Abstract
There are significant gaps in our understanding of the oral biology of the “Vulnerable” Babyrousa spp. of Indonesia. A photographic and computed tomography (CT) scanning study was carried out on 62 maxillary incisors and 114 mandibular incisors of 8 adult male Babyrousa babyrussa skulls and 14 skulls of Babyrousa celebensis. An additional group comprised 4 adult males and one subadult male, Babyrousa celebensis. The 4 maxillary incisors (102, 101, 201, and 202) and the 4 mesial mandibular incisors (302, 301, 401, and 402) were open-rooted, dentin structures. The dentine-composed lateral mandibular incisors (303 and 403) had closed roots. Maxillary incisor tooth length, and tooth root length, depth, and breadth were measured. Measurements were also made of mandibular incisor tooth and root lengths, and in 302, 301, 401, 402, the tooth depth and breadth at the apex and at 50% of the incisor length. The pulp depth and breadth of these teeth in both species decreased from the apex along the length of the tooth, while the secondary dentin depth increased. Primary dentine depth measured at the apex of the mandibular teeth did not change. Tooth wear patterns on the lingual surfaces of the incisors were attributed to mandibular yaw movements during mastication.
Introduction
The babirusa (genus Babyrousa) are suids endemic to the Indonesian island of Sulawesi and a number of smaller islands to the east, including the Sula Islands and Buru.1,2 Modern computed tomography (CT) scanning techniques have enabled recent descriptions of the roots of the premolar and molar teeth of the babirusa to be made.3,4 These descriptions have increased understanding of the oral food-processing capabilities of the babirusa and helped explain the resulting tooth wear.5,6 Other studies have drawn attention to the food-gathering behavior of the babirusa.7–12 It was interpreted that the food gathering behavior and food processing were largely carried out by the incisor teeth. These teeth have also been thought to be used for collecting leaves and other plant material for nest building. 13
There are 4 permanent incisors in the maxilla and 6 incisors in the mandible of the babirusa. 14 The modified Triadan system of veterinary dental nomenclature, 15 was adopted for this study to provide descriptions of dental location and orientation (Figure 1A and B).3,16 In 1899, the maxillary incisors and the 4 mesial mandibular incisors of the babirusa were first reported to be “completely hypselodont” (open and ever-growing roots), with only the mandibular incisors (303 and 403) closing their roots. 14 The initial enamel coating of the incisors is ephemeral, and only in very exceptional cases were small remains of enamel found on incisors. 14 The permanent incisors are therefore composed of constantly renewed dentine.
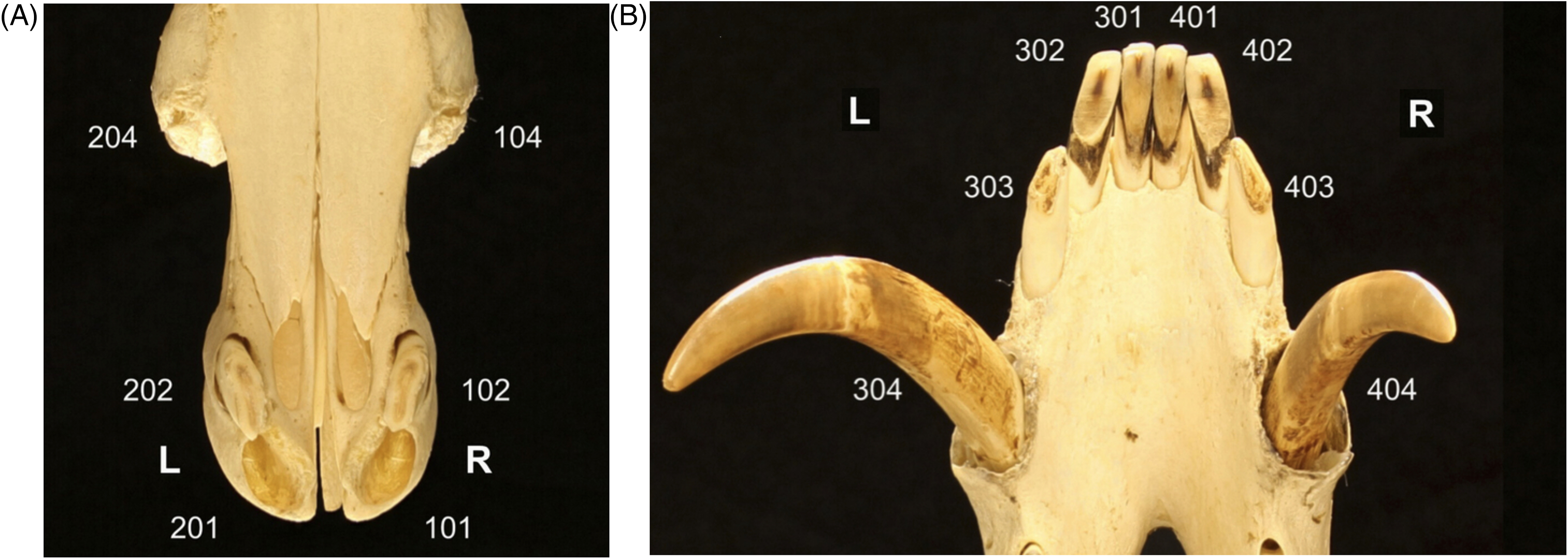
Illustration of the Modified Triadan System of Tooth Numbering Applied to (A) the Maxillary and (B) the Mandibular Incisors and Canine Teeth of a Male Babirusa (Babyrousa celebensis) (NMS.Z.2002.210.001).
Apart from these investigations carried out over 100 years ago, 14 which focused mainly on the anatomy of the deciduous teeth of the babirusa, very little information concerning the permanent incisors of the babirusa has been published. The opportunity, presented by recent CT studies of the skulls of the male babirusa,3,4 was therefore taken to examine the maxillary and mandibular incisor teeth of these specimens.
Materials and Methods
Twenty-seven babirusa (Babyrousa spp.) skulls, from 8 museum collectionsa-h (Table 1), were studied. All specimens were photographed, including close-ups of the occlusal surfaces of the teeth. Twenty-two babirusa skulls were scanned by CT (Table 1). The CT group comprised 7 adult male Babyrousa babyrussa skulls recorded as coming from Buru and one adult male Babyrousa babyrussa skull morphologically identified as coming either from Buru or the Sula Islands. The group also contained 11 adult male Babyrousa celebensis from Sulawesi, together with 3 adult male Babyrousa celebensis skulls derived from zoological collections. The group of photographed skulls comprised 4 adult male Babyrousa celebensis from Sulawesi and one subadult male Babyrousa celebensis individuals from zoological collections (Table 1). All were examined with respect to incisor tooth and incisor tooth alveolar morphology. The number of babirusa teeth CT-scanned and available for evaluation were 62 maxillary incisors and 114 mandibular incisors (n = 176).
Data From the Babyrousa babyrussa and Babyrousa celebensis Skulls.
To enable anatomical clarity in the description of observations, the dental anatomical nomenclature of the babirusa incisors (Figure 1) was derived from that published for Suidae. 17 The maxillary incisors 101 and 201 have one main cusp, the paracone with the parapreconule adjacent to it (Figure 2A and B). The maxillary incisors 102 and 202 have one main cusp, the paracone. The mandibular incisors have one cusp from which 3 ridges radiate: the precristid, endocristid, and postcristid, as shown on 301 (Figure 2C and D). The body of the incisor tooth consists almost entirely of dentine, which surrounds a central pulp cavity and one root canal.
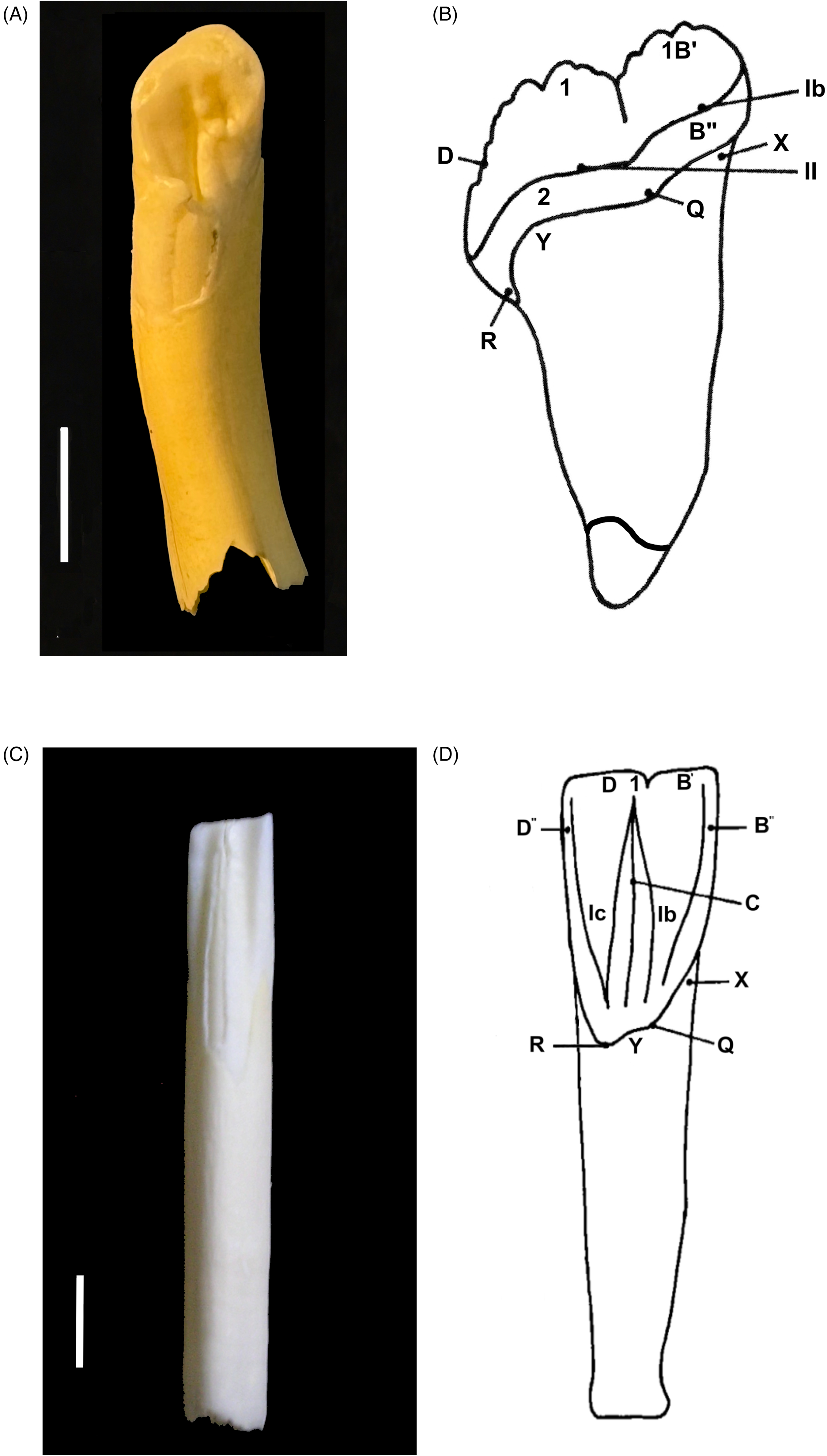
Images From a 13-Month-Old Male Babirusa (Babyrousa celebensis) (NMS.Z.1993.159.004). (A) Palatal View of Maxillary Incisor 101. (B) Maxillary Incisor Tooth. 17 Annotations: 1—Paracone; 1B′ —Parapreconule; 2—Lingual Cingulum and/or Protocone; B′′ —prestyle; D —Posterista; X—Preanticline; Y—Endoanticline; Q—Presyncline; R—Endosuncline; Ib—(Proto) Prefossa; and ll—Basal Fossa. (C) Lingual View of Mandibular Incisor 301. (D) Mandibular Incisor Tooth. 17 Annotations: 1—Protoconid; B′ —Preconulid; B′′ —Prestylid; C—Endocristid; D—Postcristid; D′—Postconulid; D′′ —Poststylid; X—Preanticlinid; Y—Endoanticlinid; Z—Postanticlinid; Q—Presynclinis; R—Endosynclinid; Ib—Prefossid; and Ic—Endofossid. Bar=5 mm.
Several of the skulls were found to have missing maxillary and mandibular teeth. Advantage was taken of these to examine the maxillary incisor alveolae (Figure 3A).
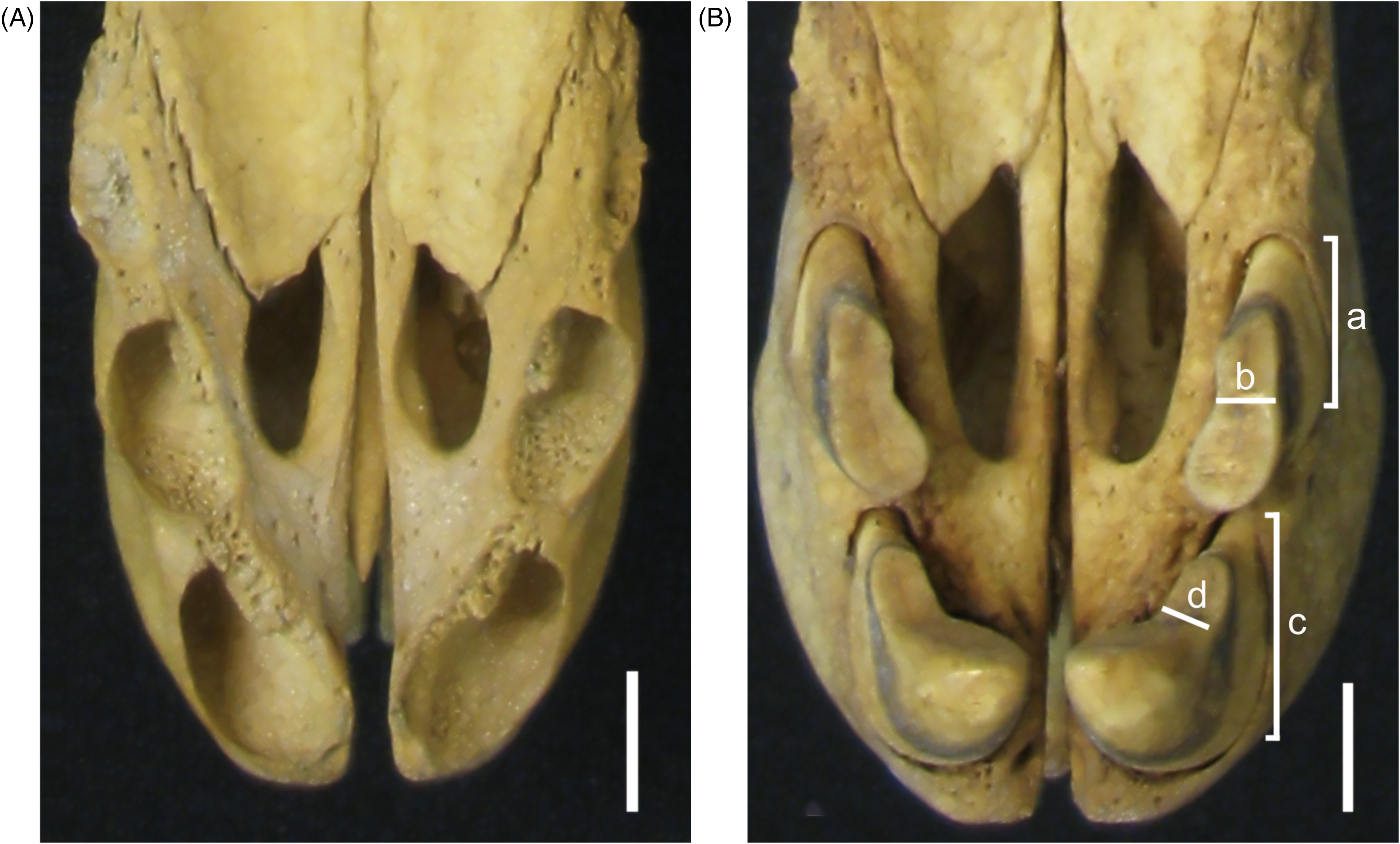
(A) Palatal Photographic View of the Maxillary Incisor Alveolae of a Young Male Babirusa (Babyrousa celebensis) (MZB.15197). (B) Palatal Photographic View of the Maxillary Incisor Teeth Situated in Their Alveolae of an Adult Male Babirusa (Babyrousa celebensis) (MZB. 6901). Annotations: a—Lateral Width of 102; b—Lateral Thickness of 102; c—Lateral Width of 101; and d—Lateral Thickness of 101. Note the Rostro-Mesial Orientation of the Teeth. Bar =10 mm.
Due to curatorial and geographical reasons (Table 1), the skulls were CT-scanned at 4 different institutions using 4 different CT scanner models. All 4 CT scanners were helical multislice scanners with very similar scan settingsi-l, with the following common settings: pitch of 0.53 to 1, 0.4 to 0.7 mm slice width, 0.2 to 0.625 mm slice interval, bone reconstruction kernel, 100 to 193 mm display field diameter, 5122 image matrix, 111 to 200 mAs, and 120 to 140 kVp.
Maxillary 101 and 201 tooth length was measured along the rostral curvature from the crown to the open apex (the extremity) of the tooth root. Maxillary 101 and 201 tooth root length was measured along the rostral curvature from the alveolar bone ridge surrounding the tooth to the open apex of the tooth root. Maxillary 102 and 202 tooth length was measured linearly from the crown to the open apex of the tooth root. Maxillary 102 and 202 tooth root length was measured linearly from the alveolar bone ridge surrounding the tooth to the open apex of the tooth root. Maxillary tooth lateral size (depth) was measured on the buccal side across the mid length of the tooth (Figure 3B). Maxillary tooth thickness (breadth) was measured at right angles to this across the mid length of the tooth (Figure 3B).
The lengths of the rod-like mandibular teeth 302, 301, 401, and 402 were measured along their ventral surfaces from the crown to the open apex of the root. The lengths of mandibular teeth 303 and 403 were measured from the crown to the closed apex of the root. Mandibular tooth root length was measured from the alveolar bone ridge surrounding the tooth to the apex of the tooth root. Mandibular tooth sizes (depth and breadth) were measured on CT two-dimensional (2D) cross-sections of the teeth at their apices and across the mid-length of the tooth. Because the cross-sections of the mandibular teeth are ovoid in shape, 5 sets of depth and breadth measurements were made of the internal structure of the mandibular teeth 302, 301, 401, and 402 at each apex, 25%, 50%, 75%, and ∼95% of the incisor tooth length.
The occlusal surfaces of the maxillary and mandibular incisors, represented by areas of tooth wear, were not measured due to the imprecision of their variable three-dimensional complexity.
Statistical analysis of variancem was undertaken on the incisor teeth of the babirusa.
Results
The morphology of the maxillary incisor teeth of Babyrousa babyrussa is very similar to that of Babyrousa celebensis (Figure 4A and B). The maxillary 101 and 201 teeth are relatively short (Table 2) with roots that curve dorso-ventrally. They grow with a mesial twist out of the incisive bone at an angle of ∼20o to 30o to the midline (Figure 3A). A valley runs caudally along their mesial surface (Figure 3B), and a shallower valley is present on the buccal side.
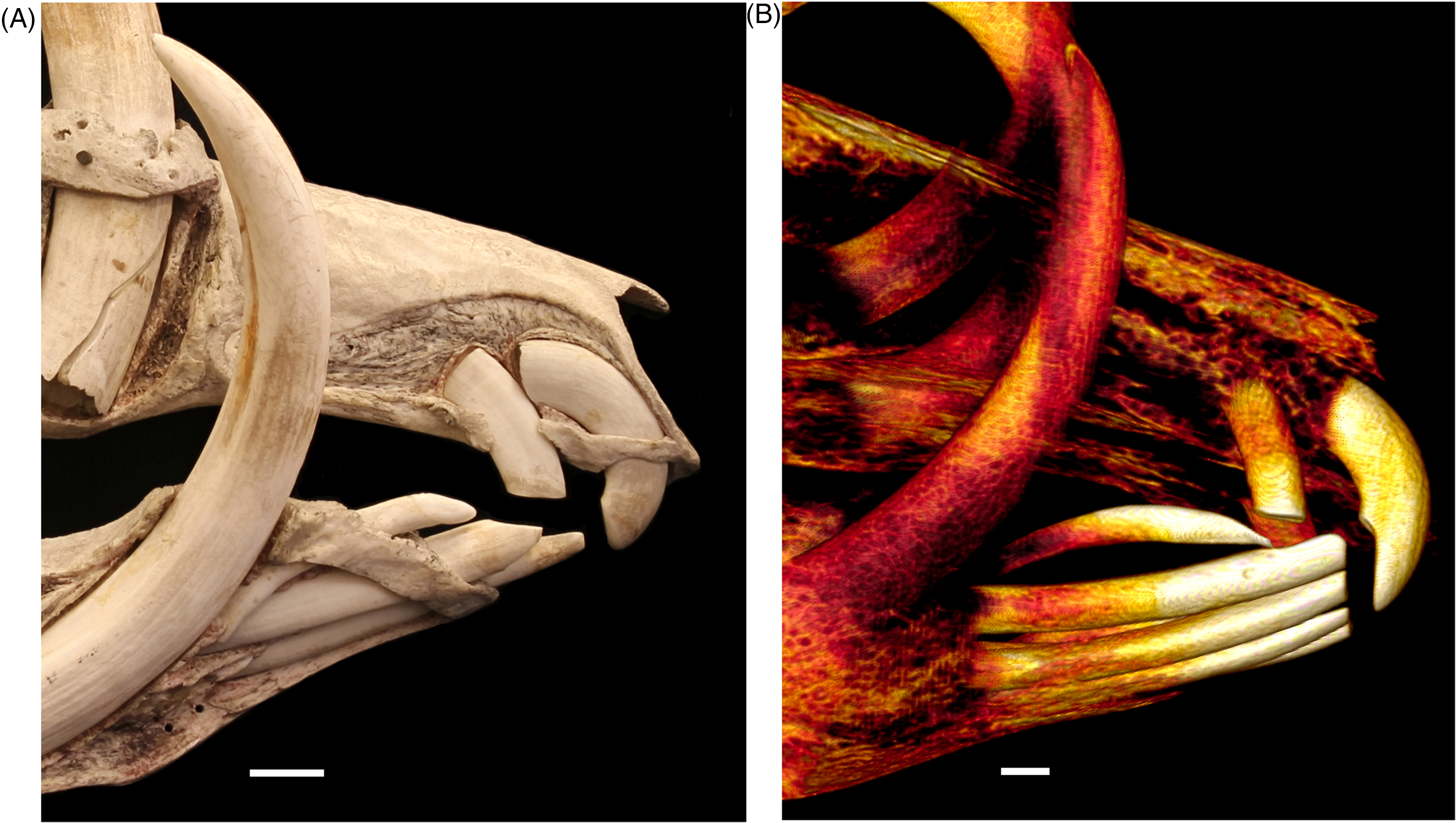
(A) Lateral Right Photographic View of the Exteriorized Maxillary Teeth 102 [Caudal] and 101 [Rostral] and Mandibular Incisor Teeth 403, 402, and 401 of an Adult Male Babirusa (Babyrousa celebensis) (OUM.05266). (B) Lateral Right Computed Tomography (CT) View of the Maxillary Teeth 102 [Caudal] and 101 [Rostral] and Mandibular Incisor Teeth 403, 402, 401, 301, and 302 of an Adult Male Babirusa (Babyrousa babyrussa) (SN.35645). Bar =10mm.
Measurements From the Babyrousa babyrussa and Babyrousa celebensis Maxillary Incisor Teeth.

Abbreviations: Av, average; Std, standard.
* P<.05; **P < .01; ***P < .001.
The occlusal surfaces of these teeth are derived in part from the crown of the tooth orientated caudally (Figures 2A, B and 4A), and partly with tooth wear from the palatal aspect of the shaft of the tooth orientated caudo-ventrally (Figures 3B and 4B). The open tooth apices (Figure 5A and B) of these teeth are orientated caudally into the incisor bone and lie rostral, medial, and dorsal to those of the maxillary 102 and 202 incisor teeth (Figure 4A and B). The latter teeth also have open roots (Figure 5B) and are shorter (P < .001), not as deep (P < .05), and thinner mesio-laterally (P < .001) than the former, with shorter tooth roots (P < .001) (Table 2). They are slightly curved dorso-ventrally (Figure 4A and B) and have a valley running caudally along their mesial surface (Figure 3B). They grow out of the incisive bone at an angle of ∼10° to 20° to the midline (Figure 3A). The occlusal surface occupies the rostral end of the tooth and is orientated ventrally (Figures 3B and 4A, B).
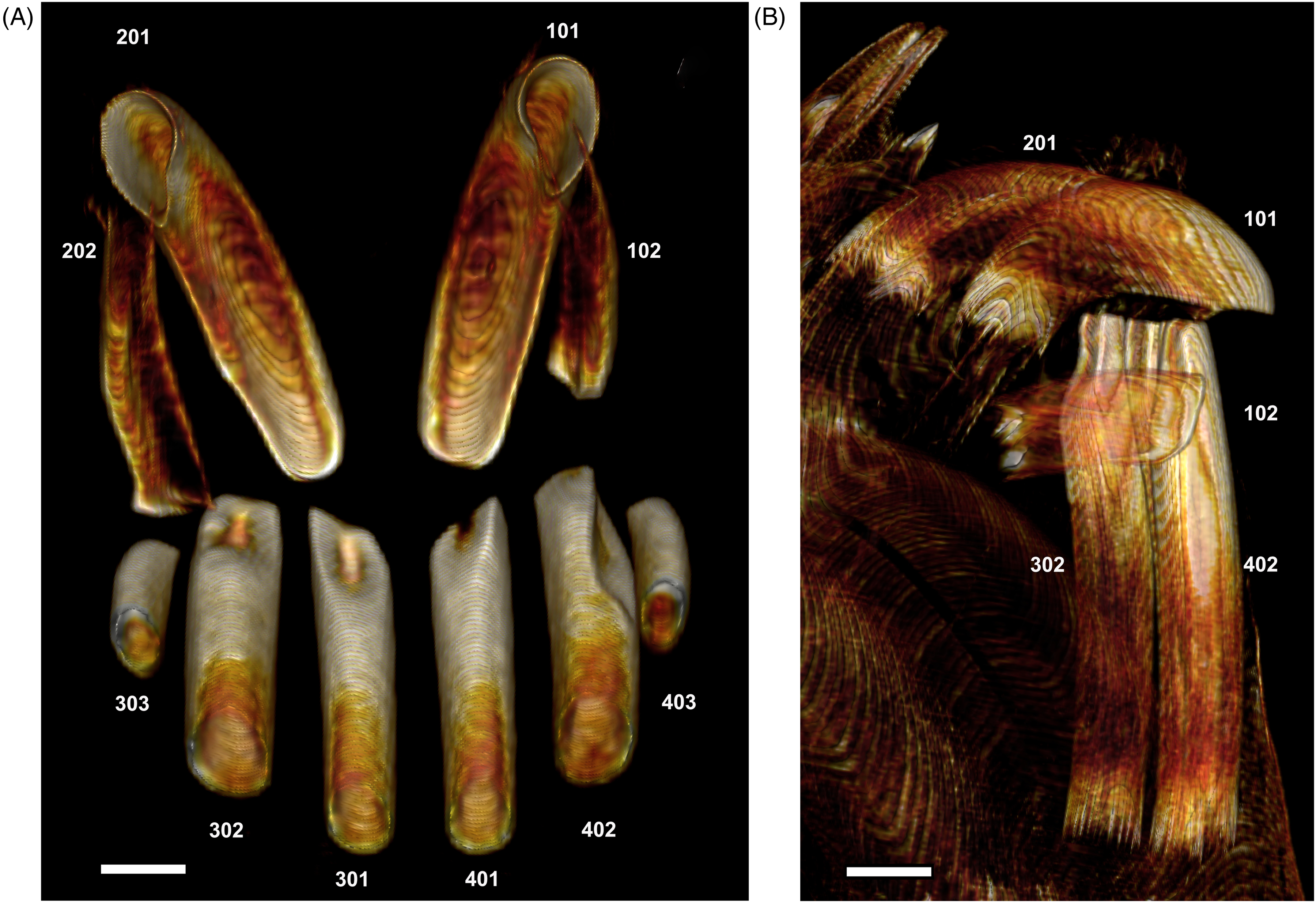
(A) Palato-Lingual Computed Tomography (CT) View of the Tubular Roots of the 6 Mandibular Incisors (Sectioned Approximately Mid-Length) and the Open-Ended Roots of the 4 Maxillary Incisors of an Adult Male Babirusa (Babyrousa babyrussa) (SNS.607). (B) Right Lateral CT View of the Open-Ended Tubular Roots of the Mandibular Incisors 302 and 402 Together with the Open-Ended Roots of the Maxillary Incisors 201, 101, and 102 (Babyrousa celebensis) (GNM.17.944). Bar=5 mm.
The 4 medial mandibular incisors 301, 302, 402, and 401 are relatively long (Table 3), slightly curved ventro-dorsally, tubular teeth, which lie approximately parallel to one another (Figures 5A, B and 6A, B). They have long tooth roots (Table 3). The occlusal surfaces of these incisors, formed largely due to wear, are orientated rostrally and dorsally (Figures 4B, 5A, B and 6B). The ovoid structure of these teeth is established at their apices, where the semivertical depth of the teeth is greater than their semihorizontal breadth (Table 3 and Figure 7A to E). This difference is maintained along the length of the teeth (Table 3 and Figure 7A to E).
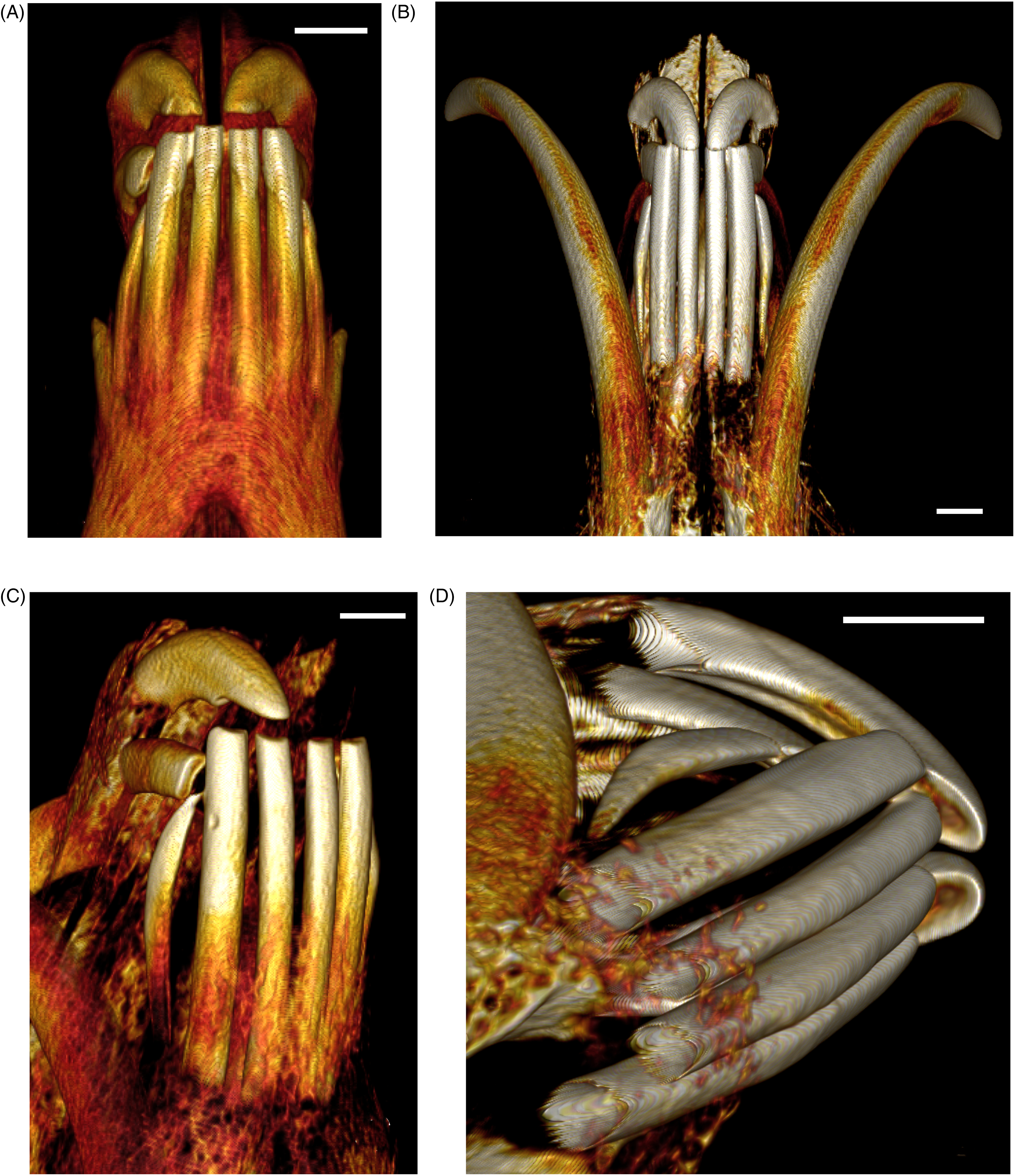
(A) Ventral Computed Tomography (CT) View of the Rod-Shaped Mandibular Incisors of a Subadult Babirusa (Babyrousa celebensis) (GNM.17.973). (B) Ventral CT View of the Rod-Shaped Mandibular Incisors of an Adult Male Babirusa (Babyrousa celebensis) (SNS.3070). (C) Oblique Right Ventral CT View of the Mandibular Incisors and Maxillary Incisors 102 and 101 of an Adult Male Babirusa (Babyrousa babyrussa) (SN.35645). (D) Oblique Right Ventral CT View of the Closed-Rooted Mandibular Incisor 403, the Open-Rooted Mandibular Incisors 402, 401, 301, and 302, and the Maxillary Incisors 102 and 101 of an Adult Male Babirusa (Babyrousa celebensis) (SNS.3070). Bar=10 mm.
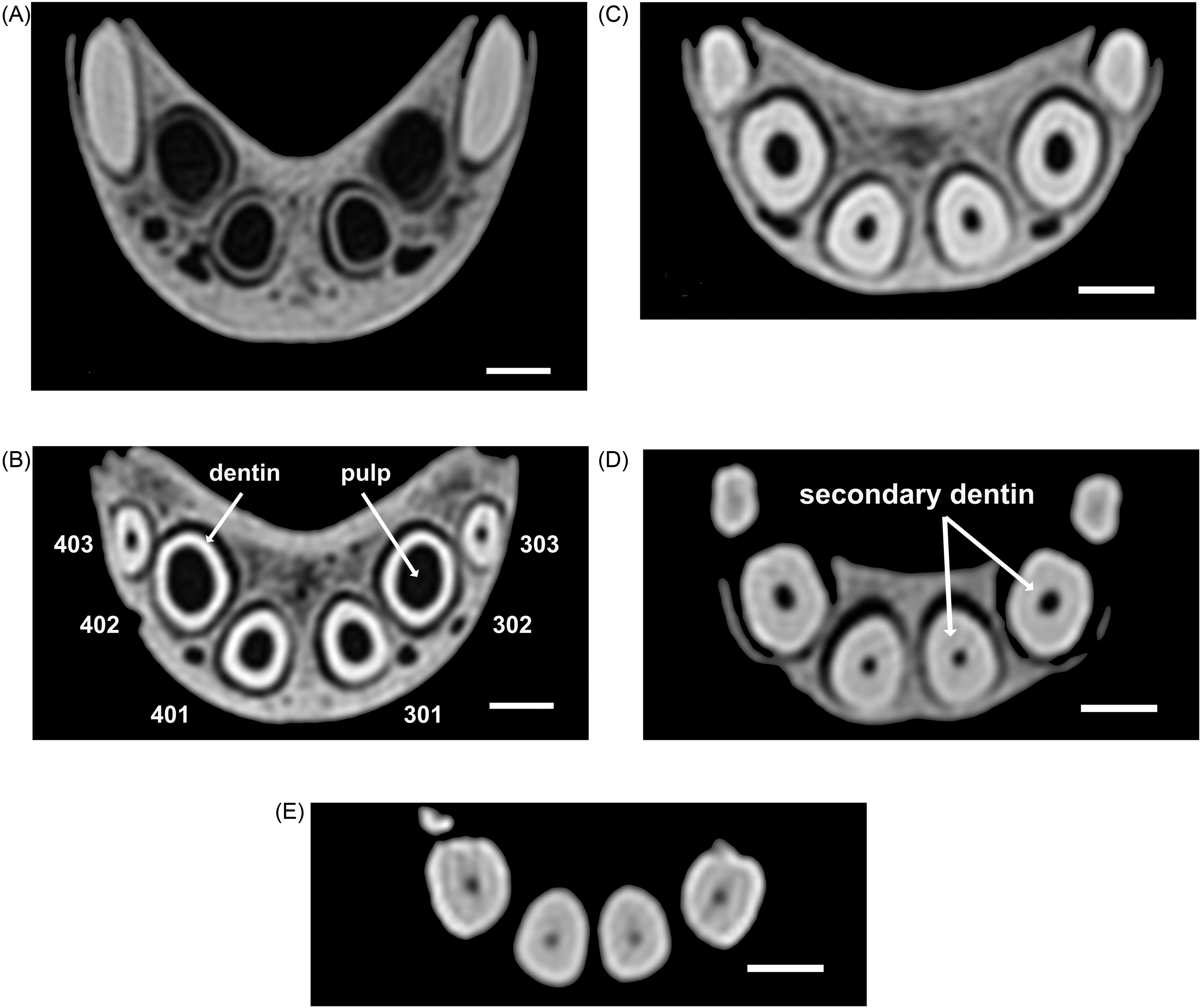
(A) Rostral Computed Tomography (CT) 2-Dimensional View of the Apices of Incisors 402, 401, 301, and 302 Dorsal and Mesial to the Mandibular Canine Teeth (404 and 304) of an Adult Male Babirusa (Babyrousa celebensis) (GNM.17.973). (B) Rostral CT 2D View of the Mandibular Incisors 403, 402, 401, 301, 302, and 303 at 25% the Length of These Teeth of an Adult Male Babirusa (Babyrousa celebensis) (GNM.17.973), Illustrating the Cross-Sectional Tooth Shapes, the Layer of Dentin, and the Enclosed Core of Pulp. (C) Rostral CT 2D View of Mandibular Incisors 403, 402, 401, 301, 302, and 303 at 50% the Length of These Teeth of an Adult Male Babirusa (Babyrousa celebensis) (GNM.17.973). (D) Rostral CT 2D View of Mandibular Incisors 403, 402, 401, 301, 302, and 303 at 75% the Length of These Teeth of an Adult Male Babirusa (Babyrousa celebensis) (GNM.17.973), Illustrating the Collar of Secondary Dentin that Has Progressively Been Forming Around the Narrowing Pulp Core. (E) Rostral CT 2D View of Mandibular Incisors 403, 402, 401, 301, and 302 at ∼95% the Length of These Teeth of an Adult Male Babirusa (Babyrousa celebensis) (GNM.17.973). Bar=5 mm.
Measurements From the Babyrousa babyrussa and Babyrousa celebensis Mandibular Incisor Teeth.
Abbreviations: Av, average; Std, standard.
*P < .05; **P < .01; ***P < .001.
The more lateral 302 and 402 teeth have larger cross-sectional dimensions (P < .001) than the adjacent, more medial, 301 and 401 incisors (Table 3).
The internal structures of these mandibular incisors change along their length. Within the layer of primary dentine, established in a nonuniform manner at the apex (Figures 5B, 6D and 7A), the pulp depths and breadths decreased progressively (P < 0.01) along the lengths of the teeth towards their cusps (Table 3). The compensatory progressive increase in the formation of secondary dentine along the incisors of both species (Table 3) is more clearly seen in Babyrousa celebensis (Figure 7A to E).
The length of the mandibular incisor tooth 302 as a proportion of the length of the mandibular ramus was 0.22 ± 0.043 (n = 16); there was no significant difference between Babyrousa babyrussa and Babyrousa celebensis in this regard.
The laterally situated 303 and 403 mandibular incisors are slender, curvilinear teeth with relatively pointed cusps, slightly enlarged mid-portions, and narrow open or closed apices (Figure 6C and D). They are shorter in length (Babyrousa babyrussa 27.8 ± 9.29 mm [n = 9] P < .01; Babyrousa celebensis 34.5 ± 13.12 mm [n = 22] P < .001) and have shorter tooth roots (Babyrousa babyrussa 27.0 ± 9.0 mm [n = 9] P < .01; Babyrousa celebensis 30.8 ± 8.15 mm [n = 22] P < .001) than the neighboring 302 and 402 mandibular incisors (Table 3). Their occlusive surface occupies the rostral end of the tooth, cusp, and shaft, and is oriented dorso-laterally (Figures 4A, B, 6C and 8B).
One aspect of the pattern of wear on the cusps of the mandibular incisors (Figures 5B, 6A to D and 7A to E) was present in both babirusa species. The ventrally facing cusps of the larger maxillary incisors 101 and 201 are situated opposite the dorso-rostrally facing cusps of the 4 mesial mandibular incisors 302, 301, 401, and 402. An additional aspect of the wear of the teeth is shown in Figure 8A and B. Almost parallel latero-medial scratch marks are apparent over the surfaces of the cusps of both the more caudal maxillary incisors 102 and 202 and on the rostro-palatal shafts of the larger maxillary incisors 101 and 201 (Figure 8A). Corresponding almost parallel latero-medial scratch wear is evident on the larger mandibular incisors 302 and 402 (Figure 8B). Polished tooth wear and similar scratch marks were evident on the incisors of both species of babirusa.
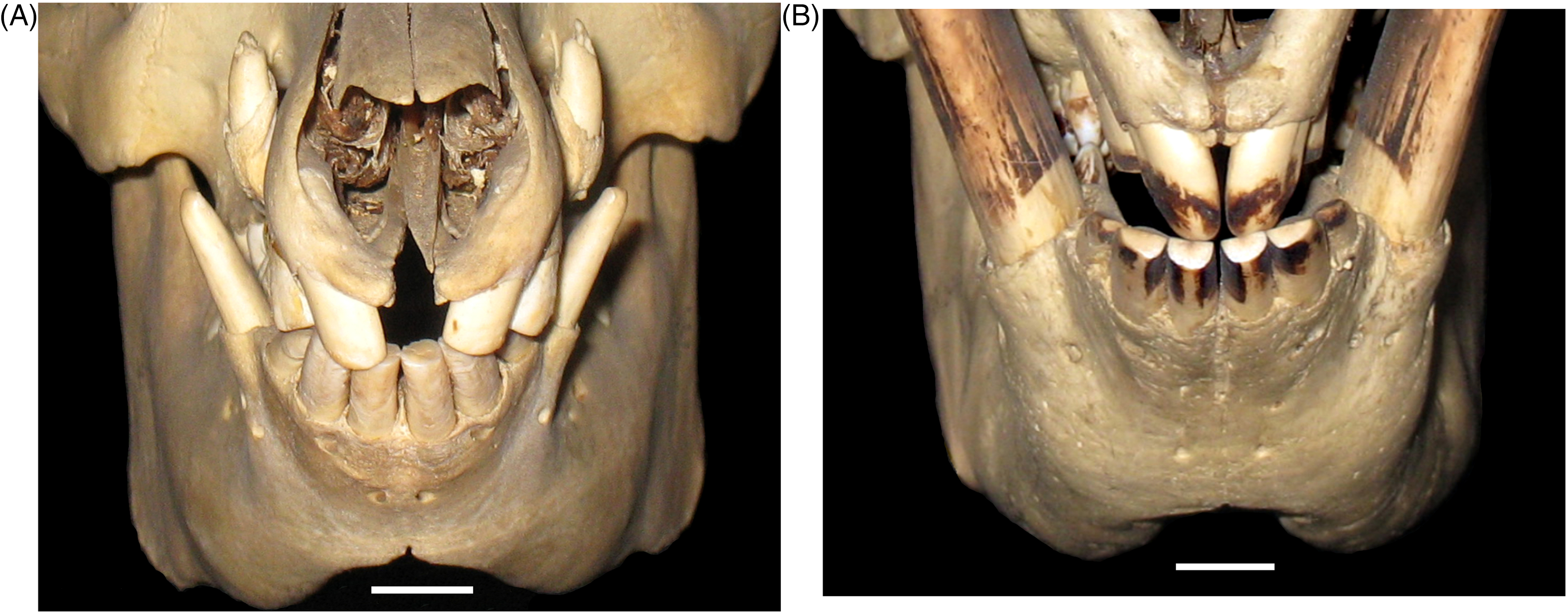
Rostral Photographic View of the Incisor Tooth Wear of Male Babirusa (Babyrousa celebensis). (A) Animal (MfN.45797) Aged 132 Days. (B) Animal (SNS.3070). Bar = 10 mm.
The apical edge of the open roots of the maxillary incisors may be relatively smooth (Figures 4A, B and 5A) or quite rough and spikey (Figures 2A, 5B and 6D). The apical structure of the open roots of the mandibular incisors can similarly vary in circumferential roughness (Figures 2C and 6D). The apices of all the mandibular incisors lay in the mandibular rami and were positioned rostral to the caudal margin of the mandibular symphysis; 16.5 ± 4.8 mm (n = 7) rostral in the case of Babyrousa babyrussa, and 21.2 ± 5.5 mm (n = 13) rostral in the case of Babyrousa celebensis.
Discussion
The present study is the first since 1899 to have closely examined the structure of the maxillary and mandibular incisor teeth in adult male babirusa (Babyrousa spp.). 14 The application of computer-tomographic-scanned illustrations and measurements has added new information to earlier descriptions of those teeth. For the first time, the sizes and masticatory relationships of the babirusas’ maxillary and mandibular incisors have been described and depicted, and the resulting pattern of tooth wear illustrated.
The anatomical arrangement of the 4 maxillary and the 4 mesial mandibular incisors in the mouth of the babirusa appears to be well adapted to their observed harvesting of fruits.7–9 Local people on Buru and the Sula Islands, Indonesia, drew attention to the various species of fig growing there, including the native Ficus heteropoda Miq, 18 as foods of the babirusa.19,20 These fig trees exhibit cauliflory, which is when the flowers and fruits are borne on the trunk instead of on branches and twigs. Cauliflory places the fruits within easy reach of babirusa standing on their hind limbs. 21 In pantropical forests, the fig is considered a keystone species because of its abundant, year-round fruiting. 22 Other plants with low-hanging fruits were also described by the people on Buru. 19 When the barbirusa graze on these tree leaves, they reach up and pull them off with their incisors.8,10
Various reports exist that indicate babirusa do not eat dirty foods, and this behavioral observation is supported by the morphology of the incisor teeth described in this study. One of the local correspondents on Taliabu said that her pet babirusa only ate clean food. 20 Another on Buru said that the babirusa did not eat dirty food. 21 Recent corroborating evidence has come from observations of babirusa in a zoological collection. These studies have documented babirusa carrying food such as sweet potato (Ipomoea batatas) that has fallen on the ground in front of their mouths and bringing it to water to wash it before eating it. 10 Other studies have shown that babirusa use their incisor teeth to selectively excavate specific regions of the white coconut mesocarp from broken young coconuts.11,12
The long rod-like shapes of the mandibular incisors 302, 301, 401, and 402 appear to derive their strength from their ovoid-shaped perimeter (Figures 5A and 7A to E) and the length of their attachments into the mandibular rami (Figures 5B and 6A to D). The mesial mandibular incisors 301 and 401 were narrower (P < .001) than the adjacent incisors 302 and 402 (Table 3). However, there was no consistent difference in their comparative lengths. The mandible functions as a rigid curved beam, 23 and studies of the mandibular symphysis of Sus domesticus indicated that the fused symphyses of older animals experienced smaller masticatory strains. 24 Studies conducted to analyze the forces placed by mastication on the rostrum and craniofacial sutures of Sus domesticus have indicated that the premaxillary and nasal bones experience lower magnitudes of force than the nearby sutures.25,26 The mesial bony protrusion of the maxillary alveoli of 101 and 201 (Figures 1A and 3A), a characteristic of the babirusa rostrum, probably acts to give support to these hypselodont teeth. Similarly, the relatively short incisors’ slightly flattened shapes (Figure 1A) and valleyed contours (Figure 2A), as well as their curved path out of the rostrum (Figure 3A), likely provide additional strength to these teeth.
There have been no reports of the male babirusa using its incisors to bite or fight with another babirusa. Agonistic behavior is performed in a different way and does not involve the use of any of its teeth, including the large lower canines.27,28
Tooth wear was anticipated following earlier studies of the wear in adult male open-rooted canine teeth. 29 The male babirusa's behavior of scent-marking trees and other substrates was likely to contribute to the erosion of the lateral and labial surfaces of the incisors.20,30 Likewise, its ploughing behavior in sand, soft soil, and wet mud, which also has a proposed scent-marking function, could have created wear on the labial surfaces of the incisors.31,32 The mouthing and chewing of muddy water in zoos and at the salt-licks on Sulawesi, Buru, and the other Sula islands was anticipated to contribute wear on the lingual surfaces of the incisors.8,32,33
Detailed scanning electron microscopic studies of babirusa teeth have revealed several different pit and scratch wear marks on the labial and lingual surfaces of babirusa incisors. 34 The pit marks on the lingual surfaces were understood to be the result of the compression of food or soil particles between the occlusal surfaces. Parallel, transverse scratch marks, comparable to those seen in the present study (Figure 9A and B), were also reported. 34 Studies of mastication in Sus domesticus have revealed that an unexpected chewing behavior was responsible for such a pattern; the lower incisors moved laterally across the midline towards one side and then back across the midline towards the other side.35–37 The possibility that the babirusa may also be able to exercise this yaw movement was investigated, and it was found to be present. 33 Videos were recorded of adult male babirusas viewed from in front of the rostrum. The mandibular yaw movements were measured by evaluating the temporal changes in the horizontal distance between the maxillary and mandibular canines. The babirusa was filmed inserting his snout into the mud, scooping up some mud, and starting to chew. The maxillary canines remained almost still, whereas the trajectory of the mandibular canines contained a horizontal component of motion. 33 The frequency of these recorded babirusa yaw movements (approximately 3 cycles per second) was comparable to that of the transverse component of mastication in domestic pigs. 35
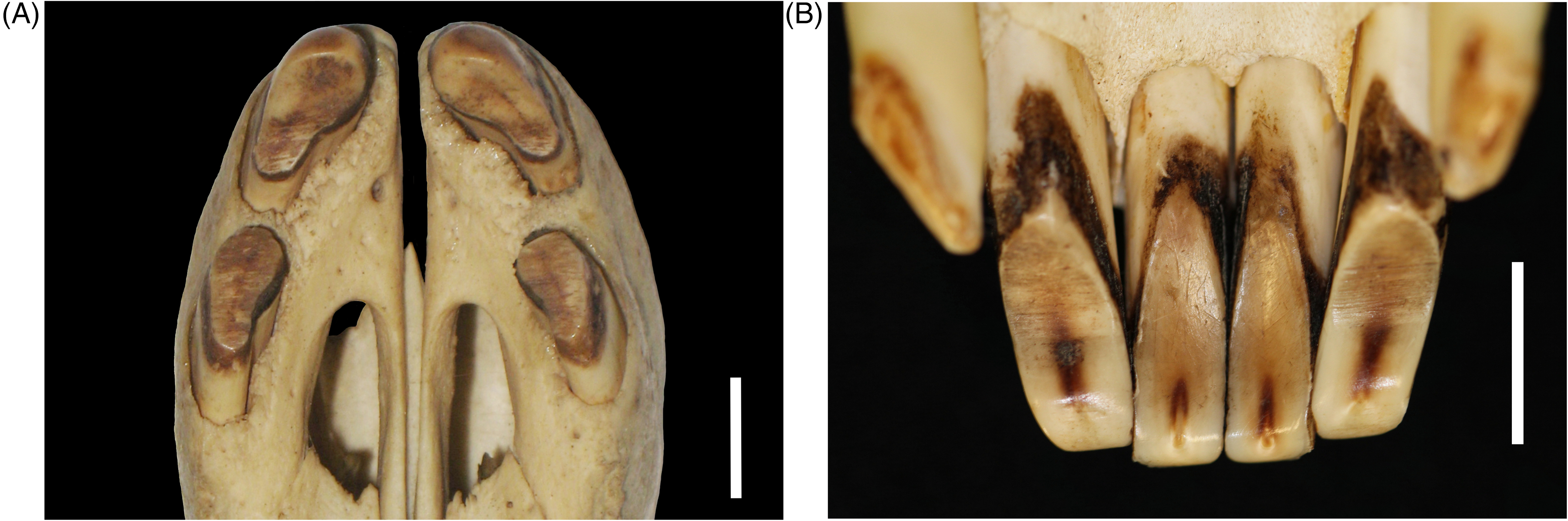
(A) Palatal Photographic View of the Maxillary Incisor Wear in a Mature Adult Male Babirusa (Babyrousa celebensis) (GNM.4.728). (B) Lingual Photographic View of the Mandibular Incisor Wear in a Young Male Adult Babirusa (Babyrousa celebensis) (NMS.Z.2002.210.001). Bar=10 mm.
The effects of such a pattern of jaw movements on the wear of the babirusa's incisor teeth can be visualized in the results of the present study. The lateral movement of the mandibular incisors 302, 301, 401, and 402 ventral to maxillary incisors 101 and 201 (Figure 10A to C) led to the flattened wear of the rostral ends of the former (Figure 8A and B). The same movement ventral to maxillary incisors 102 and 202 (Figure 10A to C) led to wear on the lingual surfaces of the mandibular incisors 302, 301, 401, and 402 (Figures 4A, B, 5A and 9B). Concomitantly, erosive wear of the labial surfaces of the maxillary incisors 101 and 201 was produced (Figures 4B, 6C, 6D and 9A) as well as on the ventral surfaces of the adjacent maxillary incisors 102 and 202 (Figure 9A and 10A to C).
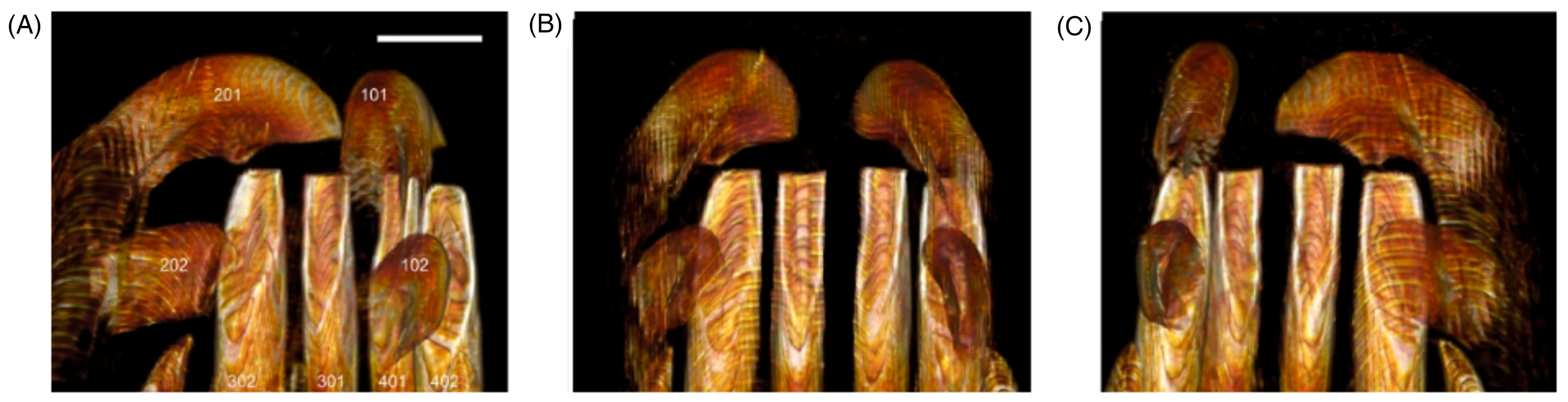
(A-C) Sequence of 3 Dorsal Computed Tomography 3-Dimensional Views of the Maxillary Incisors (L to R, 202, 201, 101, and 102) Overlying the Lingual Appearance of the Mandibular Incisors (L to R, 302, 301, 401, and 402) of an Adult Male Babirusa (Babyrousa celebensis) (GNM.4.728). These Illustrate the Nature of the Tooth Contact of the Maxillary Incisors 201 and 101 with the Rostral Ends of the 4 Medial Mandibular Incisors (302, 301, 401, and 402). They also Show the Nature of the Tooth Contact of the Cusps of the Maxillary Incisors 202 and 102 with the Lingual Surfaces of the Mandibular Incisors 302 and 402. Bar=10 mm.
The open-rooted, continual growth of the babirusa's incisors has raised questions about their rates of growth and the molecular control of growth at their apices. Significant differences in the siting of tooth growth around the perimeter of the incisor apices appeared to be present (Figures 5B, 6D and 7A). Nevertheless, these apices remained rostral to the caudal margin of the mandibular symphysis. Despite the large amount of research into the molecular regulation of tooth growth, the answers are complex and remain unclear. 38 An early histological study of the open-rooted maxillary canine tooth of the babirusa has been conducted, and narrow bands were observed within the cement layer.39,40 Although only a small amount of the dentine layer was depicted, it seemed to contain parallel dentinal tubules. 32 There appears to have been no subsequent investigations of the microscopic anatomy of babirusa teeth. However, dominant incremental enamel and dentinal markings with a daily periodicity (von Ebner lines) have been described in detail for ground sections of mandibular molar teeth (310) from wild Sus scrofa and Sus domesticus.41,42 These studies have recently been extended to analyses of dentine formation rates during crown and root growth of the mandibular molar teeth (309). 43 Differences in rates of dentin apposition were found along the vertical tooth axis, and these were dependent on the postnatal time of dentine formation. In the upper root of wild Sus scrofa, the dentine apposition rate rose from about 12.5 μm/day at 86 to 100 days of age to about 17.5 μm/day at 156 to 184 days of age before gradually declining to about 2.5 μm/day at 275 to 330 days. 43 To date, only a single measurement of dentine apposition rate in a suid incisor has been published: 3.81 ± 0.98 μm/day in Sus domesticus. 44 Taken together with the present investigation, the results of these studies suggest that analysis of dentine apposition in the mandibular incisors of the babirusa (with the appropriate permission of zoological museum collections) would yield information helpful to a better understanding of open-rooted incisor growth.
Conclusions
The structural adaptations of the open-rooted maxillary and mandibular incisors in the babirusa enable its food-gathering behavior, particularly the gathering of fruit and leaves. With the constantly growing dentin, incisors exhibit tolerance of the subsequent chewing wear at the occlusive surfaces of the teeth. The current absence of any detailed published accounts of the histology of these incisors represents an unexpected lacuna in a more complete understanding of their biology.
Materials
Senckenberg Naturhistorische Sammlungen Dresden, Germany (SNSD). National Museums Scotland, Edinburgh, Scotland (NMS). Naturmuseum Senckenberg, Frankfurt am Main, Germany (SMF). Göteborgs naturhistoriska museum, Göteborg, Sweden (GNM). Museum für Naturkunde, Berlin Germany (MfN) Museum Zoologicum Bogoriense Cibinong, Indonesia (MZB) Naturalis Biodiversity Centre Leiden, The Netherlands (NBCL) Oxford University Museum, England (OUM) The University of Edinburgh: Helical scan mode, collimator pitch of 1, 0.4 mm slice width, 0.2 mm slice interval, 120 kVp, 111 mAs, ultra-high resolution bone image reconstruction kernel (proprietary name U75u), 192 mm diameter display field of view, 5122 image matrix (64-slice CT, Somatom® Definition AS Siemens, Erlangen, Germany). The University of Giessen: Helical scan mode, collimator pitch of 0.75, 0.65 mm slice width, 0.32 mm slice interval, 140 kVp, 190 mAs, bone image reconstruction kernel (proprietary name D), 176 mm diameter display field of view, 5122 image matrix (16-slice CT, Brilliance® 16, Philips Medical Systems, Eindhoven, the Netherlands). The University of Leipzig: Helical scan mode, collimator pitch of 0.75, 0.7 mm slice width, 0.35 mm slice interval, 140 kVp, 193 mAs, bone image reconstruction kernel (proprietary name D), 100 mm diameter display field of view, 5122 image matrix, g. (6-slice CT, Brilliance® 6, Philips Medical Systems, Eindhoven, the Netherlands). Blåstjärnans Djursjukhus Göteborg: Helical scan mode, collimator pitch of 0.53125, 0.625 mm slice width, 0.312 mm slice interval, 120 kVp, 200 mAs, bone image reconstruction kernel (proprietary name Bone Plus), 182 mm diameter display field of view, 5122image matrix (16-slice CT, GE Revolution® EVO CT, GE Healthcare Japan Corporation, Tokyo, Japan). JASP Team (2020) JASP (Version 0.14.1), University of Amsterdam, Amsterdam, Netherlands.
Footnotes
Acknowledgments
The authors are thankful for the kind hospitality and assistance provided by Clara Stefen and Jens Jakobitz of the Senckenberg Naturhistorische Sammlungen Dresden, Germany, to Irina Ruf and Katrin Krohmann of the Senckenberg Forschungsinstitut und Naturmuseum Frankfurt am Main, Germany, to Ines Merseburger of the Veterinärmedizinische Fakultät, Klinik für Kleintiere, Universität Leipzig, Germany for her technical assistance and to Georg Hantke of the National Museums Scotland, Edinburgh, for photographic assistance. We are also grateful for the technical support and access to the mammal collections in each of the following museums: the National Museums Scotland, Edinburgh, Scotland; the Senckenberg Naturhistorische Sammlungen Dresden, Germany; the Senckenberg Forschungsinstitut und Naturmuseum Frankfurt am Main, Germany; and the Göteborgs naturhistoriska museum, Göteborg, Sweden; the Zoological Museum Amsterdam, The Netherlands; the Naturhistorisches Museum Basel, Switzerland; the Museum für Naturkunde, Berlin, Germany; the Museum Zoologicum Bogoriense Cibinong, Indonesia; and the Zoologisk Museum Copenhagen, Denmark. We are grateful to the University of Edinburgh and the Balloch Trust for academic support. ACK thanks the Negaunee Foundation for its generous support of a curatorial preparator at the National Museums. Scotland, who prepared some of the specimens in this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
