Abstract
A photographic and computed tomography (CT) scanning study was carried out on 295 molar teeth of 18 adult male Babyrousa babyrussa skulls and 8 skulls of Babyrousa celebensis including seven adult males and one adult female. The occlusal morphology of the permanent maxillary and mandibular molar teeth of B. babyrussa was very similar to that of B. celebensis. Most B. babyrussa maxillary molar teeth had six roots, with small numbers of teeth having four, five or seven roots. A similar pattern was suggested in B. celebensis. Mandibular molar teeth had between four and eight roots. Tooth roots of maxillary and mandibular first and second molar teeth were largely tapering, rod-like structures. The roots of the right and left maxillary third molar teeth had a more complex arrangement; some were inserted almost vertically into the maxilla; others were orientated in a more distal direction. The mesial and distal roots were splayed in appearance. The right and left mandibular third molar tooth roots retained elements of the open ‘C’ shape and were generally orientated distally. The pulp chambers were arched to fit under the main cusps in all molar teeth. Pulp canals were variable in number.
Introduction
The babirusa (genus Babyrousa) is a suid endemic to the Indonesian island of Sulawesi and a number of smaller islands to the east.1,2 Illustrations of the molar teeth of the babirusa have been previously published.3–5 Babirusa molar teeth are brachydont, in which the root is longer than the crown, and these do not grow continuously. Physical evidence of an early interest in studying the roots of these molar teeth was indicated by the half-skull and mandible of a young adult male Sulawesi babirusa (B. celebensis) (Figure 1A). In 1860 this specimen (OUM-05266) was transferred from the Christ Church College collection into the main Oxford University Museum, England (OUM) collection. Excavation of sections of the right lateral surface of the skull gave some indication of the arrangement of the lateral (buccal) roots of the molar teeth. Modern computed tomography (CT) scanning techniques have offered the opportunity to examine the roots of babirusa teeth in a number of skulls without damaging the overlying bone (Figure 1B).
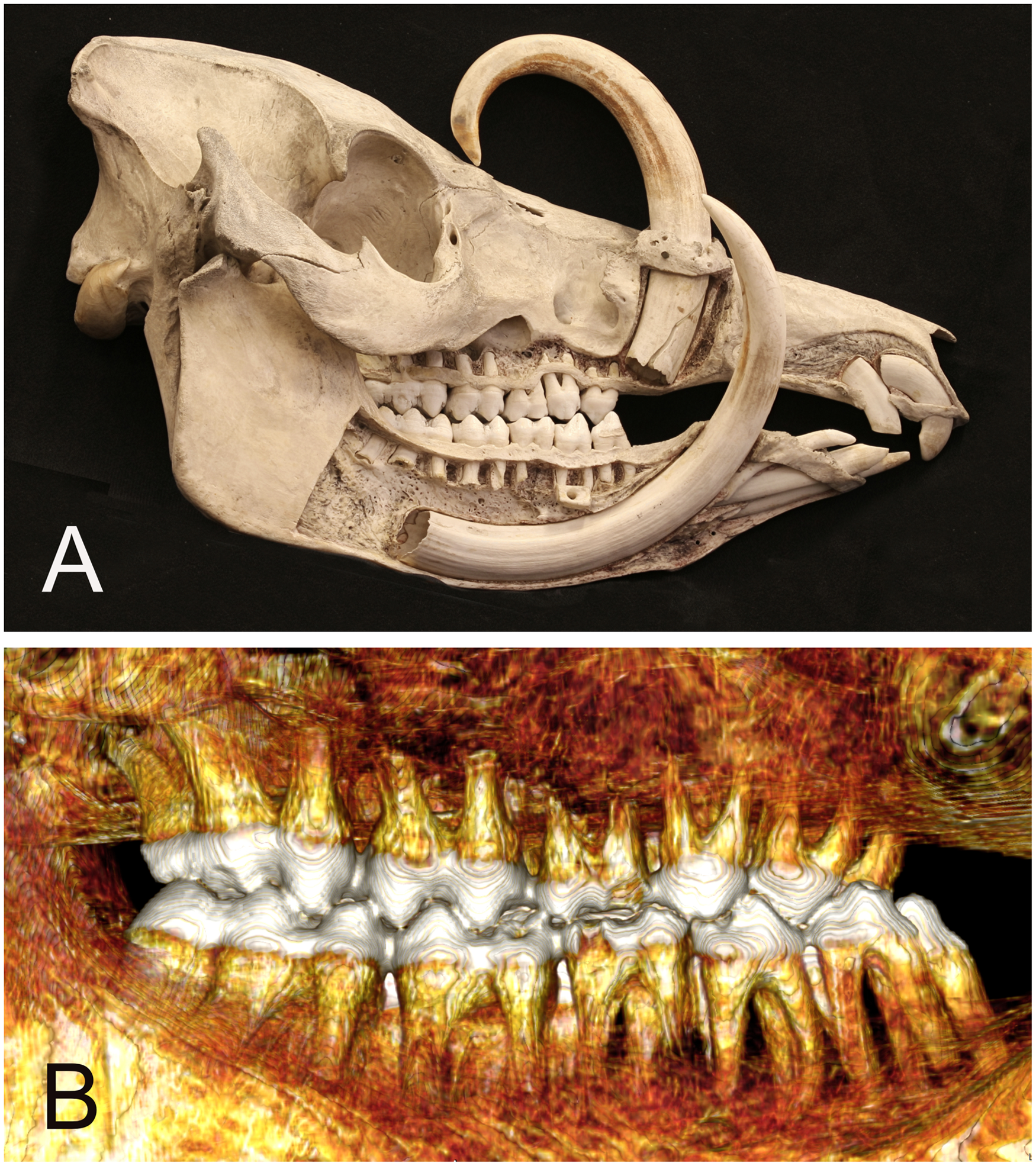
(A) Right lateral view of Sulawesi babirusa (Babyrousa celebensis) OUM 05266 illustrating anatomical exposure of the roots of the teeth. (B) Right lateral 3D CT-scan view of roots of the molar and premolar teeth of Sulawesi babirusa (Babyrousa celebensis) GNM 4.728.
During studies of the anatomy of the babirusa stomach it was found that the ingested plant material had a ‘granular appearance’. 6 Research had also revealed that the mucosal surface lining of the cardiac gland region of the stomach was very much larger than that of the Sus pigs7,8 and that it was uniquely covered in a layer of cells arranged in the form of a honeycomb. 9 Examination of the wear patterns of babirusa molar teeth showed that there was heavy wear of maxillary and mandibular first molar teeth and a relative delay in the wear of the corresponding second and third molar teeth. 6 It was also known that babirusa would select to eat fruits with hard seeds, and that these were cracked open by the molar teeth (Leus and Macdonald, 1995, unpublished observations). Recent studies have focused on the anomalous molar wear patterns found in a proportion of the (largely) North Sulawesi babirusa, B. celebensis, that seem to be regionally found in association with the presence of one particular type of wild banana, Musa balbisiana Colla. 10 This wild banana species contains 150-170 very hard, small pea-sized seeds. 10
These dietary observations prompted several questions. Could the numbers and the orientation of the roots of mandibular and maxillary molar teeth provide some form of insight into the function of babirusa molar teeth? Are the molar tooth roots anatomically arranged in some specific way to distribute the crushing force of a bite into the bony tissues of the mandibles? How might the structure of the corresponding number and arrangements of the maxillary tooth roots support the crushing and grinding motions of the occlusal surfaces against those of the mandibular teeth?
A search of the literature failed to reveal the results of any systematic study of the anatomy of the molar teeth of babirusa. As a consequence, the authors undertook a CT examination of babirusa skulls from four museum collections.a-d The skulls selected represented the babirusa species from Buru and the Sula Islands (B. babyrussa) and from the island of Sulawesi (B. celebensis). Additional skulls were accessed from OUM for specific topographical information.
The modified Triadan system of veterinary dental nomenclature, 11 as illustrated for babirusa dentition 12 was adopted for this study to provide the description of dental location and orientation. 13
Materials and Methods
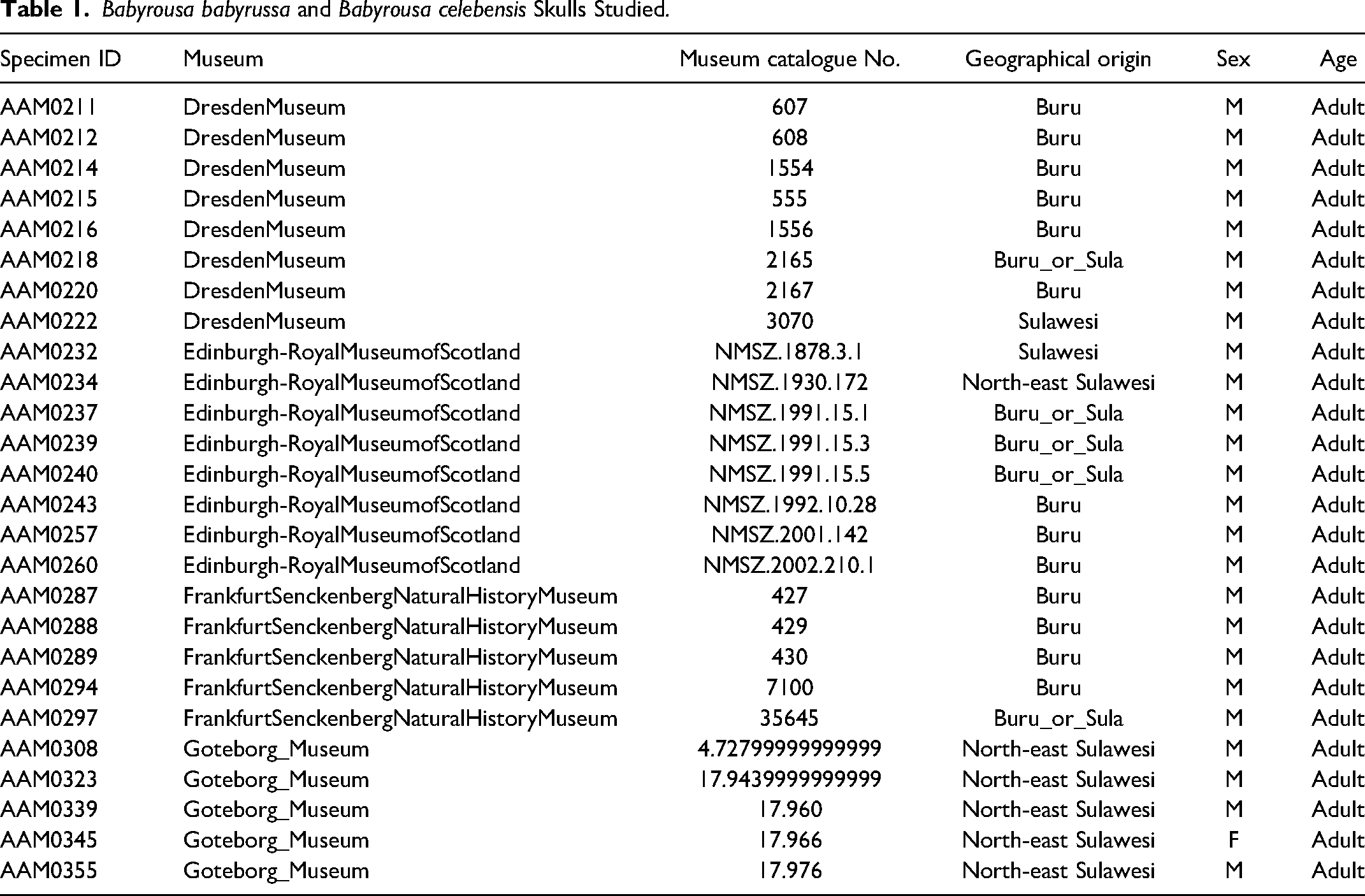
Twenty six babirusa skulls were studied. The primary research material comprised 13 adult male B. babyrussa skulls catalogued as from Buru. Five adult male B. babyrussa skulls were morphologically identified as coming either from Buru or the Sula Islands and seven adult male and one adult female B. celebensis skulls from Sulawesi (Table 1). The total number of babirusa teeth available for evaluation was 295 (Table 2). One B. babyrussa skull had no right mandibular first or second molar (409 / 410) teeth and one B. celebensis skull had no left mandibular second molar (310) tooth. Two B. babyrussa skulls had no right maxillary first molar (109) teeth and one B. celebensis skull had no 109 tooth. The mandibular tooth measurements from one male B. celebensis (AAM355) were not recorded.
Babyrousa babyrussa and Babyrousa celebensis Skulls Studied.
Babyrousa babyrussa and Babyrousa celebensis Tooth Roots.
All specimens were photographed, including close-up views of the occlusal surfaces of the teeth. To enable anatomical clarity in the description of observations, the dental anatomical nomenclature of the babirusa teeth 10 was derived from that published for Suidae. 14 The main cusps on the maxillary molars are called paracone, protocone, metacone, tetracone, and pentacone (Figure 2A). The main cusps on the mandibular molars are called protoconid, metaconid, hypoconid, entoconid, and pentaconid. A smaller sixth cusp can be seen on the left mandibular third molar (311) tooth and it is named hexaconid (Figure 2B).
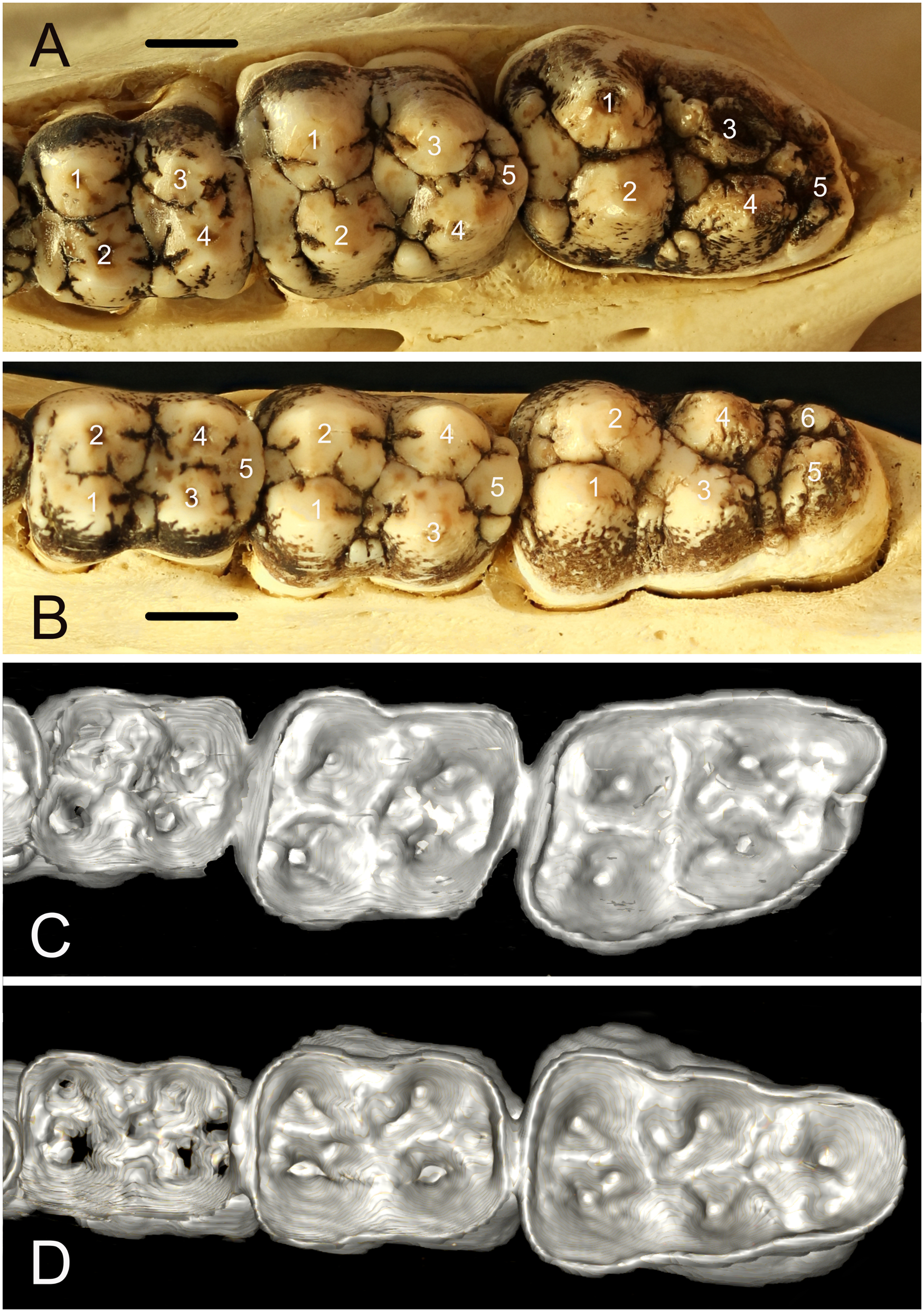
(A) Occlusal view of the left maxillary molar teeth of Buru babirusa (Babyrousa babyrussa) NMS,Z,2002.210.1. The list of symbols14 describes the cusps of the maxillary molar teeth, left to right; 209, 210 and 211: 1: paracone; 2: protocone; 3: metacone; 4: tetracone; and 5: pentacone. (bar = 10 mm). (B) Occlusal view of the left mandibular molar teeth of Buru babirusa (Babyrousa babyrussa) NMS,Z,2002.210.1. The list of symbols14 describes the cusps of the mandibular molar teeth, left to right: 309, 310 and 311. 1: protoconid; 2: metaconid; 3: hypoconid; 4: entoconid; 5: pentaconid; and 6: hexaconid. (bar = 10 mm). (C) 3D CT-scan view of the underside of the enamel layer comprising the occlusal surface of the left maxillary molar teeth of Buru babirusa (Babyrousa babyrussa) NMS,Z,2002.210.1. (D) 3D CT-scan view of the underside of the enamel layer comprising the occlusal surface of the left mandibular molar teeth of Buru babirusa (Babyrousa babyrussa) NMS,Z,2002.210.1.
For curatorial and geographical reasons (Table 1) the skulls were CT scanned at four different institutions using four different CT scanner models.e-h
Tooth length was measured from the highest point of the crown (the coronal end) to the apex (the extremity) of the tooth root. Tooth root length was measured from either the buccal or the lingual/ palatal cementoenamel junction (depending on root orientation in the CT scan) to the apex of the root. The occlusal edges of the molar tooth were formed from the enamel cusps. The various ways in which the apical ends of the right and left maxillary third molar teeth (111 / 211) more distal roots subdivided made comparative measurements unreliable. Tooth root width was measured across the mid-length of the tooth root. 12
Statistical analysis of variancei was obtained on the teeth of the adult babirusa.
Results
Occlusal Morphology
The occlusal morphology of the permanent maxillary molar teeth of B. babyrussa (Figure 2A) was very similar to that of B. celebensis. The paracone of each of the molar teeth was in the form of a buccally situated, rounded cone. On its distal surface lay the metacone. The paracone of the right and left maxillary second molar teeth (110 / 210) was bordered distally on its buccal side by a slightly smaller metacone. The protocone was situated on the mesiopalatal side of the crown and was of equivalent size to the paracone. Distal to it lay the tetracone. Distal to the protocone and the tetracone was the smaller pentacone.
Similarly, the occlusal morphology of the mandibular molar teeth of B. babyrousa (Figure 2B) was very similar to that of B. celebensis. The protoconid of each of the molar teeth was a conical, buccally situated, cusp. The adjacent, lingually situated, metaconid was approximately equivalent in size. The hypoconid and entoconid were situated distal to these, on the buccal and lingual sides respectively. The somewhat smaller pentaconid occupied the distal aspects of the left mandibular first molar tooth (309) and 409, and310/410. In the 311 and right mandibular third molar (411) teeth, the smaller again hexaconid occupied the distolingual side of the tooth adjacent to the buccally situated pentaconid.
The morphology of the dentine underlying the enamel of the occlusal surface of the maxillary and mandibular molar teeth was as indicated in Figure 2C and D respectively. The irregular cone-shapes of the dentine corresponded, in reduced form, to the occlusal morphology of the overlying enamel layers of the maxillary and mandibular molar teeth.
Number of Tooth Roots
The numbers of the measurable roots found on each molar tooth are summarised in Table 2. Small bumps and short ridges were also present on the undersides of some teeth. The majority of B. babyrussa maxillary molar teeth had six roots, with small numbers of teeth having four, five or seven roots. The four main roots were positionally designated mesiobuccal, mesiopalatal, distobuccal and distopalatal and the orientation of the additional roots was illustrated in Figure 3A. Despite the smaller sample number of B. celebensis maxillary teeth, a similar pattern may pertain in this species also. The orientation of the mandibular roots was indicated by the pattern of root alveoli in the exposed mandible (Figure 3B). The majority (60%) of B. babyrussa 309/409 and 310/410 teeth had four roots (Figure 3C and D) positionally designated mesiobuccal, mesiolingual, distobuccal and distolingual, with the remaining 40% having five roots, the fifth root designated as central. None of the 311/411 teeth in that species had four roots, with the majority (38.9%) having seven roots, including those designated as central, buccal and lingual. The 309/409 and 310/410 teeth of B. celebensis had four or five roots, while the 311/411 teeth had between six and eight roots (Table 2).
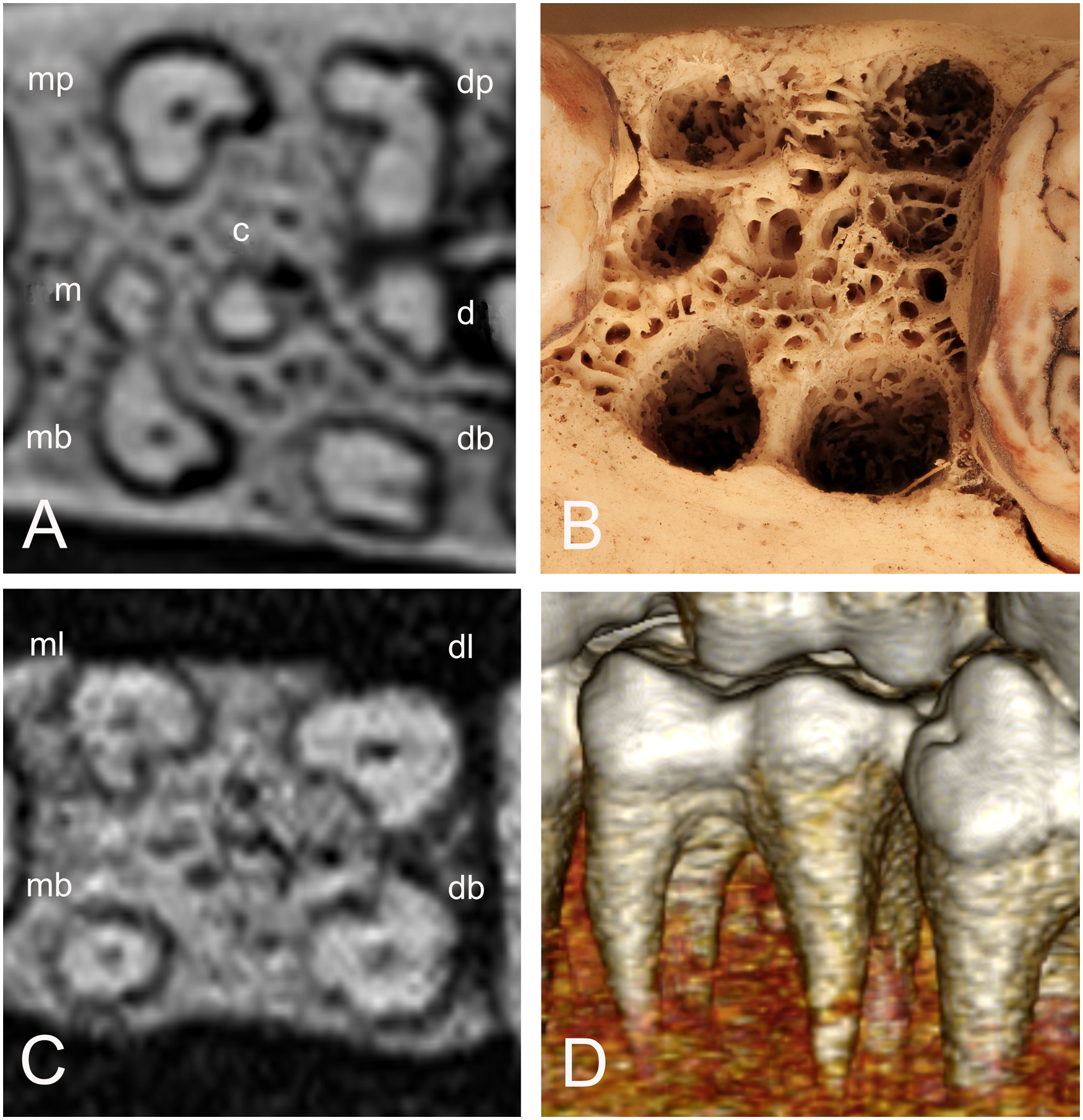
(A) Cross-section CT-scan of the 209 tooth of Buru babirusa (Babyrousa babyrussa) NMS,Z, 1991.15.5 illustrating the four main roots (mb = mesiobuccal, mp = mesiopalatal, db = distobuccal, dp = distopalatal) and the positions of three additional roots (m = mesial, c = central, d = distal). (B) Photograph of the 209 tooth alveolus of Sulawesi babirusa (Babyrousa celebensis) GNM 17.934 with openings into the bony matrix corresponding to positions of the roots described in 3A. (C) Cross-section CT-scan of the 309 tooth of Buru babirusa (Babyrousa babyrussa) NMS,Z,2001.142 illustrating the four roots (mb = mesiobuccal, ml = mesiolingual, db = distobuccal, dl = distolingual). (D) Lateral 3D CT-scan of the 309 and the protoconid and underlying mesiobuccal root of 310 of Buru or Sula babirusa (Babyrousa babyrussa) SMF 19704 DB.
Tooth Root Structure
The 109 and left maxillary first molar (209) tooth roots were largely tapering, rod-like structures (Figure 1B). These had an irregular, somewhat oval cross-section with many showing a semi-closed or open ‘C’ shaped construction (Figure 3A). The distobuccal root insertion exhibited a slight distal tilt from vertical in 75% of these teeth in B. babyrussa and in 50% of B. celebensis. The mesiobuccal roots were almost vertically inserted into the maxillae (Figure 1).
The data for the 109/209 tooth lengths, tooth root lengths and tooth root thicknesses are summarized in Table 3. In both B. babyrussa and B. celebensis the four main roots (mesiobuccal, mesiopalatal, distobuccal and distopalatal) had longer tooth and tooth root lengths than those of the mesiomedial, mesial and distomedial roots (P < .001). These four main roots in B. babyrussa were thicker than those of the mesiomedial, mesial and distomedial roots (P < .001). In B. babyrussa the mesiopalatal and distopalatal roots were thicker than the corresponding mesiobuccal and distobuccal roots (P < .001). In B. celebensis the mesiopalatal roots were thicker than the corresponding mesiobuccal roots (P < .05), and the distobuccal roots were thicker than the distomedial roots (P < .01).
Babyrousa babyrussa and Babyrousa celebensis Maxillary Molar Teeth Measurements.
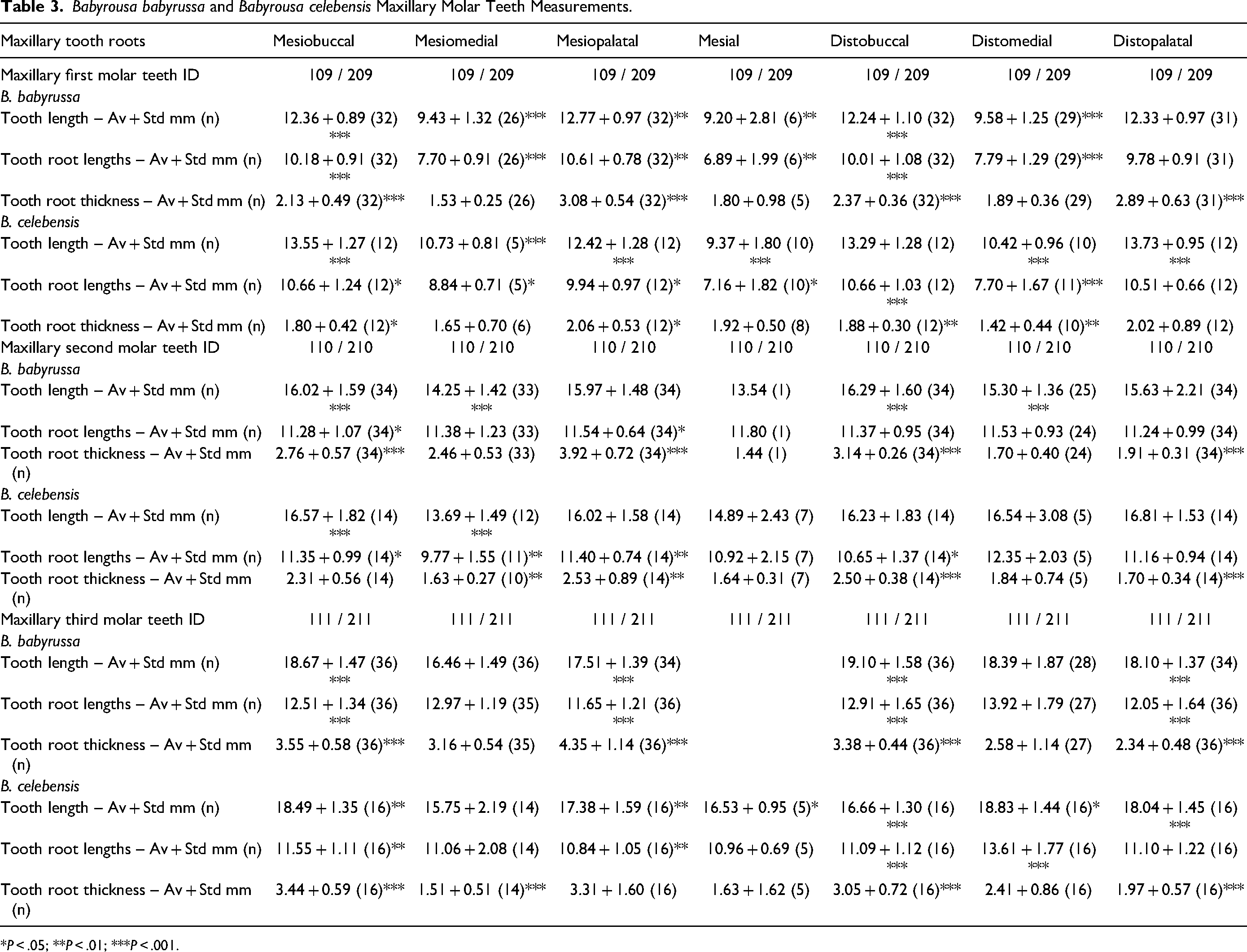
*P < .05; **P < .01; ***P < .001.
The 309/409 tooth roots were also tapering, rod-like structures (Figure 1B). The majority, 64% of the B. babyrussa, and 50% of the B. celebensis 309/409 mesiobuccal and distobuccal roots inserted almost vertically into the bone (Figure 1B). The data for the 309/409 tooth lengths, tooth root lengths and tooth root thicknesses are summarized in Table 4. In both species the central tooth and root lengths were shorter, and in B. babyrussa thinner, than each of the four main roots (P < .001). In B. babyrousa the tooth lengths of the four main roots were not significantly different from one another. However, the mesiolingual roots were longer than those of the other three main roots (P < .01) and thicker than the mesiobuccal roots (P < .001). In B. celebensis the mesiolingual tooth and root lengths were longer than those of the other three main roots (P < .001). The thickness of the roots of these teeth was not significantly different from one root to another. There were no 309/409 buccal or lingual roots in either species.
Babyrousa babyrussa and Babyrousa celebensis Mandibular Molar Teeth Measurements.
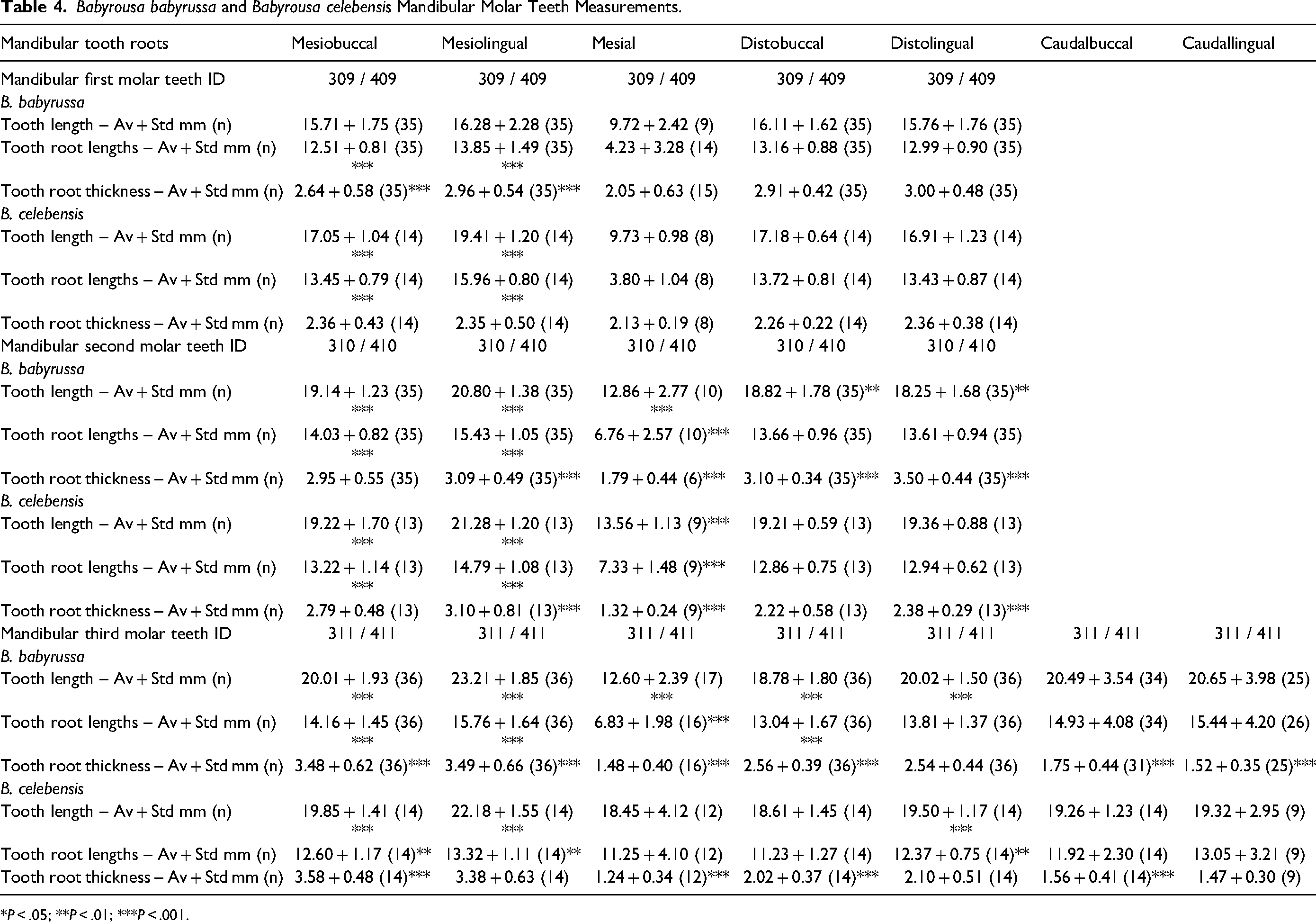
*P < .05; **P < .01; ***P < .001.
The 110/210 tooth roots were tapering, rod-like structures that were somewhat blunt-ended (Figure 1B). A slight distal tilt from vertical of the distobuccal root was also seen in the 111/211 tooth roots, where 72% of the B. babyrussa and 67% of the B. celebensis distobuccal roots inserted at an angle distally (Figure 1B). The variety of patterns of oval and closed ‘C’ roots in these teeth was note-worthy, and in particular the open ‘C’ shaped distopalatal roots (Figure 4). The data for the 110/210 teeth are summarized in Table 3. In both species, the four main roots had similar lengths but shorter mesial tooth lengths (P < .001). In B. babyrussa, the mesiopalatal root was thicker than the other mesial roots (P < .001), and the diagonally opposite distobuccal root was thicker than the other distal roots (P < .001). The mesiopalatal and mesial root thickness of the B. babyrussa was larger than that of the corresponding distopalatal and distal roots (P < .001), whereas the converse was true for the mesiobuccal and distobuccal root thickness (P < .001). Only one central root was found in B. babyrussa (Table 3). In B. celebensis the mesiopalatal and distobuccal root thicknesses are greater than that of the distopalatal root (P < .01).
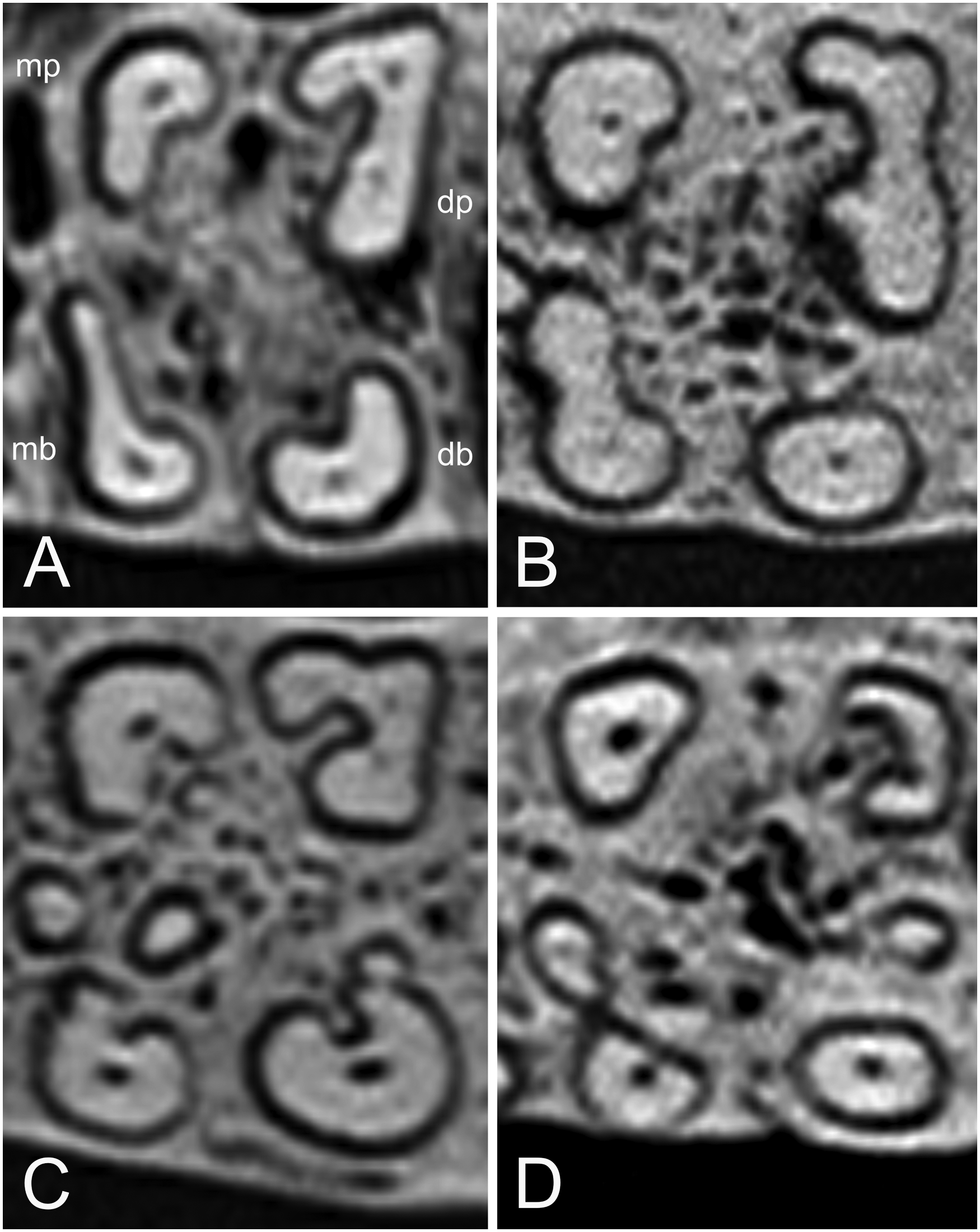
Cross-sectional CT-scans of the 210 tooth of (A) Sulawesi babirusa (Babyrousa celebensis) GNM 4.728 illustrating the four roots (mb = mesiobuccal, mp = mesiopalatal, db = distobuccal, dp = distopalatal). (B) Buru or Sula babirusa (Babyrousa babyrussa) NMSZ.1991.15.1. (C) Buru or Sula babirusa (Babyrousa babyrussa) NMS,Z,1991.15.5. (D) Buru babirusa (Babyrousa babyrussa) NMSZ.1992.10.28, to illustrate relative positions, sizes and shapes of the four main roots, and the variety of ‘C’ shapes of the distal palatal roots. Note the presence of additional roots in C and D.
The 310/410 tooth roots were rod-like structures. In both species, 67% of the 310/410 mesiobuccal and distobuccal roots were inserted almost vertically into the bone (Figure 1B). Many of the 310/410 roots showed semi-closed or open ‘C’ shaped structures (Figure 5B). In addition, 29 (80.6%) of the B. babyrussa and 10 (71.4%) of the B. celebensis 310/410 teeth had a ridge of dentine, orientated mesiodistally, descending from a central position into the bone (Figure 5). Comparable ridges were less frequently seen in 309/409 teeth (16 [44.4%] B. babyrussa and in four [28.6%] B. celebensis), in 109/209 teeth (ten [27.8%] B. babyrussa and five [35.7%] B. celebensis) and 110/210 teeth (six [16.7%] B. babyrussa and in eight [57.1%] B. celebensis). Underlying ridges of tooth dentine were also seen in a central position in 111/211 teeth (19 [52.8%] of the B. babyrussa and in ten [62.5%] of the B. celebensis) and to a lesser extent in 311/411 teeth (11 [30.6%] of the B. babyrussa and in two [12.5%] of the B. celebensis).
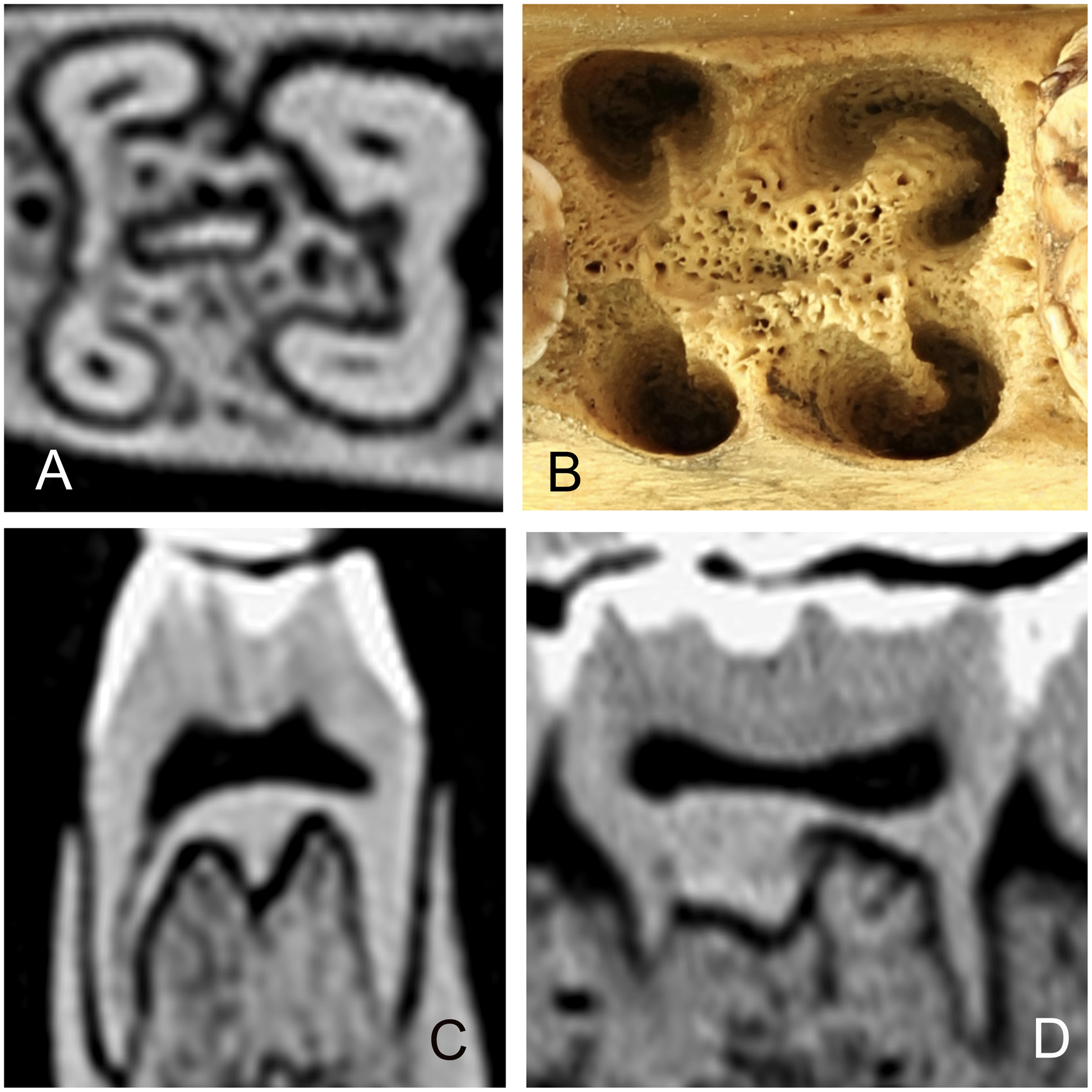
(A) Cross-sectional CT-scan of the 310 tooth in Sulawesi babirusa (Babyrousa celebensis) GNM 17.966 to illustrate the four main roots and the central ridge of bone between these roots. (B) Photograph of the 310 tooth alveolus of Sulawesi babirusa (Babyrousa celebensis) GNM 4.730 with openings into the bony matrix corresponding to positions of the roots and the bony ridge described in 5A. (C) Rostral CT-scan of the 310 tooth in Sulawesi babirusa (Babyrousa celebensis) GNM 17.966 to illustrate the bony ridge between the roots. (D) Lateral CT-scan of the 310 tooth in Sulawesi babirusa (Babyrousa celebensis) GNM 17.966 to illustrate the bony ridge between the roots.
The measurement data for the 310/410 teeth are summarized in Table 4. In both species, the mesiolingual tooth and root measurements were longer than those of the other roots (P < .001). In B. babyrussa the distolingual roots were thicker than the other roots (P < .001). In both species the central roots were about half the length of the four main roots (P < .001). The central roots were also thinner than the four main roots (P < .001). There were no 310/410 buccal or lingual roots in either species.
The structures of 111/211 tooth roots were complex. The robust and tapering rod-like mesiobuccal roots inserted almost vertically into the maxilla when viewed laterally (Figure 6). The distobuccal roots, also robust and tapering rods, were more variable with some inserting almost vertically into the maxilla (Figure 6B) and others orientated in a more distal direction (Figure 6A, C and D). The distobuccal, distal and distopalatal roots were orientated distally into the maxilla (Figures 6 and 7A, B). The mesial and distal roots were splayed in appearance when viewed in the longitudinal section (Figure 7D and E). The cross-sectional shapes of the mesial and distal roots were oval, the latter appearing more elongated in a mesiodistal direction. The cross-sectional shapes of these distal roots were variable and difficult to define. The apices of the distal roots were usually indistinct (Figures 6C, D, and 7A-C) making systematic measurement unreliable. The measurable data for the 111/211 teeth are summarized in Table 3. In B. babyrussa the mesiobuccal tooth lengths were longer than the mesial and mesiopalatal lengths, and the distobuccal tooth lengths were longer than the distal and distopalatal lengths (P < .001). However, the mesial tooth root lengths were longer than those of the mesiobuccal and mesiopalatal roots (P < .05). The distal root lengths were longer than those of the distopalatal roots (P < .001). The B. babyrussa 111/211 teeth had no central roots (Table 3). In B. celebensis the mesiobuccal root was longer than the mesiopalatal root and the distal root was longer than both distobuccal and distopalatal roots (P < .001) as well as the central and mesial roots (P < .05). In both species the mesiopalatal root thickness was greater than those of the mesial and distopalatal tooth roots (P < .001), and the distobuccal root thickness was greater than those of the distopalatal and distal roots (P < .05).
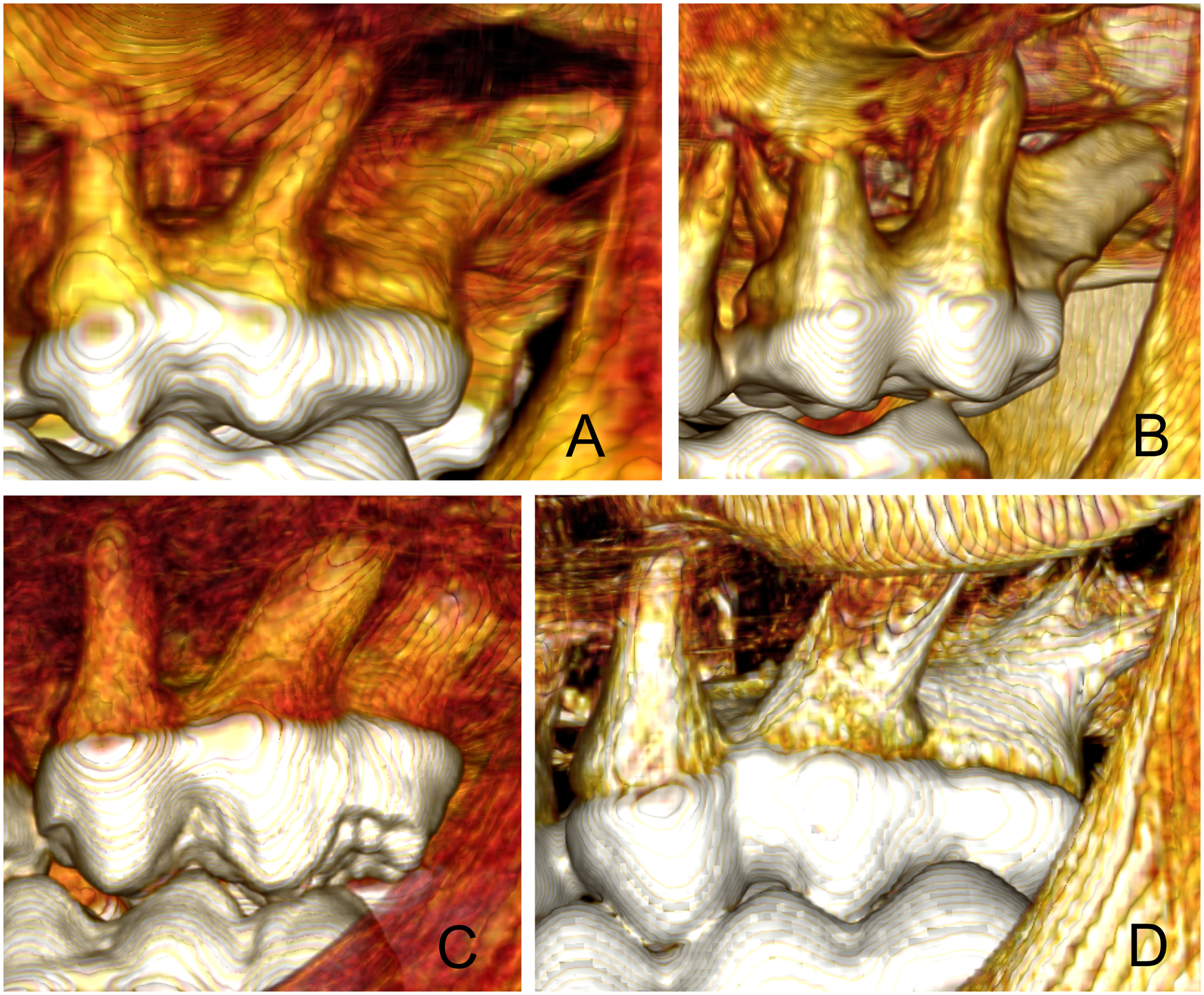
(A) 3D CT-scan view of the 211 tooth roots of Buru babirusa (Babyrousa babyrussa) SNSD 040320183 DB to illustrate the distal orientation of the distal and distobuccal roots. (B) 3D CT-scan view of the 211 tooth roots of Buru babirusa (Babyrousa babyrussa) SMF 19706 DB to illustrate the swollen shape and distal orientation of the distal root. (C) 3D CT-scan view of the 211 tooth roots of Sulawesi babirusa (Babyrousa celebensis) NMS,Z,1878.3.1 to illustrate the distal orientation and rod-like appearance of the distal and distobuccal roots. (D) 3D CT-scan view of the 211 tooth roots of Sulawesi babirusa (Babyrousa celebensis) GNM 17.960 to illustrate the distal orientation and splintered appearance of the distal and distobuccal roots.
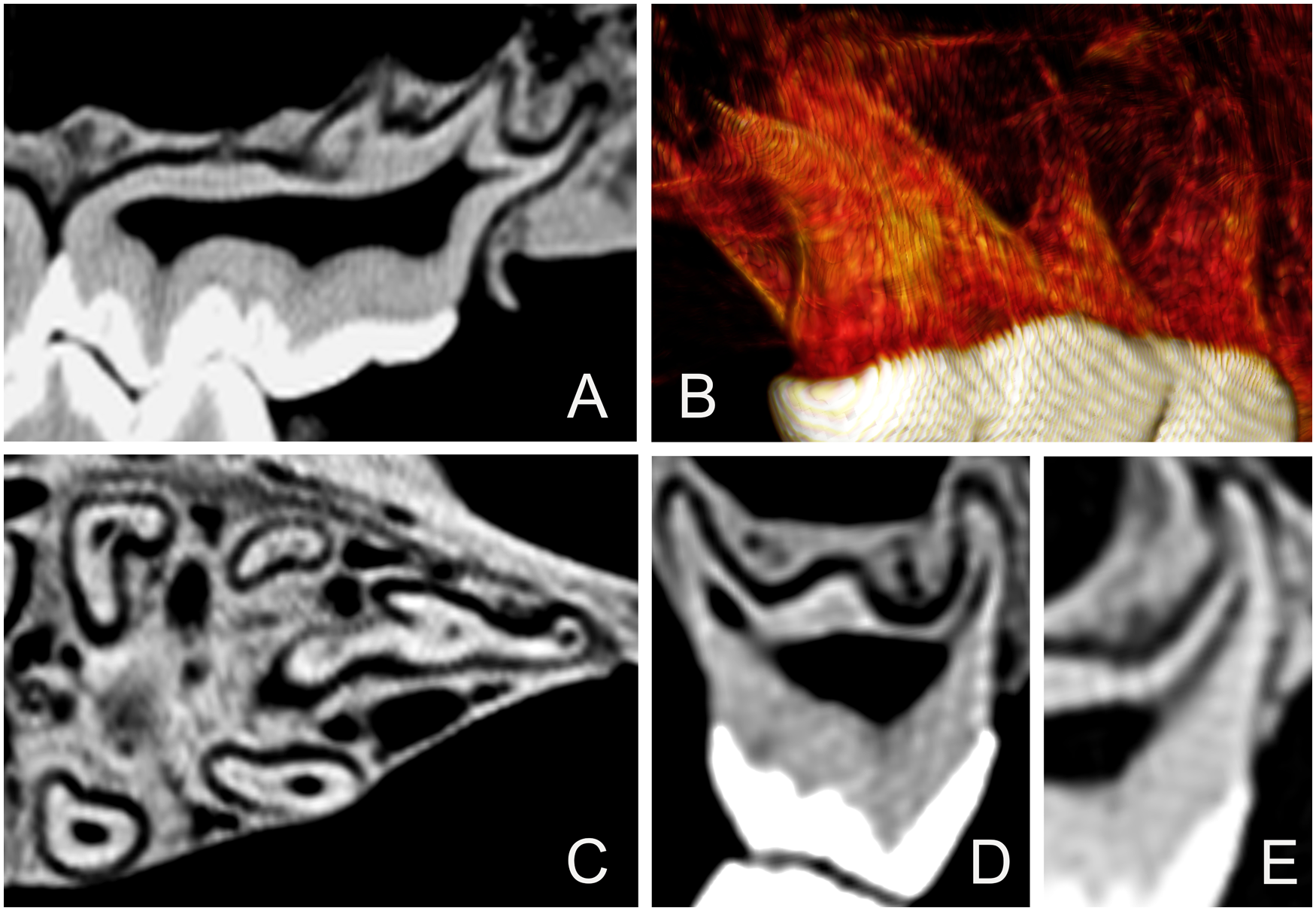
(A) Lateral-sectional CT scan of the 211 tooth root of Sulawesi babirusa (Babyrousa celebensis) GNM 17.960 to illustrate the complexity of the distal root structure. (B) 3D CT-scan view of the 211 tooth roots of Sulawesi babirusa (Babyrousa celebensis) GNM 17.960 seen from a distopalatal viewpoint. (C) Cross-sectional CT scan of the 211 tooth root of Sulawesi babirusa (Babyrousa celebensis) GNM 17.960. (D) Distal view of the longitudinal-sectional CT scan of the 211 distal roots of Sulawesi babirusa (Babyrousa celebensis) GNM 17.960 to illustrate the splayed arrangement of these roots. (E) Distal view of the longitudinal-sectional CT scan of the 211 mesiopalatal root of Sulawesi babirusa (Babyrousa celebensis) GNM 17.960.
The 311/411 tooth roots retained elements of the open ‘C’ shape (Figure 8A) and their insertions were generally blunt-ended and orientated distally (Figure 8B, D and E). The cross-sectional shapes of these roots were generally elongated oval (Figure 8A) and the apices were often bowed (Figure 8B) or splayed lingually and buccally (Figure 8C) into the bony matrix. The shapes of the distal roots were rod-like with distobuccal and distolingual roots being identified (Table 4). The data for the 311/411 teeth are summarized in Table 4.
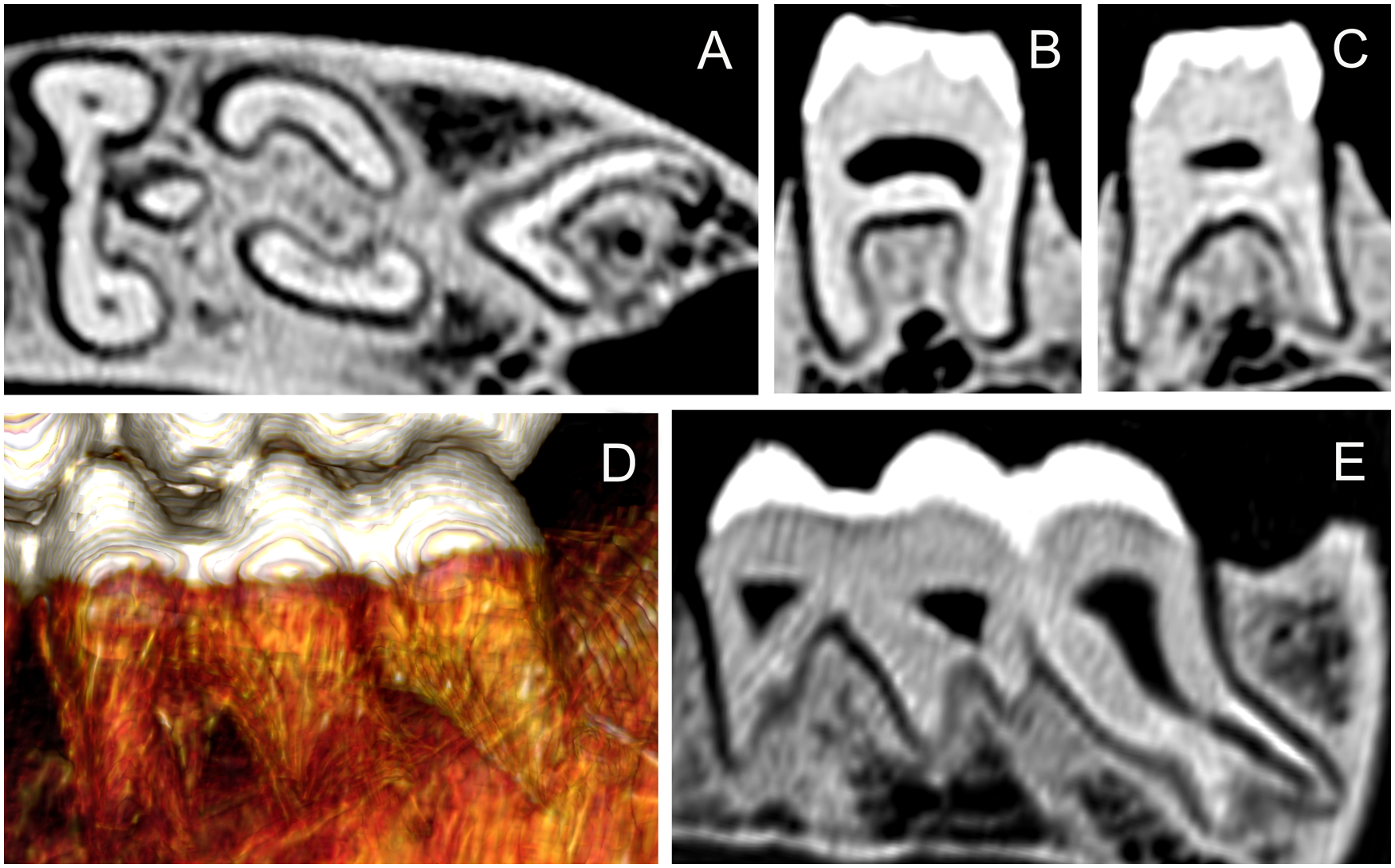
(A) Cross-sectional CT scan of the left mandibular 311 tooth root of Sulawesi babirusa (Babyrousa celebensis) GNM 4.728. (B) Mesial-sectional CT scan of the 311 distal tooth roots of Sulawesi babirusa (Babyrousa celebensis) GNM 4.728 to illustrate the mesial orientation of the roots. (C) Mesial-sectional CT scan of the 311 distal tooth roots of Sulawesi babirusa (Babyrousa celebensis) GNM 4.728 to illustrate the more distal and splayed orientation of these roots. (D) Lateral 3D CT scan of the 311 tooth root of Sulawesi babirusa (Babyrousa celebensis) GNM 4.728 to illustrate the shapes and orientation of the (left to right) mesiobuccal, buccal and distobuccal roots. (E) Lateral-sectional CT scan of the 311 tooth root of Sulawesi babirusa (Babyrousa celebensis) GNM 4.728 to illustrate the shape and distal orientation of the distobuccal root.
Within the two species, the lingual, distolingual and distobuccal teeth have similar tooth lengths. In B. babyrussa the mesiolingual tooth and root lengths are greater than that of the mesiobuccal (P < .001). The central root is less than half the lengths of the other roots; its root thickness is also thinner than those of the mesiobuccal and mesiolingual roots (P < .001). Buccal and lingual roots are thicker that distobuccal and distolingual roots (P < .001).
In B. celebensis the mesiolingual tooth and root lengths are longer than those of the mesiobuccal roots (P < .01). There are no significant differences in tooth and root lengths between the other roots. However, the mesiolingual roots were thicker than lingual and distolingual roots (P < .01). Central roots were thinner (P < .01).
Root Canal System
The pulp chamber of the 109/209 tooth was arched slightly in four places to fit under the thick dentine layer which itself was raised to fit under the four molar cusps; paracone, protocone, metacone and tetracone (Figure 2A-C). In each of the maxillary molar teeth the floor of the pulp chamber rested quite flat on the overlying dentine layer. The number of pulp canals detected in each 109/209 tooth root was one. However, B. babyrussa skulls had two pulp canals in two mesiobuccal roots and three distopalatal roots. In B. celebensis skulls, two pulp canals were detected in each of one mesiopalatal root, two distobuccal roots and five distopalatal roots, and three pulp canals were detected in one distopalatal root.
The pulp chamber of the 110/210 tooth was more obviously arched in four places to fit under the thick dentine layer which itself was raised to fit under the four main molar cusps (Figure 2A-C). The number of pulp canals detected in each 110/210 tooth root was one. However, B. babyrussa skulls had two pulp canals in eleven distopalatal roots, three in eleven distopalatal roots and four in four distopalatal roots. In B. celebensis skulls, two pulp canals were detected in each of two mesiopalatal roots, one distobuccal root and four distopalatal roots; three pulp canals were found in eight distopalatal roots; four pulp canals were found in one distopalatal root.
The pulp chamber of the 111/211 teeth was similar to that of the 110/210 teeth in that the pyramidal arching under each of the four main molar cusps (Figure 2A-C) was clearly defined (Figure 7A-D). The number of pulp canals detected in each 111/211 tooth root was generally one. However, B. babyrussa skulls had two pulp canals in each of five mesiopalatal roots, two distobuccal roots, one distopalatal root, one additional buccally oriented distal root and four palatally oriented distal roots; three pulp canals were found in one mesiopalatal root. In B. celebensis, two pulp canals were detected in each of nine mesiopalatal roots, four distobuccal roots, four distopalatal roots and two palatally oriented distal roots; three pulp canals were found in one mesiopalatal root and in three palatally oriented distal roots.
The pulp chamber of the 309/409 teeth was arched slightly in four places to fit under the thick dentine layer which itself was raised to fit under the four main molar cusps (Figure 2B-D). In each of the mandibular molar teeth the floor of the pulp chamber rested quite flat on the underlying dentine layer. The number of pulp canals detected in each B. babyrussa 309/409 tooth root was one. In B. celebensis each tooth root contained one pulp canal except for two of them being found in two mesial roots.
The pulp chamber of the 310/410 teeth was more obviously arched in four places to fit under the thick dentine layer (Figure 5C and D), which itself was raised to fit under the four main molar cusps (Figure 2B-D). The number of pulp canals detected in each 310/410 tooth root was one with the exception in B. babyrussa skulls where two were found in two distolingual roots. In B. celebensis each tooth root contained one pulp canal with the exception of two pulp canals found in each of two distobuccal roots and nine distolingual roots; three pulp canals were found in one distolingual root.
The pulp chamber of the 311/411 teeth was somewhat similar to that of the 310/410 teeth in that the pyramidal arching under each of the main molar cusps (Figure 2B-D) was clearly defined. The dentine layer overlying it was relatively thick (Figure 8B,C and E). The number of pulp canals detected in each 311/411 tooth root was one. However, B. babyrussa skulls revealed two in each of one central root, twenty-six buccal roots, twenty-one lingual roots, nine distobuccal roots and six distolingual roots; three pulp canals were found in four buccal roots, as well as in three lingual roots and two distobuccal roots. In B. celebensis each tooth root contained one pulp canal. However, two pulp canals were found in each of one central root, seven buccal roots, nine lingual roots, six distobuccal roots, and one distolingual root; three pulp canals were found in one buccal root and one distobuccal root; five pulp canals were found in one distobuccal root.
Discussion
Recent studies have described the deciduous and permanent premolar teeth of babirusa (Babyrousa spp.).12,15 The current investigation is the first since 1899-19005,16 to have closely examined the structure of the maxillary and mandibular molar teeth in the adult babirusa (Babyrousa spp.). It has extended, by illustration and measurement, the observed descriptions of those teeth and has provided additional detail regarding the anatomical variation of their roots. It has been confirmed that for both species of Babyrousa the roots of these teeth are more complex than simple rod-like structures (Figures 7 and 8). It has also quantified the various numbers of roots associated with each molar tooth (Table 2).
Studies of Sus domesticus 17 and wild Sus scrofa 18 have made available detailed comparative information on the numbers of tooth roots and their frequency of occurrence in these species. Additional tooth root information was provided for the mandible of the ‘Clawn’ strain of miniature Japanese Sus domesticus 19 and the European wild pig, Sus scrofa. 20 During development the mandibular first and second molar teeth of Phacochoerus africanus each receive 4 separate roots which are thereafter completely eroded. 16
One possible explanation for the relatively short lengths of the first molar roots of the babirusa may be that these permanent teeth erupt relatively early (∼6-8 months) when the depths of bony support provided by the growing maxilla and mandible are structurally shallow. 15 In Sus scrofa these first permanent teeth, first maxillary and mandibular molars, are usually present from about five months of age. 21 In babirusa the first molar teeth provide functional masticatory support to the three deciduous maxillary and mandibular cheek teeth until about 20-24 months of age. In Sus domesticus the maxillary and mandibular first molar teeth are present before the replacement of the deciduous teeth by the permanent premolar teeth. 22 At about 20-24 months of age the deciduous cheek teeth of the babirusa are replaced by two relatively long-rooted permanent maxillary and mandibular premolar teeth. By this time the, long rooted, second molar teeth had taken up their positions.12,15 The third molar teeth fully erupt early in the third year, at the end of the pre-adult stage of life. 23
It is likely that relative differences between the maxillary and mandibular jaw bones with regard to the amount of structural bone available to support molar tooth roots contribute to the observed differences in tooth root lengths. In Sus domesticus the densities of the bone in the maxilla and mandible differ significantly, being lower in the maxilla.24,25 This may partly explain why the maxillary molar teeth of babirusa possess more roots than those of the mandible; to increase the stability of maxillary teeth in bone of lower density. The tooth root transmits and balances occlusal forces through the periodontium to the alveolar bone. 26 In the babirusa the cheek tooth wear seen 6 suggested that the first molar teeth had been taking most of the chewing activity with less wear seen in the second and third molar teeth until later in the life of the animal. This pattern was different from that shown for Sus scrofa.27–29 In Sus scrofa there was a relative evenness of progressive wear shown by the first and second molar teeth and a lot more wear of the third molar tooth as that suid aged. The babirusa's mandibular molar teeth appeared to be rooted to function in part as ‘anvils’ for the breaking and crushing of fruits and nuts. 30 The relative pattern of wear of the occlusal surfaces of the mandibular teeth when compared with those of the maxilla tend to lend support to this hypothesis.6,10 The slight splaying of the babirusa tooth roots would contribute towards support of the molar teeth. Both male and female babirusa in zoological collections have been observed to use the molar teeth to crack open the very hard Kenari (Canarium amboinense Hochr.) nutshell and then consume the nut contained within (Leus and Macdonald, 1995, unpublished observations).
The distal orientation of the distal roots of the mandibular third molar teeth was observed in the results of the radiographic study on the Large White breed of pig. 21 This was also seen in the Japanese wild pig (Sus scrofa leucomystax). 31 In this arrangement it appeared to be acting as a ‘brace’ to the tooth to prevent the occlusal surface from being pushed distally. Some preliminary observations of mandibular yaw movements by the feeding babirusa have been recorded. 32 The dynamics of mastication have been examined in more detail in Sus domesticus,33–35 and subsequent research has shown the findings to have wider mammalian application. 36 The closure of the jaw was primarily due to the activity of the masseter and pterygoid muscles on the non-chewing side, and the temporalis muscle on the chewing side. The masseter muscles increased the crushing force on the food bolus, which was positioned in a slightly lateral position, but they also then contributed to the movement of the bolus over the mandible lingually. The anatomy of the cranial muscles supporting babirusa jaw movements has been described. 37 Further studies of their function during mastication are required.
Despite the considerable amount of work in vitro and in vivo with mice and rats, 38 it remains unknown what determines the number of roots per tooth and the direction of root formation, although they are likely to be influenced by Hertwig's epithelial root sheath. Research results indicate that an array of growth and transcription factors was expressed during the initiation of root formation, and these activators and inhibitors appear to work together to achieve a balanced signalling outcome and produce the proper patterning, number and length of dental roots during later stages of tooth morphogenesis. 38 There is some evidence that the cervical tongue, a tongue-shaped epithelial process extending from the tooth's cervical loop, is involved. 39 In rodent studies, the elongation and contact between these cervical tongues form discrete regions which resulted in the furcation zone of roots. Consequently, it has been suggested that the pattern of cervical tongues plays an important role in the determination of the numbers, lengths, and shapes of the tooth roots.39,40 Despite these and other findings, the overall mechanisms regulating dental root formation remain poorly understood.
Interspecies tooth root comparisons have been made for primates, bats and carnivores.41–43 In carnivore species, those that feed on hard food objects have larger tooth roots than those that eat soft or tough foods. 43 In bats with diets of different hardness there was a linear relationship between root size and crown size, indicating that the roots were not expanded disproportionately; instead, the entire tooth was larger in the hard diet species. 44 Given that the babirusa on the northern peninsula of Sulawesi seem to have larger 311/411 teeth than those from the Togian Islands, and that a proportion of these Sulawesi babirusa appear to be attracted to the banana (Musa balbisiana) which has as many as 150-170 hard seeds within it, perhaps a lengthy dietary relationship between (Musa balbisiana) and babirusa has contributed towards relative molar tooth size. 10 The babirusa on the Togian islands are thought to have reached there from east central Sulawesi where this species of banana does not appear to grow.45,46
Conclusion
The relative structure of the molar teeth in babirusa suggests that the main role they play is food grinding and crushing. The first molar teeth appear to provide most of the functional masticatory support in this way to the three deciduous maxillary and mandibular cheek teeth until about 20-24 months of age. Their small roots appear to be related to the shallow availability of bony material in the jaws of young babirusa. Differences in bone density and the requirement for tooth stability may partly explain why the maxillary molar teeth of babirusa possess more roots than those of the mandible. The slight splaying of the babirusa tooth roots would contribute towards support of the tooth. The root pattern of babirusa mandibular molar teeth appeared to function in support of the teeth acting as ‘anvils’ for the breaking and crushing of fruits and nuts.
Materials
Senckenberg Naturhistorische Sammlungen Dresden, Germany (SNSD)
National Museums Scotland, Edinburgh, Scotland (NMS)
Naturmuseum Senckenberg, Frankfurt am Main, Germany (SMF)
Göteborgs naturhistoriska museum, Göteborg, Sweden (GNM)
The University of Edinburgh: Helical scan mode, collimator pitch of 1, 0.4 mm slice width, 0.2 mm slice interval, 120 kVp, 111 mAs, ultra-high resolution bone image reconstruction kernel (proprietary name U75u), 192 mm diameter display field of view, 5122 image matrix (64-slice CT, Somatom® Definition AS Siemens, Erlangen, Germany).
The University of Giessen: Helical scan mode, collimator pitch of 0.75, 0.65 mm slice width, 0.32 mm slice interval, 140 kVp, 190 mAs, bone image reconstruction kernel (proprietary name D), 176 mm diameter display field of view, 5122 image matrix (16-slice CT, Brilliance® 16, Philips Medical Systems, Eindhoven, the Netherlands).
The University of Leipzig: Helical scan mode, collimator pitch of 0.75, 0.7 mm slice width, 0.35 mm slice interval, 140 kVp, 193 mAs, bone image reconstruction kernel (proprietary name D), 100 mm diameter display field of view, 5122 image matrix, g. (6-slice CT, Brilliance® 6, Philips Medical Systems, Eindhoven, the Netherlands).
Blåstjärnans Djursjukhus Göteborg: Helical scan mode, collimator pitch of 0.53125, 0.625 mm slice width, 0.312 mm slice interval, 120 kVp, 200 mAs, bone image reconstruction kernel (proprietary name Bone Plus), 182 mm diameter display field of view, 5122image matrix (16-slice CT, GE Revolution® EVO CT, GE Healthcare Japan Corporation, Tokyo, Japan).
JASP Team (2020) JASP (Version 0.14.1)
Footnotes
Acknowledgements
We are thankful for the kind hospitality and assistance provided by Clara Stefen and Jens Jakobitz of the Senckenberg Naturhistorische Sammlungen Dresden, Germany, to Irina Ruf and Katrin Krohmann of the Senckenberg Forschungsinstitut und Naturmuseum Frankfurt am Main, Germany and to Ines Merseburger of the Universität Leipzig, Veterinärmedizinische Fakultät, Klinik für Kleintiere, for her technical assistance. We are also grateful for the technical support and access to the mammal collections in each of the National Museums Scotland, Edinburgh, Scotland, the Senckenberg Naturhistorische Sammlungen Dresden, Germany, the Senckenberg Forschungsinstitut und Naturmuseum Frankfurt am Main, Germany and the Göteborgs naturhistoriska museum, Göteborg, Sweden. The assistance of Masaaki Ito with respect to the Japanese literature was also much appreciated. We are grateful to the University of Edinburgh and the Balloch Trust for academic support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
