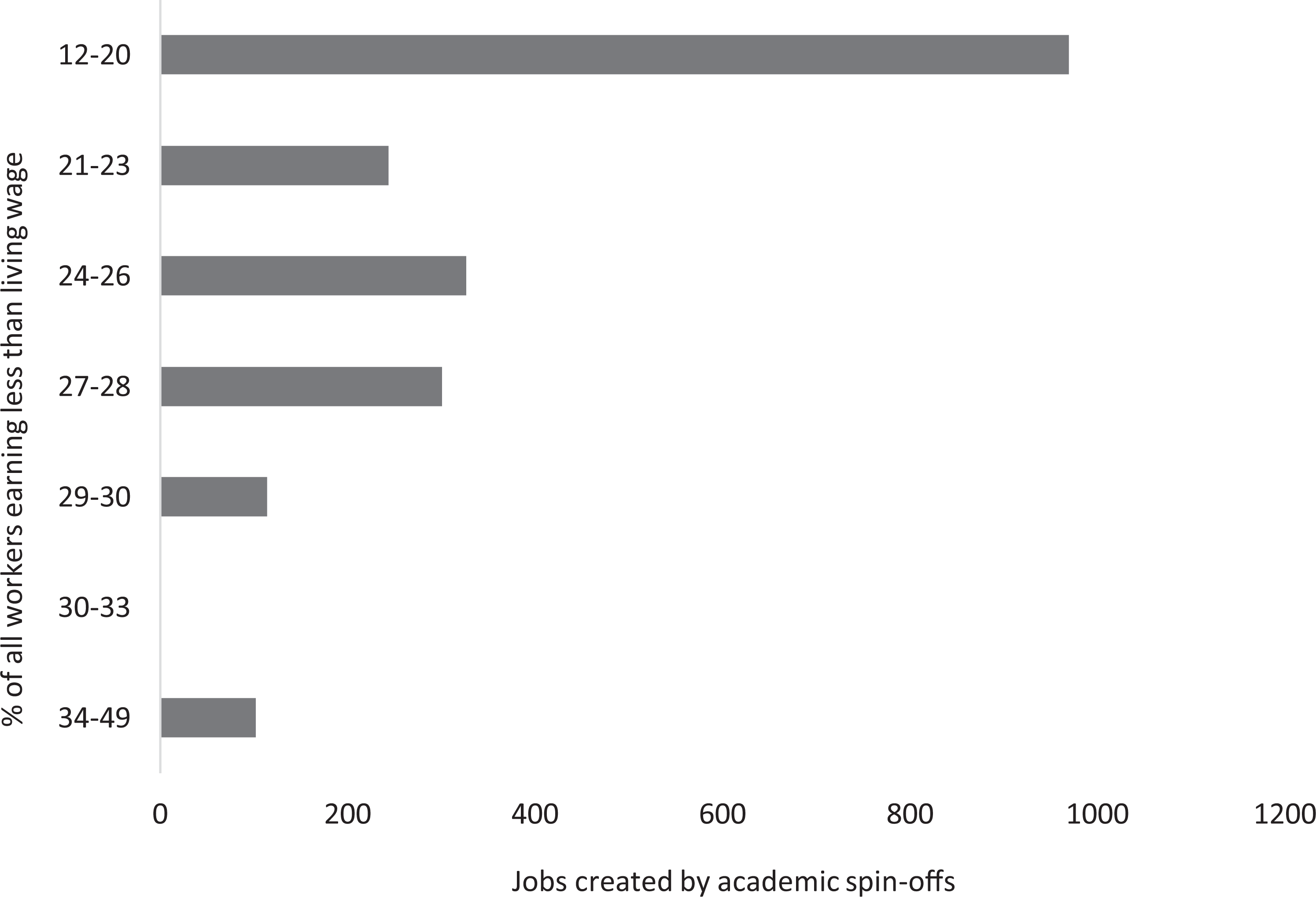
Distribution of jobs created by academic spin-offs relative to the percentage of workers earning less than the living wage.
As another illustration, we explored innovation directionality (the congruence of innovative products/services with a strategic set of collective priorities) (Weber and Rohracher, 2012) by collating the diseases targeted in case studies and categorising them using both the International Statistical Classification of Diseases and Related Health Problems (10th Revision) database and the World Health Organisation’s sustainable development goals (SDGs) (World Health Organization, 2015). SDGs are part of a triple bottom line approach to human wellbeing, a consequence of a view that current innovation systems drive medical R&D priority setting in the direction of greatest profit, but not of greatest need or of true medical benefit (Mazzucato, 2016; Sachs, 2012; UNSG, 2016). We found a preponderance of research directed towards non-communicable diseases, and in particular neoplasms (Figure 3). By contrast other SDGs, such as those relating to infectious diseases, mental health and reproductive health, appear to be underrepresented. Just 15 case studies (6%) describe work targeting orphan diseases (rare diseases affecting small populations). This is consistent with the finding of significant misalignments between health burden and R&D efforts at global and various national levels (Ràfols and Yegros, 2017).
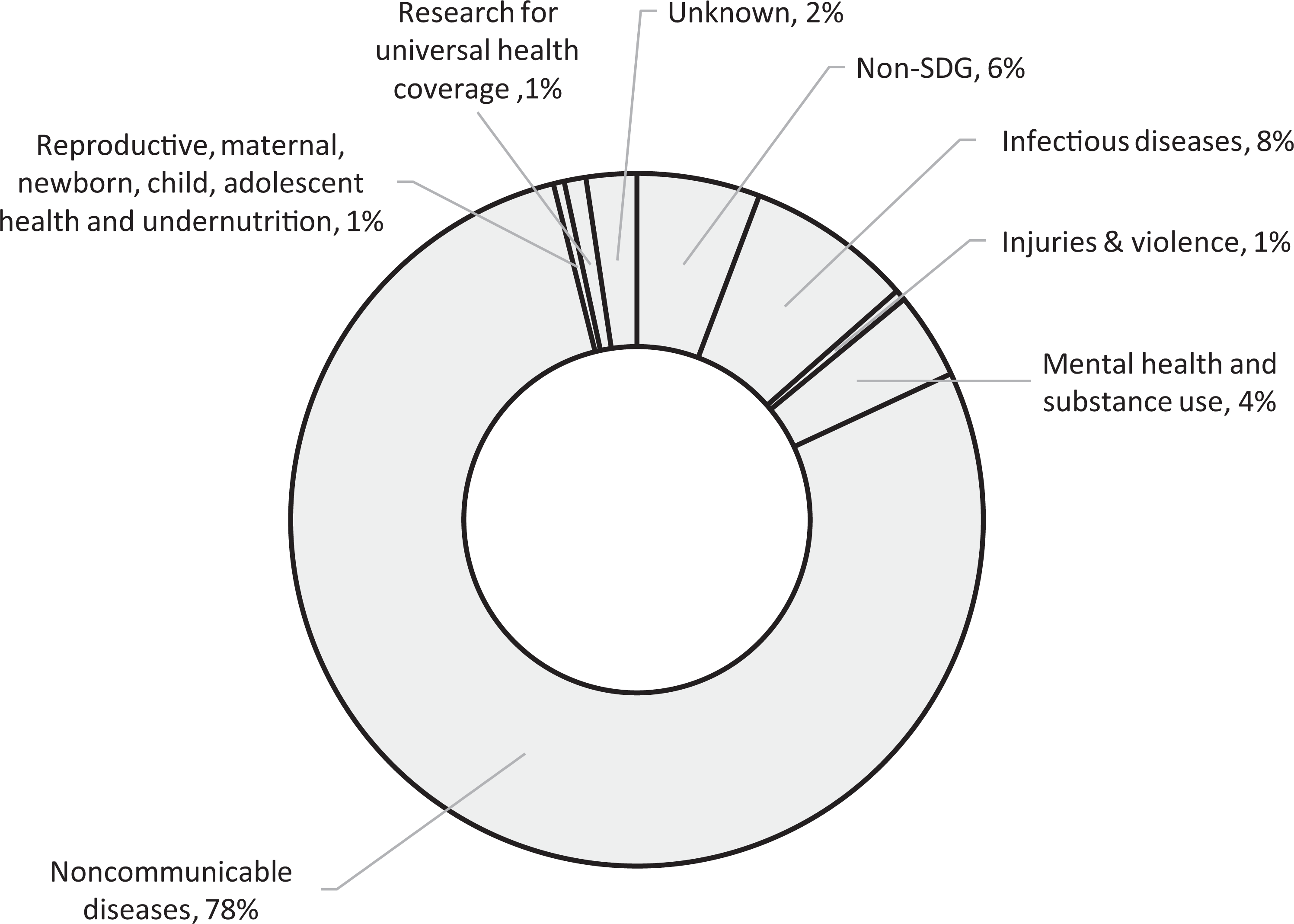
Direction of innovation. Note: n = number of case studies.
Stage 3: Integrative synthesis
Actors
Entrepreneurial academics
Entrepreneurial academics are the focus of each REF case study. These are academics who recognise that knowledge has simultaneous theoretical, practical and interdisciplinary implications (polyvalence) and who attend both to advancing academic knowledge and exploiting its practical and commercial value (Etzkowitz and Viale, 2010). Within the THS model, all lead academics in our case studies are considered as entrepreneurial, as they both published academic outputs and adopted a variety of styles to exploit knowledge value. These included venturing to form a going concern, venturing as a channel for private funding to advance research goals, industry engagement via joint research, contract research, consulting, sitting on advisory boards, membership of research consortia or simply via patent licencing.
R&D innovators
R&D innovators include teams or departments that are directly engaged in research activities leading to innovation. We assessed these by selecting a convenience sample of 100 of our 268 case study shortlist, and then determining in each case study the institutional affiliation of co-authors of papers cited as underpinning research (Steele, 2000). This method provided rigour and consistency, as case studies varied considerably in the degree to which collaborators were detailed. However, this method does underestimate the frequency of academic–industry collaborations in our case studies as these were not always accompanied by co-authored papers. We found that research activities were undertaken by five main groups: HEI-based academics, clinical researchers, industry research groups, public bodies and NGOs.
The predominant (n = 227) form of collaboration in academia was intra-institutional, including both informal interdepartmental collaboration and context-specific research centres. This is in line with the finding that entrepreneurial universities adopt hybrid forms involving both conventional hierarchical organisation with network-like mechanisms to encourage both internal and external engagement (Styhre and Lind, 2010). Interdepartmental collaborations with other UK HEIs are the next most common (n = 167), followed by collaboration with rest of the world (RoW) (n = 151) and European (n = 122) HEIs. RoW collaborations are predominantly with US HEIs, while there are very few collaborations with HEIs in emerging knowledge economies, such as China (n = 1), Singapore (n = 1), South Korea (n = 3), Brazil (n = 1), Russia (n = 2), Indonesia (n = 0) and India (n = 0). Despite the fact that many developing nations are now strategically investing to develop world-class universities to lead the shift towards knowledge economies, this had not translated into significant transnational academic collaborations in drug discovery and development up to 2013 (Altbach and Salmi, 2011; Salmi, 2009). This agrees with an earlier finding that global flows of biotechnology technological knowledge have largely bypassed developing countries (Buctuanon, 2001).
Clinical researchers represent the next most common collaborative partner. Collaborations with UK hospitals were the most common (n = 163) and appeared to be of two types – multi-centre clinical trials and smaller-scale research collaborations, often with hospitals affiliated with or local to the HEI and aimed at addressing local issues. Collaborations with European (n = 62) or RoW (n = 62) hospitals involved multi-centre clinical or field trials only.
Industry collaborations are the next most common type found. The extant literature on inter-organisational relations holds that it is primarily knowledge-intensive firms with sufficient absorptive capacity (the ability to recognise the value of new external information, assimilate it and apply it to commercial ends) that engage in collaborations (Inauen and Schenker-Wicki, 2011). Consistent with this, we found that collaborations occurred with five types of firm. First, pharmaceutical and biotechnology multinational corporations (MNCs) including GSK, Pfizer, Astra Zeneca and Novartis, undertook multiple collaborations in diverse projects. These included MNCs based in the UK (n = 26), Europe (n = 13) and the USA (n = 26). Second, multinational technology suppliers, including 3M, Phillips, Syngenta and Waters, predominantly undertook collaborations in single case studies. These included MNCs based in the UK (n = 8), Europe (n = 2) and the USA (n = 1). Third, new technology-based firms (NTBFs, independent firms less than 25 years old and active in high-technology industries) (Grilli and Murtinu, 2014) undertook single project collaborations specifically related to their technologies. These included ‘born global’ NTBFs (Oviatt and McDougall, 1994), based in Europe (n = 4) and the USA (n = 11) which sourced knowledge internationally, as well as UK-based NTBFs (n = 10). Fourth, university spin-offs founded by the focal academics of case studies (n = 25) maintained collaborative links with their home HEIs. Finally, a small number of collaborations with contract research organisations (CROs, a ‘paradigm of private science’ engaged in outsourced testing, in-house basic research and cross-firm alliances (Mirowski and Van Horn, 2005: 507)) based in the UK (n = 5), Europe (n = 1), the USA (n = 6) and Canada (n = 1) were noted.
Public bodies constituted the fourth most common R&D innovators. In the UK, these included public research institutions such as National Institute of Health Research (NIHR), Medical Research Council and Science, Technology & Facilities Council laboratories (n = 13), governmental departments (n = 2), governmental regulators such as National Institute of Biological Standards & Controls (n = 6) and public health institutions, such as the Blood Service (n = 5). In Europe (n = 29) and the RoW (n = 43), only public research and health institutions collaborated.
Finally, NGOs constitute the final R&D innovator we observed (UK n = 16, Europe n = 12, RoW n = 14). These were predominantly charitable research institutes, such as Cancer Research UK (n = 6), the Wellcome Trust (n = 2), Max Planck institutes (n = 4), the Babraham Institute (n = 2) and WiCell (n = 4). The only international NGO we encountered as co-cited author was the World Health Organisation (WHO, n = 2).
Non-R&D innovators
Non-R&D innovators include teams or departments whose function is not R&D and yet engage in innovation. In industry (e.g. production, sales, marketing, design, procurement, finance functions), these have been referred to as ‘neglected innovators’ and are often not supported by innovation policy (Hervas-Oliver et al., 2011). More broadly, non-R&D innovation may also be present in technology transfer, incubation activities, financing, creation and change of organisations. In our case studies, we identified two types of non-R&D innovator, although neither is common. First, there are clinicians who engage with focal academics in localised practice-based innovations. Such innovations are routinely neglected in studies concerning the UK’s National Health Service (NHS), which emphasise innovation outcomes from formal research projects (Savory, 2009). The second group comprises distribution companies who sign agreements with academics or spin-offs to sell and distribute new technologies.
Multi-sphere institutions
Multi-sphere institutions, or hybrids, operate at the intersection of the university, industry and government institutional spheres and synthesise elements of each sphere in their institutional design (Battilana and Lee, 2014). They encourage ‘boundary permeability’, or ease of movement across institutional boundaries, to improve effectiveness (Etzkowitz, 2012). They tend to have hierarchies with few levels and less centralised decision making to encourage flexibility and responsiveness. We identified five types: HEI supporting structures (technology transfer offices, seed capital providers), government or EU public funding organisations, academic spin-offs, venture capital firms/angel investors and non-profit organisations.
Technology transfer offices act as brokers between industry and academia, particularly in patent licensing and allocation to academic ventures (Winch and Courtney, 2007). However, no business development role is claimed for them in case studies, despite this being identified as critical to knowledge diffusion (McAdam and Marlow, 2008). More broadly, HEIs occasionally acted to provide early seed funding for academic ventures (e.g. Sheffield’s Fusion IP, Warwick Ventures, Imperial Innovations, the White Rose Seedcorn Fund).
Public funding bodies act as hybrids by unifying the institutional spheres of government and academia. Research Councils provide funding to support research excellence based on peer review but also knowledge transfer to generate economic growth, competitiveness, prosperity and wellbeing (e.g. Higher Education Innovation Funding) (HM Treasury et al., 2014). UK regional and devolved governments provide seed funding (e.g. the Scottish Co-Investment Fund, the North West Fund for Biomedical, Invest Northern Ireland) for the purposes of regional development and, while instances of this were isolated, the 2017 UK Industrial Strategy includes a greater emphasis on regional development (BEIS, 2017). Other UK public funding organisations we consider to be hybrids include the NIHR and Public Health England. The US National Institutes of Health constitutes the only foreign source of public funding for UK drug research of any consistency, while single examples of funding by the Irish and Swedish governments were identified.
Academic spin-offs, of which we identified 96, acted as multi-sphere institutions by channelling private finance to support research and tacit knowledge and technology from academia to industry to produce commercialisable innovation. Based on annual returns held by Companies House, these displayed a high degree of resilience, with just 11 ceasing trading up to 2017. Academic spin-offs appeared to be of two types, those intended to operate as going concerns and those founded with the intention that they would be acquired at some point. They included contract research organisations, firms with in-house drug discovery programmes and technology providers. Of the 96 spin-offs we identified, 56 had remained operational by 2017, while 29 had been acquired or had merged. Three could be regarded as scale-ups (average annualised growth in profit or loss greater than 20 per cent per annum over a 3 year period, and with more than 10 employees at the beginning of the observation period) (Coutu, 2014). This finding is consistent with the low representation of life sciences companies (3) in the top 50 UK fast-growth science-based companies (Royal Society, 2014). This suggests that there may be impediments to scale-up in the life sciences sector that are not experienced elsewhere.
Venture capital firms and angel investors linked academic entrepreneurs and markets by being the primary funding source of spin-offs after seed rounds (Wright et al., 2006). These mainly consisted of independent VC firms. We also identified instances of corporate venture capital via MNC subsidiaries.
Non-profit organisations may also be considered an example of multi-sphere institutions, linking academia to specific social needs which may not always overlap with political or industrial needs (Smith et al., 2011). We identified a diverse landscape of charities in our case studies, including large medical research charities (the Wellcome Trust, Cancer Research UK, the British Heart Foundation), small disease-specific charities (e.g. Lupus UK, the Alzheimer’s Disease Society, the Migraine Trust) and a small number of non-UK-based charities (e.g. the Bill and Melinda Gates Foundation, the Croucher Foundation Hong Kong, the Leukemia & Lymphoma Society). We also identified cases of the emergence of new non-profits via venture philanthropy (ReverseRett) and academic social entrepreneurship (Kidscan, Lhasa Ltd). The roles of non-profits in innovation systems have been largely neglected in academic research, although Canadian non-profits were found to act as both institutional enablers or institutional balancers, increasing or decreasing respectively a firm’s ability to innovate by shaping the networks and markets in which it participates (Dalziel, 2007). We found evidence only for an enabling role in our case studies, and identified four mechanisms by which it is undertaken within the drug discovery and development THS: provision of research funding through peer-reviewed grants, co-opting academics onto regulatory or governmental work groups, issuing of clinical treatment guidelines, and advocacy on behalf of a patient group. This is not to say that a balancing (or another as yet unidentified) role is not in operation, but that further research would be necessary to identify it.
Innovation organisers
Innovation organisers are institutions or persons occupying a key institutional position and coordinating a mix of top-down and bottom-up processes and innovation stakeholders from different organisational backgrounds to promote economic and social development and ensure agreement and support for its realisation. We identified four such actors in our case studies. The EU provided funding, networking support and strategic direction for drug discovery and innovation to foster research excellence, economic growth, regional development and social cohesion via transnational collaboration (Maassen and Stensaker, 2011). The predominant EU vehicles identified in case studies were the Framework Programmes for Research and Technological Development and the Innovative Medicines Initiative, while other programmes were also represented (PEACE II: ALFA; the European and Developing Countries Clinical Trials Partnership). The WHO provided research funding, administered procurement programmes and treatment guidelines, and fostered consensus-building to adjust national policies in areas including malaria, trauma, polio and neglected tropical diseases. The UK central government has traditionally adhered to a laissez-faire approach, funding academic research to secure societal benefits but ceding authority with regard to work allocation to the academic community under the Haldane principle (Hughes, 2011; Olssen and Peters, 2005). However, our case studies do show isolated examples of more supportive and directive interventions (Enright, 2001), including the setting of strategic direction for innovation and the establishment of funding mechanisms to support innovation. Finally, pharma and biotech MNCs acted as innovation organisers by funding research, coordinating clinical trials and bringing innovative treatments to market.
Civil society
While the Triple Helix approach has been instrumental in exploring the changing relations between academia, industry and government, it nonetheless maintains the notion of a ‘protected space’ for academia from society (Flink and Kaldewey, 2018), and therefore envisages no active role for society within the innovation system. However, as contextual knowledge becomes more important, it is argued that society’s role can no longer be neglected (Delanty, 1998). For example, patient participation (‘patient involvement in decision-making in advisory boards or committees at macro and meso levels of health care, but also […] involvement at micro level in relation to decision-making on their own care and treatment’) (World Health Organization, 2013: 8) has been identified by the WHO as a high priority. In our case studies we identified three societal actors: NGOs (discussed above), the media and patient groups. The media’s role as described in case studies was invariably passive, reporting innovative developments in healthcare. However, we did identify two mechanisms by which patient groups actively influence the innovation system, in addition to simply receiving a new drug. The first, at a meso level, is the previously-mentioned case of a parent of a child with Rett syndrome who engaged in venture philanthropy to fund research once reversibility was established. The second, at a macro level, is the political advocacy role patient groups played in securing regulatory approval and access to certain treatments.
Relationships among components
Technology transfer
Technology transfer was the core activity taking place between entrepreneurial academics, R&D and non-R&D innovators, innovation organisers and academic spin-offs within the knowledge space of the system. This was achieved through five main channels: knowledge commercialisation, academic engagement with non-academic organisations, knowledge spillovers, graduate and researcher mobility, and contributions to standards or guidelines.
Knowledge commercialisation, including patent allocation and licensing, academic venturing, merger and acquisition and joint venturing, has been widely explored in the literature, although it is often over-emphasised in policy discourse (Hughes, 2011). Patent allocation from HEIs to spin-offs and licensing to external firms were common in case studies. The acquisition of a spin-off or an R&D programme, or subsequent mergers or acquisitions of firms without direct involvement of the focal academic, were also regularly identified. Isolated examples of joint ventures involving a spin-off and an MNC or two MNCs were observed.
Academic engagement with non-academic organisations has more recently emerged as a topic for attention in the literature (Perkmann et al., 2013). There are various types, including collaborative research, contract research, co-authoring, co-patenting, consulting, secondment, informal advice, participation in work groups and advising parliamentary sub-committees. Previous research has argued that such relational forms of involvement are seen as more relevant by firms than by non-relational forms (patenting and licensing) (Perkmann et al., 2011). This is consistent with our findings for all types of R&D innovators and for non-R&D innovator clinicians. However, non-R&D innovator distribution firms engaged only in patent licensing. We also found that relational activities were generally initiated by the firm in the case of pharmaceutical and biological drugs, but often by the academic for other technologies, indicating significant differences in open innovation strategies and capabilities within these segments (Fontana et al., 2006).
Knowledge spillovers are non-market-based interactions in which firms benefit from publicly-accessible research findings rather than through their own R&D (Davies, 2008; Nelson, 2009). In our case studies we identified a number of instances of firms undertaking their own research programmes once a new therapeutic avenue or proof of concept had been established by the focal academic, generating R&D investment and employment (though both are difficult to measure) as well as new drug candidates.
Graduate and researcher mobility include the employment of an academic or student by a firm, and is regarded as a channel by which tacit knowledge moves to industry (Wright et al., 2008). Instances of academics leaving HEIs to take up full-time employment were rare in our case studies, but joint appointments (retaining an academic position while taking up a strategic advisory position in an MNC or a board appointment in a spin-off) were relatively common. Graduate mobility was more common, though was not often emphasised.
Finally, contributing to standards or guidelines was a common method by which technology was transferred both to the clinic and transnationally. This was usually under the auspices of an NGO or regulatory agency. We are aware of no research concerning this channel of technology transfer in the literature.
Substitution
Substitution arises when a gap introduced by a weakness in one institutional sphere is filled by another sphere. A known example is the gap between public or charitable research funding and venture capital funding experienced by academic ventures (Nightingale et al., 2009). We found examples of where HEI, regional/devolved government (including regional development authorities), central government and non-profit seed funding programmes had emerged to mitigate this. We also identified isolated instances of other activities that we regard as substitutions. The first was the founding of charities either by academics or patient groups to fund research for a particular cause not already served by existing charities (Kidscan, ReverseRett, Lhasa Ltd). The second was the involvement of corporate venture capital (CVC) in a number of case studies. CVC has emerged relatively recently, designed to overcome inefficiencies in the life sciences ecosystem (Greene et al., 2010). The third was a single case of an academic research group acting as the de facto research group of a firm (Funxional Therapeutics Ltd).
Networking
Networking between entrepreneurial academics, R&D innovators and multi-sphere institutions can occur in both formal and informal structures and at sub-regional, regional, national and international levels. We found that these networks tended to be of two types in our case studies. Within the knowledge space, networks (e.g. the Centre for Applied Pharmacokinetic Research; the Simcyp Consortium; the Bioconversion–Chemistry–Engineering Interface Programme; Lhasa; Division of Transduction Therapy; Neuroallianz), are directly concerned with pre-competitive applied research and innovation. EU Framework Programmes generally involve the establishment of networks of this type. Within the consensus space, networks (e.g. the Structural Genomics Consortium; the Human Toxicology Project Consortium; the Wellcome Trust Case Control Consortium; the Tay-Sachs Gene Therapy Consortium) engage in basic or user-inspired basic research and ‘blue-sky’ thinking to guide advancement in a specific field or context. A second type of consensus space network is that which is led by patient groups and emerges for the purpose of political advocacy. A third type, innovation space networks, are known to exist although are not discussed in our case studies.
Collaborative leadership
Collaborative leadership is undertaken by innovation organisers to connect people from different sectors and institutional spheres, to bridge gaps, generate consensus and balance conflicts of interest. They can integrate skills and enable people to develop competencies according to specific challenges, foster changes in thinking and practical implementation, and create new opportunities for knowledge exchange. The EU undertakes collaborative leadership largely through public–private partnerships within the Framework Programmes. The WHO does so through research, procurement and advocacy programmes designed to tackle specific social challenges. The UK central government plays a less interventionist role, limited to setting strategic innovation direction and establishing funding mechanisms. Pharma and biotech MNCs collaboratively lead in large-scale clinical trials.
Discussion
In this discussion, we illustrate how REF case studies may be used both for theory development and as a means to understand or advance insight into the UK drug discovery and development system.
Refinement of the Triple Helix system model
Our first contribution is a refinement of the THS model through a change in proposed relationships between concepts (Table 3). The THS groups actors within three main components: academia, industry and government. The roles of other actors in the system, such as non-profit organisations, the media, patient groups, supranational organisations and actors based in other countries are not well developed or are neglected (Petruzzelli et al., 2010). Our refinement aims to address this weakness and is based on the Quadruple Helix approach (Carayannis and Campbell, 2012), a development of the Triple Helix. This sees economic, political and knowledge systems interacting to form an innovation system (Carayannis and Rakhmatullin, 2014).
Triple Helix system of UK drug discovery and development.

Our refinement consists of the identification of civil society and trans- and supranational actors and the mechanisms by which they influence the Quadruple Helix, based on our empirical findings. Patients’ groups engage in substitution through venture philanthropy, and networking for the purposes of political advocacy. NGOs engage directly in technology transfer via co-authorships and the establishment of guidelines and workgroups, and indirectly through research funding and providing seed capital in the knowledge system. They also engage in networking for political advocacy, and substitution to address gaps in public or private research funding. While the media features prominently in case studies, no claims are made for its influence on any actor and its role appears limited to reporting innovative developments. We find no evidence of other mechanisms of influence by civil society actors, such as crowd funding or citizen science. Supranational organisations (the EU and WHO) engage in collaborative leadership and technology transfer (establishing research, procurement and advocacy programmes designed to tackle specific social challenges) and networking, particularly in the knowledge space.
Understanding the changing landscape of UK drug discovery and development
Our second and third contributions are to practice and concern advances in insight into the UK drug discovery and development system. The first of these concerns geographical clusters (geographically proximate groups of interconnected companies, suppliers, service providers and associated institutions in a particular field, linked by commonalities and complementarities) (Porter, 2003). Clustering is generally believed to enhance firm performance through pooling of human capital, proximity to non-traded inputs and specialised goods, easy access to markets and reduced cost associated with knowledge spillovers (Audretsch and Dohse, 2007; Audretsch and Lehmann, 2006). However, the role of geographical proximity in knowledge spillovers from universities is problematic and its importance is likely to be overstated (Davies, 2008). While facilitating knowledge transfer, geographical proximity is neither a necessary nor a sufficient condition for it to take place and indeed may have a negative impact due to technological lock-in (Boschma, 2005).
Based on an analysis of co-citation data of underpinning research and on impact case studies themselves, we found that strategic alliances, network linkages and knowledge spillovers appeared to be much more important for impact generation than geographical clusters (Zaheer and George, 2004). Indeed, geographical proximity was significant only in interactions between an academic’s HEI and a university spin-off, and between an academic’s HEI and a research hospital. This is significant because both UK policy and Big Pharma strategies have favoured geographical clustering and co-location with research-intensive HEIs in recent years (Abramovsky et al., 2007; Smith and Bagchi-Sen, 2006). Our finding suggests that, despite a strategic emphasis on co-location, firms must remain open to the wider external environment, for example through strong searching, screening and signalling processes (Fontana et al., 2006).
Our final contribution concerns transnational academic capitalism, defined as the ‘integration of [a] transnational dimension into teaching, research and service in a way that enhances transnational integration between universities and globalizing knowledge capitalism, and increases academics’ and universities’ possibilities to diversify their external funding sources transnationally’ (Kauppinen, 2012: 544). We note a strong national and Anglo-American bias in linkages between practitioners and UK academics, with few or no links to emerging knowledge economies such as China, Singapore, South Korea, Brazil, Russia, Indonesia or India. It has been suggested elsewhere that this is due to cultural and linguistic preferences and geographical constraints imposed by person-embodied exchanges and transfers of tacit knowledge (Arundel and Geuna, 2004; Patel and Pavitt, 2000). This suggests that most firms engaging in collaboration, with the possible exceptions of born-global NTBFs and MNCs, do not leverage academics’ cosmopolitan networks of colleagues established through social patterns of collaboration, collegiality and competition (Murray, 2004). If this is the case, it suggests the need for an innovation organiser to encourage link formation between UK academics and emerging economies, similar to the EU’s role in UK–European SME links and the WHO in UK–African links.
Conclusion
REF2014 case studies offer a new and important perspective on the UK’s innovation system, allowing the identification of actors and longitudinal relationships within the system. We have illustrated the utility of REF case studies in this paper by developing a novel methodology to enable the interrogation of case studies within a context of application rather than a field of research. This has enabled us both to make contributions in theory building through refinement of the Triple Helix system and to advance insight into UK drug discovery and development. Future iterations of the REF, currently envisaged to be undertaken in 2021, 2028 and beyond, offer the opportunity to undertake longitudinal studies of the drug discovery and development system as it doubtless undergoes further change following the UK withdrawal from the EU and the continued policy efforts to position the UK as a global science superpower that benefits every corner of the country.
Footnotes
Acknowledgements
The authors would like to acknowledge the invaluable support of Professor Ian Kitchen and Dr David Fox in the study evaluation phase and Professor Jackie Hunter, Professor Jamie Coleman, Professor David Webb, Dr Malcolm Skingle and Dr Dagan Lonsdale in the locating studies phase.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix 1
Keywords & search strings.
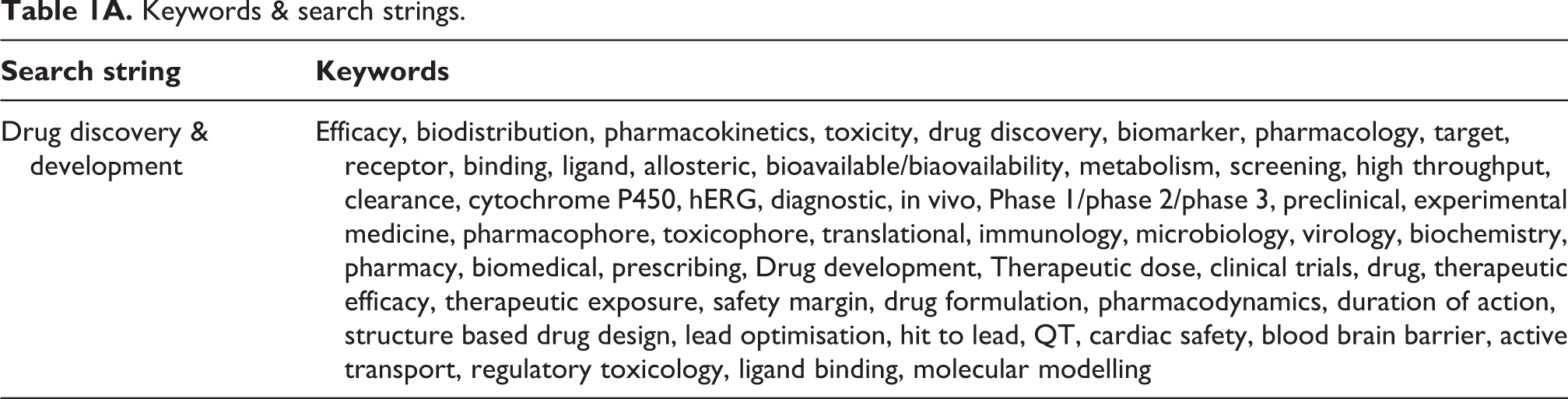
|
|
|
|---|---|
| Drug discovery & development | Efficacy, biodistribution, pharmacokinetics, toxicity, drug discovery, biomarker, pharmacology, target, receptor, binding, ligand, allosteric, bioavailable/biaovailability, metabolism, screening, high throughput, clearance, cytochrome P450, hERG, diagnostic, in vivo, Phase 1/phase 2/phase 3, preclinical, experimental medicine, pharmacophore, toxicophore, translational, immunology, microbiology, virology, biochemistry, pharmacy, biomedical, prescribing, Drug development, Therapeutic dose, clinical trials, drug, therapeutic efficacy, therapeutic exposure, safety margin, drug formulation, pharmacodynamics, duration of action, structure based drug design, lead optimisation, hit to lead, QT, cardiac safety, blood brain barrier, active transport, regulatory toxicology, ligand binding, molecular modelling |
