Abstract
Introduction
Dyslipidemia, hypertension (HTN), and Type II diabetes mellitus (T2DM) affect a large number of people and treatment represents an enormous cost to the healthcare system. They can impair the quality of life by increasing the risks of cardiovascular disease, renal disease, and mortality. 1 It is estimated that 30-60% of patients with T2DM have dyslipidemia.1,2 Studies have shown that simultaneous control of glycemic levels monitored via hemoglobin A1c (HbA1c), blood pressure (BP), and lipids reduce cardiovascular events instead of only one factor alone. According to ACC/AHA guidelines, it is recommended that patients with both type 1 and type 2 diabetes between 40-75 years of age be treated with statin therapy as prevention for cardiovascular events. 3 Uncontrolled glycemic levels, lipids, and BP can increase the risk of myocardial infarction or stroke. Hence, it is crucial to control HbA1c, BP, and LDL to prevent cardiovascular events.
Health plans use a 90-day-supply of chronic medications to improve the Center for Medicare and Medicaid Services (CMS) metrics for filling prescriptions and attaining higher Star Ratings. However, the adherence rate is a surrogate marker of disease state goal attainment. The previous research on how 90-day-supply can improve adherence lacks the evidence that it improves reaching the minimal biomarkers for chronic disease management. A 90-day-supply refill was shown to improve persistence, 4 but it does not mean that it improves reaching controlled clinical biomarkers. This study was completed to assess refill patterns and clinical biomarkers. If the adherence rates calculated by the proportion of days covered (PDC) are higher with 90-day-supply the assumption is clinical markers should be controlled more frequently.
The purpose of this study was to determine if a 90-day-supply resulted in the clinical endpoints of patients were controlled more often when patients received 90-day-supply rather than less than 90-day-supply prescriptions during one calendar year in a defined sample of patients. The specific aim was to determine if 90-day-supply prescriptions were related to better adherence as defined by CMS and disease state control using HbA1c, BP, and LDL as clinical biomarkers markers. Injectable medications for diabetic patients are not included in the Star Ratings System and were not included in this analysis. The study is specific to oral hypoglycemic agents (OHA), an angiotensin-converting enzyme inhibitor/ angiotensin II receptor blockers (ACEI/ARB), and/or statins.
Study Method
Design
This was a retrospective study with data obtained from patient insurance claims and electronic medical record reviews. The study was approved by the Human Subjects Protection Program of the university and the healthcare medical group.
Subjects
Patients in a health plan defined cohort with dual coverage with Medicare and Medicaid were reviewed. These patients were low-income and high utilizers of healthcare. Each patient had a university primary care physician or nurse practitioner. The cohort was provided at home services to improve their care and close Medicare gaps. Patients were prescribed an OHA, an ACEI/ARB, and/or a statin between January 1, 2017, and December 31, 2017, and had clinical markers for HbA1c, BP, or LDL values documented between January 1, 2017, and March 30, 2018. Individuals without clinical markers or day-supply of medication were excluded.
Measures
The University Health Plan Quality Improvement (QI) data were provided to the pharmacist with the PDC. These data included gender, age, and primary language spoken. The QI data did not include self-reported ethnicity. These data were divided into those adherent or not adherent according to health plan (the QI definition of adherence was having the PDC ≥ 85% during one calendar year and was used for this project). These data were also divided into the ones prescribed an OHA, an ACEI/ARB, and/or a statin from January 1, 2017 through December 31, 2017.
The QI data for these patients required a review of the insurance claims data for each patient. The pharmacist manually reviewed each profile and divided patients into a group getting a 90-day-supply throughout the year and a group getting less than a 90-day-supply throughout the year. Patients in the less than a 90-day-supply group received either a 30-day-supply for 12 months, 45-60-day-supply for 12 months, and/or a mixture of 30 and 90-day-supply for 12 months.
The pharmacist completed a manual electronic medical record review to record the HbA1c, BP, and LDL from January 1, 2017, and March 30, 2018. The March 30, 2018 date was used to ensure there were at least three months from the last potential prescription refill of a medication that a clinic visit, and laboratory monitoring measurement was possible. Any patient without the biomarker related to the specific medication class to make an assessment was excluded.
Data Collection
QI data were provided to the pharmacist. An electronic medical record and insurance claims data review was completed by the pharmacist to extract clinical data and day-supply not available in the QI data. Insurance claims data were processed through a proprietary company that was accessible to the pharmacist to ensure that prescriptions were filled, the dates filled, and the quantity. The electronic medical records used were a university-wide, proprietary company that captures all office visit and inpatient admissions for patients seen with in the health system. Laboratory and vital sign data were available. Only the clinical biomarker data were collected. All the data were de-identified and entered in an Excel spreadsheet (Microsoft).
Data Analysis
The entire sample was divided into those patients prescribed an OHA, ACEI/ARB, and statin into separate groups since every patient did not receive all three medication drug classes. Clinical biomarkers were separated into categorical data using the minimum values for controlled or not controlled based on the CMS Star Ratings System. These were as follows: HbA1c <9% and ≥ 9% (OHA), BP <140/90 mm Hg and ≥ 140/90 mm Hg (ACEI/ARB), and/or LDL <100 mg/dl or ≥ 100 mg/dl (statin). Since the standard for hypertension control is having an elevated SBP or DBP, the data were collected such that either SBP or DBP being elevated was determined to be above the minimum threshold.
The patients were categorized into those obtaining 90-day-supply and less than 90-day-supply and had a PDC of <85% or ≥85%. Having PDC ≥85% was the QI definition of adherence. Continuous data were summarized using means and standard deviations and groups were compared using a two-sample t-test assuming independent samples and unequal variances. The separate drug classes were analyzed by the category of adherence and the minimal value for control of the specific disease state. A 2 × 2 comparison was performed using Chi-square analysis for each medication class with the dichotomy of days-supply and minimal biomarker attainment. A P-value ≤ .05 was used to determine the statistical significance of the rates of days-supply of prescriptions and biomarker control. All statistical tests were performed using data analysis tools in Excel (Microsoft).
Results
Of the 273 patients reviewed, 251 met all inclusion criteria, and 22 were excluded due to missing data. The 251 patients were sorted into those with OHA, ACEI/ARB, and statin prescriptions. Patients were analyzed separately and could be in more than one analysis. A total of 105 patients were prescribed an OHA, 158 were prescribed an ACEI/ARB, and 175 patients were prescribed a statin.
Characteristics of Study Subjects.

The percentage of the patients in the OHA, ACEI/ARB, and statin groups that were prescribed a 90-day-supply of medications were 55%, 58%, and 42%, respectively. The percentage of all patients who had PDC ≥85% was 65%, 63%, and 68% for OHA, ACEI/ARB, and statin groups. The range of adherence rates for the three drug classes was similar (OHA 21-100%; ACEI/ARB 25-100%; statin 24-100%). For each drug class, there was a statistically significant difference between those patients receiving a 90-day-supply throughout the year and having a PDC of ≥ 85%.
The HbA1c, BP, and LDL-C for the groups receiving a 90-day supply and less than 90-day-supply were, respectively, 7.4 ± 1.6%, and 7.9 ± 1.7%, 137.2 ± 18.0/77.0 ± 11.0 mm Hg and 133.7 ± 20.6/76.3 ± 13.6 mm Hg, and 88.1 ± 38.2 mg/dl and 96.2 ± 45.5 mm/dl. There were no statistical differences in the HbA1c, SBP, DBP, and LDL between 90-day-supply and less than 90-day-supply.
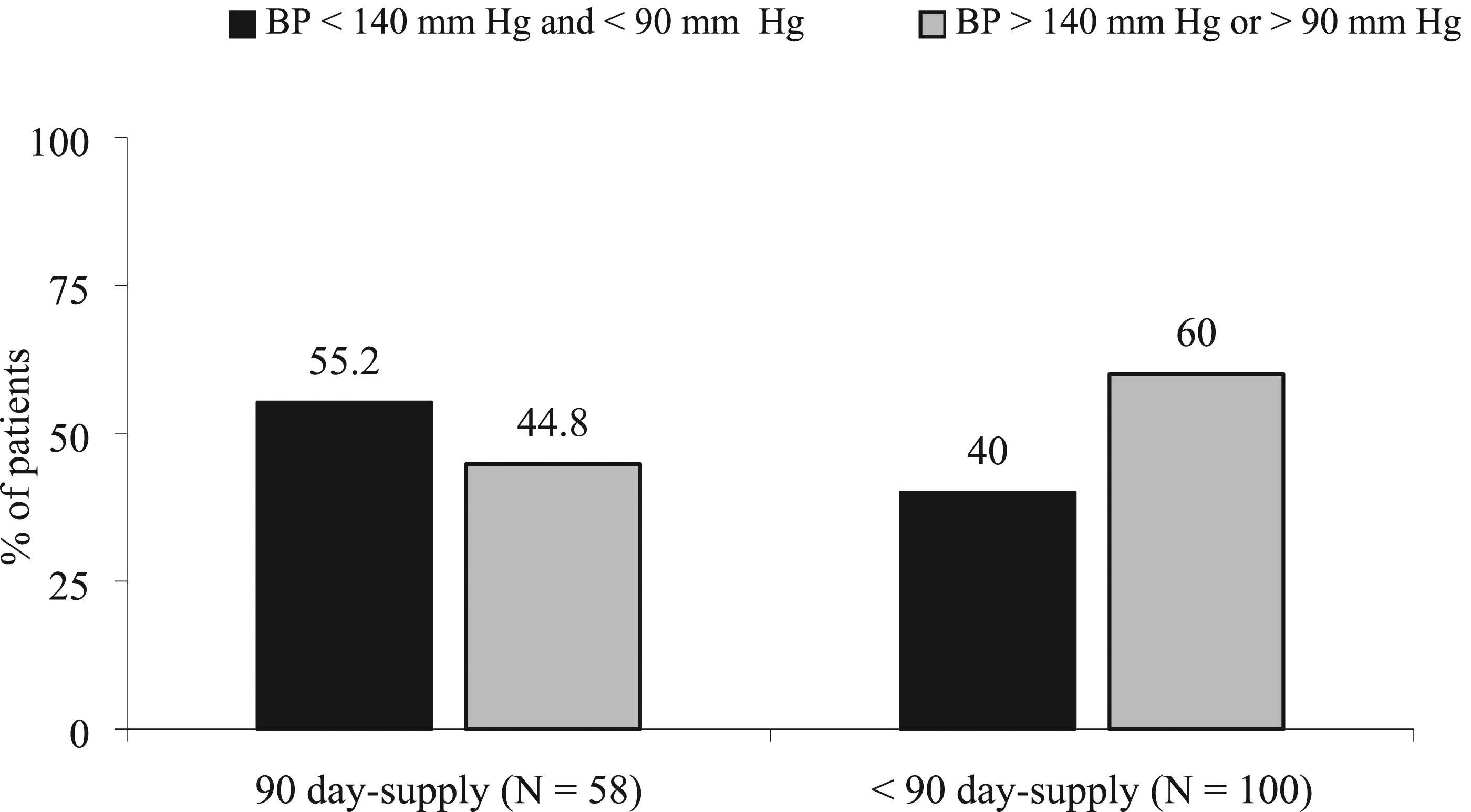
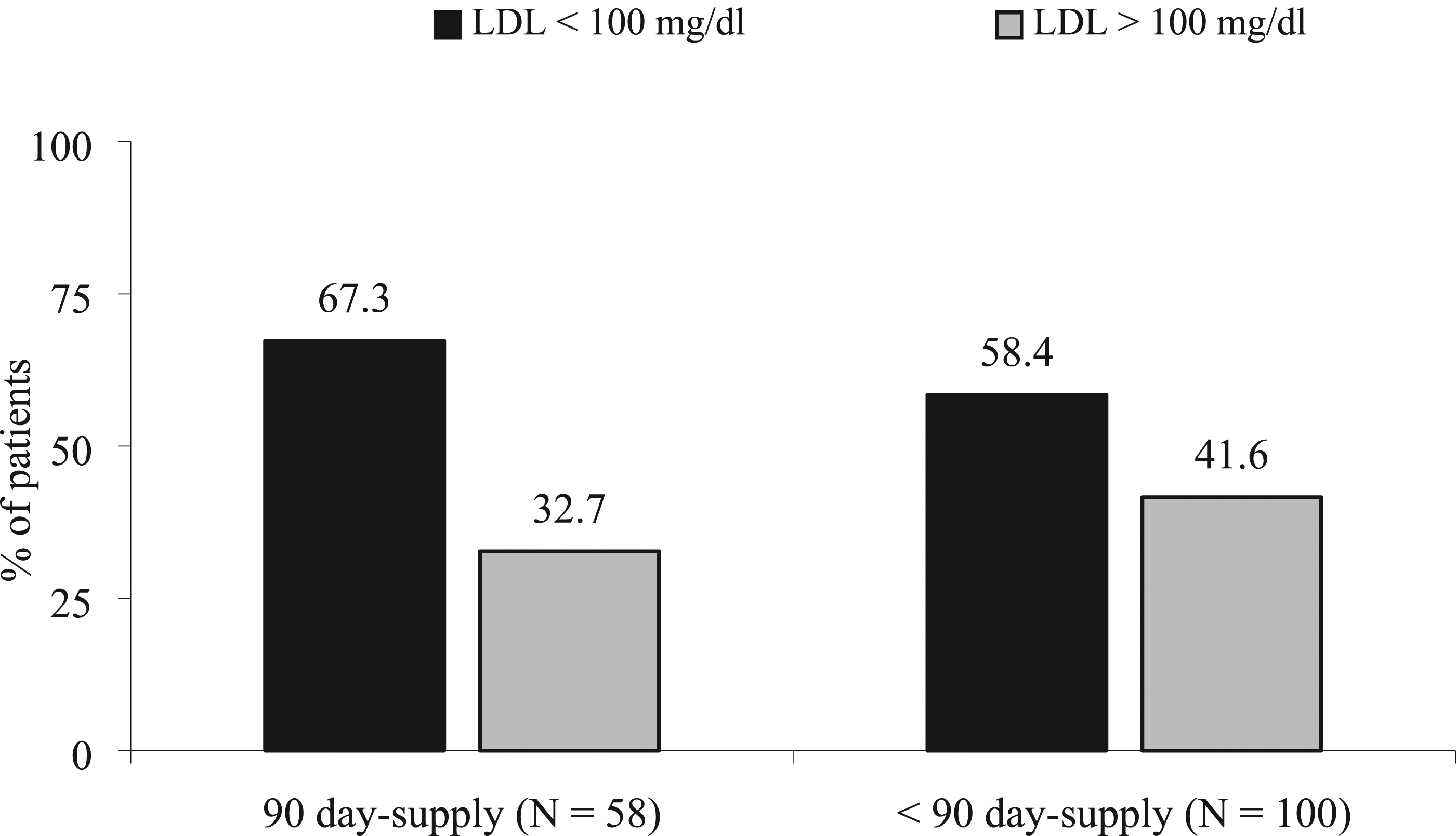
Overall, a total of 81.0% of patients prescribed an OHA had an HbA1c of ≤ 9%, 45.6% had an SBP of < 140 and DBP < 90 mm Hg in the ACEI/ARB patients, and 63.4% of the patients treated with a statin had an LDL < 100 mg/dl. The comparison of receiving a 90-day-supply all year or less than 90-day-supply and attaining the minimal biomarker for the three drug classes are shown in Figures 1–3. Each of the drug class groups had non-statistical differences between receiving a 90-day-supply throughout the year vs less than 90-days-supply and reaching the minimum goal biological marker (P > .05 for each drug class). Comparison of Day-Supply on Hemoglobin A1c (HbA1c). P = .36. Comparison of Day-Supply on Systolic Blood Pressure (SBP). P = .065. Comparison of Day-Supply on Low-Density Lipoprotein (LDL). P = .22.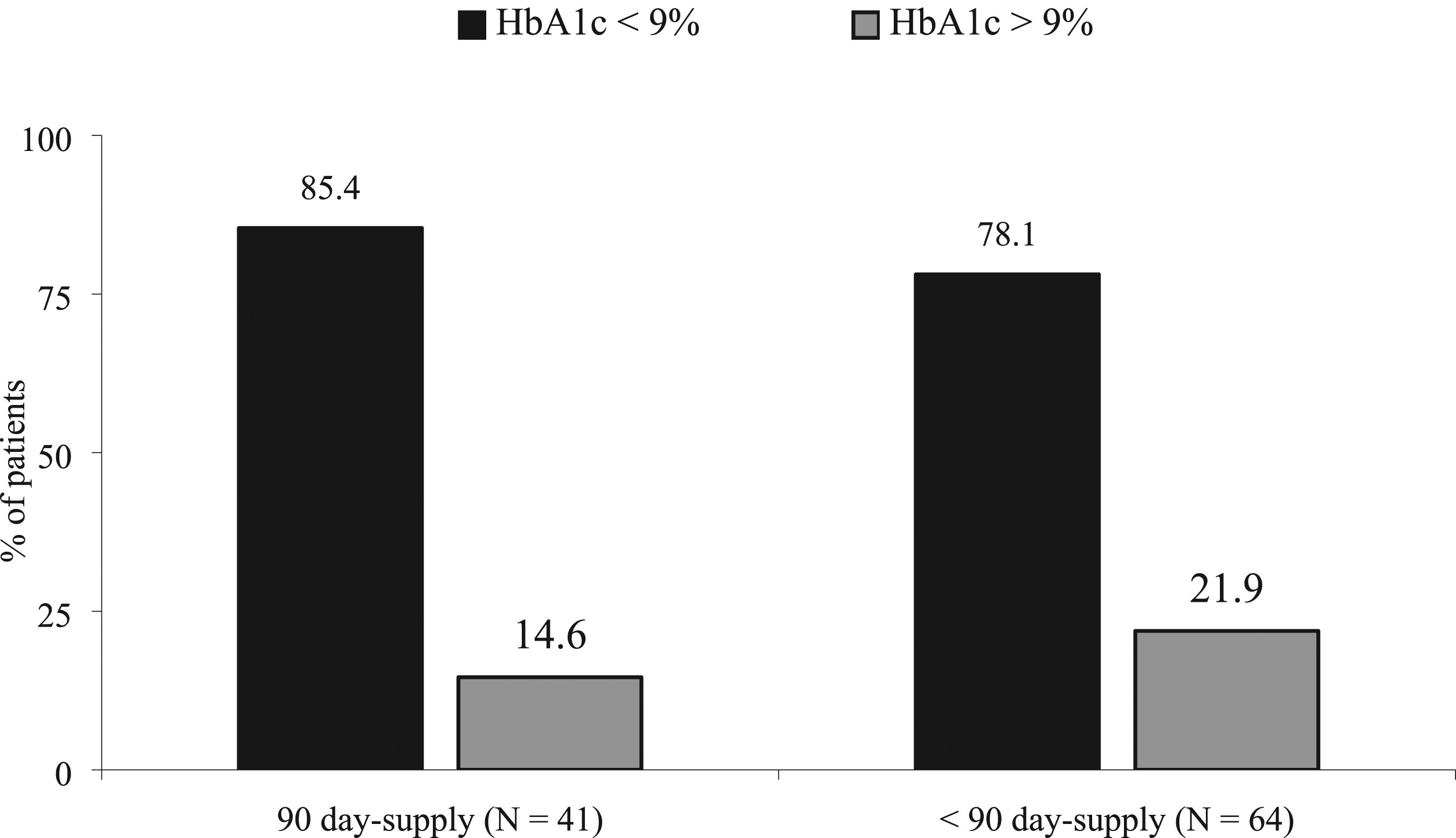
Discussion
The primary findings of this study are that receiving a 90-day-supply of the three drug classes did result in a statistically higher proportion of patients with PDC ≥85%. However, the 90-day-supply was not statistically different than less than 90-day-supply for minimally controlling HbA1c, BP, and LDL. Previous research has found that a 90-day-supply improved the rate of persistence measured by PDC for the CMS Star Ratings medications. 5 These data show the same results. The rate of adequate adherence seen in the 90-day-supply group is a surrogate marker for disease state management. The dichotomy between a PDC of ≥ 85% being statistically greater when a 90-day-supply is used, did not show the minimal clinical biomarker was reached more frequently when a 90-day-supply was used. The PDC is a surrogate marker and these results suggest the surrogate marker is inadequate. A 90-day-supply of medication may improve persistence for patients who have barriers to accessing pharmacies such as lack of transportation or a pharmacy in close vicinity. 6 However, if patients have not reached appropriate clinical endpoints within the disease state goals, it diminishes the utility of a longer day supply. In these results, between 14.6% and 60% of the patients prescribed the targeted drug classes did not attain the minimum clinical biomarker threshold. These results might also support that when obtaining a refill of medications, the pharmacist should also ensure that the underlying disease state is optimally treated. This appears to be an unmet need. It also points out a large gap in care in our health care system.
Other avenues that can help improve adherence such as comprehensive medication reviews, prescription synchronization, automatic refills, home visits, and education have had favorable outcomes. 7 An alerting program, mHealth, or mobile Health intervention for adherence (mAdherence) had a significant impact on adherence behaviors. The impact on adherence was seen in 15 of the 27 randomized controlled trials (RCTs). Significant improvements between groups were reported in 16 of 41 studies (39%) had significant improvements between groups on clinical outcomes. 8 However, it is also important to consider that patients in a lower socioeconomic group might not have access to be able to use technology that will support improving medication-taking behavior.
Non-adherence is a missed opportunity for pharmacies to generate revenue that might improve the quality of care for patients. Since the rate of non-adherence was substantial (about 30% of patients), identifying ways to improve adherence should be developed. Payers should include payment for additional pharmacist’s clinical services that will directly benefit the health plans and patients.
One consideration is the synchronization of all medications to reduce the need to fill prescriptions multiple times during any frequency of day-supply. Synchronization did improve the PDC in a recent meta-analysis of 9 studies. 9 This should be a consideration for all patients regardless of the day-supply of medications.
Knowing which patients are non-adherent might substantially improve the PDC. Identifying non-adherent patients can lead to automatic refills. This approach was shown to improve CMS Star Ratings. 10 Further, it seems possible with computerized prescription records to begin anticipating when refills are needed, and contacting patients could enhance medication-taking behavior. It might also identify a potential medication-related problem (e.g., adverse effect) by contacting patients.
Future research should focus on developing mechanisms to improve persistence, adherence, reaching appropriate clinical endpoints, and optimal outcomes. Using surrogate markers for assessing health outcomes must be shown to be reliable. These results show it is not. Barriers to taking medication include language and health literacy. Pharmacists need to determine if patients understand why they are taking medications and what they should expect when being treated. Lower literacy patients may benefit from larger font medication lists and the times they should be taken. Pictograms for when medications are taken might help the patients with the lowest health literacy. 11 These could be color-coded to help the patient understand treatment (e.g., heart medications could be in red).
Most pharmacy operations lack clinical data and most prescribers do not have prescription fill data in a readily retrievable form. Pharmacists, prescribers, patients, caregivers, and payers should work collaboratively to eliminate these gaps in care and identify patients who are not persistent and seek ways that ensure medication-taking behavior is improved. This can be done by having timely data alerting pharmacists to patients needing refills of chronic medications. Pharmacists should review all medications at the time of filling/refilling any prescriptions to increase the opportunity to identify when prescriptions need to be refilled. Additionally, pharmacists and prescribers should work collaboratively to reach minimal endpoints of therapy to reduce the rate of complications of diabetes, hypertension, and hyperlipidemia. For example, point of care testing and/or reviewing BP, HbA1c, glucose, and/or LDL can be done at the pharmacy or at home visits.
Limitations
There were several limitations of this study. The first is that the study was a retrospective analysis. A limitation inherent in retrospective studies is the accuracy of the data extraction. The QI data could not be independently validated, but the laboratory test results and blood pressures were verified from the manual electronic medical record review. It is possible that the sample size being small is a limitation. This study assessed 100% of a dual eligible cohort and that is the strength. However, the cohort was moderately sized. It would require an increase of more than 15% of the ACEI/ARB patients and more than a 50% of the OHA and statin patients to have reached statistical significance in the analysis. Another limitation is the findings may not be generalizable to other populations. These data were collected from a cohort of Medicare and Medicaid dual-eligible patients seen in a university clinic and who are from a lower socioeconomic segment of the population. These patients may not be generalizable to other Medicare groups.
Conclusion
The PDC was higher for a 90-day-supply vs a less than 90-day-supply. However, the disease state management minimum goals were statistically not different with a 90-day-supply of medication vs a less than 90-day-supply of medication for OHA, ACEI/ARB, and or statins. This points to the discordant nature of CMS efforts. Payers should financially support such efforts as a way to improve persistence and adherence. Since the clinical endpoint is more relevant to outcomes, the attainment of goal clinical biomarkers should be combined with the PDC in assessing Medicare Star Ratings. Future studies should assess larger groups to verify the results of this study and include reaching the minimum biomarker status within the PDC.
Footnotes
Authors’ Note
At the time of this project, Dr. Ameli was a Doctor of Pharmacy student at the R. Ken Coit College of Pharmacy, The University of Arizona.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
