Abstract
Introduction
Iron deficiency is a contributor to morbidity and mortality in patients with heart failure (HF).1,2 Intravenous (IV) iron repletion in patients with heart failure with reduced ejection fraction (HFrEF) leads to a reduction in heart failure hospitalizations, increase in functional capacity, and improvement in quality of life.2-4 Iron deficiency persists in patients after continuous flow left ventricular assist device (CF-LVAD) implantation and often results in concomitant anemia, leading to increases in hospitalizations and mortality.5,6 A recent small series showed benefit of IV iron administration after CF-LVAD implant through the resolution of iron deficiency and anemia, however the IV iron was administered months to years following device placement. 7
Based on recommendations from the ACC/AHA 2022 Guideline for Management of Heart Failure, our institution routinely manages iron deficiency in patients hospitalized with HF through IV iron repletion. 8 Due to the potential benefit, in 2017 this practice was extended to include any CF-LVAD patient meeting iron deficiency criteria. More recently, diagnosis of iron deficiency and administration of IV iron (when indicated) have been completed during the index CF-LVAD implant admission, after surgical placement of the device but prior to discharge. We conducted a retrospective cohort analysis to compare outcomes between iron-deficient patients who did and did not receive IV iron repletion prior to discharge from Abbott HeartMate III™ (HM3) CF-LVAD placement.
Materials and Methods
All patients that underwent HM3 implant at our institution between January 2015 and June 2020 that met criteria for iron-deficiency (serum ferritin <100 ng/mL or ferritin 100 – 299 ng/mL and Tsat< 20%) were identified. Patients were included if they had a documented pre-CF-LVAD Minnesota Living with Heart Failure Questionnaire (MLWHFQ) and 6 Minute Walking Distance (6MWD), a documented post-CF-LVAD MLWHFQ or 6MWD recorded within 180 days of discharge from CF-LVAD implant, as well as a serum ferritin and transferrin saturation (Tsat) recorded post-CF-LVAD implant during the index admission. Patients were excluded if they received IV iron outside of the index admission, required dialysis at time of index discharge, required blood transfusion(s) for a clinically significant bleeding event within 180 days following device implant, or if they expired or received transplant prior to outpatient follow-up. Patients were stratified into 2 groups: iron deficient patients who received IV iron and those who did not receive IV iron. Those in the IV iron group received sodium ferric gluconate in 250 mg increments every 12 hours until total deficit was corrected. 9 Iron deficits were determined by the Ganzoni equation.
The analysis included 2 co-primary endpoints: change in MLWHFQ and 6MWD from baseline to outpatient follow-up occurring within 180 days from discharge. Secondary outcomes included change in New York Heart Association (NYHA) class from baseline to outpatient follow-up, and laboratory resolution of iron deficiency and anemia. Resolution of iron deficiency was defined as no longer meeting the criteria for iron deficiency. Anemia was defined as a hemoglobin of less than 13 g/dL for males and 12 g/dL for females. Resolution of anemia was defined as no longer meeting the criteria for anemia. To assess safety, charts of patients receiving IV iron were reviewed for documentation of infusion related reactions, defined as the occurrence of hypotension (systolic blood pressure decline of >20 mmHg from baseline), anaphylaxis, shortness of breath, urticaria, facial flushing, myalgias, and/or joint pain.
Primary outcome analysis was performed using the ANCOVA model adjusting for baseline MLWHFQ and 6MWD. Secondary outcomes were analyzed using Fisher’s exact test. A
Results
Baseline Characteristics.
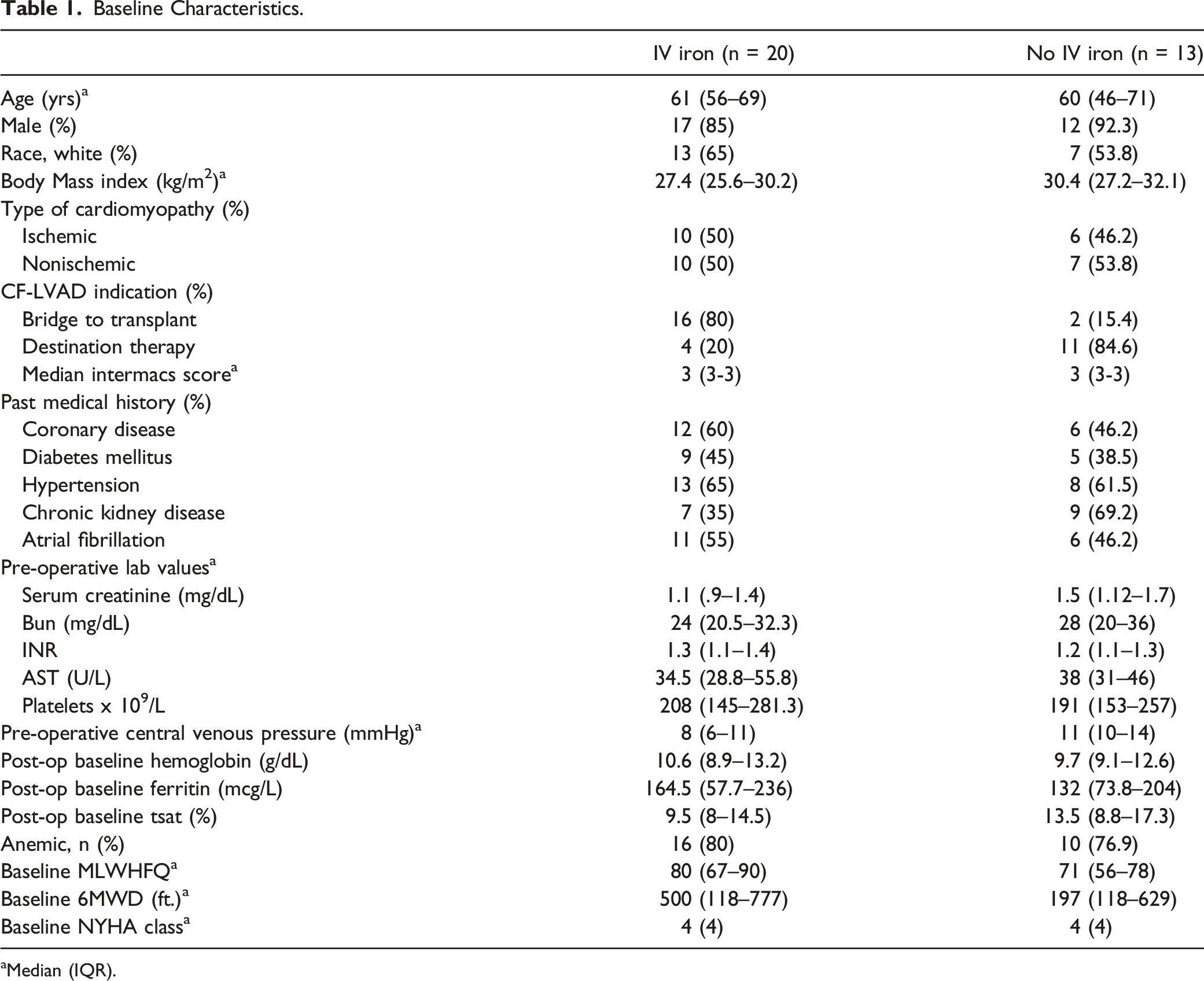
aMedian (IQR).
Study Outcomes.
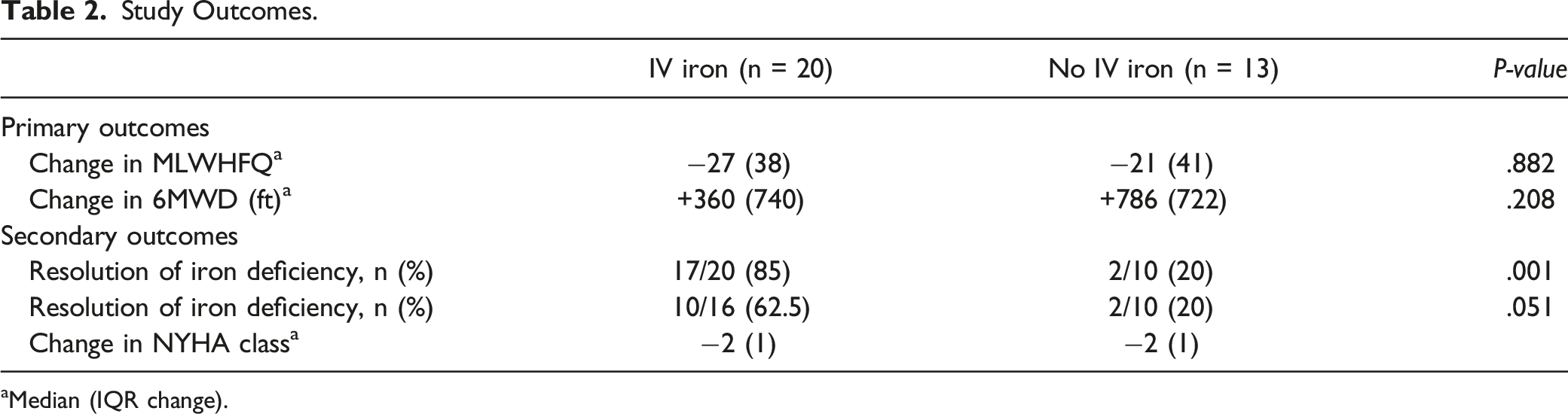
aMedian (IQR change).
Discussion
To our knowledge, this is the first study to evaluate the effect of IV iron given during the index admission following CF-LVAD implant. We found that while patients receiving IV iron did not experience an early improvement in quality of life or functional capacity when compared to those who did not receive IV iron, a significantly greater number of patients receiving IV iron experienced resolution of their iron deficiency at early outpatient follow-up. The majority of them also demonstrated early resolution of their anemia. The most likely explanation for the lack of early functional status improvement attributable to IV iron replacement is the very recent implantation of the CF-LVAD, which has been proven to lead to significant improvement in MLWHFQ and 6MWD over the same time period. However, the exact influence of the CF-LVAD cannot be determined by the findings of our study.
Our study has several important limitations. First, this was a small, single center study with short follow-up. Eighty percent of patients in the IV iron group received a LVAD as bridge to transplant, indicating a potentially healthier patient population than those not receiving IV iron, as evidenced by a higher baseline 6MWD. While not statistically significant, the numerically lower baseline 6MWD in the no IV iron group allowed for a larger increase during the follow-up period, possibly obscuring the impact of early IV iron replacement. A larger number of patients in the no IV iron group were diagnosed with chronic kidney disease than the IV iron group, which may have put that group at higher risk of anemia. However, since baseline rates of anemia were similar (76.9% vs 80%), it is unlikely this difference had any impact on the overall outcome of this study. Post-operative infections were not assessed as a safety outcome in our study. While the data regarding risk of infection post intravenous iron infusion is inconclusive, the risk of infection following IV iron infusions in the CF-LVAD population cannot be determined by this study. Most patients not receiving IV iron had HM3s implanted prior to 2017. It is possible that changes in clinical management over the time period of this study may have had an unmeasured impact on early post-operative outcomes.
Our results demonstrate the safety of early post-operative administration of IV iron in the CF-LVAD population and its efficacy in correcting iron deficiency. It remains unclear whether this therapy has a meaningful impact on patients’ early post-operative quality of life and functional capacity. However, while not proved by our data, given that failure to resolve iron deficiency after LVAD is associated with poor outcomes, it stands to reason that an early, effective iron replacement protocol such as the 1 described above here may confer benefits. There may also be other benefits of early inpatient repletion of iron stores in the population, such as reducing the amount of blood products required by bridge to transplant patients to prevent alloantibody formation and also avoiding the high costs of outpatient IV iron administration. Future prospective studies are needed to clarify the optimal timing and magnitude of symptomatic benefit, if any, of IV iron repletion in the post-CF-LVAD patient population.
Footnotes
Author’s Contributions
Ryan M. Vesper (ryan.vesper@nm.org): Concept/design, data analysis/interpretation, drafting article, approval of article, statistics, and data collection, Lauren Kemp (lauren.kemp@conehealth.com): Concept/design, critical revision, Prashanth Iyer (piyer97@gmail.com): Concept/design, data analysis/interpretation, statistics, critical revision, Katelynn Granger (grangerK@musc.edu): Data collection, Christina T. Doligalski (Chrissy.doligalski@unchealth.unc.edu): Concept/design, data analysis/interpretation, critical revision, Mirnela Byku (mirnela.byku@med.unc.edu): critical revision, Ian B. Hollis (ian.hollis@unchealth.unc.edu): Concept/design, data analysis/interpretation, drafting article, approval of article, critical revision.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Disclosure
Dr. Byku currently serves as an advisory board member for Abbott Laboratories.
